Submitted:
31 March 2026
Posted:
03 April 2026
You are already at the latest version
Abstract
Keywords:
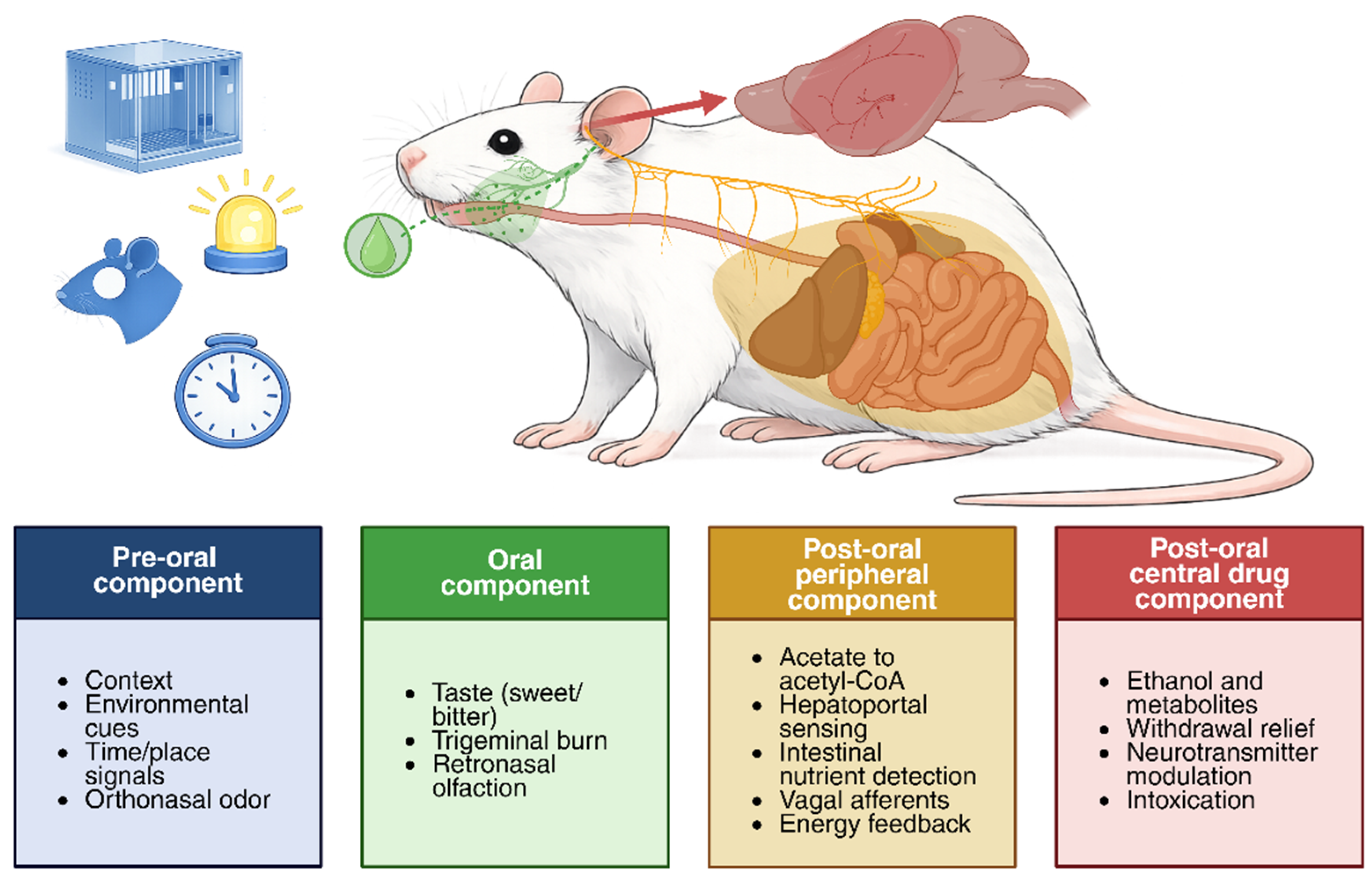
1. Introduction: A Multi-Reward Component Framework for Ethanol Intake
2. Evidence for Distinct Reward Components Across Rodent Models
2.1. Evidence for Post-Oral Reward Components
2.1.1. The Post-Oral Peripheral Reward and Central Drug Reward Components Together
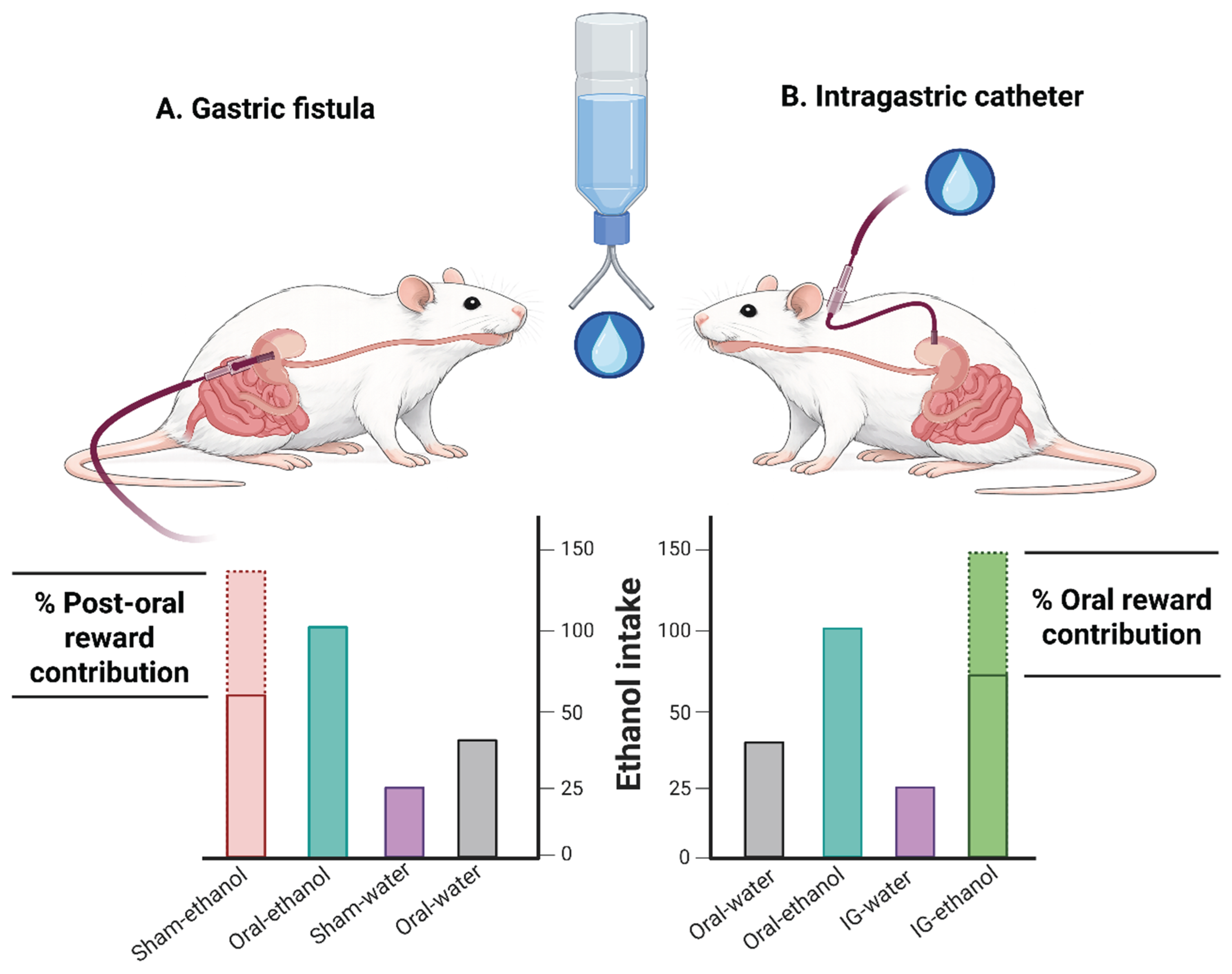
2.1.1.1. The Post-Oral Components Without the Oral Component: Intragastric Self-Infusion
2.1.1.2. The Post-Oral Components Without the Oral Component and Peripheral Gastric Post-Oral Reward: Intravenous Self-Infusion
2.1.2. The Post-Oral Central Drug Reward Component
2.1.2.1. Pharmacological Regulation by Internal Cues: Titration and Drug Discrimination
2.1.2.2. Pharmacological Consequences of Sustained Intake: Physical Dependence
2.1.2.3. The Role of Ethanol and Its Metabolites
2.1.3. The Post-Oral Peripheral Component
2.1.4. Conclusion on Post-Oral Reward Components
2.2. Evidence for Oral Reward Component
2.2.1. The Oral Reward Compotnent Without Post-Oral Components: Sham Drinking with Gastric Fistula
2.2.2. Ethanol Preference over Palatable Alternatives
2.2.3. Ethanol Drinking Persistence Despite Quinine-Adulteration
2.2.4. Oral Reward Through Enhanced Palatability: Taste Reactivity and Brief Access Licking
2.2.5. Ethanol Intake Despite the Absence of Specific Taste Receptors
2.2.6. Conclusion on the Oral Reward Component
2.3. Evidence for the Pre-Oral Reward Component
2.4. Additional Modulatory Factors Shaping Ethanol Intake
2.5. Heterogeneity of Reward Components Across Models and Implications for Translation
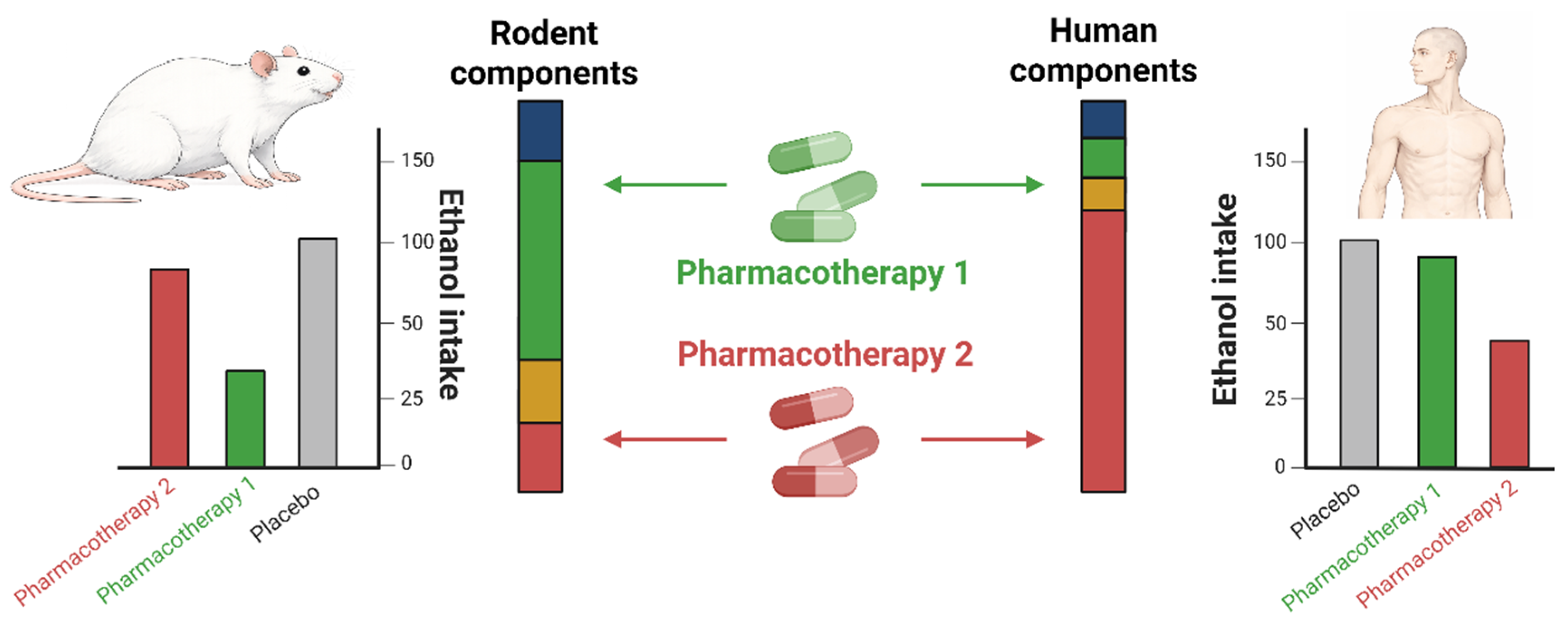
3. A Reward-Component Framework for AUD Pharmacotherapy: Alignment and Multi-Component Action
3.1. Translational Failures as Cases of Component Misalignment
3.1.1. CRF1 Receptor Antagonists
3.1.2. Histamine H3 Receptor Inverse Agonists
3.2. The Reward Components Modified by Clinically Used Pharmacotherapies for AUD
3.2.1. Naltrexone
3.2.2. Disulfiram
3.2.3. Acamprosate
3.3. Emerging and Repurposed Pharmacotherapies
3.4. Conclusion: Toward Component-Aligned Pharmacotherapy
4. Future Directions: Experimental Strategies to Dissociate the Reward Components Controlling Ethanol Intake
5. Conclusion
6. Outstanding Questions for the Field
- Which reward component(s) primarily control high intake in each rodent line, and how do their relative contributions change with experience, dependence, and testing conditions?
