Submitted:
02 April 2026
Posted:
03 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
- Without nanobubble injection
- With air-based nanobubble injection
- With pure oxygen-assisted nanobubble injection
2.1. Experimental Setup
2.2. Operating Conditions
2.3. Measurement Methods
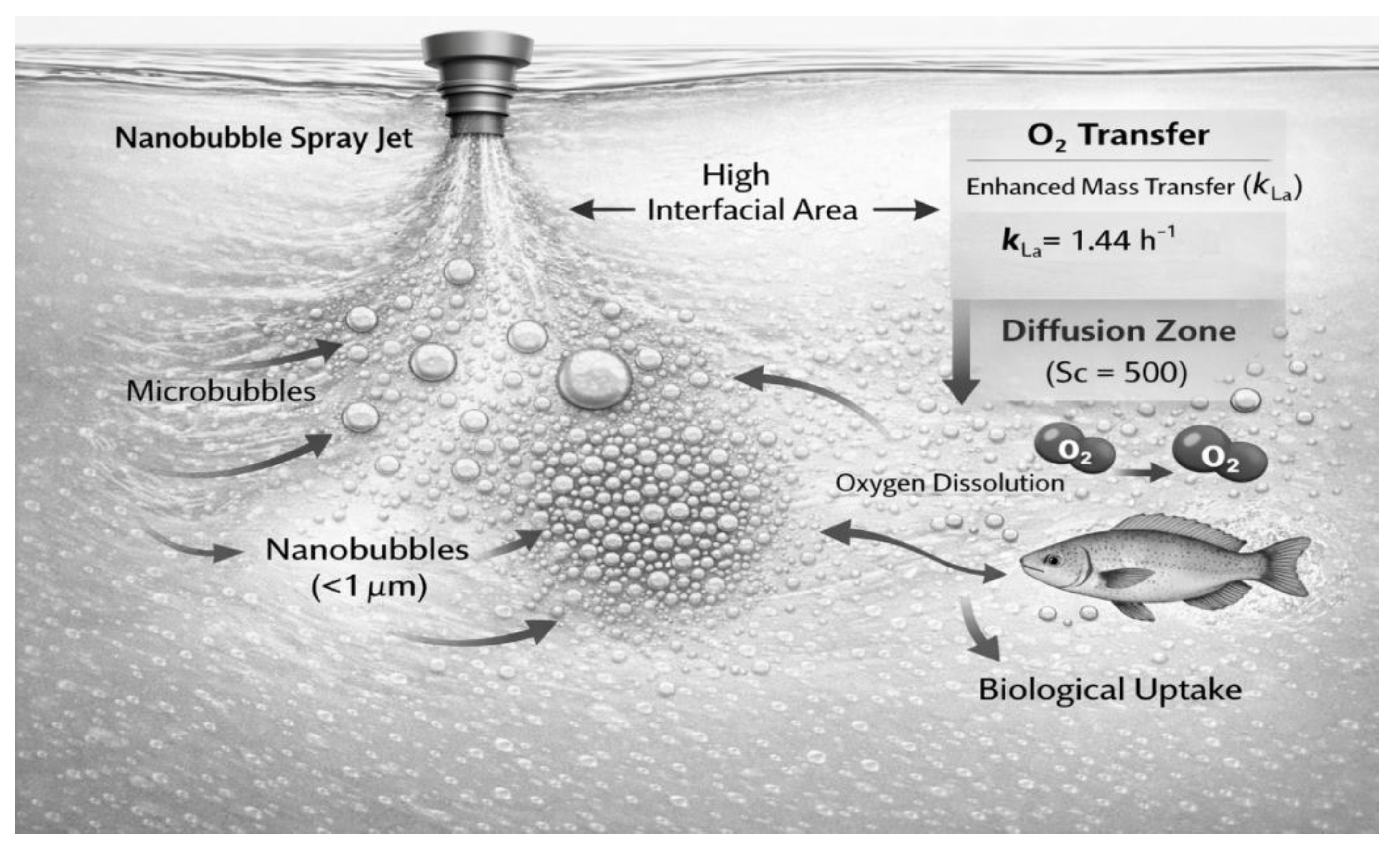
2.4. Oxygen Transfer Analysis
2.5. Specific oxygen transfer efficiency (SOTE)
3. Results
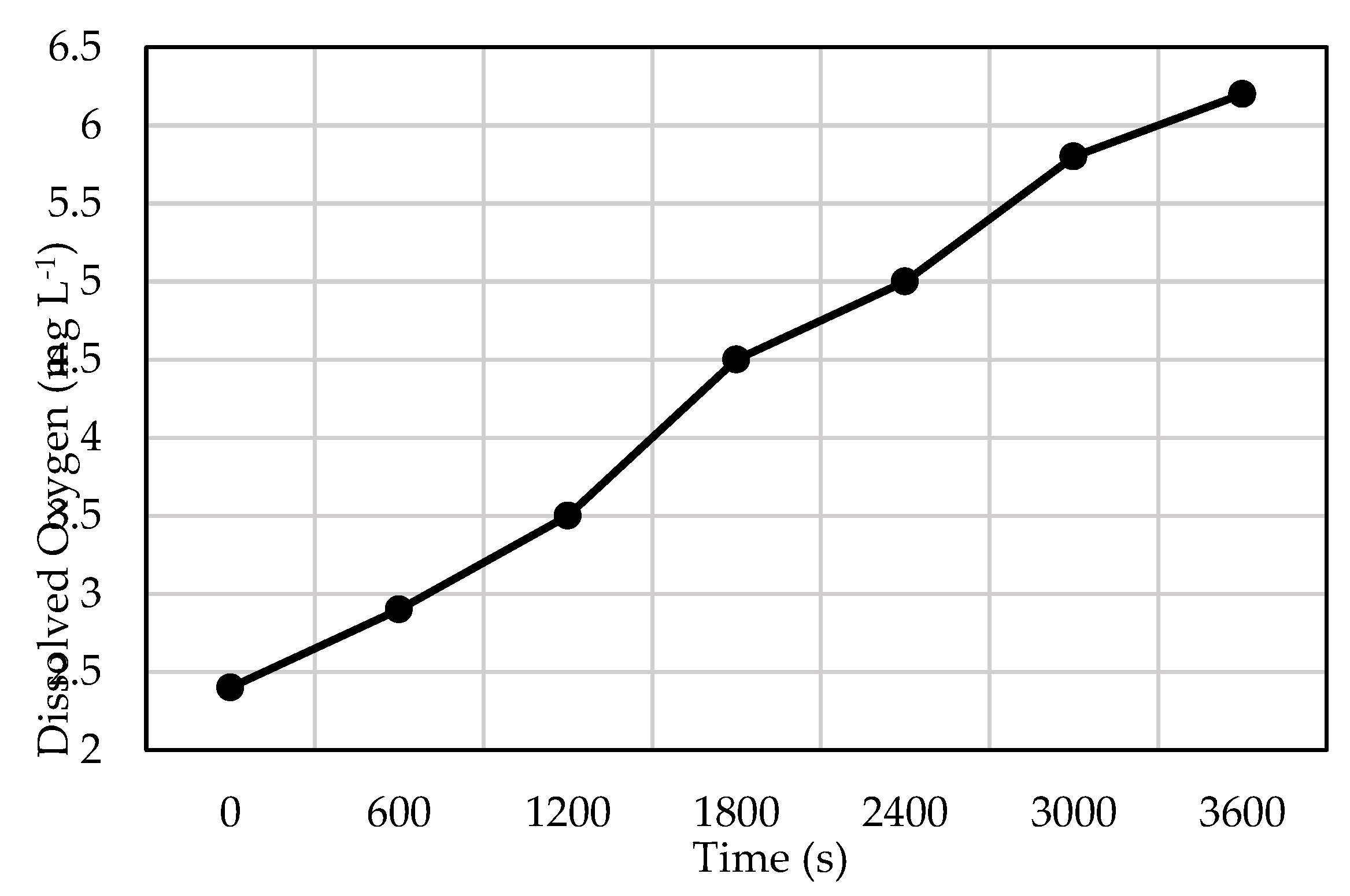
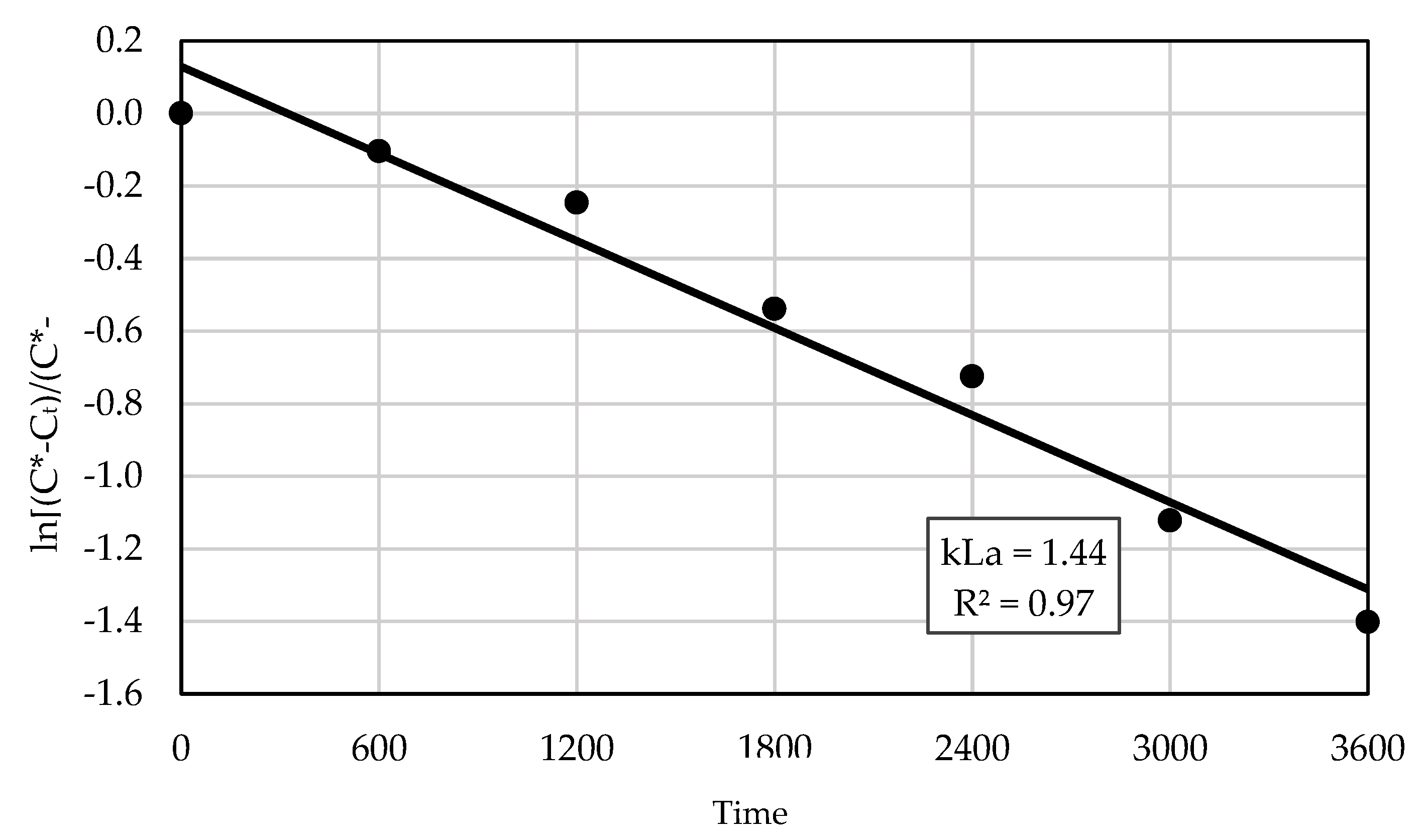
3.1. Overall Oxygen Transfer Behavior and Kinetics
3.2. Energy Performance (SOTE)
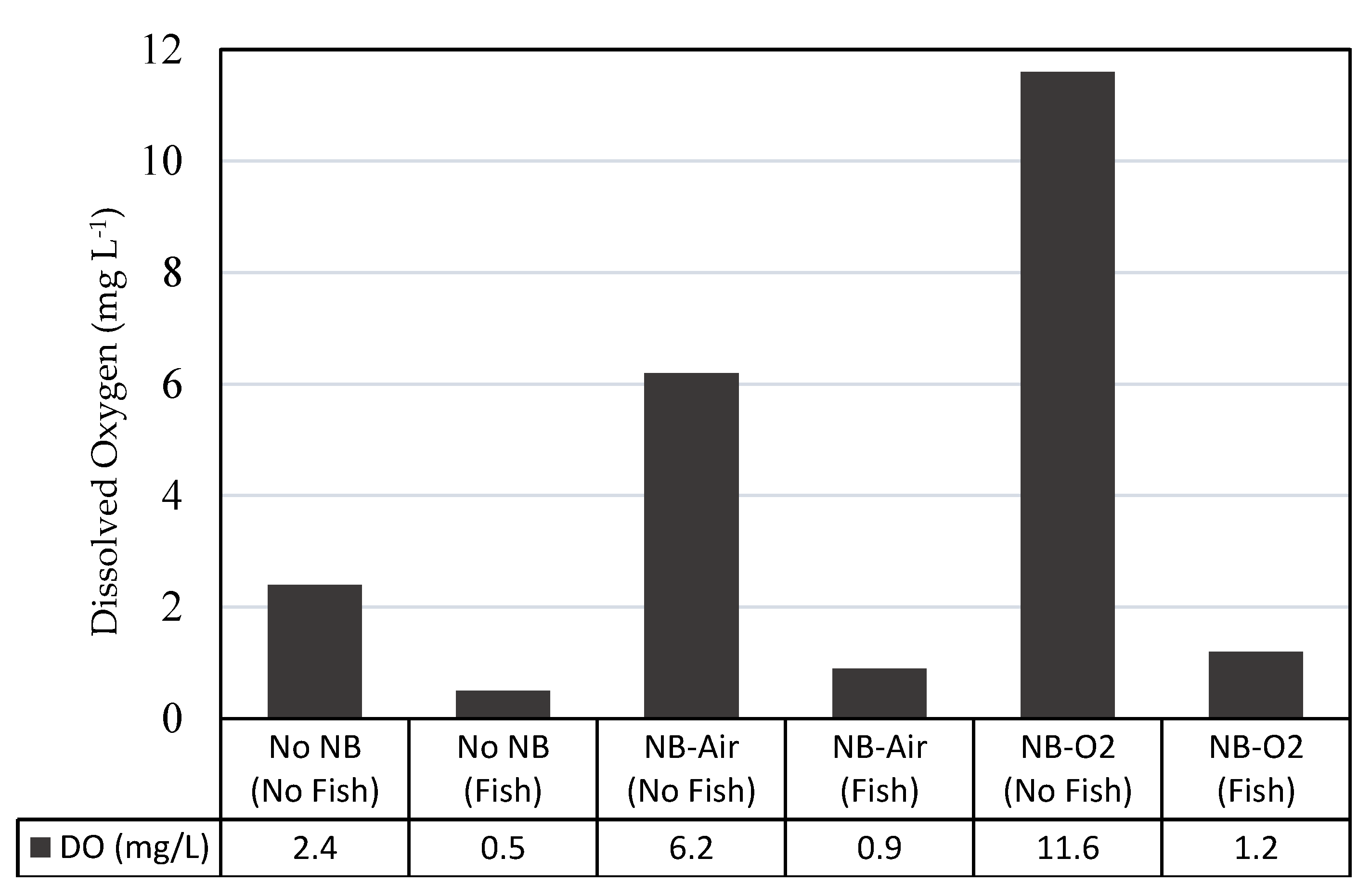
3.3. Effect of Gas Composition and Nanobubble Characteristics
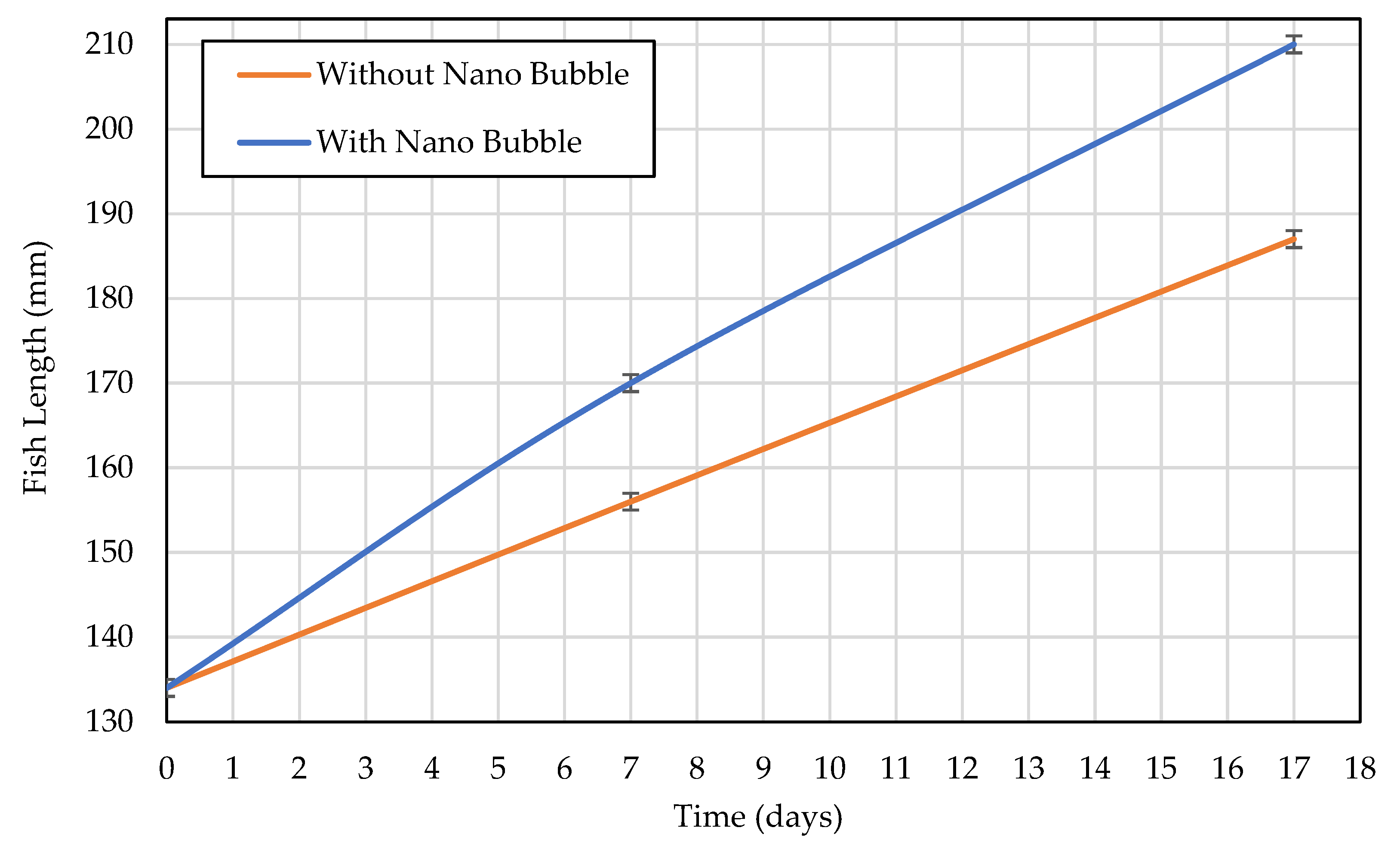
3.4. Biological Validation of Oxygen Transfer Enhancement
3.5. Hydrodynamic and Dimensionless Analysis
3.6. Comparative Performance Evaluation
3.7. Experimental Uncertainty
3.7.1. Uncertainty of Oxygen Mass Transfer
3.7.2. Uncertainty of SOTE
4. Discussion
4.1. Overall Oxygen Transfer Behavior and Kinetics
4.2. Energy Performance Interpretation
4.3. Role of Gas Composition and Nanobubbles
