Submitted:
31 March 2026
Posted:
02 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Portable Electrochemical Sensor Platform
2.2. Electrode Preparation and Electrochemical Characterization
2.3. Telemedicine Integration in Simulated Gynecology Workflows
3. Results
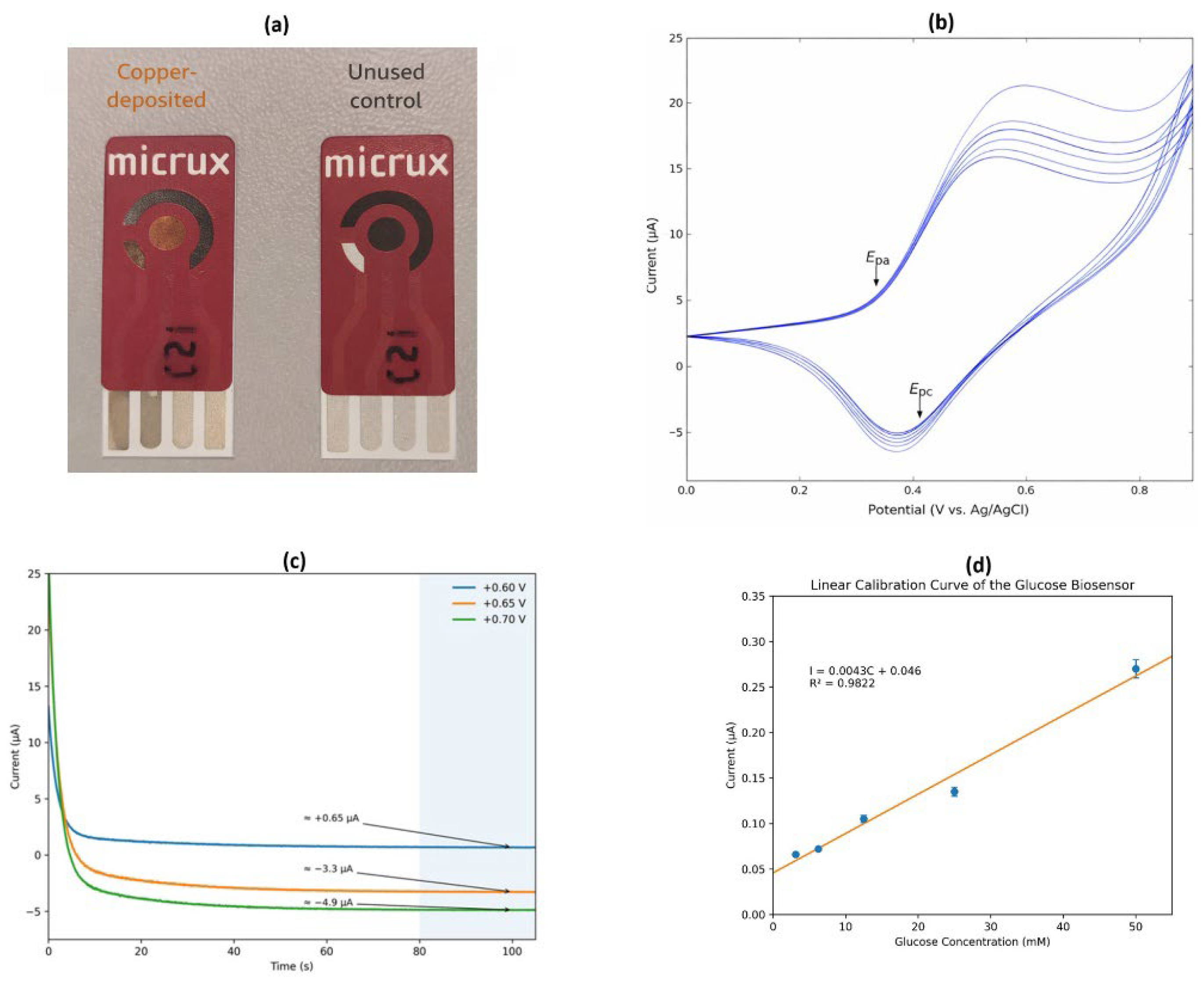
3.1. Electrochemical Performance of the Cu-Modified Electrode
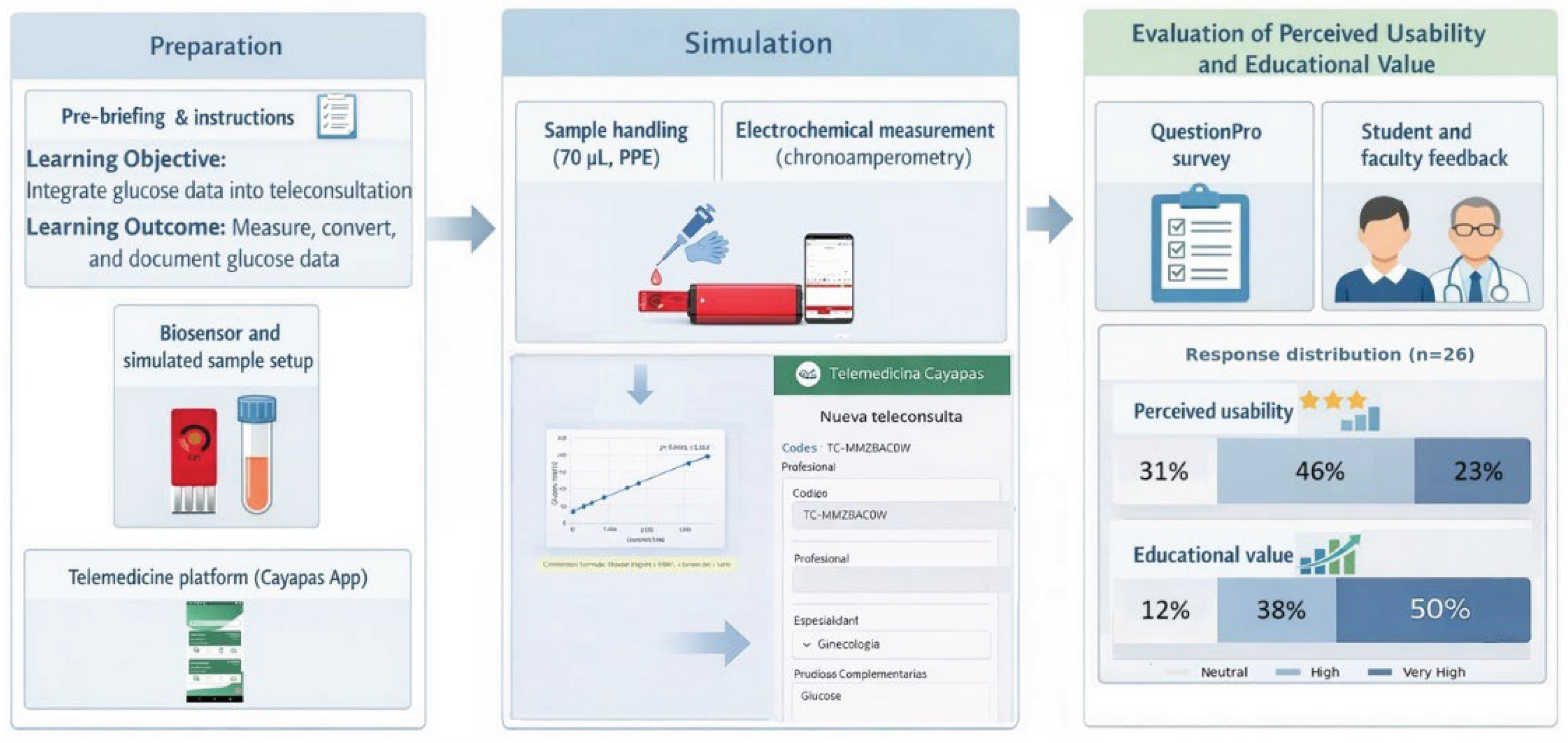
3.2. Results of Telemedicine Integration in Simulated Gynecology Workflows
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Ag | Silver |
| Ag/AgCl | Silver/Silver Chloride |
| AE | Auxiliary Electrode |
| CNTs | Carbon Nanotubes |
| CuO | Copper(II) Oxide |
| CV | Cyclic Voltammetry |
| LOD | Limit of Detection |
| LOQ | Limit of Quantification |
| MOF | Metal–Organic Framework |
| NaOH | Sodium Hydroxide |
| PBS | Phosphate-Buffered Saline |
| RE | Reference Electrode |
| RSD | Relative Standard Deviation |
| WE | Working Electrode |
| CA | Chronoamperometry |
| SPCE | Screen-Printed Carbon Electrode |
| GUI | Graphical User Interface |
| R2 | Coefficient of Determination |
| mg/dL | Milligrams per Deciliter |
References
- International Diabetes Federation, I. IDF Diabetes Atlas Available online: https://diabetesatlas.org/ (accessed on 19 March 2026).