- Can we identify preclinical models in which ethanol intake is primarily controlled by the post-oral central drug reward component, and do such models improve the prediction of pharmacotherapy outcomes from preclinical studies to clinical AUD?
- Can orthogonal experimental designs (e.g., oral vs intragastric vs sham drinking, or intravenous vs intragastric) reliably dissociate reward components and yield reproducible estimates of their relative contribution across laboratories?
- Which reward components are causally modified by clinically used, repurposed, and emerging pharmacotherapies, and to what extent do these component profiles differ between rodents and humans?
- How large, persistent and behaviorally meaningful is ethanol’s peripheral component in standard and high-drinking lines?
- Why is intravenous ethanol a weak reinforcer in rats, and what does this reveal about the relative importance of oral reward , post-oral peripheral reward, and post-oral central drug reward components?
- Are there sex differences in the relative contribution of reward components to ethanol drinking?
Author Contributions
Acknowledgments
Competing interests
References
- MacKillop, J; Agabio, R; Feldstein Ewing, SW; Heilig, M; Kelly, JF; Leggio, L; et al. Hazardous drinking and alcohol use disorders. Nat Rev Dis Prim 2022, 8(1), 80. [Google Scholar] [CrossRef]
- Guerrin, CGJ; Tesselaar, DRM; Booij, J; Schellekens, AFA; Homberg, JR. Precision medicine in substance use disorders: Integrating behavioral, environmental, and biological insights. Neurosci Biobehav Rev [Internet] 2025, 176, 106311. Available online: https://www.sciencedirect.com/science/article/pii/S0149763425003124. [CrossRef]
- Witkiewitz, K; Litten, RZ; Leggio, L. Advances in the science and treatment of alcohol use disorder. Sci Adv 2019, 5(9), eaax4043. [Google Scholar] [CrossRef] [PubMed]
- Spanagel, R. Animal models of addiction. Dialogues Clin Neurosci 2017, 19(3), 247–58. [Google Scholar] [CrossRef] [PubMed]
- Kwako, LE; Spagnolo, PA; Schwandt, ML; Thorsell, A; George, DT; Momenan, R; et al. The corticotropin releasing hormone-1 (CRH1) receptor antagonist pexacerfont in alcohol dependence: a randomized controlled experimental medicine study. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol 2015, 40(5), 1053–63. [Google Scholar] [CrossRef]
- Schwandt, ML; Cortes, CR; Kwako, LE; George, DT; Momenan, R; Sinha, R; et al. The CRF1 Antagonist Verucerfont in Anxious Alcohol-Dependent Women: Translation of Neuroendocrine, But not of Anti-Craving Effects. Neuropsychopharmacology [Internet] 2016, 41(12), 2818–29. [Google Scholar] [CrossRef]
- Spierling, SR; Zorrilla, EP. Don’t stress about CRF: assessing the translational failures of CRF(1)antagonists. Psychopharmacology (Berl) 2017, 234(9–10), 1467–81. [Google Scholar] [CrossRef] [PubMed]
- Le Foll, B; Naassila, M; Jeanblanc, J; Hendershot, CS; Chavarria, J; Calmels, T; et al. Histamine H3 Receptor as a target for alcohol use disorder: challenging the predictability of animal models for clinical translation in drug development. Transl Psychiatry [Internet] 2026. [Google Scholar] [CrossRef]
- Samson, HH; Files, FJ; Denning, C; Marvin, S. Comparison of alcohol-preferring and nonpreferring selectively bred rat lines. I. Ethanol initiation and limited access operant self-administration. Alcohol Clin Exp Res 1998, 22(9), 2133–46. [Google Scholar]
- Tabakoff, B; Hoffman, PL. Animal models in alcohol research. Alcohol Res Heal J Natl Inst Alcohol Abus Alcohol. 2000, 24(2), 77–84. [Google Scholar]
- Oberlin, B; Best, C; Matson, L; Henderson, A; Grahame, N. Derivation and characterization of replicate high- and low-alcohol preferring lines of mice and a high-drinking crossed HAP line. Behav Genet. 2011, 41(2), 288–302. [Google Scholar] [CrossRef] [PubMed]
- Bell, RL; Rodd-Henricks, ZA; Webster, AA; Lumeng, L; Li, TK; McBride, WJ; et al. Heart rate and motor-activating effects of orally self-administered ethanol in alcohol-preferring (P) rats. Alcohol Clin Exp Res. 2002, 26(8), 1162–70. [Google Scholar] [CrossRef] [PubMed]
- Li, TK; Lumeng, L; Doolittle, DP. Selective breeding for alcohol preference and associated responses. Behav Genet. 1993, 23(2), 163–70. [Google Scholar] [CrossRef]
- McBride, WJ; Rodd, ZA; Bell, RL; Lumeng, L; Li, TK. The alcohol-preferring (P) and high-alcohol-drinking (HAD) rats – Animal models of alcoholism. Alcohol [Internet] 2014, 48(3), 209–15. Available online: https://www.sciencedirect.com/science/article/pii/S0741832913001778. [CrossRef]
- Bell, RL; Rodd, ZA; Lumeng, L; Murphy, JM; McBride, WJ. The alcohol-preferring P rat and animal models of excessive alcohol drinking. Addict Biol. 2006, 11(3–4), 270–88. [Google Scholar] [CrossRef]
- Weber, LA; Yee, DM; Small, DM; Petzschner, FH. The interoceptive origin of reinforcement learning. Trends Cogn Sci. 2025, 29(9), 840–54. [Google Scholar] [CrossRef] [PubMed]
- Wilson, DF; Matschinsky, FM. Ethanol metabolism: The good, the bad, and the ugly. Med Hypotheses [Internet] 2020, 140, 109638. Available online: https://www.sciencedirect.com/science/article/pii/S0306987720300797. [CrossRef]
- de Araujo, IE; Schatzker, M; Small, DM. Rethinking Food Reward. Annu Rev Psychol 2020, 71, 139–64. [Google Scholar] [CrossRef]
- Sclafani, A. From appetite setpoint to appetition: 50years of ingestive behavior research. Physiol Behav 2018, 192, 210–7. [Google Scholar] [CrossRef]
- Waller, MB; McBride, WJ; Gatto, GJ; Lumeng, L; Li, TK. Intragastric Self-Infusion of Ethanol by Ethanol-Preferring and -Nonpreferring Lines of Rats. Science (80- ) [Internet] 1984, 225(4657), 78–80. [Google Scholar] [CrossRef]
- Murphy, JM; Waller, MB; Gatto, GJ; McBride, WJ; Lumeng, L; Li, TK. Effects of fluoxetine on the intragastric self-administration of ethanol in the alcohol preferring P line of rats. Alcohol [Internet] 1988, 5(4), 283–6. Available online: https://www.sciencedirect.com/science/article/pii/0741832988900663. [CrossRef]
- Elizalde, G; Sclafani, A. Flavor preferences conditioned by intragastric polycose infusions: a detailed analysis using an electronic esophagus preparation. Physiol Behav 1990, 47(1), 63–77. [Google Scholar] [CrossRef]
- Drucker, DB; Ackroff, K; Sclafani, A. Flavor preference produced by intragastric polycose infusions in rats using a concurrent conditioning procedure. Physiol Behav [Internet] 1993, 54(2), 351–5. Available online: https://www.sciencedirect.com/science/article/pii/003193849390122V. [CrossRef] [PubMed]
- Ackroff, K; Sclafani, A. Flavor quality and ethanol concentration affect ethanol-conditioned flavor preferences. Pharmacol Biochem Behav [Internet] 2002, 74(1), 229–40. Available online: https://www.sciencedirect.com/science/article/pii/S0091305702009875. [CrossRef]
- Fidler, TL; Clews, TW; Cunningham, CL. Reestablishing an intragastric ethanol self-infusion model in rats. Alcohol Clin Exp Res. 2006, 30(3), 414–28. [Google Scholar] [CrossRef]
- Ackroff, K; Sclafani, A. Flavor preferences conditioned by intragastric infusion of ethanol in rats. Pharmacol Biochem Behav 2001, 68(2), 327–38. [Google Scholar] [CrossRef]
- Fidler, TL; Dion, AM; Powers, MS; Ramirez, JJ; Mulgrew, JA; Smitasin, PJ; et al. Intragastric self-infusion of ethanol in high- and low-drinking mouse genotypes after passive ethanol exposure. Genes Brain Behav 2011, 10(3), 264–75. [Google Scholar] [CrossRef] [PubMed]
- Windisch, KA; Kosobud, AEK; Czachowski, CL. Intravenous alcohol self-administration in the P rat. Alcohol [Internet] 2014, 48(5), 419–25. Available online: https://www.sciencedirect.com/science/article/pii/S0741832914000901. [CrossRef]
- Hyytiä, P; Schulteis, G; Koob, GF. Intravenous heroin and ethanol self-administration by alcohol-preferring AA and alcohol-avoiding ANA rats. Psychopharmacology (Berl) [Internet] 1996, 125(3), 248–54. [Google Scholar] [CrossRef]
- Lê, AD; Kalant, H. Intravenous self-administration of alcohol in rats-problems with translation to humans. Addict Biol. 2017, 22(6), 1665–81. [Google Scholar] [CrossRef]
- McClearn, GE; Rodgers, DA. Differences in Alcohol Preference among Inbred Strains of Mice. Q J Stud Alcohol [Internet] Available from. 1959, 20(4), 691–5. [Google Scholar] [CrossRef]
