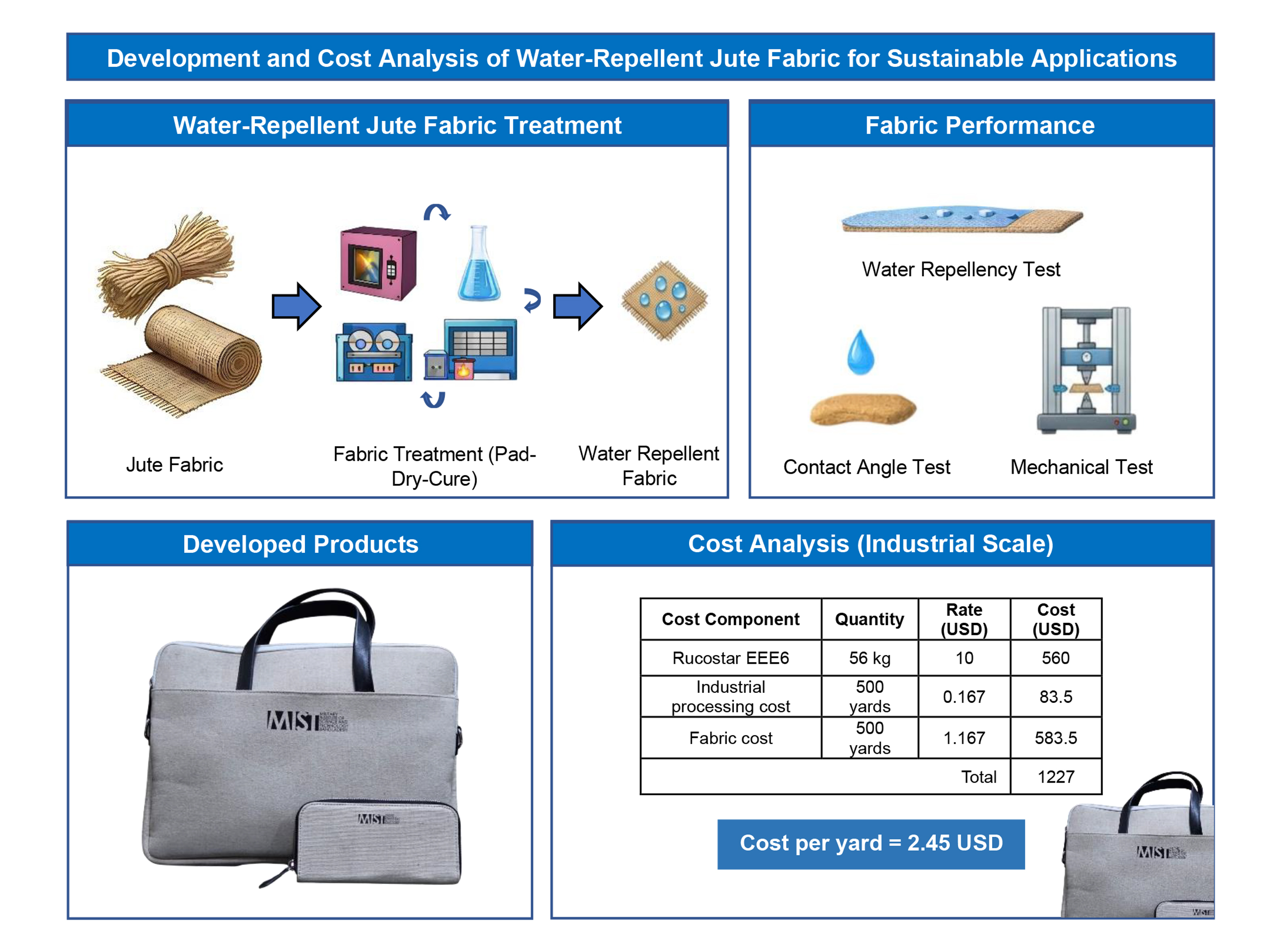
1. Introduction
Jute is a natural lignocellulosic fiber, extracted from the bast fibers of plants belonging to the genus
Corchorus, essentially
Corchorus capsularis and
Corchorus olitorius. It is one of the most abundant, least expensive, and eco-friendly natural fibers that is extensively grown in such countries as Bangladesh and India [
1]. Jute is also biodegradable, renewable, compostable and that is why jute is also an attractive substitute to the synthetic materials. Jute materials are biodegradable under the influence of microorganisms to form carbon dioxide, water, and biomass without polluting the environment, and this process is referred to as bio-degradation. On the other hand, plastic is an artificial polymer, a bulk of which is petrochemical material and is commonly used in packaging due to its light weight, durability and immense resistance to water. Plastic packaging mediums such as polyethylene and polypropylene are typically used as a shopping bag, wrapping food and as a food storage medium [
2]. However, plastic materials cannot be biodegraded and may stay in the environment for hundreds of years, posing a great environmental and pollution threat to the surroundings and the oceans. Therefore, the sustainable alternatives are in high demand. Jute can be used as an alternative to the plastic packaging material since it is biodegradable and natural [
3]. Jute is predominantly biodegradable because of its chemical composition which is primarily cellulose (approximately 60 to 65 percent), hemicellulose (20 to 25 percent) and lignin (10 to 15 percent). The hydroxyl (-OH) groups of cellulose and hemicellulose are numerous, which makes jute susceptible to microbial decay, and thus in this manner, this material can be disposed in an environmentally-friendly manner [
4,
5].
