Submitted:
31 March 2026
Posted:
02 April 2026
You are already at the latest version
Abstract
Keywords:
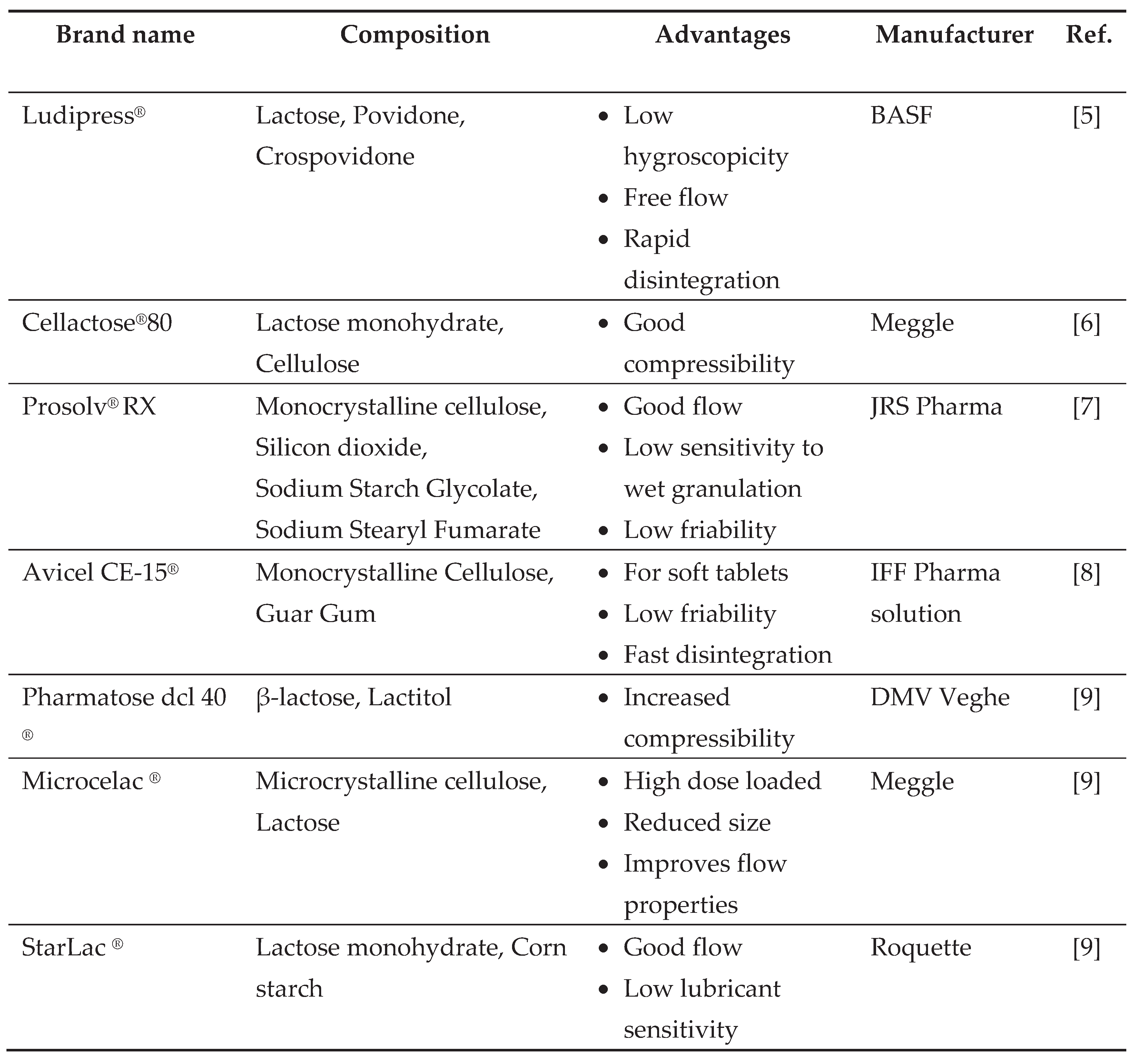
1. Introduction
2. Materials and Methods
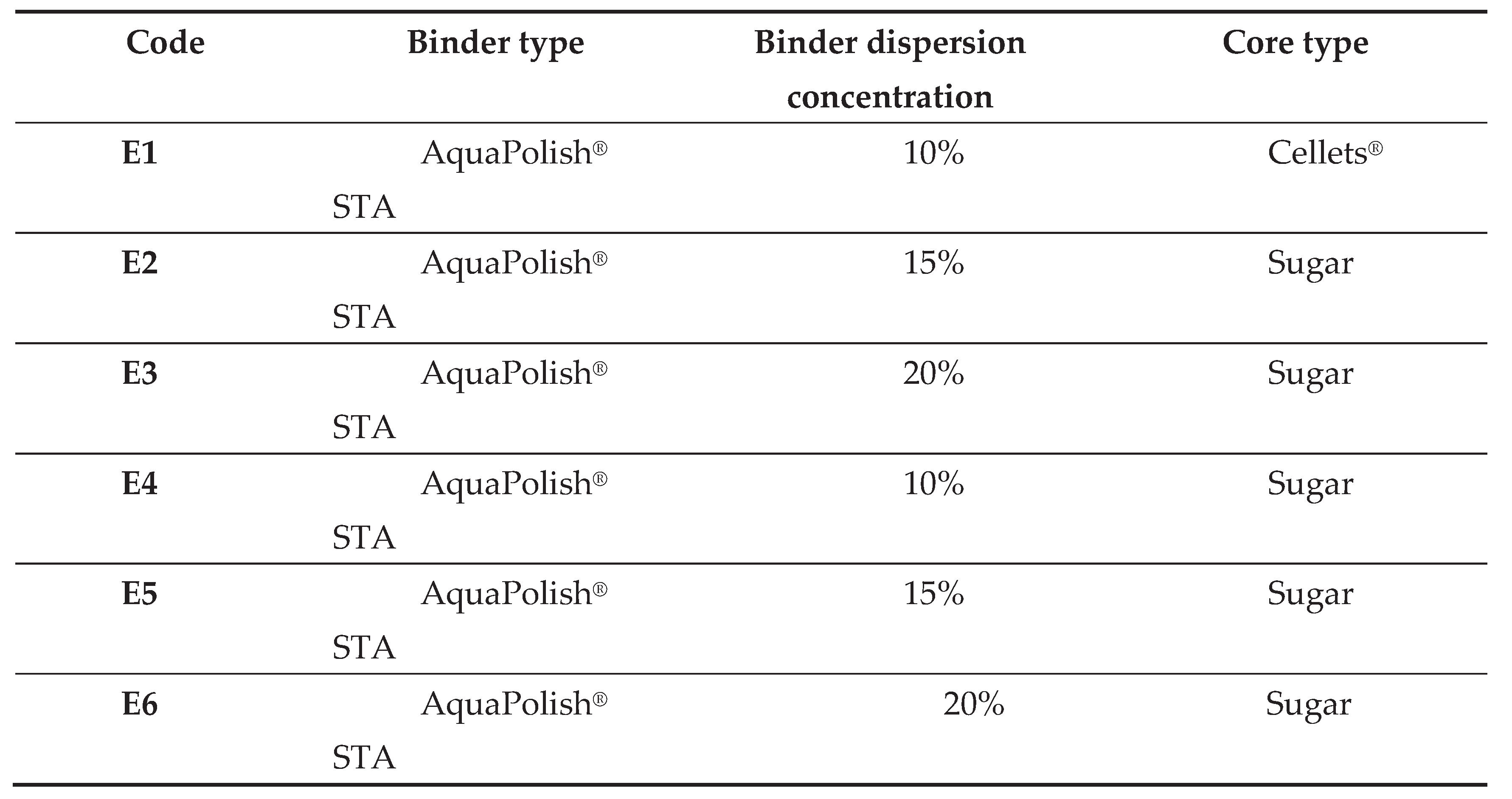
2.1. Granules Manufacturing Steps
2.2. Evaluation of the Granules Obtained by Means of the SeDeM Expert System.
2.2.1. Particle Size
2.2.2. Compressibility
2.2.3. Flow Property Assessment
2.2.4. Lubricity/Dosage Evaluation
2.2.5. Lubricity/Stability Assessment
2.2.6. Parameter Conversion into Radius Values
2.2.7. Acquiring the SeDeM Diagram
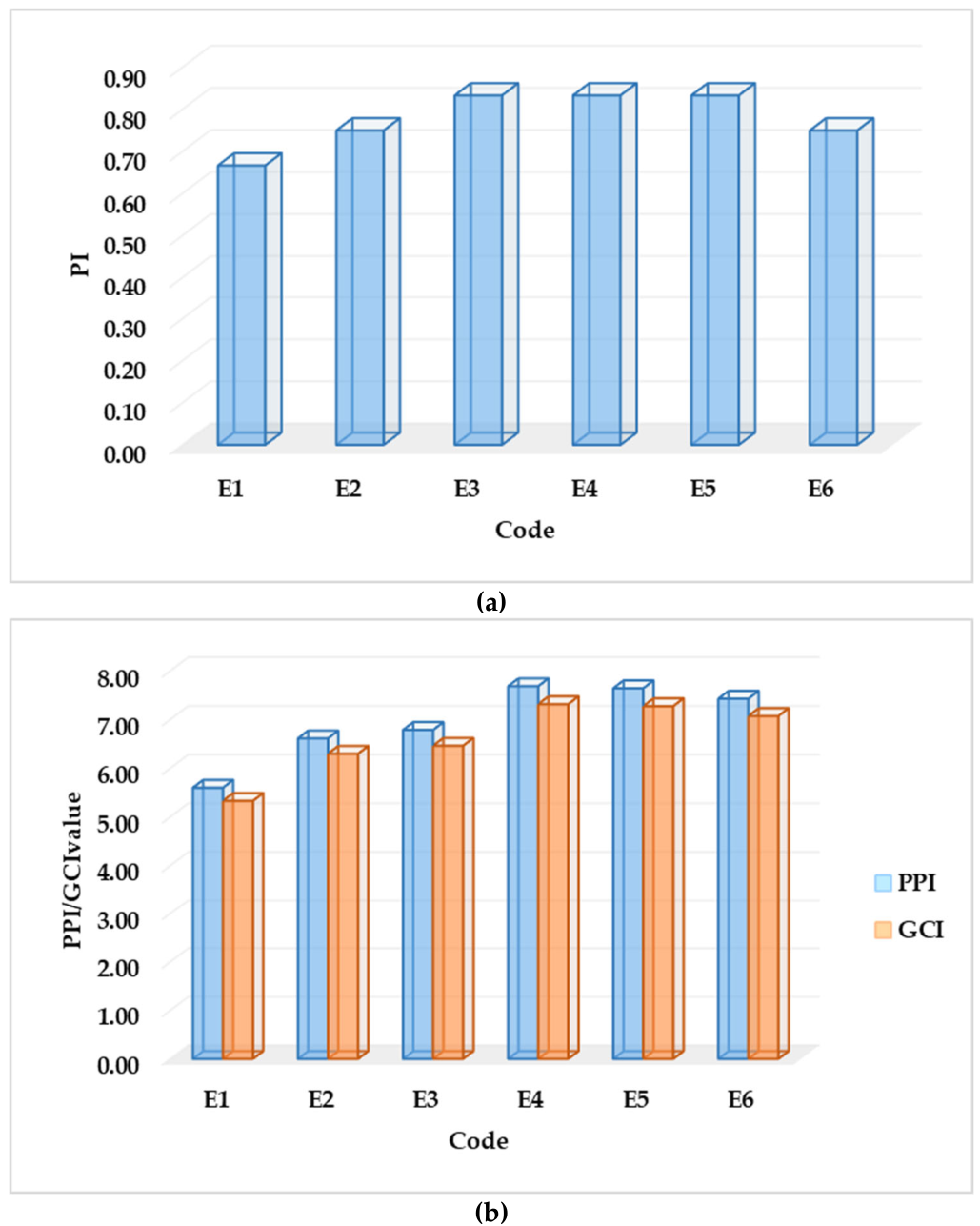
2.2.8. Assessment of the Parameter Index (PI), Parameter Profile Index (PPI) and Good Compressibility Index (GCI)
2.3. Uncoated Tablets Manufacturing Using the Granules Developed
2.4. Uncoated Tablet Quality Assessment
2.4.1. Mechanical Property—Tablet Friability
2.4.2. Mechanical Property—Resistance to Crushing
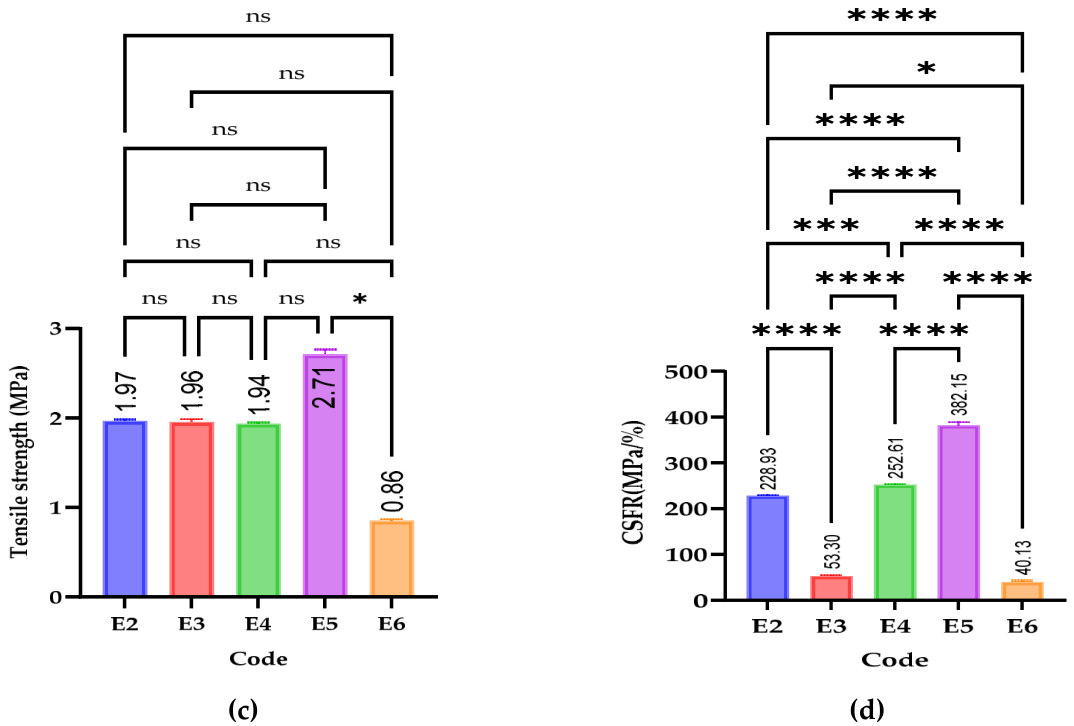
2.4.3. Tensile Strength
2.4.4. Crushing Strength—Friability Ratio
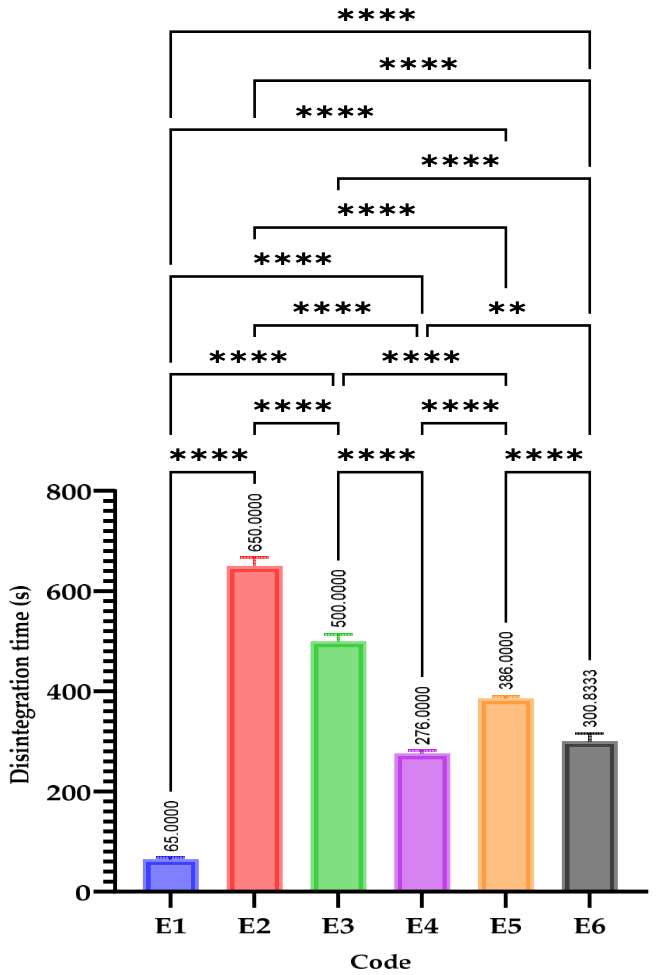
2.4.5. Tablet Disintegration
2.4.6. Determination of Tablet Diameter and Thickness
2.5. Statistical Evaluation
3. Results and Discussions