- Grahame, NJ; Cunningham, CL. Intravenous Ethanol Self-administration in C57BL/6J and DBA/2J Mice. Alcohol Clin Exp Res [Internet] Available from. 1997, 21(1), 56–62. [Google Scholar] [CrossRef]
- He, XX; Nebert, DW; Vasiliou, V; Zhu, H; Shertzer, HG. Genetic differences in alcohol drinking preference between inbred strains of mice. Pharmacogenetics 1997, 7(3), 223–33. [Google Scholar] [CrossRef]
- Waller, MB; McBride, WJ; Lumeng, L; Li, TK. Effects of intravenous ethanol and of 4-methylpyrazole on alcohol drinking in alcohol-preferring rats. Pharmacol Biochem Behav [Internet] 1982, 17(4), 763–8. Available online: https://www.sciencedirect.com/science/article/pii/0091305782903598. [CrossRef]
- Carnicella, S; Yowell, Q V; Ron, D. Regulation of Operant Oral Ethanol Self-Administration: A Dose–Response Curve Study in Rats. Alcohol Clin Exp Res [Internet] Available from. 2011, 35(1), 116–25. [Google Scholar] [CrossRef]
- Hodge, CW; Cox, AA; Bratt, AM; Camarini, R; Iller, K; Kelley, SP; et al. The discriminative stimulus properties of self-administered ethanol are mediated by GABA(A) and NMDA receptors in rats. Psychopharmacology (Berl) 2001, 154(1), 13–22. [Google Scholar] [CrossRef] [PubMed]
- Shelton, KL; Macenski, MJ. Discriminative stimulus effects of self-administered ethanol. Behav Pharmacol 1998, 9(4), 329–36. [Google Scholar] [CrossRef] [PubMed]
- Waller, MB; McBride, WJ; Lumeng, L; Li, TK. Induction of dependence on ethanol by free-choice drinking in alcohol-preferring rats. Pharmacol Biochem Behav [Internet] 1982, 16(3), 501–7. Available online: https://www.sciencedirect.com/science/article/pii/0091305782904592. [CrossRef]
- Dyr, W; Kostowski, W. Warsaw high-preferring (WHP) and Warsaw low-preferring (WLP) lines of rats selectively bred for high and low voluntary ethanol intake: preliminary phenotypic characterization. Alcohol 2008, 42(3), 161–70. [Google Scholar] [CrossRef]
- Israel, Y; Quintanilla, ME; Karahanian, E; Rivera-Meza, M; Herrera-Marschitz, M. The “first hit” toward alcohol reinforcement: role of ethanol metabolites. Alcohol Clin Exp Res. 2015, 39(5), 776–86. [Google Scholar] [CrossRef]
- Gatto, GJ; McBride, WJ; Murphy, JM; Lumeng, L; Li, TK. Ethanol self-infusion into the ventral tegmental area by alcohol-preferring rats. Alcohol [Internet] 1994, 11(6), 557–64. Available online: https://www.sciencedirect.com/science/article/pii/0741832994900833. [CrossRef]
- Rodd, ZA; Bell, RL; Melendez, RI; Kuc, KA; Lumeng, L; Li, TK; et al. Comparison of intracranial self-administration of ethanol within the posterior ventral tegmental area between alcohol-preferring and Wistar rats. Alcohol Clin Exp Res. 2004, 28(8), 1212–9. [Google Scholar] [CrossRef]
- Engleman, EA; Ding, ZM; Oster, SM; Toalston, JE; Bell, RL; Murphy, JM; et al. Ethanol is self-administered into the nucleus accumbens shell, but not the core: evidence of genetic sensitivity. Alcohol Clin Exp Res. 2009, 33(12), 2162–71. [Google Scholar] [CrossRef]
- Rodd, ZA; Bell, RL; Zhang, Y; Murphy, JM; Goldstein, A; Zaffaroni, A; et al. Regional Heterogeneity for the Intracranial Self-Administration of Ethanol and Acetaldehyde within the Ventral Tegmental Area of Alcohol-Preferring (P) Rats: Involvement of Dopamine and Serotonin. Neuropsychopharmacology [Internet] Available from. 2005, 30(2), 330–8. [Google Scholar] [CrossRef]
- Rodd-Henricks, ZA; Melendez, RI; Zaffaroni, A; Goldstein, A; McBride, WJ; Li, TK. The reinforcing effects of acetaldehyde in the posterior ventral tegmental area of alcohol-preferring rats. Pharmacol Biochem Behav 2002, 72(1–2), 55–64. [Google Scholar] [CrossRef]
- Rodd, ZA; Bell, RL; Kuc, KA; Murphy, JM; Lumeng, L; McBride, WJ. Effects of concurrent access to multiple ethanol concentrations and repeated deprivations on alcohol intake of high-alcohol-drinking (HAD) rats. Addict Biol. 2009, 14(2), 152–64. [Google Scholar] [CrossRef]
- Deehan, GA; Hauser, SR; Wilden, JA; Truitt, WA; Rodd, ZA. Elucidating the biological basis for the reinforcing actions of alcohol in the mesolimbic dopamine system: the role of active metabolites of alcohol. Front Behav Neurosci [Internet]. 2013, pp. 7–2013. Available online: https://www.frontiersin.org/journals/behavioral-neuroscience/articles/10.3389/fnbeh.2013.00104.
- Deehan, GAJ; Engleman, EA; Ding, ZM; McBride, WJ; Rodd, ZA. Microinjections of acetaldehyde or salsolinol into the posterior ventral tegmental area increase dopamine release in the nucleus accumbens shell. Alcohol Clin Exp Res. 2013, 37(5), 722–9. [Google Scholar] [CrossRef]
- Brown, ZW; Amit, Z; Rockman, GE. Intraventricular self-administration of acetaldehyde, but not ethanol, in naive laboratory rats. Psychopharmacology (Berl) 1979, 64(3), 271–6. [Google Scholar] [CrossRef]
- Smith, BR; Amit, Z; Splawinsky, J. Conditioned place preference induced by intraventricular infusions of acetaldehyde. Alcohol. 1984, 1(3), 193–5. [Google Scholar] [CrossRef] [PubMed]
- Quintanilla, ME; Rivera-Meza, M; Berrios-Cárcamo, PA; Bustamante, D; Buscaglia, M; Morales, P; et al. Salsolinol, free of isosalsolinol, exerts ethanol-like motivational/sensitization effects leading to increases in ethanol intake. Alcohol 2014, 48(6), 551–9. [Google Scholar] [CrossRef] [PubMed]
- Melis, M; Enrico, P; Peana, AT; Diana, M. Acetaldehyde mediates alcohol activation of the mesolimbic dopamine system. Eur J Neurosci 2007, 26(10), 2824–33. [Google Scholar] [CrossRef]
- Hipólito, L; Martí-Prats, L; Sánchez-Catalán, MJ; Polache, A; Granero, L. Induction of conditioned place preference and dopamine release by salsolinol in posterior VTA of rats: Involvement of μ-opioid receptors. Neurochem Int [Internet] 2011, 59(5), 559–62. Available online: https://www.sciencedirect.com/science/article/pii/S0197018611001720. [CrossRef]
- Xie, G; Hipólito, L; Zuo, W; Polache, A; Granero, L; Krnjevic, K; et al. Salsolinol stimulates dopamine neurons in slices of posterior ventral tegmental area indirectly by activating μ-opioid receptors. J Pharmacol Exp Ther. 2012, 341(1), 43–50. [Google Scholar] [CrossRef]
- Rodd, ZA; Bell, RL; Zhang, Y; Goldstein, A; Zaffaroni, A; McBride, WJ; et al. Salsolinol produces reinforcing effects in the nucleus accumbens shell of alcohol-preferring (P) rats. Alcohol Clin Exp Res. 2003, 27(3), 440–9. [Google Scholar] [CrossRef] [PubMed]
- Duncan, C; Deitrich, RA. A critical evaluation of tetrahydroisoquinoline induced ethanol preference in rats. Pharmacol Biochem Behav 1980, 13(2), 265–81. [Google Scholar] [CrossRef]
- Myers, RD; Melchior, CL. Differential actions on voluntary alcohol intake of tetrahydroisoquinolines or a β-carboline infused chronically in the ventricle of the rat. Pharmacol Biochem Behav [Internet] 1977, 7(4), 381–92. Available online: https://www.sciencedirect.com/science/article/pii/0091305777902350. [CrossRef] [PubMed]
- Matsuzawa, S; Suzuki, T; Misawa, M. Involvement of mu-opioid receptor in the salsolinol-associated place preference in rats exposed to conditioned fear stress. Alcohol Clin Exp Res. 2000, 24(3), 366–72. [Google Scholar]
- Hipólito, L; Sánchez-Catalán, MJ; Zornoza, T; Polache, A; Granero, L. Locomotor stimulant effects of acute and repeated intrategmental injections of salsolinol in rats: role of mu-opioid receptors. Psychopharmacology (Berl) 2010, 209(1), 1–11. [Google Scholar] [CrossRef] [PubMed]
- Starkey, JA; Mechref, Y; Muzikar, J; McBride, WJ; Novotny, M V. Determination of salsolinol and related catecholamines through on-line preconcentration and liquid chromatography/atmospheric pressure photoionization mass spectrometry. Anal Chem. 2006, 78(10), 3342–7. [Google Scholar] [CrossRef]
- Koechling, UM; Amit, Z. Effects of 3-amino-1,2,4-triazole on brain catalase in the mediation of ethanol consumption in mice. Alcohol [Internet] 1994, 11(3), 235–9. Available online: https://www.sciencedirect.com/science/article/pii/0741832994900361. [CrossRef]
- C.M.G. A, Z. A. The effect of 3-amino-1,2,4-triazole on voluntary ethanol consumption: Evidence for brain catalase involvement in the mechanism of action. Neuropharmacology [Internet] 1992, 31(7), 709–12. Available online: https://www.sciencedirect.com/science/article/pii/002839089290150N. [CrossRef]
- Tampier, L; Quintanilla, ME; Mardones, J. Effects of aminotriazole on ethanol, water, and food intake and on brain catalase in UChA and UChB rats. Alcohol [Internet] 1995, 12(4), 341–4. Available online: https://www.sciencedirect.com/science/article/pii/074183299500014I. [CrossRef]