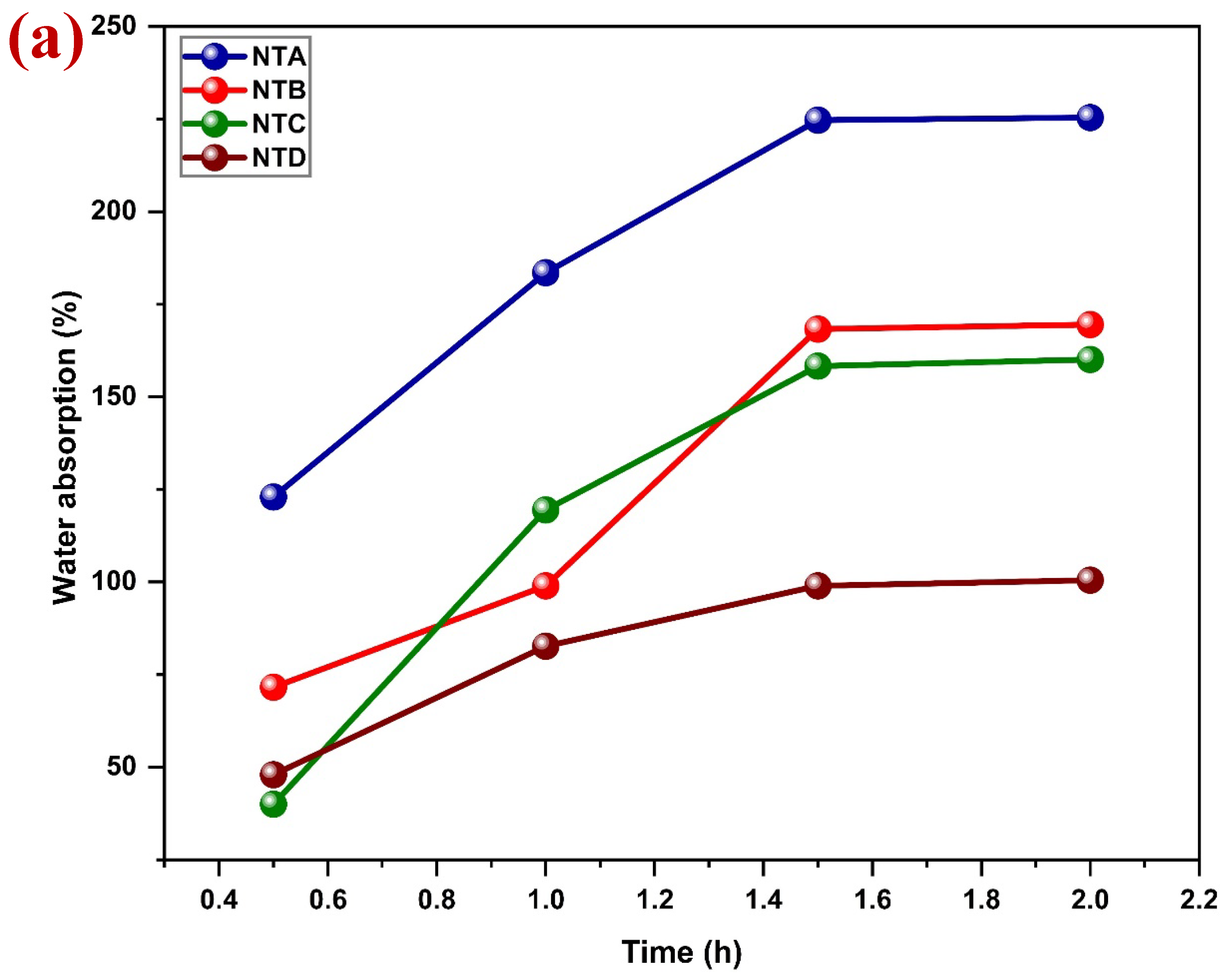
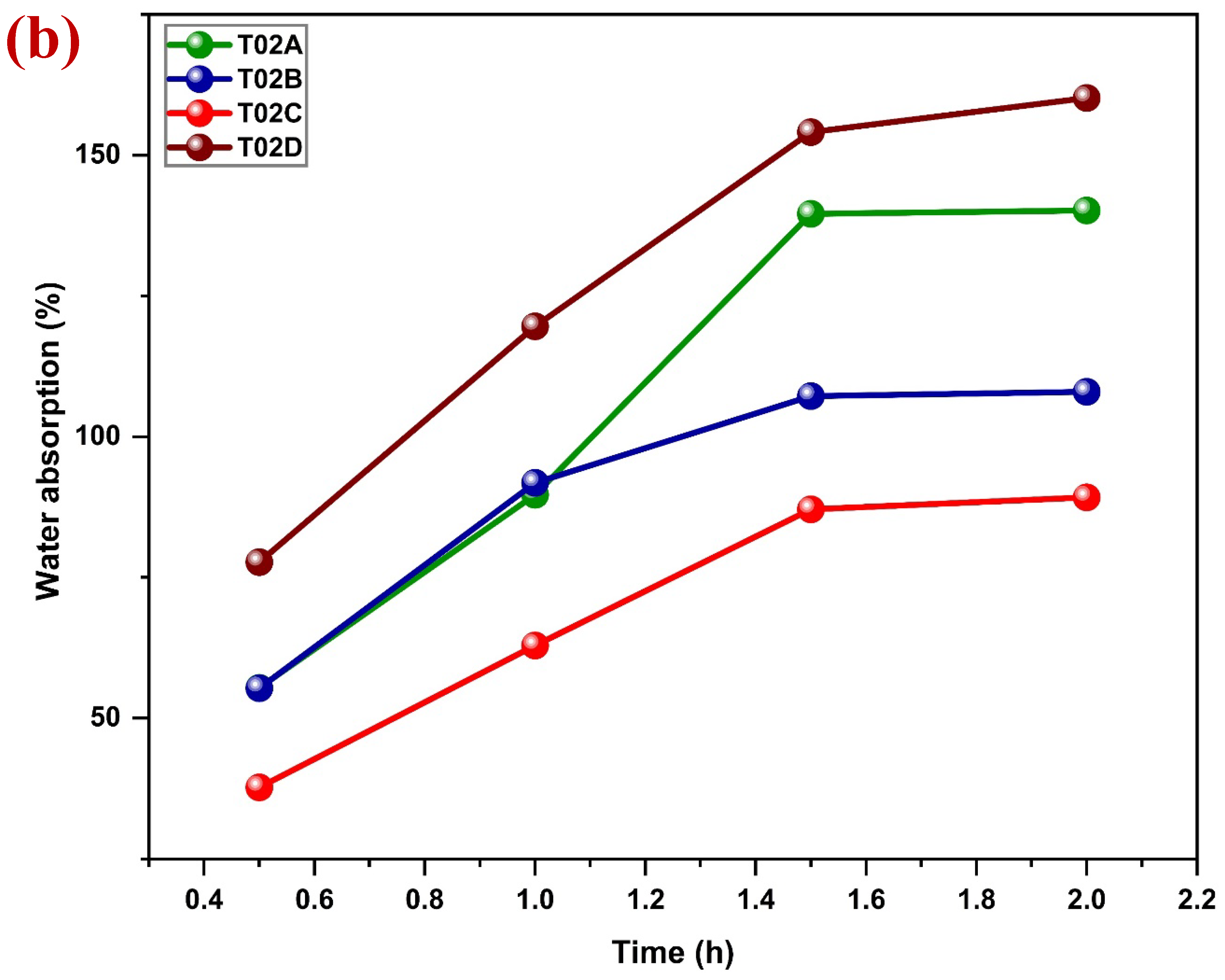
Although jute is environmentally friendly, biodegradable in its untreated form, it cannot be used directly as an alternative to plastic packaging materials. The main disadvantage of jute is connected with its chemical structure. The main components of jute (cellulose and hemicellulose) have large numbers of hydrophilic hydroxyl (-OH) groups. This makes the hydroxyl groups easily interact as hydrogen bonds with the water molecules and thus high moisture absorbency. As a result, untreated jute has high water absorption, fiber expansion, and lack of dimensional stability as well as mechanical property degradation [
6,
7]. On the other hand, plastic materials are water resistant and their absorption of moisture is very low, but they are not biodegradable and last longer in the environment. Therefore, to convert jute into practical packaging materials, such as shopping bags, it is necessary to impart durable water-resistant properties while maintaining its biodegradable nature [
8]. Water repellency in jute fabrics is typically achieved through chemical treatments, including paraffin wax, silicone-based finishes, polyurethane coatings, and fluorocarbon resins. Fluorocarbon-based finishes are particularly favored due to their low surface energy, strong hydrophobicity, and high wear resistance [
9,
10]. Commonly used commercial water-repellent agents are Nuva N2114 and Scotchgard [
11]. In this study, we employed Rucostar EEE6 (C
6-Fluorocarbon resin with hyperbranched polymers in a hydrocarbon matrix) for water-repellent treatment in the jute fabrics.