4.4. Biological Implications
4.5. Hydrodynamic Interpretation
4.6. Comparative Interpretation
4.7. Engineering Interpretation and Uncertainty
5. Conclusions
Acknowledgments
References
- Boyd, C. E., Torrans, E. L., & Tucker, C. S. (2017). Dissolved Oxygen and Aeration in Ictalurid Catfish Aquaculture. Journal of the World Aquaculture Society, 49(1), 7–70. [CrossRef]
- Elnady, M., Elwahed, R. A., & Gad, G. (2016). Effect of Dietary Protein Levels on Dissolved Oxygen Dynamics and Growth Performance of Nile Tilapia. Egyptian Journal of Agricultural Sciences, 67(1), 42–53. [CrossRef]
- Yanuhar, U., Musa, M., Evanuarini, H., Wuragil, D. K., & Permata, F. S. (2022). Water Quality in Koi Fish (Cyprinus Carpio) Concrete Ponds with Filtration in Nglegok District, Blitar Regency. Universal Journal of Agricultural Research, 10(6), 814–820. [CrossRef]
- Balami, S. (2021). Recirculation Aquaculture Systems: Components, Advantages, and Drawbacks. Tropical Agroecosystems, 2(2), 104–109. [CrossRef]
- Abdullah, S. (2019). Environmental Impacts of Commercial Shrimp Farming in Coastal Zone of Bangladesh and Approaches for Sustainable Management. International Journal of Environmental Sciences & Natural Resources, 20(3). [CrossRef]
- Laktuka, K., Kalnbaļķīte, A., Sniega, L., Logins, K., & Lauka, D. (2023). Towards the Sustainable Intensification of Aquaculture: Exploring Possible Ways Forward. Sustainability, 15(24), 16952. [CrossRef]
- Dayrit, G. B., Cruz, E. M. V, Rodkhum, C., Mabrok, M., Ponza, P., & Santos, M. D. (2023). Potential Influence of Shading in Freshwater Ponds on the Water Quality Parameters and the Hematological and Biochemical Profiles of Nile Tilapia (Oreochromis Niloticus Linnaeus, 1758). Fishes, 8(6), 322. [CrossRef]
- Han, P., Lu, Q., Fan, L., & Zhou, W. (2019). A Review on the Use of Microalgae for Sustainable Aquaculture. Applied Sciences, 9(11), 2377. [CrossRef]
- Agus, M., Darmanto, Y. S., & Prayitno, S. B. (2016). The Dynamics of Water Quality Soft-Shelled Crab Aquaculture in Pemalang Regency, Middle Java, Indonesia. International Journal of Science and Research (Ijsr), 5(2), 1448–1451. [CrossRef]