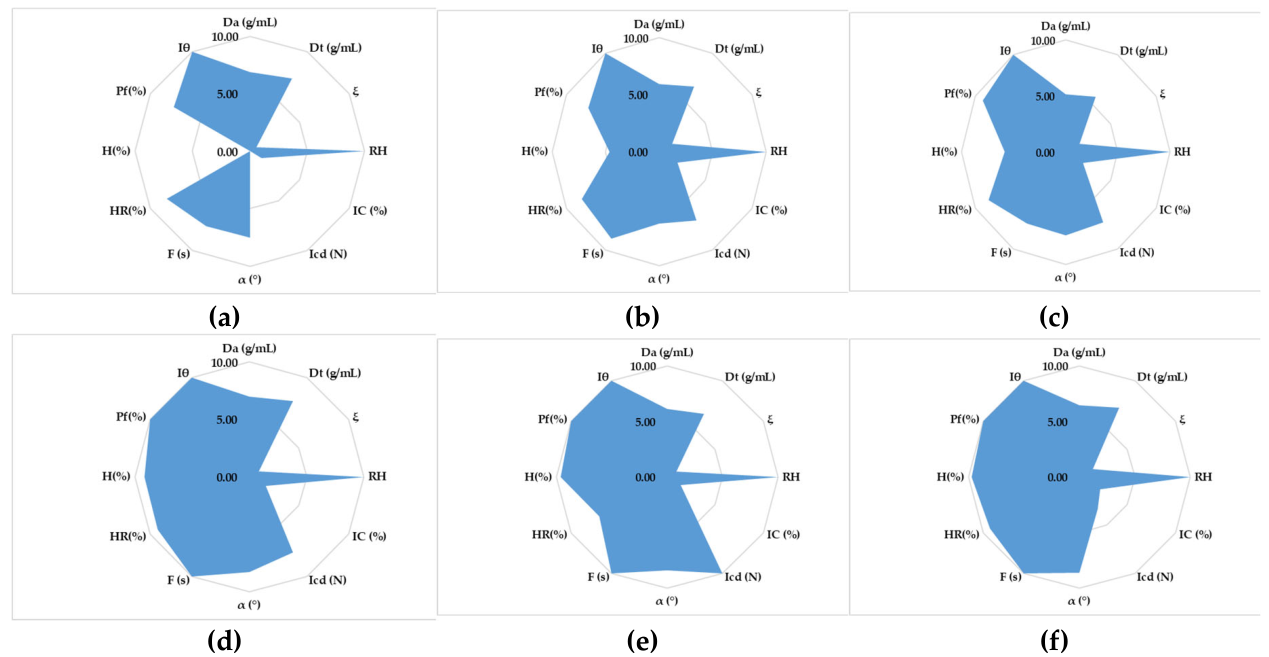
3.1. Evaluation of the Granules by Means of the SeDeM Specific Incidence Factors
3.1.1. Particle Size
3.1.2. Compressibility Incidence Factor
3.1.3. Flow Property Evaluation
3.1.4. Evaluation of Lubricity/Dosage
3.1.5. Lubricity/Stability Assessment
3.2. Mathematical Evaluation of the Proposed Granular Excipients
3.3. Tablets’ Quality Assessment
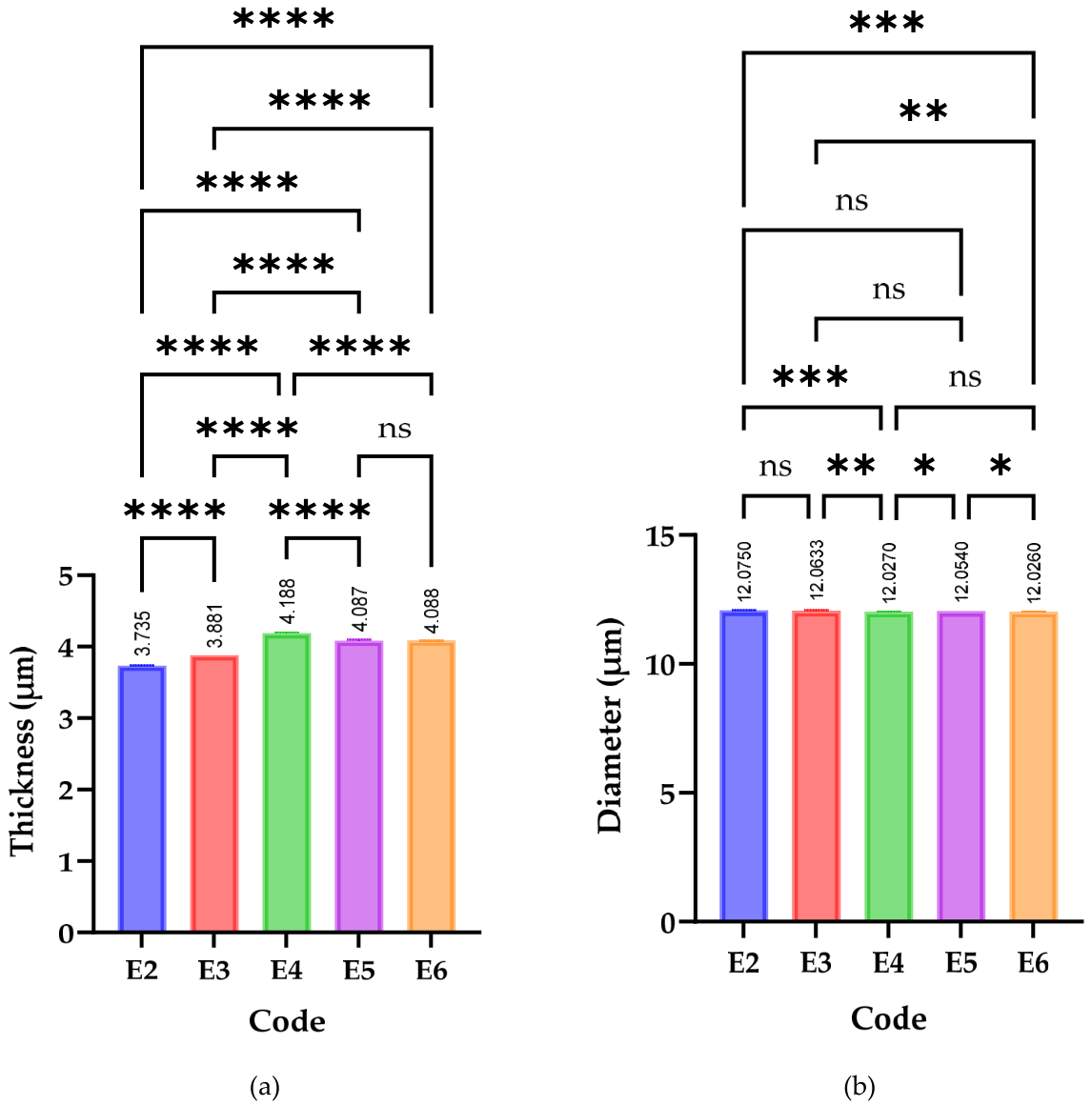
3.3.1. Dimensional Parameters (Thickness and Diameter)
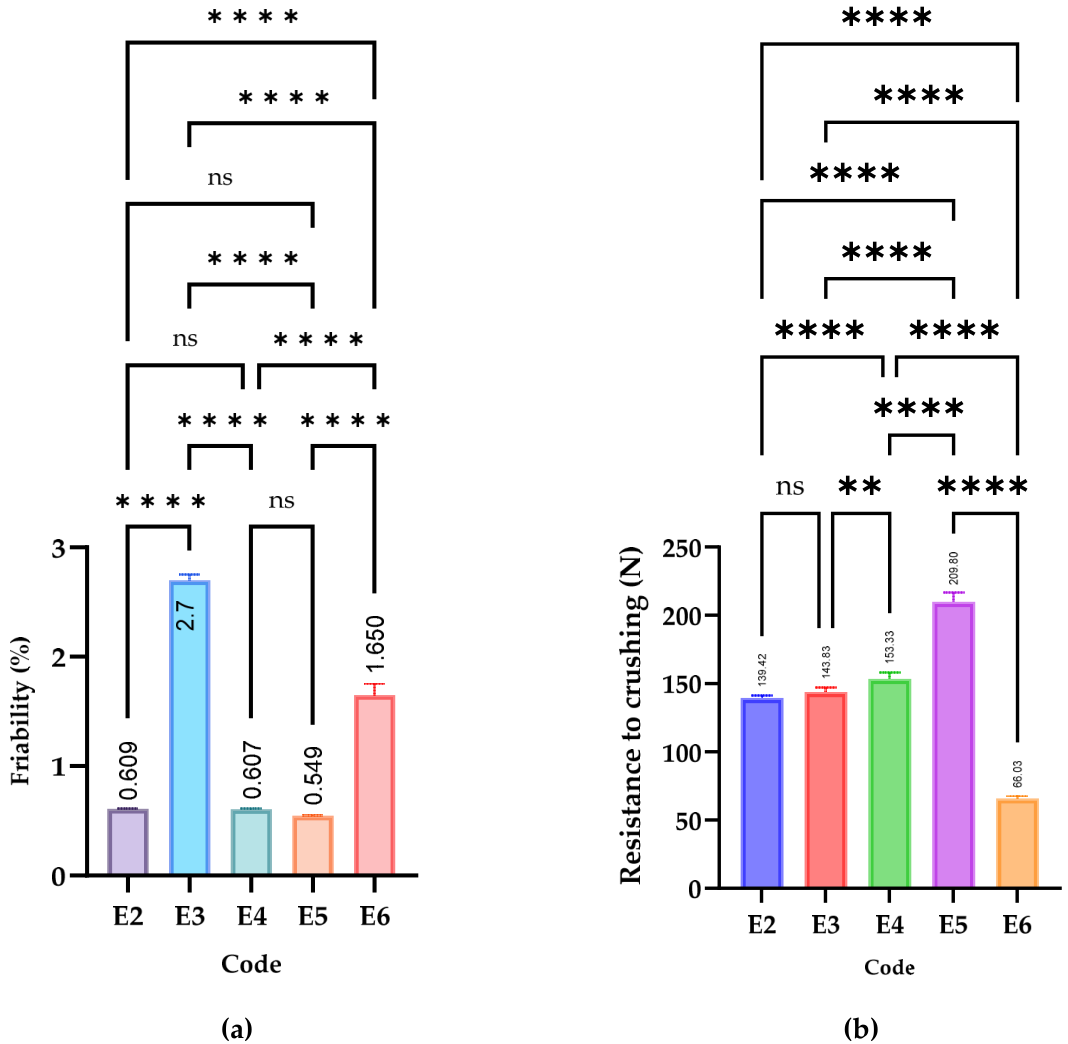
3.3.2. Mechanical Parameters
3.4. Tablet Disintegration
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| IPEC | International Pharmaceutical Excipients Council |
| SeDeM | Sediment Delivery Model |
| API | Active pharmaceutical ingredient |
| Da | Bulk density (g/mL) |
| Dt | Tapped density (g/mL) |
| Ie | Porosity |
| CI | Carr Index |
| Icd | Cohesion Index |
| HR | Hausner ratio |
| α | Angle of repose |
| t’’ | Flowability (parameter) |
| %HR | Loss on drying (%) |
| %H | Hygroscopicity |
| %Pf | Particles < 50 µm |
| Iθ | Homogeneity index |
| Ph. Eur. 12 | European Pharmacopeia 12th Edition |
| v | value range |
| r | Radius value |
| PI | Parameter Index |