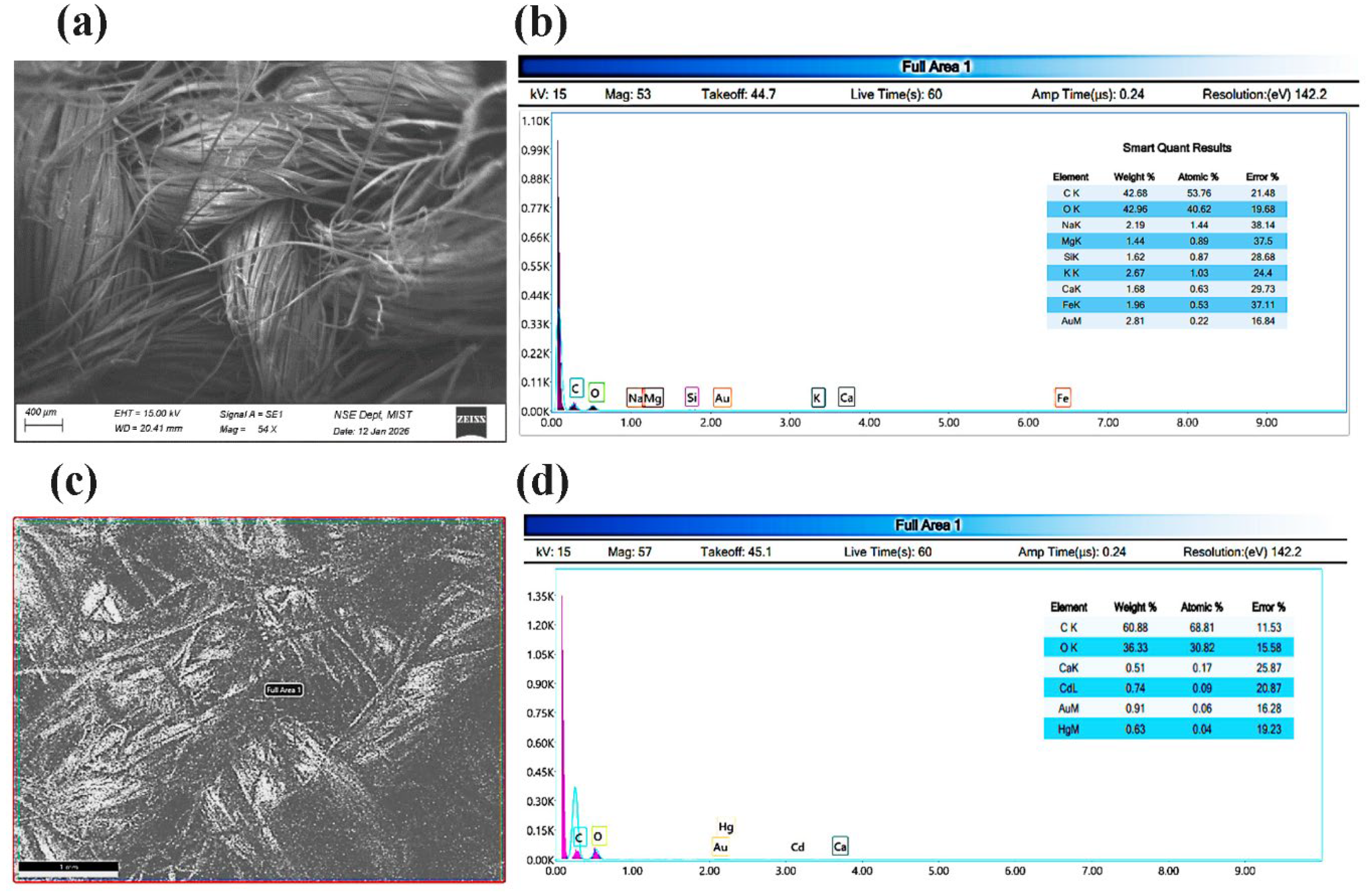
Scanning Electron Microscopy (SEM) is usually employed examine and compare the morphology of the surface of the textile fibers prior to and following the chemical treatment. SEM offers high-resolution microstructural images, which can be used to study the structure of the fibers and surface roughness, depth of coating, and the existence of the deposited finishing layers [
12,
13]. It is able to detect structural components in jute fabrics, which are cellulose microfibrils, lignin-rich surface layers, hemicellulosic matrix regions, and natural surface impurities as well as physical defects such as fibrillation, voids as well as fiber breakage. Besides morphological analysis. SEM and elemental analysis allow one to detect chemical elements on the surface of the fiber. As jute is mainly formed by cellulose, hemicellulose, and lignin, the main elements that can be detected are carbon (C) and oxygen (O), whereas minor elements can also be identified such as calcium (Ca), magnesium (Mg), potassium (K), silicon (Si), and trace iron (Fe), are also present in natural minerals. Following the chemical treatment, other elements can be present and will appear depending on which finishing agent was used, i.e., fluorine (F) with fluorocarbon resin or silicon (Si) with silicone-based coatings, which proves successful surface modification [
14]. The process of creating continuous layers of polymer, sealing the pores of the surface, flattening the surface of the fiber, and the introduction of new elements can thus be well observed and confirmed.
