Submitted:
31 March 2026
Posted:
02 April 2026
Read the latest preprint version here
Abstract
Keywords:
Introduction
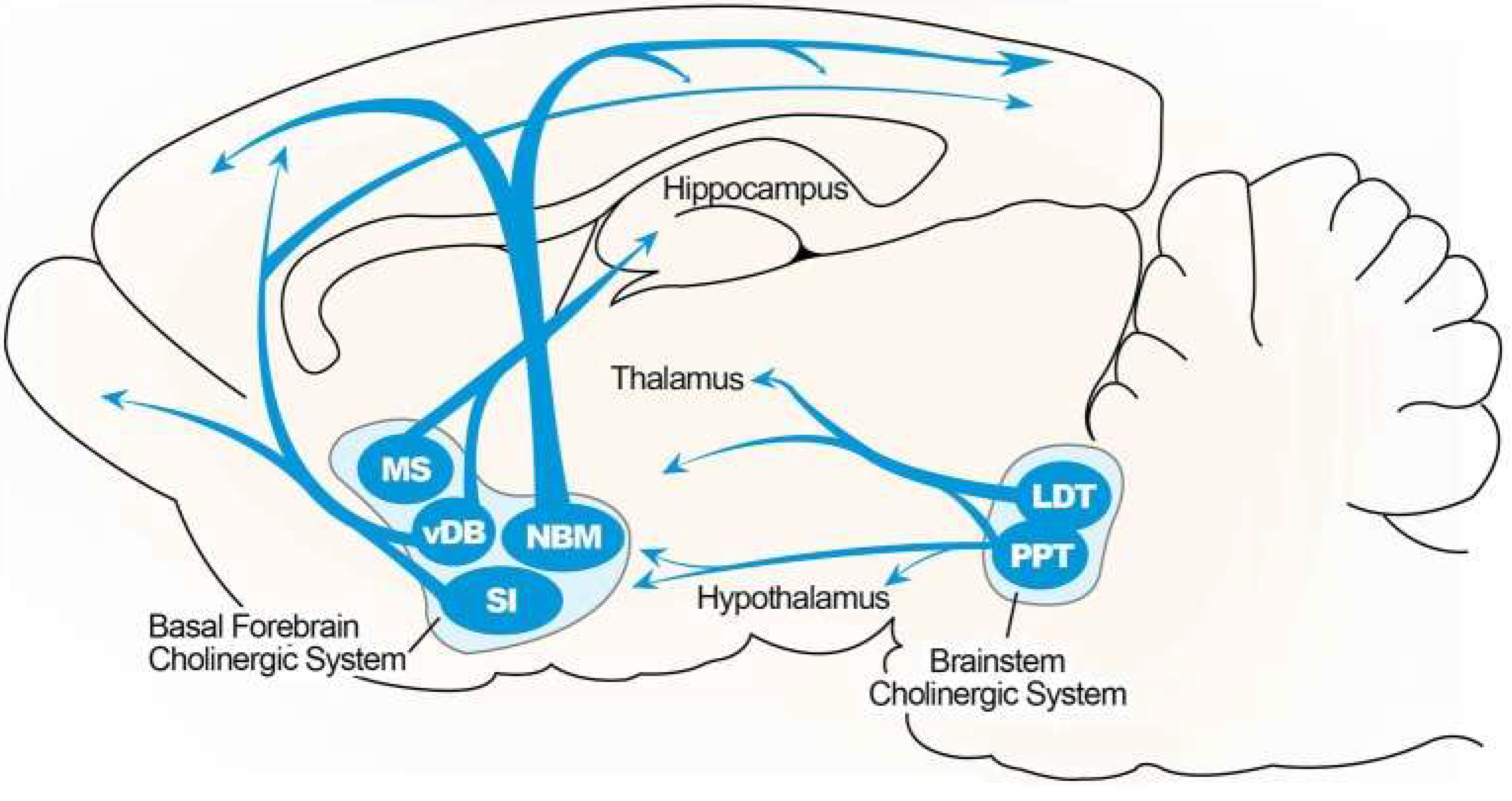
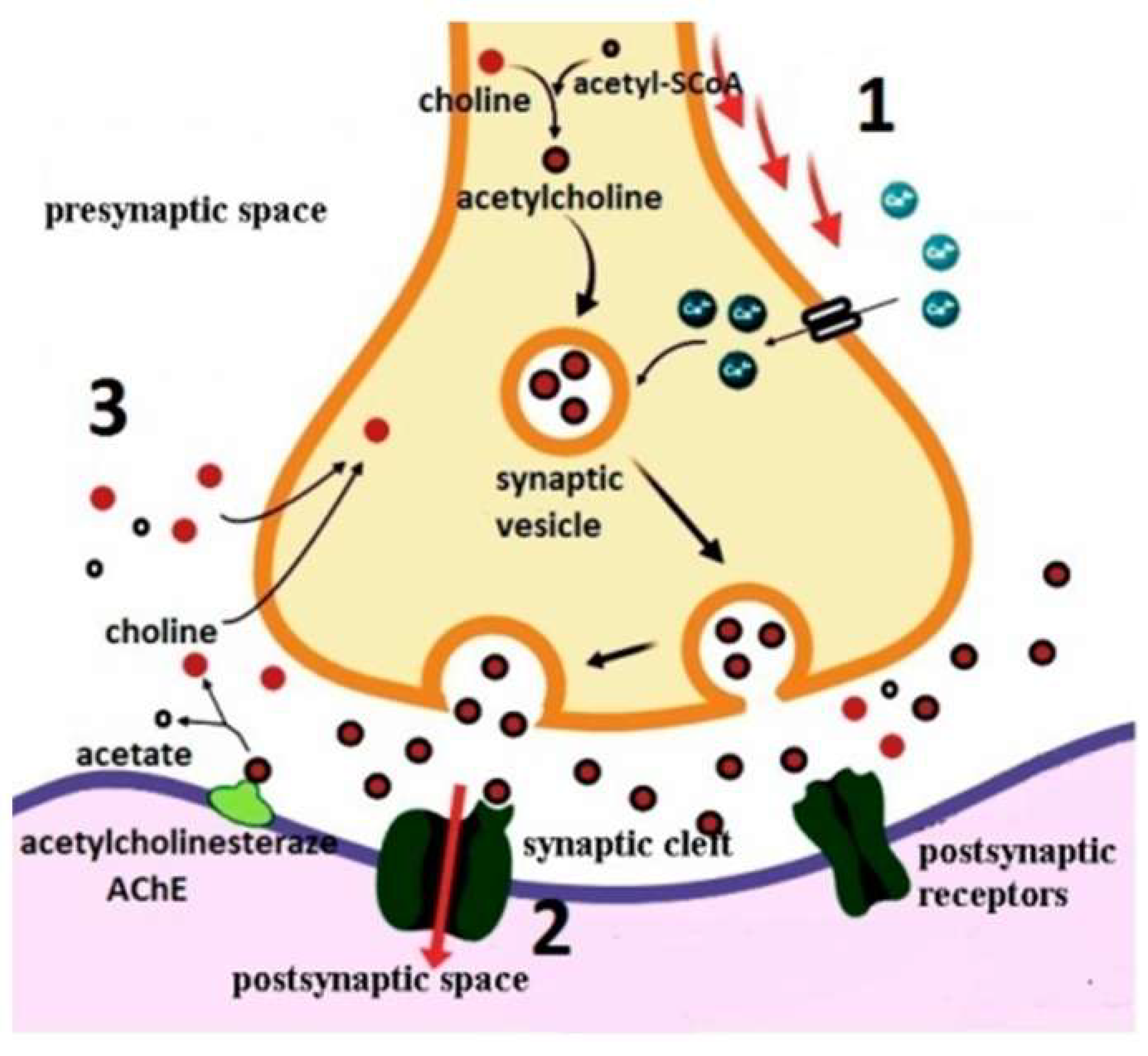
Cholinergic Hypothesis
Aβ
Tau
Endocrine Drivers of Female-Related Alzheimer’s Disease Vulnerability
Follicle-Stimulating Hormone and Its Role in Alzheimer’s Disease Pathogenesis
Convergent Pathways of Ovarian and Neurobiological Aging
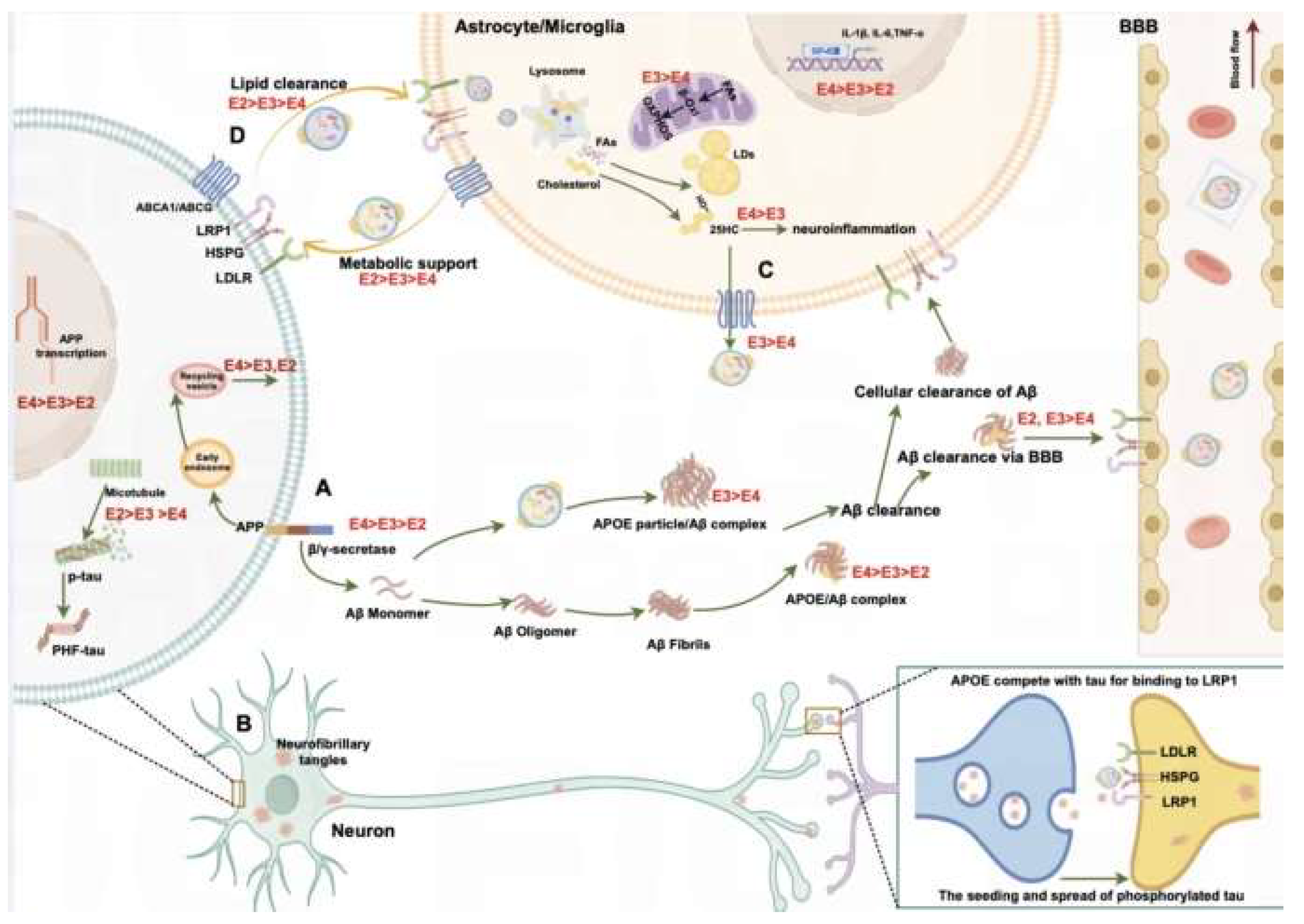
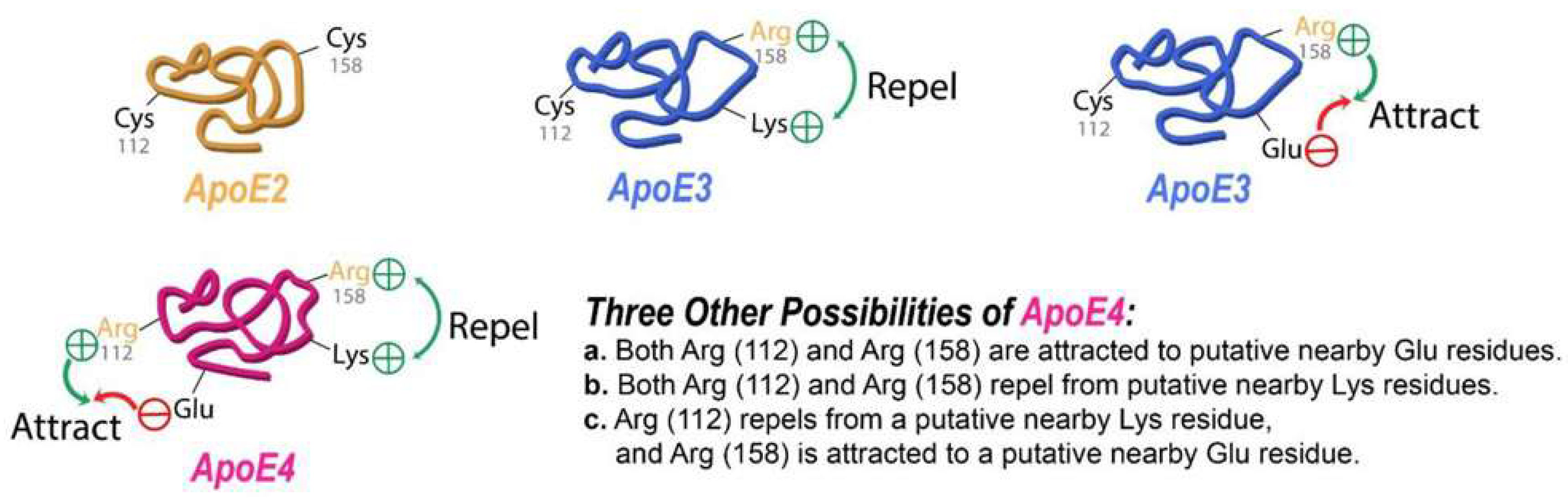
The Role of APOE Variants in AD Pathobiology
| Term | Scale | Structure | Solubility | Stability | Primary Biological Relevance | Toxicity | Relationship to AD |
| Monomers | Molecular | Single Aβ peptides | Soluble | Unstable | Baseline physiological state | Low | Not directly pathogenic |
| Early-stage aggregates (oligomers) | Molecular-nanoscale | Small, flexible clusters (dimers, trimers, etc.) | Soluble | Transient | Synaptic signaling disruption, membrane interaction | Highest | Best correlate with cognitive decline |
| Protofibrils | Nanoscale | Elongated, partially ordered assemblies | Semi-soluble | Intermediate | Transitional species between oligomers and fibrils | High | Contribute to early pathology |
| Mature fibrils | Nanoscale–microscale | Highly ordered β-sheet polymers | Insoluble | Stable | Structural end-products of aggregation | Moderate–low | Build plaques but are not main toxic species |
| Plaques | Tissue-level | Macroscopic extracellular deposits of fibrils + cellular debris | Insoluble | Very stable | Histopathological hallmark | Variable | Poor correlation with symptoms |
Differential Hormonal Regulation of Apolipoprotein E
Metabolic and Lipid Regulation of Apolipoprotein E
A Note on Other Apolipoprotein Variants
Stepwise Neurobiological Dysregulation Driven by Elevated FSH in Alzheimer’s Disease
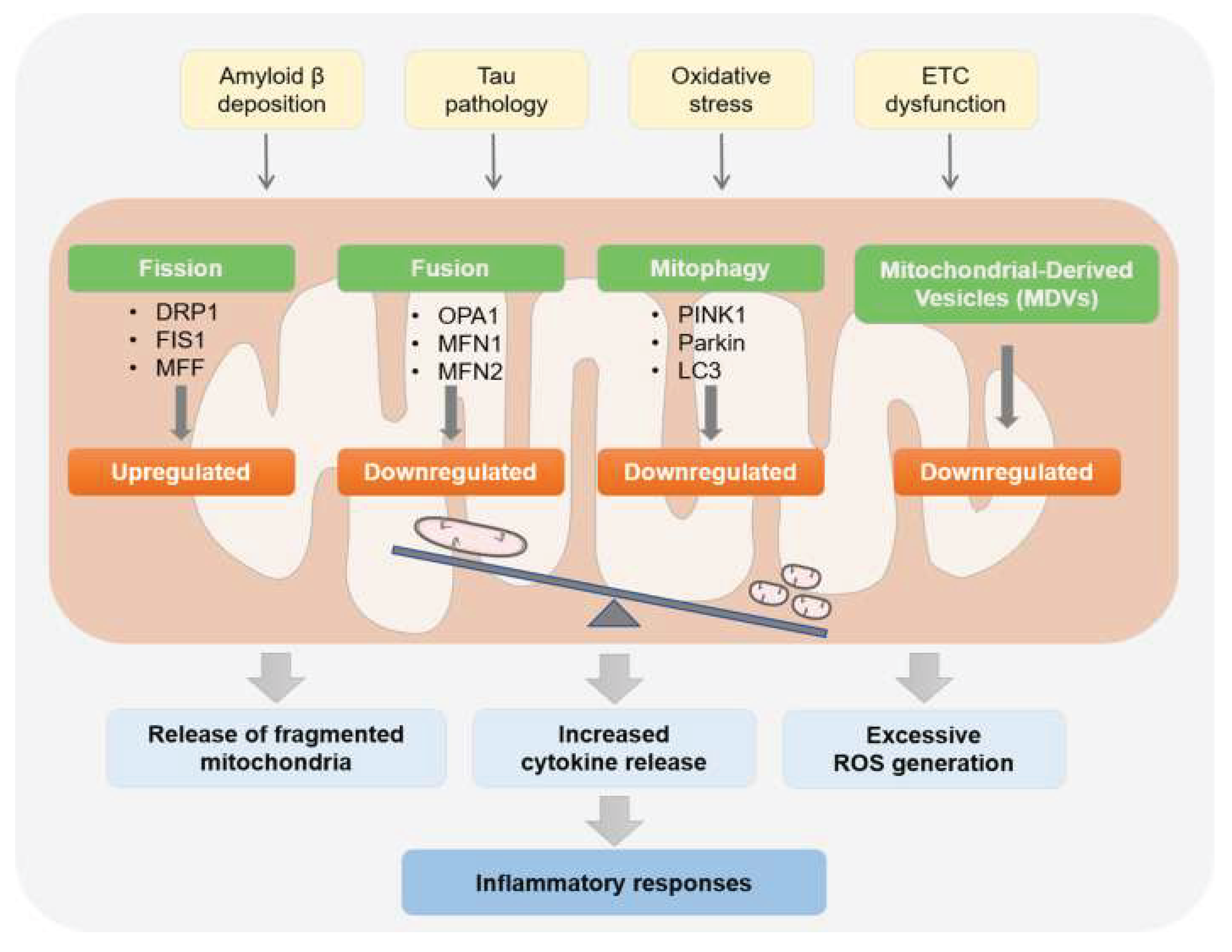
Elevated FSH Disrupts Oxidative Phosphorylation (OXPHOS) in Aging Neurons, Shifting Cellular Energetics Toward Dysfunction
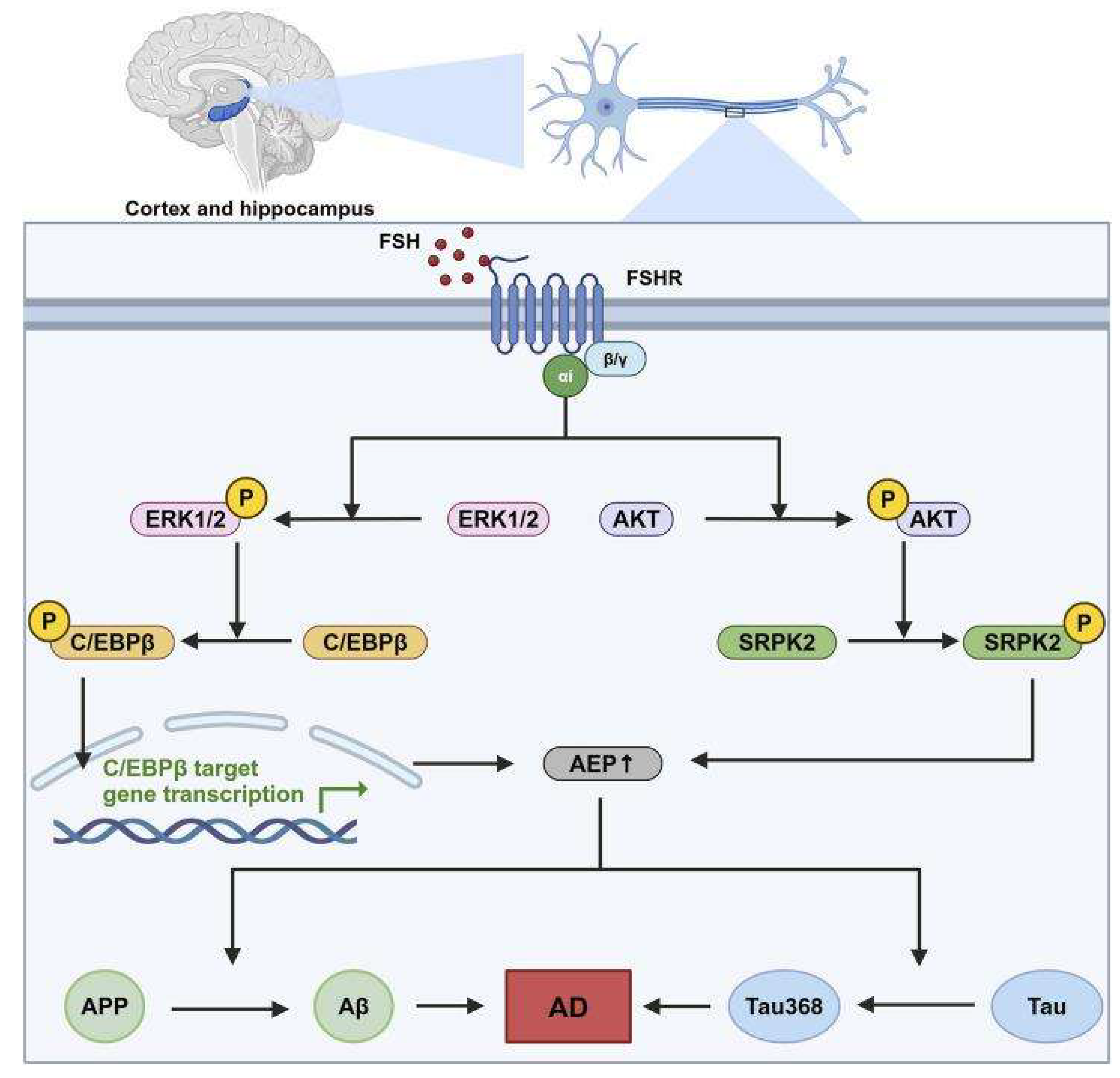
FSH-Mediated Enhancement of ApoE4 as a Barrier to Autophagy: Implications for Proteinopathy
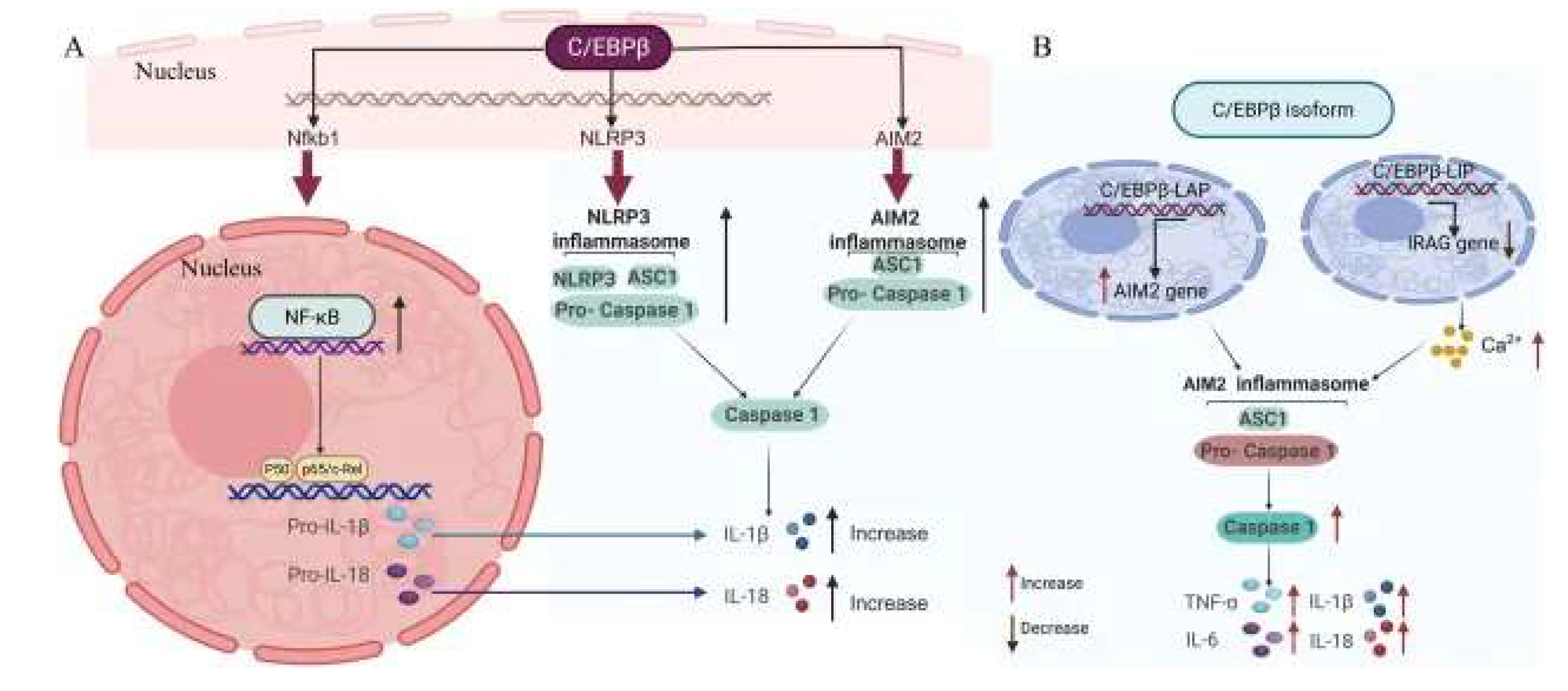
FSH-Mediated Augmentation of Glial Activation in Alzheimer’s Disease
FSH-Mediated Disruption of the Blood-Brain Barrier
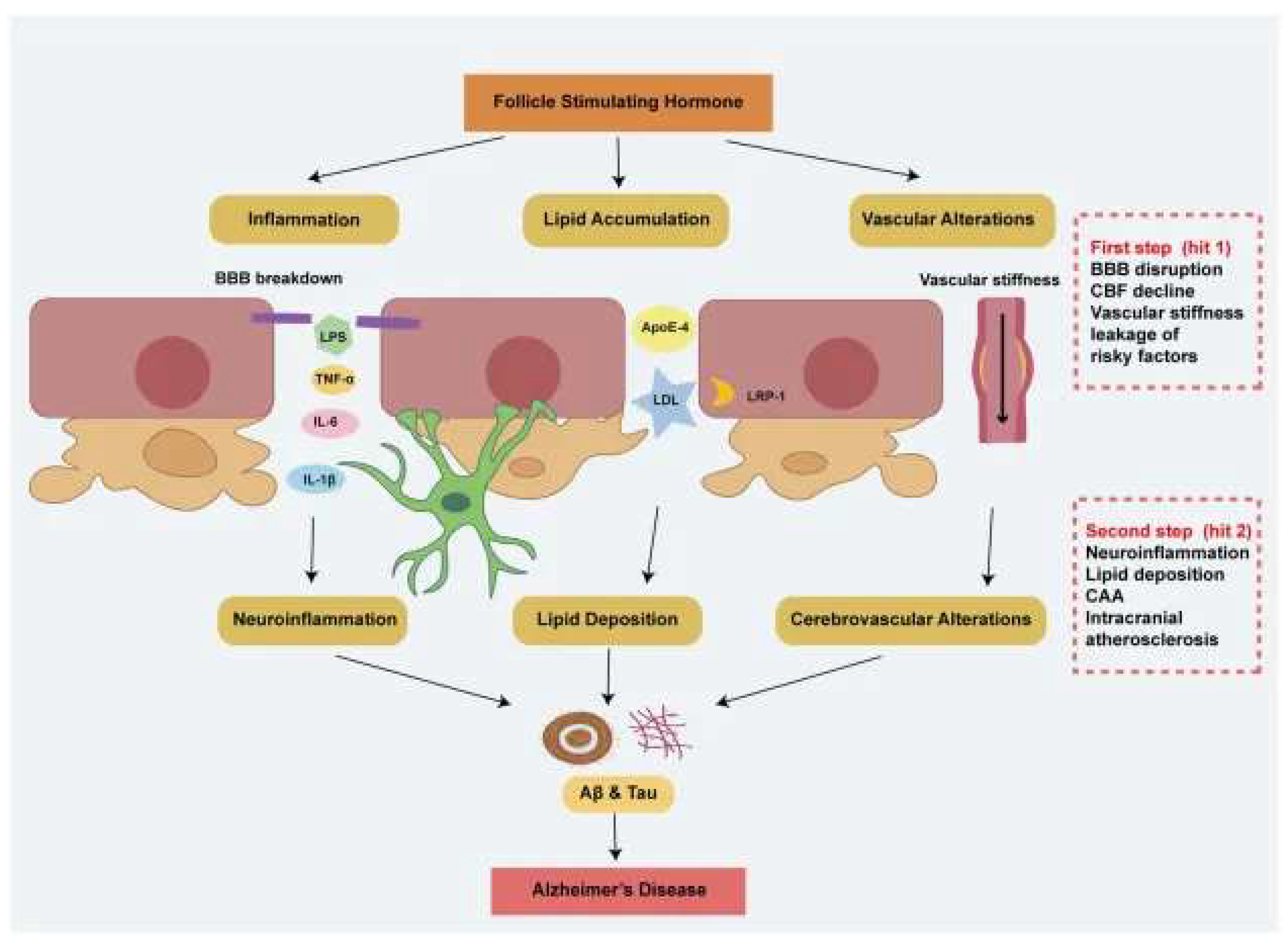
Integrative Mechanistic Framework: How Reproductive Aging in Biological Females Reconfigures Alzheimer’s Disease Risk
Endocrine and Metabolic Targets: Translational Opportunities in Alzheimer’s Disease
Therapeutic Targeting of FSH in Alzheimer’s Disease
Hormone Therapy and Estrogen Receptor Modulation
Targeting Metabolic Perturbations in Alzheimer’s Disease
Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Abeer, M. I.; Abdulhasan, A.; Haguar, Z.; Narayanaswami, V. Isoform-specific modification of apolipoprotein E by 4-hydroxynonenal: protective role of apolipoprotein E3 against oxidative species. The FEBS journal 2023, 290(11), 3006–3025. [Google Scholar] [CrossRef]
- Ali, N.; Sohail, R.; Jaffer, S. R.; Siddique, S.; Kaya, B.; Atowoju, I.; Imran, A.; Wright, W.; Pamulapati, S.; Choudhry, F.; Akbar, A.; Khawaja, U. A. The Role of Estrogen Therapy as a Protective Factor for Alzheimer's Disease and Dementia in Postmenopausal Women: A Comprehensive Review of the Literature. Cureus 2023, 15(8), e43053. [Google Scholar] [CrossRef]
- Alkhalifa, A. E.; Al-Ghraiybah, N. F.; Odum, J.; Shunnarah, J. G.; Austin, N.; Kaddoumi, A. Blood-Brain Barrier Breakdown in Alzheimer's Disease: Mechanisms and Targeted Strategies. International journal of molecular sciences 2023, 24(22), 16288. [Google Scholar] [CrossRef]
- Almansoub, H. A. M. M.; Tang, H.; Wu, Y.; Wang, D. Q.; Mahaman, Y. A. R.; Wei, N.; Almansob, Y. A. M.; He, W.; Liu, D. Tau Abnormalities and the Potential Therapy in Alzheimer's Disease. Journal of Alzheimer's disease: JAD 2019, 67(1), 13–33. [Google Scholar] [CrossRef]
- Almey, A.; Cannell, E.; Bertram, K.; Filardo, E.; Milner, T. A.; Brake, W. G. Medial prefrontal cortical estradiol rapidly alters memory system bias in female rats: ultrastructural analysis reveals membrane-associated estrogen receptors as potential mediators. Endocrinology 2014, 155(11), 4422–4432. [Google Scholar] [CrossRef] [PubMed]
- Alquezar, C.; Arya, S.; Kao, A. W. Tau Post-translational Modifications: Dynamic Transformers of Tau Function, Degradation, and Aggregation. Frontiers in neurology 2021, 11, 595532. [Google Scholar] [CrossRef] [PubMed]
- Alzheimer's disease facts and figures. Alzheimer's & dementia: the journal of the Alzheimer's Association 2024, 20(5), 3708–3821. [CrossRef]
- Andy, C.; Nerattini, M.; Jett, S.; Carlton, C.; Zarate, C.; Boneu, C.; Fauci, F.; Ajila, T.; Battista, M.; Pahlajani, S.; Christos, P.; Fink, M. E.; Williams, S.; Brinton, R. D.; Mosconi, L. Systematic review and meta-analysis of the effects of menopause hormone therapy on cognition. Frontiers in endocrinology 2024, 15, 1350318. [Google Scholar] [CrossRef]
- Apátiga-Pérez, R.; Soto-Rojas, L. O.; Campa-Córdoba, B. B.; Luna-Viramontes, N. I.; Cuevas, E.; Villanueva-Fierro, I.; Ontiveros-Torres, M. A.; Bravo-Muñoz, M.; Flores-Rodríguez, P.; Garcés-Ramirez, L.; de la Cruz, F.; Montiel-Sosa, J. F.; Pacheco-Herrero, M.; Luna-Muñoz, J. Neurovascular dysfunction and vascular amyloid accumulation as early events in Alzheimer's disease. Metabolic brain disease 2022, 37(1), 39–50. [Google Scholar] [CrossRef] [PubMed]
- Ayton, S.; Bush, A. I. β-amyloid: The known unknowns. Ageing research reviews 2021, 65, 101212. [Google Scholar] [CrossRef]
- Baek, M. S.; Cho, H.; Lee, H. S.; Lee, J. H.; Ryu, Y. H.; Lyoo, C. H. Effect of APOE ε4 genotype on amyloid-β and tau accumulation in Alzheimer’s disease. Alzheimer’s Research & Therapy 2020, 12, 140. [Google Scholar] [CrossRef]
- Ball, A. B.; Jones, A. E.; Nguyễn, K. B.; Rios, A.; Marx, N.; Hsieh, W. Y.; Yang, K.; Desousa, B. R.; Kim, K. K. O.; Veliova, M.; Del Mundo, Z. M.; Shirihai, O. S.; Benincá, C.; Stiles, L.; Bensinger, S. J.; Divakaruni, A. S. Pro-inflammatory macrophage activation does not require inhibition of oxidative phosphorylation. EMBO reports 2025, 26(4), 982–1002. [Google Scholar] [CrossRef] [PubMed]
- Baragetti, A.; Catapano, A. L.; Magni, P. Multifactorial Activation of NLRP3 Inflammasome: Relevance for a Precision Approach to Atherosclerotic Cardiovascular Risk and Disease. International journal of molecular sciences 2020, 21(12), 4459. [Google Scholar] [CrossRef] [PubMed]
- Bermejo, P.; Martín-Aragón, S.; Benedí, J.; Susín, C.; Felici, E.; Gil, P.; Villar, Á. M. Peripheral levels of glutathione and protein oxidation as markers in the development of Alzheimer’s disease from Mild Cognitive Impairment. Free Radical Research 2008, 42(2), 162–170. [Google Scholar] [CrossRef] [PubMed]
- Berry, A. S.; Harrison, T. M. New perspectives on the basal forebrain cholinergic system in Alzheimer's disease. Neuroscience and biobehavioral reviews 2023, 150, 105192. [Google Scholar] [CrossRef] [PubMed]
- Beckman, M.; Knox, K.; Koneval, Z.; Smith, C.; Jayadev, S.; Barker-Haliski, M. Loss of presenilin 2 age-dependently alters susceptibility to acute seizures and kindling acquisition. Neurobiology of disease 2020, 136, 104719. [Google Scholar] [CrossRef] [PubMed]
- Bethlehem, R. A. I.; Seidlitz, J.; White, S. R.; Vogel, J. W.; Anderson, K. M.; Adamson, C.; Adler, S.; Alexopoulos, G. S.; Anagnostou, E.; Areces-Gonzalez, A.; Astle, D. E.; Auyeung, B.; Ayub, M.; Bae, J.; Ball, G.; Baron-Cohen, S.; Beare, R.; Bedford, S. A.; Benegal, V.; Beyer, F.; Alexander-Bloch, A. F. Brain charts for the human lifespan. Nature 2022, 604(7906), 525–533. [Google Scholar] [CrossRef]
- Bjørklund, G.; Aaseth, J.; Dadar, M.; Chirumbolo, S. Molecular Targets in Alzheimer's Disease. Molecular neurobiology 2019, 56(10), 7032–7044. [Google Scholar] [CrossRef]
- Brosseron, F.; Krauthausen, M.; Kummer, M.; Heneka, M. T. Body fluid cytokine levels in mild cognitive impairment and Alzheimer's disease: a comparative overview. Molecular neurobiology 2014, 50(2), 534–544. [Google Scholar] [CrossRef]
- Bronzuoli, M. R.; Facchinetti, R.; Valenza, M.; Cassano, T.; Steardo, L.; Scuderi, C. Astrocyte Function Is Affected by Aging and Not Alzheimer's Disease: A Preliminary Investigation in Hippocampi of 3xTg-AD Mice. Frontiers in pharmacology 2019, 10, 644. [Google Scholar] [CrossRef]
- Burkhardt, M. S.; Foster, J. K.; Laws, S. M.; Baker, L. D.; Craft, S.; Gandy, S. E.; Stuckey, B. G.; Clarnette, R.; Nolan, D.; Hewson-Bower, B.; Martins, R. N. Oestrogen replacement therapy may improve memory functioning in the absence of APOE epsilon4. Journal of Alzheimer's disease: JAD 2004, 6(3), 221–228. [Google Scholar] [CrossRef]
- Butterfield, D. A.; Mattson, M. P. Apolipoprotein E and oxidative stress in brain with relevance to Alzheimer's disease. Neurobiology of disease 2020, 138, 104795. [Google Scholar] [CrossRef]
- Casarini, L.; Crépieux, P. Molecular Mechanisms of Action of FSH. Frontiers in endocrinology 2019, 10, 305. [Google Scholar] [CrossRef]
- Castellani, R. J.; Jamshidi, P.; Plascencia-Villa, G.; Perry, G. The Amyloid Cascade Hypothesis: A Conclusion in Search of Support. The American journal of pathology 2025, 195(11), 1988–1997. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Fang, H. Q.; Liu, J. T.; Chang, S. Y.; Cheng, L. B.; Sun, M. X.; Feng, J. R.; Liu, Z. M.; Zhang, Y. H.; Rosen, C. J.; Liu, P. Atlas of Fshr expression from novel reporter mice. eLife 2025, 13, RP93413. [Google Scholar] [CrossRef]
- Chen, J.; Li, Q.; Wang, J. Topology of human apolipoprotein E3 uniquely regulates its diverse biological functions. Proceedings of the National Academy of Sciences of the United States of America 2011, 108(36), 14813–14818. [Google Scholar] [CrossRef]
- Chen, J.; Sun, J.; Hu, Y.; Wan, X.; Wang, Y.; Gao, M.; Liang, J.; Liu, T.; Sun, X. MicroRNA-191-5p ameliorates amyloid-β1-40 -mediated retinal pigment epithelium cell injury by suppressing the NLRP3 inflammasome pathway. FASEB journal: official publication of the Federation of American Societies for Experimental Biology 2021, 35(4), e21184. [Google Scholar] [CrossRef]
- Chen, T. H.; Koh, K. Y.; Lin, K. M.; Chou, C. K. Mitochondrial Dysfunction as an Underlying Cause of Skeletal Muscle Disorders. International journal of molecular sciences 2022, 23(21), 12926. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Jin, H.; Chen, J.; Li, J.; Găman, M. A.; Zou, Z. The multifaceted roles of apolipoprotein E4 in Alzheimer's disease pathology and potential therapeutic strategies. Cell death discovery 2025, 11(1), 312. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y. J.; Lin, C. H.; Lane, H. Y. From Menopause to Neurodegeneration-Molecular Basis and Potential Therapy. International journal of molecular sciences 2021, 22(16), 8654. [Google Scholar] [CrossRef]
- Chong, C. M.; Ke, M.; Tan, Y.; Huang, Z.; Zhang, K.; Ai, N.; Ge, W.; Qin, D.; Lu, J. H.; Su, H. Presenilin 1 deficiency suppresses autophagy in human neural stem cells through reducing γ-secretase-independent ERK/CREB signaling. Cell death & disease 2018, 9(9), 879. [Google Scholar] [CrossRef]
- Christodoulou, R. C.; Eller, D.; Papageorgiou, P. S.; Angelopoulou, E.; Vassiliou, E.; Papageorgiou, S. G. Metabolic Dysfunction in Alzheimer's Disease: Brain Glucose Hypometabolism as an Early Precursor to Amyloid and Tau Pathology. Journal of clinical medicine 2026, 15(5), 1884. [Google Scholar] [CrossRef]
- Cobley, J. N.; Fiorello, M. L.; Bailey, D. M. 13 reasons why the brain is susceptible to oxidative stress. Redox biology 2018, 15, 490–503. [Google Scholar] [CrossRef]
