Submitted:
01 April 2026
Posted:
02 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Literature Search Strategy and Scope
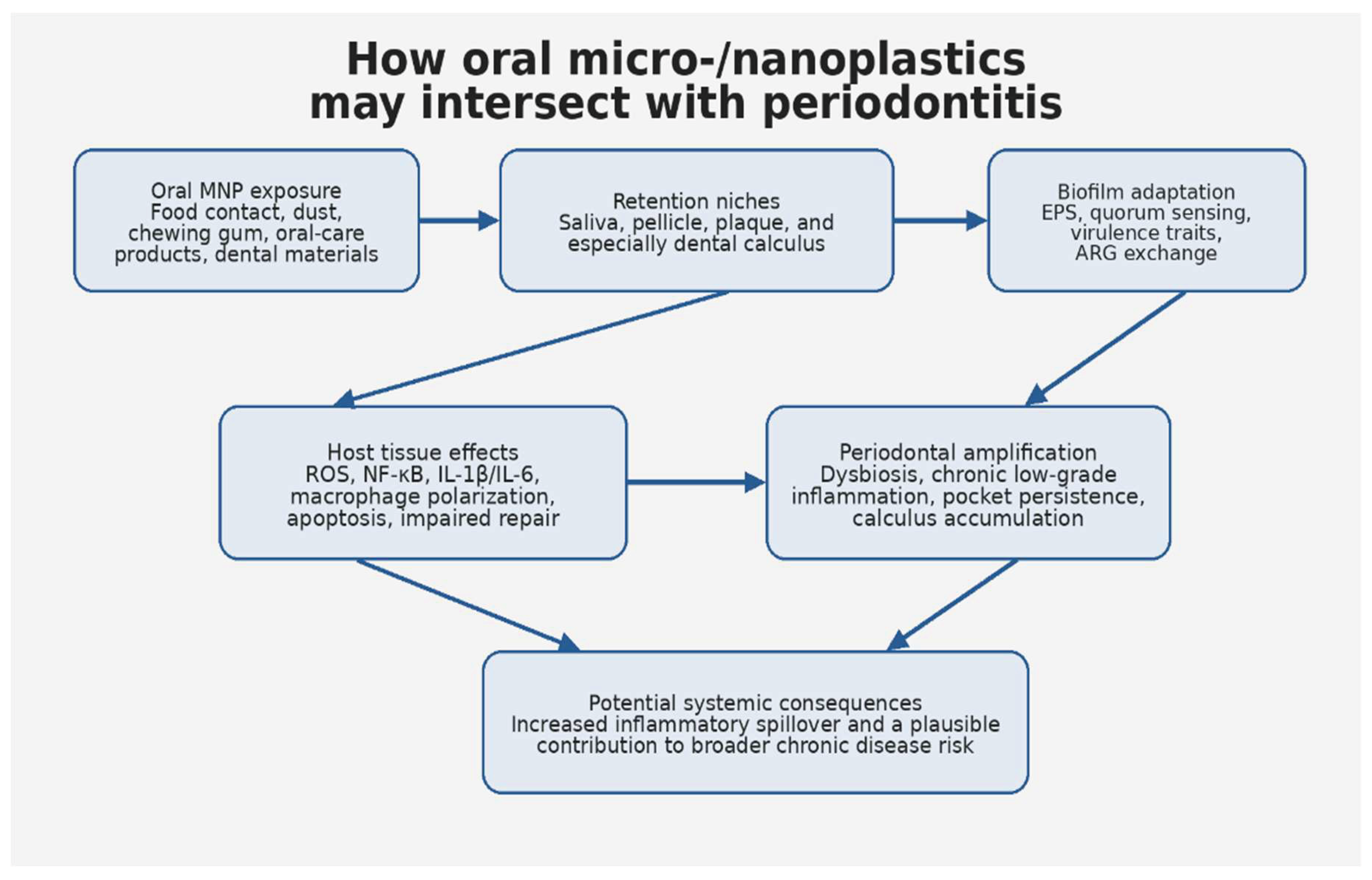
3. Periodontitis as a Chronic Low-Grade Inflammatory Interface
4. The Oral Cavity as an Exposure Portal and Local Source of Micro-/Nanoplastics
5. Direct Evidence for Oral Retention: Saliva, Plaque, and Dental Calculus
6. Mechanistic Convergence Between Micro-/Nanoplastic Toxicology and Periodontal Pathobiology
7. The Oral Biofilm as a Potential Plastic Reservoir and Pathogenic Amplifier
8. Bacterial Degradation of Plastics and the Special Case of Dental Polymers
9. Micro-/Nanoplastics, Chronic Disease, and the Possible Oral-Systemic Bridge
10. Evidence Synthesis: What Is Established, What Is Plausible, and What Remains Unresolved
11. Priority Research Agenda for Environmental Oral Microbiology
12. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Belstrøm, D.; Constancias, F.; Drautz-Moses, D. I.; Schuster, S. C.; Veleba, M.; Mahé, F.; Givskov, M. Periodontitis associates with species-specific gene expression of the oral microbiota. NPJ Biofilms and Microbiomes 2021, 7(1), 76. [Google Scholar] [CrossRef]
- Bishop, B.; Webber, W. S.; Atif, S. M.; Ley, A.; Pankratz, K. A.; Kostelecky, R.; Colgan, S. P.; Dinarello, C. A.; Zhang, W.; Li, S. Micro- and nano-plastics induce inflammation and cell death in human cells. Frontiers in Immunology 2025, 16, 1528502. [Google Scholar] [CrossRef] [PubMed]
- Bourbia, M.; Ma, D.; Cvitkovitch, D. G.; Santerre, J. P.; Finer, Y. Cariogenic bacteria degrade dental resin composites and adhesives. Journal of Dental Research 2013, 92(11), 989–994. [Google Scholar] [CrossRef] [PubMed]
- Bruno, A.; Dovizio, M.; Milillo, C.; Aruffo, E.; Pesce, M.; Gatta, M.; Chiacchiaretta, P.; Di Carlo, P.; Ballerini, P. Orally ingested micro- and nano-plastics: A hidden driver of inflammatory bowel disease and colorectal cancer. Cancers 2024, 16(17), 3079. [Google Scholar] [CrossRef]
- Cecoro, G.; Annunziata, M.; Iuorio, M. T.; Nastri, L.; Guida, L. Periodontitis, low-grade inflammation and systemic health: A scoping review. Medicina 2020, 56(6), 272. [Google Scholar] [CrossRef]
- Chen, J.; Cheng, Y.; Fu, R.; Chen, X.; Zhang, P.; Lu, Y.; Liu, B.; Chen, P.; Wang, J.; Cao, H.; Gu, J.; Chen, H.; Jiang, Z.; Li, T.; Zhang, J.; Chen, B.; Cao, G. Multiomics reveals nonphagocytosable microplastics induce colon inflammatory injury via bile acid-gut microbiota interactions and barrier dysfunction. ACS Applied Materials & Interfaces 2025, 17(31), 44138–44159. [Google Scholar] [CrossRef]
- Delaviz, Y.; Finer, Y.; Santerre, J. P. Biodegradation of resin composites and adhesives by oral bacteria and saliva: A rationale for new material designs that consider the clinical environment and treatment challenges. Dental Materials 2014, 30(1), 16–32. [Google Scholar] [CrossRef]
- Di Spirito, F.; Folliero, V.; Di Palo, M. P.; De Benedetto, G.; Aulisio, L.; Martina, S.; Rinaldi, L.; Franci, G. Micro- and nanoplastics and the oral cavity: Implications for oral and systemic health, dental practice, and the environment—A narrative review. Journal of Functional Biomaterials 2025, 16(9), 332. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.; Chen, R.; Xie, X.; Chen, Z.; Yang, D.; Hao, C.; Wang, S. AIM2-driven inflammation in periodontitis: Mechanisms and systemic implications. Journal of Inflammation Research 2025, 18, 6983–6997. [Google Scholar] [CrossRef] [PubMed]
