Submitted:
01 April 2026
Posted:
02 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2.1. Marine Collagen Hydrolysates and Their Growing Role as Beauty Supplements
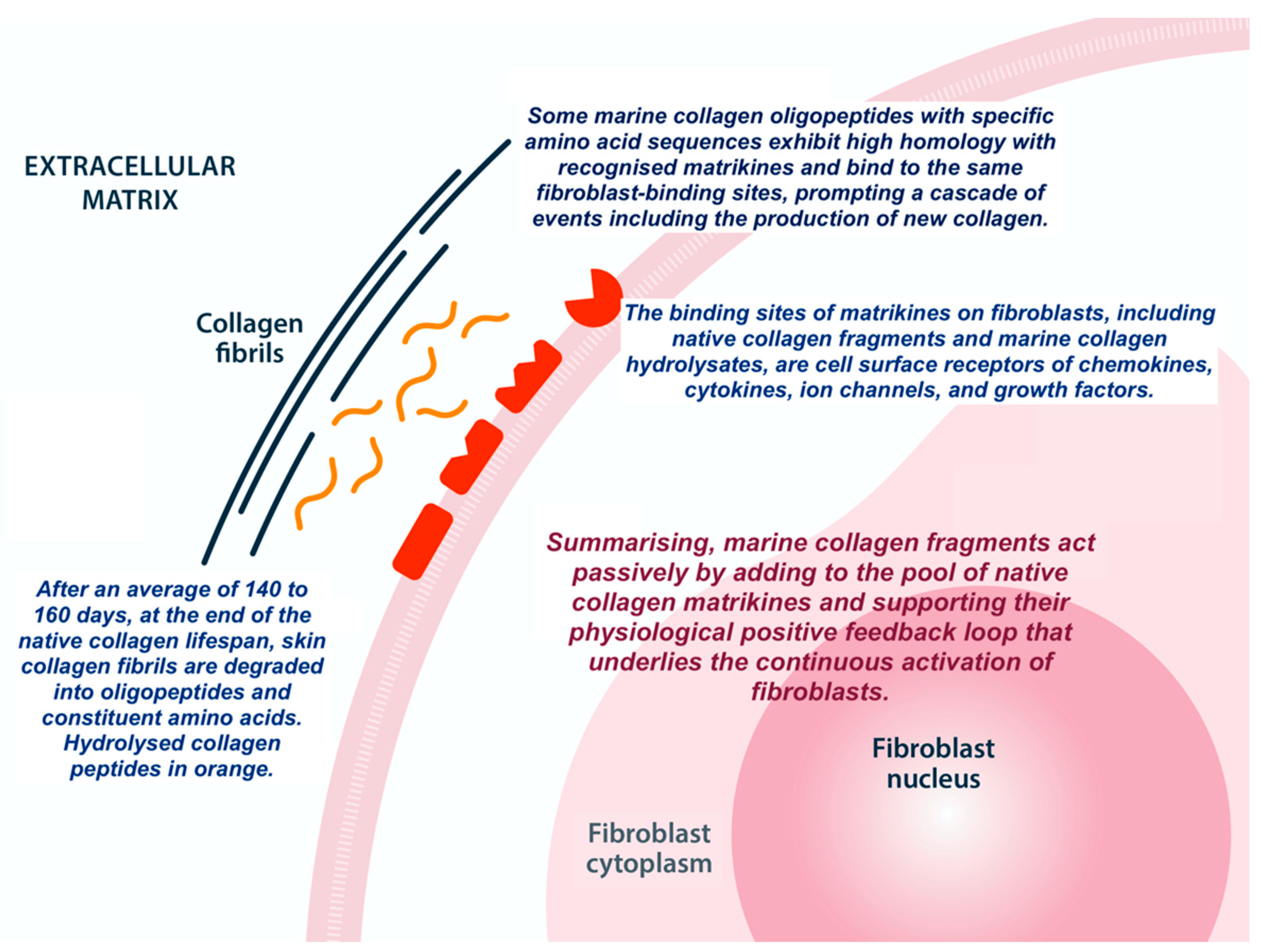
2.2. A Role for Marine Collagen Oligopeptides in Chronic Wound Healing Is Soundly Based
2. Material and Methods
- Inclusion only of well-described, methodologically sound, and reproducible studies, published in PubMed-indexed journals with notable impact factors.
- Exclusion of all studies of questionable quality published in non-indexed, non-peer-reviewed journals.