- Rotzinger, S; Smith, BR; Amit, Z. Catalase inhibition attenuates the acquisition of ethanol and saccharin-quinine consumption in laboratory rats. Behav Pharmacol 1994, 5(2), 203–9. [Google Scholar] [CrossRef]
- Ledesma, JC; Baliño, P; Aragon, CMG. Reduction in central H2O2 levels prevents voluntary ethanol intake in mice: a role for the brain catalase-H2O2 system in alcohol binge drinking. Alcohol Clin Exp Res. 2014, 38(1), 60–7. [Google Scholar] [CrossRef]
- Karahanian, E; Rivera-Meza, M; Tampier, L; Quintanilla, ME; Herrera-Marschitz, M; Israel, Y. Long-term inhibition of ethanol intake by the administration of an aldehyde dehydrogenase-2 (ALDH2)-coding lentiviral vector into the ventral tegmental area of rats. Addict Biol. 2015, 20(2), 336–44. [Google Scholar] [CrossRef] [PubMed]
- Tampier, L; Quintanilla, ME; Karahanian, E; Rivera-Meza, M; Herrera-Marschitz, M; Israel, Y. The alcohol deprivation effect: marked inhibition by anticatalase gene administration into the ventral tegmental area in rats. Alcohol Clin Exp Res. 2013, 37(8), 1278–85. [Google Scholar] [CrossRef] [PubMed]
- Quintanilla, ME; Tampier, L; Karahanian, E; Rivera-Meza, M; Herrera-Marschitz, M; Israel, Y. Reward and relapse: complete gene-induced dissociation in an animal model of alcohol dependence. Alcohol Clin Exp Res. 2012, 36(3), 517–22. [Google Scholar] [CrossRef]
- Quintanilla, ME; Israel, Y; Sapag, A; Tampier, L. The UChA and UChB rat lines: metabolic and genetic differences influencing ethanol intake. Addict Biol. 2006, 11(3–4), 310–23. [Google Scholar] [CrossRef]
- Tampier, L; Quintanilla, ME. Involvement of brain ethanol metabolism on acute tolerance development and on ethanol consumption in alcohol-drinker (UChB) and non-drinker (UChA) rats. Addict Biol. 2003, 8(3), 279–86. [Google Scholar] [CrossRef]
- Karahanian, E; Quintanilla, ME; Tampier, L; Rivera-Meza, M; Bustamante, D; Gonzalez-Lira, V; et al. Ethanol as a Prodrug: Brain Metabolism of Ethanol Mediates its Reinforcing Effects. Alcohol Clin Exp Res [Internet] Available from. 2011, 35(4), 606–12. [Google Scholar] [CrossRef]
- Rivera-Meza, M; Vásquez, D; Quintanilla, ME; Lagos, D; Rojas, B; Herrera-Marschitz, M; et al. Activation of mitochondrial aldehyde dehydrogenase (ALDH2) by ALDA-1 reduces both the acquisition and maintenance of ethanol intake in rats: A dual mechanism? Neuropharmacology 2019, 146, 175–83. [Google Scholar] [CrossRef]
- Orrico, A; Hipólito, L; Sánchez-Catalán, MJ; Martí-Prats, L; Zornoza, T; Granero, L; et al. Efficacy of D-penicillamine, a sequestering acetaldehyde agent, in the prevention of alcohol relapse-like drinking in rats. Psychopharmacology (Berl) 2013, 228(4), 563–75. [Google Scholar] [CrossRef]
- Font, L; Aragon, CMG; Miquel, M. Voluntary ethanol consumption decreases after the inactivation of central acetaldehyde by d-penicillamine. Behav Brain Res. 2006, 171(1), 78–86. [Google Scholar] [CrossRef]
- Quintanilla, ME; Callejas, O; Tampier, L. Aversion to acetaldehyde: differences in low-alcohol-drinking (UChA) and high-alcohol-drinking (UChB) rats. Alcohol [Internet] 2002, 26(2), 69–74. Available online: https://www.sciencedirect.com/science/article/pii/S0741832901001975. [CrossRef] [PubMed]
- Deems, DA; Oetting, RL; Sherman, JE; Garcia, J. Hungry, but not thirsty, rats prefer flavors paired with ethanol. Physiol Behav [Internet] 1986, 36(1), 141–4. Available online: https://www.sciencedirect.com/science/article/pii/0031938486900879. [CrossRef]
- Sherman, JE; Hickis, CF; Rice, AG; Rusiniak, KW; Garcia, J. Preferences and aversions for stimuli paired with ethanol in hungry rats. Anim Learn Behav [Internet] Available from. 1983, 11(1), 101–6. [Google Scholar] [CrossRef]
- de Vaca, SC; Carr, KD. Food Restriction Enhances the Central Rewarding Effect of Abused Drugs. J Neurosci [Internet] 1998, 18, 7502–10. Available online: https://api.semanticscholar.org/CorpusID:15955552. [CrossRef]
- Ackroff, K; Rozental, D; Sclafani, A. Ethanol-conditioned flavor preferences compared with sugar- and fat-conditioned preferences in rats. Physiol Behav. 2004, 81(4), 699–713. [Google Scholar] [CrossRef] [PubMed]
- Lucas, F; Sclafani, A; Ackroff, K; Sclafani, A. Flavor preferences conditioned by intragastric fat infusions in rats. Pharmacol Biochem Behav 1989, 46(2), 327–38. [Google Scholar] [CrossRef]
- Pérez, C; Lucas, F; Sclafani, A. Increased flavor acceptance and preference conditioned by the postingestive actions of glucose. Physiol Behav. 1998, 64(4), 483–92. [Google Scholar] [CrossRef]
- Lankford, MF; Roscoe, AK; Pennington, SN; Myers, RD. Drinking of high concentrations of ethanol versus palatable fluids in alcohol-preferring (P) rats: valid animal model of alcoholism. Alcohol [Internet] 1991, 8(4), 293–9. Available online: https://www.sciencedirect.com/science/article/pii/074183299190417U. [CrossRef]
- Mehiel, R; Bolles, RC. Learned flavor preferences based on caloric outcome. Anim Learn Behav [Internet] Available from. 1984, 12(4), 421–7. [Google Scholar] [CrossRef]
- Samson, HH; Roehrs, TA; Tolliver, GA. Ethanol reinforced responding in the rat: A concurrent analysis using sucrose as the alternate choice. Pharmacol Biochem Behav [Internet] 1982, 17(2), 333–9. Available online: https://www.sciencedirect.com/science/article/pii/0091305782900880. [CrossRef]
- Files, FJ; Samson, HH; Brice, GT. Sucrose, ethanol, and sucrose/ethanol reinforced responding under variable-interval schedules of reinforcement. Alcohol Clin Exp Res. 1995, 19(5), 1271–8. [Google Scholar] [CrossRef] [PubMed]
- Ren, X; Ferreira, JG; Zhou, L; Shammah-Lagnado, SJ; Yeckel, CW; de Araujo, IE. Nutrient selection in the absence of taste receptor signaling. J Neurosci Off J Soc Neurosci. 2010, 30(23), 8012–23. [Google Scholar] [CrossRef]
- Lucas, F; Sclafani, A. Differential reinforcing and satiating effects of intragastric fat and carbohydrate infusions in rats. Physiol Behav 1999, 66(3), 381–8. [Google Scholar] [CrossRef]
- Sclafani, A; Cardieri, C; Tucker, K; Blusk, D; Ackroff, K. Intragastric glucose but not fructose conditions robust flavor preferences in rats. Am J Physiol. 1993, 265 2 Pt 2, R320–5. [Google Scholar] [CrossRef] [PubMed]
- Rowland, NE; Morian, KR. Sham intake of ethanol in “P” rats. Alcohol [Internet] 1994, 11(2), 163–5. Available online: https://www.sciencedirect.com/science/article/pii/0741832994900582. [CrossRef]
- Rowland, NE; Barnett, M. Sham ingestion of alcohol in rats. Alcohol. 1992, 9(1), 75–7. [Google Scholar] [CrossRef]
- Russell, RN; McBride, WJ; Lumeng, L; Li, TK; Murphy, JM. Apomorphine and 7-OH DPAT reduce ethanol intake of P and HAD rats. Alcohol [Internet] 1996, 13(5), 515–9. Available online: https://www.sciencedirect.com/science/article/pii/0741832995000623. [CrossRef]
- Toalston, JE; Oster, SM; Kuc, KA; Pommer, TJ; Murphy, JM; Lumeng, L; et al. Effects of alcohol and saccharin deprivations on concurrent ethanol and saccharin operant self-administration by alcohol-preferring (P) rats. Alcohol 2008, 42(4), 277–84. [Google Scholar] [CrossRef]
- Lankford, MF; Myers, RD. Genetics of alcoholism: Simultaneous presentation of a chocolate drink diminishes alcohol preference in high drinking HAD rats. Pharmacol Biochem Behav [Internet] 1994, 49(2), 417–25. Available online: https://www.sciencedirect.com/science/article/pii/009130579490443X. [CrossRef]
- Colombo, G; Agabio, R; Diaz, G; Fà, M; Lobina, C; Reali, R; et al. Sardinian alcohol-preferring rats prefer chocolate and sucrose over ethanol. Alcohol [Internet] 1997, 14(6), 611–5. Available online: https://www.sciencedirect.com/science/article/pii/S074183299700075X. [CrossRef]
- Tampier, L; Quintanilla, ME. Effect of concurrent saccharin intake on ethanol consumption by high-alcohol-drinking (UChB) rats. Addict Biol. 2009, 14(3), 276–82. [Google Scholar] [CrossRef] [PubMed]
- Loi, B; Lobina, C; Maccioni, P; Fantini, N; Carai, MAM; Gessa, GL; et al. Increase in Alcohol Intake, Reduced Flexibility of Alcohol Drinking, and Evidence of Signs of Alcohol Intoxication in Sardinian Alcohol-Preferring Rats Exposed to Intermittent Access to 20% Alcohol. Alcohol Clin Exp Res [Internet] Available from. 2010, 34(12), 2147–54. [Google Scholar] [CrossRef]