- Cohen, J.; Torres, C. Astrocyte senescence: Evidence and significance. Aging cell 2019, 18(3), e12937. [Google Scholar] [CrossRef]
- Combs, B.; Mueller, R. L.; Morfini, G.; Brady, S. T.; Kanaan, N. M. Tau and Axonal Transport Misregulation in Tauopathies. Advances in experimental medicine and biology 2019, 1184, 81–95. [Google Scholar] [CrossRef]
- Corraliza-Gomez, M.; Sanchez, D.; Ganfornina, M. D. Lipid-Binding Proteins in Brain Health and Disease. Frontiers in neurology 2019, 10, 1152. [Google Scholar] [CrossRef]
- Cortes-Canteli, M.; Iadecola, C. Alzheimer's Disease and Vascular Aging: JACC Focus Seminar. Journal of the American College of Cardiology 2020, 75(8), 942–951. [Google Scholar] [CrossRef] [PubMed]
- Coughlan, G. T.; Betthauser, T. J.; Boyle, R.; Koscik, R. L.; Klinger, H. M.; Chibnik, L. B.; Jonaitis, E. M.; Yau, W. W.; Wenzel, A.; Christian, B. T.; Gleason, C. E.; Saelzler, U. G.; Properzi, M. J.; Schultz, A. P.; Hanseeuw, B. J.; Manson, J. E.; Rentz, D. M.; Johnson, K. A.; Sperling, R.; Johnson, S. C.; Buckley, R. F. Association of Age at Menopause and Hormone Therapy Use With Tau and β-Amyloid Positron Emission Tomography. JAMA neurology 2023, 80(5), 462–473. [Google Scholar] [CrossRef] [PubMed]
- Craig, M. C.; Maki, P. M.; Murphy, D. G. The Women's Health Initiative Memory Study: findings and implications for treatment. The Lancet. Neurology 2005, 4(3), 190–194. [Google Scholar] [CrossRef] [PubMed]
- Dani, M.; Wood, M.; Mizoguchi, R.; Fan, Z.; Walker, Z.; Morgan, R.; Hinz, R.; Biju, M.; Kuruvilla, T.; Brooks, D. J.; Edison, P. Microglial activation correlates in vivo with both tau and amyloid in Alzheimer's disease. Brain: a journal of neurology 2018, 141(9), 2740–2754. [Google Scholar] [CrossRef] [PubMed]
- Dasadhikari, S.; Ghosh, S.; Pal, S.; Knowles, T. P. J.; Garai, K. A single fibril study reveals that ApoE inhibits the elongation of Aβ42 fibrils in an isoform-dependent manner. Communications chemistry 2025, 8(1), 133. [Google Scholar] [CrossRef] [PubMed]
- Deane, R.; Sagare, A.; Hamm, K.; Parisi, M.; Lane, S.; Finn, M. B.; Holtzman, D. M.; Zlokovic, B. V. apoE isoform-specific disruption of amyloid beta peptide clearance from mouse brain. The Journal of clinical investigation 2008, 118(12), 4002–4013. [Google Scholar] [CrossRef] [PubMed]
- Deming, Y.; Filipello, F.; Cignarella, F.; Cantoni, C.; Hsu, S.; Mikesell, R.; Li, Z.; Del-Aguila, J. L.; Dube, U.; Farias, F. G.; Bradley, J.; Budde, J.; Ibanez, L.; Fernandez, M. V.; Blennow, K.; Zetterberg, H.; Heslegrave, A.; Johansson, P. M.; Svensson, J.; Nellgård, B.; Cruchaga, C. The MS4A gene cluster is a key modulator of soluble TREM2 and Alzheimer's disease risk. Science translational medicine 2019, 11(505), eaau2291. [Google Scholar] [CrossRef]
- Depypere, H.; Vergallo, A.; Lemercier, P.; Lista, S.; Benedet, A.; Ashton, N.; Cavedo, E.; Zetterberg, H.; Blennow, K.; Vanmechelen, E.; Hampel, H.; Neurodegeneration Precision Medicine Initiative (NPMI). Menopause hormone therapy significantly alters pathophysiological biomarkers of Alzheimer's disease. Alzheimer's & dementia: the journal of the Alzheimer's Association 2023, 19(4), 1320–1330. [Google Scholar] [CrossRef]
- Descalzi, G; Gao, V; Steinman, MQ; Suzuki, A; Alberini, CM. Lactate from astrocytes fuels learning-induced mRNA translation in excitatory and inhibitory neurons. Commun Biol. 2019, 2, 247. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Doering, S.; McKay, N. S.; Jana, N.; Dombrowski, K.; McCullough, A.; Millar, P. R.; Hobbs, D. A.; Agrawal, R.; Flores, S.; Llibre-Guerra, J. J.; Huey, E. D.; Ances, B. M.; Xiong, C.; Aschenbrenner, A. J.; Hassenstab, J.; Morris, J. C.; Gordon, B. A.; Benzinger, T. L. S. Domain-specific cognitive impairment is differentially affected by Alzheimer disease tau pathologic burden and spread. Imaging neuroscience (Cambridge, Mass.) 2024, 2, imag-2-00405. [Google Scholar] [CrossRef]
- Driscoll, D. M.; Schreiber, J. R.; Schmit, V. M.; Getz, G. S. Regulation of apolipoprotein E synthesis in rat ovarian granulosa cells. The Journal of biological chemistry 1985, 260(15), 9031–9038. [Google Scholar] [CrossRef]
- Duffy, D. M.; Ko, C.; Jo, M.; Brannstrom, M.; Curry, T. E. Ovulation: Parallels With Inflammatory Processes. Endocrine reviews 2019, 40(2), 369–416. [Google Scholar] [CrossRef]
- Erten-Lyons, D.; Dodge, H. H.; Woltjer, R.; Silbert, L. C.; Howieson, D. B.; Kramer, P.; Kaye, J. A. Neuropathologic basis of age-associated brain atrophy. JAMA neurology 2013, 70(5), 616–622. [Google Scholar] [CrossRef]
- Eskandari-Sedighi, G.; Crichton, M.; Zia, S.; Gomez-Cardona, E.; Cortez, L. M.; Patel, Z. H.; Takahashi-Yamashiro, K.; St Laurent, C. D.; Sidhu, G.; Sarkar, S.; Aghanya, V.; Sim, V. L.; Tan, Q.; Julien, O.; Plemel, J. R.; Macauley, M. S. Alzheimer's disease associated isoforms of human CD33 distinctively modulate microglial cell responses in 5XFAD mice. Molecular neurodegeneration 2024, 19(1), 42. [Google Scholar] [CrossRef]
- Fedeli, C.; Filadi, R.; Rossi, A.; Mammucari, C.; Pizzo, P. PSEN2 (presenilin 2) mutants linked to familial Alzheimer disease impair autophagy by altering Ca2+ homeostasis. Autophagy 2019, 15(12), 2044–2062. [Google Scholar] [CrossRef]
- Fedele, E. Anti-Amyloid Therapies for Alzheimer's Disease and the Amyloid Cascade Hypothesis. International journal of molecular sciences 2023, 24(19), 14499. [Google Scholar] [CrossRef]
- Feng, Y.; He, D.; Yao, Z.; Klionsky, D. J. The machinery of macroautophagy. Cell research 2014, 24(1), 24–41. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, M. T.; Iulita, M. F.; Cavedo, E.; Chiesa, P. A.; Schumacher Dimech, A.; Santuccione Chadha, A.; Baracchi, F.; Girouard, H.; Misoch, S.; Giacobini, E.; Depypere, H.; Hampel, H. & Women’s Brain Project and the Alzheimer Precision Medicine Initiative (2018). Sex differences in Alzheimer disease - the gateway to precision medicine. Nature reviews. Neurology 14(8), 457–469. [CrossRef]
- Fleischer, A. W.; Schalk, J. C.; Wetzel, E. A.; Hanson, A. M.; Sem, D. S.; Donaldson, W. A.; Frick, K. M. Long-term oral administration of a novel estrogen receptor beta agonist enhances memory and alleviates drug-induced vasodilation in young ovariectomized mice. Hormones and behavior 2021, 130, 104948. [Google Scholar] [CrossRef] [PubMed]
- Frieden, C.; Garai, K. Concerning the structure of apoE. Protein science: a publication of the Protein Society 2013, 22(12), 1820–1825. [Google Scholar] [CrossRef] [PubMed]
- Frieden, C.; Wang, H.; Ho, C. M. W. A mechanism for lipid binding to apoE and the role of intrinsically disordered regions coupled to domain-domain interactions. Proceedings of the National Academy of Sciences of the United States of America 2017, 114(24), 6292–6297. [Google Scholar] [CrossRef]
- Fruhwürth, S.; Zetterberg, H.; Paludan, S. R. Microglia and amyloid plaque formation in Alzheimer's disease - Evidence, possible mechanisms, and future challenges. Journal of neuroimmunology 2024, 390, 578342. [Google Scholar] [CrossRef]
- Fung, Y. L.; Ng, K. E. T.; Vogrin, S. J.; Meade, C.; Ngo, M.; Collins, S. J.; Bowden, S. C. Comparative Utility of Manual versus Automated Segmentation of Hippocampus and Entorhinal Cortex Volumes in a Memory Clinic Sample. Journal of Alzheimer's disease: JAD 2019, 68(1), 159–171. [Google Scholar] [CrossRef] [PubMed]
- Gabbouj, S.; Ryhänen, S.; Marttinen, M.; Wittrahm, R.; Takalo, M.; Kemppainen, S.; Martiskainen, H.; Tanila, H.; Haapasalo, A.; Hiltunen, M.; Natunen, T. Altered Insulin Signaling in Alzheimer's Disease Brain - Special Emphasis on PI3K-Akt Pathway. Frontiers in neuroscience 2019, 13, 629. [Google Scholar] [CrossRef] [PubMed]
- Ganfornina, M. D.; Do Carmo, S.; Lora, J. M.; Torres-Schumann, S.; Vogel, M.; Allhorn, M.; González, C.; Bastiani, M. J.; Rassart, E.; Sanchez, D. Apolipoprotein D is involved in the mechanisms regulating protection from oxidative stress. Aging cell 2008, 7(4), 506–515. [Google Scholar] [CrossRef]
- Gao, C.; Jiang, J.; Tan, Y.; Chen, S. Microglia in neurodegenerative diseases: mechanism and potential therapeutic targets. Signal transduction and targeted therapy 2023, 8(1), 359. [Google Scholar] [CrossRef]
- Gawriluk, T. R.; Hale, A. N.; Flaws, J. A.; Dillon, C. P.; Green, D. R.; Rucker, E. B., 3rd. Autophagy is a cell survival program for female germ cells in the murine ovary. Reproduction (Cambridge, England) 2011, 141(6), 759–765. [Google Scholar] [CrossRef] [PubMed]
- Gazestani, V.; Kamath, T.; Nadaf, N. M.; Dougalis, A.; Burris, S. J.; Rooney, B.; Junkkari, A.; Vanderburg, C.; Pelkonen, A.; Gomez-Budia, M.; Välimäki, N. N.; Rauramaa, T.; Therrien, M.; Koivisto, A. M.; Tegtmeyer, M.; Herukka, S. K.; Abdulraouf, A.; Marsh, S. E.; Hiltunen, M.; Nehme, R.; Macosko, E. Z. Early Alzheimer's disease pathology in human cortex involves transient cell states. Cell 2023, 186(20), 4438–4453.e23. [Google Scholar] [CrossRef]
- Getz, G. S.; Reardon, C. A. Apoprotein E as a lipid transport and signaling protein in the blood, liver, and artery wall. Journal of lipid research 2009, 50 Suppl(Suppl), S156–S161. [Google Scholar] [CrossRef] [PubMed]
- Gonos, E. S.; Kapetanou, M.; Sereikaite, J.; Bartosz, G.; Naparło, K.; Grzesik, M.; Sadowska-Bartosz, I. Origin and pathophysiology of protein carbonylation, nitration and chlorination in age-related brain diseases and aging. Aging 2018, 10(5), 868–901. [Google Scholar] [CrossRef] [PubMed]
- Goodall, E. F.; Wang, C.; Simpson, J. E.; Baker, D. J.; Drew, D. R.; Heath, P. R.; Saffrey, M. J.; Romero, I. A.; Wharton, S. B. Age-associated changes in the blood-brain barrier: comparative studies in human and mouse. Neuropathology and applied neurobiology 2018, 44(3), 328–340. [Google Scholar] [CrossRef]
- Grammatikopoulou, M. G.; Goulis, D. G.; Gkiouras, K.; Theodoridis, X.; Gkouskou, K. K.; Evangeliou, A.; Dardiotis, E.; Bogdanos, D. P. To Keto or Not to Keto? A Systematic Review of Randomized Controlled Trials Assessing the Effects of Ketogenic Therapy on Alzheimer Disease. Advances in nutrition (Bethesda, Md.) 2020, 11(6), 1583–1602. [Google Scholar] [CrossRef] [PubMed]
- Gyparaki, M. T.; Arab, A.; Sorokina, E. M.; Santiago-Ruiz, A. N.; Bohrer, C. H.; Xiao, J.; Lakadamyali, M. Tau forms oligomeric complexes on microtubules that are distinct from tau aggregates. Proceedings of the National Academy of Sciences of the United States of America 2021, 118(19), e2021461118. [Google Scholar] [CrossRef]
- Hagihara, H.; Miyakawa, T. Decreased Brain pH Correlated With Progression of Alzheimer Disease Neuropathology: A Systematic Review and Meta-Analyses of Postmortem Studies. The international journal of neuropsychopharmacology 2024, 27(10), pyae047. [Google Scholar] [CrossRef]
- Hall, J. E.; Lavoie, H. B.; Marsh, E. E.; Martin, K. A. Decrease in gonadotropin-releasing hormone (GnRH) pulse frequency with aging in postmenopausal women. The Journal of clinical endocrinology and metabolism 2000, 85(5), 1794–1800. [Google Scholar] [CrossRef]
- Halliday, M.; Mallucci, G. R. Review: Modulating the unfolded protein response to prevent neurodegeneration and enhance memory. Neuropathology and applied neurobiology 2015, 41(4), 414–427. [Google Scholar] [CrossRef]
- Hamilton, C.; Anand, P. K. Right place, right time: localisation and assembly of the NLRP3 inflammasome. F1000Research 2019, 8, F1000 Faculty Rev–676. [Google Scholar] [CrossRef]
- Hampel, H.; Mesulam, M. M.; Cuello, A. C.; Khachaturian, A. S.; Vergallo, A.; Farlow, M. R.; Snyder, P. J.; Giacobini, E.; Khachaturian, Z. S. Revisiting the Cholinergic Hypothesis in Alzheimer's Disease: Emerging Evidence from Translational and Clinical Research. The journal of prevention of Alzheimer's disease 2019, 6(1), 2–15. [Google Scholar] [CrossRef] [PubMed]