- Genitsaridi, I.; Ke, C.; Salim, A.; Sajjadi, S.F.; Tomic, D.; James, S.; Thirunavukkarasu, S.; Issaka, A.; Chen, L.; Basit, A.; et al. 11th Edition of the IDF Diabetes Atlas: Global, Regional, and National Diabetes Prevalence Estimates for 2024 and Projections for 2050. The Lancet Diabetes & Endocrinology 2025, 14, 149. [CrossRef]
- Félix, C.; López-Flecher, M.; Vega, M.; Andrango, K.; Andrango, S.; Parise-Vasco, J.M.; Angamarca-Iguago, J.; Simancas-Racines, D.; López-Jaramillo, P.; Bangdiwala, S.I.; et al. Self-Reported Prevalence of Chronic Non-Communicable Diseases Concerning Socioeconomic and Educational Factors: Analysis of the PURE-Ecuador Cohort. Global Heart 2025, 20, 29. [CrossRef]
- Tufiño, A.; Teneda, A.C.C. The Ecuadorian Health System by 2023. Medwave 2023, 23. [CrossRef]
- Pokrywka, P.; Kamińska, H.; Jarosz-Chobot, P. Importance of Simulation-Based Medical Education and Its Application in Diabetology. Pediatric Endocrinology Diabetes and Metabolism 2024, 30, 45. [CrossRef]
- Karachaliou, F.; Simatos, G.; Simatou, A. The Challenges in the Development of Diabetes Prevention and Care Models in Low-Income Settings. Frontiers in Endocrinology 2020, 11, 518. [CrossRef]
- Wei, M.; Qiao, Y.; Zhao, H.; Liang, J.; Li, T.; Luo, Y.; Lu, S.; Shi, X.; Lu, W.; Sun, X. Electrochemical Non-Enzymatic Glucose Sensors: Recent Progress and Perspectives. Chemical Communications 2020, 56, 14553. [CrossRef]
- Sajeevan, A.; Sukumaran, R.A.; Panicker, L.R.; Goud, K.Y. Trends in Ready-to-Use Portable Electrochemical Sensing Devices for Healthcare Diagnosis. Microchimica Acta 2025, 192, 80. [CrossRef]
- Saha, T.; Caño, R.D.; Mahato, K.; Paz, E.D. la; Chen, C.; Ding, S.; Yin, L.; Wang, J. Wearable Electrochemical Glucose Sensors in Diabetes Management: A Comprehensive Review. Chemical Reviews 2023, 123, 7854. [CrossRef]
- Govindaraj, M.; Srivastava, A.; Muthukumaran, M.K.; Tsai, P.; Lin, Y.; Raja, B.K.; Rajendran, J.; Ponnusamy, V.K.; Selvi, J.A. Current Advancements and Prospects of Enzymatic and Non-Enzymatic Electrochemical Glucose Sensors. International Journal of Biological Macromolecules 2023, 253, 126680.
- Jiang, D.; Liu, T.; Chu, Z.; Wang, Y. Advances in Nanostructured Material-Based Non-Enzymatic Electrochemical Glucose Sensors. Analytical Methods 2023, 15, 6344. [CrossRef]
- Phetsang, S.; Kidkhunthod, P.; Chanlek, N.; Jakmunee, J.; Mungkornasawakul, P.; Ounnunkad, K. Copper/Reduced Graphene Oxide Film Modified Electrode for Non-Enzymatic Glucose Sensing Application. Scientific Reports 2021, 11, 9302. [CrossRef]
- Sridara, T.; Upan, J.; Saianand, G.; Tuantranont, A.; Karuwan, C.; Jakmunee, J. Non-Enzymatic Amperometric Glucose Sensor Based on Carbon Nanodots and Copper Oxide Nanocomposites Electrode. Sensors 2020, 20, 808. [CrossRef]
- Pour, S.R.S.; Calabria, D.; Emamiamin, A.; Lazzarini, E.; Pace, A.; Guardigli, M.; Zangheri, M.; Mirasoli, M. Electrochemical vs. Optical Biosensors for Point-of-Care Applications: A Critical Review. Chemosensors 2023, 11, 546.