- Francis, D. L.; Reddy, S. S. P. Microplastics in the pathogenesis of periodontal diseases: A narrative review. Annals of Global Health 2025, 91(1), 69. [Google Scholar] [CrossRef]
- Garcia, M. A.; Liu, R.; Nihart, A.; El Hayek, E.; Castillo, E.; Barrozo, E. R.; Suter, M. A.; Bleske, B.; Scott, J.; Forsythe, K.; Gonzalez-Estrella, J.; Aagaard, K. M.; Campen, M. J. Quantitation and identification of microplastics accumulation in human placental specimens using pyrolysis gas chromatography mass spectrometry. Toxicological Sciences 2024, 199(1), 81–88. [Google Scholar] [CrossRef] [PubMed]
- Hajishengallis, G. Periodontitis: From microbial immune subversion to systemic inflammation. Nature Reviews Immunology 2015, 15(1), 30–44. [Google Scholar] [CrossRef]
- Hajishengallis, G.; Chavakis, T. Local and systemic mechanisms linking periodontal disease and inflammatory comorbidities. Nature Reviews Immunology 2021, 21(7), 426–440. [Google Scholar] [CrossRef]
- Howard, S. A.; de Dios, R.; Maslova, E.; Myridakis, A.; Miller, T. H.; McCarthy, R. R. Pseudomonas aeruginosa clinical isolates can encode plastic-degrading enzymes that allow survival on plastic and augment biofilm formation. Cell Reports 2025, 44(5), 115650. [Google Scholar] [CrossRef]
- Huang, P.; Li, Z.; Liu, R.; Bartlam, M.; Wang, Y. Polystyrene nanoparticles induce biofilm formation in Pseudomonas aeruginosa. Journal of Hazardous Materials 2024, 469, 133950. [Google Scholar] [CrossRef]
- Hsiao, H.-Y.; Nien, C.-Y.; Shiu, R.-F.; Chin, W.-C.; Yen, T.-H. Microplastic and nanoplastic exposure and risk of diabetes mellitus. World Journal of Clinical Cases 2025, 13(3), 98110. [Google Scholar] [CrossRef]
- Jung, J.-S.; Kook, J.-K.; Park, S.-N.; Lim, Y. K.; Choi, G. H.; Kim, S.; Ji, S. Salivary microbiota reflecting changes in subgingival microbiota. Microbiology Spectrum 2024, 12(11), e01030-24. [Google Scholar] [CrossRef]
- Leslie, H. A.; van Velzen, M. J. M.; Brandsma, S. H.; Vethaak, A. D.; Garcia-Vallejo, J. J.; Lamoree, M. H. Discovery and quantification of plastic particle pollution in human blood. Environment International 2022, 163, 107199. [Google Scholar] [CrossRef]
- Liu, S.; Wang, C.; Yang, Y.; Du, Z.; Li, L.; Zhang, M.; Ni, S.; Yue, Z.; Yang, K.; Wang, Y.; Li, X.; Yang, Y.; Qin, Y.; Li, J.; Yang, Y.; Zhang, M. Microplastics in three types of human arteries detected by pyrolysis-gas chromatography/mass spectrometry (Py-GC/MS). Journal of Hazardous Materials 2024, 469, 133855. [Google Scholar] [CrossRef] [PubMed]
- Marashdeh, M. Q.; Gitalis, R.; Levesque, C.; Finer, Y. Enterococcus faecalis hydrolyzes dental resin composites and adhesives. Journal of Endodontics 2018, 44(4), 609–613. [Google Scholar] [CrossRef] [PubMed]
- Mazurek-Mochol, M.; Bonsmann, T.; Mochol, M.; Poniewierska-Baran, A.; Pawlik, A. The role of interleukin 6 in periodontitis and its complications. International Journal of Molecular Sciences 2024, 25(4), 2146. [Google Scholar] [CrossRef]