- Timme, NM; Linsenbardt, D; Timm, M; Galbari, T; Cornwell, E; Lapish, C. Alcohol-preferring P rats exhibit aversion-resistant drinking of alcohol adulterated with quinine. Alcohol [Internet] 2020, 83, 47–56. Available online: https://www.sciencedirect.com/science/article/pii/S0741832919301235. [CrossRef]
- Brasser, SM; Silbaugh, BC; Ketchum, MJ; Olney, JJ; Lemon, CH. Chemosensory responsiveness to ethanol and its individual sensory components in alcohol-preferring, alcohol-nonpreferring and genetically heterogeneous rats. Addict Biol [Internet] Available from. 2012, 17(2), 423–36. [Google Scholar] [CrossRef]
- Seif, T; Chang, SJ; Simms, JA; Gibb, SL; Dadgar, J; Chen, BT; et al. Cortical activation of accumbens hyperpolarization-active NMDARs mediates aversion-resistant alcohol intake. Nat Neurosci [Internet] Available from. 2013, 16(8), 1094–100. [Google Scholar] [CrossRef]
- Sneddon, EA; Schuh, KM; Fennell, KA; Grahame, NJ; Radke, AK. Crossed high alcohol preferring mice exhibit aversion-resistant responding for alcohol with quinine but not footshock punishment. Alcohol 2022, 105, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Houck, CA; Carron, CR; Millie, LA; Grahame, NJ. Innate and Acquired Quinine-Resistant Alcohol, but not Saccharin, Drinking in Crossed High-Alcohol-Preferring Mice. Alcohol Clin Exp Res. 2019, 43(11), 2421–30. [Google Scholar] [CrossRef]
- Bice, PJ; Kiefer, SW. Taste Reactivity in Alcohol Preferring and Nonpreferring Rats. Alcohol Clin Exp Res [Internet] Available from. 1990, 14(5), 721–7. [Google Scholar] [CrossRef]
- Lemon, CH; Wilson, DM; Brasser, SM. Differential neural representation of oral ethanol by central taste-sensitive neurons in ethanol-preferring and genetically heterogeneous rats. J Neurophysiol [Internet] Available from. 2011, 106(6), 3145–56. [Google Scholar] [CrossRef] [PubMed]
- Tampier, L; Quintanilla, ME. Saccharin consumption and the effect of a long-term exposure to a sweetened alcoholic solution in high- (UChB) and low- (UChA) alcohol-drinking rats. Alcohol [Internet] 2005, 37(1), 47–52. Available online: https://www.sciencedirect.com/science/article/pii/S0741832905001667. [CrossRef] [PubMed]
- Agabio, R; Carai, MA; Lobina, C; Pani, M; Reali, R; Bourov, I; et al. Dissociation of ethanol and saccharin preference in sP and sNP rats. Alcohol Clin Exp Res. 2000, 24(1), 24–9. [Google Scholar] [CrossRef] [PubMed]
- Dyr, W; Wyszogrodzka, E; Mierzejewski, P; Bieńkowski, P. Drinking of flavored solutions by high preferring (WHP) and low preferring (WLP) alcohol-drinking rats. Pharmacol Reports [Internet] Available from. 2014, 66(1), 28–33. [Google Scholar] [CrossRef]
- Badia-Elder, NE; Kiefer, SW. Taste Reactivity in Alcohol-Preferring AA and Alcohol-Avoiding ANA Rats. Alcohol [Internet] 1999, 18(2), 159–63. Available online: https://www.sciencedirect.com/science/article/pii/S0741832998000792. [CrossRef]
- Sinclair, JD; Kampov-Polevoy, A; Stewart, R; Li, TK. Taste preferences in rat lines selected for low and high alcohol consumption. Alcohol [Internet] 1992, 9(2), 155–60. Available online: https://www.sciencedirect.com/science/article/pii/0741832992900278. [CrossRef]
- Kampov-Polevoy, AB; Garbutt, JC; Janowsky, DS. Association between preference for sweets and excessive alcohol intake: a review of animal and human studies. Alcohol Alcohol. 1999, 34(3), 386–95. [Google Scholar] [CrossRef]
- Bachmanov, AA; Tordoff, MG; Beauchamp, GK. Ethanol consumption and taste preferences in C57BL/6ByJ and 129/J mice. Alcohol Clin Exp Res 1996, 20(2), 201–6. [Google Scholar] [CrossRef]
- Oberlin, BG; Grahame, NJ. High-alcohol preferring mice are more impulsive than low-alcohol preferring mice as measured in the delay discounting task. Alcohol Clin Exp Res. 2009, 33(7), 1294–303. [Google Scholar] [CrossRef]
- Crabbe, JC; Spence, SE; Brown, LL; Metten, P. Alcohol preference drinking in a mouse line selectively bred for high drinking in the dark. Alcohol [Internet] 2011, 45(5), 427–40. Available online: https://www.sciencedirect.com/science/article/pii/S0741832910002193. [CrossRef]
- Barkley-Levenson, AM; Crabbe, JC. High drinking in the dark mice: a genetic model of drinking to intoxication. Alcohol 2014, 48(3), 217–23. [Google Scholar] [CrossRef]
- Koh, AP; Dando, R. Bitter and Sweet Diets Alter Taste Response and Alcohol Consumption Behavior in Mice. Nutrients 2025, 17. [Google Scholar] [CrossRef]
- Goodwin, FL; Amit, Z. Relative taste thresholds for ethanol, saccharin, and quinine solutions in three strains of rats nonselected for ethanol: a comparative study. Exp Clin Psychopharmacol 2000, 8(2), 216–24. [Google Scholar] [CrossRef]
- Goodwin, FL; Amit, Z. Do taste factors contribute to the mediation of ethanol intake? Ethanol and saccharin-quinine intake in three rat strains. Alcohol Clin Exp Res. 1998, 22(4), 837–44. [Google Scholar] [CrossRef]
- Loney, GC; Meyer, PJ. Brief Exposures to the Taste of Ethanol (EtOH) and Quinine Promote Subsequent Acceptance of EtOH in a Paradigm that Minimizes Postingestive Consequences. Alcohol Clin Exp Res. 2018, 42(3), 589–602. [Google Scholar] [CrossRef]
- Lemon, CH; Brasser, SM; Smith, D V. Alcohol activates a sucrose-responsive gustatory neural pathway. J Neurophysiol. 2004, 92(1), 536–44. [Google Scholar] [CrossRef] [PubMed]
- Vetter, I; Wyse, BD; Roberts-Thomson, SJ; Monteith, GR; Cabot, PJ. Mechanisms involved in potentiation of transient receptor potential vanilloid 1 responses by ethanol. Eur J Pain [Internet] Available from. 2008, 12(4), 441–54. [Google Scholar] [CrossRef] [PubMed]
- Simon, SA; de Araujo, IE. The salty and burning taste of capsaicin. J Gen Physiol. 2005, 125(6), 531–4. [Google Scholar] [CrossRef] [PubMed]
- Trevisani, M; Smart, D; Gunthorpe, MJ; Tognetto, M; Barbieri, M; Campi, B; et al. Ethanol elicits and potentiates nociceptor responses via the vanilloid receptor-1. Nat Neurosci 2002, 5(6), 546–51. [Google Scholar] [CrossRef]
- Blednov, YA; Walker, D; Martinez, M; Levine, M; Damak, S; Margolskee, RF. Perception of sweet taste is important for voluntary alcohol consumption in mice. Genes Brain Behav 2008, 7(1), 1–13. [Google Scholar] [CrossRef] [PubMed]
- Brasser, SM; Norman, MB; Lemon, CH. T1r3 taste receptor involvement in gustatory neural responses to ethanol and oral ethanol preference. Physiol Genomics 2010, 41(3), 232–43. [Google Scholar] [CrossRef] [PubMed]
- Beeler, JA; McCutcheon, JE; Cao, ZFH; Murakami, M; Alexander, E; Roitman, MF; et al. Taste uncoupled from nutrition fails to sustain the reinforcing properties of food. Eur J Neurosci 2012, 36(4), 2533–46. [Google Scholar] [CrossRef]
- Tellez, LA; Ren, X; Han, W; Medina, S; Ferreira, JG; Yeckel, CW; et al. Glucose utilization rates regulate intake levels of artificial sweeteners. J Physiol [Internet] Available from. 2013, 591(22), 5727–44. [Google Scholar] [CrossRef]
- de Araujo, IE; Oliveira-Maia, AJ; Sotnikova, TD; Gainetdinov, RR; Caron, MG; Nicolelis, MAL; et al. Food Reward in the Absence of Taste Receptor Signaling. Neuron [Internet] Available from. 2008, 57(6), 930–41. [Google Scholar] [CrossRef] [PubMed]
- Sclafani, A; Glendinning, JI. Flavor preferences conditioned in C57BL/6 mice by intragastric carbohydrate self-infusion. Physiol Behav. 2003, 79(4–5), 783–8. [Google Scholar] [CrossRef]
- Sclafani, A. Oral and postoral determinants of food reward. Physiol Behav [Internet] 2004, 81(5), 773–9. Available online: https://www.sciencedirect.com/science/article/pii/S0031938404001817. [CrossRef]
- Holman, GL. Intragastric reinforcement effect. J Comp Physiol Psychol. 1969, 69(3), 432–41. [Google Scholar] [CrossRef]
- Bell, RL; Stewart, RB; Woods, JE, 2nd; Lumeng, L; Li, TK; Murphy, JM; et al. Responsivity and development of tolerance to the motor impairing effects of moderate doses of ethanol in alcohol-preferrfile:///C:/Users/cypgue/Downloads/29240979.nbibing (P) and -nonpreferring (NP) rat lines. Alcohol Clin Exp Res 2001, 25(5), 644–50. [Google Scholar]
- Katner, SN; Kerr, TM; Weiss, F. Ethanol anticipation enhances dopamine efflux in the nucleus accumbens of alcohol-preferring (P) but not Wistar rats. Behav Pharmacol. 1996, 7(7), 669–74. [Google Scholar] [CrossRef]
- Melendez, RI; Rodd-Henricks, ZA; Engleman, EA; Li, TK; McBride, WJ; Murphy, JM. Microdialysis of dopamine in the nucleus accumbens of alcohol-preferring (P) rats during anticipation and operant self-administration of ethanol. Alcohol Clin Exp Res. 2002, 26(3), 318–25. [Google Scholar] [CrossRef] [PubMed]