| PPI | Parameter Profile Index |
| GCI | Good Compressibility Index |
| f | reliability factor |
| CSFR | Crushing strength/friability ratio |
References
- Qelliny, M. R.; Mustafa, W. W.; Fatease, A. A.; Alamri, A. H.; Alany, R.; Abdelkader, H. Biofunctional Excipients: Their Emerging Role in Overcoming the Inherent Poor Biopharmaceutical Characteristics of Drugs. Pharmaceutics 2025, 17 (5), 598. [CrossRef]
- Pockle, R. D.; Masareddy, R. S.; Patil, A. S.; Patil, P. D. A Comprehensive Review on Pharmaceutical Excipients. Ther. Deliv. 2023, 14 (7), 443–458. [CrossRef]
- Goel, R.; Bhardwaj, S.; Bana, S. Pharmaceutical Excipients. In Dosage Forms, Formulation Developments and Regulations; Elsevier, 2024; pp 311–348. [CrossRef]
- Available online: https://ipec-federation.org/ (accessed 2026-03-04).
- Ludipress®. Available online: https://www.pharmaexcipients.com/product/ludipress/ (accessed 2026-03-04).
- Cellactose® 80. Available online: https://www.meggle-excipients.com/products/cellactose-80 (accessed 2026-03-04).
- PROSOLV® RX. Available online: https://www.jrspharma.com/pharma_en/products/excipients/prosolv-rx.php (accessed 2026-03-04).