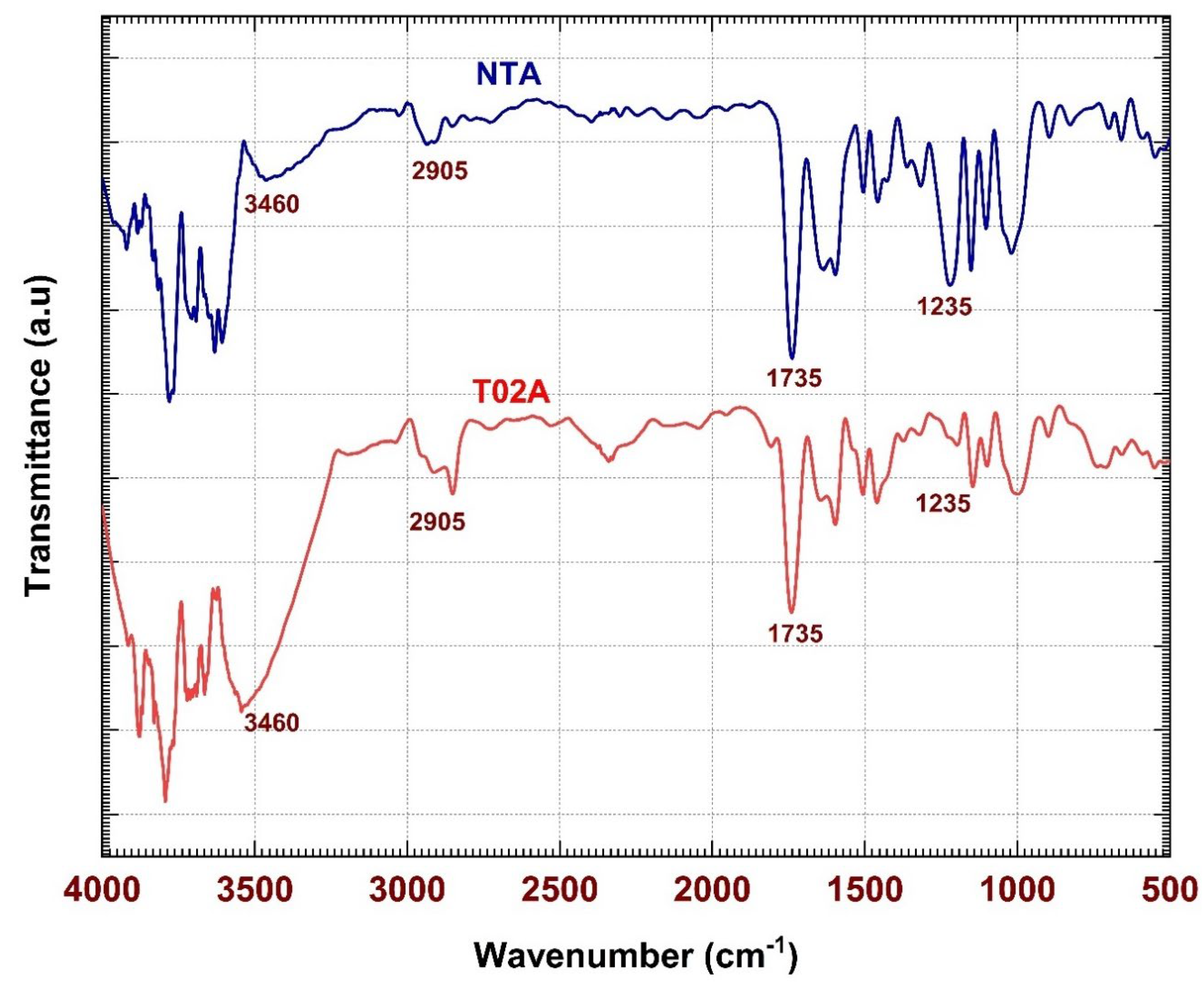
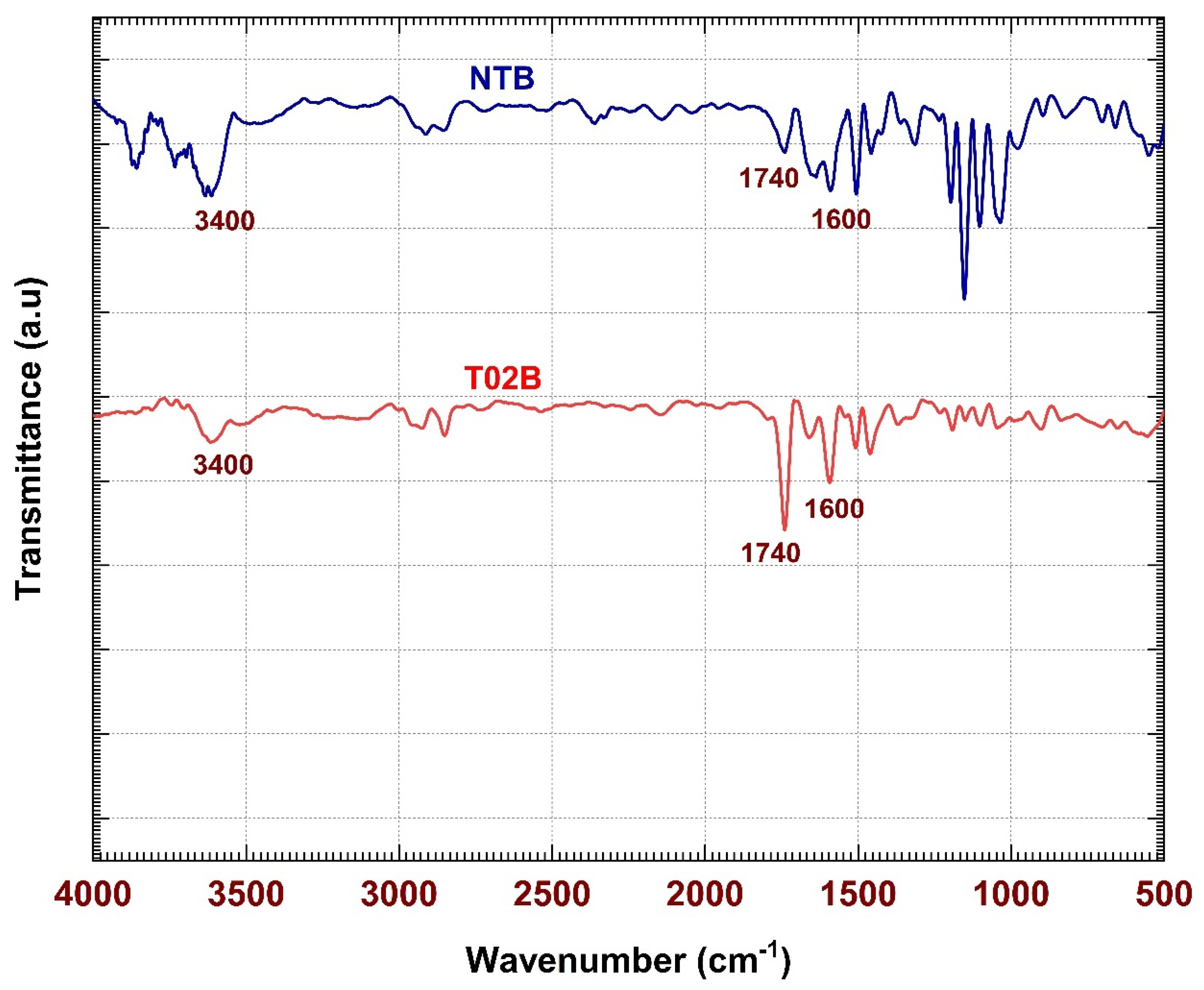
The FTIR spectroscopy is an effective tool used to study the chemical structure and functional groups of the textile materials [
15,
16]. FTIR analysis incorporates characteristic bands of absorption with respect to specific molecular vibrations, and as a result, the interaction of chemicals between fibers and finishing agents can be detected. This method is also helpful especially in distinguishing between physically deposited and chemically bonded finishes on the surface of the fibers. The intensity of changes in the peaks, or alternation of the hydroxyl group absorption bands may indicate the chemical modification of cellulose [
17]. Thus, it will be necessary to verify the creation of new bonds and to see how the process of applying a water-repellent finishing works.
Tensile strength test is regularly performed to test the mechanical performance of textile materials. It is used to determine the maximum force required to break a fabric, which can determine the strength, elongation, and structural integrity of the fabric [
18,
19,
20]. The chemical treatment can potentially interfere with fiber bonding and internal structure. So, tensile testing is required to guarantee that finishing procedures do not undermine durability. In packaging applications, tensile strength has to be sufficient to resist handling and load bearing requirements.
