Submitted:
31 March 2026
Posted:
01 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
Study Population
Diagnostic Evaluation
Treatment Approach
Target Volume Definition
Post-Treatment Evaluation and Follow-Up
Statistical Methods
Results
Patient, Tumor, and Treatment Characteristics
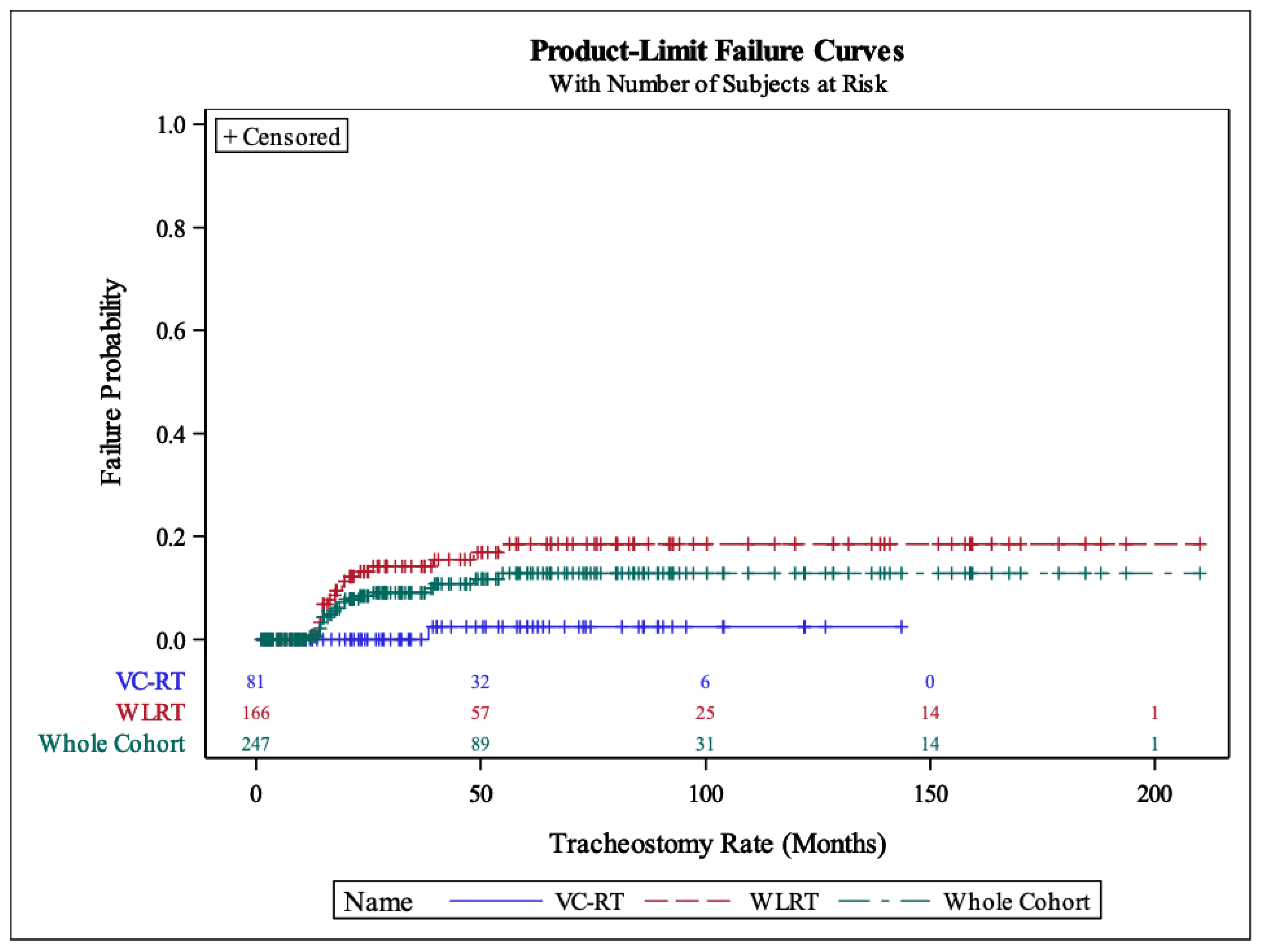
Tracheostomy Rate
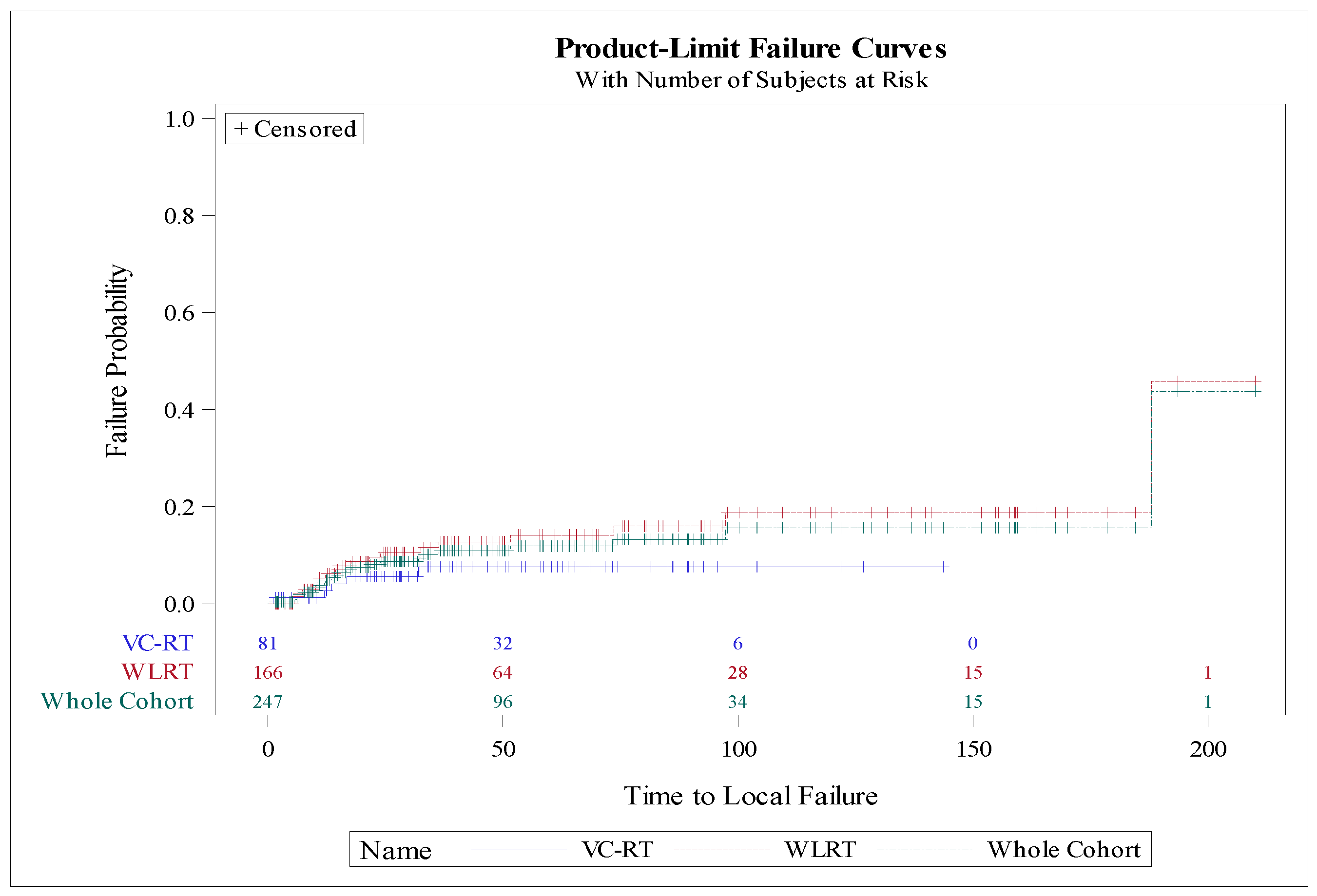
Local Failure
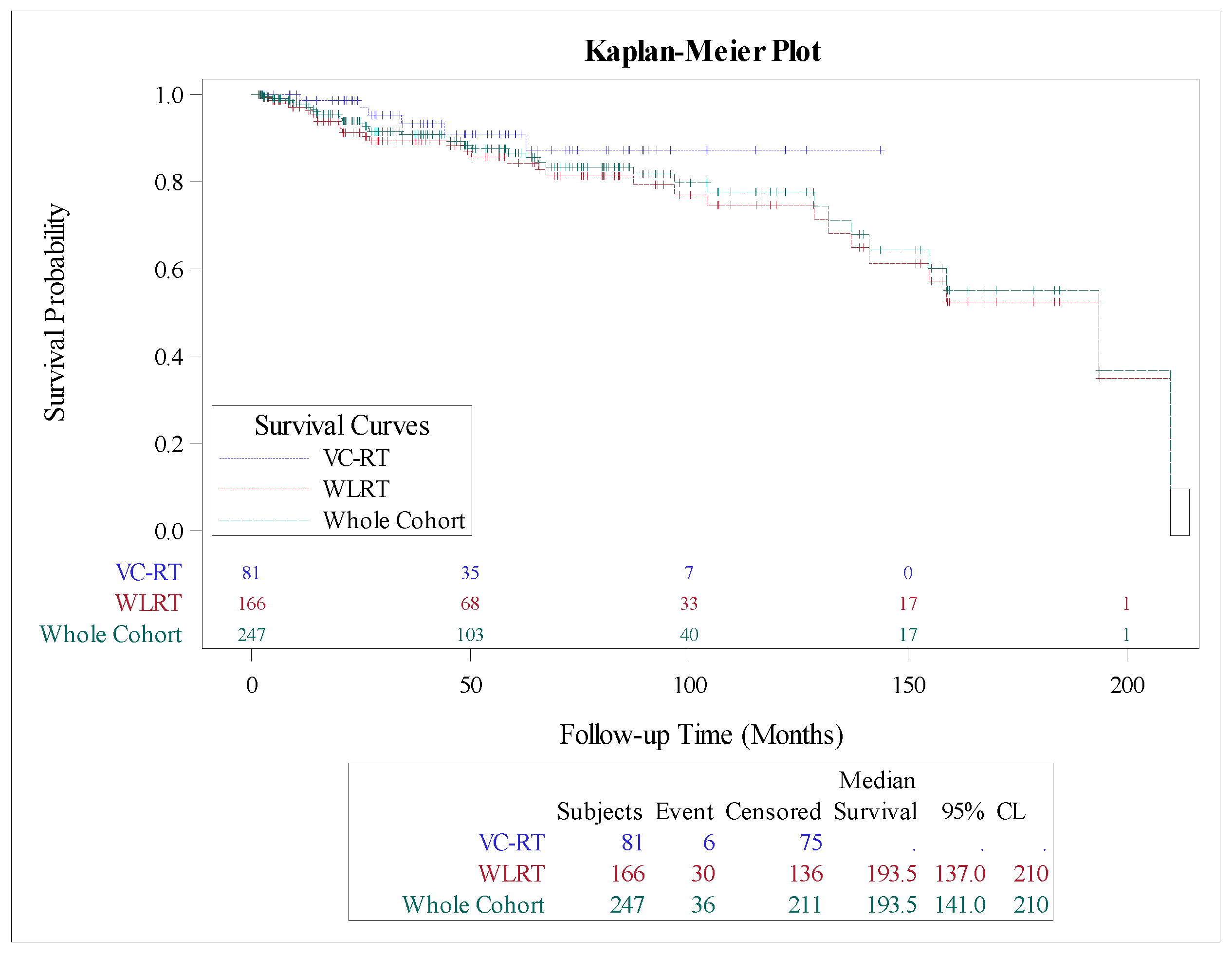
Overall Survival
Toxicity
Discussion
Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ESGC: Early-stage glottic cancer |
| WLRT: Whole-larynx radiotherapy |
| VC-RT: Vocal cord–only radiotherapy |
| LC: Local control |
| OS : Overall survival |
References
- Peng PY, Lin CY, Liao CT, Wang HM, Huang SF, Tsai Y Te, et al. Outcome of T2 Glottic Cancer Treated with Radiotherapy Alone or Concurrent Chemo-Radiotherapy. Cancers (Basel) [Internet]. 2025 Feb 1 [cited 2026 Mar 16];17(4). Available from: https://pubmed.ncbi.nlm.nih.gov/40002305/. [CrossRef]
- Kinshuck AJ, Shenoy A, Jones TM. Voice outcomes for early laryngeal cancer. Curr Opin Otolaryngol Head Neck Surg [Internet]. 2017 Jun 1 [cited 2026 Mar 16];25(3):211–6. Available from: https://pubmed.ncbi.nlm.nih.gov/28277333/. [CrossRef]
- Fink DS, McWhorter AJ. Trans-oral laser microsurgery in the management of glottic cancer. Oper Tech Otolaryngol - Head Neck Surg [Internet]. 2024 Jun 1 [cited 2026 Mar 16];35(2):149–57. Available from: https://www.optecoto.com/action/showFullText?pii=S1043181024000307. [CrossRef]
- Cetinayak O, Dogan E, Kuru A, Akturk N, Aydin B, Umay C, et al. Outcome of Early-Stage Glottic Laryngeal Carcinoma Patients Treated with Radical Radiotherapy Using Different Techniques. J Oncol [Internet]. 2019 [cited 2026 Mar 16];2019. Available from: https://pubmed.ncbi.nlm.nih.gov/31781218/. [CrossRef]