- Vo, T. T. E., Ko, H., Huh, J., & Park, N. (2021). Overview of Solar Energy for Aquaculture: The Potential and Future Trends. Energies, 14(21), 6923. [CrossRef]
- Chellapandi, P. (2021). Development of Top-Dressing Automation Technology for Sustainable Shrimp Aquaculture in India. Discover Sustainability, 2(1). [CrossRef]
- Beg, M. M., Roy, S. M., Kar, A., Mukherjee, C. K., Bhagat, S., & Tanveer, M. (2024). Study on Recirculating Aquaculture System (RAS) in Organic Fish Production. Iop Conference Series Earth and Environmental Science, 1391(1), 12013. [CrossRef]
- Mauladani, S., Rahmawati, A. I., Absirin, M. F., Saputra, R. N., Pratama, A. F., Hidayatullah, A., Dwiarto, A., Syarif, A. F., Junaedi, H., Cahyadi, D., Saputra, H. K. H., Prabowo, W. T., Kartamiharja, U. K. A., Noviyanto, A., & Rochman, N. T. (2020). Economic Feasibility Study of Litopenaeus Vannamei Shrimp Farming: Nanobubble Investment in Increasing Harvest Productivity. Jurnal Akuakultur Indonesia, 19(1), 30–38. [CrossRef]
- Tran, N. L. H., Lam, T. Q., Duong, P. V. Q., Doan, L., Vu, M. P., Nguyen, K. H. P., & Nguyen, K. T. (2023). Review on the Significant Interactions Between Ultrafine Gas Bubbles and Biological Systems. Langmuir, 40(1), 984–996. [CrossRef]