Surface wetting resistance of fabrics is measured by the spray rating test, which is commonly done based on the AATCC Method 22. The test is used to determine the effectiveness of a fabric in an exposure to a controlled water spray when it has to be resistant to water penetration [
21]. The wetting pattern is compared visually to a standardized rating scale with a rating of 100 being total surface water repellency. Spray test has been extensively utilized in the studies of textile finishing due to the simulation of the actual conditions of being exposed to rain or splashing water [
22,
23].
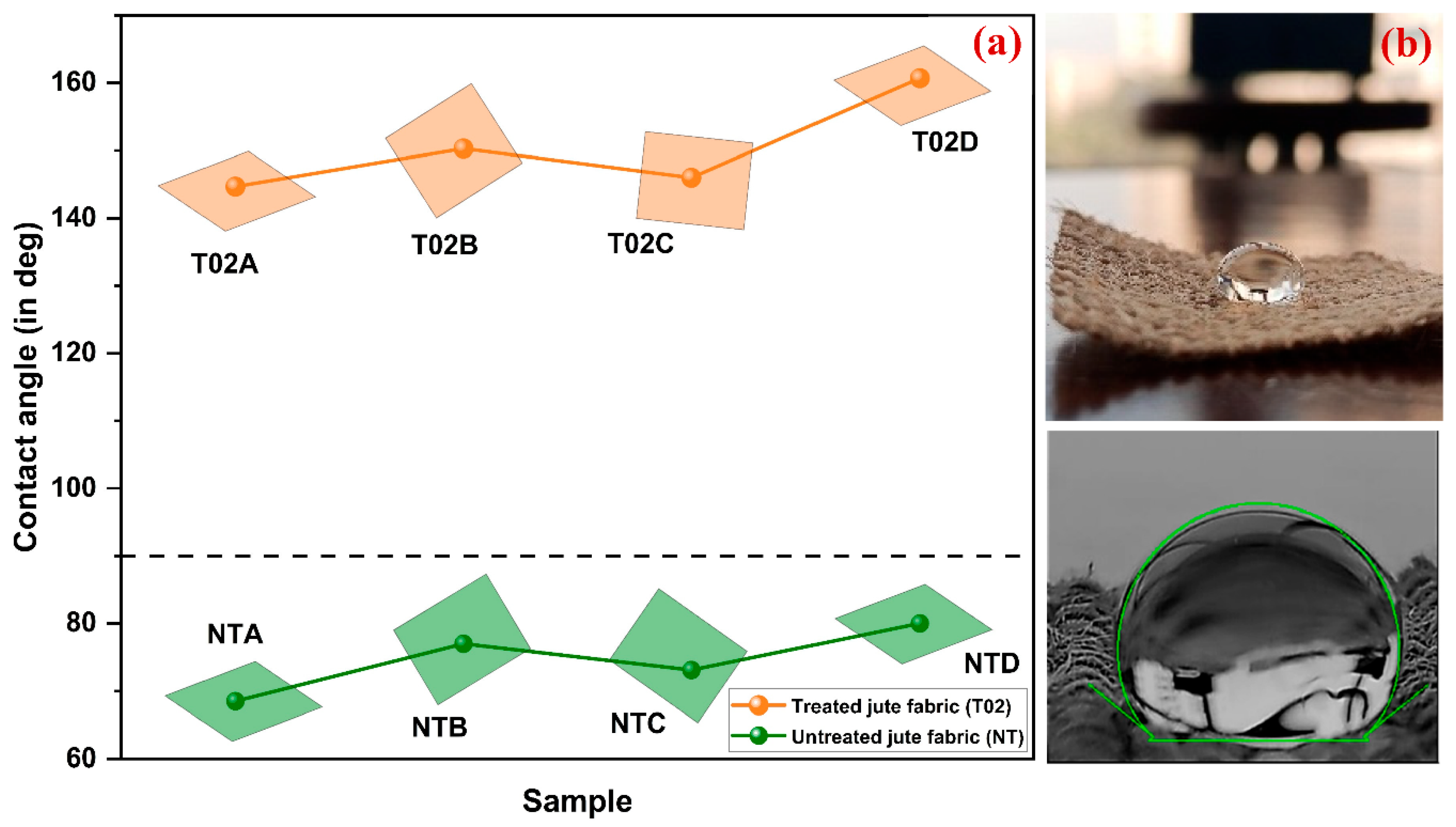
Contact angle test is a quantitative test that is taken to know the wettability of a material surface. It is the angle which is created between a droplet of liquid and a solid surface [
24]. Hydrophobic surface is characterized by a contact angle of more than 90 and Hydrophilic surface is characterized by a contact angle of less than 90 degrees [
25,
26]. This is a direct measure of the energy modification of the surface and is commonly used to measure the performance of water-repellent treatments.