- Xu, J.; Yan, Z.; Liu, Q. Smartphone-Based Electrochemical Systems for Glucose Monitoring in Biofluids: A Review. Sensors 2022, 22, 5670. [CrossRef]
- Campuzano, S.; Montiel, V.R.; Torrente-Rodríguez, R.M.; Pingarrón, J.M. Bioelectroanalytical Technologies for Advancing the Frontiers To Democratize Personalized Desired Health. Analytical Chemistry 2025, 97, 11371. [CrossRef]
- Martins, D.; Lewerenz, S.; Carmo, A.; Martins, H. Interoperability of Telemonitoring Data in Digital Health Solutions: A Scoping Review. Frontiers in Digital Health 2025, 7, 1502260. [CrossRef]
- Khan, A.; DeVoe, E.; Andreescu, S. Carbon-Based Electrochemical Biosensors as Diagnostic Platforms for Connected Decentralized Healthcare. Sensors & Diagnostics 2023, 2, 529. [CrossRef]
- El-Rashidy, N.; El–Sappagh, S.; Islam, S.M.R.; El-Bakry, H.M.; Abdelrazek, S. Mobile Health in Remote Patient Monitoring for Chronic Diseases: Principles, Trends, and Challenges. Diagnostics 2021, 11, 607. [CrossRef]
- Costa-Rama, E.; Fernández-Abedul, M.T. Paper-Based Screen-Printed Electrodes: A New Generation of Low-Cost Electroanalytical Platforms. Biosensors 2021, 11, 51. [CrossRef]
- MicruX Technologies. Screen-Printed Carbon Electrodes ED-S1PE-C21 – Technical Specifications. Available online: https://www.micruxfluidic.com (accessed on 10 March 2026).
- MicruX ECSens Bipot Guide, M. MicruX® ECSens® Bipot – Quick Installation Guide Available online: https://www.micruxfluidic.com (accessed on 16 March 2026).
- Gao, W.; Zhou, X.; Heinig, N.F.; Thomas, J.P.; Zhang, L.; Leung, K.T. Non-enzymatic Saliva-Range Glucose Sensing Using Electrodeposited Cuprous Oxide Nanocubes on a Graphene Strip. ACS Applied Nano Materials 2021, 4, 4790. [CrossRef]
- Jerigová, M.; Odziomek, M.; López-Salas, N. “We Are Here!” Oxygen Functional Groups in Carbons for Electrochemical Applications. ACS Omega 2022, 7, 11544. [CrossRef]
- Ahmed, J.; Islam, Md.N.; Faisal, M.; Algethami, J.S.; Rahman, M.M.; Maiyalagan, T.; Hasnat, M.A.; Harraz, F.A. Efficient Oxidation of Hydrazine over Electrochemically Activated Glassy Carbon Electrode Surface: Kinetics and Sensing Performance. Diamond and Related Materials 2024, 145, 111115. [CrossRef]
- Guati, C.; Gómez-Coma, L.; Fallanza, M.; Ortíz, I. Non-Enzymatic Amperometric Glucose Screen-Printed Sensors Based on Copper and Copper Oxide Particles. Applied Sciences 2021, 11, 10830. [CrossRef]
- Nurani, D.A.; Wibowo, R.; Fajri, I.F.E. Non-Enzymatic Glucose Sensor Based on Electrodeposited Copper on Carbon Paste Electrode (Cu/CPE). AIP conference proceedings 2016, 1729, 20056. [CrossRef]
- Yao, B.; Ren, X.; Feng, C.; Qiao, H.; Qi, X. Enhanced Glucose Sensing Performance of P-n Cu2O Homojunction Promoted by Cu(I)/Cu(III) Redox Process. Journal of Electroanalytical Chemistry 2023, 940, 117500. [CrossRef]
- Franco, F.F.; Hogg, R.A.; Manjakkal, L. Cu2O-Based Electrochemical Biosensor for Non-Invasive and Portable Glucose Detection. Biosensors 2022, 12, 174. [CrossRef]
- Vasquez-Cevallos, L.; Mitchell, A.; Muñoz-Hernández, S.; Herranz, Á.; García-Mingo, A.; Martin, P.; Castro, M.; Soto-Rodriguez, P.E.D.; Parrales–Bravo, F.; Caicedo–Quiroz, R. Educational and Clinical Applications of a Web- and Android-Based Telemedicine Platform to Expand Rural Health Care in Ecuador. Telemedicine Reports 2025, 6, 67. [CrossRef]
- Vasquez-Cevallos, L.; Bobokova, J.; González-Granda, P.V.; Iniesta-Chamorro, J.M.; Aguilera, E.J.G.; Hernando, M.E. Design and Technical Validation of a Telemedicine Service for Rural Healthcare in Ecuador. Telemedicine Journal and e-Health 2017, 24, 544. [CrossRef]
- Vasquez-Cevallos, L.; García-Mingo, A.; Martin, P.; Muñoz-Hernández, S.; Herranz, Á.; Caicedo–Quiroz, R.; Robalino, G.R.G.; Estrada, R. Technical Development and Effectiveness of a Telemedicine Service on Amazon Web Services (AWS) for Ecuador’s Rural Communities. In World Congress on Medical Physics and Biomedical Engineering, September 7 - 12, 2009, Munich, Germany; Springer Nature, 2025; p. 250.