- Hammond, T. R.; Marsh, S. E.; Stevens, B. Immune Signaling in Neurodegeneration. Immunity 2019, 50(4), 955–974. [Google Scholar] [CrossRef] [PubMed]
- Hara, Y.; McKeehan, N.; Dacks, P. A.; Fillit, H. M. Evaluation of the Neuroprotective Potential of N-Acetylcysteine for Prevention and Treatment of Cognitive Aging and Dementia. The journal of prevention of Alzheimer's disease 2017, 4(3), 201–206. [Google Scholar] [CrossRef] [PubMed]
- Harrison, T. M.; La Joie, R.; Maass, A.; Baker, S. L.; Swinnerton, K.; Fenton, L.; Mellinger, T. J.; Edwards, L.; Pham, J.; Miller, B. L.; Rabinovici, G. D.; Jagust, W. J. Longitudinal tau accumulation and atrophy in aging and alzheimer disease. Annals of neurology 2019, 85(2), 229–240. [Google Scholar] [CrossRef] [PubMed]
- Hatters, D. M.; Budamagunta, M. S.; Voss, J. C.; Weisgraber, K. H. Modulation of apolipoprotein E structure by domain interaction: differences in lipid-bound and lipid-free forms. The Journal of biological chemistry 2005, 280(40), 34288–34295. [Google Scholar] [CrossRef]
- Heilig, E. A.; Xia, W.; Shen, J.; Kelleher, R. J., 3rd. A presenilin-1 mutation identified in familial Alzheimer disease with cotton wool plaques causes a nearly complete loss of gamma-secretase activity. The Journal of biological chemistry 2010, 285(29), 22350–22359. [Google Scholar] [CrossRef]
- Henderson, V. W.; Benke, K. S.; Green, R. C.; Cupples, L. A.; Farrer, L. A.; MIRAGE Study Group. Postmenopausal hormone therapy and Alzheimer's disease risk: interaction with age. Journal of neurology, neurosurgery, and psychiatry 2005, 76(1), 103–105. [Google Scholar] [CrossRef]
- Henderson, V. W.; Paganini-Hill, A.; Emanuel, C. K.; Dunn, M. E.; Buckwalter, J. G. Estrogen replacement therapy in older women. Comparisons between Alzheimer's disease cases and nondemented control subjects. Archives of neurology 1994, 51(9), 896–900. [Google Scholar] [CrossRef] [PubMed]
- Heneka, M. T.; Kummer, M. P.; Stutz, A.; Delekate, A.; Schwartz, S.; Vieira-Saecker, A.; Griep, A.; Axt, D.; Remus, A.; Tzeng, T. C.; Gelpi, E.; Halle, A.; Korte, M.; Latz, E.; Golenbock, D. T. NLRP3 is activated in Alzheimer's disease and contributes to pathology in APP/PS1 mice. Nature 2013, 493(7434), 674–678. [Google Scholar] [CrossRef]
- Hickman, S. E.; Kingery, N. D.; Ohsumi, T. K.; Borowsky, M. L.; Wang, L. C.; Means, T. K.; El Khoury, J. The microglial sensome revealed by direct RNA sequencing. Nature neuroscience 2013, 16(12), 1896–1905. [Google Scholar] [CrossRef]
- Hilton Boon, M.; Thomson, H.; Shaw, B.; Akl, E. A.; Lhachimi, S. K.; López-Alcalde, J.; Klugar, M.; Choi, L.; Saz-Parkinson, Z.; Mustafa, R. A.; Langendam, M. W.; Crane, O.; Morgan, R. L.; Rehfuess, E.; Johnston, B. C.; Chong, L. Y.; Guyatt, G. H.; Schünemann, H. J.; Katikireddi, S. V.; GRADE Working Group. Challenges in applying the GRADE approach in public health guidelines and systematic reviews: a concept article from the GRADE Public Health Group. Journal of clinical epidemiology 2021, 135, 42–53. [Google Scholar] [CrossRef]
- Hornung, V.; Ablasser, A.; Charrel-Dennis, M.; Bauernfeind, F.; Horvath, G.; Caffrey, D. R.; Latz, E.; Fitzgerald, K. A. AIM2 recognizes cytosolic dsDNA and forms a caspase-1-activating inflammasome with ASC. Nature 2009, 458(7237), 514–518. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z. H.; Gu, D.; Mazzone, T. Oleic acid modulates the post-translational glycosylation of macrophage ApoE to increase its secretion. The Journal of biological chemistry 2004, 279(28), 29195–29201. [Google Scholar] [CrossRef]
- Iaccarino, L.; Tammewar, G.; Ayakta, N.; Baker, S. L.; Bejanin, A.; Boxer, A. L.; Gorno-Tempini, M. L.; Janabi, M.; Kramer, J. H.; Lazaris, A.; Lockhart, S. N.; Miller, B. L.; Miller, Z. A.; O'Neil, J. P.; Ossenkoppele, R.; Rosen, H. J.; Schonhaut, D. R.; Jagust, W. J.; Rabinovici, G. D. Local and distant relationships between amyloid, tau and neurodegeneration in Alzheimer's Disease. NeuroImage. Clinical 2017, 17, 452–464. [Google Scholar] [CrossRef]
- Ioannou, K.; Bucci, M.; Tzortzakakis, A.; Savitcheva, I.; Nordberg, A.; Chiotis, K. & Alzheimer’s Disease Neuroimaging Initiative Tau PET positivity predicts clinically relevant cognitive decline driven by Alzheimer's disease compared to comorbid cases; proof of concept in the ADNI study. Molecular psychiatry 2025, 30(2), 587–599. [Google Scholar] [CrossRef] [PubMed]
- Islam, S.; Noorani, A.; Sun, Y.; Michikawa, M.; Zou, K. Multi-functional role of apolipoprotein E in neurodegenerative diseases. Frontiers in aging neuroscience 2025, 17, 1535280. [Google Scholar] [CrossRef] [PubMed]
- Ismail, R.; Parbo, P.; Madsen, L. S.; Hansen, A. K.; Hansen, K. V.; Schaldemose, J. L.; Kjeldsen, P. L.; Stokholm, M. G.; Gottrup, H.; Eskildsen, S. F.; Brooks, D. J. The relationships between neuroinflammation, beta-amyloid and tau deposition in Alzheimer's disease: a longitudinal PET study. Journal of neuroinflammation 2020, 17(1), 151. [Google Scholar] [CrossRef]
- Jamerlan, A.; Hulme, J. From Evasion to Collapse: The Kinetic Cascade of TDP-43 and the Failure of Proteostasis. International Journal of Molecular Sciences 2026, 27(3), 1136. [Google Scholar] [CrossRef]
- Jia, M.; Dahlman-Wright, K.; Gustafsson, J. Å. Estrogen receptor alpha and beta in health and disease. Best practice & research. Clinical endocrinology & metabolism 2015, 29(4), 557–568. [Google Scholar] [CrossRef]
- Jiang, Q.; Lee, C. Y.; Mandrekar, S.; Wilkinson, B.; Cramer, P.; Zelcer, N.; Mann, K.; Lamb, B.; Willson, T. M.; Collins, J. L.; Richardson, J. C.; Smith, J. D.; Comery, T. A.; Riddell, D.; Holtzman, D. M.; Tontonoz, P.; Landreth, G. E. ApoE promotes the proteolytic degradation of Abeta. Neuron 2008, 58(5), 681–693. [Google Scholar] [CrossRef]
- Jones, G. H.; Vecera, C. M.; Pinjari, O. F.; Machado-Vieira, R. Inflammatory signaling mechanisms in bipolar disorder. Journal of biomedical science 2021, 28(1), 45. [Google Scholar] [CrossRef] [PubMed]
- Jung, E. S.; Choi, H.; Mook-Jung, I. Decoding microglial immunometabolism: a new frontier in Alzheimer's disease research. Molecular neurodegeneration 2025, 20(1), 37. [Google Scholar] [CrossRef] [PubMed]
- Kalenga, C. Z.; Hay, J. L.; Boreskie, K. F.; Duhamel, T. A.; MacRae, J. M.; Metcalfe, A.; Nerenberg, K. A.; Robert, M.; Ahmed, S. B. The Association Between Route of Post-menopausal Estrogen Administration and Blood Pressure and Arterial Stiffness in Community-Dwelling Women. Frontiers in cardiovascular medicine 2022, 9, 913609. [Google Scholar] [CrossRef]
- Kanekiyo, T.; Cirrito, J. R.; Liu, C. C.; Shinohara, M.; Li, J.; Schuler, D. R.; Shinohara, M.; Holtzman, D. M.; Bu, G. Neuronal clearance of amyloid-β by endocytic receptor LRP1. The Journal of neuroscience: the official journal of the Society for Neuroscience 2013, 33(49), 19276–19283. [Google Scholar] [CrossRef]
- Kang, J. H.; Grodstein, F. Postmenopausal hormone therapy, timing of initiation, APOE and cognitive decline. Neurobiology of aging 2012, 33(7), 1129–1137. [Google Scholar] [CrossRef]
- Kang, J. H.; Weuve, J.; Grodstein, F. Postmenopausal hormone therapy and risk of cognitive decline in community-dwelling aging women. Neurology 2004, 63(1), 101–107. [Google Scholar] [CrossRef] [PubMed]
- Kantarci, K.; Lowe, V. J.; Lesnick, T. G.; Tosakulwong, N.; Bailey, K. R.; Fields, J. A.; Shuster, L. T.; Zuk, S. M.; Senjem, M. L.; Mielke, M. M.; Gleason, C.; Jack, C. R.; Rocca, W. A.; Miller, V. M. Early Postmenopausal Transdermal 17β-Estradiol Therapy and Amyloid-β Deposition. Journal of Alzheimer's disease: JAD 2016, 53(2), 547–556. [Google Scholar] [CrossRef]
- Kaushik, S.; Cuervo, A. M. The coming of age of chaperone-mediated autophagy. Nature reviews. Molecular cell biology 2018, 19(6), 365–381. [Google Scholar] [CrossRef] [PubMed]
- Kermath, B. A.; Riha, P. D.; Woller, M. J.; Wolfe, A.; Gore, A. C. Hypothalamic molecular changes underlying natural reproductive senescence in the female rat. Endocrinology 2014, 155(9), 3597–3609. [Google Scholar] [CrossRef]
- Kerr, J. S.; Adriaanse, B. A.; Greig, N. H.; Mattson, M. P.; Cader, M. Z.; Bohr, V. A.; Fang, E. F. Mitophagy and Alzheimer's Disease: Cellular and Molecular Mechanisms. Trends in neurosciences 2017, 40(3), 151–166. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Jung, U. J.; Kim, S. R. The Crucial Role of the Blood-Brain Barrier in Neurodegenerative Diseases: Mechanisms of Disruption and Therapeutic Implications. Journal of clinical medicine 2025, 14(2), 386. [Google Scholar] [CrossRef]
- Kim, S. M.; Sultana, F.; Sims, S.; Gimenez-Roig, J.; Laurencin, V.; Pallapati, A.; Rojekar, S.; Frolinger, T.; Zhou, W.; Gumerova, A.; Macdonald, A.; Ryu, V.; Lizneva, D.; Korkmaz, F.; Yuen, T.; Zaidi, M. FSH, bone, belly and brain. The Journal of endocrinology 2024, 262(1), e230377. [Google Scholar] [CrossRef]
- Kim, Y. J.; Soto, M.; Branigan, G. L.; Rodgers, K.; Brinton, R. D. Association between menopausal hormone therapy and risk of neurodegenerative diseases: Implications for precision hormone therapy. Alzheimer's & dementia (New York, N. Y.) 2021, 7(1), e12174. [Google Scholar] [CrossRef]
- Knox, E. G.; Aburto, M. R.; Clarke, G.; Cryan, J. F.; O'Driscoll, C. M. The blood-brain barrier in aging and neurodegeneration. Molecular psychiatry 2022, 27(6), 2659–2673. [Google Scholar] [CrossRef]
- Kreisl, W. C.; Lyoo, C. H.; McGwier, M.; Snow, J.; Jenko, K. J.; Kimura, N.; Corona, W.; Morse, C. L.; Zoghbi, S. S.; Pike, V. W.; McMahon, F. J.; Turner, R. S.; Innis, R. B.; Biomarkers Consortium PET Radioligand Project Team. In vivo radioligand binding to translocator protein correlates with severity of Alzheimer's disease. Brain: a journal of neurology 2013, 136 Pt 7, 2228–2238. [Google Scholar] [CrossRef]
- Kim, S. M.; Sultana, F.; Sims, S.; Gimenez-Roig, J.; Laurencin, V.; Pallapati, A.; Rojekar, S.; Frolinger, T.; Zhou, W.; Gumerova, A.; Macdonald, A.; Ryu, V.; Lizneva, D.; Korkmaz, F.; Yuen, T.; Zaidi, M. FSH, bone, belly and brain. The Journal of endocrinology 2024, 262(1), e230377. [Google Scholar] [CrossRef]
- Krishnadas, N.; Doré, V.; Groot, C.; Lamb, F.; Bourgeat, P.; Burnham, S. C.; Huang, K.; Goh, A. M. Y.; Masters, C. L.; Villemagne, V. L.; Rowe, C. C.; AIBL research group. Mesial temporal tau in amyloid-β-negative cognitively normal older persons. Alzheimer's research & therapy 2022, 14(1), 51. [Google Scholar] [CrossRef]
- Kockx, M.; Traini, M.; Kritharides, L. Cell-specific production, secretion, and function of apolipoprotein E. Journal of molecular medicine (Berlin, Germany) 2018, 96(5), 361–371. [Google Scholar] [CrossRef] [PubMed]
- Kolnes, A. J.; Øystese, K. A. B.; Olarescu, N. C.; Ringstad, G.; Berg-Johnsen, J.; Casar-Borota, O.; Bollerslev, J.; Jørgensen, A. P. FSH Levels Are Related to E-cadherin Expression and Subcellular Location in Nonfunctioning Pituitary Tumors. The Journal of clinical endocrinology and metabolism 2020, 105(8), 2587–2594. [Google Scholar] [CrossRef] [PubMed]
- Kontaxi, C.; Piccardo, P.; Gill, A. C. Lysine-Directed Post-translational Modifications of Tau Protein in Alzheimer's Disease and Related Tauopathies. Frontiers in molecular biosciences 2017, 4, 56. [Google Scholar] [CrossRef] [PubMed]
- Koutsodendris, N.; Nelson, M. R.; Rao, A.; Huang, Y. Apolipoprotein E and Alzheimer’s disease: Findings, hypotheses, and potential mechanisms. Annual Review of Pathology: Mechanisms of Disease 17 2022, 73–99. [Google Scholar] [CrossRef]