A common pre-test of water resistance of textile materials is the drop test. It is a qualitative method that is easy to use to assess water penetration behavior [
27]. The procedure of this test is to place a drop of water on the fabric material and the time of absorption is recorded. Absorption-resistant and droplet-shaped materials are regarded as water-repellent, and the opposite suggests hydrophilicity [
28,
29].
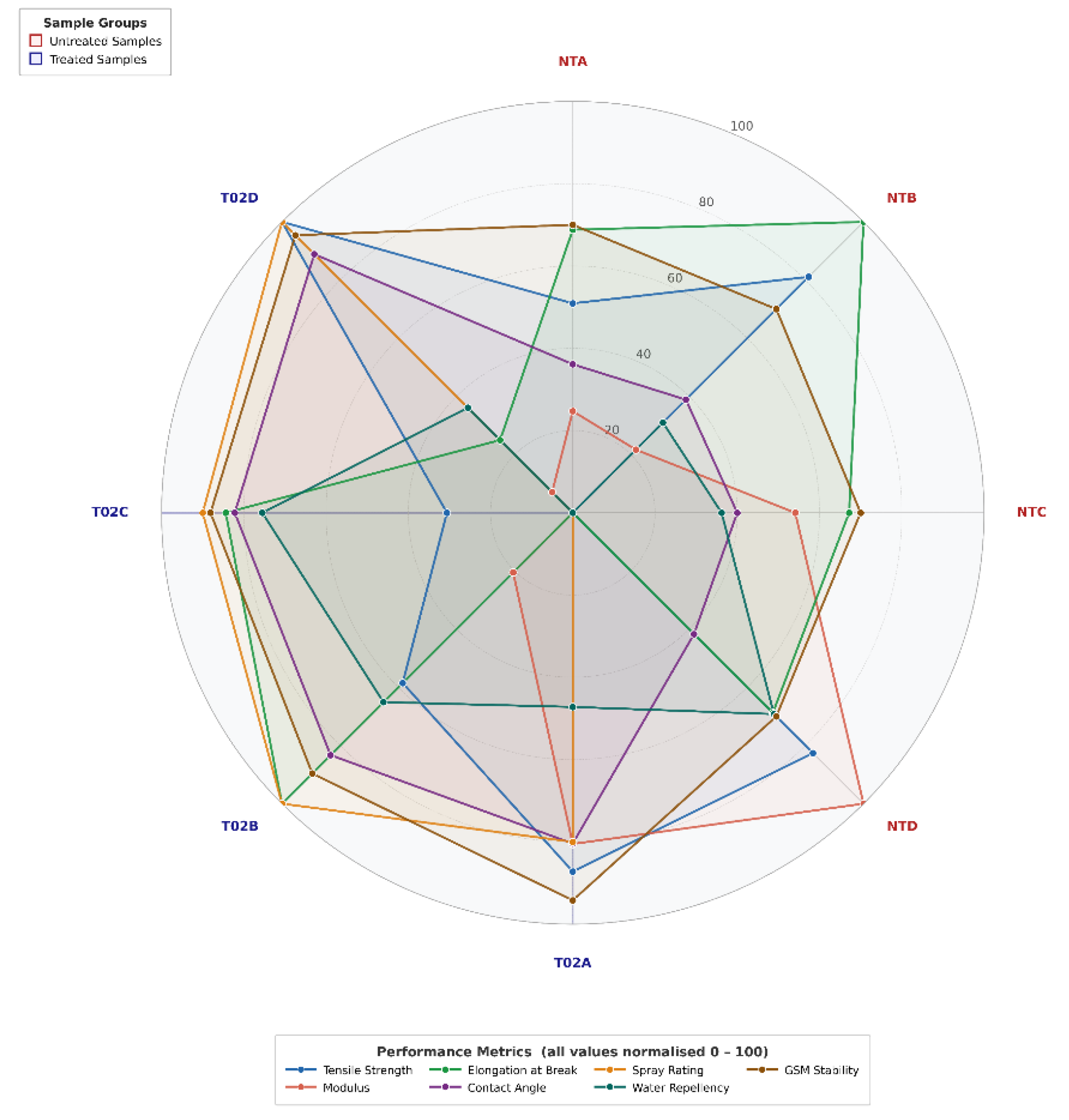
This study presents a thorough analysis of fluorocarbon-based water-repellent jute fabrics, demonstrating enhanced hydrophobicity while preserving mechanical strength and natural texture, highlighting their potential as sustainable alternatives to plastic-based materials.