- Montenegro Raudales, J. L.; Yoshimura, A.; Sm, Z.; Kaneko, T.; Ozaki, Y.; Ukai, T.; Miyazaki, T.; Latz, E.; Hara, Y. Dental calculus stimulates interleukin-1β secretion by activating NLRP3 inflammasome in human and mouse phagocytes. PLOS ONE 2016, 11(9), e0162865. [Google Scholar] [CrossRef]
- Nijenhuis, W.; Houthuijs, K. J.; Brits, M.; van Velzen, M. J. M.; Brandsma, S. H.; Lamoree, M. H.; Béen, F. M. Improved multivariate quantification of plastic particles in human blood using non-targeted pyrolysis GC-MS. Journal of Hazardous Materials 2025, 489, 137584. [Google Scholar] [CrossRef]
- Ormsby, M. J.; Woodford, L.; White, H. L.; Fellows, R.; Quilliam, R. S. The plastisphere can protect Salmonella Typhimurium from UV stress under simulated environmental conditions. Environmental Pollution 2024, 358, 124464. [Google Scholar] [CrossRef]
- Pant, U.; Tate, J.; Liu, X.; Birse, N.; Elliott, C.; Cao, C. From automated Raman to cost-effective nanoparticle-on-film (NPoF) SERS spectroscopy: A combined approach for assessing micro- and nanoplastics released into the oral cavity from chewing gum. Journal of Hazardous Materials 2025, 486, 136978. [Google Scholar] [CrossRef]
- Saha, U.; Jena, S.; Simnani, F. Z.; Singh, D.; Choudhury, A.; Naser, S. S.; Lenka, S. S.; Kirti, A.; Nandi, A.; Sinha, A.; Patro, S.; Kujawska, M.; Suar, M.; Kaushik, N. K.; Ghosh, A.; Verma, S. K. The unseen perils of oral-care products generated micro/nanoplastics on human health. Ecotoxicology and Environmental Safety 2025, 290, 117526. [Google Scholar] [CrossRef]
- Singh, J.; Khalichi, P.; Cvitkovitch, D. G.; Santerre, J. P. Composite resin degradation products from BisGMA monomer modulate the expression of genes associated with biofilm formation and other virulence factors in Streptococcus mutans. Journal of Biomedical Materials Research Part A 2009, 88A(2), 551–560. [Google Scholar] [CrossRef] [PubMed]
- Skaba, D.; Fiegler-Rudol, J.; Dembicka-Mączka, D.; Wiench, R. Nanoplastics and immune disruption: A systematic review of exposure routes, mechanisms, and health implications. International Journal of Molecular Sciences 2025, 26(11), 5228. [Google Scholar] [CrossRef] [PubMed]
- Torrungruang, K.; Vathesatogkit, P.; Mahanonda, R.; Thienpramuk, L. Periodontitis and hypertension are linked through systemic inflammation: A 5-year longitudinal study. Journal of Clinical Periodontology 2024, 51(5), 536–546. [Google Scholar] [CrossRef] [PubMed]
- Velsko, I. M.; Overmyer, K. A.; Speller, C.; Klaus, L.; Collins, M. J.; Loe, L.; Frantz, L. A. F.; Sankaranarayanan, K.; Lewis, C. M., Jr.; Rodriguez Martinez, J. B.; Chaves, E.; Coon, J. J.; Larson, G.; Warinner, C. The dental calculus metabolome in modern and historic samples. Metabolomics 2017, 13(11), 134. [Google Scholar] [CrossRef]
- Wang, H.; Chen, H.; Ruan, C.; Liao, J.; Schwarz, C.; Shi, B.; Alvarez, P. J. J.; Yu, P. Nanoplastics induce prophage activation and quorum sensing to enhance biofilm mechanical and chemical resilience. Water Research 2026, 288 Pt B, 124712. [Google Scholar] [CrossRef]