- Korkmaz, F.; Sims, S.; Sen, F.; Sultana, F.; Laurencin, V.; Cullen, L.; Pallapati, A.; Liu, A.; Chen, R.; Rojekar, S.; Pevnev, G.; Cheliadinova, U.; Vasilyeva, D.; Burganova, G.; Macdonald, A.; Saxena, M.; Goosens, K.; Rosen, C. J.; Barak, O.; Lizneva, D.; Zaidi, M. Gene-dose-dependent reduction of Fshr expression improves spatial memory deficits in Alzheimer's mice. Molecular psychiatry 2025, 30(5), 2119–2126. [Google Scholar] [CrossRef] [PubMed]
- Laakkonen, E. K.; Karppinen, J. E.; Lehti, S.; Lee, E.; Pesonen, E.; Juppi, H. K.; Kujala, U. M.; Haapala, E. A.; Aukee, P.; Laukkanen, J. A.; Ihalainen, J. K. Associations of Sex Hormones and Hormonal Status With Arterial Stiffness in a Female Sample From Reproductive Years to Menopause. Frontiers in endocrinology 2021, 12, 765916. [Google Scholar] [CrossRef]
- La Joie, R.; Visani, A. V.; Lesman-Segev, O. H.; Baker, S. L.; Edwards, L.; Iaccarino, L.; Soleimani-Meigooni, D. N.; Mellinger, T.; Janabi, M.; Miller, Z. A.; Perry, D. C.; Pham, J.; Strom, A.; Gorno-Tempini, M. L.; Rosen, H. J.; Miller, B. L.; Jagust, W. J.; Rabinovici, G. D. Association of APOE4 and Clinical Variability in Alzheimer Disease With the Pattern of Tau- and Amyloid-PET. Neurology 2021, 96(5), e650–e661. [Google Scholar] [CrossRef]
- Lamontagne-Kam, D.; Ulfat, A. K.; Hervé, V.; Vu, T. M.; Brouillette, J. Implication of tau propagation on neurodegeneration in Alzheimer's disease. Frontiers in neuroscience 2023, 17, 1219299. [Google Scholar] [CrossRef]
- Laughlin, G. A.; Kritz-Silverstein, D.; Barrett-Connor, E. Endogenous oestrogens predict 4-year decline in verbal fluency in postmenopausal women: the Rancho Bernardo Study. Clinical endocrinology 2010, 72(1), 99–106. [Google Scholar] [CrossRef]
- Lee, J. H.; Yang, D. S.; Goulbourne, C. N.; Im, E.; Stavrides, P.; Pensalfini, A.; Chan, H.; Bouchet-Marquis, C.; Bleiwas, C.; Berg, M. J.; Huo, C.; Peddy, J.; Pawlik, M.; Levy, E.; Rao, M.; Staufenbiel, M.; Nixon, R. A. Faulty autolysosome acidification in Alzheimer's disease mouse models induces autophagic build-up of Aβ in neurons, yielding senile plaques. Nature neuroscience 2022, 25(6), 688–701. [Google Scholar] [CrossRef]
- Li, B.; Xie, Z.; Wang, M.; Nie, S.; Qian, Z.; Meng, X.; Liu, X.; Kang, S. S.; Ye, K. Neuronal C/EBPβ Shortens the Lifespan via Inactivating NAMPT. Advanced science (Weinheim, Baden-Wurttemberg, Germany) 2025, 12(21), e2414871. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Ling, Y.; Kuang, H.; 122. Research progress on FSH-FSHR signaling in the pathogenesis of non-reproductive diseases. Frontiers in cell and developmental biology 2024, 12, 1506450. [Google Scholar] [CrossRef]
- Li, R.; Wang, X.; He, P. The most prevalent rare coding variants of TREM2 conferring risk of Alzheimer's disease: A systematic review and meta-analysis. Experimental and therapeutic medicine 2021, 21(4), 347. [Google Scholar] [CrossRef] [PubMed]
- Li, J. W.; Zong, Y.; Cao, X. P.; Tan, L.; Tan, L. Microglial priming in Alzheimer's disease. Annals of translational medicine 2018, 6(10), 176. [Google Scholar] [CrossRef] [PubMed]
- Li, X; Fang, C; Li, Y; Xiong, X; Xu, X; Gu, L. Glycolytic reprogramming during microglial polarization in neurological diseases. Front Immunol 2025, 16, 1648887. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lin, Y.; Im, H.; Diem, L. T.; Ham, S. Characterizing the structural and thermodynamic properties of Aβ42 and Aβ40. Biochemical and biophysical research communications 2019, 510(3), 442–448. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Xia, X.; Wang, Y.; Zheng, J. C. Mitochondrial dysfunction in microglia: a novel perspective for pathogenesis of Alzheimer's disease. Journal of neuroinflammation 2022, 19(1), 248. [Google Scholar] [CrossRef]
- Lizneva, D.; Rahimova, A.; Kim, S. M.; Atabiekov, I.; Javaid, S.; Alamoush, B.; Taneja, C.; Khan, A.; Sun, L.; Azziz, R.; Yuen, T.; Zaidi, M. FSH Beyond Fertility. Frontiers in endocrinology 2019, 10, 136. [Google Scholar] [CrossRef] [PubMed]
- Lindner, K.; Gavin, A. C. Isoform- and cell-state-specific APOE homeostasis and function. Neural regeneration research 2024, 19(11), 2456–2466. [Google Scholar] [CrossRef]
- Lim, S. H. Y.; Hansen, M.; Kumsta, C. Molecular Mechanisms of Autophagy Decline during Aging. Cells 2024, 13(16), 1364. [Google Scholar] [CrossRef]
- Lish, A. M.; Grogan, E. F. L.; Benoit, C. R.; Pearse, R. V., 2nd; Heuer, S. E.; Luquez, T.; Orme, G. A.; Galle, P. C.; Milinkeviciute, G.; Green, K. N.; Alexander, K. D.; Fancher, S. B.; Stern, A. M.; Fujita, M.; Bennett, D. A.; Seyfried, N. T.; De Jager, P. L.; Menon, V.; Young-Pearse, T. L. CLU alleviates Alzheimer's disease-relevant processes by modulating astrocyte reactivity and microglia-dependent synaptic density. Neuron 2025, 113(12), 1925–1946.e11. [Google Scholar] [CrossRef] [PubMed]
- Liu, C. C.; Zhao, N.; Fu, Y.; Wang, N.; Linares, C.; Tsai, C. W.; Bu, G. ApoE4 Accelerates Early Seeding of Amyloid Pathology. Neuron 2017, 96(5), 1024–1032.e3. [Google Scholar] [CrossRef]
- Liu, C.; Liu, J.; Wang, Y. Y.; Xu, S. F.; Yu, L. M. APOE Lipoprotein Particles: Pathophysiology, Therapy, and the Crosstalk in Alzheimer's Disease and Cardiovascular Disease. Molecular neurobiology 2025, 63(1), 325. [Google Scholar] [CrossRef]
- Liu, H.; Zhao, X.; Chen, J.; Win, Y. Y.; Cai, J. Unnatural foldamers as inhibitors of Aβ aggregation via stabilizing the Aβ helix. Chemical communications (Cambridge, England) 2025, 61(24), 4586–4594. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Hao, M.; Zhang, J.; Chen, Z.; Zhou, J.; Wang, C.; Zhang, H.; Wang, J. FSHR-mTOR-HIF1 signaling alleviates mouse follicles from AMPK-induced atresia. Cell reports 2023, 42(10), 113158. [Google Scholar] [CrossRef]
- Liu, P. P.; Xie, Y.; Meng, X. Y.; Kang, J. S. History and progress of hypotheses and clinical trials for Alzheimer's disease. Signal transduction and targeted therapy 2019, 4, 29. [Google Scholar] [CrossRef]
- Lo, C. H.; Sachs, J. N. The role of wild-type tau in Alzheimer's disease and related tauopathies. Journal of life sciences (Westlake Village, Calif.) 2020, 2(4), 1–17. [Google Scholar] [CrossRef]
- Luo, H. B.; Xia, Y. Y.; Shu, X. J.; Liu, Z. C.; Feng, Y.; Liu, X. H.; Yu, G.; Yin, G.; Xiong, Y. S.; Zeng, K.; Jiang, J.; Ye, K.; Wang, X. C.; Wang, J. Z. SUMOylation at K340 inhibits tau degradation through deregulating its phosphorylation and ubiquitination. Proceedings of the National Academy of Sciences of the United States of America 2014, 111(46), 16586–16591. [Google Scholar] [CrossRef]
- Luo, X.; Liang, J.; Lei, X.; Sun, F.; Gong, M.; Liu, B.; Zhou, Z. C/EBPβ in Alzheimer's disease: An integrative regulator of pathological mechanisms. Brain research bulletin 2025, 221, 111198. [Google Scholar] [CrossRef]
- Lutsenko, S.; Roy, S.; Tsvetkov, P. Mammalian copper homeostasis: physiological roles and molecular mechanisms. Physiological reviews 2025, 105(1), 441–491. [Google Scholar] [CrossRef] [PubMed]
- Mahley, R. W.; Weisgraber, K. H.; Huang, Y. Apolipoprotein E: structure determines function, from atherosclerosis to Alzheimer's disease to AIDS. Journal of lipid research 2009, 50 Suppl(Suppl), S183–S188. [Google Scholar] [CrossRef]
- Ma, J.; Liu, C.; Yang, Y.; Yu, J.; Yang, J.; Yu, S.; Zhang, J.; Huang, L. C/EBPβ Acts Upstream of NF-κB P65 Subunit in Ox-LDL-Induced IL-1β Production by Macrophages. Cellular physiology and biochemistry: international journal of experimental cellular physiology, biochemistry, and pharmacology 2018, 48(4), 1605–1615. [Google Scholar] [CrossRef]
- Mak, E.; Bethlehem, R. A. I.; Romero-Garcia, R.; Cervenka, S.; Rittman, T.; Gabel, S.; Surendranathan, A.; Bevan-Jones, R. W.; Passamonti, L.; Vázquez Rodríguez, P.; Su, L.; Arnold, R.; Williams, G. B.; Hong, Y. T.; Fryer, T. D.; Aigbirhio, F. I.; Rowe, J. B.; O'Brien, J. T. In vivo coupling of tau pathology and cortical thinning in Alzheimer's disease. Alzheimer's & dementia (Amsterdam, Netherlands) 2018, 10, 678–687. [Google Scholar] [CrossRef]
- Maki, P. M.; Drogos, L. L.; Rubin, L. H.; Banuvar, S.; Shulman, L. P.; Geller, S. E. Objective hot flashes are negatively related to verbal memory performance in midlife women. Menopause (New York, N.Y.) 2008, 15(5), 848–856. [Google Scholar] [CrossRef] [PubMed]
- Malpetti, M.; Joie, R.; Rabinovici, G. D. Tau Beats Amyloid in Predicting Brain Atrophy in Alzheimer Disease: Implications for Prognosis and Clinical Trials. Journal of nuclear medicine: official publication, Society of Nuclear Medicine 2022, 63(6), 830–832. [Google Scholar] [CrossRef] [PubMed]
- Mathiisen, T. M.; Lehre, K. P.; Danbolt, N. C.; Ottersen, O. P. The perivascular astroglial sheath provides a complete covering of the brain microvessels: an electron microscopic 3D reconstruction. Glia 2010, 58(9), 1094–1103. [Google Scholar] [CrossRef] [PubMed]
- Mattson, M. P. Involvement of GABAergic interneuron dysfunction and neuronal network hyperexcitability in Alzheimer's disease: Amelioration by metabolic switching. International review of neurobiology 2020, 154, 191–205. [Google Scholar] [CrossRef]
- Melville, M.; He, L.; Desai, R.; Nyamayaro, P.; Fox, C.; Kothari, K. U.; Condron, P.; Miao, M.; Hickey, M.; Spector, A. Menopause hormone therapy and risk of mild cognitive impairment or dementia: a systematic review and meta-analysis. The lancet. Healthy longevity 2025, 6(12), 100803. [Google Scholar] [CrossRef]
- McClure, C.; McPeak, M. B.; Youssef, D.; Yao, Z. Q.; McCall, C. E.; El Gazzar, M. Stat3 and C/EBPβ synergize to induce miR-21 and miR-181b expression during sepsis. Immunology and cell biology 2017, 95(1), 42–55. [Google Scholar] [CrossRef] [PubMed]
- McCoy, N. D.; Gawrys, S. P.; Mackintosh, S. G.; et al. Ovarian somatic tissue rejuvenates circulating apolipoproteins and promotes cognitive health in postreproductive female mice. In GeroScience; 2025. [Google Scholar] [CrossRef]
- Milkovic, L.; Zarkovic, N.; Marusic, Z.; Zarkovic, K.; Jaganjac, M. The 4-Hydroxynonenal-Protein Adducts and Their Biological Relevance: Are Some Proteins Preferred Targets? Antioxidants (Basel, Switzerland) 2023, 12(4), 856. [Google Scholar] [CrossRef]
- Milner, T. A.; Ayoola, K.; Drake, C. T.; Herrick, S. P.; Tabori, N. E.; McEwen, B. S.; Warrier, S.; Alves, S. E. Ultrastructural localization of estrogen receptor beta immunoreactivity in the rat hippocampal formation. The Journal of comparative neurology 2005, 491(2), 81–95. [Google Scholar] [CrossRef] [PubMed]
- MohanKumar, S. M. J.; Balasubramanian, P.; Subramanian, M.; MohanKumar, P. S. Chronic estradiol exposure - harmful effects on behavior, cardiovascular and reproductive functions. Reproduction (Cambridge, England) 2018, 156(5), R169–R186. [Google Scholar] [CrossRef] [PubMed]
- Monterey, M. D.; Wei, H.; Wu, X.; Wu, J. Q. The Many Faces of Astrocytes in Alzheimer's Disease. Frontiers in neurology 2021, 12, 619626. [Google Scholar] [CrossRef]
- Monsorno, K.; Ginggen, K.; Ivanov, A.; Buckinx, A.; Lalive, A. L.; Tchenio, A.; Benson, S.; Vendrell, M.; D'Alessandro, A.; Beule, D.; Pellerin, L.; Mameli, M.; Paolicelli, R. C. Loss of microglial MCT4 leads to defective synaptic pruning and anxiety-like behavior in mice. Nature communications 2023, 14(1), 5749. [Google Scholar] [CrossRef] [PubMed]
- Morris, J. C.; Roe, C. M.; Xiong, C.; Fagan, A. M.; Goate, A. M.; Holtzman, D. M.; Mintun, M. A. APOE predicts amyloid-beta but not tau Alzheimer pathology in cognitively normal aging. Annals of neurology 2010, 67(1), 122–131. [Google Scholar] [CrossRef] [PubMed]
- Mosconi, L.; Andy, C.; Nerattini, M.; et al. Systematic Review and Meta-analysis of Menopause Hormone Therapy (MHT) and the Risk of Alzheimer’s Disease and All-cause Dementia: Effects of MHT Characteristics, Location, and APOE-4 Status. Curr Obstet Gynecol Rep 2025, 14, 6. [Google Scholar] [CrossRef]
