Submitted:
01 April 2026
Posted:
02 April 2026
You are already at the latest version
Abstract
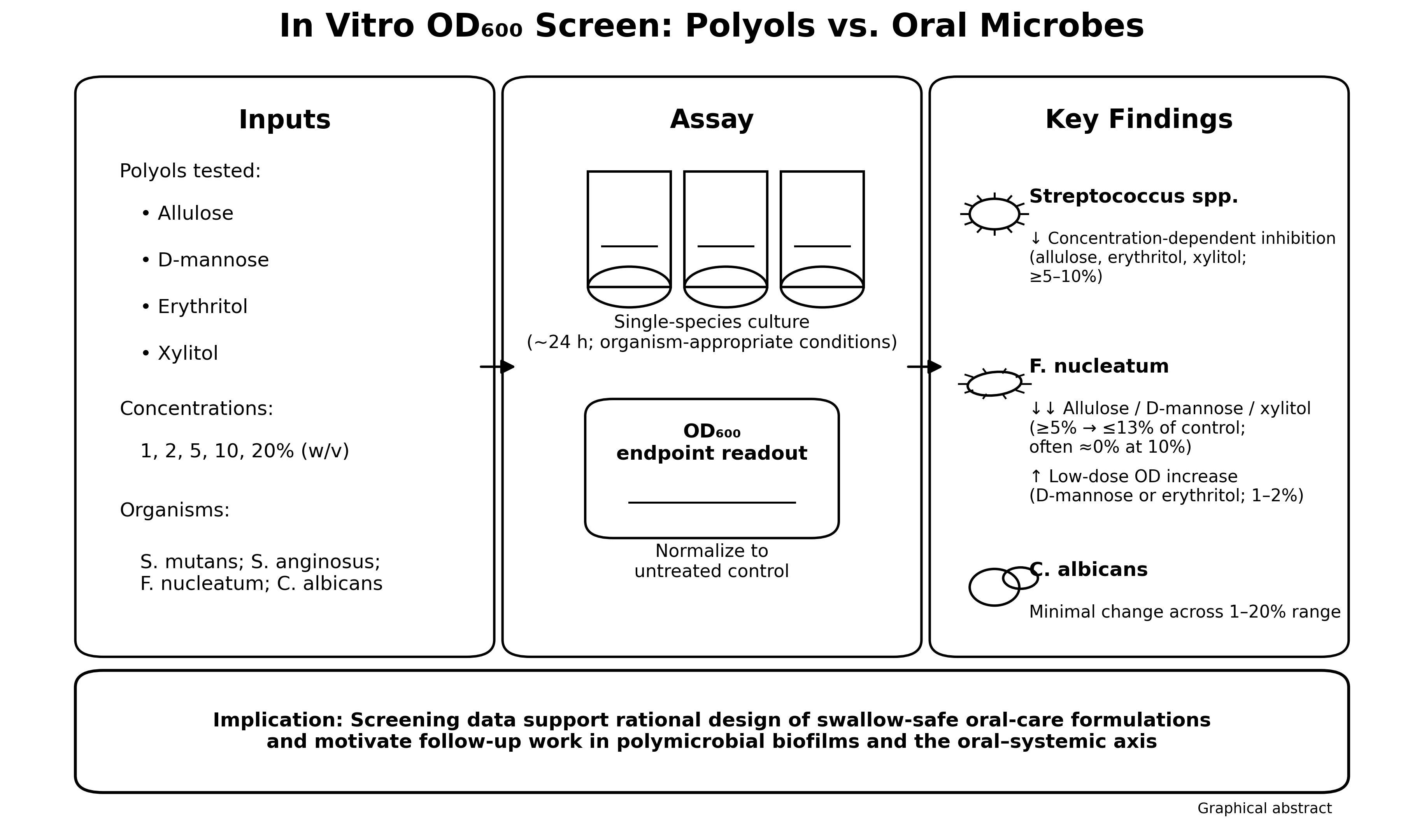
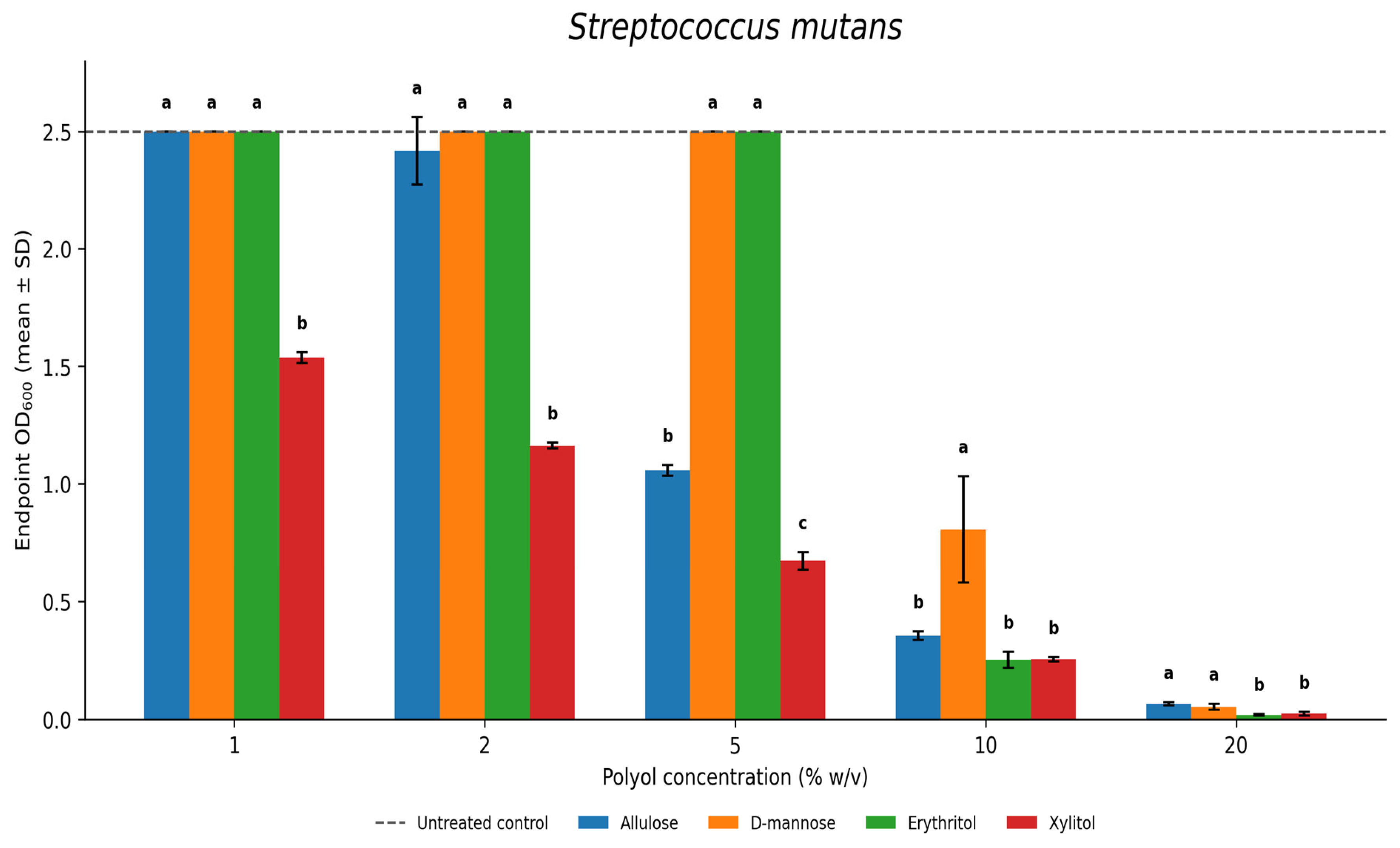
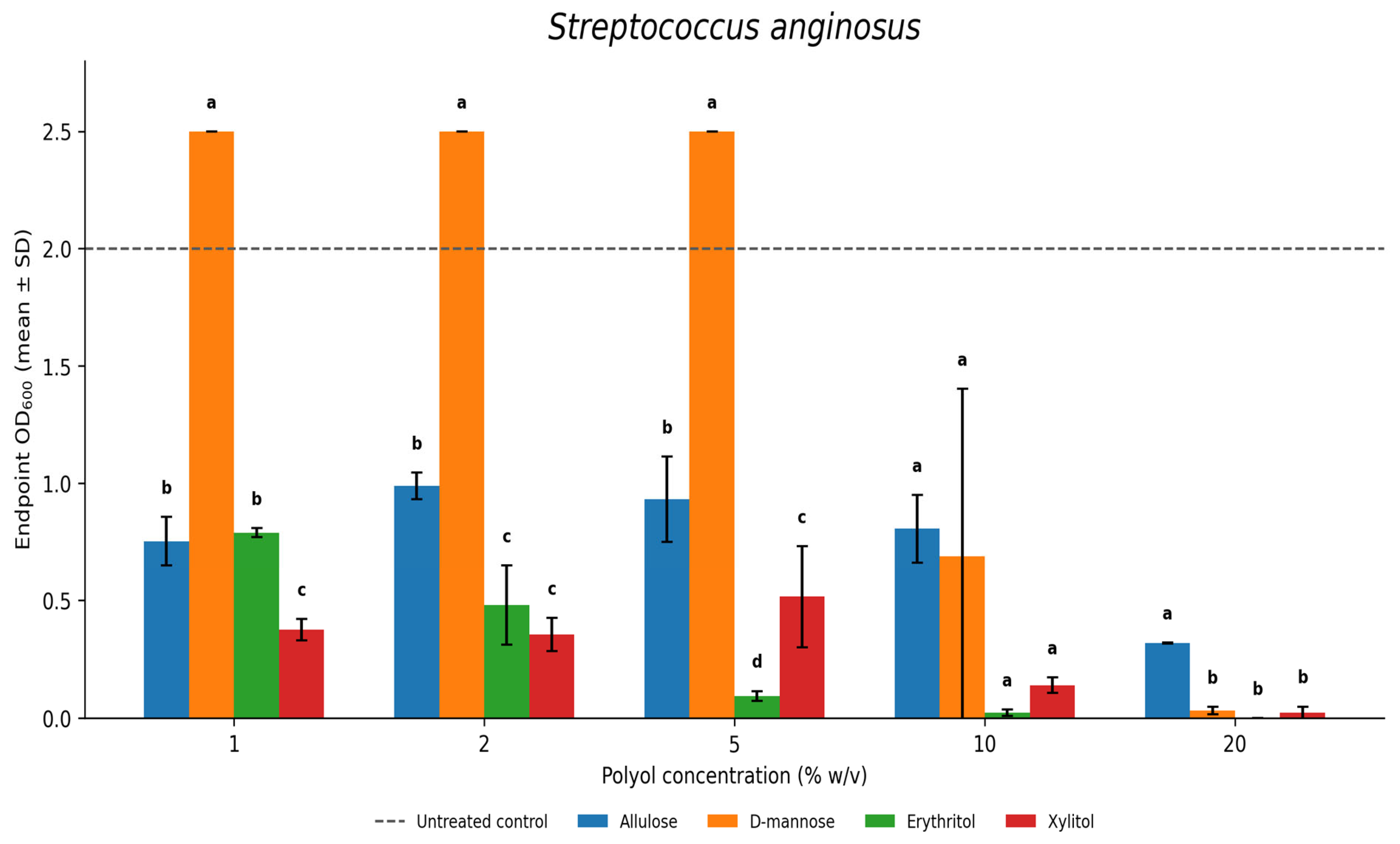
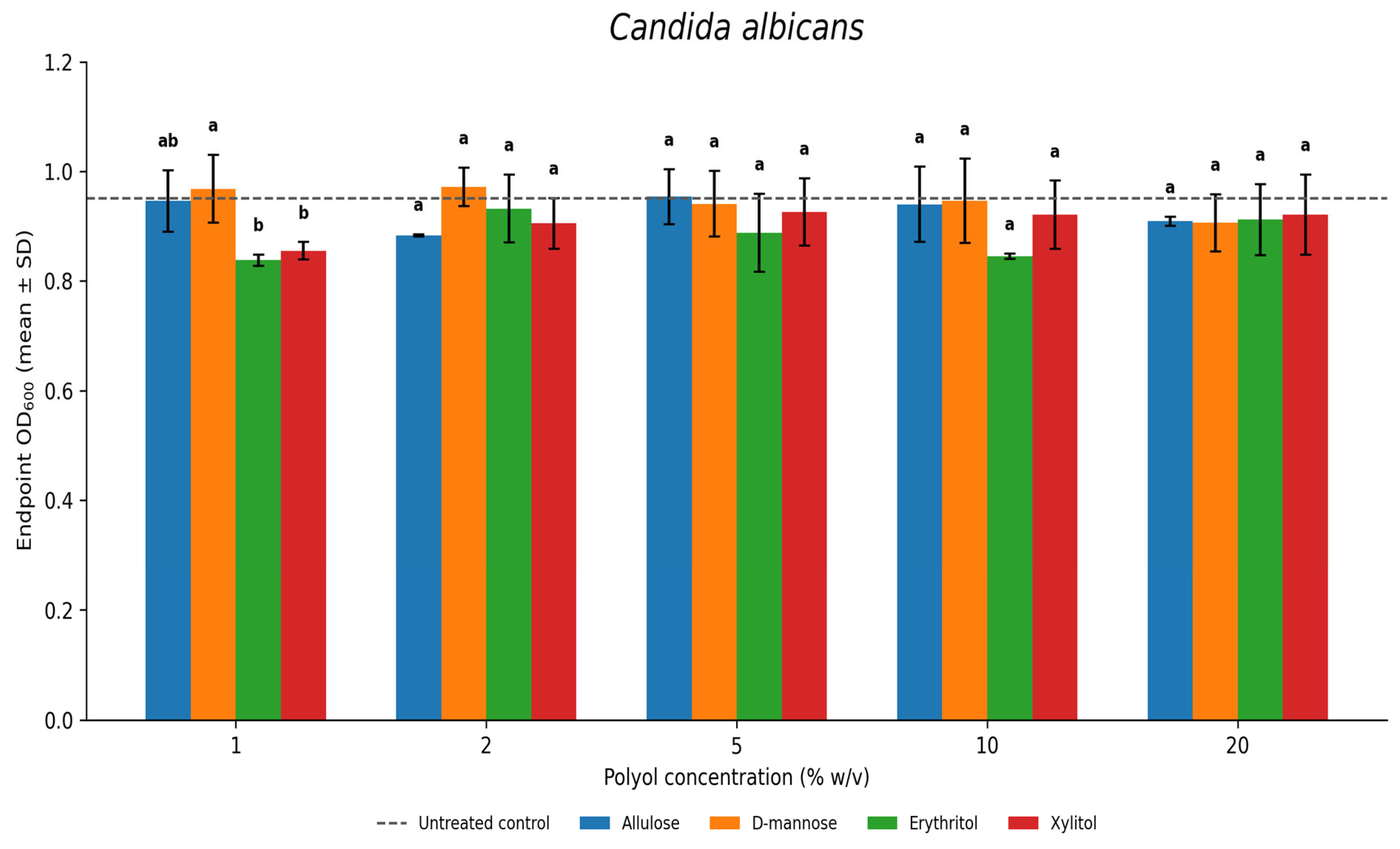
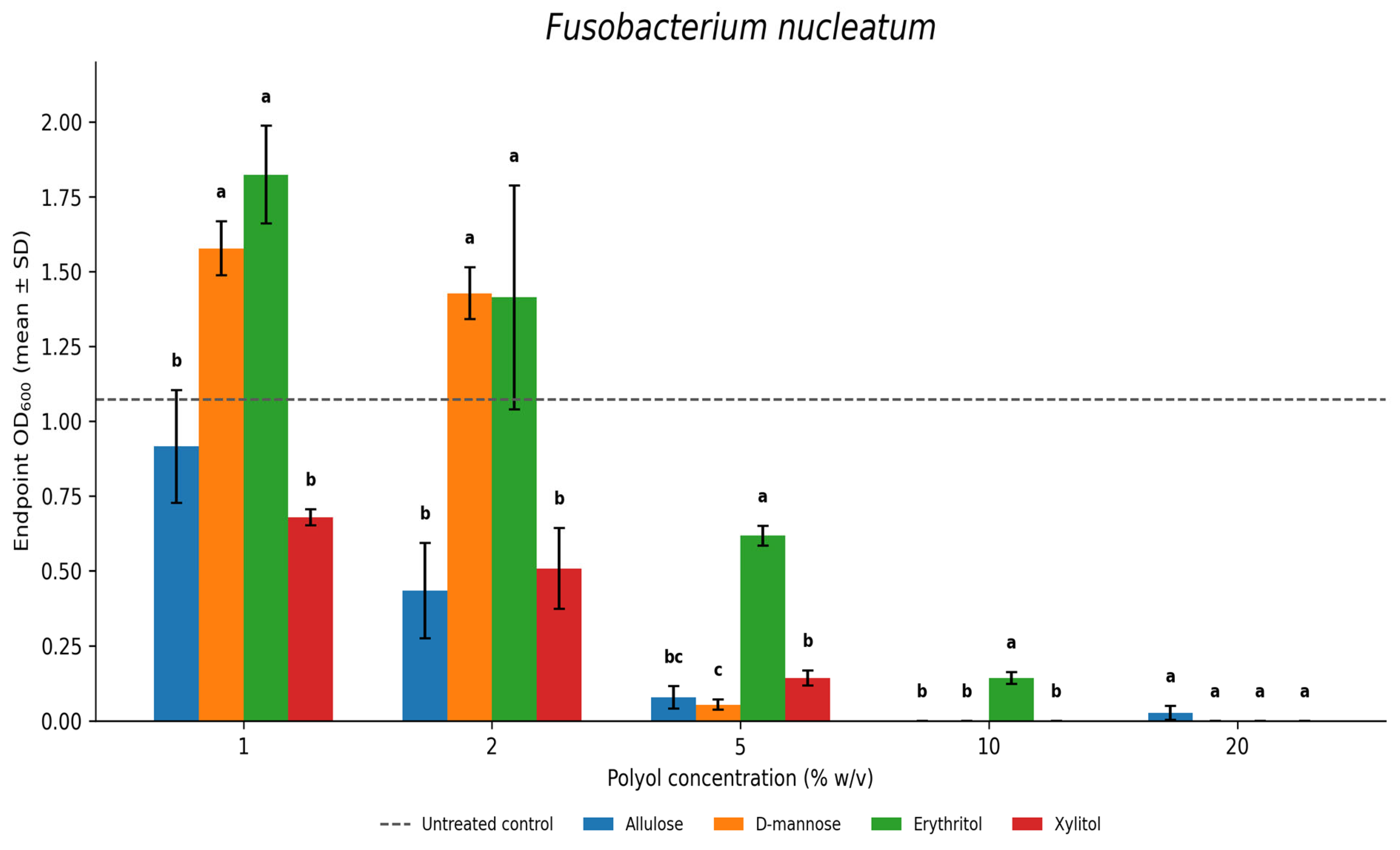
Polyols are widely used as non-cariogenic sweeteners in foods and oral care products, yet their comparative activity against diverse oral microbes and their potential relevance to the oral–systemic axis remain incompletely defined. Here, we performed an in vitro, optical-density (OD)-based screening of four polyols—allulose, D-mannose, erythritol, and xylitol—against Streptococcus mutans, Streptococcus anginosus, Candida albicans, and Fusobacterium nucleatum. Cultures were grown with polyols at 1–20% (w/v), and OD600 was recorded at organism-specific endpoints (~24 h). Allulose, erythritol, and xylitol produced strong, concentration-dependent