- Nghĩa, N. H., Van, P. T., Giang, P. T., Hanh, N. T., St-Hilaire, S., & Domingos, J. A. (2021). Control Of Vibrioparahaemolyticus(AHPND Strain) and Improvement of Water Quality Using Nanobubble Technology. Aquaculture Research, 52(6), 2727–2739. [CrossRef]
- Dien, L. T., Linh, N. V, Sangpo, P., Senapin, S., St-Hilaire, S., Rodkhum, C., & Dong, H. T. (2021). Ozone Nanobubble Treatments Improve Survivability of Nile Tilapia (Oreochromis Niloticus) Challenged With a Pathogenic Multi-drug-resistant Aeromonas Hydrophila. Journal of Fish Diseases, 44(9), 1435–1447. [CrossRef]
- Wang, S., Liu, Y., Lyu, T., Pan, G., & Li, P. (2020). Aquatic Macrophytes in Morphological and Physiological Responses to the Nanobubble Technology Application for Water Restoration. Acs Es&t Water, 1(2), 376–387. [CrossRef]
- Domingos, J. A., Huang, Q., Liu, H., Dong, H. T., Khongcharoen, N., Van, P. T., Nghĩa, N. H., Giang, P. T., Viet, P. T., & St-Hilaire, S. (2021). Air-Nanobubbles Ineffective to Reduce Pathogenic Bacteria in Fresh and Brackish Waters. [CrossRef]