- Mosconi, L.; Rahman, A.; Diaz, I.; Wu, X.; Scheyer, O.; Hristov, H. W.; Vallabhajosula, S.; Isaacson, R. S.; de Leon, M. J.; Brinton, R. D. Increased Alzheimer's risk during the menopause transition: A 3-year longitudinal brain imaging study. PloS one 2018, 13(12), e0207885. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, Y. A. Reproductive aging in biological females: mechanisms and immediate consequences. Frontiers in endocrinology 2025, 16, 1658592. [Google Scholar] [CrossRef]
- Nerattini, M.; Jett, S.; Andy, C.; Carlton, C.; Zarate, C.; Boneu, C.; Battista, M.; Pahlajani, S.; Loeb-Zeitlin, S.; Havryulik, Y.; Williams, S.; Christos, P.; Fink, M.; Brinton, R. D.; Mosconi, L. Systematic review and meta-analysis of the effects of menopause hormone therapy on risk of Alzheimer's disease and dementia. Frontiers in aging neuroscience 2023, 15, 1260427. [Google Scholar] [CrossRef] [PubMed]
- Neth, B. J.; Mintz, A.; Whitlow, C.; Jung, Y.; Solingapuram Sai, K.; Register, T. C.; Kellar, D.; Lockhart, S. N.; Hoscheidt, S.; Maldjian, J.; Heslegrave, A. J.; Blennow, K.; Cunnane, S. C.; Castellano, C. A.; Zetterberg, H.; Craft, S. Modified ketogenic diet is associated with improved cerebrospinal fluid biomarker profile, cerebral perfusion, and cerebral ketone body uptake in older adults at risk for Alzheimer's disease: a pilot study. Neurobiology of aging 2020, 86, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Nixon, R. A.; Wegiel, J.; Kumar, A.; Yu, W. H.; Peterhoff, C.; Cataldo, A.; Cuervo, A. M. Extensive involvement of autophagy in Alzheimer disease: an immuno-electron microscopy study. Journal of neuropathology and experimental neurology 2005, 64(2), 113–122. [Google Scholar] [CrossRef] [PubMed]
- Nixon, R. A.; Yang, D. S. Autophagy failure in Alzheimer's disease--locating the primary defect. Neurobiology of disease 2011, 43(1), 38–45. [Google Scholar] [CrossRef] [PubMed]
- Noble, W.; Hanger, D. P.; Miller, C. C.; Lovestone, S. The importance of tau phosphorylation for neurodegenerative diseases. Frontiers in neurology 2013, 4, 83. [Google Scholar] [CrossRef] [PubMed]
- Nordengen, K.; Kirsebom, B. E.; Henjum, K.; Selnes, P.; Gísladóttir, B.; Wettergreen, M.; Torsetnes, S. B.; Grøntvedt, G. R.; Waterloo, K. K.; Aarsland, D.; Nilsson, L. N. G.; Fladby, T. Glial activation and inflammation along the Alzheimer's disease continuum. Journal of neuroinflammation 2019, 16(1), 46. [Google Scholar] [CrossRef]
- Oliveira, T. P. D.; Morais, A. L. B.; Dos Reis, P. L. B.; Palotás, A.; Vieira, L. B. A Potential Role for the Ketogenic Diet in Alzheimer's Disease Treatment: Exploring Pre-Clinical and Clinical Evidence. Metabolites 2023, 14(1), 25. [Google Scholar] [CrossRef]
- Okello, A.; Edison, P.; Archer, H. A.; Turkheimer, F. E.; Kennedy, J.; Bullock, R.; Walker, Z.; Kennedy, A.; Fox, N.; Rossor, M.; Brooks, D. J. Microglial activation and amyloid deposition in mild cognitive impairment: a PET study. Neurology 2009, 72(1), 56–62. [Google Scholar] [CrossRef]
- Oku, M.; Sakai, Y. Three Distinct Types of Microautophagy Based on Membrane Dynamics and Molecular Machineries. BioEssays: news and reviews in molecular, cellular and developmental biology 2018, 40(6), e1800008. [Google Scholar] [CrossRef]
- Oriá, R. B.; de Almeida, J. Z.; Moreira, C. N.; Guerrant, R. L.; Figueiredo, J. R. Apolipoprotein E Effects on Mammalian Ovarian Steroidogenesis and Human Fertility. Trends in endocrinology and metabolism: TEM 2020, 31(11), 872–883. [Google Scholar] [CrossRef]
- Ossenkoppele, R.; Lyoo, C. H.; Sudre, C. H.; van Westen, D.; Cho, H.; Ryu, Y. H.; Choi, J. Y.; Smith, R.; Strandberg, O.; Palmqvist, S.; Westman, E.; Tsai, R.; Kramer, J.; Boxer, A. L.; Gorno-Tempini, M. L.; La Joie, R.; Miller, B. L.; Rabinovici, G. D.; Hansson, O. Distinct tau PET patterns in atrophy-defined subtypes of Alzheimer's disease. Alzheimer's & dementia: the journal of the Alzheimer's Association 2020, 16(2), 335–344. [Google Scholar] [CrossRef]
- Ossenkoppele, R.; Smith, R.; Mattsson-Carlgren, N.; Groot, C.; Leuzy, A.; Strandberg, O.; Palmqvist, S.; Olsson, T.; Jögi, J.; Stormrud, E.; Cho, H.; Ryu, Y. H.; Choi, J. Y.; Boxer, A. L.; Gorno-Tempini, M. L.; Miller, B. L.; Soleimani-Meigooni, D.; Iaccarino, L.; La Joie, R.; Baker, S.; Hansson, O. Accuracy of Tau Positron Emission Tomography as a Prognostic Marker in Preclinical and Prodromal Alzheimer Disease: A Head-to-Head Comparison Against Amyloid Positron Emission Tomography and Magnetic Resonance Imaging. JAMA neurology 2021, 78(8), 961–971. [Google Scholar] [CrossRef] [PubMed]
- Parbo, P.; Ismail, R.; Hansen, K. V.; Amidi, A.; Mårup, F. H.; Gottrup, H.; Brændgaard, H.; Eriksson, B. O.; Eskildsen, S. F.; Lund, T. E.; Tietze, A.; Edison, P.; Pavese, N.; Stokholm, M. G.; Borghammer, P.; Hinz, R.; Aanerud, J.; Brooks, D. J. Brain inflammation accompanies amyloid in the majority of mild cognitive impairment cases due to Alzheimer's disease. Brain: a journal of neurology 2017, 140(7), 2002–2011. [Google Scholar] [CrossRef] [PubMed]
- Parcon, P. A.; Balasubramaniam, M.; Ayyadevara, S.; Jones, R. A.; Liu, L.; Shmookler Reis, R. J.; Barger, S. W.; Mrak, R. E.; Griffin, W. S. T. Apolipoprotein E4 inhibits autophagy gene products through direct, specific binding to CLEAR motifs. Alzheimer's & dementia: the journal of the Alzheimer's Association 2018, 14(2), 230–242. [Google Scholar] [CrossRef]
- Pallapati, A. R.; Korkmaz, F.; Rojekar, S.; Sims, S.; Misra, A.; Gimenez-Roig, J.; Gangadhar, A.; Laurencin, V.; Gumerova, A.; Cheliadinova, U.; Sultana, F.; Vasilyeva, D.; Cullen, L.; Schuermann, J.; Munitz, J.; Kannangara, H.; Parte, S.; Pevnev, G.; Burganova, G.; Tumoglu, Z.; Zaidi, M. Efficacy and safety of a therapeutic humanized FSH-blocking antibody in obesity and Alzheimer's disease models. The Journal of clinical investigation 2025, 135(17), e182702. [Google Scholar] [CrossRef]
- Palmer, J. E.; Wilson, N.; Son, S. M.; Obrocki, P.; Wrobel, L.; Rob, M.; Takla, M.; Korolchuk, V. I.; Rubinsztein, D. C. Autophagy, aging, and age-related neurodegeneration. Neuron 2025, 113(1), 29–48. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.; Jeon, W. K.; Bizon, J. L.; Han, J. S. Interaction of basal forebrain cholinergic neurons with the glucocorticoid system in stress regulation and cognitive impairment. Frontiers in aging neuroscience 2015, 7, 43. [Google Scholar] [CrossRef] [PubMed]
- Paul, S. M.; Yohn, S. E.; Popiolek, M.; Miller, A. C.; Felder, C. C. Muscarinic Acetylcholine Receptor Agonists as Novel Treatments for Schizophrenia. The American journal of psychiatry 2022, 179(9), 611–627. [Google Scholar] [CrossRef]
- Pearson, H. A.; Peers, C. Physiological roles for amyloid beta peptides. The Journal of physiology 2006, 575 Pt 1, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Phillips, M. C. L.; Deprez, L. M.; Mortimer, G. M. N.; Murtagh, D. K. J.; McCoy, S.; Mylchreest, R.; Gilbertson, L. J.; Clark, K. M.; Simpson, P. V.; McManus, E. J.; Oh, J. E.; Yadavaraj, S.; King, V. M.; Pillai, A.; Romero-Ferrando, B.; Brinkhuis, M.; Copeland, B. M.; Samad, S.; Liao, S.; Schepel, J. A. C. Randomized crossover trial of a modified ketogenic diet in Alzheimer's disease. Alzheimer's research & therapy 2021, 13(1), 51. [Google Scholar] [CrossRef]
- Piccarducci, R.; Giacomelli, C.; Bertilacchi, M. S.; Benito-Martinez, A.; Di Giorgi, N.; Daniele, S.; Signore, G.; Rocchiccioli, S.; Vilar, M.; Marchetti, L.; Martini, C. Apolipoprotein E ε4 triggers neurotoxicity via cholesterol accumulation, acetylcholine dyshomeostasis, and PKCε mislocalization in cholinergic neuronal cells. Biochimica et biophysica acta. Molecular basis of disease 2023, 1869(7), 166793. [Google Scholar] [CrossRef] [PubMed]
- Pires, M.; Rego, A. C. Apoe4 and Alzheimer's Disease Pathogenesis-Mitochondrial Deregulation and Targeted Therapeutic Strategies. International journal of molecular sciences 2023, 24(1), 778. [Google Scholar] [CrossRef] [PubMed]
- Planche, V.; Manjon, J. V.; Mansencal, B.; Lanuza, E.; Tourdias, T.; Catheline, G.; Coupé, P. Structural progression of Alzheimer's disease over decades: the MRI staging scheme. Brain communications 2022, 4(3), fcac109. [Google Scholar] [CrossRef] [PubMed]
- Pourhadi, N.; Mørch, L. S.; Holm, E. A.; Torp-Pedersen, C.; Meaidi, A. Menopausal hormone therapy and dementia: nationwide, nested case-control study. BMJ (Clinical research ed.) 2023, 381, e072770. [Google Scholar] [CrossRef]
- Price, M. S.; Rastegari, E.; Gupta, R.; Vo, K.; Moore, T. I.; Venkatachalam, K. Intracellular lactate dynamics in Drosophila neurons. iScience 2025, 28(10), 113462. [Google Scholar] [CrossRef] [PubMed]
- Qi, G.; Mi, Y.; Shi, X.; Gu, H.; Brinton, R. D.; Yin, F. ApoE4 Impairs Neuron-Astrocyte Coupling of Fatty Acid Metabolism. Cell reports 2021, 34(1), 108572. [Google Scholar] [CrossRef]
- Quick, J. D.; Silva, C.; Wong, J. H.; Lim, K. L.; Reynolds, R.; Barron, A. M.; Zeng, J.; Lo, C. H. Lysosomal acidification dysfunction in microglia: an emerging pathogenic mechanism of neuroinflammation and neurodegeneration. Journal of neuroinflammation 2023, 20(1), 185. [Google Scholar] [CrossRef]
- Rahman, A.; Jackson, H.; Hristov, H.; Isaacson, R. S.; Saif, N.; Shetty, T.; Etingin, O.; Henchcliffe, C.; Brinton, R. D.; Mosconi, L. Sex and Gender Driven Modifiers of Alzheimer's: The Role for Estrogenic Control Across Age, Race, Medical, and Lifestyle Risks. Frontiers in aging neuroscience 2019, 11, 315. [Google Scholar] [CrossRef]
- Rauchmann, B. S.; Brendel, M.; Franzmeier, N.; Trappmann, L.; Zaganjori, M.; Ersoezlue, E.; Morenas-Rodriguez, E.; Guersel, S.; Burow, L.; Kurz, C.; Haeckert, J.; Tatò, M.; Utecht, J.; Papazov, B.; Pogarell, O.; Janowitz, D.; Buerger, K.; Ewers, M.; Palleis, C.; Weidinger, E.; Perneczky, R. Microglial Activation and Connectivity in Alzheimer Disease and Aging. Annals of neurology 2022, 92(5), 768–781. [Google Scholar] [CrossRef]
- Reddy, K.; Cusack, C. L.; Nnah, I. C.; Khayati, K.; Saqcena, C.; Huynh, T. B.; Noggle, S. A.; Ballabio, A.; Dobrowolski, R. Dysregulation of Nutrient Sensing and CLEARance in Presenilin Deficiency. Cell reports 2016, 14(9), 2166–2179. [Google Scholar] [CrossRef]
- Riedel, B. C.; Thompson, P. M.; Brinton, R. D. Age, APOE and sex: Triad of risk of Alzheimer's disease. The Journal of steroid biochemistry and molecular biology 2016, 160, 134–147. [Google Scholar] [CrossRef] [PubMed]
- Riku, Y.; Brion, J. P.; Ando, K.; Uchihara, T.; Iwasaki, Y. The Determinant of Tau Spreading in Alzheimer's Disease: Dependent on Senile Plaque, Neural Circuits, or Spatial Proximity? International journal of molecular sciences 2025, 26(24), 12088. [Google Scholar] [CrossRef]
- Robbins, M.; Clayton, E.; Kaminski Schierle, G. S. Synaptic tau: A pathological or physiological phenomenon? Acta neuropathologica communications 2021, 9(1), 149. [Google Scholar] [CrossRef]
- Rocca, M. S.; Pannella, M.; Bayraktar, E.; Marino, S.; Bortolozzi, M.; Di Nisio, A.; Foresta, C.; Ferlin, A. Extragonadal function of follicle-stimulating hormone: Evidence for a role in endothelial physiology and dysfunction. Molecular and cellular endocrinology 2024, 594, 112378. [Google Scholar] [CrossRef] [PubMed]
- Rocca, W. A.; Kantarci, K.; Faubion, S. S. Risks and benefits of hormone therapy after menopause for cognitive decline and dementia: A conceptual review. Maturitas 2024, 184, 108003. [Google Scholar] [CrossRef] [PubMed]
