Submitted:
02 April 2026
Posted:
03 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Description of the Work and Literature Search Strategy
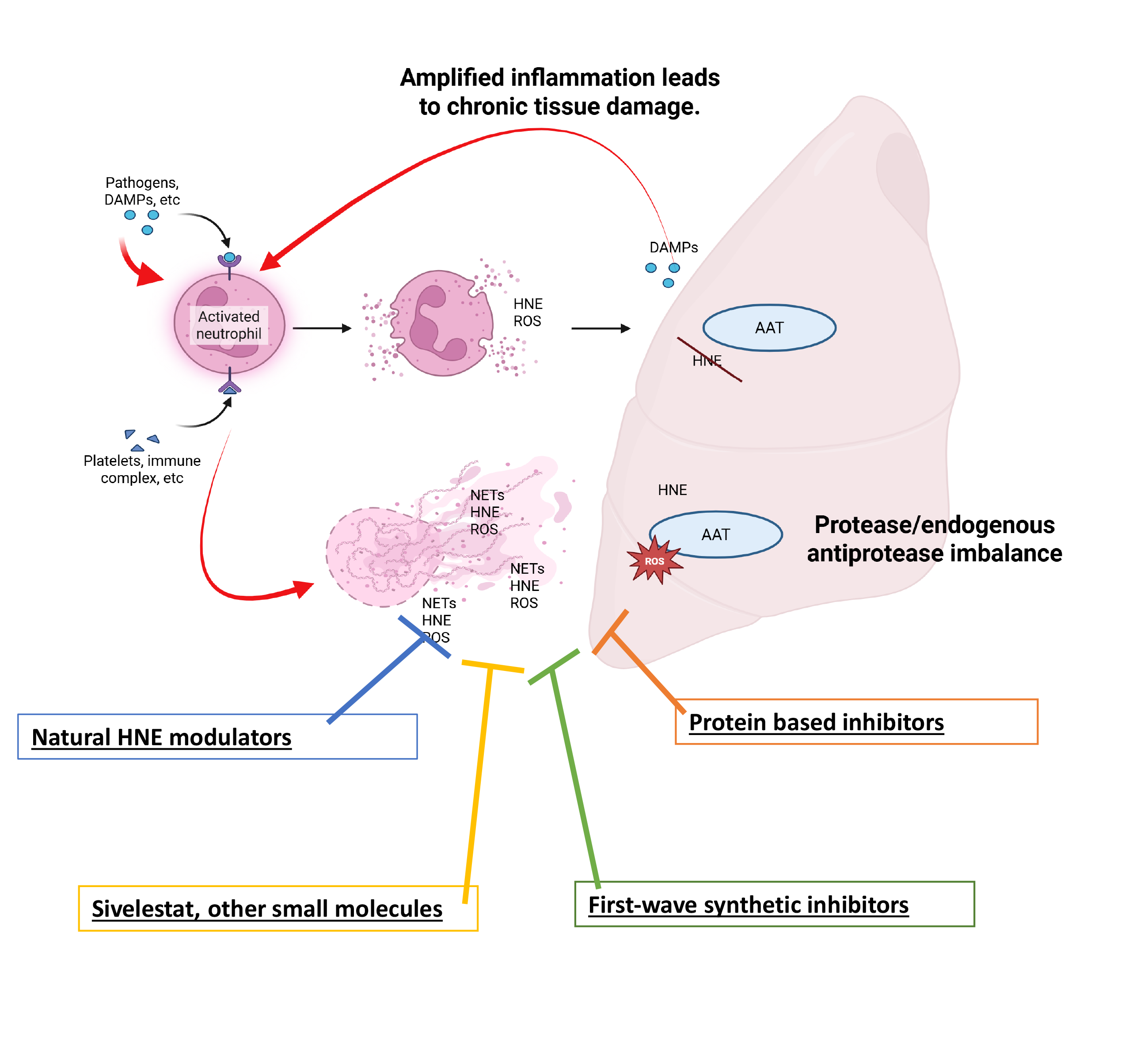
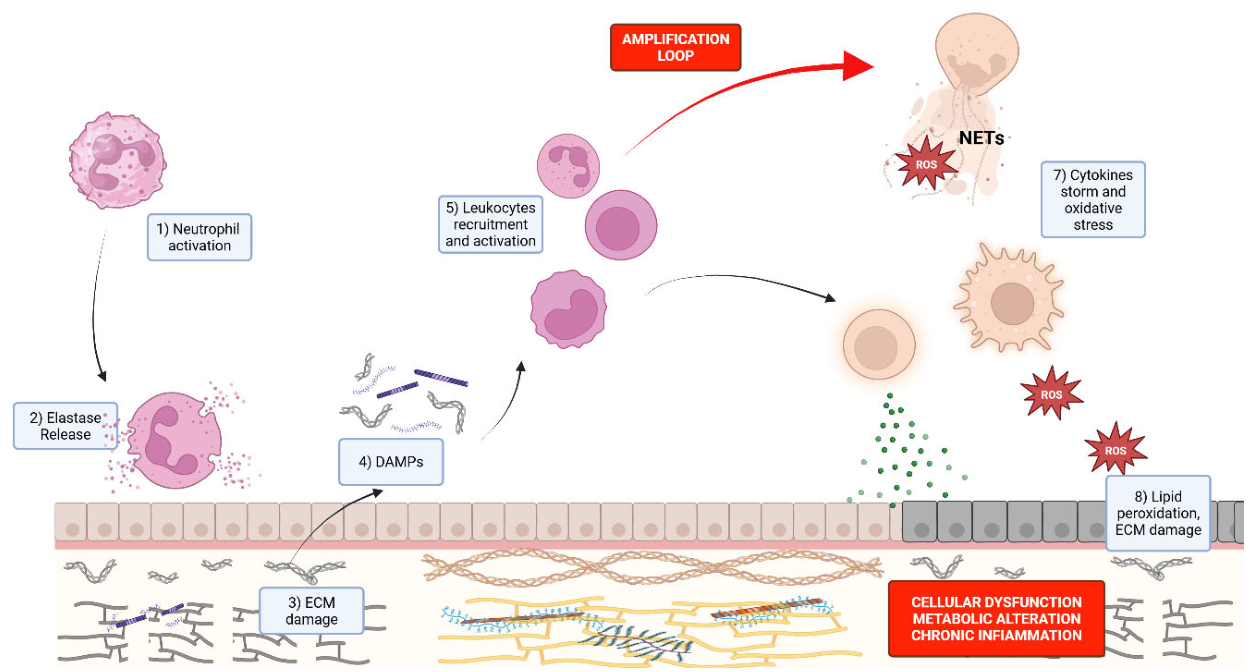
3. Neutrophils, Elastase and the “Protein Storm”
4. Factors Contributing to Inadequate Inhibition of HNE by Endogenous Inhibitors
5. Why Nature-Inspired Inhibition Has Not Solved the HNE Problem?
6. Synthetic Inhibitors of HNE as Drug Candidates: Definition and General Concepts
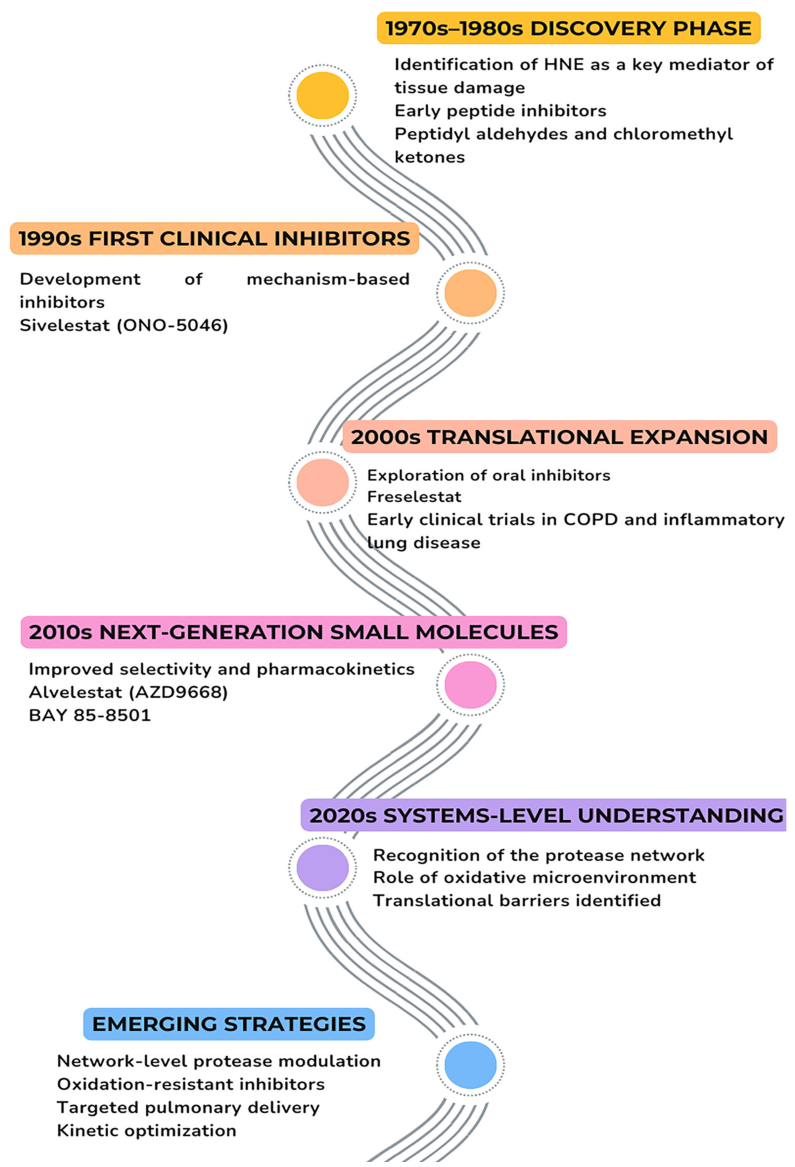
7. First-Wave Synthetic Strategies Targeting HNE
8. Emergence of Modern Synthetic HNE Inhibitors
9. Sivelestat and the Protease Inhibition Paradox: Success in Chronic Disease, Struggle in Acute Critical Care
9.1. Protease Inhibition in Chronic Diseases vs. Acute Critical Care
9.2. The Temporal and Stratification Mismatch
9.3. Reinterpreting the Sivelestat Experience
10. From First-Generation Lessons to Next-Generation Design
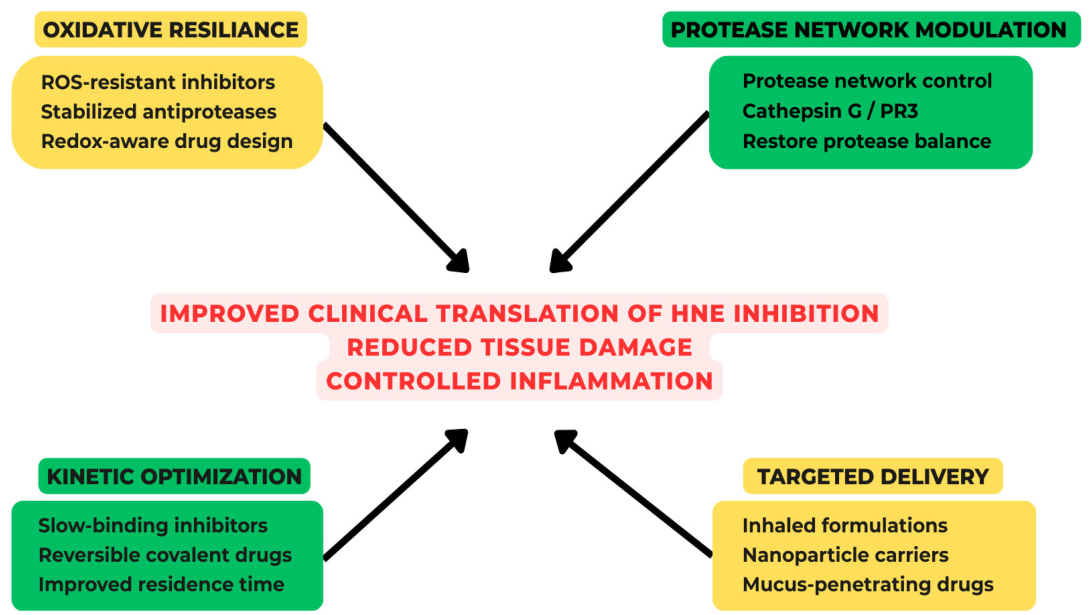
11. Overcoming First-Generation Limitations: Design Drivers for Next-Generation HNE Inhibitors
12. Small-Molecule Optimization Trajectories: From Transitional Scaffolds to Clinically Advanced Candidate
12.1. Transitional Chemical Space and Early Rational Optimization
12.2. Second-Generation Refinement: The Case of Alvelestat
12.3. High-Affinity Optimization and Clinical Advancement: BAY 85-8501
13. Translational Lessons and Pharmacological Differentiation Across Small-Molecule HNE Inhibitors
14. Scaffold Diversification and Medicinal Chemistry Expansion in HNE Inhibitor Development
15. Beyond Small Molecules: Peptide-Based and Biologically Inspired Inhibition Strategies
16. Emerging Combination or Multi-Mechanism Strategies
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAT | α1-antitrypsin |
| AATD | α1-antitrypsin deficiency |
| ARDS | Acute respiratory distress syndrome |
| CF | Cystic fibrosis |
| COPD | Chronic obstructive pulmonary diseases |