3. Results
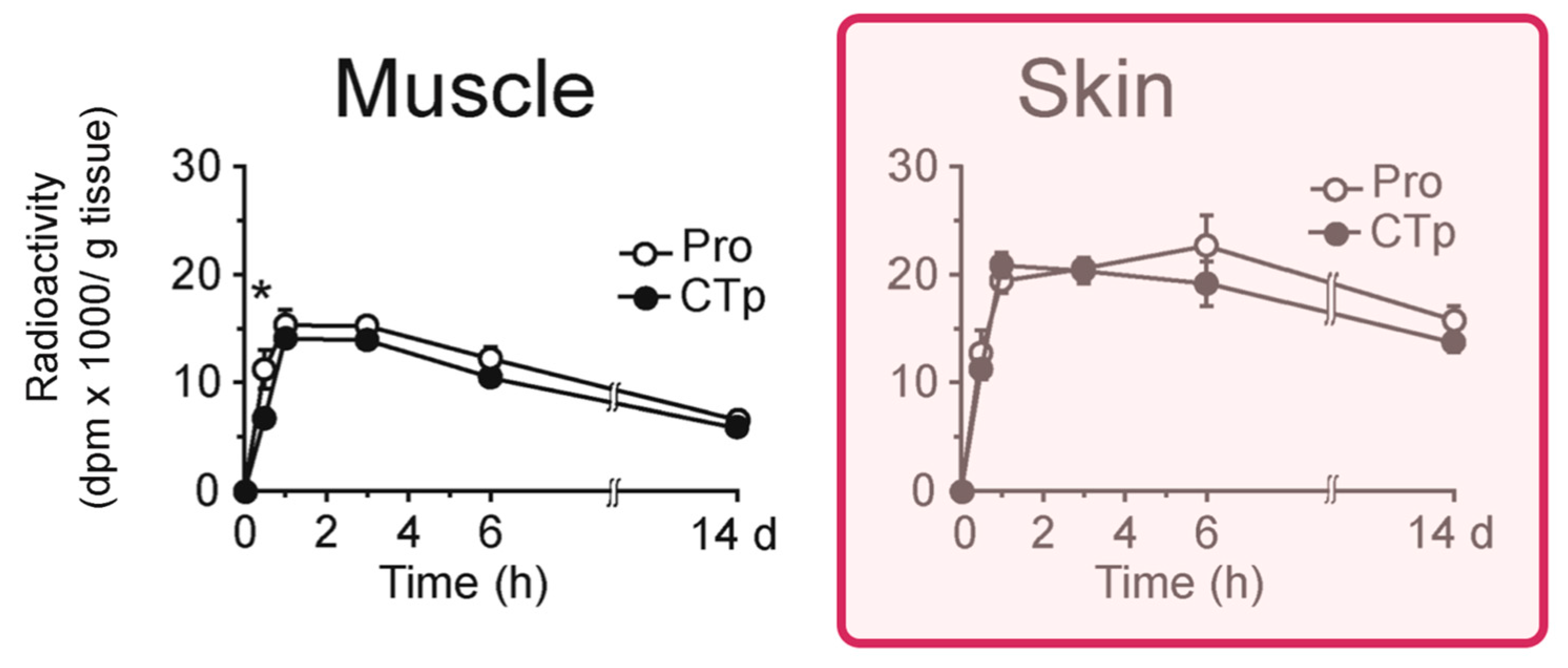
3.1. Safety and Oral-Route Bioavailability of Hydrolyzed Marine Collagens
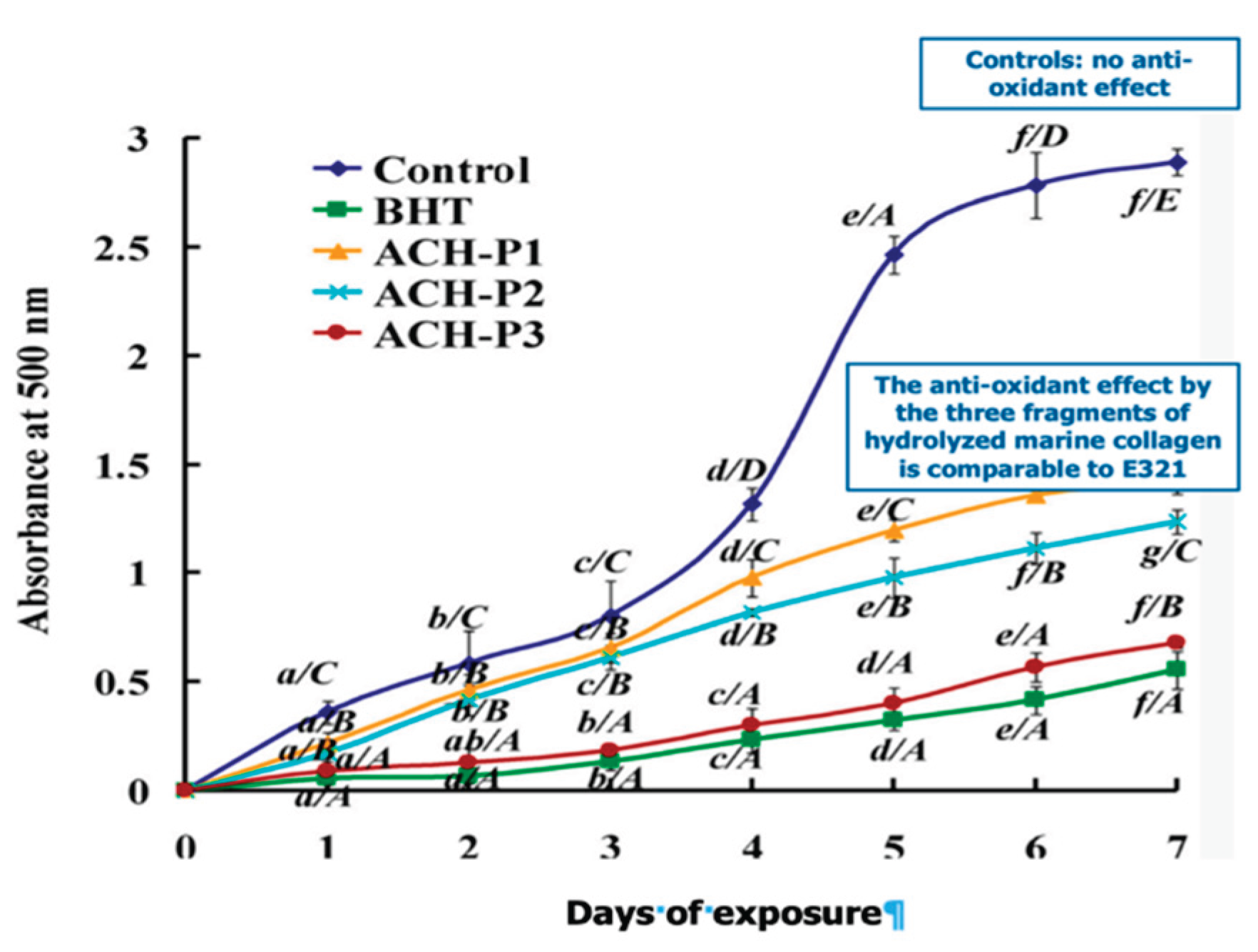
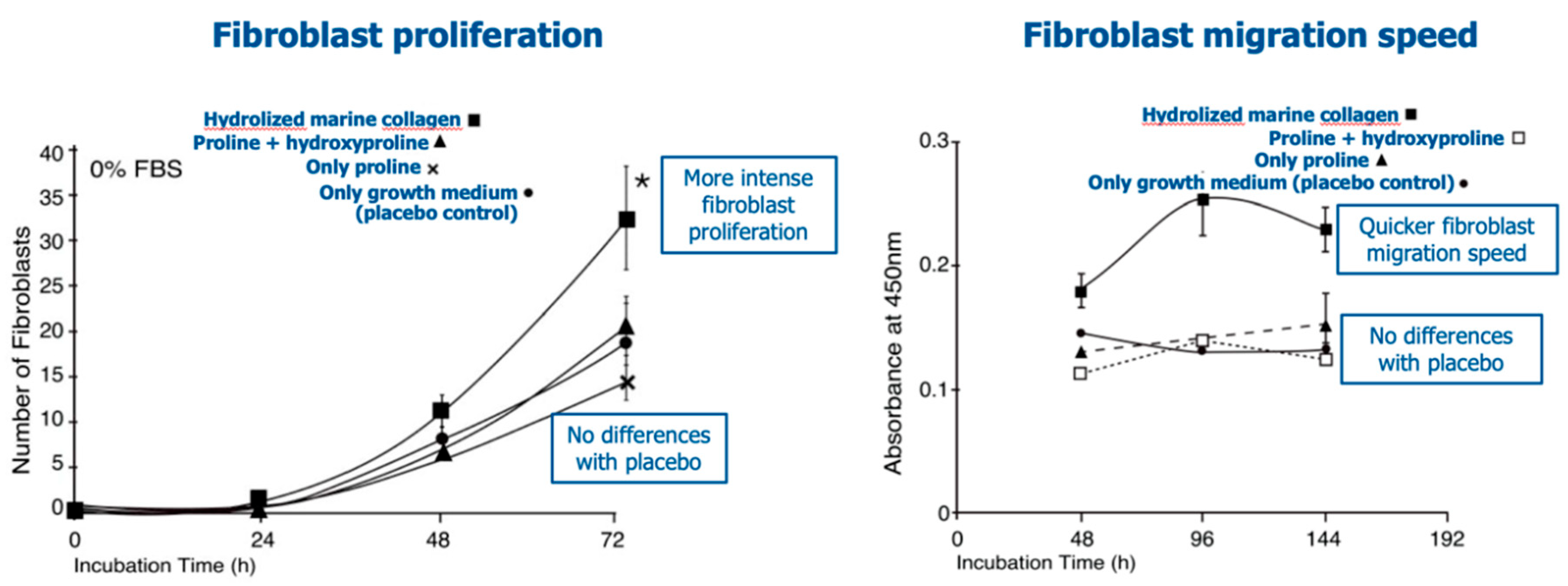
3.2. The Biological Properties of Hydrolyzed Marine Collagens
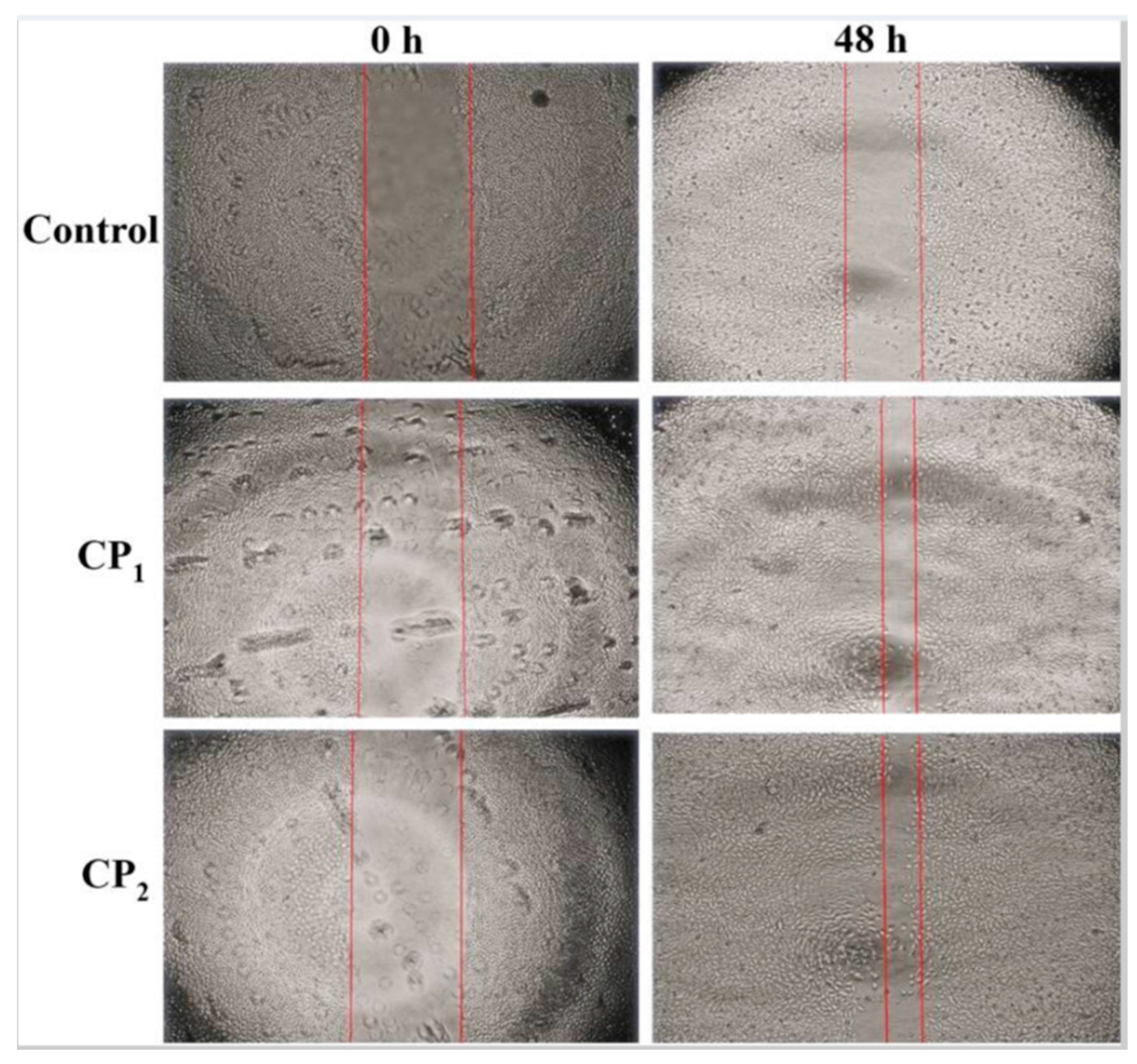
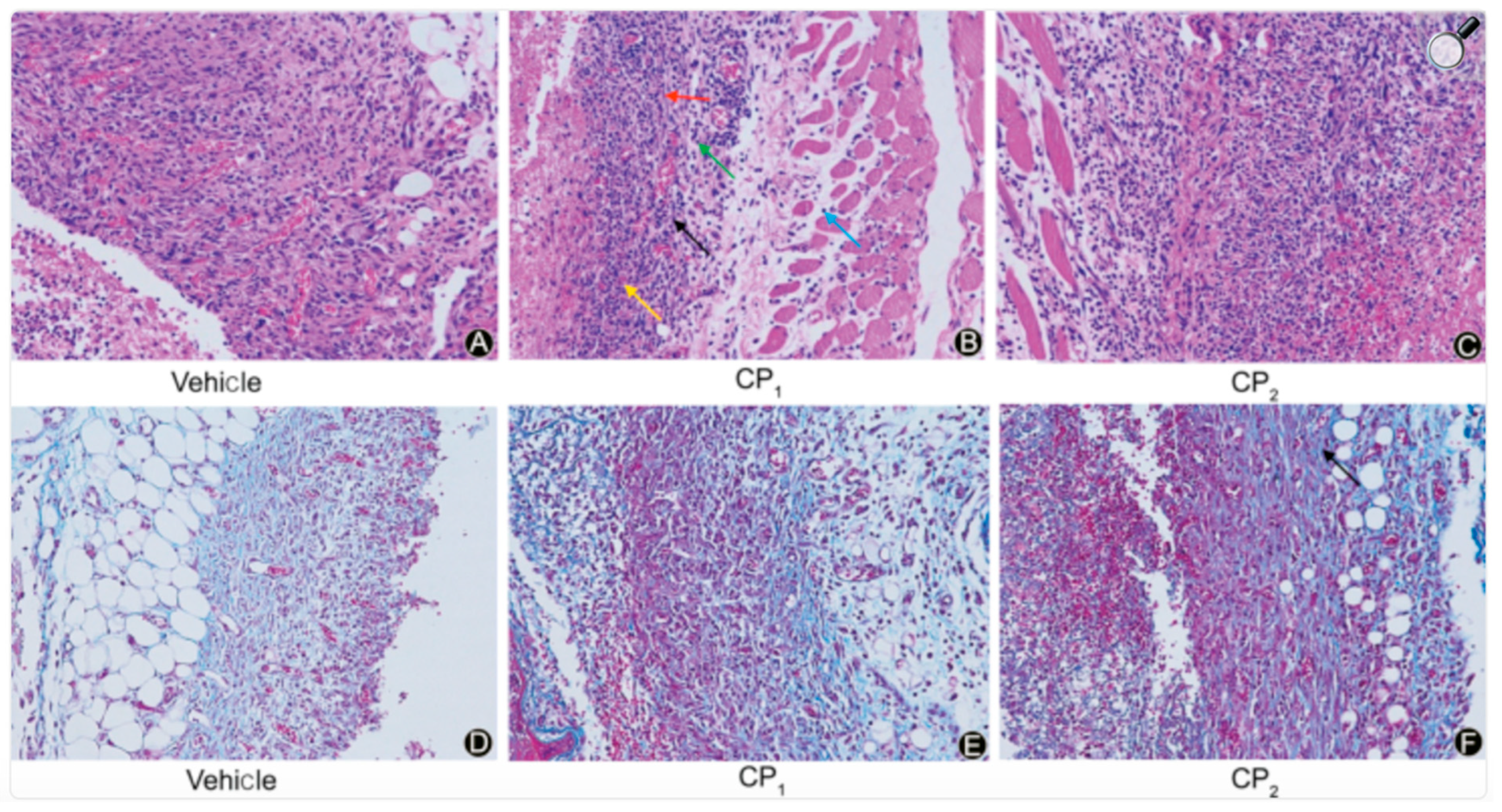
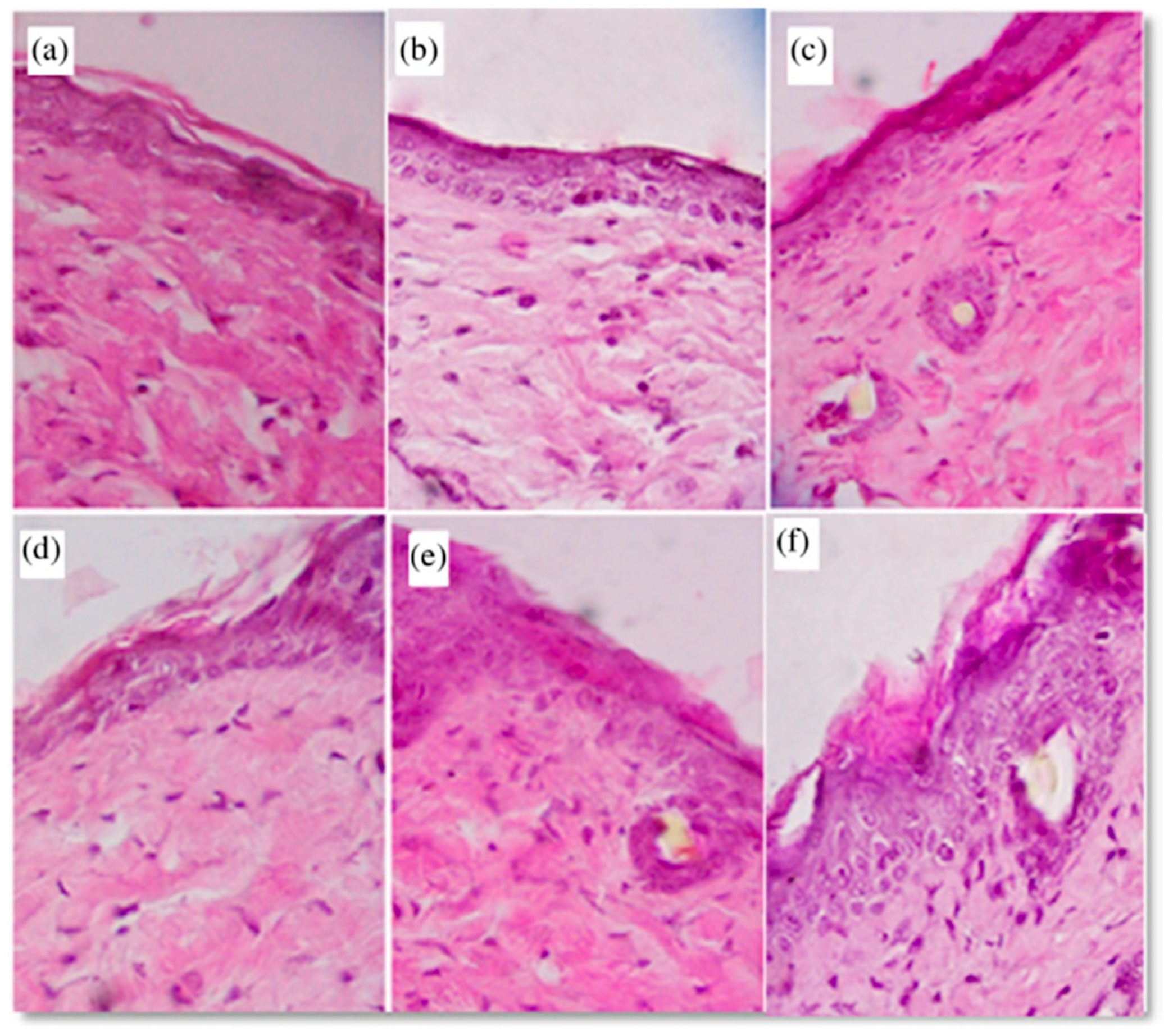
3.3. The Promising Rationale for Hydrolyzed Marine Collagens in Wound Care
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Global Beauty Supplements Market Outlook, 2024; Research and Markets: Dublin, Ireland, September 2024.
- Bartoletti, E.; Cavallini, C.; Klinger, M.E.A.; Lim, T.S.; Llorca Pérez, V.M.; Raichi, M. Hydrolyzed Marine Collagen: Emerging Evidence of Benefits Vva The Oral Route - Review And Insights For Medical Aesthetics Practitioners. Clin Cosmet Investig Dermatol. 2025, 18, 3339–3356. [Google Scholar] [CrossRef]
- Ma, X.; Chuang, P.H.; Tseng, Y.H.; Wang, X.; Ma, Z.; Chen, H.; Zhai, W.; Yang, W.; Meng, Z.; Xu, J. Progress in Research on Animal Collagen Peptides: Preparation, Bioactivity, and Application. Molecules. 2025, 30, 3061. [Google Scholar] [CrossRef]
- Coppola, D.; Oliviero, M.; Vitale, G.A.; Lauritano, C.; D’Ambra, I.; Iannace, S.; de Pascale, D. Marine Collagen from Alternative and Sustainable Sources: Extraction, Processing and Applications. Mar Drugs. 2020, 18, 214. [Google Scholar] [CrossRef]