- Myers, RD; Ice, JC; Piercy, KT. Odor of specific alcohol solutions selectively alters locomotor activity of alcohol-drinking P rats. Physiol Behav 1997, 61(1), 71–5. [Google Scholar] [CrossRef] [PubMed]
- Pomrenze, MB; Fetterly, TL; Winder, DG; Messing, RO. The Corticotropin Releasing Factor Receptor 1 in Alcohol Use Disorder: Still a Valid Drug Target? Alcohol Clin Exp Res 2017, 41(12), 1986–99. [Google Scholar] [CrossRef] [PubMed]
- Hansson, AC; Cippitelli, A; Sommer, WH; Fedeli, A; Björk, K; Soverchia, L; et al. Variation at the rat Crhr1 locus and sensitivity to relapse into alcohol seeking induced by environmental stress. Proc Natl Acad Sci U S A 2006, 103(41), 15236–41. [Google Scholar] [CrossRef]
- Chu, K; Koob, GF; Cole, M; Zorrilla, EP; Roberts, AJ. Dependence-induced increases in ethanol self-administration in mice are blocked by the CRF1 receptor antagonist antalarmin and by CRF1 receptor knockout. Pharmacol Biochem Behav 2007, 86(4), 813–21. [Google Scholar] [CrossRef]
- Gehlert, DR; Cippitelli, A; Thorsell, A; Lê, AD; Hipskind, PA; Hamdouchi, C; et al. 3-(4-Chloro-2-morpholin-4-yl-thiazol-5-yl)-8-(1-ethylpropyl)-2,6-dimethyl-imidazo [1,2-b]pyridazine: a novel brain-penetrant, orally available corticotropin-releasing factor receptor 1 antagonist with efficacy in animal models of alcoholism. J Neurosci Off J Soc Neurosci. 2007, 27(10), 2718–26. [Google Scholar] [CrossRef]
- Richardson, HN; Zhao, Y; Fekete, ÉM; Funk, CK; Wirsching, P; Janda, KD; et al. MPZP: A novel small molecule corticotropin-releasing factor type 1 receptor (CRF1) antagonist. Pharmacol Biochem Behav [Internet] 2008, 88(4), 497–510. Available online: https://www.sciencedirect.com/science/article/pii/S0091305707003218. [CrossRef]
- Funk, CK; Zorrilla, EP; Lee, MJ; Rice, KC; Koob, GF. Corticotropin-releasing factor 1 antagonists selectively reduce ethanol self-administration in ethanol-dependent rats. Biol Psychiatry 2007, 61(1), 78–86. [Google Scholar] [CrossRef]
- Sabino, V; Cottone, P; Koob, GF; Steardo, L; Lee, MJ; Rice, KC; et al. Dissociation between opioid and CRF1 antagonist sensitive drinking in Sardinian alcohol-preferring rats. Psychopharmacology (Berl) 2006, 189(2), 175–86. [Google Scholar] [CrossRef]
- Sommer, WH; Rimondini, R; Hansson, AC; Hipskind, PA; Gehlert, DR; Barr, CS; et al. Upregulation of voluntary alcohol intake, behavioral sensitivity to stress, and amygdala crhr1 expression following a history of dependence. Biol Psychiatry 2008, 63(2), 139–45. [Google Scholar] [CrossRef]
- Diaz-Granados, JL; Graham, DL. The effects of continuous and intermittent ethanol exposure in adolesence on the aversive properties of ethanol during adulthood. Alcohol Clin Exp Res. 2007, 31(12), 2020–7. [Google Scholar] [CrossRef] [PubMed]
- Flores-Ramirez, FJ; Matzeu, A; Sánchez-Marín, L; Martin-Fardon, R. Blockade of corticotropin-releasing factor-1 receptors in the infralimbic cortex prevents stress-induced reinstatement of alcohol seeking in male Wistar rats: Evidence of interaction between CRF(1) and orexin receptor signaling. Neuropharmacology 2022, 210, 109046. [Google Scholar] [CrossRef]
- Chen, YW; Fiscella, KA; Bacharach, SZ; Tanda, G; Shaham, Y; Calu, DJ. Effect of yohimbine on reinstatement of operant responding in rats is dependent on cue contingency but not food reward history. Addict Biol. 2015, 20(4), 690–700. [Google Scholar] [CrossRef]
- Marinelli, PW; Funk, D; Juzytsch, W; Harding, S; Rice, KC; Shaham, Y; et al. The CRF1 receptor antagonist antalarmin attenuates yohimbine-induced increases in operant alcohol self-administration and reinstatement of alcohol seeking in rats. Psychopharmacology (Berl) 2007, 195(3), 345–55. [Google Scholar] [CrossRef]
- Liu, X; Weiss, F. Additive effect of stress and drug cues on reinstatement of ethanol seeking: exacerbation by history of dependence and role of concurrent activation of corticotropin-releasing factor and opioid mechanisms. J Neurosci Off J Soc Neurosci. 2002, 22(18), 7856–61. [Google Scholar] [CrossRef]
- Panula, P. Histamine, histamine H(3) receptor, and alcohol use disorder. Br J Pharmacol 2020, 177(3), 634–41. [Google Scholar] [CrossRef]
- Brown, RE; Stevens, DR; Haas, HL. The physiology of brain histamine. Prog Neurobiol 2001, 63(6), 637–72. [Google Scholar] [CrossRef]
- Yoshimoto, R; Miyamoto, Y; Shimamura, K; Ishihara, A; Takahashi, K; Kotani, H; et al. Therapeutic potential of histamine H3 receptor agonist for the treatment of obesity and diabetes mellitus. Proc Natl Acad Sci U S A 2006, 103(37), 13866–71. [Google Scholar] [CrossRef]
- Costa, A; Micioni Di Bonaventura, E; Botticelli, L; Eramo, B; Gaetani, S; Passani, MB; et al. Brain histaminergic system: An emerging target for the treatment of feeding and eating-related disorders. Pharmacol Res. 2025, 221, 107949. [Google Scholar] [CrossRef]
- Ishizuka, T; Yamatodani, A. Integrative role of the histaminergic system in feeding and taste perception. Front Syst Neurosci. 2012, 6, 44. [Google Scholar] [CrossRef] [PubMed]
- Xu, L; Lin, W; Zheng, Y; Wang, Y; Chen, Z. The Diverse Network of Brain Histamine in Feeding: Dissect its Functions in a Circuit-Specific Way. Curr Neuropharmacol. 2024, 22(2), 241–59. [Google Scholar] [CrossRef]
- Nuutinen, S; Vanhanen, J; Mäki, T; Panula, P. Histamine h3 receptor: a novel therapeutic target in alcohol dependence? Front Syst Neurosci 2012, 6, 36. [Google Scholar] [CrossRef]
- Nuutinen, S; Vanhanen, J; Pigni, MC; Panula, P. Effects of histamine H3 receptor ligands on the rewarding, stimulant and motor-impairing effects of ethanol in DBA/2J mice. Neuropharmacology 2011, 60(7–8), 1193–9. [Google Scholar] [CrossRef]
- Nuutinen, S; Lintunen, M; Vanhanen, J; Ojala, T; Rozov, S; Panula, P. Evidence for the role of histamine H3 receptor in alcohol consumption and alcohol reward in mice. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol 2011, 36(10), 2030–40. [Google Scholar] [CrossRef]
- Treesukosol, Y; Ishizuka, T; Yamamoto, T; Yamatodani, A. The effect of taste stimuli on histamine release in the anterior hypothalamus of rats. Brain Res [Internet] 2003, 964(1), 51–5. Available online: https://www.sciencedirect.com/science/article/pii/S0006899302040611. [CrossRef] [PubMed]
- Nuutinen, S; Mäki, T; Rozov, S; Bäckström, P; Hyytiä, P; Piepponen, P; et al. Histamine H3 receptor antagonist decreases cue-induced alcohol reinstatement in mice. Neuropharmacology [Internet] 2016, 106, 156–63. Available online: https://www.sciencedirect.com/science/article/pii/S0028390815002646. [CrossRef] [PubMed]
- Hendershot, CS; Wardell, JD; Samokhvalov, A V; Rehm, J. Effects of naltrexone on alcohol self-administration and craving: meta-analysis of human laboratory studies. Addict Biol. 2017, 22(6), 1515–27. [Google Scholar] [CrossRef]
- McPheeters, M; O’Connor, EA; Riley, S; Kennedy, SM; Voisin, C; Kuznacic, K; et al. Pharmacotherapy for Alcohol Use Disorder: A Systematic Review and Meta-Analysis. JAMA 2023, 330(17), 1653–65. [Google Scholar] [CrossRef]
- Leventhal, L; Kirkham, TC; Cole, JL; Bodnar, RJ. Selective actions of central mu and kappa opioid antagonists upon sucrose intake in sham-fed rats. Brain Res. 1995, 685(1–2), 205–10. [Google Scholar] [CrossRef]
- Leventhal, L; Bodnar, RJ. Different central opioid receptor subtype antagonists modify maltose dextrin and deprivation-induced water intake in sham feeding and sham drinking rats. Brain Res. 1996, 741(1–2), 300–8. [Google Scholar] [CrossRef]
- Kirkham, TC; Cooper, SJ. Attenuation of sham feeding by naloxone is stereospecific: evidence for opioid mediation of orosensory reward. Physiol Behav. 1988, 43(6), 845–7. [Google Scholar] [CrossRef]
- Coonfield, DL; Hill, KG; Kaczmarek, HJ; Ferraro, FM, 3rd; Kiefer, SW. Low doses of naltrexone reduce palatability and consumption of ethanol in outbred rats. Alcohol 2002, 26(1), 43–7. [Google Scholar] [CrossRef]
- Beczkowska, IW; Koch, JE; Bostock, ME; Leibowitz, SF; Bodnar, RJ. Central opioid receptor subtype antagonists differentially reduce intake of saccharin and maltose dextrin solutions in rats. Brain Res. 1993, 618(2), 261–70. [Google Scholar] [CrossRef]
