Submitted:
02 April 2026
Posted:
03 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
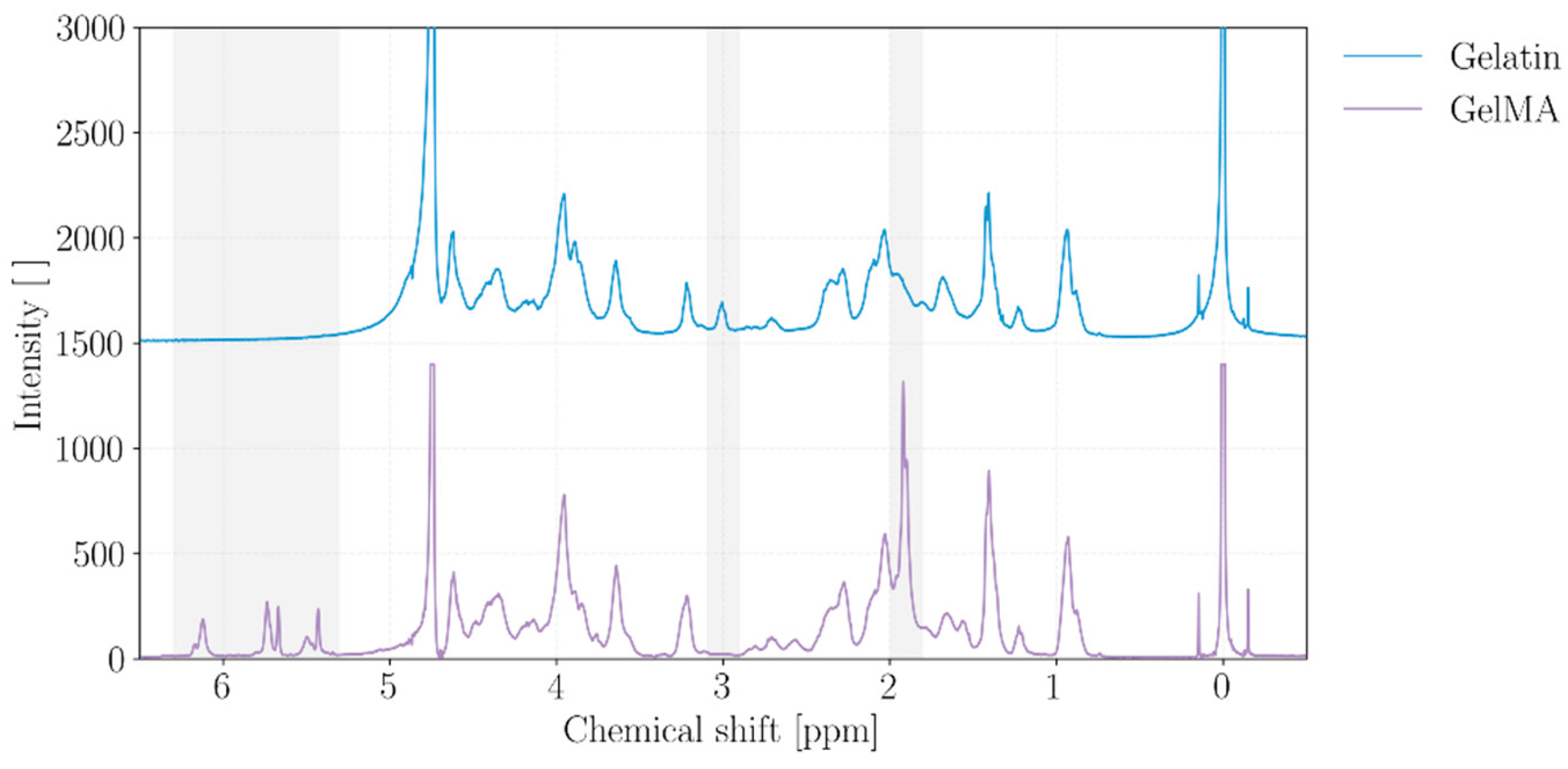
2.1. GelMA Characterizazion
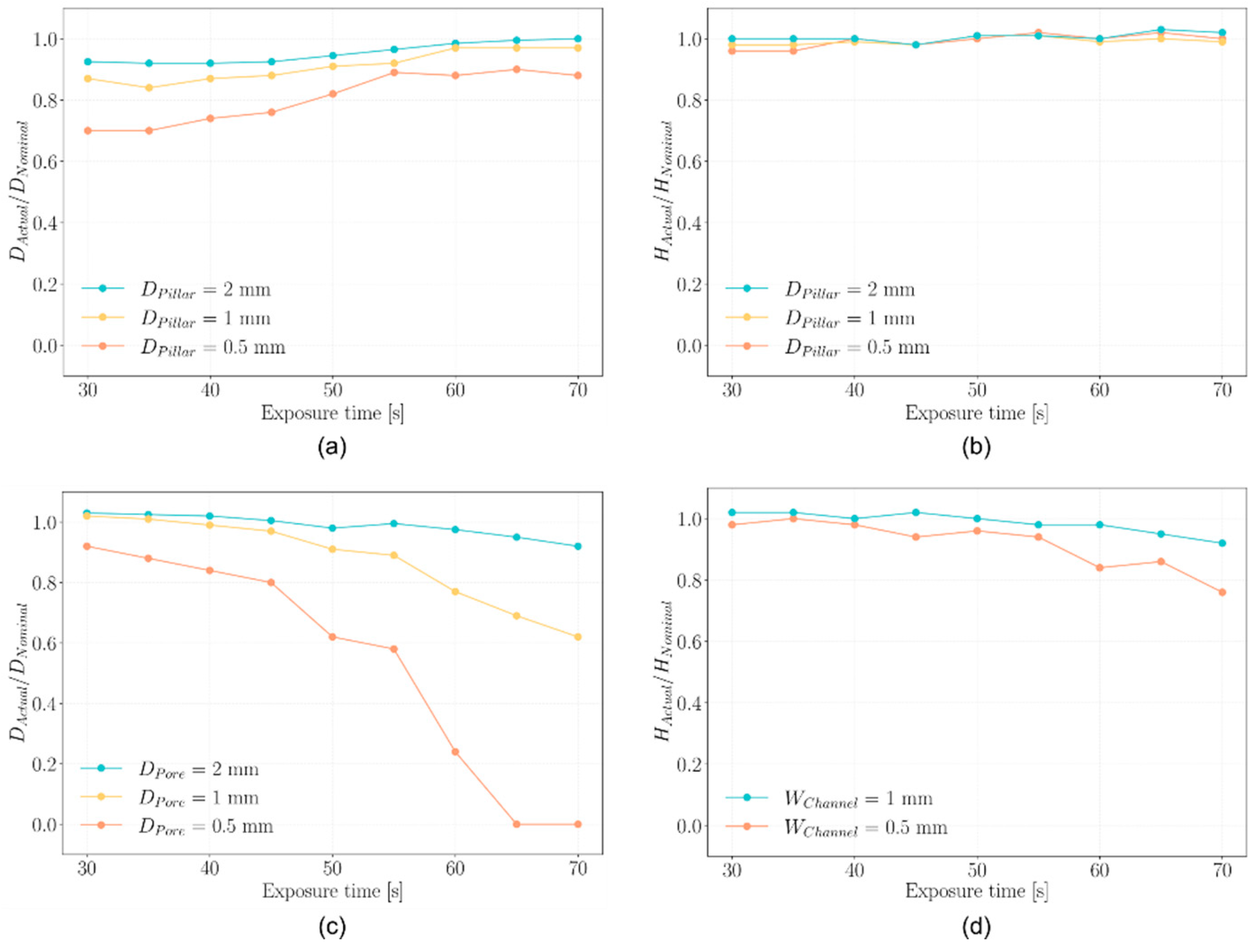
2.2. Printability Assessment
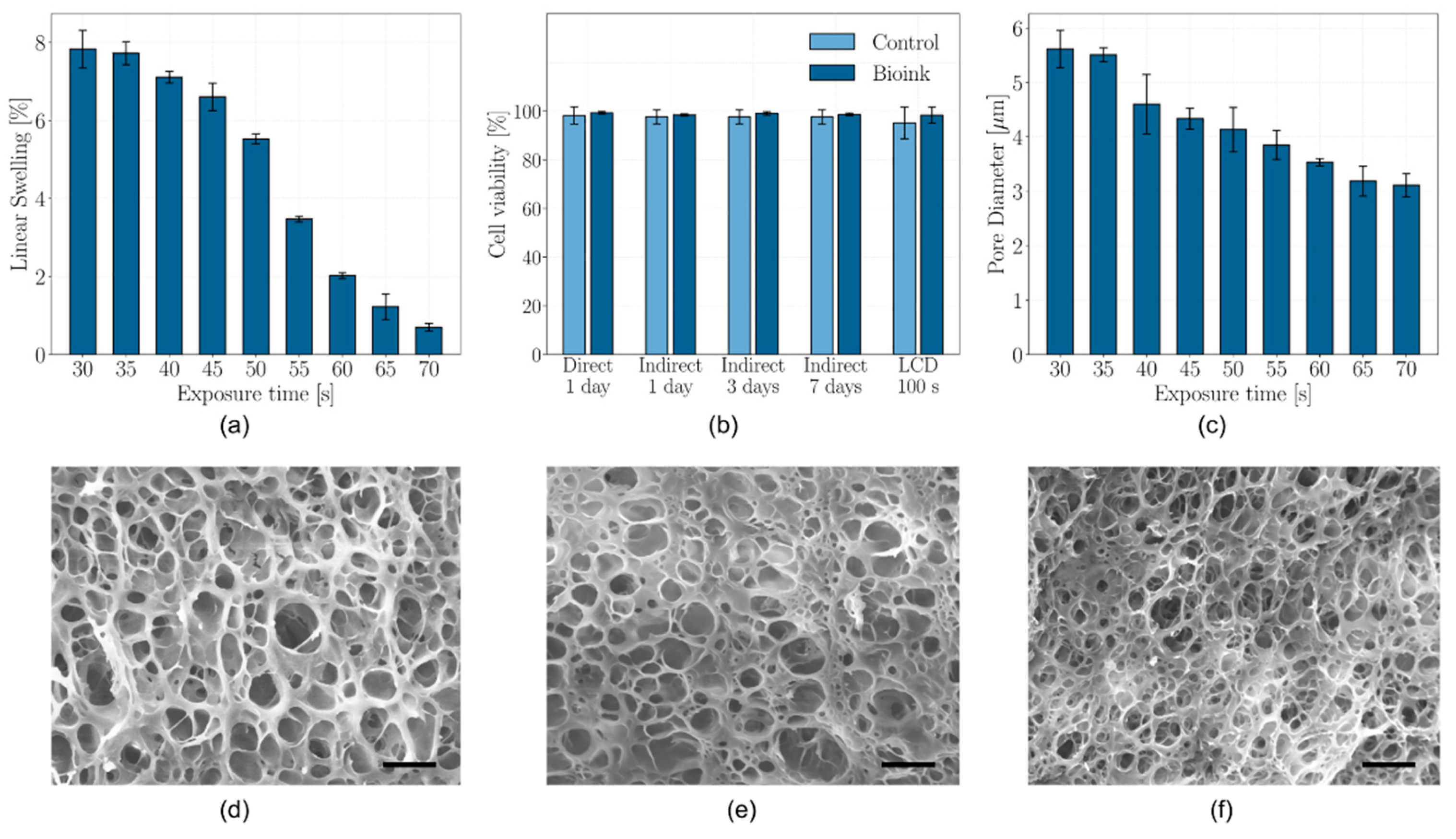
2.3. Swelling Characterization
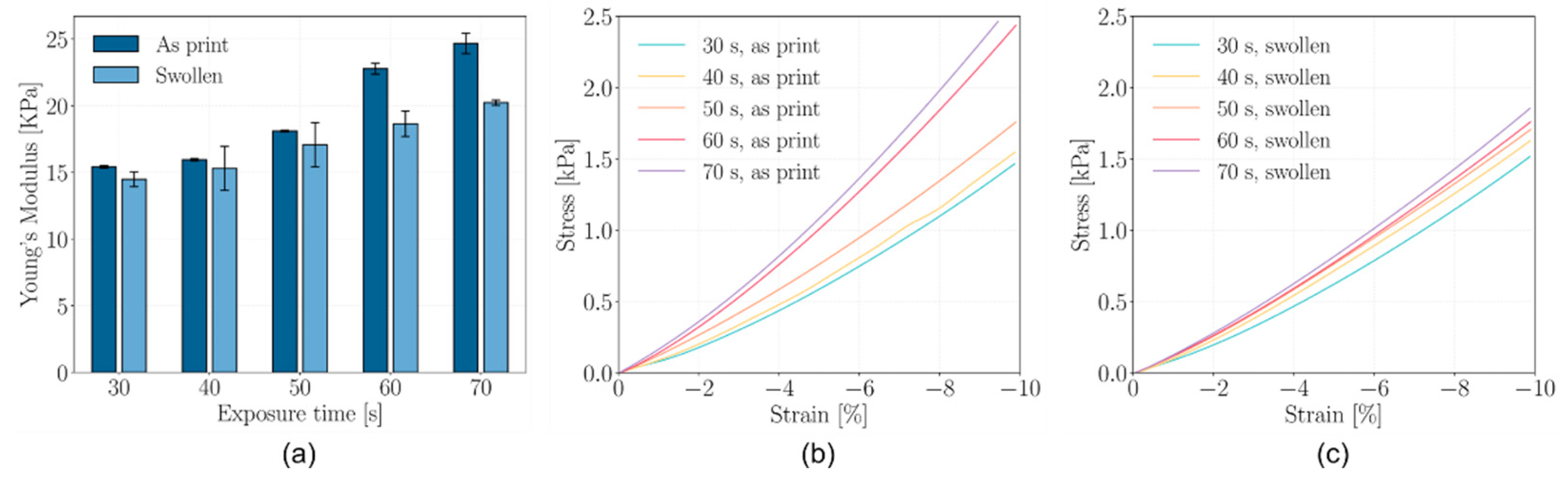
2.4. Mechanical Properties
2.5. Microstructure Characterization
2.6. Cell Viability
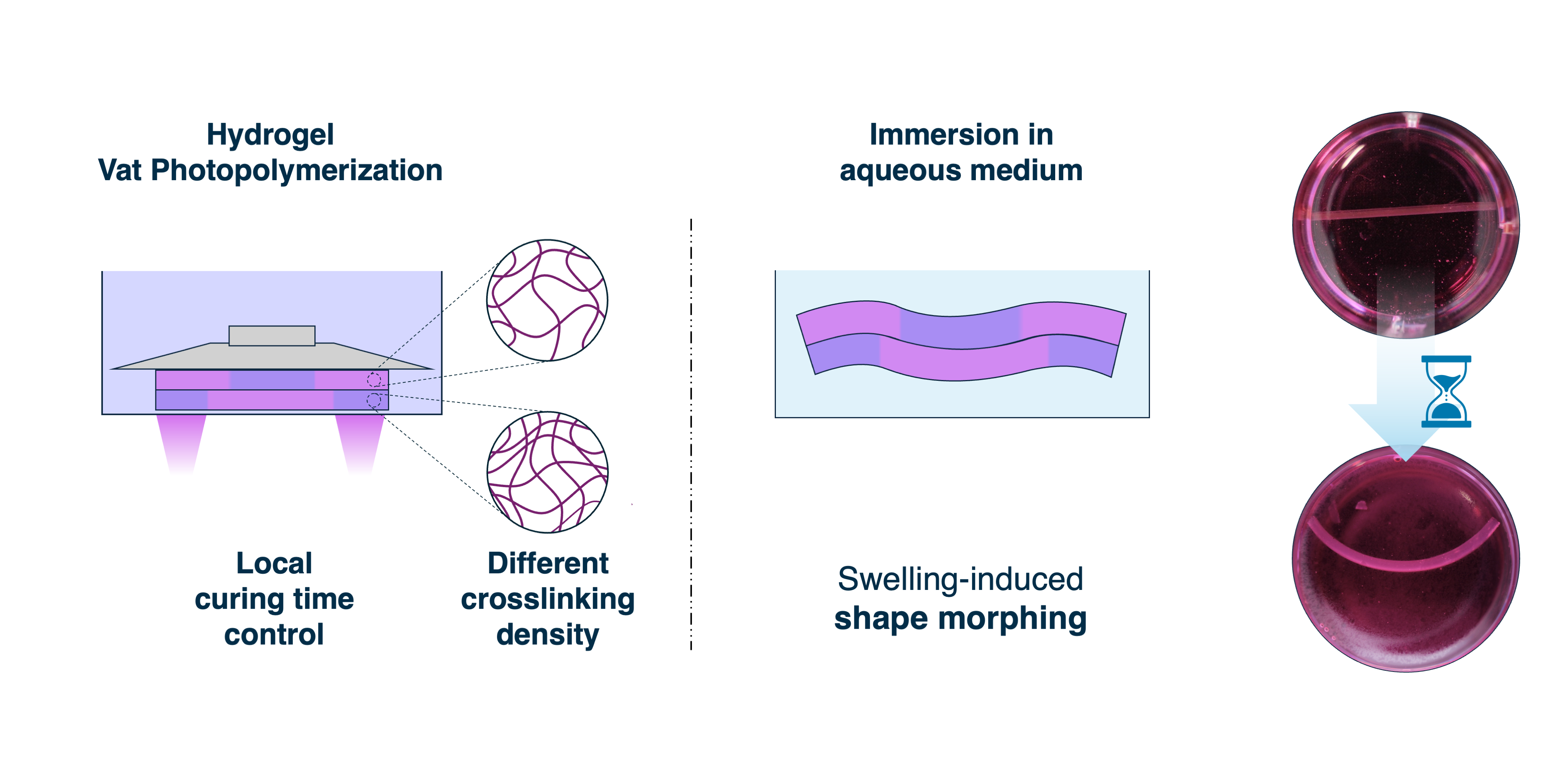
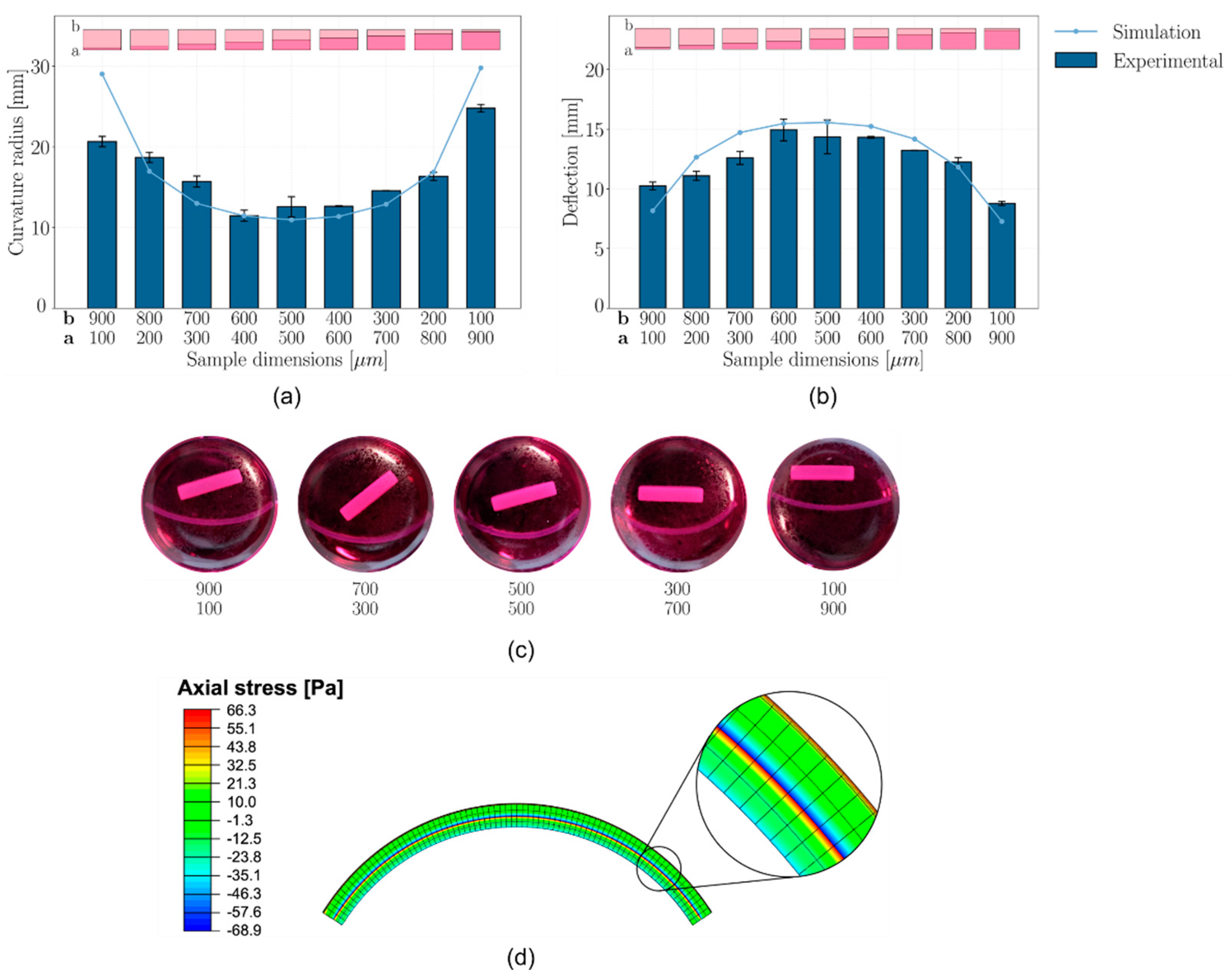
2.7. Shape Morphing
2.8. Shape Morphing Simulation
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. GelMA Synthesis and Characterization
4.3. Hydrogel Formulation
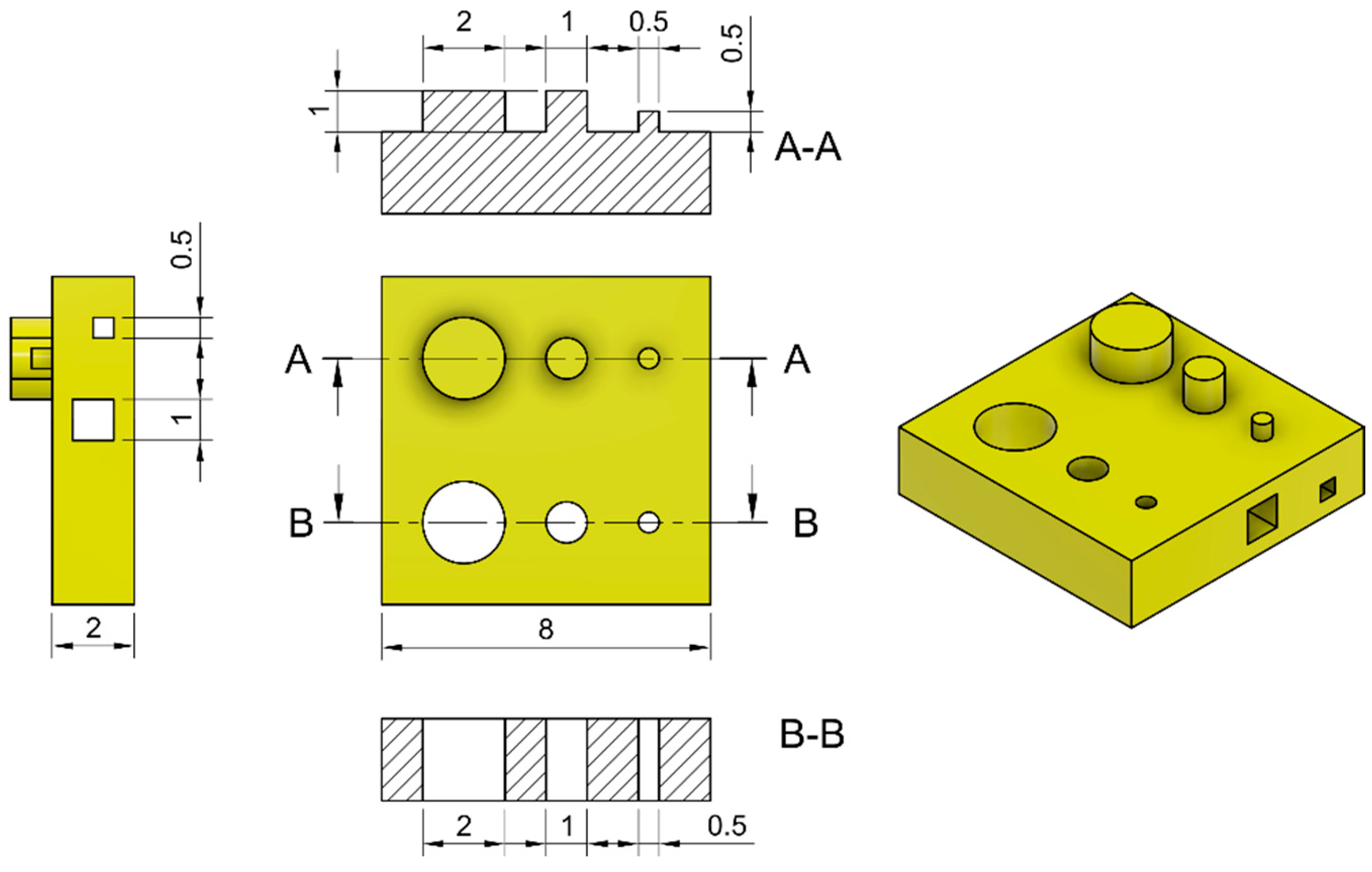
4.4. Printability Assessment
4.5. Swelling Characterization
4.6. Mechanical Properties Characterization
4.7. Microstructure Characterization
4.8. Cell viability Assessment
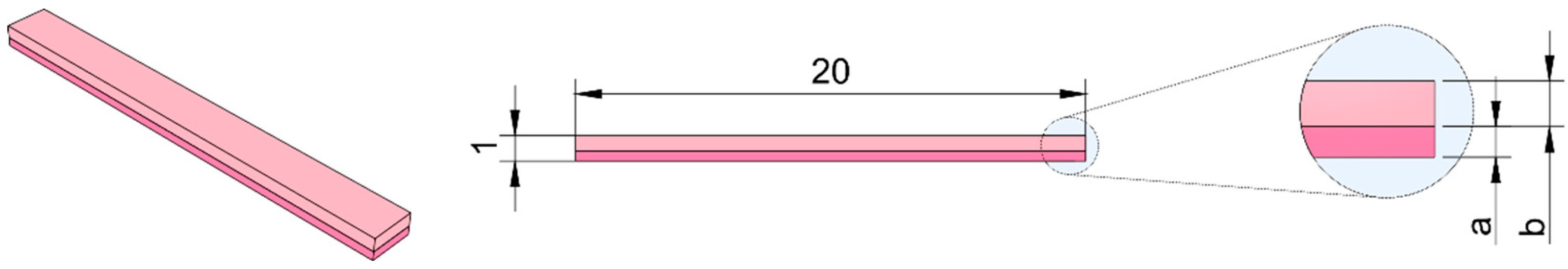
4.9. Shape Morphing
4.10. Shape Morphing Simulation
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Murphy, S. V; Atala, A. 3D Bioprinting of Tissues and Organs. Nat. Biotechnol. 2014, 32, 773–785. [CrossRef]
- Lee, W.; Hong, Y.; Dai, G. 3D Bioprinting of Vascular Conduits for Pediatric Congenital Heart Repairs. Translational Research 2019, 211, 35–45. [CrossRef]
- Hölzl, K.; Lin, S.; Tytgat, L.; Van Vlierberghe, S.; Gu, L.; Ovsianikov, A. Bioink Properties before, during and after 3D Bioprinting. Biofabrication 2016, 8, 032002. [CrossRef]
- Mania, S.; Banach-Kopeć, A.; Maciejewska, N.; Czerwiec, K.; Słonimska, P.; Deptuła, M.; Baczyński-Keller, J.; Pikuła, M.; Sachadyn, P.; Tylingo, R. From Bioink to Tissue: Exploring Chitosan-Agarose Composite in the Context of Printability and Cellular Behaviour. Molecules 2024, 29, 4648. [CrossRef]
- Kirillova, A.; Maxson, R.; Stoychev, G.; Gomillion, C.T.; Ionov, L. 4D Biofabrication Using Shape-Morphing Hydrogels. Advanced Materials 2017, 29. [CrossRef]