- Roda, A. R.; Serra-Mir, G.; Montoliu-Gaya, L.; Tiessler, L.; Villegas, S. Amyloid-beta peptide and tau protein crosstalk in Alzheimer's disease. Neural regeneration research 2022, 17(8), 1666–1674. [Google Scholar] [CrossRef] [PubMed]
- Roe, J. M.; Vidal-Piñeiro, D.; Sørensen, Ø.; Grydeland, H.; Leonardsen, E. H.; Iakunchykova, O.; Pan, M.; Mowinckel, A.; Strømstad, M.; Nawijn, L.; Milaneschi, Y.; Andersson, M.; Pudas, S.; Bråthen, A. C. S.; Kransberg, J.; Falch, E. S.; Øverbye, K.; Kievit, R. A.; Ebmeier, K. P.; Lindenberger, U.; Wang, Y. Brain change trajectories in healthy adults correlate with Alzheimer's related genetic variation and memory decline across life. Nature communications 2024, 15(1), 10651. [Google Scholar] [CrossRef]
- Roelofs, E. J.; Dengel, D. R.; Wang, Q.; Hodges, J. S.; Steinberger, J.; Baker, K. S. The Role of Follicle-stimulating Hormone in Vascular Dysfunction Observed in Hematopoietic Cell Transplant Recipients. Journal of pediatric hematology/oncology 2022, 44(3), e695–e700. [Google Scholar] [CrossRef]
- Rosu, G. C.; Catalin, B.; Balseanu, T. A.; Laurentiu, M.; Claudiu, M.; Kumar-Singh, S.; Daniel, P. Inhibition of Aquaporin 4 Decreases Amyloid Aβ40 Drainage Around Cerebral Vessels. Molecular neurobiology 2020, 57(11), 4720–4734. [Google Scholar] [CrossRef]
- Ruiz, J.; Kouiavskaia, D.; Migliorini, M.; Robinson, S.; Saenko, E. L.; Gorlatova, N.; Li, D.; Lawrence, D.; Hyman, B. T.; Weisgraber, K. H.; Strickland, D. K. The apoE isoform binding properties of the VLDL receptor reveal marked differences from LRP and the LDL receptor. Journal of lipid research 2005, 46(8), 1721–1731. [Google Scholar] [CrossRef]
- Rutherford, S.; Fraza, C.; Dinga, R.; Kia, S. M.; Wolfers, T.; Zabihi, M.; Berthet, P.; Worker, A.; Verdi, S.; Andrews, D.; Han, L. K.; Bayer, J. M.; Dazzan, P.; McGuire, P.; Mocking, R. T.; Schene, A.; Sripada, C.; Tso, I. F.; Duval, E. R.; Chang, S. E.; Marquand, A. F. Charting brain growth and aging at high spatial precision. eLife 2022, 11, e72904. [Google Scholar] [CrossRef] [PubMed]
- Saleh, R. N. M.; Hornberger, M.; Ritchie, C. W.; Minihane, A. M. Hormone replacement therapy is associated with improved cognition and larger brain volumes in at-risk APOE4 women: results from the European Prevention of Alzheimer's Disease (EPAD) cohort. Alzheimer's research & therapy 2023, 15(1), 10. [Google Scholar] [CrossRef]
- Schmitz, T. W.; Mur, M.; Aghourian, M.; Bedard, M. A.; Spreng, R. N.; Alzheimer’s Disease Neuroimaging Initiative. Longitudinal Alzheimer's Degeneration Reflects the Spatial Topography of Cholinergic Basal Forebrain Projections. Cell reports 2018, 24(1), 38–46. [Google Scholar] [CrossRef]
- Schwabe, M. R.; Fleischer, A. W.; Kuehn, R. K.; Chaudhury, S.; York, J. M.; Sem, D. S.; Donaldson, W. A.; LaDu, M. J.; Frick, K. M. The novel estrogen receptor beta agonist EGX358 and APOE genotype influence memory, vasomotor, and anxiety outcomes in an Alzheimer's mouse model. Frontiers in aging neuroscience 2024, 16, 1477045. [Google Scholar] [CrossRef]
- Sengupta, U.; Kayed, R. Amyloid β, Tau, and α-Synuclein aggregates in the pathogenesis, prognosis, and therapeutics for neurodegenerative diseases. Progress in neurobiology 2022, 214, 102270. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Pozo, A.; Frosch, M. P.; Masliah, E.; Hyman, B. T. Neuropathological alterations in Alzheimer disease. Cold Spring Harbor perspectives in medicine 2011, 1(1), a006189. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Pozo, A.; Das, S.; Hyman, B. T. APOE and Alzheimer's disease: advances in genetics, pathophysiology, and therapeutic approaches. The Lancet. Neurology 2021, 20(1), 68–80. [Google Scholar] [CrossRef] [PubMed]
- Sierra-Magro, A.; Bartolome, F.; Lozano-Muñoz, D.; Alarcón-Gil, J.; Gine, E.; Sanz-SanCristobal, M.; Alonso-Gil, S.; Cortes-Canteli, M.; Carro, E.; Pérez-Castillo, A.; Morales-García, J. A. C/EBPβ Regulates TFAM Expression, Mitochondrial Function and Autophagy in Cellular Models of Parkinson's Disease. International journal of molecular sciences 2023, 24(2), 1459. [Google Scholar] [CrossRef]
- Shaerzadeh, F.; Motamedi, F.; Khodagholi, F. Inhibition of akt phosphorylation diminishes mitochondrial biogenesis regulators, tricarboxylic acid cycle activity and exacerbates recognition memory deficit in rat model of Alzheimer's disease. Cellular and molecular neurobiology 2014, 34(8), 1223–1233. [Google Scholar] [CrossRef]
- Shaikh, S. B.; Nicholson, L. F. Effects of chronic low dose rotenone treatment on human microglial cells. Molecular neurodegeneration 2009, 4, 55. [Google Scholar] [CrossRef]
- Shen, J.; Kelleher, R. J., 3rd. The presenilin hypothesis of Alzheimer's disease: evidence for a loss-of-function pathogenic mechanism. Proceedings of the National Academy of Sciences of the United States of America 2007, 104(2), 403–409. [Google Scholar] [CrossRef]
- Shete, N.; Calabrese, J.; Tonetti, D. A. Revisiting Estrogen for the Treatment of Endocrine-Resistant Breast Cancer: Novel Therapeutic Approaches. Cancers 2023, 15(14), 3647. [Google Scholar] [CrossRef]
- Short, R. A.; Bowen, R. L.; O'Brien, P. C.; Graff-Radford, N. R. Elevated gonadotropin levels in patients with Alzheimer disease. Mayo Clinic proceedings 2001, 76(9), 906–909. [Google Scholar] [CrossRef]
- Shughrue, P. J.; Lane, M. V.; Scrimo, P. J.; Merchenthaler, I. Comparative distribution of estrogen receptor-alpha (ER-alpha) and beta (ER-beta) mRNA in the rat pituitary, gonad, and reproductive tract. Steroids 1998, 63(10), 498–504. [Google Scholar] [CrossRef] [PubMed]
- Shumaker, S. A.; Legault, C.; Rapp, S. R.; Thal, L.; Wallace, R. B.; Ockene, J. K.; Hendrix, S. L.; Jones, B. N., 3rd; Assaf, A. R.; Jackson, R. D.; Kotchen, J. M.; Wassertheil-Smoller, S.; Wactawski-Wende, J.; WHIMS Investigators. Estrogen plus progestin and the incidence of dementia and mild cognitive impairment in postmenopausal women: the Women's Health Initiative Memory Study: a randomized controlled trial. JAMA 2003, 289(20), 2651–2662. [Google Scholar] [CrossRef] [PubMed]
- Shumaker, S. A.; Legault, C.; Kuller, L.; Rapp, S. R.; Thal, L.; Lane, D. S.; Fillit, H.; Stefanick, M. L.; Hendrix, S. L.; Lewis, C. E.; Masaki, K.; Coker, L. H.; Women's Health Initiative Memory Study. Conjugated equine estrogens and incidence of probable dementia and mild cognitive impairment in postmenopausal women: Women's Health Initiative Memory Study. JAMA 2004, 291(24), 2947–2958. [Google Scholar] [CrossRef]
- Silva, I.; Silva, J.; Ferreira, R.; Trigo, D. Glymphatic system, AQP4, and their implications in Alzheimer's disease. Neurological research and practice 2021, 3(1), 5. [Google Scholar] [CrossRef]
- Sinha, S.; Lieberburg, I. Cellular mechanisms of beta-amyloid production and secretion. Proceedings of the National Academy of Sciences of the United States of America 1999, 96(20), 11049–11053. [Google Scholar] [CrossRef] [PubMed]
- Sirois, J.; Richards, J. S. Transcriptional regulation of the rat prostaglandin endoperoxide synthase 2 gene in granulosa cells. Evidence for the role of a cis-acting C/EBP beta promoter element. The Journal of biological chemistry 1993, 268(29), 21931–21938. [Google Scholar] [CrossRef] [PubMed]
- Singleton, E.; Hansson, O.; Pijnenburg, Y. A. L.; La Joie, R.; Mantyh, W. G.; Tideman, P.; Stomrud, E.; Leuzy, A.; Johansson, M.; Strandberg, O.; Smith, R.; Berendrecht, E.; Miller, B. L.; Iaccarino, L.; Edwards, L.; Strom, A.; Wolters, E. E.; Coomans, E.; Visser, D.; Golla, S. S. V.; Ossenkoppele, R. Heterogeneous distribution of tau pathology in the behavioural variant of Alzheimer's disease. In Journal of neurology, neurosurgery, and psychiatry; Advance online publication, 2021; Volume 92, 8, pp. 872–880. [Google Scholar] [CrossRef]
- Sinsky, J.; Pichlerova, K.; Hanes, J. Tau Protein Interaction Partners and Their Roles in Alzheimer's Disease and Other Tauopathies. International journal of molecular sciences 2021, 22(17), 9207. [Google Scholar] [CrossRef] [PubMed]
- Sochocka, M.; Diniz, B. S.; Leszek, J. Inflammatory Response in the CNS: Friend or Foe? Molecular neurobiology 2017, 54(10), 8071–8089. [Google Scholar] [CrossRef] [PubMed]
- Smitz, J. E.; Cortvrindt, R. G. The earliest stages of folliculogenesis in vitro. Reproduction (Cambridge, England) 2002, 123(2), 185–202. [Google Scholar] [CrossRef]
- Spina, E.; Ferrari, R. R.; Pellegrini, E.; Colombo, M.; Poloni, T. E.; Guaita, A.; Davin, A. Mitochondrial Alterations, Oxidative Stress, and Therapeutic Implications in Alzheimer's Disease: A Narrative Review. Cells 2025, 14(3), 229. [Google Scholar] [CrossRef]
- Stanciu, G. D.; Luca, A.; Rusu, R. N.; Bild, V.; Beschea Chiriac, S. I.; Solcan, C.; Bild, W.; Ababei, D. C. Alzheimer's Disease Pharmacotherapy in Relation to Cholinergic System Involvement. Biomolecules 2019, 10(1), 40. [Google Scholar] [CrossRef] [PubMed]
- Stanciu, G. D.; Luca, A.; Rusu, R. N.; Bild, V.; Beschea Chiriac, S. I.; Solcan, C.; Bild, W.; Ababei, D. C. Alzheimer's Disease Pharmacotherapy in Relation to Cholinergic System Involvement. Biomolecules 2019, 10(1), 40. [Google Scholar] [CrossRef] [PubMed]
- Staurenghi, E.; Giannelli, S.; Testa, G.; Sottero, B.; Leonarduzzi, G.; Gamba, P. Cholesterol Dysmetabolism in Alzheimer's Disease: A Starring Role for Astrocytes? Antioxidants (Basel, Switzerland) 2021, 10(12), 1890. [Google Scholar] [CrossRef]
- Stefaniak, O.; Dobrzyńska, M.; Drzymała-Czyż, S.; Przysławski, J. Diet in the Prevention of Alzheimer's Disease: Current Knowledge and Future Research Requirements. Nutrients 2022, 14(21), 4564. [Google Scholar] [CrossRef]
- Stuchell-Brereton, M. D.; Zimmerman, M. I.; Miller, J. J.; Mallimadugula, U. L.; Incicco, J. J.; Roy, D.; Smith, L. G.; Cubuk, J.; Baban, B.; DeKoster, G. T.; Frieden, C.; Bowman, G. R.; Soranno, A. Apolipoprotein E4 has extensive conformational heterogeneity in lipid-free and lipid-bound forms. Proceedings of the National Academy of Sciences of the United States of America 2023, 120(7), e2215371120. [Google Scholar] [CrossRef] [PubMed]
- St-Onge, F.; Chapleau, M.; Breitner, J. C.; Villeneuve, S.; Binette, A. P. Tau accumulation and its spatial progression across the Alzheimer's disease spectrum. medRxiv: the preprint server for health sciences 2023, 23290880. [Google Scholar] [CrossRef] [PubMed]
- Stouffer, K. M.; Chen, C.; Kulason, S.; Xu, E.; Witter, M. P.; Ceritoglu, C.; Albert, M. S.; Mori, S.; Troncoso, J.; Tward, D. J.; Miller, M. I.; Alzheimer’s Disease Neuroimaging Initiative. Early amygdala and ERC atrophy linked to 3D reconstruction of rostral neurofibrillary tau tangle pathology in Alzheimer's disease. NeuroImage. Clinical 2023, 38, 103374. [Google Scholar] [CrossRef]
- Straccia, M.; Gresa-Arribas, N.; Dentesano, G.; Ejarque-Ortiz, A.; Tusell, J. M.; Serratosa, J.; Solà, C.; Saura, J. Pro-inflammatory gene expression and neurotoxic effects of activated microglia are attenuated by absence of CCAAT/enhancer binding protein β. Journal of neuroinflammation 2011, 8, 156. [Google Scholar] [CrossRef]
- Suresh, S.; Singh S, A.; Rushendran, R.; Vellapandian, C.; Prajapati, B. Alzheimer's disease: the role of extrinsic factors in its development, an investigation of the environmental enigma. Frontiers in neurology 2023, 14, 1303111. [Google Scholar] [CrossRef]
- Song, Y. J.; Li, S. R.; Li, X. W.; Chen, X.; Wei, Z. X.; Liu, Q. S.; Cheng, Y. The Effect of Estrogen Replacement Therapy on Alzheimer's Disease and Parkinson's Disease in Postmenopausal Women: A Meta-Analysis. Frontiers in neuroscience 2020, 14, 157. [Google Scholar] [CrossRef]
- Sung, Y. F.; Tsai, C. T.; Kuo, C. Y.; Lee, J. T.; Chou, C. H.; Chen, Y. C.; Chou, Y. C.; Sun, C. A. Use of Hormone Replacement Therapy and Risk of Dementia: A Nationwide Cohort Study. Neurology 2022, 99(17), e1835–e1842. [Google Scholar] [CrossRef]