- Chary, K., Brigolin, D., & Callier, M. D. (2022). Farm-scale Models in Fish Aquaculture – An Overview of Methods and Applications. Reviews in Aquaculture, 14(4), 2122–2157. [CrossRef]
- Jia, M., Farid, M. U., Kharraz, J. A., Kumar, N. M., Chopra, S. S., Jang, A., Chew, Y. M. J., Khanal, S. K., Chen, G., & An, A. K. (2023). Nanobubbles in Water and Wastewater Treatment Systems: Small Bubbles Making Big Difference. Water Research, 245, 120613. [CrossRef]
- American Public Health Association (APHA), (2017). Standard Methods for the Examination of Water and Wastewater (23rd ed.). Washington, DC: APHA.
- Metcalf & Eddy Inc. George Tchobanoglous et al. Wastewater Engineering: Treatment and Resource Recovery, McGraw-Hill Education (2014). ISBN: 978-0-07-340118-8.
- Lekang, O. I. (2007). Aquaculture Engineering. Blackwell Publishing.
- ASCE 18-96: Standard Guidelines for In-Process Oxygen Transfer Testing.
- Chemical Engineering Journal, Zhang et al., (2016) — Microbubble mass transfer enhancement.
- Separation and Purification Technology, Li et al., (2019) — Nanobubble oxygen dissolution.