- Yang, G.H.; Yeo, M.; Koo, Y.W.; Kim, G.H. 4D Bioprinting: Technological Advances in Biofabrication. Macromol. Biosci. 2019, 19. [CrossRef]
- Hospodiuk, M.; Dey, M.; Sosnoski, D.; Ozbolat, I.T. The Bioink: A Comprehensive Review on Bioprintable Materials. Biotechnol. Adv. 2017, 35, 217–239. [CrossRef]
- Cui, X.; Li, J.; Hartanto, Y.; Durham, M.; Tang, J.; Zhang, H.; Hooper, G.; Lim, K.; Woodfield, T. Advances in Extrusion 3D Bioprinting: A Focus on Multicomponent Hydrogel-Based Bioinks. Adv. Healthc. Mater. 2020, 9. [CrossRef]
- Lee, M.; Rizzo, R.; Surman, F.; Zenobi-Wong, M. Guiding Lights: Tissue Bioprinting Using Photoactivated Materials. Chem. Rev. 2020, 120, 10950–11027. [CrossRef]
- Guida, L.; Cavallaro, M.; Levi, M. Advancements in High-Resolution 3D Bioprinting: Exploring Technological Trends, Bioinks and Achieved Resolutions. Bioprinting 2024, 44, e00376. [CrossRef]
- Gillispie, G.; Prim, P.; Copus, J.; Fisher, J.; Mikos, A.G.; Yoo, J.J.; Atala, A.; Lee, S.J. Assessment Methodologies for Extrusion-Based Bioink Printability. Biofabrication 2020, 12, 022003. [CrossRef]
- Ribezzi, D.; Zegwaart, J.; Van Gansbeke, T.; Tejo-Otero, A.; Florczak, S.; Aerts, J.; Delrot, P.; Hierholzer, A.; Fussenegger, M.; Malda, J.; et al. Multi-material Volumetric Bioprinting and Plug-and-play Suspension Bath Biofabrication via Bioresin Molecular Weight Tuning and via Multiwavelength Alignment Optics. Advanced Materials 2025, 37. [CrossRef]
- Li, Y.-C.; Zhang, Y.S.; Akpek, A.; Shin, S.R.; Khademhosseini, A. 4D Bioprinting: The next-Generation Technology for Biofabrication Enabled by Stimuli-Responsive Materials. Biofabrication 2016, 9, 012001. [CrossRef]
- Shen, J.; Du, S.; Xu, Z.; Gan, T.; Handschuh-Wang, S.; Zhang, X. Anti-Freezing, Non-Drying, Localized Stiffening, and Shape-Morphing Organohydrogels. Gels 2022, 8, 331. [CrossRef]