- Adeel M, Faisal M, Rashid A, Rasheed S, Hussain R, Malik KI, et al. Outcomes of definitive radiotherapy for early laryngeal cancer in terms of survival and patterns of failure. J Laryngol Otol [Internet]. 2019 [cited 2026 Mar 16];133(12):1087–91. Available from: https://pubmed.ncbi.nlm.nih.gov/31791441/. [CrossRef]
- Yamazaki H, Nishiyama K, Tanaka E, Koizumi M, Chatani M. Radiotherapy for early glottic carcinoma (T1N0M0): Results of prospective randomized study of radiation fraction size and overall treatment time. Int J Radiat Oncol Biol Phys [Internet]. 2006 Jan 1 [cited 2025 Apr 28];64(1):77–82. Available from: https://pubmed.ncbi.nlm.nih.gov/16169681/. [CrossRef]
- Bahig H, Nguyen-Tan PF, Filion É, Roberge D, Thanomsack P, de Guise J, et al. Larynx motion considerations in partial larynx volumetric modulated arc therapy for early glottic cancer. J Med Imaging Radiat Oncol [Internet]. 2017 Oct 1 [cited 2026 Mar 16];61(5):666–73. [CrossRef]
- Mulholland GB, Zhang H, Nguyen NTA, Tkacyzk N, Seikaly H, O’Connell D, et al. Optimal detection of hypothyroidism in early stage laryngeal cancer treated with radiotherapy. J Otolaryngol Head Neck Surg [Internet]. 2015 [cited 2026 Mar 16];44(1). Available from: https://pubmed.ncbi.nlm.nih.gov/26362315/. [CrossRef]
- Tans L, Al-Mamgani A, Kwa SLS, Elbers JBW, Keskin-Cambay F, Sewnaik A, et al. Single vocal cord irradiation for early-stage glottic cancer: Excellent local control and favorable toxicity profile. Oral Oncol. 2022 Apr 1;127. [CrossRef]
- Ding C, Lee P, Jiang SB. SU-E-T-233: Cyberknife Versus Linac IMRT for Dose Comparision in Hypofractionated Hemi Larynx Irradiation of Early Stage True Vocal Cord Cancer: A Dosimetric Study. Med Phys. 2015 Jun;42(6Part15):3386–3386. [CrossRef]