- Geahchan, S.; Baharlouei, P.; Rahman, A. Marine Collagen: A Promising Biomaterial for Wound Healing & Skin Anti-Aging. Mar Drugs. 2022, 20, 61. [Google Scholar] [CrossRef]
- Ekmektzoglou, K.A.; Zografos, G.C. A Concomitant Review of the Effects of Diabetes Mellitus and Hypothyroidism in Wound Healing. World J Gastroenterol. 2006, 12, 2721–2729. [Google Scholar] [CrossRef] [PubMed]
- Goulding, V. The Effects of Diabetes on Collagen within Wound Healing. Diabetic Foot Journal. 2015, 18, 75–80. [Google Scholar]
- Huang, Y.; Kyriakides, T.R. The Role of Extracellular Matrix in The Pathophysiology of Diabetic Wounds. Matrix Biol Plus. 2020, 6-7, 100037. [Google Scholar] [CrossRef]
- Wang, P.; Sun, J.; Wang, Y.; Ren, B.; Liu, Y.; Liu, Y.; Chen, L.; Yu, L.; Dai, T.; Yu, L.; Dong, Z. Promotion of DFU Wound Healing via BRG1-COL16A1 Axis in Fibroblasts. Adv Sci (Weinh). 2026, e16687. [Google Scholar] [CrossRef]
- Li, X.; He, Z.; Long, C.; Chen, M.; Chen, M.; Zhang, L.; Luo, Z; Tian, J. Single-Cell Dissection of Fibroblast Heterogeneity in Diabetic Ulcers: Platelet-Rich Plasma (PRP) Therapy Activates Core Regenerative Programs via PLAGL1/RUNX2/ZKSCAN7 Networks. Front Biosci (Landmark Ed). 2025, 30, 47450. [Google Scholar] [CrossRef]
- Sugihara, F.; Inoue, N.; Venkateswarathirukumara, S. Ingestion of Bioactive Collagen Hydrolysates Enhanced Pressure Ulcer Healing in a Randomized Double-Blind Placebo-Controlled Clinical Study. Sci Rep. 2018, 8, 11403. [Google Scholar] [CrossRef]
