Submitted:
02 April 2026
Posted:
07 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Health Risks Across Mission Durations
2.1. Short-Duration Missions (30 Days)
2.2. Medium-Duration Missions (180 Days)
2.3. One-Year Missions
2.4. Multi-Year, Mars-Class Missions (2+ Years)
3. Health Risk Matrix: Diagnosis, Therapy, Monitoring
4. Preventive Countermeasures and Predictive Monitoring
5. AI-Powered Onboard Physician and Surgical Robotics
6. Engineering Architecture: MERIDIAN System
7. Dual-Use Potential for Low-Income Nations
- 1)
- ease of use and self explanatory,
- 2)
- providing automation and decision support based on monitoring data and a prediction engine,
- 3)
- low manufacturing and operating cost,
- 4)
- small footprint and energy efficiency,
- 5)
- local fabrication and maintenance possibilities,
- 6)
- connectivity-aware medical and operational AI capabilities,
- 7)
- high data security and individual accounts for each user,
- 8)
- health status metrics and progress indicators together with recommendations,
- 9)
- connection to a local sensor element (e.g. a smartphone with camera) with a dashboard,
- 10)
- and the ability to send / retrieve data and connect to emergency services.
8. Conclusion and Next Steps
9. Engineering / Innovator Development Agenda
10. Addendum - Abbreviations
References
- Nicogossian, A.E.; Williams, R.S.; Huntoon, C.L.; Doarn, C.R.; Polk, J.D. Space Physiology and Medicine: From Evidence to Practice, 4 ed.; Springer: New York, 2016. [Google Scholar] [CrossRef]
- Williams, D.; et al. Acclimation during space flight: Effects on human physiology. Canadian Medical Association Journal 2009, 180, 1317–1323. [Google Scholar] [CrossRef]
- Buckey, J.C. Space Physiology; Oxford University Press: Oxford, 2006. [Google Scholar]
- Hargens, A.R.; Vico, L. Long-duration bed rest as an analog to microgravity. Journal of Applied Physiology 2016, 120, 891–903. [Google Scholar] [CrossRef]
- Balasubramaniam, S.; et al. Space medicine in the era of long-duration exploration missions. npj Microgravity 2020, 6, 32. [Google Scholar]
- Smith, T.; Peterman, A.; Donoviel, D. Exponential Medicine: Challenges of Human Spaceflight Bringing Innovations for Earth—A Case Study. In Novel Innovation Design for the Future of Health; Friebe, M., Ed.; Springer: Cham, 2022. [Google Scholar] [CrossRef]
- Nguyen, T.H. Space Healthtech: Innovation Base for Longevity. In Novel Innovation Design for the Future of Health; Friebe, M., Ed.; Springer: Cham, 2022. [Google Scholar] [CrossRef]
- Farmer, P.E.; et al. Expansion of cancer care and control in countries of low and middle income: A call to action. The Lancet 2010, 376, 1186–1193. [Google Scholar] [CrossRef] [PubMed]
- Meara, J.G.; et al. Global surgery 2030: Evidence and solutions for achieving health, welfare, and economic development. The Lancet 2015, 386, 569–624. [Google Scholar] [CrossRef] [PubMed]
- Mindel, S.; et al. Artificial intelligence applications for point-of-care ultrasound in low-resource settings: A scoping review. Ultrasound in Medicine & Biology 2024, 50, 601–616. [Google Scholar] [CrossRef]
- Savonen, B.L. 3D Printing in Low Resource Healthcare Settings. Doctoral dissertation, Michigan Technological University, 2020. [Google Scholar]
- Hughson, R.L.; et al. Cardiovascular adaptations to long-duration spaceflight: Lessons from the ISS. Physiological Reviews 2018, 98, 599–626. [Google Scholar] [CrossRef]