suppression of streptococcal growth at ≥5–10%, whereas C. albicans showed minimal changes across the tested range. F. nucleatum was highly sensitive to allulose, D-mannose, and xylitol at ≥5% (reducing OD to ≤13% of untreated control), while low concentrations of D-mannose and erythritol increased OD above control, suggesting species-specific utilization or stress responses. One-way ANOVA with Tukey’s HSD post hoc testing supported significant between-polyol differences for most concentrations in Streptococcus spp. and F. nucleatum. Collectively, these results identify polyol- and taxon-specific growth phenotypes that can inform the formulation of swallow-safe oral hygiene products and motivate follow-up work in polymicrobial biofilm models and clinical studies targeting oral inflammation and downstream systemic risk.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Microorganisms and Culture Conditions
2.2. Polyols and Preparation
2.3. Optical Density Growth Assay
2.4. Data Processing and Normalization
2.5. Statistical Analysis
3. Results
3.1. Streptococcus mutans
3.2. Streptococcus anginosus
3.3. Candida albicans
3.4. Fusobacterium nucleatum
4. Discussion
4.1. Summary of Main Findings
4.2. Relationship to Prior Literature on Xylitol and Erythritol
4.3. Emerging Evidence for Allulose and D-Mannose in Oral Microbial Modulation
4.4. Implications for Oral Biofilms and Formulation Design
4.5. Strengthening the Oral–Systemic Discussion: Mechanistic Pathways and Organism-Specific Relevance
4.6. Safety Considerations for Swallow-Safe Oral Products
4.7. Limitations
4.8. Future Directions
| Systemic condition / pathway | Example oral microbial connection (selected examples) |
| Cardiometabolic inflammation and atherosclerotic risk | Periodontal inflammation can increase systemic inflammatory mediators and endotoxemia; episodic bacteremia and oral–gut axis are proposed routes [20,21,22,23,24,25]. |