- Sibilla, S.; Godfrey, M.; Brewer, S.; Budh-Raja, A.; Genovese, L. An Overview of the Beneficial Effects of Hydrolyzed Collagen as a Nutraceutical on Skin Properties: Scientific Background and Clinical Studies. Open Nutraceut J. 2015, 8, 29–42. [Google Scholar] [CrossRef]
- Watanabe-Kamiyama, M.; Shimizu, M.; Kamiyama, S.; Taguchi, Y.; Sone, H.; Morimatsu, F.; Shirakawa, H.; Furukawa, Y.; Koma, M. Absorption and Effectiveness of Orally Administered Low-Molecular-Weight Collagen Hydrolysate in Rats. J Agric Food Chem. 2010, 58, 835–841. [Google Scholar] [CrossRef]
- Jafari, H.; Lista, A.; Siekapen, M.M.; Ghaffari-Bohlouli, P.; Nie, L.; Alimoradi, H.; Shavandi, A. Fish Collagen: Extraction, Characterisation, and Applications for Biomaterials Engineering. Polymers (Basel). 2020, 12, 2230. [Google Scholar] [CrossRef]
- Islam, J.; Solval, K.E.M. Recent Advancements in Marine Collagen: Exploring New Sources, Processing Approaches, and Nutritional Applications. Mar Drugs. 2025, 23, 190. [Google Scholar] [CrossRef]
- Salvatore, L.; Gallo, N.; Natali, M.L.; Campa, L.; Lunetti, P.; Madaghiele, M.; Blasi, F.S.; Corallo, A.; Capobianco, L.; Sannino, A. Marine Collagen and Its Derivatives: Versatile and Sustainable Bio-Resources for Healthcare. Mater Sci Eng C Mater Biol Appl. 2020, 113, 110963. [Google Scholar] [CrossRef]