- Avicel® CE-15. Available online: https://www.bsce.co.il/product-page/avicel-ce-15 (accessed 2026-03-04).
- Grathwohl, T. Contribution of Particle Design Research to the Development of Patient-Centric Dosage Forms. Pharma Excipients. Available online: https://www.pharmaexcipients.com/news/particle-design-patient-centric/ (accessed 2026-03-04).
- Bano, G.; Dhenge, R. M.; Diab, S.; Goodwin, D. J.; Gorringe, L.; Ahmed, M.; Elkes, R.; Zomer, S. Streamlining the Development of an Industrial Dry Granulation Process for an Immediate Release Tablet with Systems Modelling. Chem. Eng. Res. Des. 2022, 178, 421–437. [CrossRef]
- Ranjan, O. P.; Kumbhar, A. P. Dry and Wet Granulation. In Polymers for Oral Drug Delivery Technologies; Elsevier, 2025; pp 463–494. [CrossRef]
- Monaco, D.; Omar, C.; Reynolds, G. K.; Tajarobi, P.; Litster, J. D.; Salman, A. D. Drying in a Continuous Wet Granulation Line: Investigation of Different End of Drying Control Methods. Powder Technol. 2021, 392, 157–166. [CrossRef]
- Vadaga, A. K.; Gudla, S. S.; Nareboina, G. S. K.; Gubbala, H.; Golla, B. Comprehensive Review on Modern Techniques of Granulation in Pharmaceutical Solid Dosage Forms. Intell. Pharm. 2024, 2 (5), 609–629. [CrossRef]