- Bertino, M; Beauchamp, GK; Engelman, K. Naltrexone, an opioid blocker, alters taste perception and nutrient intake in humans. Am J Physiol. 1991, 261 1 Pt 2, R59–63. [Google Scholar] [CrossRef]
- Arbisi, PA; Billington, CJ; Levine, AS. The Effect of Naltrexone on Taste Detection and Recognition Threshold. Appetite [Internet] 1999, 32(2), 241–9. Available online: https://www.sciencedirect.com/science/article/pii/S0195666398902173. [CrossRef]
- Lanz, J; Biniaz-Harris, N; Kuvaldina, M; Jain, S; Lewis, K; Fallon, BA. Disulfiram: Mechanisms, Applications, and Challenges. Antibiot (Basel, Switzerland) 2023, (3), 12. [Google Scholar] [CrossRef]
- Suh, JJ; Pettinati, HM; Kampman, KM; O’Brien, CP. The status of disulfiram: a half of a century later. J Clin Psychopharmacol. 2006, 26(3), 290–302. [Google Scholar] [CrossRef] [PubMed]
- Shen, WW. Anticraving therapy for alcohol use disorder: A clinical review. Neuropsychopharmacol reports 2018, 38(3), 105–16. [Google Scholar] [CrossRef]
- Skinner, MD; Lahmek, P; Pham, H; Aubin, HJ. Disulfiram efficacy in the treatment of alcohol dependence: a meta-analysis. PLoS One 2014, 9(2), e87366. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, CH; Pedersen, B; Tønnesen, H. The efficacy of disulfiram for the treatment of alcohol use disorder. Alcohol Clin Exp Res. 2011, 35(10), 1749–58. [Google Scholar] [CrossRef]
- Gaval-Cruz, M; Weinshenker, D. mechanisms of disulfiram-induced cocaine abstinence: antabuse and cocaine relapse. Mol Interv. 2009, 9(4), 175–87. [Google Scholar] [CrossRef]
- Schroeder, JP; Cooper, DA; Schank, JR; Lyle, MA; Gaval-Cruz, M; Ogbonmwan, YE; et al. Disulfiram attenuates drug-primed reinstatement of cocaine seeking via inhibition of dopamine β-hydroxylase. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol 2010, 35(12), 2440–9. [Google Scholar] [CrossRef]
- Cooper, DA; Kimmel, HL; Manvich, DF; Schmidt, KT; Weinshenker, D; Howell, LL. Effects of pharmacologic dopamine β-hydroxylase inhibition on cocaine-induced reinstatement and dopamine neurochemistry in squirrel monkeys. J Pharmacol Exp Ther. 2014, 350(1), 144–52. [Google Scholar] [CrossRef]
- Lei, Y; Tang, L; Chen, Q; Wu, L; He, W; Tu, D; et al. Disulfiram ameliorates nonalcoholic steatohepatitis by modulating the gut microbiota and bile acid metabolism. Nat Commun [Internet] Available from. 2022, 13(1), 6862. [Google Scholar] [CrossRef]
- Tampier, L; Quintanilla, ME; Israel, Y. Tolerance to disulfiram induced by chronic alcohol intake in the rat. Alcohol Clin Exp Res. 2008, 32(6), 937–41. [Google Scholar] [CrossRef]
- Maisel, NC; Blodgett, JC; Wilbourne, PL; Humphreys, K; Finney, JW. Meta-analysis of naltrexone and acamprosate for treating alcohol use disorders: when are these medications most helpful? Addiction 2013, 108(2), 275–93. [Google Scholar] [CrossRef] [PubMed]
- Kalk, NJ; Lingford-Hughes, AR. The clinical pharmacology of acamprosate. Br J Clin Pharmacol 2014, 77(2), 315–23. [Google Scholar] [CrossRef] [PubMed]
- Spanagel, R; Vengeliene, V; Jandeleit, B; Fischer, WN; Grindstaff, K; Zhang, X; et al. Acamprosate produces its anti-relapse effects via calcium. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol 2014, 39(4), 783–91. [Google Scholar] [CrossRef] [PubMed]
- Dahchour, A; De Witte, P. Acamprosate decreases the hypermotility during repeated ethanol withdrawal. Alcohol 1999, 18(1), 77–81. [Google Scholar] [CrossRef]
- Cole, JC; Littleton, JM; Little, HJ. Acamprosate, but not naltrexone, inhibits conditioned abstinence behaviour associated with repeated ethanol administration and exposure to a plus-maze. Psychopharmacology (Berl) 2000, 147(4), 403–11. [Google Scholar] [CrossRef]
- Cowen, MS; Adams, C; Kraehenbuehl, T; Vengeliene, V; Lawrence, AJ. The acute anti-craving effect of acamprosate in alcohol-preferring rats is associated with modulation of the mesolimbic dopamine system. Addict Biol. 2005, 10(3), 233–42. [Google Scholar] [CrossRef]
- Heyser, CJ; Schulteis, G; Durbin, P; Koob, GF. Chronic acamprosate eliminates the alcohol deprivation effect while having limited effects on baseline responding for ethanol in rats. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol 1998, 18(2), 125–33. [Google Scholar] [CrossRef] [PubMed]
- Kurokawa, K; Mizuno, K; Shibasaki, M; Higashioka, M; Oka, M; Hirouchi, M; et al. Acamprosate suppresses ethanol-induced place preference in mice with ethanol physical dependence. J Pharmacol Sci. 2013, 122(4), 289–98. [Google Scholar] [CrossRef]
- Quertemont, E; Brabant, C; De Witte, P. Acamprosate reduces context-dependent ethanol effects. Psychopharmacology (Berl) 2002, 164(1), 10–8. [Google Scholar] [CrossRef] [PubMed]
- Bachteler, D; Economidou, D; Danysz, W; Ciccocioppo, R; Spanagel, R. The effects of acamprosate and neramexane on cue-induced reinstatement of ethanol-seeking behavior in rat. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol 2005, 30(6), 1104–10. [Google Scholar] [CrossRef]
- Czachowski, CL; Delory, MJ. Acamprosate and naltrexone treatment effects on ethanol and sucrose seeking and intake in ethanol-dependent and nondependent rats. Psychopharmacology (Berl) 2009, 204(2), 335–48. [Google Scholar] [CrossRef] [PubMed]
- Le Magnen, J; Tran, G; Durlach, J; Martin, C. Dose-dependent suppression of the high alcohol intake of chronically intoxicated rats by Ca-acetyl homotaurinate. Alcohol. 1987, 4(2), 97–102. [Google Scholar] [CrossRef]
- Le Magnen, J; Tran, G; Durlach, J. Lack of effects of Ca-acetyl homotaurinate on chronic and acute toxicities of ethanol in rats. Alcohol. 1987, 4(2), 103–8. [Google Scholar] [CrossRef]
- Jerlhag, E. GLP-1 Receptor Agonists: Promising Therapeutic Targets for Alcohol Use Disorder. Endocrinology 2025, 166(4). [Google Scholar] [CrossRef]
- Klausen, MK; Jensen, ME; Møller, M; Le Dous, N; Jensen, AMØ; Zeeman, VA; et al. Exenatide once weekly for alcohol use disorder investigated in a randomized, placebo-controlled clinical trial. JCI insight 2022, 7(19). [Google Scholar] [CrossRef]
- Hendershot, CS; Bremmer, MP; Paladino, MB; Kostantinis, G; Gilmore, TA; Sullivan, NR; et al. Once-Weekly Semaglutide in Adults With Alcohol Use Disorder: A Randomized Clinical Trial. JAMA psychiatry 2025, 82(4), 395–405. [Google Scholar] [CrossRef] [PubMed]
- Eshraghi, R; Ghadimi, DJ; Montazerinamin, S; Bahrami, A; Kachela, Y; Rezasoltani, M; et al. Effects of glucagon-like peptide-1 receptor agonists on alcohol consumption: a systematic review and meta-analysis. EClinicalMedicine 2025, 90, 103645. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Megido, C; Thomsen, M. Sex-dependent divergence in the effects of GLP-1 agonist exendin-4 on alcohol reinforcement and reinstatement in C57BL/6J mice. Psychopharmacology (Berl) 2023, 240(6), 1287–98. [Google Scholar] [CrossRef] [PubMed]
- Vallöf, D; Maccioni, P; Colombo, G; Mandrapa, M; Jörnulf, JW; Egecioglu, E; et al. The glucagon-like peptide 1 receptor agonist liraglutide attenuates the reinforcing properties of alcohol in rodents. Addict Biol. 2016, 21(2), 422–37. [Google Scholar] [CrossRef]
- Eren-Yazicioglu, CY; Yigit, A; Dogruoz, RE; Yapici-Eser, H. Can GLP-1 Be a Target for Reward System Related Disorders? A Qualitative Synthesis and Systematic Review Analysis of Studies on Palatable Food, Drugs of Abuse, and Alcohol. Front Behav Neurosci. 2020, 14, 614884. [Google Scholar] [CrossRef]
- Zheng, Z; Zong, Y; Ma, Y; Tian, Y; Pang, Y; Zhang, C; et al. Glucagon-like peptide-1 receptor: mechanisms and advances in therapy. Signal Transduct Target Ther 2024, 9(1), 234. [Google Scholar] [CrossRef]
- Jensen, ME; Galli, A; Thomsen, M; Jensen, KL; Thomsen, GK; Klausen, MK; et al. Glucagon-like peptide-1 receptor regulation of basal dopamine transporter activity is species-dependent. Neurochem Int. 2020, 138, 104772. [Google Scholar] [CrossRef]
- Vallöf, D; Kalafateli, AL; Jerlhag, E. Brain region specific glucagon-like peptide-1 receptors regulate alcohol-induced behaviors in rodents. Psychoneuroendocrinology 2019, 103, 284–95. [Google Scholar] [CrossRef]
- Allingbjerg, ML; Hansen, SN; Secher, A; Thomsen, M. Glucagon-like peptide-1 receptors in nucleus accumbens, ventral hippocampus, and lateral septum reduce alcohol reinforcement in mice. Exp Clin Psychopharmacol 2023, 31(3), 612–20. [Google Scholar] [CrossRef]