| EGFR | Epidermal growth factor receptor |
| DAMPs | Damage associated molecular patterns |
| HNE | Human neutrophil elastase |
| NETs | Neutrophil extracellular trap |
| ROS | Reactive oxygen species |
| SLPI | Secretory leukoprotease inhibitor |
| 4-HNE | 4-hydroxy-2-nonenal |
References
- Nie, J.; Zhou, L.; Tian, W.; Liu, X.; Yang, L.; Yang, X.; Zhang, Y.; Wei, S.; Wang, D.W.; Wei, J. Deep insight into cytokine storm: from pathogenesis to treatment. Signal Transduct. Target. Ther. 2025, 10, 112. [CrossRef]
- Voynow, J.A.; Shinbashi, M. Neutrophil elastase and chronic lung disease. Biomolecules 2021, 11, 1065. [CrossRef]
- Kelly-Robinson, G.A.; Reihill, J.A.; Lundy, F.T.; McGarvey, L.P.; Lockhart, J.C.; Litherland, G.J.; Thornbury, K.D.; Martin, S.L. The serpin superfamily and their role in the regulation and dysfunction of serine protease activity in COPD and other chronic lung diseases. Int. J. Mol. Sci. 2021, 22, 6351. [CrossRef]
- Viglio, S.; Bak, E.G.; Schouten, I.G.M.; Iadarola, P.; Stolk, J. Protease-specific biomarkers to analyse protease inhibitors for emphysema associated with alpha-1 antitrypsin deficiency: an overview of current approaches. Int. J. Mol. Sci. 2021, 22, 1065. [CrossRef]
- Liu, X.; Ucakar, B.; Vanvarenberg, K.; Marbaix, E.; Vanbever, R. Impact of administration route and PEGylation on alpha-1 antitrypsin augmentation therapy. J. Control. Release 2025, 382, 113643. [CrossRef]
- Oriano, M.; Amati, F.; Gramegna, A.; De Soyza, A.; Mantero, M.; Sibila, O.; Chotirmall, S.H.; Voza, A.; Marchisio, P.; Blasi, F.; et al. Protease–antiprotease imbalance in bronchiectasis. Int. J. Mol. Sci. 2021, 22, 5996. [CrossRef]
- Lucas, S.D.; Costa, E.; Guedes, R.C.; Moreira, R. Targeting COPD: advances on low-molecular-weight inhibitors of human neutrophil elastase. Med. Res. Rev. 2013, 33, E73–E101. [CrossRef]
- Tousif, M.I.; Nazir, M.; Riaz, N.; Saleem, M.; Tauseef, S.; Azam, S.M.; Arfan Yawer, M.; Zengin, G. Terpenoids as human neutrophil elastase inhibitors: a comprehensive review. ChemBioChem 2023, 24, e202300346. [CrossRef]