- Shiblee, M.N.I.; Ahmed, K.; Kawakami, M.; Furukawa, H. 4D Printing of Shape-Memory Hydrogels for Soft-Robotic Functions. Adv. Mater. Technol. 2019, 4. [CrossRef]
- Rovelli, R.; Cecchini, B.; Zavagna, L.; Azimi, B.; Ricci, C.; Esin, S.; Milazzo, M.; Batoni, G.; Danti, S. Emerging Multiscale Biofabrication Approaches for Bacteriotherapy. Molecules 2024, 29, 533. [CrossRef]
- Lanzalaco, S.; Armelin, E. Poly(N-Isopropylacrylamide) and Copolymers: A Review on Recent Progresses in Biomedical Applications. Gels 2017, 3, 36. [CrossRef]
- Morouço, P.; Azimi, B.; Milazzo, M.; Mokhtari, F.; Fernandes, C.; Reis, D.; Danti, S. Four-Dimensional (Bio-)Printing: A Review on Stimuli-Responsive Mechanisms and Their Biomedical Suitability. Applied Sciences 2020, 10, 9143. [CrossRef]
- Loukelis, K.; Helal, Z.A.; Mikos, A.G.; Chatzinikolaidou, M. Nanocomposite Bioprinting for Tissue Engineering Applications. Gels 2023, 9, 103. [CrossRef]
- Farrukh, A.; Nayab, S. Shape Memory Hydrogels for Biomedical Applications. Gels 2024, 10, 270. [CrossRef]
- Siddique, M.F.; Omar, F.K.; Al-Marzouqi, A.H. Design and Application of Stimuli-Responsive Hydrogels for 4D Printing: A Review of Adaptive Materials in Engineering. Gels 2026, 12, 138. [CrossRef]
- Singh, J.; Nayak, P. PH-responsive Polymers for Drug Delivery: Trends and Opportunities. Journal of Polymer Science 2023, 61, 2828–2850. [CrossRef]
- Londoño-Berrío, M.; Pérez-Buitrago, S.; Ortiz-Trujillo, I.C.; Hoyos-Palacio, L.M.; Orozco, L.Y.; López, L.; Zárate-Triviño, D.G.; Capobianco, J.A.; Mena-Giraldo, P. Cytotoxicity and Genotoxicity of Azobenzene-Based Polymeric Nanocarriers for Phototriggered Drug Release and Biomedical Applications. Polymers (Basel). 2022, 14, 3119. [CrossRef]