- Quezada, C.A.I.; Pérez, J.I.F.; Mingo, A.G.; Martín, P. de C.S. Evaluation of a Telemedicine Tool for the Training of Rotating Interns. 2025, 1. [CrossRef]
- Vasquez-Cevallos, L.; Parpacen-Briones, L.; Del-Pino-Bazan, F.; Moran-Chaguay, H.; Diaz-Mora, P.; Zevallos, J.C.; Estrada, R.; Soto-Rodriguez, P.E.D.; Muñoz-Hernández, S.; Herranz, Á. Evaluation of Mixed Reality Technologies in Telemedicine-Assisted Childbirth Simulations. Procedia Computer Science 2024, 251, 438. [CrossRef]
- Rodríguez, K.A.M.; Benaglio, C.; Sepúlveda, J.I.O. Early Integration of Clinical Simulation in Medical Students: A Progressive Experience Using SimZones. Journal of Medical Education and Curricular Development 2025, 12. [CrossRef]
- Geetha, M.; Maurya, M.R.; Al-Máadeed, S.; Muthalif, A.A.; Sadasivuni, K.K. High-Precision Non-enzymatic Electrochemical Glucose Sensing Based on CNTs/CuO Nanocomposite. Journal of Electronic Materials 2022, 51, 4905. [CrossRef]
- Zhao, C.; Tang, X.; Zhao, J.; Cao, J.; Jiang, Z.; Qin, J. MOF Derived Core-Shell CuO/C with Temperature-Controlled Oxygen-Vacancy for Real Time Analysis of Glucose. Journal of Nanobiotechnology 2022, 20. [CrossRef]
- Koukouviti, E.; Plessas, A.K.; Pagkali, V.; Economou, A.; Papaefstathiou, G.S.; Kokkinos, C. 3D-Printed Electrochemical Glucose Device with Integrated Fe(II)-MOF Nanozyme. Microchimica Acta 2023, 190. [CrossRef]
- Jiménez-Rodríguez, A.; Sotelo, E.; Martı́nez, L.; Huttel, Y.; González, M.U.; Mayoral, Á.; García-Martín, J.M.; Videa, M.; Cholula-Díaz, J.L. Green Synthesis of Starch-Capped Cu2O Nanocubes and Their Application in the Direct Electrochemical Detection of Glucose. RSC Advances 2021, 11, 13711. [CrossRef]
- Bouamra, B.; Chakroun, K.; Bustos, E.M. de; Dobson, J.; Rouge, J.-A.; Moulin, T. Simulation-Based Teaching of Telemedicine for Future Users of Teleconsultation and Tele-Expertise: Feasibility Study. JMIR Medical Education 2021, 7. [CrossRef]
- Wu, J.; Liu, H.; Chen, W.; Ma, B.; Ju, H. Device Integration of Electrochemical Biosensors. Nature Reviews Bioengineering 2023, 1, 346. [CrossRef]
- Kim, J.; Jeong, J.; Ko, S.H. Electrochemical Biosensors for Point-of-Care Testing. Bio-Design and Manufacturing 2024, 7, 548. [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