- Salim, I.; Olowosulu, A. K.; Abdulsamad, A.; Gwarzo, M. S.; Khalid, G. M.; Ahmad, N. T.; Eichie, F. E.; Kurfi, F. S. Application of SeDeM Expert System in the Development of Novel Directly Compressible Co-Processed Excipients via Co-Processing. Future J. Pharm. Sci. 2021, 7 (1), 135. [CrossRef]
- Singh, I.; Thakur, A. K.; Bala, R.; Madan, R. SeDeM Expert System, an Innovative Tool for Developing Directly Compressible Tablets: A Review. Curr. Drug Res. Rev. 2021, 13 (1), 16–24. [CrossRef]
- Castañeda Hernández, O.; Domínguez-Robles, J.; Caraballo, I.; Bernad, M. J.; Melgoza Contreras, L. M. Comparison between Polymeric Excipients Using SeDeM Expert System in Combination with Mathematical Modeling and Quality Control Tools. J. Drug Deliv. Sci. Technol. 2023, 86, 104750. [CrossRef]
- Kotsur, Yu. M.; Flisjuk, E. V. Application of the SeDeM Method for Optimization of Tablet Formulations (A Review). Pharm. Chem. J. 2021, 55 (3), 290–294. [CrossRef]
- Shukla, A. K.; Yadav, V. K.; Verma, M.; Kanaujia, K. A.; Jaiswal, A.; Gupta, V. Expert Systems in Preformulation and Formulation Development with Special Reference to SeDeM System: An Innovative, Problem Solving, Intelligent and Optimization Algorithm Tool. Curr. Indian Sci. 2024, 02, e2210299X338978. [CrossRef]