- Ricard-Blum, S.; Vallet, S.D. Fragments Generated upon Extracellular Matrix Remodeling: Biological Regulators and Potential Drugs. Matrix Biol. 2019, 75–76, 170–189. [Google Scholar] [CrossRef]
- Kisling, A.; Lust, R.M.; Katwa, L.C. What is the Role of Peptide Fragments of Collagen I and IV In Health and Disease? Life Sci. 2019, 228, 30–34. [Google Scholar] [CrossRef]
- Hu, Z.; Yang, P.; Zhou, C.; Li, S.; Hong, P. Marine Collagen Peptides from the Skin of Nile Tilapia (Oreochromis Niloticus): Characterization and Wound Healing Evaluation. Mar Drugs. 2017, 15, 102. [Google Scholar] [CrossRef]
- Liang, J.; Pei, X.; Zhang, Z.; Wang, N.; Wang, J.; Li, Y. The Protective Effects of Long-Term Oral Administration of Marine Collagen Hydrolysate from Chum Salmon on Collagen Matrix Homeostasis in the Chronologically Aged Skin of Sprague-Dawley Male Rats. J Food Sci. 2010, 75, H230–H238. [Google Scholar] [CrossRef]
- Kim, D.U.; Chung, H.C.; Choi, J.; Sakai, Y.; Lee, B.Y. Oral intake of Low-Molecular-Weight Collagen Peptide Improves Hydration, Elasticity, and Wrinkling in Human Skin: A Randomized, Double-Blind, Placebo-Controlled Study. Nutrients. 2018, 10, 826. [Google Scholar] [CrossRef]
- Aragona, S.E.; Mereghetti, G.; Ciprandi, G. LumiHeal® and Anti-Inflammatory Regenerative Medicine (AIMED): A New Management Strategy for Shortening Wound Healing and Effective Outcomes, from Repair 1.0 to Wound Regeneration 4.0. J Biol Regul Homeost Agents. 2022, 36, 87(S2)–95(S2). [Google Scholar] [CrossRef]