- Amirthalingam, S.; Rajendran, A.K.; Moon, Y.G.; Hwang, N.S. Stimuli-Responsive Dynamic Hydrogels: Design, Properties and Tissue Engineering Applications. Mater. Horiz. 2023, 10, 3325–3350. [CrossRef]
- Gugulothu, S.B.; Chatterjee, K. Visible Light-Based 4D-Bioprinted Tissue Scaffold. ACS Macro Lett. 2023, 12, 494–502. [CrossRef]
- Takashima, Y.; Hatanaka, S.; Otsubo, M.; Nakahata, M.; Kakuta, T.; Hashidzume, A.; Yamaguchi, H.; Harada, A. Expansion–Contraction of Photoresponsive Artificial Muscle Regulated by Host–Guest Interactions. Nat. Commun. 2012, 3, 1270. [CrossRef]
- Municoy, S.; Álvarez Echazú, M.I.; Antezana, P.E.; Galdopórpora, J.M.; Olivetti, C.; Mebert, A.M.; Foglia, M.L.; Tuttolomondo, M. V.; Alvarez, G.S.; Hardy, J.G.; et al. Stimuli-Responsive Materials for Tissue Engineering and Drug Delivery. Int. J. Mol. Sci. 2020, 21, 4724. [CrossRef]
- Stoychev, G.; Puretskiy, N.; Ionov, L. Self-Folding All-Polymer Thermoresponsive Microcapsules. Soft Matter 2011, 7, 3277. [CrossRef]
- Lv, C.; Xia, H.; Shi, Q.; Wang, G.; Wang, Y.; Chen, Q.; Zhang, Y.; Liu, L.; Sun, H. Sensitively Humidity-Driven Actuator Based on Photopolymerizable PEG-DA Films. Adv. Mater. Interfaces 2017, 4. [CrossRef]
- Zhang, L.; Liang, H.; Jacob, J.; Naumov, P. Photogated Humidity-Driven Motility. Nat. Commun. 2015, 6, 7429. [CrossRef]
- Mandal, A.; Chatterjee, K. Emerging Trends in Humidity-Responsive 4D Bioprinting. Chemical Engineering Journal 2023, 455, 140550. [CrossRef]
- Cui, C.; Kim, D.-O.; Pack, M.Y.; Han, B.; Han, L.; Sun, Y.; Han, L.-H. 4D Printing of Self-Folding and Cell-Encapsulating 3D Microstructures as Scaffolds for Tissue-Engineering Applications. Biofabrication 2020, 12, 045018. [CrossRef]
- Díaz-Payno, P.J.; Kalogeropoulou, M.; Muntz, I.; Kingma, E.; Kops, N.; D’Este, M.; Koenderink, G.H.; Fratila-Apachitei, L.E.; van Osch, G.J.V.M.; Zadpoor, A.A. Swelling-Dependent Shape-Based Transformation of a Human Mesenchymal Stromal Cells-Laden 4D Bioprinted Construct for Cartilage Tissue Engineering. Adv. Healthc. Mater. 2023, 12. [CrossRef]