| Infective endocarditis | Oral streptococci are common etiologic agents; routine activities can seed transient bacteremia [27]. |
| Cerebrovascular disease | Cnm+ S. mutans strains have been associated with cerebrovascular pathology, including cerebral microbleeds [26]. |
| Diabetes and metabolic dysregulation | Bidirectional links between periodontal inflammation and glycemic control have been proposed; oral–gut axis and systemic cytokines are implicated [20,21,22,23,24,25]. |
| Adverse pregnancy outcomes | Periodontitis and oral dysbiosis have been associated with adverse outcomes in observational and meta-analytic studies [36]. |
| Neurodegeneration | Oral pathogens and periodontal inflammation have been investigated in relation to Alzheimer’s disease; microbial and inflammatory mechanisms are discussed in translational work [35,36]. |
| Non-alcoholic fatty liver disease | Oral dysbiosis may influence the gut–liver axis through swallowed microbes and immune modulation [38]. |
| Gastrointestinal disease and malignancy-associated microbiomes | F. nucleatum has been discussed in gastrointestinal disease contexts and cancer-associated microbiomes, motivating interest in oral reservoirs [32,33]. |
| Opportunistic invasive infections | The Streptococcus anginosus group can participate in deep-seated abscess formation following translocation [27]. |
| Peri-implant and periodontal tissue destruction | F. nucleatum contributes to periodontal/peri-implant biofilms and can modulate host immune responses [29,30]. |
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Centers for Disease Control and Prevention. 2024 Oral Health Surveillance Report: Selected Findings. Available online: https://www.cdc.gov/oral-health/php/2024-oral-health-surveillance-report/ (accessed on 11 February 2026).