- Figuera-Figuera, A.; Suñé-Pou, M.; Pérez-Lozano, P.; García-Montoya, E.; Amela-Navarro, J.; Suñé-Negre, J. M. SeDeM as a Tool to Validate Drug Substance Manufacturing Processes and Assess Scalability and Suitability for Direct Compression: Supplier Screening. Pharmaceutics 2023, 15 (8), 2034. [CrossRef]
- Vlad, R.-A.; Antonoaea, P.; Todoran, N.; Muntean, D.-L.; Rédai, E. M.; Silași, O. A.; Tătaru, A.; Bîrsan, M.; Imre, S.; Ciurba, A. Pharmacotechnical and Analytical Preformulation Studies for Cannabidiol Orodispersible Tablets. Saudi Pharm. J. 2021, 29 (9), 1029–1042. [CrossRef]
- Khan, A. Optimization of the Process Variables of Roller Compaction, on the Basis of Granules Characteristics (Flow, Mechanical Strength, and Disintegration Behavior): An Application of SeDeM-ODT Expert System. Drug Dev. Ind. Pharm. 2019, 45 (9), 1537–1546. [CrossRef]
- European Pharmacopoeia—New online-only 12th Edition—European Directorate for the Quality of Medicines & HealthCare—EDQM. European Directorate for the Quality of Medicines & HealthCare. https://www.edqm.eu/en/european-pharmacopoeia-new-online-only-12th-edition (accessed 2026-03-05).