- Jamal, M.; Kadam, S.S.; Xiao, R.; Jivan, F.; Onn, T.; Fernandes, R.; Nguyen, T.D.; Gracias, D.H. Bio-Origami Hydrogel Scaffolds Composed of Photocrosslinked PEG Bilayers. Adv. Healthc. Mater. 2013, 2, 1142–1150. [CrossRef]
- Kwag, H.R.; Serbo, J. V.; Korangath, P.; Sukumar, S.; Romer, L.H.; Gracias, D.H. A Self-Folding Hydrogel In Vitro Model for Ductal Carcinoma. Tissue Eng. Part C Methods 2016, 22, 398–407. [CrossRef]
- Lee, Y. Bin; Jeon, O.; Lee, S.J.; Ding, A.; Wells, D.; Alsberg, E. Induction of Four-Dimensional Spatiotemporal Geometric Transformations in High Cell Density Tissues via Shape-Changing Hydrogels. Adv. Funct. Mater. 2021, 31. [CrossRef]
- Ding, A.; Jeon, O.; Cleveland, D.; Gasvoda, K.L.; Wells, D.; Lee, S.J.; Alsberg, E. Jammed Micro-Flake Hydrogel for Four-Dimensional Living Cell Bioprinting. Advanced Materials 2022, 34. [CrossRef]
- Ding, A.; Lee, S.J.; Ayyagari, S.; Tang, R.; Huynh, C.T.; Alsberg, E. 4D Biofabrication via Instantly Generated Graded Hydrogel Scaffolds. Bioact. Mater. 2022, 7, 324–332. [CrossRef]
- Shirahama, H.; Lee, B.H.; Tan, L.P.; Cho, N.-J. Precise Tuning of Facile One-Pot Gelatin Methacryloyl (GelMA) Synthesis. Sci. Rep. 2016, 6, 31036. [CrossRef]
- Zhu, M.; Wang, Y.; Ferracci, G.; Zheng, J.; Cho, N.-J.; Lee, B.H. Gelatin Methacryloyl and Its Hydrogels with an Exceptional Degree of Controllability and Batch-to-Batch Consistency. Sci. Rep. 2019, 9, 6863. [CrossRef]
- Yu, C.; Schimelman, J.; Wang, P.; Miller, K.L.; Ma, X.; You, S.; Guan, J.; Sun, B.; Zhu, W.; Chen, S. Photopolymerizable Biomaterials and Light-Based 3D Printing Strategies for Biomedical Applications. Chem. Rev. 2020, 120, 10695–10743. [CrossRef]