- Eke, P.I.; Dye, B.A.; Wei, L.; Thornton-Evans, G.O.; Genco, R.J. Prevalence of Periodontitis in Adults in the United States: 2009 and 2010. J. Dent. Res. 2012, 91, 914–920. [CrossRef]
- Eke, P.I.; Wei, L.; Borgnakke, W.S.; Thornton-Evans, G.; Zhang, X.; Lu, H.; McGuire, L.C.; Genco, R.J. Periodontitis Prevalence in Adults ≥65 Years of Age, in the USA. Periodontol. 2000 2016, 72, 76–95. [CrossRef]
- Milgrom, P.; Ly, K.A.; Rothen, M. Xylitol and Its Vehicles for Public Health Needs. Adv. Dent. Res. 2009, 21, 44–47. [CrossRef]
- Burt, B.A. The Use of Sorbitol- and Xylitol-Sweetened Chewing Gum in Caries Control. J. Am. Dent. Assoc. 2006, 137, 190–196. [CrossRef]
- Janakiram, C., Deepan Kumar, C. V., & Joseph, J. (2017). Xylitol in preventing dental caries: A systematic review and meta-analyses. Journal of natural science, biology, and medicine, 8(1), 16–21. [CrossRef]
- Knuuttila, M.L.E.; Mäkinen, K.K. Effect of Xylitol on the Growth and Metabolism of Streptococcus mutans. Caries Res. 1975, 9, 177–189. [CrossRef]
- Söderling, E.M. Xylitol, Mutans Streptococci, and Dental Plaque. Adv. Dent. Res. 2009, 21, 74–78. [CrossRef]
- Loimaranta, V.; Mazurel, D.; Deng, D.; Söderling, E. Xylitol and Erythritol Inhibit Real-Time Biofilm Formation of Streptococcus mutans. BMC Microbiol. 2020, 20, 184. [CrossRef]
- Bradshaw, D. J., & Marsh, P. D. (1994). Effect of sugar alcohols on the composition and metabolism of a mixed culture of oral bacteria grown in a chemostat. Caries research, 28(4), 251–256. [CrossRef]
- Mäkinen, K.K.; Isotupa, K.P.; Kivilompolo, T.; Mäkinen, P.L.; Toivanen, J.; Söderling, E. Comparison of Erythritol and Xylitol Saliva Stimulants in the Control of Dental Plaque and Mutans Streptococci. Caries Res. 2001, 35, 129–135. [CrossRef]
- Mäkinen, K.K.; Saag, M.; Isotupa, K.P.; Olak, J.; Nõmmela, R.; Söderling, E.; Mäkinen, P.L. Similarity of the Effects of Erythritol and Xylitol on Some Risk Factors of Dental Caries. Caries Res. 2005, 39, 207–215. [CrossRef]
- Kõljalg, S.; Smidt, I.; Chakrabarti, A.; Bosscher, D.; Mändar, R. Exploration of Singular and Synergistic Effect of Xylitol and Erythritol on Causative Agents of Dental Caries. Sci. Rep. 2020, 10, 6297. [CrossRef]
- Salli, K.; Lehtinen, M.J.; Tiihonen, K.; Ouwehand, A.C. Xylitol’s Health Benefits beyond Dental Health: A Comprehensive Review. Nutrients 2019, 11, 1813. [CrossRef]
- Mäkinen, K. K., & Söderling, E. (1980). A quantitative study of mannitol, sorbitol, xylitol, and xylose in wild berries and commercial fruits. Journal of Food Science, 45(2), 367–371, 374. doi.abs/10.1111/j.1365-2621.1980.tb02616.x.