- Bicakci BC, Mustafayev TZ, Cetinayak O, Igdem S, Birgi SD, Meydan D, et al. Outcomes of carotid sparing intensity-modulated radiotherapy for early stage glottic cancer in 201 patients: Multicenter study of Turkish Radiation Oncology Society/TROD-01-007. Head Neck. 2022 Aug 1;44(8):1825–32. [CrossRef]
- Brouwer CL, Steenbakkers RJHM, Bourhis J, Budach W, Grau C, Grégoire V, et al. CT-based delineation of organs at risk in the head and neck region: DAHANCA, EORTC, GORTEC, HKNPCSG, NCIC CTG, NCRI, NRG Oncology and TROG consensus guidelines. Radiother Oncol [Internet]. 2015 Oct 1 [cited 2026 Mar 16];117(1):83–90. Available from: https://pubmed.ncbi.nlm.nih.gov/26277855/. [CrossRef]
- Mucha-Małecka A, Chrostowska A, Urbanek K, Małecki K. Prognostic factors in patients with T1 glottic cancer treated with radiotherapy. Strahlenther Onkol [Internet]. 2019 Sep 1 [cited 2026 Mar 16];195(9):792–804. Available from: https://pubmed.ncbi.nlm.nih.gov/31214734/. [CrossRef]
- Rosenthal DI, Fuller CD, Barker JL, Mason B, Garcia JA, Lewin JS, et al. Simple Carotid-Sparing Intensity-Modulated Radiotherapy Technique and Preliminary Experience for T1-2 Glottic Cancer. Int J Radiat Oncol Biol Phys [Internet]. 2010 Jun 1 [cited 2026 Mar 16];77(2):455–61. Available from: https://pubmed.ncbi.nlm.nih.gov/19679406/. [CrossRef]
- Tali TA, Amin F, Dar JA, Sofi SR, Sofi MA, Khan NA, et al. Treatment Outcome of Early-stage Laryngeal Carcinoma Treated with Radical Radiotherapy Using Different Techniques: A Retrospective Study. Asian Pacific J Cancer Care. 2023;8(2):269–73. [CrossRef]