- Jariwala, N.; Ozols, M.; Bell, M.; Bradley, E.; Gilmore, A.; Debelle, L.; Sherratt, M.J. Matrikines as Mediators of Tissue Remodelling. Adv Drug Deliv Rev. 2022, 185, 114240. [Google Scholar] [CrossRef]
- Ma, Y.; Kleinbeck, K.; Kao, W.J. Extracellular Matrix-Derived Tripeptide Proline-Glycine-Proline Inhibits Keratinocyte Proliferation and Migration. Wound Repair Regen. 2011, 19, 718–726. [Google Scholar] [CrossRef]
- Kwon, Y.W.; Heo, S.C.; Lee, T.W.; Park, G.T.; Yoon, J.W.; Jang, I.H.; Kim, S.C.; Ko, H.C.; Ryu, Y.; Kang, H.; et al. N-Acetylated Proline-Glycine-Proline Accelerates Cutaneous Wound Healing and Neovascularisation by Human Endothelial Progenitor Cells. Sci Rep. 2017, 7, 43057. [Google Scholar] [CrossRef]
- Dang, Q.F.; Liu, H.; Yan, J.Q.; Liu, C.S.; Liu, Y.; Li, J.J. Characterisation of Collagen from Haddock Skin and Wound Healing Properties of its Hydrolysates. Biomed Mater. 2015, 10, 015022. [Google Scholar] [CrossRef]
- Wang, J.; Xu, M.; Liang, R.; Zhao, M.; Zhang, Z.; Li, Y. Oral Administration of Marine Collagen Peptides Prepared from Chum Salmon (Oncorhynchus keta) Improves Wound Healing Following Cesarean Section in Rats. Food Nutr Res. 2015, 59, 26411. [Google Scholar] [CrossRef]
- Yang, F.; Jin, S.; Tang, Y. Marine Collagen Peptides Promote Cell Proliferation Of NIH-3T3 Fibroblasts via NF-κB Signaling Pathway. Molecules. 2019, 24, 4201. [Google Scholar] [CrossRef]