- Han, S.; Rajitha, K.; Park, S.; Lim, J.; Jung, H.-Y.; Kim, J.; Kim, D. Unveiling the Impact of Allulose on Oral Microbiota and Biofilm Formation via a Cariogenic Potential Assessment Platform. Front. Cell. Infect. Microbiol. 2025, 15, 1670139. [CrossRef]
- Ruby, J.D.; Momeni, S.S.; Wu, H. The Effect of Allulose, Sucralose, and Xylitol on Streptococcus mutans Acid Production. JADA Foundat. Sci. 2025, 4, 100052. [CrossRef]
- Kranjčec, B.; Papeš, D.; Altarac, S. D-Mannose Powder for Prophylaxis of Recurrent Urinary Tract Infections in Women: A Randomized Clinical Trial. World J. Urol. 2014, 32, 79–84. [CrossRef]
- Hayward, G.; Mort, S.; Hay, A.D.; et al. d-Mannose for Prevention of Recurrent Urinary Tract Infection among Women: A Randomized Clinical Trial. JAMA Intern. Med. 2024, 184, 619–628. [CrossRef]
- Rajasekaran, J.J.; Krishnamurthy, H.K.; Bosco, J.; Jayaraman, V.; Krishna, K.; Wang, T.; Bei, K. Oral Microbiome: A Review of Its Impact on Oral and Systemic Health. Microorganisms 2024, 12, 1797. [CrossRef]
- Kim, M.-Y.; Pang, E.-K. Relationship between Periodontitis and Systemic Health Conditions: A Narrative Review. Ewha Med. J. 2025, 48, e27. [CrossRef]
- Xi, M.; Ruan, Q.; Zhong, S.; Li, J.; Qi, W.; Xie, C.; Wang, X.; Abuduxiku, N.; Ni, J. Periodontal Bacteria Influence Systemic Diseases through the Gut Microbiota. Front. Cell. Infect. Microbiol. 2024, 14, 1478362. [CrossRef]
- Jiménez De Nunzio, S.; Portal-Núñez, S.; Arias Macías, C.M.; et al. Does a Dysbiotic Oral Microbiome Trigger the Risk of Chronic Inflammatory Disease? Curr. Treat. Options Allergy 2023, 10, 364–383. [CrossRef]
- Sahni, V.; Shankar, A. Oral Health and Its Expanding Role in Systemic Disease, Cancer Outcomes, and Public Health. Lancet Reg. Health—Southeast Asia 2025, 100627. [CrossRef]
- Murray, P. E., Coffman, J. A., & Garcia-Godoy, F. (2024). Oral Pathogens’ Substantial Burden on Cancer, Cardiovascular Diseases, Alzheimer’s, Diabetes, and Other Systemic Diseases: A Public Health Crisis-A Comprehensive Review. Pathogens (Basel, Switzerland), 13(12), 1084. [CrossRef]
- Hosoki, S.; Saito, S.; Tonomura, S.; et al. Oral Carriage of Streptococcus mutans Harboring the cnm Gene Relates to an Increased Incidence of Cerebral Microbleeds. Stroke 2021, 52, 373–381. [CrossRef]
- Seo, H.; Hyun, J.; Kim, H.; et al. Risk and Outcome of Infective Endocarditis in Streptococcal Bloodstream Infections according to Streptococcal Species. Microbiol. Spectr. 2023, 11, e01049-23. [CrossRef]
- Pilarczyk-Zurek, M.; Sitkiewicz, I.; Koziel, J. The Clinical View on Streptococcus anginosus Group—Opportunistic Pathogens Coming Out of Hiding. Front. Microbiol. 2022, 13, 956677. [CrossRef]
- Signat, B.; Roques, C.; Poulet, P.; Duffaut, D. Role of Fusobacterium nucleatum in Periodontal Health and Disease. Curr. Issues Mol. Biol. 2011, 13, 25–36. [CrossRef]
- Chen, Y.; Shi, T.; Li, Y.; Huang, L.; Yin, D. Fusobacterium nucleatum: The Opportunistic Pathogen of Periodontal and Peri-Implant Diseases. Front. Microbiol. 2022, 13, 860149. [CrossRef]
- Abreu, M.T.; Peek, R.M., Jr. Gastrointestinal Malignancy and the Microbiome. Gastroenterology 2014, 146, 1534–1546.e3. [CrossRef]
- Zhou J, Ma Q, Liang J, Pan Y, Chen Y, Yu S, Liu Y, Zhang Q, Li Y, and Zou J (2025). smu_1558c-mediated regulation of growth and biofilm formation in Streptococcus mutans. Front. Microbiol. 15:1507928. [CrossRef]
- Stevenson, K. et al. General calibration of microbial growth in microplate readers. Sci. Rep. 6, 38828; (2016). [CrossRef]