- Zheng, YJ; Soegiharto, C; Au, HCT; Valentino, K; Le, GH; Wong, S; et al. A systematic review on the role of glucagon-like peptide-1 receptor agonists on alcohol-related behaviors: potential therapeutic strategy for alcohol use disorder. Acta Neuropsychiatr 2025, 37, e51. [Google Scholar] [CrossRef]
- Jensterle, M; Rizzo, M; Janez, A. Glucagon-Like Peptide 1 and Taste Perception: From Molecular Mechanisms to Potential Clinical Implications. Int J Mol Sci 2021, 22(2). [Google Scholar] [CrossRef]
- Khan, R; Doty, RL. GLP-1 receptor agonists significantly impair taste function. Physiol Behav 2025, 291, 114793. [Google Scholar] [CrossRef]
- Trapp, S; Brierley, DI. Brain GLP-1 and the regulation of food intake: GLP-1 action in the brain and its implications for GLP-1 receptor agonists in obesity treatment. Br J Pharmacol. 2022, 179(4), 557–70. [Google Scholar] [CrossRef] [PubMed]
- Gibbons, C; Blundell, J; Tetens Hoff, S; Dahl, K; Bauer, R; Baekdal, T. Effects of oral semaglutide on energy intake, food preference, appetite, control of eating and body weight in subjects with type 2 diabetes. Diabetes Obes Metab 2021, 23(2), 581–8. [Google Scholar] [CrossRef]
- Holt, MK; Richards, JE; Cook, DR; Brierley, DI; Williams, DL; Reimann, F; et al. Preproglucagon Neurons in the Nucleus of the Solitary Tract Are the Main Source of Brain GLP-1, Mediate Stress-Induced Hypophagia, and Limit Unusually Large Intakes of Food. Diabetes 2019, 68(1), 21–33. [Google Scholar] [CrossRef]
- Shin, YK; Martin, B; Golden, E; Dotson, CD; Maudsley, S; Kim, W; et al. Modulation of taste sensitivity by GLP-1 signaling. J Neurochem. 2008, 106(1), 455–63. [Google Scholar] [CrossRef]
- Doval-Caballero, JLE; Ferreira-Hermosillo, A; Eugenio-Ponce, GD; García-Sáenz, MR; Ibarra-Salce, R; Tenorio-Rojo, AP; et al. Potential role of glucagon like peptide 1 in taste receptors. Front Endocrinol (Lausanne) 2025, 16, 1683419. [Google Scholar] [CrossRef]
- Bettadapura, S; Dowling, K; Jablon, K; Al-Humadi, AW; le Roux, CW. Changes in food preferences and ingestive behaviors after glucagon-like peptide-1 analog treatment: techniques and opportunities. Int J Obes (Lond) 2025, 49(3), 418–26. [Google Scholar] [CrossRef] [PubMed]
- Martin, B; Dotson, CD; Shin, YK; Ji, S; Drucker, DJ; Maudsley, S; et al. Modulation of taste sensitivity by GLP-1 signaling in taste buds. Ann N Y Acad Sci. 2009, 1170, 98–101. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, M; Dencker, D; Wörtwein, G; Weikop, P; Egecioglu, E; Jerlhag, E; et al. The glucagon-like peptide 1 receptor agonist Exendin-4 decreases relapse-like drinking in socially housed mice. Pharmacol Biochem Behav 2017, 160, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Suchankova, P; Yan, J; Schwandt, ML; Stangl, BL; Caparelli, EC; Momenan, R; et al. The glucagon-like peptide-1 receptor as a potential treatment target in alcohol use disorder: evidence from human genetic association studies and a mouse model of alcohol dependence. Transl Psychiatry 2015, 5(6), e583. [Google Scholar] [CrossRef]
- Aranäs, C; Edvardsson, CE; Shevchouk, OT; Zhang, Q; Witley, S; Blid Sköldheden, S; et al. Semaglutide reduces alcohol intake and relapse-like drinking in male and female rats. EBioMedicine 2023, 93, 104642. [Google Scholar] [CrossRef]
- Hoffman, PL; de Guglielmo, G; Vengeliene, V; Kunze, W; Lebonville, CL; Swinny, JD; et al. Engaging Gut-to-Brain Signalling to Treat Alcohol Use Disorder. Addict Biol. 2026, 31(3), e70144. [Google Scholar] [CrossRef]
- Young, WG; Deutsch, JA. The construction, surgical implantation, and use of gastric catheters and a pyloric cuff. J Neurosci Methods 1981, 3(4), 377–84. [Google Scholar] [CrossRef]
- Rauhofer, EA; Smith, GP; Gibbs, J. Acute blockade of gastric emptying and meal size in rats. Physiol Behav. 1993, 54(5), 881–4. [Google Scholar] [CrossRef]
- Erickson, CK. Lowering of blood ethanol by activated carbon products in rats and dogs. Alcohol [Internet] 1993, 10(2), 103–7. Available online: https://www.sciencedirect.com/science/article/pii/0741832993900886. [CrossRef] [PubMed]
- Matsuda, H; Shimoda, H; Ninomiya, K; Yoshikawa, M. INHIBITORY MECHANISM OF COSTUNOLIDE, A SESQUITERPENE LACTONE ISOLATED FROM LAURUS NOBILIS, ON BLOOD-ETHANOL ELEVATION IN RATS: INVOLVEMENT OF INHIBITION OF GASTRIC EMPTYING AND INCREASE IN GASTRIC JUICE SECRETION. Alcohol Alcohol [Internet] Available from. 2002, 37(2), 121–7. [Google Scholar] [CrossRef] [PubMed]
- Colombo, G; Agabio, R; Lobina, C; Reali, R; Morazzoni, P; Bombardelli, E; et al. Salvia miltiorrhiza Extract Inhibits Alcohol Absorption, Preference, and Discrimination in sP Rats. Alcohol [Internet] 1999, 18(1), 65–70. Available online: https://www.sciencedirect.com/science/article/pii/S074183299800069X. [CrossRef]
- Lee, YJ; Pantuck, CB; Pantuck, EJ. Effect of ginseng on plasma levels of ethanol in the rat. Planta Med. 1993, 59(1), 17–9. [Google Scholar] [CrossRef]
- Crabbe, JC; Metten, P; Savarese, AM; Ozburn, AR; Schlumbohm, JP; Spence, SE; et al. Ethanol Conditioned Taste Aversion in High Drinking in the Dark Mice. Brain Sci. 2019, 9(1). [Google Scholar] [CrossRef] [PubMed]
- Gimenez-Gomez, P; Le, T; Zinter, M; M’Angale, P; Duran-Laforet, V; Freels, TG; et al. Suppression of binge alcohol drinking by an inhibitory neuronal ensemble in the mouse medial orbitofrontal cortex. Nat Neurosci [Internet] Available from. 2025, 28(8), 1741–52. [Google Scholar] [CrossRef] [PubMed]
- McDougle, M; de Araujo, A; Singh, A; Yang, M; Braga, I; Paille, V; et al. Separate gut-brain circuits for fat and sugar reinforcement combine to promote overeating. Cell Metab [Internet] 2024, 36(2), 393–407.e7. Available online: https://www.sciencedirect.com/science/article/pii/S1550413123004667. [CrossRef] [PubMed]
| Pharmacotherapy | Primary target | Reward component(s) plausibly modified | Translational observation |
|---|---|---|---|
| CRF1 receptor antagonists | CRF1 blockade (stress circuitry). | Post-oral state (stress/withdrawal); pre-oral (stress-triggered seeking); modulatory stress responsivity (vapor-dependent configuration). | Clinical efficacy: no meaningful benefit on craving/drinking despite target engagement in trials. Preclinical efficacy: effective in vapor/withdrawal-dependent escalation; minimal effects on baseline drinking. |
| Histamine H3 inverse agonist | H3 receptor inverse agonism (increase histaminergic tone; arousal/feeding modulation). | Post-oral peripheral; post-oral central drug (neuromodulatory gain); oral; pre-oral conditioned. | Clinical efficacy: no reduction in heavy drinking days; Preclinical efficacy: reduced intake/seeking across operant/relapse-like procedures (incl. vapor paradigms). |
| Naltrexone | µ-opioid receptor antagonism. | Oral; post-oral central drug. | Clinical efficacy: modest, reproducible reductions in heavy drinking in subsets. Preclinical efficacy: reduces ethanol intake, but also intake of palatable non-alcohol rewards (incl. sham-feeding paradigms). |
| Disulfiram | ALDH inhibition (acetaldehyde accumulation; aversive state). | Post-oral aversive constraint; pre-oral (avoidance/adherence expectancy); Post-oral central drug (DBH inhibition); Stress/arousal modulation. | Clinical efficacy: efficacy strongly depends on supervision/adherence; limited unsupervised durability. Preclinical efficacy: history/procedure dependent; can lose efficacy after chronic exposure in some lines. |
| Acamprosate | Withdrawal-state stabilization (E/I modulation, calcium-dependent; mechanism unresolved). | Post-oral state (withdrawal/negative); pre-oral (reduced cue-driven relapse). | Clinical efficacy: moderate efficacy for abstinence/relapse prevention, especially detoxified individuals. Preclinical efficacy: more reliable in dependence/withdrawal-structured paradigms; variable effects on baseline intake. |
| GLP-1 receptor agonists | GLP-1 receptor activation (gut–brain satiety + reward-circuit modulation). | Post-oral peripheral (satiety/visceral state, gastric emptying); post-oral central drug (mesolimbic modulation); Oral (taste/food preference); pre-oral (cue responding). | Clinical efficacy: emerging signals (semaglutide: reduced craving/some drinking outcomes; exenatide mixed). Preclinical efficacy: reduces intake/operant responding and some relapse-like measures; often with parallel feeding effects depending on protocol. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