- Bencherif, S.A.; Srinivasan, A.; Horkay, F.; Hollinger, J.O.; Matyjaszewski, K.; Washburn, N.R. Influence of the Degree of Methacrylation on Hyaluronic Acid Hydrogels Properties. Biomaterials 2008, 29, 1739–1749. [CrossRef]
- Lin, J.-T.; Liu, H.-W.; Chen, K.-T.; Cheng, D.-C. Modeling the Optimal Conditions for Improved Efficacy and Crosslink Depth of Photo-Initiated Polymerization. Polymers (Basel). 2019, 11, 217. [CrossRef]
- Pariskar, A.; Sharma, P.K.; Murty, U.S.; Banerjee, S. Effect of Tartrazine as Photoabsorber for Improved Printing Resolution of 3D Printed “Ghost Tablets”: Non-Erodible Inert Matrices. J. Pharm. Sci. 2023, 112, 1020–1031. [CrossRef]
- He, N.; Wang, X.; Shi, L.; Li, J.; Mo, L.; Chen, F.; Huang, Y.; Liu, H.; Zhu, X.; Zhu, W.; et al. Photoinhibiting via Simultaneous Photoabsorption and Free-Radical Reaction for High-Fidelity Light-Based Bioprinting. Nat. Commun. 2023, 14, 3063. [CrossRef]
- Cavallo, A.; Radaelli, G.; Al Kayal, T.; Mero, A.; Mezzetta, A.; Guazzelli, L.; Soldani, G.; Losi, P. Optimization of Gelatin and Crosslinker Concentrations in a Gelatin/Alginate-Based Bioink with Potential Applications in a Simplified Skin Model. Molecules 2025, 30, 649. [CrossRef]
- Nichol, J.W.; Koshy, S.T.; Bae, H.; Hwang, C.M.; Yamanlar, S.; Khademhosseini, A. Cell-Laden Microengineered Gelatin Methacrylate Hydrogels. Biomaterials 2010, 31, 5536–5544. [CrossRef]
- Schuurman, W.; Levett, P.A.; Pot, M.W.; van Weeren, P.R.; Dhert, W.J.A.; Hutmacher, D.W.; Melchels, F.P.W.; Klein, T.J.; Malda, J. Gelatin-Methacrylamide Hydrogels as Potential Biomaterials for Fabrication of Tissue-Engineered Cartilage Constructs. Macromol. Biosci. 2013, 13, 551–561. [CrossRef]
- Li, Z.; Liu, Z.; Ng, T.Y.; Sharma, P. The Effect of Water Content on the Elastic Modulus and Fracture Energy of Hydrogel. Extreme Mech. Lett. 2020, 35, 100617. [CrossRef]