- Cohen, H.S.; et al. Vestibular and cognitive effects of spaceflight. Journal of Vestibular Research 2010, 20, 1–9. [Google Scholar] [CrossRef]
- Crucian, B.; et al. Immune system dysregulation during spaceflight: Potential countermeasures for deep-space exploration missions. Frontiers in Immunology 2014, 5, 267. [Google Scholar] [CrossRef]
- Vico, L.; Hargens, A. Skeletal changes during and after long-duration spaceflight. The Lancet Diabetes & Endocrinology 2018, 6, 829–838. [Google Scholar] [CrossRef]
- Fitts, R.H.; et al. Prolonged space flight-induced alterations in the structure and function of human skeletal muscle fibres. The Journal of Physiology 2010, 588, 3567–3592. [Google Scholar] [CrossRef]
- Lee, A.G.; et al. Spaceflight-associated neuro-ocular syndrome (SANS). Eye 2020, 34, 1164–1170. [Google Scholar] [CrossRef]
- Macias, B.R.; et al. Spaceflight-associated neuro-ocular syndrome (SANS) and the potential role of head-down tilt bed rest. npj Microgravity 2020, 6, 7. [Google Scholar] [CrossRef]
- Cucinotta, F.A.; Durante, M. Cancer risk from exposure to galactic cosmic rays: Implications for space exploration by human beings. The Lancet Oncology 2006, 7, 431–435. [Google Scholar] [CrossRef] [PubMed]
- Levy, J.; et al. Deep vein thrombosis in space. The New England Journal of Medicine 2020, 382, 89–90. [Google Scholar] [CrossRef]
- Van Ombergen, A.; et al. Brain tissue-volume changes in cosmonauts. The New England Journal of Medicine 2018, 379, 1678–1680. [Google Scholar] [CrossRef] [PubMed]
- Roberts, D.R.; et al. Effects of spaceflight on astronaut brain structure as indicated on MRI. The New England Journal of Medicine 2019, 380, 1647–1656. [Google Scholar] [CrossRef]
- LeBlanc, A.; et al. Bisphosphonates as a supplement to exercise to protect bone during long-duration spaceflight. Osteoporosis International 2013, 24, 2105–2114. [Google Scholar] [CrossRef]
- Meck, J.V.; et al. Episodes of supraventricular tachycardia in astronauts during spaceflight. The American Journal of Cardiology 2001, 87, 547–550. [Google Scholar] [CrossRef]
- Cucinotta, F.A.; et al. Space radiation and cataracts in astronauts. Radiation Research 2001, 156, 460–466. [Google Scholar] [CrossRef]
- Kanas, N.; Manzey, D. Space Psychology and Psychiatry, 2 ed.; Springer: New York, 2008. [Google Scholar]
- Sargsyan, A.E.; et al. Ultrasound for spaceflight and remote environments. Critical Ultrasound Journal 2005, 7, 1–10. Please verify year, journal volume, and DOI; the supplied metadata appears inconsistent. [CrossRef]
- Cosper, P.F.; et al. Optimizing healthcare in space: The role of ultrasound imaging in long-duration missions. npj Microgravity 2024, 10, 15. [Google Scholar] [CrossRef]
- Smith, S.M.; et al. Benefits for bone from resistance exercise and nutrition in long-duration spaceflight. Journal of Bone and Mineral Research 2012, 27, 1896–1906. [Google Scholar] [CrossRef]
- Loehr, J.A.; et al. Physical training for long-duration spaceflight. Aerospace Medicine and Human Performance 2015, 86, A14–A23. [Google Scholar] [CrossRef]
- Schreurs, A.S.; et al. Dietary countermeasure mitigates simulated spaceflight-induced bone loss and radiation damage. Scientific Reports 2020, 10, 4173. [Google Scholar] [CrossRef]