- Amin MB, Greene FL, Edge SB, Compton CC, Gershenwald JE, Brookland RK, et al. The Eighth Edition AJCC Cancer Staging Manual: Continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J Clin [Internet]. 2017 Mar [cited 2026 Mar 16];67(2):93–9. Available from: https://pubmed.ncbi.nlm.nih.gov/28094848/. [CrossRef]
- Mohamad I, Salem A, Abu Hejleh T, Wadi-Ramahi S, Qandeel M, El Khatib S, et al. Peer-assisted Learning: Intensity-modulated Radiotherapy Transition in Developing Countries. Clin Oncol [Internet]. 2017 Oct 1 [cited 2026 Mar 17];29(10):689–95. Available from: https://pubmed.ncbi.nlm.nih.gov/28647432/. [CrossRef]
- Naunheim MR, Garneau J, Park C, Carroll L, Goldberg L, Woo P. Voice Outcomes After Radiation for Early-Stage Laryngeal Cancer. J Voice [Internet]. 2020 May 1 [cited 2026 Mar 17];34(3):460–4. Available from: https://pubmed.ncbi.nlm.nih.gov/30611594/. [CrossRef]
- Razavian NB, D’Agostino RB, Shenker RF, Hughes RT. Intensity-Modulated Radiation Therapy for Early-Stage Squamous Cell Carcinoma of the Glottic Larynx: A Systematic Review and Meta-Analysis. Int J Radiat Oncol Biol Phys [Internet]. 2023 Nov 1 [cited 2026 Mar 17];117(3):652–63. Available from: https://pubmed.ncbi.nlm.nih.gov/37150263/. [CrossRef]
- Linden SML, Philippens MEP, Sher DJ, Sanders ME, de Bree R, Rijken JA, et al. Risk of dysfunctional larynx after radiotherapy for early-stage glottic laryngeal cancer: A systematic review and meta-analysis. Radiother Oncol [Internet]. 2026 Jan 1 [cited 2026 Mar 17];214. Available from: https://researchinformation.umcutrecht.nl/en/publications/risk-of-dysfunctional-larynx-after-radiotherapy-for-early-stage-g/fingerprints/. [CrossRef]
- Yamazaki H, Suzuki G, Nakamura S, Yoshida K, Konishi K, Teshima T, et al. Radiotherapy for laryngeal cancer-technical aspects and alternate fractionation. J Radiat Res [Internet]. 2017 Jul 1 [cited 2026 Mar 17];58(4):495–508. Available from: https://pubmed.ncbi.nlm.nih.gov/28898958/. [CrossRef]