- Mira, P., Yeh, P., & Hall, B. G. (2022). Estimating microbial population data from optical density. PloS one, 17(10), e0276040. [CrossRef]
- Meng, C., Bai, C., Brown, T. D., Hood, L. E., & Tian, Q. (2018). Human Gut Microbiota and Gastrointestinal Cancer. Genomics, proteomics & bioinformatics, 16(1), 33–49. [CrossRef]
- Dominy, S. S., Lynch, C., Ermini, F., Benedyk, M., Marczyk, A., Konradi, A., Nguyen, M., Haditsch, U., Raha, D., Griffin, C., Holsinger, L. J., Arastu-Kapur, S., Kaba, S., Lee, A., Ryder, M. I., Potempa, B., Mydel, P., Hellvard, A., Adamowicz, K., Hasturk, H., … Potempa, J. (2019). Porphyromonas gingivalis in Alzheimer’s disease brains: Evidence for disease causation and treatment with small-molecule inhibitors. Science advances, 5(1), eaau3333. [CrossRef]
- Ryder, M.I. Porphyromonas gingivalis and Alzheimer’s Disease: Recent Findings and Potential Therapies. J. Periodontol. 2020, 91, S45–S49. [CrossRef]
- Castaño-Suárez, L., Paternina-Mejía, G.Y., Vásquez-Olmos, L.D. et al. Linking Periodontitis to Adverse Pregnancy Outcomes: a Comprehensive Review and Meta-analysis. Curr Oral Health Rep 11, 125–137 (2024). [CrossRef]
- Kuraji, R., Sekino, S., Kapila, Y., & Numabe, Y. (2021). Periodontal disease-related nonalcoholic fatty liver disease and nonalcoholic steatohepatitis: An emerging concept of oral-liver axis. Periodontology 2000, 87(1), 204–240. [CrossRef]
- Mäkinen K. K. (2016). Gastrointestinal Disturbances Associated with the Consumption of Sugar Alcohols with Special Consideration of Xylitol: Scientific Review and Instructions for Dentists and Other Health-Care Professionals. International journal of dentistry, 2016, 5967907. [CrossRef]
- Bordier, V.; Teysseire, F.; Senner, F.; et al. Absorption and Metabolism of the Natural Sweeteners Erythritol and Xylitol in Humans: A Dose-Ranging Study. Int. J. Mol. Sci. 2022, 23, 9867. [CrossRef]
- Wölnerhanssen, B. K., Meyer-Gerspach, A. C., Beglinger, C., & Islam, M. S. (2020). Metabolic effects of the natural sweeteners xylitol and erythritol: A comprehensive review. Critical reviews in food science and nutrition, 60(12), 1986–1998. [CrossRef]
- U.S. Food and Drug Administration. GRAS Notice (GRN) No. 893: D-Allulose (Agency Response Letter), 2019. Available online: https://www.fda.gov/media/138901/download (accessed on 11 February 2026).
- Bloomer, R. J., Pence, J., Hellenbrand, J., Davis, A., Davis, S., Stockton, M., & Martin, K. R. (2024). Randomized Trial to Assess the Safety and Tolerability of Daily Intake of an Allulose Amino Acid-Based Hydration Beverage in Men and Women. Nutrients, 16(11), 1766. [CrossRef]
- Uhari, M.; Kontiokari, T.; Koskela, M.; Niemelä, M. Xylitol Chewing Gum in Prevention of Acute Otitis Media: Double Blind Randomised Trial. BMJ 1996, 313, 1180–1184. [CrossRef]
- Persaud, N., Azarpazhooh, A., Keown-Stoneman, C., Birken, C. S., Isaranuwatchai, W., Maguire, J. L., Mamdani, M., Allen, C., Mason, D., Kowal, C., Jaleel, M., Bazeghi, F., Thorpe, K. E., Laupacis, A., & Parkin, P. C. (2024). Xylitol for the prevention of acute otitis media episodes in children aged 1-5 years: a randomised controlled trial. Archives of disease in childhood, 109(2), 121–124. [CrossRef]
- Witkowski, M.; et al. Xylitol Is Prothrombotic and Associated with Cardiovascular Risk. Eur. Heart J. 2024, 45, 2439–2452. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