- Trainor, D.A. Synthetic inhibitors of human neutrophil elastase. Trends Pharmacol. Sci. 1987, 8, 303–307. [CrossRef]
- Ohbayashi, H. Current synthetic inhibitors of human neutrophil elastase in 2005. Expert Opin. Ther. Pat. 2005, 15, 759–771. [CrossRef]
- Crocetti, L.; Quinn, M.T.; Schepetkin, I.A.; Giovannoni, M.P. A patenting perspective on human neutrophil elastase inhibitors (2014–2018) and their therapeutic applications. Expert Opin. Ther. Pat. 2019, 29, 555–578. [CrossRef]
- Ocampo-Gallego, J.S.; Pedroza-Escobar, D.; Caicedo-Ortega, R.A.; Berumen-Murra, M.T.; Novelo-Aguirre, A.L.; de Sotelo-León, R.D.; Delgadillo-Guzmán, D. Human neutrophil elastase inhibitors: classification, biological-synthetic sources and their relevance in related diseases. Fundam. Clin. Pharmacol. 2024, 38, 13–32. [CrossRef]
- Wang, Z.; Chen, F.; Zhai, R.; Zhang, L.; Su, L.; Lin, X.; Thompson, T.; Christiani, D.C. Plasma neutrophil elastase and elafin imbalance is associated with acute respiratory distress syndrome development. PLoS ONE 2009, 4, e4380. [CrossRef]
- Cagnone, M.; Piloni, D.; Ferrarotti, I.; Di Venere, M.; Viglio, S.; Magni, S.; Bardoni, A.; Salvini, R.; Fumagalli, M.; Iadarola, P.; et al. Balance between proteases and α1-antitrypsin in bronchoalveolar lavage fluid of lung transplant recipients. High-Throughput 2019, 8, 5. [CrossRef]
- Barrera, G.; Pizzimenti, S.; Ciamporcero, E.S.; Daga, M.; Ullio, C.; Arcaro, A.; Cetrangolo, G.P.; Ferretti, C.; Dianzani, C.; Lepore, A.; et al. Role of 4-hydroxynonenal-protein adducts in human diseases. Antioxid. Redox Signal. 2015, 22, 1681–1702. [CrossRef]
- Prasad, B. Chronic obstructive pulmonary disease (COPD). Int. J. Pharm. Res. Technol. 2020, 10, 67–71.
- Galiniak, S.; Mołoń, M.; Rachel, M. Links between disease severity, bacterial infections and oxidative stress in cystic fibrosis. Antioxidants 2022, 11, 887. [CrossRef]