- Bahig H, Rosenthal DI, Nguyen-Tan FP, Fuller DC, Yuan Y, Hutcheson KA, et al. Vocal-cord Only vs. Complete Laryngeal radiation (VOCAL): a randomized multicentric Bayesian phase II trial. BMC Cancer [Internet]. 2021 Dec 1 [cited 2026 Mar 17];21(1). Available from: https://pubmed.ncbi.nlm.nih.gov/33888069/. [CrossRef]
- Gangsaas A, Astreinidou E, Quint S, Levendag PC, Heijmen B. Cone-beam computed tomography-guided positioning of laryngeal cancer patients with large interfraction time trends in setup and nonrigid anatomy variations. Int J Radiat Oncol Biol Phys [Internet]. 2013 Oct 1 [cited 2026 Mar 17];87(2):401–6. Available from: https://pubmed.ncbi.nlm.nih.gov/23958149/. [CrossRef]
- Al-Mamgani A, Kwa SLS, Tans L, Moring M, Fransen D, Mehilal R, et al. Single Vocal Cord Irradiation: Image Guided Intensity Modulated Hypofractionated Radiation Therapy for T1a Glottic Cancer: Early Clinical Results. Int J Radiat Oncol Biol Phys [Internet]. 2015 Oct 1 [cited 2026 Mar 17];93(2):337–43. Available from: https://pubmed.ncbi.nlm.nih.gov/26264629/. [CrossRef]
- Megwalu UC, Sikora AG. Survival outcomes in advanced laryngeal cancer. JAMA Otolaryngol Head Neck Surg [Internet]. 2014 Sep 1 [cited 2026 Mar 17];140(9):855–60. Available from: https://pubmed.ncbi.nlm.nih.gov/25144163/. [CrossRef]
- Pakkanen P, Irjala H, Ilmarinen T, Halme E, Lindholm P, Mäkitie A, et al. Survival and Larynx Preservation in Early Glottic Cancer: A Randomized Trial Comparing Laser Surgery and Radiation Therapy. Int J Radiat Oncol Biol Phys [Internet]. 2022 May 1 [cited 2026 Mar 17];113(1):96–100. Available from: https://pubmed.ncbi.nlm.nih.gov/35164976/. [CrossRef]