- Masson, P.; et al. Advanced biosensors for monitoring astronauts’ health during long-duration space missions. Biosensors and Bioelectronics 2018, 111, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Quintero, V.; et al. Wearable biosensors for health monitoring: Advances in materials and integration. Nanoscale Horizons 2025, 10, 456–476. [Google Scholar] [CrossRef]
- Kramer, A.H.; et al. Optic nerve sheath diameter and radiologic measures of raised intracranial pressure. Journal of Neuroimaging 2013, 23, 78–82. [Google Scholar] [CrossRef]
- Li, X.; et al. Microfluidic point-of-care testing for space and remote environments. Lab on a Chip 2019, 19, 3210–3225. [Google Scholar] [CrossRef]
- Friebe, M. Reverse Innovation: Circumvent Digital Health Transformation Issues. In Novel Innovation Design for the Future of Health; Friebe, M., Ed.; Springer: Cham, 2022. [Google Scholar] [CrossRef]
- Friebe, M.; Illanes, A. Advancements in Medical Imaging and Image-Guided Procedures: A Potential—Or Rather Likely—Paradigm Shift in Diagnosis and Therapy: Understand Disruption and Take Advantage of It! Applied Sciences 2023, 13, 9218. [Google Scholar] [CrossRef]
- Crum, W.; et al. AI-driven clinical decision support for deep space missions: Lessons from early NASA deployments. npj Digital Medicine 2025, 8, 10. DOI appears incomplete; please verify. [CrossRef]
- D., P.; et al. Robot-assisted surgery in space: pros and cons. A review from the surgeon’s point of view. NPJ microgravity 2021, 7, 56. [Google Scholar] [CrossRef]
- Satava, R.M.; et al. Surgical robotics and telesurgery in space medicine. Surgical Endoscopy 2001, 15, 1192–1195. [Google Scholar] [CrossRef]
- Loukas, M.; et al. Robot-assisted surgery in space: Pros and cons. npj Microgravity 2022, 8, 18. [Google Scholar] [CrossRef]
- Freeman, R.J.; et al. Design and evaluation of a multifunctional surgical device for space medicine. Acta Astronautica 2020, 171, 103–111. [Google Scholar] [CrossRef]
- Savonen, B.L.; Pearce, J.M. The competitive advantage of using 3D-printing in low-resource healthcare settings. Proceedings of the Proceedings of the Design Society: DESIGN Conference 2020, Vol. 1, 2541–2550. [Google Scholar]
- Bahadur, S.; et al. AI-enabled obstetric point-of-care ultrasound as an emerging tool in low-resource settings: A scoping review. Global Health: Science and Practice 2025, 13, e2025012. [Google Scholar]
- Ryan, K.; et al. AI-assisted ultrasound: Bridging innovation and access in low-resource settings. Global Health Innovation 2024, 2, 145–154. [Google Scholar]
- Moy, L.; et al. Overview of artificial intelligence in point-of-care ultrasound: New horizons for global health. Advances in Interventional Technology 2024, 6, 1–15. [Google Scholar]
- Topol, E.; et al. Overcoming barriers in the use of artificial intelligence in point-of-care ultrasound. npj Digital Medicine 2025, 8, 22. [Google Scholar] [CrossRef]
- Arent, K.; et al. Advancements in Medical Imaging and Image-GuideSelected Topics in Design and Application of a Robot for Remote Medical Examination with the Use of Ultrasonography and Ascultation from the Perspective of the REMEDI Project. Journal of Automation, Mobile Robotics and Intelligent Systems 2017, 11, 82–94. [Google Scholar] [CrossRef]
- Friebe, M. Haptic and Palpation Sensing for Robotic Surgery: Engineering Perspectives on Design and Integration. Sensors 2026, 26, 1126. [Google Scholar] [CrossRef] [PubMed]