- Meyer, N.J.; Gattinoni, L.; Calfee, C.S. Acute respiratory distress syndrome. Lancet 2021, 398, 622–637. [CrossRef]
- Balogh, E.; Veale, D.J.; McGarry, T.; Orr, C.; Szekanecz, Z.; Ng, C.T.; Fearon, U.; Biniecka, M. Oxidative stress impairs energy metabolism in rheumatoid arthritis. Arthritis Res. Ther. 2018, 20, 95. [CrossRef]
- Ferreira, A.V.; Perelshtein, I.; Perkas, N.; Gedanken, A.; Cunha, J.; Cavaco-Paulo, A. Detection of human neutrophil elastase on wound dressings as marker of inflammation. Appl. Microbiol. Biotechnol. 2017, 101, 1443–1454. [CrossRef]
- Cipak Gasparovic, A.; Milkovic, L.; Borovic Sunjic, S.; Zarkovic, N. Cancer growth regulation by 4-hydroxynonenal. Free Radic. Biol. Med. 2017, 111, 226–234. [CrossRef]
- Korkmaz, B.; Attucci, S.; Jourdan, M.L.; Juliano, L.; Gauthier, F. Inhibition of neutrophil elastase by α1-protease inhibitor at neutrophil surface. J. Immunol. 2005, 175, 3329–3339. [CrossRef]
- Janciauskiene, S.; Wrenger, S.; Immenschuh, S.; Olejnicka, B.; Greulich, T.; Welte, T.; Chorostowska-Wynimko, J. The Multifaceted Effects of Alpha1-Antitrypsin on Neutrophil Functions. Front. Pharmacol. 2018, 9, 341. [CrossRef]
- Taggart, C.; Cervantes-Laurean, D.; Kim, G.; McElvaney, N.G.; Wehr, N.; Moss, J.; Levine, R.L. Oxidation of α1-antitrypsin causes loss of activity. J. Biol. Chem. 2000, 275, 27258–27265. [CrossRef]
- Li, Z.; Alam, S.; Wang, J.; Sandstrom, C.S.; Janciauskiene, S.J.; Mahadeva, R. Oxidized α1-antitrypsin stimulates MCP-1 release. Am. J. Physiol. Lung Cell. Mol. Physiol. 2009, 297, L388–L400. [CrossRef]
- Brand, P.; Schulte, M.; Wencker, M.; Herpich, C.H.; Klein, G.; Hanna, K.; Meyer, T. Lung deposition of inhaled alpha1-proteinase inhibitor. Eur. Respir. J. 2009, 34, 354–360. [CrossRef]
- Casaloti, L.G.; Santos, J.A.N. Inhibition of elastase by Sedum dendroideum extracts. Multidiscip. Sci. J. 2019, 7, 140–147. [CrossRef]
- Kacem, R. Phenolic compounds from medicinal plants as Natural anti-elastase products for the therapy of pulmonary emphysema. J. Med. Plants Res. 2013, 7, 3499–3507.