- Hut AR, Boia ER, Para D, Iovanescu G, Horhat D, Mikša L, et al. Laryngeal Cancer in the Modern Era: Evolving Trends in Diagnosis, Treatment, and Survival Outcomes. J Clin Med [Internet]. 2025 May 1 [cited 2026 Mar 17];14(10). Available from: https://pubmed.ncbi.nlm.nih.gov/40429363/. [CrossRef]
- MacKenzie R, Franssen E, Balogh J, Birt D, Gilbert R. The Prognostic Significance of Tracheostomy in Carcinoma of the Larynx Treated With Radiotherapy and Surgery for Salvage. Int J Radiat Oncol [Internet]. 1998 Apr 1 [cited 2026 Mar 17];41(1):43–51. Available from: https://www.sciencedirect.com/science/article/abs/pii/S0360301698000303. [CrossRef]
- Mendenhall WM, Strojan P, Lee AWM, Rinaldo A, Eisbruch A, Ng WT, et al. Radiotherapy in the management of glottic squamous cell carcinoma. Head Neck [Internet]. 2020 Dec 1 [cited 2026 Mar 17];42(12):3558–67. Available from: https://pubmed.ncbi.nlm.nih.gov/32896071/. [CrossRef]
- Sanguineti G, Adapala P, Endres EJ, Brack C, Fiorino C, Sormani MP, et al. Dosimetric Predictors of Laryngeal Edema. Int J Radiat Oncol Biol Phys [Internet]. 2007 Jul 1 [cited 2026 Mar 16];68(3):741–9. Available from: https://pubmed.ncbi.nlm.nih.gov/17398024/. [CrossRef]
- Rancati T, Fiorino C, Sanguineti G. NTCP modeling of subacute/late laryngeal edema scored by fiberoptic examination. Int J Radiat Oncol Biol Phys [Internet]. 2009 Nov 1 [cited 2026 Mar 28];75(3):915–23. Available from: https://pubmed.ncbi.nlm.nih.gov/19747783/. [CrossRef]
- Inoue T, Inoue T, Chatani M, Teshima T. Irradiated volume and arytenoid edema after radiotherapy for T1 glottic carcinoma. 1992 [cited 2026 Mar 28];168(1):23–6. Available from: https://inis.iaea.org/records/22xzc-9ew60.