- Gülbağ, S.; Yılmaz Usta, D.; Gültekin, H. E.; Oktay, A. N.; Demirtaş, Ö.; Karaküçük, A.; Çelebi, N. New Perspective to Develop Memantine Orally Disintegrating Tablet Formulations: SeDeM Expert System. Pharm. Dev. Technol. 2018, 23 (5), 512–519. [CrossRef]
- Wan, S.; Yang, R.; Zhang, H.; Li, X.; Gu, M.; Guan, T.; Ren, J.; Sun, H.; Dai, C. Application of the SeDeM Expert System in Studies for Direct Compression Suitability on Mixture of Rhodiola Extract and an Excipient. AAPS PharmSciTech 2019, 20 (3), 105. [CrossRef]
- Hamman, H.; Hamman, J.; Wessels, A.; Scholtz, J.; Steenekamp, J. H. Development of Multiple-Unit Pellet System Tablets by Employing the SeDeM Expert Diagram System I: Pellets with Different Sizes. Pharm. Dev. Technol. 2018, 23 (7), 706–714. [CrossRef]
- Aguilar-Díaz, J. E.; García-Montoya, E.; Pérez-Lozano, P.; Suñé-Negre, J. M.; Miñarro, M.; Ticó, J. R. SeDeM Expert System a New Innovator Tool to Develop Pharmaceutical Forms. Drug Dev. Ind. Pharm. 2014, 40 (2), 222–236. [CrossRef]
- Haleem, R. M.; Salem, M. Y.; Fatahallah, F. A.; Abdelfattah, L. E. Quality in the Pharmaceutical Industry—A Literature Review. Saudi Pharm. J. 2015, 23 (5), 463–469. [CrossRef]
- AlSwayeh, R.; Alvi, S. N.; Hammami, M. M. Quality Assessment of Nine Paracetamol 500 Mg Tablet Brands Marketed in Saudi Arabia. BMC Res. Notes 2021, 14 (1), 254. [CrossRef]
- Newton, J. M. The Calculation of the Tensile Strength of Tablets. J. Pharm. Pharmacol. 1974, 26 (3), 215–216. [CrossRef]
- Halenius, A.; Lakio, S.; Antikainen, O.; Hatara, J.; Yliruusi, J. Fast Tablet Tensile Strength Prediction Based on Non-Invasive Analytics. AAPS PharmSciTech 2014, 15 (3), 781–791. [CrossRef]
- Adedokun, M. O.; Ayorinde, J. O.; Odeniyi, M. A. Compressional, Mechanical and Release Properties of a Novel Gum in Paracetamol Tablet Formulations. Curr. Issues Pharm. Med. Sci. 2014, 27 (3), 187–194. [CrossRef]
- Vlad, R.-A.; Pintea, C.; Chirteș, D.-A.; Antonoaea, P.; Rédai, E. M.; Todoran, N.; Bîrsan, M.; Ciurba, A. The Influence of the Intergranular Superdisintegrant Performance on New Drotaverine Orodispersible Tablet Formulations. Pharmaceutics 2023, 15 (8), 2147. [CrossRef]
- Vlad, R.-A.; Antonoaea, P.; Todoran, N.; Rédai, E.-M.; Bîrsan, M.; Muntean, D.-L.; Imre, S.; Hancu, G.; Farczádi, L.; Ciurba, A. Development and Evaluation of Cannabidiol Orodispersible Tablets Using a 23-Factorial Design. Pharmaceutics 2022, 14 (7), 1467. [CrossRef]
- Suñé-Negre, J. M.; Roig, M.; Fuster, R.; Hernández, C.; Ruhí, R.; García-Montoya, E.; Pérez-Lozano, P.; Miñarro, M.; Ticó, J. R. New Classification of Directly Compressible (DC) Excipients in Function of the SeDeM Diagarm Expert System. Int. J. Pharm. 2014, 470 (1–2), 15–27. [CrossRef]
- Suñé-Negre, J. M.; Roig, M.; Fuster, R.; Hernández, C.; Ruhí, R.; García-Montoya, E.; Pérez-Lozano, P.; Miñarro, M.; Ticó, J. R. New Classification of Directly Compressible (DC) Excipients in Function of the SeDeM Diagarm Expert System. Int. J. Pharm. 2014, 470 (1–2), 15–27. [CrossRef]
- Salish, K.; Maurer, R.; Mao, C. Risks of Powder Flow Obstruction in Hopper and Bin Discharge in Solid Dosage Form Manufacture Should Be Predicted Under The Active Stress State. J. Pharm. Sci. 2024, 113 (3), 688–698. [CrossRef]
- Suñé-Negre, J.; Pérez-Lozano, P.; Miñarro, M.; Roig, M.; Fuster, R.; Hernández, C.; Ruhí, R.; García-Montoya, E.; Ticó, J. Application of the SeDeM Diagram and a New Mathematical Equation in the Design of Direct Compression Tablet Formulation. Eur. J. Pharm. Biopharm. 2008, 69 (3), 1029–1039. [CrossRef]