- Wittenauer, J.; Mäckle, S.; Sußmann, D.; Schweiggert-Weisz, U.; Carle, R. Polyphenols from grape pomace inhibit elastase. Fitoterapia 2015, 101, 179–187. [CrossRef]
- Tan, X.F.; Kim, D.W.; Song, Y.H.; Kim, J.Y.; Yuk, H.J.; Wang, Y.; Curtis-Long, M.J.; Park, K.H. Human neutrophil elastase inhibitory potential of flavonoids from Campylotropis hirtella and their kinetics. J. Enzyme Inhib. Med. Chem. 2016, 31, 16–22. [CrossRef]
- Uddin, Z.; Li, Z.; Song, Y.H.; Kim, J.Y.; Park, K.H. Visconata inhibits human neutrophil elastase. Tetrahedron Lett. 2017, 58, 2507–2511. [CrossRef]
- Saleem, M.; Nazir, M.; Hussain, H.; Tousif, M.I.; Elsebai, M.F.; Riaz, N.; Akhtar, N. Natural Phenolics as Inhibitors of the Human Neutrophil Elastase (HNE) Release: An Overview of Natural Anti-inflammatory Discoveries during Recent Years. Anti-Inflamm. Anti-Allergy Agents Med. Chem. 2018, 17, 70–94. [CrossRef]
- Tang, Y.; Wang, Y.; Pei, Z.; Li, W.; Zhang, D.; Liu, L.; Kong, L.; Liu, S.; Jiang, X.; Ma, H. A serine protease inhibitor from Musca domestica larva exhibits inhibitory activity against elastase and chymotrypsin. Biotechnol. Lett. 2016, 38, 1147–1153. [CrossRef]
- Luan, N.; Zhao, Q.; Duan, Z.; Ji, M.; Xing, M.; Zhu, T.; Mwangi, J.; Rong, M.; Liu, J.; Lai, R. Identification and Characterization of ShSPI, a Kazal-Type Elastase Inhibitor from the Venom of Scolopendra hainanum. Toxins 2019, 11, 708. [CrossRef]
- Wan, H.; Lee, K.S.; Kim, B.Y.; Yuan, M.; Zhan, S.; You, H.; Li, J.; Jin, B.R. A spider (Araneus ventricosus) chymotrypsin inhibitor that acts as an elastase inhibitor and a microbial serine protease inhibitor. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2013, 165, 36–41. [CrossRef]
- Ahmad, S.; Saleem, M.; Riaz, N.; Lee, Y.S.; Diri, R.; Noor, A.; Almasri, D.; Bagalagel, A.; Elsebai, M.F. The Natural Polypeptides as Significant Elastase Inhibitors. Front. Pharmacol. 2020, 11, 688. [CrossRef]
- Marinaccio, L.; Stefanucci, A.; Scioli, G.; Della Valle, A.; Zengin, G.; Cichelli, A.; Mollica, A. Peptide Human Neutrophil Elastase Inhibitors from Natural Sources: An Overview. Int. J. Mol. Sci. 2022, 23, 2924. [CrossRef]
- Cantin, A.M.; Woods, D.E. Aerosolized prolastin suppresses bacterial proliferation. Am. J. Respir. Crit. Care Med. 1999, 160, 1130–1135. [CrossRef]
- Gottlieb, D.J.; Luisetti, M.; Stone, P.J.; Allegra, L.; Cantey-Kiser, J.M.; Grassi, C.; Snider, G.L. Short-term supplementation therapy in AAT deficiency. Am. J. Respir. Crit. Care Med. 2000, 162, 2069–2072. [CrossRef]
- McCarthy, C.; Dimitrov, B.D. Augmentation therapy for alpha-1 antitrypsin deficiency. COPD 2010, 7, 234–236. [CrossRef]
- Strange, C. Anti-proteases and alpha-1 antitrypsin augmentation therapy. Respir. Care 2018, 63, 690–698. [CrossRef]
- Griese, M.; Scheuch, G. Delivery of alpha-1 antitrypsin to airways. Ann. Am. Thorac. Soc. 2016, 13, S346–S351. [CrossRef]
- Korkmaz, B.; Horwitz, M.S.; Jenne, D.E.; Gauthier, F. Neutrophil elastase, proteinase 3, and cathepsin G as therapeutic targets in human diseases. Pharmacol. Rev. 2010, 62, 726–759. [CrossRef]
- Powers, J.C.; Asgian, J.L.; Ekici, O.D.; James, K.E. Irreversible inhibitors of serine, cysteine, and threonine proteases. Chem. Rev. 2002, 102, 4639–4750. [CrossRef]
- Hedstrom, L. Serine protease mechanism and specificity. Chem. Rev. 2002, 102, 4501–4524. [CrossRef]
- Bode, W.; Huber, R. Structural basis of the endoproteinase-protein inhibitor interaction. Biochim. Biophys. Acta 2000, 1477, 241–252. [CrossRef]
- Turk, B. Targeting proteases: successes, failures and future prospects. Nat. Rev. Drug Discov. 2006, 5, 785–799. [CrossRef]
- Edwards, P.D.; Bernstein, P.R. Synthetic inhibitors of elastase. Med. Res. Rev. 1994, 14, 127–194. [CrossRef]
- Edwards, P.D.; Zottola, M.A.; Davis, M.; Williams, J.; Tuthill, P.A. Peptidyl inhibitors of human neutrophil elastase. J. Med. Chem. 1995, 38, 3972–3982. [CrossRef]
- Cregge, R.J.; Durham, S.L.; Farr, R.A.; Gallion, S.L.; Hare, C.M.; Hoffman, R.V.; Janusz, M.J.; Kim, H.O.; Koehl, J.R.; Mehdi, S.; et al. Inhibition of human neutrophil elastase. 4. Design, synthesis, X-ray crystallographic analysis, and structure-activity relationships for a series of P2-modified, orally active peptidyl pentafluoroethyl ketones. J. Med. Chem. 1998, 41, 2461–2480. [CrossRef]
- Sato, F.; Inoue, Y.; Omodani, T.; Imano, K.; Okazaki, H.; Takemura, T.; Komiya, M. Design and synthesis of peptide-based carboxylic acid-containing transition-state inhibitors of human neutrophil elastase. Bioorg. Med. Chem. Lett. 2002, 12, 551–555. [CrossRef]