- Heikenfeld, J.; et al. Universal flexible wearable biosensors for noninvasive health monitoring. ACS Applied Materials & Interfaces 2024, 16, 12345–12360. [Google Scholar] [CrossRef]
- Barbazzeni, B. Value Propositions for Future Health Developments: Digital, Portable, Connected, Experience-Enhancing, Supportive, Patient-Centric, and Affordable. In Novel Innovation Design for the Future of Health; Friebe, M., Ed.; Springer: Cham, 2022. [Google Scholar] [CrossRef]

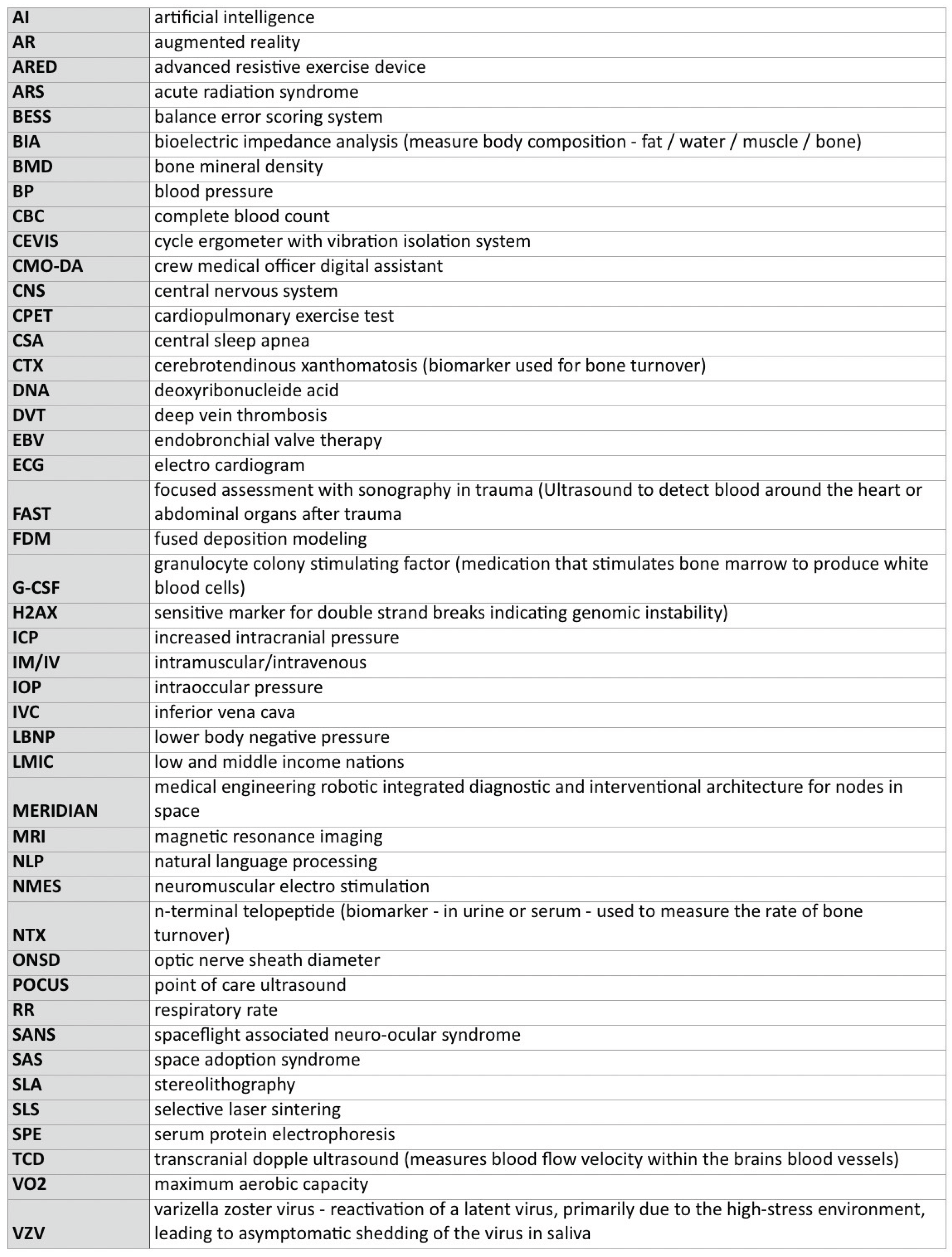
| Condition | Onset | In-space diagnosis | In-space therapy | Monitoring |
|---|---|---|---|---|
| Space Adaptation Syndrome | Days 1–4 | Clinical: nausea score, nystagmus, vestibular tests | Promethazine, scopolamine, fluid resupply | Symptom diary, vestibular testing |
| Cardiovascular deconditioning | 30+ days | Cardiac POCUS, ECG, BP | LBNP, aerobic exercise, fluid/salt loading | VO2 max (CPET), Holter, weekly POCUS |
| Muscle atrophy | 30+ days | Muscle ultrasound, BIA, grip strength | ARED resistance exercise, protein, NMES | Serial muscle ultrasound, dynamometry |
| Bone loss | 30+ days | Ultrasound densitometry, bone markers | Bisphosphonates, high-load exercise, Ca/Vit D | Bone biomarkers, cortical ultrasound |
| SANS / ICP elevation | 60+ days | Ocular POCUS (ONSD), fundoscopy, vision | LBNP, positioning, acetazolamide | Weekly ONSD, acuity, tonometry |
| DVT / thromboembolism | 60+ days | Venous POCUS | Anticoagulation, compression, ambulation | Serial POCUS, D-dimer |
| Radiation injury / ARS | Ongoing | Dosimetry, CBC | G-CSF, antioxidants, storm shelter | CBC, DNA damage markers |
| Immune / viral reactivation | 30+ days | PCR, CBC differential | Antivirals, immune support | Serial viral load, lymphocytes |
| Brain structural change | 180+ days | ONSD, TCD | Posture, LBNP, ICP-lowering therapy | Monthly TCD, ocular POCUS |
| Psychological deterioration | 90+ days | Psychometrics, behavior monitoring | AI-assisted CBT, sleep hygiene, light therapy | Weekly cognitive and mood tests |
| Renal calculi | 60+ days | Renal POCUS | Hydration, thiazides, citrate | Serial renal ultrasound |
| Orthostatic intolerance | Post-flight | Stand test, HR/BP | Fluid loading, compression, fludrocortisone | Daily standing HR/BP |
| Acute trauma / appendicitis | Any | FAST POCUS, exam | Antibiotics, robotic-assisted surgery | Labs, serial exam |
| Data domain | Specific inputs | Modality |
|---|---|---|
| Continuous vitals | ECG, SpO2, HR, RR, skin temp, BP | Wearable biosensors |
| Metabolic / lab | CBC, CRP, cortisol, glucose, creatinine, viral PCR, bone markers | Microfluidic POC |
| Imaging | POCUS video (cardiac, lung, abdominal, ocular, vascular) | Handheld ultrasound + AI |
| Ophthalmologic | ONSD, fundus images, IOP | POCUS + fundoscope |
| Neurological | TCD waveform, cognitive scores, pupillometry | TCD + tablet tests |
| Radiation | Cumulative dose, SPE alerts | Personal dosimeter |
| Behavioral | Sleep (actigraphy), mood scores, speech features | Wearables + NLP |
| Genomic | Baseline variants, epigenetic clock | Sequencing |
| Environmental | CO2, humidity, temp, particulates | Cabin sensors |
| Symptom NLP | Self-reported symptoms, questionnaires | Voice/text interface |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