- Sun, Y. Y.; Wang, Z.; Huang, H. C. Roles of ApoE4 on the Pathogenesis in Alzheimer's Disease and the Potential Therapeutic Approaches. Cellular and molecular neurobiology 2023, 43(7), 3115–3136. [Google Scholar] [CrossRef]
- Song, Z. H.; Yu, H. Y.; Wang, P.; Mao, G. K.; Liu, W. X.; Li, M. N.; Wang, H. N.; Shang, Y. L.; Liu, C.; Xu, Z. L.; Sun, Q. Y.; Li, W. Germ cell-specific Atg7 knockout results in primary ovarian insufficiency in female mice. Cell death & disease 2015, 6(1), e1589. [Google Scholar] [CrossRef]
- Sweeney, M. D.; Montagne, A.; Sagare, A. P.; Nation, D. A.; Schneider, L. S.; Chui, H. C.; Harrington, M. G.; Pa, J.; Law, M.; Wang, D. J. J.; Jacobs, R. E.; Doubal, F. N.; Ramirez, J.; Black, S. E.; Nedergaard, M.; Benveniste, H.; Dichgans, M.; Iadecola, C.; Love, S.; Bath, P. M.; Zlokovic, B. V. Vascular dysfunction-The disregarded partner of Alzheimer's disease. Alzheimer's & dementia: the journal of the Alzheimer's Association 2019, 15(1), 158–167. [Google Scholar] [CrossRef]
- Tate, M.; Wijeratne, H. R. S.; Kim, B.; Philtjens, S.; You, Y.; Lee, D. H.; Gutierrez, D. A.; Sharify, D.; Wells, M.; Perez-Cardelo, M.; Doud, E. H.; Fernandez-Hernando, C.; Lasagna-Reeves, C.; Mosley, A. L.; Kim, J. Deletion of miR-33, a regulator of the ABCA1-APOE pathway, ameliorates neuropathological phenotypes in APP/PS1 mice. Alzheimer's & dementia: the journal of the Alzheimer's Association 2024, 20(11), 7805–7818. [Google Scholar] [CrossRef]
- Taxier, L. R.; Philippi, S. M.; Fleischer, A. W.; York, J. M.; LaDu, M. J.; Frick, K. M. APOE4 homozygote females are resistant to the beneficial effects of 17β-estradiol on memory and CA1 dendritic spine density in the EFAD mouse model of Alzheimer's disease. Neurobiology of aging 2022a, 118, 13–24. [Google Scholar] [CrossRef]
- Taxier, L. R.; Philippi, S. M.; York, J. M.; LaDu, M. J.; Frick, K. M. The detrimental effects of APOE4 on risk for Alzheimer's disease may result from altered dendritic spine density, synaptic proteins, and estrogen receptor alpha. Neurobiology of aging 2022b, 112, 74–86. [Google Scholar] [CrossRef] [PubMed]
- Takata, F.; Nakagawa, S.; Matsumoto, J.; Dohgu, S. Blood-Brain Barrier Dysfunction Amplifies the Development of Neuroinflammation: Understanding of Cellular Events in Brain Microvascular Endothelial Cells for Prevention and Treatment of BBB Dysfunction. Frontiers in cellular neuroscience 2021, 15, 661838. [Google Scholar] [CrossRef]
- Tang, Y.; Cai, D. Hypothalamic inflammation and GnRH in aging development. Cell cycle (Georgetown, Tex.) 2013, 12(17), 2711–2712. [Google Scholar] [CrossRef]
- Thakur, M. K.; Ghosh, S. Age and sex dependent alteration in presenilin expression in mouse cerebral cortex. Cellular and molecular neurobiology 2007, 27(8), 1059–1067. [Google Scholar] [CrossRef]
- Therriault, J.; Pascoal, T. A.; Lussier, F. Z.; Tissot, C.; Chamoun, M.; Bezgin, G.; Servaes, S.; Benedet, A. L.; Ashton, N. J.; Karikari, T. K.; Lantero-Rodriguez, J.; Kunach, P.; Wang, Y. T.; Fernandez-Arias, J.; Massarweh, G.; Vitali, P.; Soucy, J. P.; Saha-Chaudhuri, P.; Blennow, K.; Zetterberg, H.; Rosa-Neto, P. Biomarker modeling of Alzheimer's disease using PET-based Braak staging. Nature aging 2022, 2(6), 526–535. [Google Scholar] [CrossRef]
- Thomas, S. N.; Funk, K. E.; Wan, Y.; Liao, Z.; Davies, P.; Kuret, J.; Yang, A. J. Dual modification of Alzheimer's disease PHF-tau protein by lysine methylation and ubiquitylation: a mass spectrometry approach. Acta neuropathologica 2012, 123(1), 105–117. [Google Scholar] [CrossRef] [PubMed]
- Thurston, R. C.; Maki, P.; Chang, Y.; Wu, M.; Aizenstein, H. J.; Derby, C. A.; Karikari, T. K. Menopausal vasomotor symptoms and plasma Alzheimer disease biomarkers. American journal of obstetrics and gynecology 2024, 230(3), 342.e1–342.e8. [Google Scholar] [CrossRef]
- Tian, Z.; Fan, J.; Zhao, Y.; Bi, S.; Si, L.; Liu, Q. Estrogen receptor beta treats Alzheimer's disease. Neural regeneration research 2013, 8(5), 420–426. [Google Scholar] [CrossRef] [PubMed]
- Troutwine, B. R.; Hamid, L.; Lysaker, C. R.; Strope, T. A.; Wilkins, H. M. Apolipoprotein E and Alzheimer's disease. Acta pharmaceutica Sinica. B 2022, 12(2), 496–510. [Google Scholar] [CrossRef]
- Tong, J. H.; Gong, S. Q.; Zhang, Y. S.; Dong, J. R.; Zhong, X.; Wei, M. J.; Liu, M. Y. Association of Circulating Apolipoprotein AI Levels in Patients With Alzheimer's Disease: A Systematic Review and Meta-Analysis. Frontiers in aging neuroscience 2022, 14, 899175. [Google Scholar] [CrossRef]
- Tsukada, J.; Yoshida, Y.; Kominato, Y.; Auron, P. E. The CCAAT/enhancer (C/EBP) family of basic-leucine zipper (bZIP) transcription factors is a multifaceted highly-regulated system for gene regulation. Cytokine 2011, 54(1), 6–19. [Google Scholar] [CrossRef]
- Twarowski, B.; Herbet, M. Inflammatory Processes in Alzheimer's Disease-Pathomechanism, Diagnosis and Treatment: A Review. International journal of molecular sciences 2023, 24(7), 6518. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, S.; Gupta, P.; Saini, A. S.; Kaushal, C.; Sharma, S. The peroxisome proliferator-activated receptor: A family of nuclear receptors role in various diseases. Journal of advanced pharmaceutical technology & research 2011, 2(4), 236–240. [Google Scholar] [CrossRef]
- Uilenbroek, J. T.; Tiller, R.; de Jong, F. H.; Vels, F. Specific suppression of follicle-stimulating hormone secretion in gonadectomized male and female rats with intrasplenic ovarian transplants. The Journal of endocrinology 1978, 78(3), 399–406. [Google Scholar] [CrossRef]
- van Helmond, Z.; Miners, J. S.; Kehoe, P. G.; Love, S. Higher soluble amyloid beta concentration in frontal cortex of young adults than in normal elderly or Alzheimer's disease. Brain pathology (Zurich, Switzerland) 2010, 20(4), 787–793. [Google Scholar] [CrossRef]
- van den Hurk, R.; Zhao, J. Formation of mammalian oocytes and their growth, differentiation and maturation within ovarian follicles. Theriogenology 2005, 63(6), 1717–1751. [Google Scholar] [CrossRef]
- Venegas, C.; Kumar, S.; Franklin, B. S.; Dierkes, T.; Brinkschulte, R.; Tejera, D.; Vieira-Saecker, A.; Schwartz, S.; Santarelli, F.; Kummer, M. P.; Griep, A.; Gelpi, E.; Beilharz, M.; Riedel, D.; Golenbock, D. T.; Geyer, M.; Walter, J.; Latz, E.; Heneka, M. T. Microglia-derived ASC specks cross-seed amyloid-β in Alzheimer's disease. Nature 2017, 552(7685), 355–361. [Google Scholar] [CrossRef]
- Verghese, P. B.; Castellano, J. M.; Garai, K.; Wang, Y.; Jiang, H.; Shah, A.; Bu, G.; Frieden, C.; Holtzman, D. M. ApoE influences amyloid-β (Aβ) clearance despite minimal apoE/Aβ association in physiological conditions. Proceedings of the National Academy of Sciences of the United States of America 2013, 110(19), E1807–E1816. [Google Scholar] [CrossRef] [PubMed]
- Verma, M.; Vats, A.; Taneja, V. Toxic species in amyloid disorders: Oligomers or mature fibrils. Annals of Indian Academy of Neurology 2015, 18(2), 138–145. [Google Scholar] [CrossRef]
- Vogel, J. W.; Young, A. L.; Oxtoby, N. P.; Smith, R.; Ossenkoppele, R.; Strandberg, O. T.; La Joie, R.; Aksman, L. M.; Grothe, M. J.; Iturria-Medina, Y.; Alzheimer’s Disease Neuroimaging Initiative; Pontecorvo, M. J.; Devous, M. D.; Rabinovici, G. D.; Alexander, D. C.; Lyoo, C. H.; Evans, A. C.; Hansson, O. Four distinct trajectories of tau deposition identified in Alzheimer's disease. Nature medicine 2021, 27(5), 871–881. [Google Scholar] [CrossRef] [PubMed]
- Voloboueva, LA; Emery, JF; Sun, X; Giffard, RG. Inflammatory response of microglial BV-2 cells includes a glycolytic shift and is modulated by mitochondrial glucose-regulated protein 75/mortalin. FEBS Lett. 2013, 587(6), 756–62. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Vinogradova, Y; Dening, T; Hippisley-Cox, J; Taylor, L; Moore, M; Coupland, C; et al. Use of menopausal hormone therapy and risk of dementia: nested case-control studies using QResearch and CPRD databases. BMJ 2021, 374, n2182. [Google Scholar] [CrossRef]
- Wahrle, S. E.; Jiang, H.; Parsadanian, M.; Kim, J.; Li, A.; Knoten, A.; Jain, S.; Hirsch-Reinshagen, V.; Wellington, C. L.; Bales, K. R.; Paul, S. M.; Holtzman, D. M. Overexpression of ABCA1 reduces amyloid deposition in the PDAPP mouse model of Alzheimer disease. The Journal of clinical investigation 2008, 118(2), 671–682. [Google Scholar] [CrossRef]
- Walczak-Nowicka, Ł. J.; Herbet, M. Acetylcholinesterase Inhibitors in the Treatment of Neurodegenerative Diseases and the Role of Acetylcholinesterase in their Pathogenesis. International journal of molecular sciences 2021, 22(17), 9290. [Google Scholar] [CrossRef]
- Wang, J.; Li, Y.; Xia, Y. C/EBPβ as a master regulator of inflammasome signaling in neurodegenerative diseases: mechanisms and therapeutic implications. Frontiers in immunology 2025, 16, 1656165. [Google Scholar] [CrossRef]
- Wang, S-M; Jeong, C; Um, YH; Kang, DW; Kim, S; Lee, S; Lee, CU; Aizenstein, HJ; Baek, K-H; Lim, HK. Follicle-stimulating hormone linked to cognitive decline and amyloid burden in postmenopausal women. Front. Aging Neurosci. 2026, 17, 1697255. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Chen, G.; Schindler, S. E.; Christensen, J.; McKay, N. S.; Liu, J.; Wang, S.; Sun, Z.; Hassenstab, J.; Su, Y.; Flores, S.; Hornbeck, R.; Cash, L.; Cruchaga, C.; Fagan, A. M.; Tu, Z.; Morris, J. C.; Mintun, M. A.; Wang, Y.; Benzinger, T. L. S. Baseline Microglial Activation Correlates With Brain Amyloidosis and Longitudinal Cognitive Decline in Alzheimer Disease. Neurology(R) neuroimmunology & neuroinflammation 2022, 9(3), e1152. [Google Scholar] [CrossRef]
- Wang, S.; Liao, Z.; Zhang, Q.; Han, X.; Liu, C.; Wang, J. Mitochondrial dysfunction in Alzheimer's disease: a key frontier for future targeted therapies. Frontiers in immunology 2025, 15, 1484373. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Shao, X.; Yu, L.; Sun, J.; Yin, X. S.; Chen, Z.; Xu, Y.; Wang, N.; Zhang, D.; Qiu, W.; Liu, F.; Ma, C. Changes in the pH value of the human brain in Alzheimer's disease pathology correlated with CD68-positive microglia: a community-based autopsy study in Beijing, China. Molecular brain 2025, 18(1), 10. [Google Scholar] [CrossRef]
- Wang, W.; Huang, J.; Qian, S.; Zheng, Y.; Yu, X.; Jiang, T.; Ai, R.; Hou, J.; Ma, E.; Cai, J.; He, H.; Wang, X.; Xie, C. Amyloid-β but not tau accumulation is strongly associated with longitudinal cognitive decline. CNS neuroscience & therapeutics 2024, 30(7), e14860. [Google Scholar] [CrossRef]
- Wang, Y.; Hu, H.; Liu, X.; Guo, X. Hypoglycemic medicines in the treatment of Alzheimer's disease: Pathophysiological links between AD and glucose metabolism. Frontiers in pharmacology 2023, 14, 1138499. [Google Scholar] [CrossRef]
- Wang, J.; Dickson, D. W.; Trojanowski, J. Q.; Lee, V. M. The levels of soluble versus insoluble brain Abeta distinguish Alzheimer's disease from normal and pathologic aging. Experimental neurology 1999, 158(2), 328–337. [Google Scholar] [CrossRef]
- Wang, Y.; Yutuc, E.; Griffiths, W. J. Neuro-oxysterols and neuro-sterols as ligands to nuclear receptors, GPCRs, ligand-gated ion channels and other protein receptors. British journal of pharmacology 2021, 178(16), 3176–3193. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, H.; Iqbal, M.; Zheng, J.; Wines-Samuelson, M.; Shen, J. Partial loss of presenilin impairs age-dependent neuronal survival in the cerebral cortex. The Journal of neuroscience: the official journal of the Society for Neuroscience 2014, 34(48), 15912–15922. [Google Scholar] [CrossRef]