- Ghosh, R.N.; Thomas, J.; B. R., V.; N. G., D.; Janardanan, A.; Namboothiri, P.K.; Peter, M. An Insight into Synthesis, Properties and Applications of Gelatin Methacryloyl Hydrogel for 3D Bioprinting. Mater. Adv. 2023, 4, 5496–5529. [CrossRef]
- Schiavi, A.; Cuccaro, R.; Troia, A. Functional Mechanical Attributes of Natural and Synthetic Gel-Based Scaffolds in Tissue Engineering: Strain-Stiffening Effects on Apparent Elastic Modulus and Compressive Toughness. J. Mech. Behav. Biomed. Mater. 2022, 126, 105066. [CrossRef]
- Walejewska, E.; Melchels, F.P.W.; Paradiso, A.; McCormack, A.; Szlazak, K.; Olszewska, A.; Srebrzynski, M.; Swieszkowski, W. Tuning Physical Properties of GelMA Hydrogels through Microarchitecture for Engineering Osteoid Tissue. Biomacromolecules 2024, 25, 188–199. [CrossRef]
- Chen, Y.; Lin, R.; Qi, H.; Yang, Y.; Bae, H.; Melero-Martin, J.M.; Khademhosseini, A. Functional Human Vascular Network Generated in Photocrosslinkable Gelatin Methacrylate Hydrogels. Adv. Funct. Mater. 2012, 22, 2027–2039. [CrossRef]
- Bova, L.; Maggiotto, F.; Micheli, S.; Giomo, M.; Sgarbossa, P.; Gagliano, O.; Falcone, D.; Cimetta, E. A Porous Gelatin Methacrylate-Based Material for 3D Cell-Laden Constructs. Macromol. Biosci. 2023, 23. [CrossRef]
- He, J.; Sun, Y.; Gao, Q.; He, C.; Yao, K.; Wang, T.; Xie, M.; Yu, K.; Nie, J.; Chen, Y.; et al. Gelatin Methacryloyl Hydrogel, from Standardization, Performance, to Biomedical Application. Adv. Healthc. Mater. 2023, 12. [CrossRef]
- Ghosh, R.N.; Thomas, J.; B. R., V.; N. G., D.; Janardanan, A.; Namboothiri, P.K.; Peter, M. An Insight into Synthesis, Properties and Applications of Gelatin Methacryloyl Hydrogel for 3D Bioprinting. Mater. Adv. 2023, 4, 5496–5529. [CrossRef]
- Loh, Q.L.; Choong, C. Three-Dimensional Scaffolds for Tissue Engineering Applications: Role of Porosity and Pore Size. Tissue Eng. Part B Rev. 2013, 19, 485–502. [CrossRef]