- Zhou, M.; Ren, G.; Zhang, B.; Ma, F.; Fan, J.; Qiu, Z. Screening and identification of a novel antidiabetic peptide from collagen hydrolysates of Chinese giant salamander skin: Network pharmacology, inhibition kinetics and protection of IR-HepG2 cells. Food Funct. 2022, 13(6), 3329–3342. [Google Scholar] [CrossRef]
- Natsir, H.; Dali, S.; Arif, A.R.; Sartika; Leliani. Activity and kinetics of α-glucosidase inhibition by collagen hydrolysate from Thunnus albacares bone. J Phys Conf Ser. 2019, 1341, 032015. [Google Scholar] [CrossRef]
- Hatanaka, T.; Kawakami, K.; Uraji, M. Inhibitory effect of collagen-derived tripeptides on dipeptidylpeptidase-IV activity. J Enzyme Inhib Med Chem. 2014, 29, 823–828. [Google Scholar] [CrossRef]
- Kusuma, R.J.; Ermamilia, A.; Halimah, S.N.; Pradani, N.F.; Sholikha, I. Selar (Selar crumenophthalmus) fish protein hydrolysate has antidiabetic properties possibly through GLP-1. Curr Nutr Food Sci. 2020, 17, 516–522. [Google Scholar] [CrossRef]
- Zheng, H.; Zhao, L.; Xie, Y; Tan, Y. Purification identification of novel dipeptidyl peptidase IV inhibitory peptides derived from bighead carp (Hypophthalmichthys nobilis). Foods. 2024, 13, 2644. [Google Scholar] [CrossRef]
- Devasia, S.; Kumar, S.; Stephena, P.S.; Inoue, N.; Sugihara, F.; Koizumi, S.; Matsushita, A.; Sriraam, V.T. A double blind, randomised, four-arm clinical study to evaluate the safety, efficacy, and tolerability of collagen peptide as a nutraceutical therapy in the management of type II diabetes mellitus. J Diabetes Metab. 2019, 10, 1–7. [Google Scholar] [CrossRef]