- Warunek, J.; Warunek, S.; Calderon, M.; Franks, J.; Watkins, S.; Turnquist, H.; Al-Jewair, T. Orthodontic derived microplastics impact macrophage differentiation and homeostasis. Progress in Orthodontics 2026, 27(1), 3. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Yi, Y.; Yang, B.; Cui, X.; Chen, F.; Wu, G. Polyethylene: An identified component of human dental calculus triggers cytotoxicity and inflammatory responses in gingival fibroblasts. Environment International 2025, 203, 109759. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Dong, Z.; Zhang, S.; Ma, J.; Liu, S. Microplastic biofilm as hotspots of antibiotic resistance genes and potential pathogens. NPJ Biofilms and Microbiomes 2026, 12(1), 24. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zhang, G.; Zhang, D.; Zhu, N.; Bo, J.; Meng, X.; Chen, Y.; Qin, Y.; Liu, H.; Li, W. Microplastic biofilms promote the horizontal transfer of antibiotic resistance genes in estuarine environments. Marine Environmental Research 2024, 202, 106777. [Google Scholar] [CrossRef]
| Claim | Current evidence base | Strength of support | Interpretation |
|---|---|---|---|
| Human oral retention of MNPs occurs | Direct human evidence | Moderate-to-high | Microplastics detected in human dental calculus; strongest direct oral retention evidence (Wu et al., 2025). |
| MNPs can activate periodontal cell inflammatory pathways | Direct in vitro periodontal-cell evidence | Moderate | Polyethylene reduced gingival fibroblast viability, impaired migration, and activated NF-κB/IL-1β/IL-6 signaling (Wu et al., 2025). |
| The oral cavity is a relevant exposure and generation site | Direct oral exposure and dental material evidence | Moderate | Chewing gum, oral-care products, orthodontic materials, and dental polymers can release particles or polymer by-products (Pant et al., 2025; Saha et al., 2025; Warunek et al., 2026). |
| Plastic-associated biofilms can become more pathogenic or resilient | Strong non-oral experimental evidence; oral extrapolation | Moderate for general plastisphere biology; low-to-moderate for oral-specific translation | Biofilm promotion, EPS increase, quorum sensing activation, ARG transfer, and pathogen persistence reported in environmental systems (Huang et al., 2024; Zhou et al., 2024; Wang et al., 2026; Zhang et al., 2026). |
| Oral microbes degrade dental polymers, and polymer by-products can reshape biofilms | Direct oral microbiology and dental material evidence | High for methacrylate dental polymers | Cariogenic bacteria and E. faecalis degrade resins; BisGMA degradation products alter S. mutans virulence-related gene expression (Bourbia et al., 2013; Marashdeh et al., 2018; Singh et al., 2009). |
| Environmental MNP exposure causes human periodontitis | No direct longitudinal clinical proof | Insufficient | Currently unproven; supported mainly by mechanistic plausibility and limited direct oral evidence (Francis & Reddy, 2025). |
| Pathway | MNP-associated evidence | Periodontal relevance | Implication |
|---|---|---|---|
| Oxidative stress and NF-κB activation | Inflammatory signaling and redox imbalance after MNP exposure in oral and non-oral systems | Central to periodontal tissue destruction and host dysregulation | Shared pathway likely to intensify chronic inflammatory signaling |