- Whitmer, R. A.; Quesenberry, C. P.; Zhou, J.; Yaffe, K. Timing of hormone therapy and dementia: the critical window theory revisited. Annals of neurology 2011, 69(1), 163–169. [Google Scholar] [CrossRef]
- Weber, D.; Davies, M. J.; Grune, T. Determination of protein carbonyls in plasma, cell extracts, tissue homogenates, isolated proteins: Focus on sample preparation and derivatization conditions. Redox biology 2015, 5, 367–380. [Google Scholar] [CrossRef]
- Wei, L; Yang, X; Wang, J; Wang, Z; Wang, Q; Ding, Y; Yu, A. H3K18 lactylation of senescent microglia potentiates brain aging and Alzheimer's disease through the NFκB signaling pathway. J Neuroinflammation 2023, 20(1), 208. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wei, Y.; Liu, M.; Wang, D. The propagation mechanisms of extracellular tau in Alzheimer's disease. Journal of neurology 2022, 269(3), 1164–1181. [Google Scholar] [CrossRef]
- Wilson, A. C.; Clemente, L.; Liu, T.; Bowen, R. L.; Meethal, S. V.; Atwood, C. S. Reproductive hormones regulate the selective permeability of the blood-brain barrier. Biochimica et biophysica acta 2008, 1782(6), 401–407. [Google Scholar] [CrossRef]
- Wirth, M.; Oh, H.; Mormino, E. C.; Markley, C.; Landau, S. M.; Jagust, W. J. The effect of amyloid β on cognitive decline is modulated by neural integrity in cognitively normal elderly. Alzheimer's & dementia: the journal of the Alzheimer's Association 2013, 9(6), 687–698.e1. [Google Scholar] [CrossRef]
- Wiseman, F. K.; Al-Janabi, T.; Hardy, J.; Karmiloff-Smith, A.; Nizetic, D.; Tybulewicz, V. L.; Fisher, E. M.; Strydom, A. A genetic cause of Alzheimer disease: mechanistic insights from Down syndrome. Nature reviews. Neuroscience 2015, 16(9), 564–574. [Google Scholar] [CrossRef]
- Wood Alexander, M.; Honer, W. G.; Saloner, R.; Galea, L. A. M.; Bennett, D. A.; Rabin, J. S.; Casaletto, K. B. The interplay between age at menopause and synaptic integrity on Alzheimer's disease risk in women. Science advances 2025, 11(10), eadt0757. [Google Scholar] [CrossRef] [PubMed]
- Wu, D. M.; Liu, J. P.; Liu, J.; Ge, W. H.; Wu, S. Z.; Zeng, C. J.; Liang, J.; Liu, K.; Lin, Q.; Hong, X. W.; Sun, Y. E.; Lu, J. Immune pathway activation in neurons triggers neural damage after stroke. Cell reports 2023, 42(11), 113368. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Zhang, Y.; Zhao, C.; Li, Q.; Zhang, J.; Han, J.; Xu, Z.; Li, J.; Ma, Y.; Wang, P.; Xu, H. Disruption of C/EBPβ-Clec7a axis exacerbates neuroinflammatory injury via NLRP3 inflammasome-mediated pyroptosis in experimental neuropathic pain. Journal of translational medicine 2022, 20(1), 583. [Google Scholar] [CrossRef]
- Wu, J.; Carlock, C.; Zhou, C.; Nakae, S.; Hicks, J.; Adams, H. P.; Lou, Y. IL-33 is required for disposal of unnecessary cells during ovarian atresia through regulation of autophagy and macrophage migration. Journal of immunology (Baltimore, Md.: 1950) 2015, 194(5), 2140–2147. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q. L.; Yang, X.; Luo, J. X.; Liu, L.; Zhou, Y.; Lu, M. H. Microglia energy metabolism: A new perspective on Alzheimer's disease treatment. Journal of the neurological sciences 2025, 475, 123585. [Google Scholar] [CrossRef]
- Wu, X. L.; Piña-Crespo, J.; Zhang, Y. W.; Chen, X. C.; Xu, H. X. Tau-mediated Neurodegeneration and Potential Implications in Diagnosis and Treatment of Alzheimer's Disease. Chinese medical journal 2017, 130(24), 2978–2990. [Google Scholar] [CrossRef]
- Wu, Z.; Wang, Z. H.; Liu, X.; Zhang, Z.; Gu, X.; Yu, S. P.; Keene, C. D.; Cheng, L.; Ye, K. Traumatic brain injury triggers APP and Tau cleavage by delta-secretase, mediating Alzheimer's disease pathology. Progress in neurobiology 2020, 185, 101730. [Google Scholar] [CrossRef] [PubMed]
- Wynne, M. E.; Ogunbona, O.; Lane, A. R.; Gokhale, A.; Zlatic, S. A.; Xu, C.; Wen, Z.; Duong, D. M.; Rayaprolu, S.; Ivanova, A.; Ortlund, E. A.; Dammer, E. B.; Seyfried, N. T.; Roberts, B. R.; Crocker, A.; Shanbhag, V.; Petris, M.; Senoo, N.; Kandasamy, S.; Claypool, S. M.; Faundez, V. APOE expression and secretion are modulated by mitochondrial dysfunction. eLife 2023, 12, e85779. [Google Scholar] [CrossRef]
- Xia, Y.; Qadota, H.; Wang, Z. H.; Liu, P.; Liu, X.; Ye, K. X.; Matheny, C. J.; Berglund, K.; Yu, S. P.; Drake, D.; Bennett, D. A.; Wang, X. C.; Yankner, B. A.; Benian, G. M.; Ye, K. Neuronal C/EBPβ/AEP pathway shortens life span via selective GABAnergic neuronal degeneration by FOXO repression. Science advances 2022, 8(13), eabj8658. [Google Scholar] [CrossRef]
- Xia, Z.; Prescott, E. E.; Urbanek, A.; Wareing, H. E.; King, M. C.; Olerinyova, A.; Dakin, H.; Leah, T.; Barnes, K. A.; Matuszyk, M. M.; Dimou, E.; Hidari, E.; Zhang, Y. P.; Lam, J. Y. L.; Danial, J. S. H.; Strickland, M. R.; Jiang, H.; Thornton, P.; Crowther, D. C.; Ohtonen, S.; De, S. Co-aggregation with Apolipoprotein E modulates the function of Amyloid-β in Alzheimer's disease. Nature communications 2024, 15(1), 4695. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.; Kang, S. S.; Wang, Z.; Liu, X.; Kuo, T. C.; Korkmaz, F.; Padilla, A.; Miyashita, S.; Chan, P.; Zhang, Z.; Katsel, P.; Burgess, J.; Gumerova, A.; Ievleva, K.; Sant, D.; Yu, S. P.; Muradova, V.; Frolinger, T.; Lizneva, D.; Iqbal, J.; Ye, K. FSH blockade improves cognition in mice with Alzheimer's disease. Nature 2022, 603(7901), 470–476. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.; Kang, S. S.; Wang, M.; Wang, Z.; Xia, Y.; Liao, J.; Liu, X.; Yu, S. P.; Zhang, Z.; Ryu, V.; Yuen, T.; Zaidi, M.; Ye, K. FSH and ApoE4 contribute to Alzheimer's disease-like pathogenesis via C/EBPβ/δ-secretase in female mice. Nature communications 2023, 14(1), 6577. [Google Scholar] [CrossRef]
- Xiong, Y. S.; Liu, F. F.; Liu, D.; Huang, H. Z.; Wei, N.; Tan, L.; Chen, J. G.; Man, H. Y.; Gong, C. X.; Lu, Y.; Wang, J. Z.; Zhu, L. Q. Opposite effects of two estrogen receptors on tau phosphorylation through disparate effects on the miR-218/PTPA pathway. Aging cell 2015, 14(5), 867–877. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Pang, Y.; Fan, X. Mitochondria in oxidative stress, inflammation and aging: from mechanisms to therapeutic advances. Signal transduction and targeted therapy 2025, 10(1), 190. [Google Scholar] [CrossRef]
- Xue, Y.; Zuo, S.; Wang, F.; Qi, X. From hormones to neurodegeneration: how FSH drives Alzheimer's disease. Frontiers in aging neuroscience 2025, 17, 1578439. [Google Scholar] [CrossRef]
- Yaffe, K.; Haan, M.; Byers, A.; Tangen, C.; Kuller, L. Estrogen use, APOE, and cognitive decline: evidence of gene-environment interaction. Neurology 2000, 54(10), 1949–1954. [Google Scholar] [CrossRef]
- Yang, D. S.; Stavrides, P.; Mohan, P. S.; Kaushik, S.; Kumar, A.; Ohno, M.; Schmidt, S. D.; Wesson, D. W.; Bandyopadhyay, U.; Jiang, Y.; Pawlik, M.; Peterhoff, C. M.; Yang, A. J.; Wilson, D. A.; St George-Hyslop, P.; Westaway, D.; Mathews, P. M.; Levy, E.; Cuervo, A. M.; Nixon, R. A. Therapeutic effects of remediating autophagy failure in a mouse model of Alzheimer disease by enhancing lysosomal proteolysis. Autophagy 2011, 7(7), 788–789. [Google Scholar] [CrossRef] [PubMed]
- Yao, Q.; Long, C.; Yi, P.; Zhang, G.; Wan, W.; Rao, X.; Ying, J.; Liang, W.; Hua, F. C/EBPβ: A transcription factor associated with the irreversible progression of Alzheimer's disease. CNS neuroscience & therapeutics 2024, 30(4), e14721. [Google Scholar] [CrossRef]
- Yang, H; Mo, N; Tong, L; Dong, J; Fan, Z; Jia, M; Yue, J; Wang, Y. Microglia lactylation in relation to central nervous system diseases. Neural Regen Res 2025, 20(1), 29–40. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yang, Z.; Klionsky, D. J. Eaten alive: a history of macroautophagy. Nature cell biology 2010, 12(9), 814–822. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.; Zhao, C.; Qiu, Y.; Zhou, Z.; Bao, J.; Qian, W. Dendritic/Post-synaptic Tau and Early Pathology of Alzheimer's Disease. Frontiers in molecular neuroscience 2021, 14, 671779. [Google Scholar] [CrossRef]
- Yoshiyama, Y.; Higuchi, M.; Zhang, B.; Huang, S. M.; Iwata, N.; Saido, T. C.; Maeda, J.; Suhara, T.; Trojanowski, J. Q.; Lee, V. M. Synapse loss and microglial activation precede tangles in a P301S tauopathy mouse model. Neuron 2007, 53(3), 337–351. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Nagasu, H.; Murakami, T.; Hoang, H.; Broderick, L.; Hoffman, H. M.; Horng, T. Inflammasome activation leads to Caspase-1-dependent mitochondrial damage and block of mitophagy. Proceedings of the National Academy of Sciences of the United States of America 2014, 111(43), 15514–15519. [Google Scholar] [CrossRef]
- Yuk, J. S.; Lee, J. S.; Park, J. H. Menopausal hormone therapy and risk of dementia: health insurance database in South Korea-based retrospective cohort study. Frontiers in aging neuroscience 2023, 15, 1213481. [Google Scholar] [CrossRef]
- Zaidi, M.; Ye, K. A pituitary hormone has a key role in Alzheimer's disease. In Nature; Advance online publication, 2022. [Google Scholar] [CrossRef]
- Zeng, Q.; Li, K.; Luo, X.; Wang, S.; Xu, X.; Jiaerken, Y.; Liu, X.; Hong, L.; Hong, H.; Li, Z.; Fu, Y.; Zhang, T.; Chen, Y.; Liu, Z.; Huang, P.; Zhang, M.; for behalf of Alzheimer's Disease Neuroimaging Initiative (ADNI). The association of enlarged perivascular space with microglia-related inflammation and Alzheimer's pathology in cognitively normal elderly. Neurobiology of disease 2022, 170, 105755. [Google Scholar] [CrossRef]
- Zhang, G.; Li, J.; Purkayastha, S.; Tang, Y.; Zhang, H.; Yin, Y.; Li, B.; Liu, G.; Cai, D. Hypothalamic programming of systemic aging involving IKK-β, NF-κB and GnRH. Nature 2013, 497(7448), 211–216. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wei, W.; Zhao, M.; Ma, L.; Jiang, X.; Pei, H.; Cao, Y.; Li, H. Interaction between Aβ and Tau in the Pathogenesis of Alzheimer's Disease. International journal of biological sciences 2021, 17(9), 2181–2192. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, Y.; Wang, J.; Xia, Y.; Zhang, J.; Chen, L. Recent advances in Alzheimer's disease: Mechanisms, clinical trials and new drug development strategies. Signal transduction and targeted therapy 2024, 9(1), 211. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X. W.; Zhu, X. X.; Tang, D. S.; Lu, J. H. Targeting autophagy in Alzheimer's disease: Animal models and mechanisms. Zoological research 2023, 44(6), 1132–1145. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Xu, C.; Sun, J.; Shen, H. M.; Wang, J.; Yang, C. Impairment of the autophagy-lysosomal pathway in Alzheimer's diseases: Pathogenic mechanisms and therapeutic potential. Acta pharmaceutica Sinica. B 2022, 12(3), 1019–1040. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Yao, W.; Li, C.; Wu, W.; Li, Q.; Liu, H. Administration of follicle-stimulating hormone induces autophagy via upregulation of HIF-1α in mouse granulosa cells. Cell death & disease 2017, 8(8), e3001. [Google Scholar] [CrossRef]
- Zou, H.; Chen, M.; Wang, X.; Yu, J.; Li, X.; Xie, Y.; Liu, J.; Liu, M.; Xu, L.; Zhang, Q.; Tian, X.; Zhang, F.; Guo, B. C/EBPβ isoform-specific regulation of podocyte pyroptosis in lupus nephritis-induced renal injury. The Journal of pathology 2023, 261(3), 269–285. [Google Scholar] [CrossRef]
- Zsido, R. G.; Heinrich, M.; Slavich, G. M.; Beyer, F.; Kharabian Masouleh, S.; Kratzsch, J.; Raschpichler, M.; Mueller, K.; Scharrer, U.; Löffler, M.; Schroeter, M. L.; Stumvoll, M.; Villringer, A.; Witte, A. V.; Sacher, J. Association of Estradiol and Visceral Fat With Structural Brain Networks and Memory Performance in Adults. JAMA network open 2019, 2(6), e196126. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