- Boyd, C.E. and Tucker, C.S. (1998) Pond Aquaculture and Water Quality Management. Kluwer Academic Pub., London, 44-48. [CrossRef]
- D Rosso, LE Larson, MK Stenstrom, Aeration of large-scale municipal wastewater treatment plants: state of the art (2008), Water Science and Technology,.
- Frank R. Spellman, The Science of Water, Concepts and Applications, 2nd Ed., (2007). [CrossRef]
- Bird, R., Stewart, W. and Lightfoot, E. (2002) Transport Phenomena. 2nd Edition, John Wiley and Sons, New York.
- Incropera, F. P., DeWitt, D. P., Bergman, T. L., & Lavine, A. S. (2007). Fundamentals of Heat and Mass Transfer (6th ed.). Hoboken, NJ: John Wiley & Sons.
- Coulson, J.M., and Richardson, J.F. (2005). “Chemical Engineering”, 4th ed, vol. 6, Pergamon Press, Oxford.
- Hideki Tsuge, (2014), Micro- and Nanobubbles: Fundamentals and Applications, Pan Stanford Publishing.
- Holman, J. P. (2012). Experimental Methods for Engineers (8th ed.). McGraw-Hill Education.
- Taylor, J. R. (1997). An Introduction to Error Analysis: The Study of Uncertainties in Physical Measurements (2nd ed.). University Science Books.
- ASME PTC 19.1 (1998). Test Uncertainty, Supplement to ASME Performance Test Codes.
- Boyd, C.E. (2020). Water Quality: An Introduction (2nd ed.). Springer.
- ASCE 2-06 (2007). Measurement of Oxygen Transfer in Clean Water (ASCE/EWRI 2-06). Reston, VA: American Society of Civil Engineers.
- Agarwal, A., Ng, W.J. and Liu, Y. (2011) Principle and Applications of Microbubble and Nanobubble Technology for Water Treatment. Chemosphere, 84, 1175-1180. [CrossRef]
- Michael B. Timmons; James M. Ebeling (2013), Recirculating Aquaculture, 3rd Edition, Ithaca Publishing Company, LLC.
- Xu, Y., Li, L., Lou, S., Tian, J., Sun, S.-H., Li, X., & Li, Y. (2022). Effects of Nano-Aerators on Microbial Communities and Functions in the Water, Sediment, and Shrimp Intestine in Litopenaeus Vannamei Aquaculture Ponds. Microorganisms, 10(7), 1302. [CrossRef]