- Elsharief MM, Hassouna AH, Shouman T, Mosallam A, El-Badrawy A, Shawky A, et al. Single vocal cord irradiation (SVCI) vs whole laryngeal radiotherapy in the treatment of T1aN0 glottic cancer A prospective randomized trial. Radiother Oncol [Internet]. 2026 Mar [cited 2026 Mar 28];216:111376. Available from: https://pubmed.ncbi.nlm.nih.gov/41520731/. [CrossRef]
- Zumsteg ZS, Riaz N, Jaffery S, Hu M, Gelblum D, Zhou Y, et al. Carotid sparing intensity-modulated radiation therapy achieves comparable locoregional control to conventional radiotherapy in T1-2N0 laryngeal carcinoma. Oral Oncol [Internet]. 2015 Jul 1 [cited 2026 Mar 17];51(7):716–23. Available from: https://pubmed.ncbi.nlm.nih.gov/25958831/. [CrossRef]
- Treechairusame T, Dee EC, Cao C, Wu Y, Yu Y, Gelblum D, et al. Partial vs Whole Laryngeal Radiotherapy for Clinical Stage T1-2N0M0/Tis Laryngeal Carcinoma. JAMA Otolaryngol Head Neck Surg [Internet]. 2025 Nov 13 [cited 2026 Mar 28];151(11):1071–9. Available from: https://pubmed.ncbi.nlm.nih.gov/41037280/. [CrossRef]
- Plummer C, Henderson RD, O’Sullivan JD, Read SJ. Ischemic stroke and transient ischemic attack after head and neck radiotherapy: A review. Stroke [Internet]. 2011 Sep [cited 2026 Mar 16];42(9):2410–8. [CrossRef]
- Swisher-Mcclure S, Mitra N, Lin A, Ahn P, Wan F, O’Malley B, et al. Risk of fatal cerebrovascular accidents after external beam radiation therapy for early-stage glottic laryngeal cancer. Head Neck [Internet]. 2014 May 1 [cited 2026 Mar 16];36(5):611–6. [CrossRef]
- Chera BS, Amdur RJ, Morris CG, Mendenhall WM. Carotid-sparing intensity-modulated radiotherapy for early-stage squamous cell carcinoma of the true vocal cord. Int J Radiat Oncol Biol Phys [Internet]. 2010 Aug 1 [cited 2026 Mar 17];77(5):1380–5. Available from: https://pubmed.ncbi.nlm.nih.gov/19942356/.