- Annabi, N.; Nichol, J.W.; Zhong, X.; Ji, C.; Koshy, S.; Khademhosseini, A.; Dehghani, F. Controlling the Porosity and Microarchitecture of Hydrogels for Tissue Engineering. Tissue Eng. Part B Rev. 2010, 16, 371–383. [CrossRef]
- Lee, B.H.; Shirahama, H.; Cho, N.-J.; Tan, L.P. Efficient and Controllable Synthesis of Highly Substituted Gelatin Methacrylamide for Mechanically Stiff Hydrogels. RSC Adv. 2015, 5, 106094–106097. [CrossRef]
- Claaßen, C.; Claaßen, M.H.; Truffault, V.; Sewald, L.; Tovar, G.E.M.; Borchers, K.; Southan, A. Quantification of Substitution of Gelatin Methacryloyl: Best Practice and Current Pitfalls. Biomacromolecules 2018, 19, 42–52. [CrossRef]
- Van Den Bulcke, A.I.; Bogdanov, B.; De Rooze, N.; Schacht, E.H.; Cornelissen, M.; Berghmans, H. Structural and Rheological Properties of Methacrylamide Modified Gelatin Hydrogels. Biomacromolecules 2000, 1, 31–38. [CrossRef]
- Wu, S.-D.; Hsu, S. 4D Bioprintable Self-Healing Hydrogel with Shape Memory and Cryopreserving Properties. Biofabrication 2021, 13, 045029. [CrossRef]
- Meschini, S.; Pellegrini, E.; Maestri, C.A.; Condello, M.; Bettotti, P.; Condello, G.; Scarpa, M. In Vitro Toxicity Assessment of Hydrogel Patches Obtained by Cation-induced Cross-linking of Rod-like Cellulose Nanocrystals. J. Biomed. Mater. Res. B Appl. Biomater. 2020, 108, 687–697. [CrossRef]
- Iervolino, F.; Belgio, B.; Bonessa, A.; Potere, F.; Suriano, R.; Boschetti, F.; Mantero, S.; Levi, M. Versatile and Non-Cytotoxic GelMA-Xanthan Gum Biomaterial Ink for Extrusion-Based 3D Bioprinting. Bioprinting 2023, 31, e00269. [CrossRef]
| Parameter | Value |
| Layer height | 50 μm |
| Bottom layers | 0 |
| Exposure time | 20 to 70 s |
| Resting time after retraction | 0.8 s |
| Lifting distance | 3.0 + 1.4 mm |
| Lifting speed | 15 + 55 mm/min |
| Retract distance | 2.3 + 2.1 mm |
| Retract speed | 145 + 45 mm/min |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