- Imaki, K.; Okada, T.; Nakayama, Y.; Nagao, Y.; Kobayashi, K.; Sakai, Y.; Mohri, T.; Amino, T.; Nakai, H.; Kawamura, M. Non-peptidic inhibitors of human neutrophil elastase: the design and synthesis of sulfonanilide-containing inhibitors. Bioorg. Med. Chem. 1996, 4, 2115–2134. [CrossRef]
- Ohmoto, K.; Yamamoto, T.; Okuma, M.; Horiuchi, T.; Imanishi, H.; Odagaki, Y.; Kawabata, K.; Sekioka, T.; Hirota, Y.; Matsuoka, S.; et al. Development of orally active nonpeptidic inhibitors of human neutrophil elastase. J. Med. Chem. 2001, 44, 1268–1285. [CrossRef]
- Kawabata, K.; Suzuki, M.; Sugitani, M.; Imaki, K.; Toda, M.; Miyamoto, T. ONO-5046, a novel inhibitor of human neutrophil elastase. Biochem. Biophys. Res. Commun. 1991, 177*, 814–820. [CrossRef]
- Kawabata, K.; Moore, A.R.; Willoughby, D.A. Impaired activity of protease inhibitors towards neutrophil elastase bound to human articular cartilage. Ann. Rheum. Dis. 1996, 55, 248–252. [CrossRef]
- Navia, M.A.; McKeever, B.M.; Springer, J.P.; Lin, T.Y.; Williams, H.R.; Fluder, E.M.; Dorn, C.P.; Hoogsteen, K. Structure of human neutrophil elastase in complex with a peptide chloromethyl ketone inhibitor at 1.84-A resolution. Proc. Natl. Acad. Sci. USA 1989, 86, 7–11. [CrossRef]
- Aikawa, N.; Kawasaki, Y. Clinical utility of the neutrophil elastase inhibitor sivelestat for the treatment of acute respiratory distress syndrome. Ther. Clin. Risk Manag. 2014, 10, 621–629. [CrossRef]
- Kido, T.; Muramatsu, K.; Yatera, K.; Asakawa, T.; Otsubo, H.; Kubo, T.; Fujino, Y.; Matsuda, S.; Mayumi, T.; Mukae, H. Efficacy of early sivelestat administration on acute lung injury and acute respiratory distress syndrome. Respirology 2017, 22, 708–713. [CrossRef]
- Iwata, K.; Doi, A.; Ohji, G.; Oka, H.; Oba, Y.; Takimoto, K.; Igarashi, W.; Gremillion, D.H.; Shimada, T. Effect of neutrophil elastase inhibitor (sivelestat sodium) in the treatment of acute lung injury (ALI) and acute respiratory distress syndrome (ARDS): a systematic review and meta-analysis. Intern. Med. 2010, 49, 2423–2432. [CrossRef]
- Sahebnasagh, A.; Saghafi, F.; Safdari, M.; Khataminia, M.; Sadremomtaz, A.; Talaei, Z.; Rezai Ghaleno, H.; Bagheri, M.; Habtemariam, S.; Avan, R. Neutrophil elastase inhibitor (sivelestat) may be a promising therapeutic option for management of acute lung injury/acute respiratory distress syndrome or disseminated intravascular coagulation in COVID-19. J. Clin. Pharm. Ther. 2020, 45, 1515–1519. [CrossRef]
- Ding, Q.; Wang, Y.; Yang, C.; Tuerxun, D.; Yu, X. Effect of Sivelestat in the Treatment of Acute Lung Injury and Acute Respiratory Distress Syndrome: A Systematic Review and Meta-Analysis. Intensive Care Res. 2023, 1–10. [CrossRef]
- De Clercq, E. The design of drugs for HIV and HCV. Nat. Rev. Drug Discov. 2007, 6, 1001–1018. [CrossRef]
- Lin, C.; Kwong, A.D.; Perni, R.B. Discovery and development of VX-950, a novel, covalent, and reversible inhibitor of hepatitis C virus NS3.4A serine protease. Infect. Disord. Drug Targets 2006, 6, 3–16. [CrossRef]
- Papayannopoulos, V. Neutrophil extracellular traps in immunity and disease. Nat. Rev. Immunol. 2018, 18, 134–147. [CrossRef]
- Shapiro, S.D. Proteolysis in the lung. Eur. Respir. J. Suppl. 2003, 44, 30s–32s. [CrossRef]
- Calfee, C.S.; Delucchi, K.; Parsons, P.E.; Thompson, B.T.; Ware, L.B.; Matthay, M.A.; NHLBI ARDS Network. Subphenotypes in acute respiratory distress syndrome: latent class analysis of data from two randomised controlled trials. Lancet Respir. Med. 2014, 2, 611–620. [CrossRef]
- Matthay, M.A.; Zemans, R.L.; Zimmerman, G.A.; Arabi, Y.M.; Beitler, J.R.; Mercat, A.; Herridge, M.; Randolph, A.G.; Calfee, C.S. Acute respiratory distress syndrome. Nat. Rev. Dis. Primers 2019, 5, 18. [CrossRef]
- Bos, L.D.J.; Artigas, A.; Constantin, J.M.; Hagens, L.A.; Heijnen, N.; Laffey, J.G.; Meyer, N.; Papazian, L.; Pisani, L.; Schultz, M.J.; et al. Precision medicine in acute respiratory distress syndrome: workshop report and recommendations for future research. Eur. Respir. Rev. 2021, 30, 200317. [CrossRef]
- Matera, M.G.; Rogliani, P.; Ora, J.; Calzetta, L.; Cazzola, M. Investigational elastase inhibitors for ARDS. Expert Opin. Investig. Drugs 2023, 32, 793–802. [CrossRef]
- Li, Y.; Zhao, J.; Wei, J.; Zhang, Y.; Zhang, H.; Li, Y.; Liao, T.; Hu, Y.; Yuan, B.; Zhang, X.; et al. Neutrophil elastase inhibitor (Sivelestat) in the treatment of acute respiratory distress syndrome induced by COVID-19: a multicenter retrospective cohort study. Respir. Res. 2025, 26, 28. [CrossRef]
- Tremblay, G.M.; Janelle, M.F.; Bourbonnais, Y. Anti-inflammatory activity of neutrophil elastase inhibitors. Curr. Opin. Investig. Drugs 2003, 4, 556–565.