- Mohamad I, Abu Taha S, Bushehri A, Youssef B, Ozyar E, Alotain I, et al. Global Disparities and Trends in Radiotherapy for Early-Stage Glottic Cancer. 2026 Mar 9 [cited 2026 Mar 28]; Available from: https://www.preprints.org/manuscript/202603.0691/v1.
| Characteristic | Total (n=247) | WLRT (n=166) | VC-RT (n=81) | P value |
|---|---|---|---|---|
| Follow-up, median (range), (months) | 38.2(1.61-192) | 42.1(1.6-194) | 43.4(1.6-144) | 0.21 |
| Age median (range), (years) | 59(30-92) | 50(30-92) | 57(32-90) | 0.19 |
| Gender | ||||
| Female | 6(2.4%) | 3(1.8%) | 3(3.7%) | 0.397 |
| Male | 241 (97.6%) | 163(98.2%) | 78(96.3%) | |
| Performance Status (ECOG) | ||||
| 0 | 210(85%) | 150(90.4%) | 60(74.1%) | 0.002 |
| 1 | 34(13.8%) | 15(9%) | 19(23.4%) | |
| 2 | 3(1.2%) | 1(0.06%) | 2(2.5%) | |
| Smoking History | ||||
| No | 52(21.1%) | 29(17.5%) | 23(28.4%) | 0.048 |
| Yes | 195(78.9%) | 137(82.5%) | 58(71.6%) | |
| Alcohol Use | ||||
| No | 236(95.5%) | 159(95.8%) | 77(95.1%) | 0.754 |
| Yes | 11(4.5%) | 7(4.2%) | 4(4.9%) | |
| cT- category | ||||
| Tis | 3 (1.2%) | 3 (1.8%) | 0 | <0.001 |
| T1a | 200 (81.0%) | 137 (82.5%) | 63 (77.8%) | |
| T1b | 17 (6.9%) | 17 (10.2%) | 0 | |
| T2 | 27 (10.9%) | 9 (5.4%) | 18 (22.2%) | |
| Overall Stage group | ||||
| Stage 0 | 3 (1.2%) | 3 (1.8%) | 0 | <0.001 |
| Stage I | 217 (87.9%) | 154 (92.8%) | 63 (77.8%) | |
| Stage II | 27 (10.9%) | 9 (5.4%) | 18 (22.2%) | |
| Radiotherapy Technique | ||||
| 2D RT | ||||
| 3DCRT | 144 (58.3%) | 119 (71.7%) | 25 (30.9%) | <0.001 |
| IMRT | 103 (41.7%) | 47 (28.3%) | 56 (69.1%) | |
| Fractionation | ||||
| Conventional | ||||
| 66Gy/33fraction/5 fractions/week | 29(80.6%) | 4(57.1%) | 25(86.2%) | 0.001 |
| 70Gy/35frcations/5 fractions/week | 7(19.4%) | 3(42.9%) | 4(13.8%) | |
| Accelerated Conventional | ||||
| 66Gy/33fraction/6 fractions/week | 26(89.7%) | 17(89.5%) | 9(90.0%) | |
| 70Gy/35frcations/6 fractions/week | 3(10.3%) | 2(10.5%) | 1(10.0%) | |
| Accelerated Hypofractionation | ||||
| 51Gy/20fractions/5fraction/week | 2(1.2%) | 2(5.0%) | 0 | |
| 60Gy/25fractions/5fractions/week | 8(4.9%) | 1(2.5%) | 7(5.7%) | |
| 63Gy/28fractions/5 fractions/week | 138(85.2%) | 26(65.0%) | 112(91.8%) | |
| 65.25Gy/29 fractions/5 fractions/week | 14(8.6%) | 11(27.5%) | 3(2.5%) | |
| Tracheostomy, irrespective of the underlying cause | ||||
| No | 211(85.4%) | 132(79.5%) | 79(97.5%) | 0.001 |
| Yes | 36(14.6%) | 34(20.5%) | 2(2.5%) | |
| Tracheostomy, based on the underlying cause | ||||
| Edema | 20(100%) | 20(100%) | 0 | 0.001 |
| Recurrence | 211(85.4%) | 132(79.5%) | 79(97.5%) | |
| Salvage laryngectomy | ||||
| No | 227(91.9%) | 150(90.4%) | 77(95.1%) | 0.23 |
| Yes | 20(8.1%) | 16(16.6%) | 4(4.9%) | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