- Saurí, J.; Millán, D.; Suñé-Negre, J. M.; Pérez-Lozano, P.; Sarrate, R.; Fàbregas, A.; Carrillo, C.; Miñarro, M.; Ticó, J. R.; García-Montoya, E. The Use of the SeDeM Diagram Expert System for the Formulation of Captopril SR Matrix Tablets by Direct Compression. Int. J. Pharm. 2014, 461 (1–2), 38–45. [CrossRef]
- Sun, C. C. Mechanism of Moisture Induced Variations in True Density and Compaction Properties of Microcrystalline Cellulose. Int. J. Pharm. 2008, 346 (1–2), 93–101. [CrossRef]
- Mahours, G. M.; Shaaban, D. E. Z.; Shazly, G. A.; Auda, S. H. The Effect of Binder Concentration and Dry Mixing Time on Granules, Tablet Characteristics and Content Uniformity of Low Dose Drug in High Shear Wet Granulation. J. Drug Deliv. Sci. Technol. 2017, 39, 192–199. [CrossRef]
- Köster, C.; Kleinebudde, P. Evaluation of Binders in Twin-Screw Wet Granulation—Optimization of Tabletability. Int. J. Pharm. 2024, 659, 124290. [CrossRef]
- (Brniak, W.; Jachowicz, R.; Pelka, P. The Practical Approach to the Evaluation of Methods Used to Determine the Disintegration Time of Orally Disintegrating Tablets (ODTs). Saudi Pharm. J. 2015, 23 (4), 437–443. [CrossRef]
 |
| Parameter | Wet Granulation | Dry Granulation | Ref. |
| Basic principle | Uses a binder to agglomerate powder particles | Uses compaction | [10,11,12,13] |
| Binder requirement | Requires liquid binder solution | No liquid binder (may use dry binder) | |
| Moisture involvement | Yes | No | |
| Heat exposure | Requires drying | No drying step required | |
| Suitable for heat-sensitive drugs | Limited suitability | Highly suitable | |
| Suitable for moisture-sensitive drugs | Not suitable | Suitable | |
| Equipment | High-shear granulator, fluid bed granulator, dryer | Roller compactor or slugging press | |
| Process steps | Mixing → Wet massing → Granulation → Drying → Milling | Mixing → Compaction (slugging/roller) → Milling | |
| Cost | Higher (more steps, energy for drying) | Lower (fewer steps) | |
| Time consumption | Longer | Shorter | |
| Granule properties | Generally, more uniform and stronger | Less dense, may show more fines | |
| Flow properties | Usually improved significantly | Improved, but sometimes less than wet | |
| Tablet hardness | Typically higher | May be lower depending on compaction | |
| Scale-up complexity | More complex | Relatively simpler | |
| Industrial preference | Widely used for difficult formulations | Preferred for moisture/heat-sensitive APIs |
 |
| Excipient | Role | Amount (g) (w/w%) | ||||||
| E1 | E2 | E3 | E4 | E5 | E6 | |||
| Microcrystalline cellulose | Filler | 93 | 93 | 93 | - | - | - | |
| Lactose | Filler | - | - | - | 94 | 94 | 94 | |
| Pruv® | Lubricant | 1 | 1 | 1 | 1 | 1 | 1 | |
| Sodium alginate | Disintegrant | 1 | 1 | 1 | - | - | - | |
| Sorbitol | Sweetener | 5 | 5 | 5 | 5 | 5 | 5 | |
| Dispersion of AquaPolish®STA | Binder | quantity sufficient (q.s.) | q.s. | q.s. | q.s. | q.s. | q.s. | |
| Final mixture mass (g) | 100 | 100 | 100 | 100 | 100 | 100 | ||
| Parameters | Value Range (v) | Radius range (r) | Factors Applied to Limit Value |
| Da | 0–1 g/mL | 0–10 | 10 × v |
| Dt | 0–1 g/mL | 0–10 | 10 × v |
| ξ | 0–1.2 | 0–10 | (10 × v) / 1.2 |
| CI | 0–50% | 0–10 | v / 5 |
| Icd | 0–200 N | 0–10 | v / 20 |
| HR | 3–1 | 0–10 | (30—10 × v) / 2 |
| α | 50–0° | 0–10 | 10—(v / 5) |
| t” | 20–0 | 0–10 | 10—(v / 2) |
| %RH | 0–10% | 0–10 | 10—v |
| %H | 20–0% | 0–10 | 10—(v / 2) |
| Pf | 50–0% | 0–10 | 10—(v / 5) |
| Iθ | 0–0.02 | 0–10 | 500 × v |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