| Inflammasome activity and IL-1β/IL-6 release | Gingival fibroblast and human cell studies show cytokine upregulation; calculus itself activates NLRP3 | IL-1β and IL-6 are core mediators of periodontitis and oral-systemic inflammation | Particle exposure may amplify an already activated periodontal cytokine network |
| Impaired migration, apoptosis, and wound repair | Polyethylene reduced fibroblast viability and migration; barrier injury was reported in gut models | Delayed epithelial/connective tissue repair supports lesion chronicity | MNPs may hinder resolution after periodontal injury or therapy |
| Macrophage and T-cell polarization | Orthodontic particles altered macrophage homeostasis; non-oral models show immune imbalance | Periodontitis is shaped by maladaptive innate and adaptive immunity | Combined exposure may favor persistent proinflammatory cell states |
| Biofilm EPS production, quorum sensing, and ARG transfer | Plastic particles enhance EPS, virulence traits, and horizontal gene transfer in environmental biofilms | Oral biofilms depend on matrix architecture, signaling, and coaggregation | Potential oral plastisphere mechanism requiring direct subgingival testing |
| Barrier dysfunction and translocation potential | Systemic tissue studies support internal exposure and barrier injury | Periodontal tissues already form an inflamed, permeable interface | Could increase the dissemination of inflammatory mediators, microbes, or particles |
| Research question | Recommended design | Key measurements | Why it matters |
|---|---|---|---|
| How common are MNPs in periodontal compartments? | Cross-sectional, contamination-controlled clinical sampling | Saliva, supra-/subgingival plaque, GCF, calculus, tissue; polymer fingerprinting; dental-material inventory | Establishes prevalence and source attribution by periodontal status |
| Where are particles located within oral biofilms? | Spatial imaging and correlative spectroscopy | Raman/FTIR/Py-GC-MS plus microscopy; matrix localization; particle size and charge | Determines whether particles are embedded in pathogenic biofilm microdomains |
| Do MNPs worsen dysbiosis or host injury? | Multi-species oral biofilm and host co-culture models | EPS, quorum sensing, virulence genes, invasion, cytokines, osteoimmune readouts | Tests causality in orally relevant systems rather than environmental surrogates |
| Are external and dentistry-derived plastics biologically distinct? | Comparative polymer-source studies | Environmental weathered particles versus orthodontic, restorative, and oral-care derived particles | Separates environmental exposure from treatment-related polymer burdens |
| Does oral plastic burden predict periodontal progression? | Prospective longitudinal cohorts | Periodontal parameters, plastic burden, inflammatory biomarkers, restorations, diet, smoking, therapy history | Moves the field from plausibility to temporality and risk estimation |
| Can intervention reduce plastic-associated inflammatory burden? | Clinical or translational intervention studies | Debridement, source reduction, lower-shedding materials, post-treatment polymer measurements | Provides immediate preventive and materials-science relevance |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