- Stevens, T.; Ekholm, K.; Gränse, M.; Lindahl, M.; Kozma, V.; Jungar, C.; Ottosson, T.; Falk-Håkansson, H.; Churg, A.; Wright, J.L.; et al. AZD9668: pharmacological characterization of a novel oral inhibitor of neutrophil elastase. J. Pharmacol. Exp. Ther. 2011, 339, 313–320. [CrossRef]
- Gunawardena, K.A.; Gullstrand, H.; Perrett, J. Pharmacokinetics and safety of AZD9668, an oral neutrophil elastase inhibitor, in healthy volunteers and patients with COPD. Int. J. Clin. Pharmacol. Ther. 2013, 51, 288–304. [CrossRef]
- Stockley, R.; De Soyza, A.; Gunawardena, K.; Perrett, J.; Forsman-Semb, K.; Entwistle, N.; Snell, N. Phase II study of a neutrophil elastase inhibitor (AZD9668) in patients with bronchiectasis. Respir. Med. 2013, 107, 524–533. [CrossRef]
- Wells, J.M.; Titlestad, I.L.; Tanash, H.; Turner, A.M.; Chapman, K.R.; Hatipoğlu, U.Ş.; Goldklang, M.P.; D’’Armiento, J.M.; Pirozzi, C.S.; Drummond, M.B.; et al. Two randomised controlled phase 2 studies of the oral neutrophil elastase inhibitor alvelestat in alpha-1 antitrypsin deficiency. Eur. Respir. J. 2025, 66, 2501019. [CrossRef]
- von Nussbaum, F.; Li, V.M.; Allerheiligen, S.; Anlauf, S.; Bärfacker, L.; Bechem, M.; Delbeck, M.; Fitzgerald, M.F.; Gerisch, M.; Gielen-Haertwig, H.; et al. Freezing the Bioactive Conformation to Boost Potency: The Identification of BAY 85-8501, a Selective and Potent Inhibitor of Human Neutrophil Elastase for Pulmonary Diseases. ChemMedChem 2015, 10, 1163–1173. [CrossRef]
- Watz, H.; Nagelschmitz, J.; Kirsten, A.; Pedersen, F.; van der Mey, D.; Schwers, S.; Bandel, T.J.; Rabe, K.F. Safety and efficacy of the human neutrophil elastase inhibitor BAY 85-8501 for the treatment of non-cystic fibrosis bronchiectasis: A randomized controlled trial. Pulm. Pharmacol. Ther. 2019, 56, 86–93. [CrossRef]
- Kuraki, T.; Ishibashi, M.; Takayama, M.; Shiraishi, M.; Yoshida, M. A novel oral neutrophil elastase inhibitor (ONO-6818) inhibits human neutrophil elastase-induced emphysema in rats. Am. J. Respir. Crit. Care Med. 2002, 166, 496–500. [CrossRef]
- Yoshimura, Y.; Hiramatsu, Y.; Sato, Y.; Homma, S.; Enomoto, Y.; Jikuya, T.; Sakakibara, Y. ONO-6818, a novel, potent neutrophil elastase inhibitor, reduces inflammatory mediators during simulated extracorporeal circulation. Ann. Thorac. Surg. 2003, 76, 1234–1239. [CrossRef]
- Ohmoto, K.; Okuma, M.; Yamamoto, T.; Kijima, H.; Sekioka, T.; Kitagawa, K.; Yamamoto, S.; Tanaka, K.; Kawabata, K.; Sakata, A.; et al. Design and synthesis of new orally active inhibitors of human neutrophil elastase. Bioorg. Med. Chem. 2001, 9, 1307–1323. [CrossRef]
- Stockley, R.A. Neutrophils and the pathogenesis of COPD. Chest 2002, 121, 151S–155S. [CrossRef]
- Attucci, S.; Gauthier, A.; Korkmaz, B.; Delépine, P.; Martino, M.F.; Saudubray, F.; Diot, P.; Gauthier, F. EPI-hNE4, a proteolysis-resistant inhibitor of human neutrophil elastase and potential anti-inflammatory drug for treating cystic fibrosis. J. Pharmacol. Exp. Ther. 2006, 318, 803–809. [CrossRef]
- Mac Sweeney, R.; McAuley, D.F. Do Nonventilatory Strategies for Acute Lung Injury and ARDS Work? In Evidence-Based Practice of Critical Care; Saunders: Philadelphia, PA, USA, 2010; pp. 73–81. [CrossRef]
- Barnes, P.J. Oxidative stress-based therapeutics in COPD. Redox Biol. 2020, 33, 101544. [CrossRef]
- Kang, H.; Seo, J.; Yang, E.J.; Choi, I.H. Silver Nanoparticles Induce Neutrophil Extracellular Traps Via Activation of PAD and Neutrophil Elastase. Biomolecules 2021, 11, 317. [CrossRef]
- Kim, J.; Sahay, G. Nanomedicine hitchhikes on neutrophils to the inflamed lung. Nat. Nanotechnol. 2022, 17, 1–2. [CrossRef]
- Rezaei, N.; Zadory, M.; Babity, S.; Marleau, S.; Brambilla, D. Therapeutic applications of nanoparticles targeting neutrophil and extracellular traps. J. Control. Release 2023, 358, 636–653. [CrossRef]
- Li, H.; Li, C.; Fu, C.; Wang, Y.; Liang, T.; Wu, H.; Wu, C.; Wang, C.; Sun, T.; Liu, S. Innovative nanoparticle-based approaches for modulating neutrophil extracellular traps in diseases: from mechanisms to therapeutics. J. Nanobiotechnology 2025, 23, 88. [CrossRef]