- Alexiadou, K.; Doupis, J. Management of diabetic foot ulcers. Diabetes Ther. 2012, 3, 4. [Google Scholar] [CrossRef]
- Dewi, D.A.R.; Arimuko, A.; Norawati, L.; Yenny, S.W.; Setiasih, N.L.; Perdiyana, A.; Arkania, N.; Nadhira, F.; Wiliantari, N. Exploring the impact of hydrolyzed collagen oral supplementation on skin rejuvenation: A systematic review and meta-analysis. Cureus. 2023, 15, e50231. [Google Scholar] [CrossRef]
- Mokari-Yamchi, A.; Davoodi, S.H. Collagen supplementation for skin health: A mechanistic systematic review. J Cosmet Dermatol. 2020, 19, 2820–2829. [Google Scholar] [CrossRef]
- Ravindran, R.; Pizzol, D.; López-Gil, JF.; Rahmati, M.; Boyer, L.; Fond, G.; Butler, L.; Stellato, A.; Gawronska, J.; Barnett, Y.; et al. Collagen supplementation for skin and musculoskeletal health: An umbrella review of meta-analyses on elasticity, hydration, and structural outcomes. Aesthet Surg J Open Forum. 2026, 8, ojag018. [Google Scholar] [CrossRef]
- Myung, S.K.; Park, Y. Effects of collagen supplements on skin aging: A systematic review and meta-analysis of randomized controlled trials. Am J Med. 2025, 138, 1264–1277. [Google Scholar] [CrossRef]
- Shanks, N.; Greek, R.; Greek, J. Are animal models predictive for humans? Philos Ethics Humanit Med. 2009, 4, 2. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