- Das, S. K., Mondal, B., Sarkar, U. K., Das, B. K., & Borah, S. (2022). Understanding and Approaches Towards Circular Bio-economy of Wastewater Reuse in Fisheries and Aquaculture in India: An Overview. Reviews in Aquaculture, 15(3), 1100–1114. [CrossRef]
- English, N. J. (2022). Sustainable Exploitation and Commercialization of Ultradense Nanobubbles: Reinventing Liquidity. Acs Sustainable Chemistry & Engineering, 10(11), 3383–3386. [CrossRef]
- Mehrim, A. I., & Refaey, M. M. (2023). An Overview of the Implication of Climate Change on Fish Farming in Egypt. Sustainability, 15(2), 1679. [CrossRef]
- Nisar, U., Peng, D., Mu, Y., & Sun, Y. (2022). A Solution for Sustainable Utilization of Aquaculture Waste: A Comprehensive Review of Biofloc Technology and Aquamimicry. Frontiers in Nutrition, 8. [CrossRef]
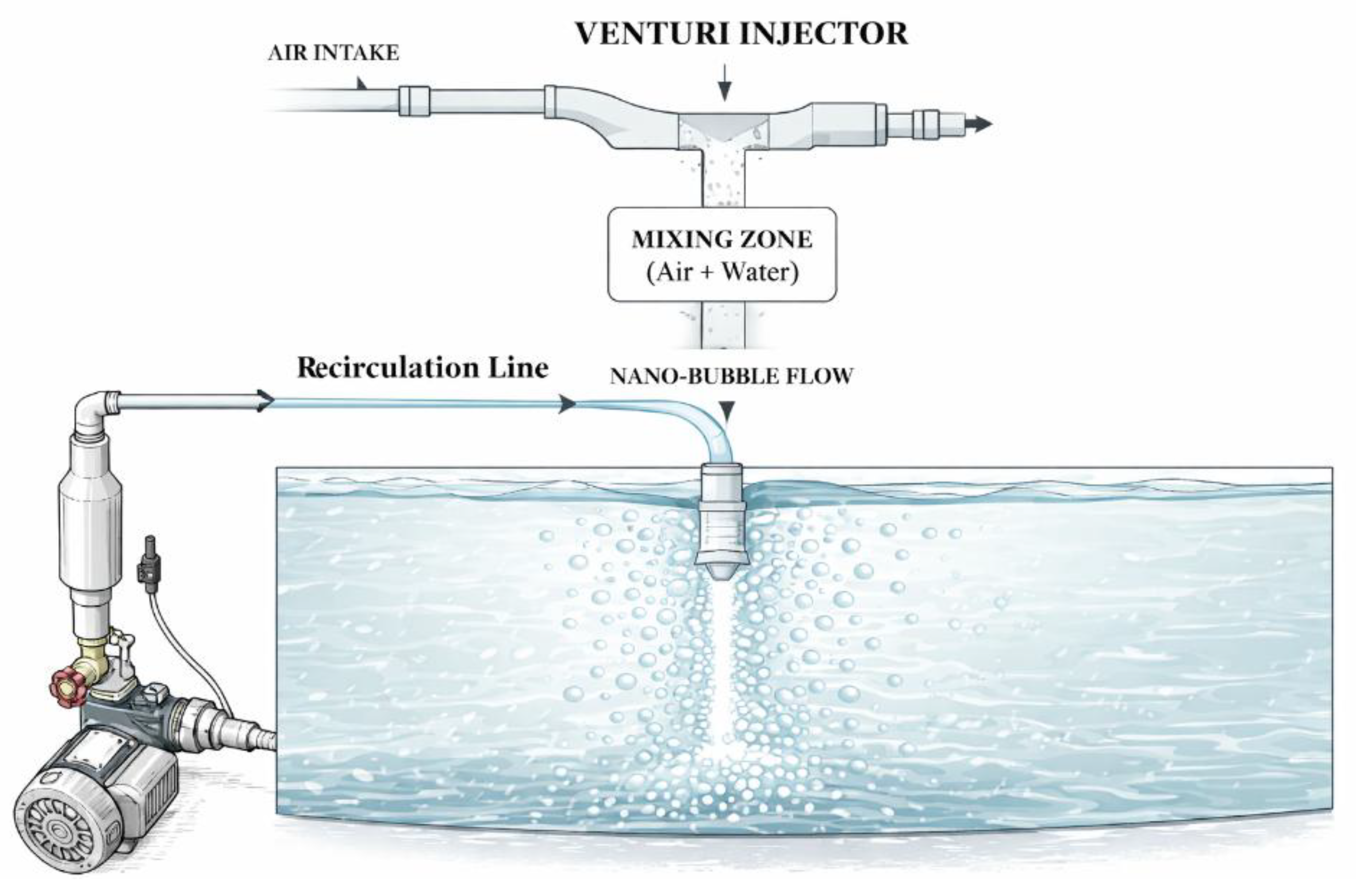
| No. | Parameter | Instrument/Specification | Accuracy/Resolution |
|---|---|---|---|
| 1. | Pump Power (P) | Shimizu PS-128 BIT (125 W) | ±9% |
| 2. | Dissolved Oxygen (DO) | DO-5512SD meter | ±0.1 mg L⁻¹ |
| 3. | Initial DO (C₀) | 2.4 mg L⁻¹ | derived from DO meter resolution |
| 4. | Final DO (Cₜ) | 6.2 mg L⁻¹ | derived from DO meter resolution |
| 5. | Pond Volume (V) | 2512 L (D = 2 m, H = 0.8 m) | ±3% |
| 6. | Measurement time (t) | 3600 s | ±0.1 s |
| 7. | Flowrate (Q) | 0.16 (L/s) | used for hydrodynamic analysis |
| System Type | kLa (h⁻¹) | SOTE (gO₂/kWh) | Scale | Reference |
|---|---|---|---|---|
| Fine bubble diffuser | 0.5 – 5.0 | 800 – 1500 | Wastewater plant | [27,36] |
| Mechanical surface aerator | 0.3 – 2.0 | 500 – 1200 | Large pond | [26] |
| Coarse bubble aerator | 0.2 – 1.5 | 400 – 900 | Municipal WWTP | [21] |
| Venturi injector | 0.1 – 0.8 | 200 – 600 | Small systems | [37] |
| Industrial nanobubble system | 0.5 – 3.0 | 600 – 1300 | Treatment plant | [32] |
| Microbubble generator | 0.4 – 2.5 | 500 – 1100 | Lab-scale | [38] |
| Air pump | 0.05 – 0.3 | 50 – 200 | Small-scale air pump | [39] |
| Nanobubble spray system (this study) | ≈ 1.44 | ≈ 76.4 | 2.5 m³ pond | Present study |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