- Lee, H.J.; Lee, N.K.; Kim, J.; Kim, J.; Seo, D.; Shin, H.E.; Kim, J.; Ahn, J.H.; Kim, S.N.; Kim, H.S.; et al. Sequential nanoparticle therapy targeting neutrophil hyperactivation to prevent neutrophil-induced pulmonary fibrosis. J. Nanobiotechnology 2025, 23, 381. [CrossRef]
| Property | Endogenous Inhibitors (AAT, SLPI, Elafin) | Natural Small-Molecule Inhibitors | Synthetic Small-Molecule Inhibitors |
Peptide or Biologic Inhibitors |
| Molecular size | Large proteins | Small–medium | Small | Medium-sized |
| Selectivity for HNE | High | Variable | Tunable, often high | Very high |
| Oxidative stability | Poor (susceptible to ROS) | Variable | Optimizable | Moderate |
| Tissue penetration | Limited | Often limited | Generally good tissue penetration | Limited |
| Pharmacokinetics | Short half-life | Rapid metabolism | Optimizable | Limited stability |
| Immunogenicity risk | Low–moderate | Low | Low | Potential risk |
| Manufacturing complexity | High | Moderate | Moderate | High |
| Clinical success | Partial (AAT therapy) | Minimal | Moderate (regional approvals, clinical trials) | Limited |
| Main limitation | Oxidative inactivation and limited compartment access | Low potency and metabolic instability | Context-dependent efficacy and translational challenges | Delivery constraints and metabolic instability |
| Compound | Type | Clinical Development/ Indication | Key Advantages | Main Limitations | Reference |
| Early peptidyl ketones/aldehydes | Peptide-derived inhibitors | Preclinical | High potency; early mechanistic validation | Poor pharmacokinetics; toxicity | [49,50,51] |
| α-ketoheterocycles/oxadiazoles | Synthetic small molecules | Preclinical | Improved selectivity compared with early inhibitors | Limited clinical translation | [52,53] |
| Sivelestat (ONO-5046) | Small-molecule inhibitor | Approved in Japan/Korea for ARDS; trials elsewhere | First clinically approved HNE inhibitor; proof of target validity | Short half-life; continuous infusion; limited global efficacy | [54,57,58,59,60,61,70] |
| Alvelestat (AZD9668) | Oral small-molecule inhibitor | Phase II trials in COPD, bronchiectasis, AAT deficiency | Oral bioavailability; high selectivity; suitable for chronic therapy | Limited improvement in lung function endpoints | [72,73,74,75] |
| BAY 85-8501 | Potent small-molecule inhibitor | Phase IIa bronchiectasis | Very high potency and selectivity; good tolerability | Limited clinical efficacy demonstrated | [76,77] |
| Freselestat (ONO-6818) | Small-molecule inhibitor (sivelestat lineage) | Phase II (discontinued) in COPD, AAT deficiency | Oral activity; improved scaffold stability | Liver toxicity signals; program discontinued | [78,79] |
| Depelestat (DX-890 / EPI-hNE4) | Recombinant peptide biologic | Phase I–II ARDS, cystic fibrosis | High specificity; mimics endogenous inhibition | Poor tissue penetration; proteolytic instability | [82,83] |
| Limiting Factor | Mechanism | Consequence | Clinical Implication |
| Biological redundancy | Compensatory activity of other proteases | Partial pathway suppression | Limited efficacy in complex syndromes (e.g., ARDS) |
| Enzyme compartmentalization | Membrane-bound, NET-associated, or matrix-bound HNE becomes inaccessible | Incomplete target engagement | Persistent elastase activity despite therapy |
| Oxidative microenvironment | ROS modify inhibitors and endogenous antiproteases | Reduced inhibitory capacity | Reduced effectiveness in inflamed tissues |
| Pharmacokinetic–pharmacodynamic mismatch | Insufficient drug concentration at disease site | Suboptimal inhibition | Failure despite potent in vitro activity |
| Timing of intervention | Intervention after irreversible tissue damage | Limited reversibility | Poor outcomes in acute disease |
| Patient heterogeneity | Variable neutrophil burden and disease endotypes | Diluted treatment effect | Negative or inconclusive trials |
| Biomarker limitations | Lack of validated biomarkers of elastase activity | Poor patient selection | Inefficient clinical trial design |
| Delivery constraints | Poor penetration into mucus or inflamed tissue | Reduced local exposure | Particularly relevant in lung diseases |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




