Submitted:
01 April 2026
Posted:
01 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
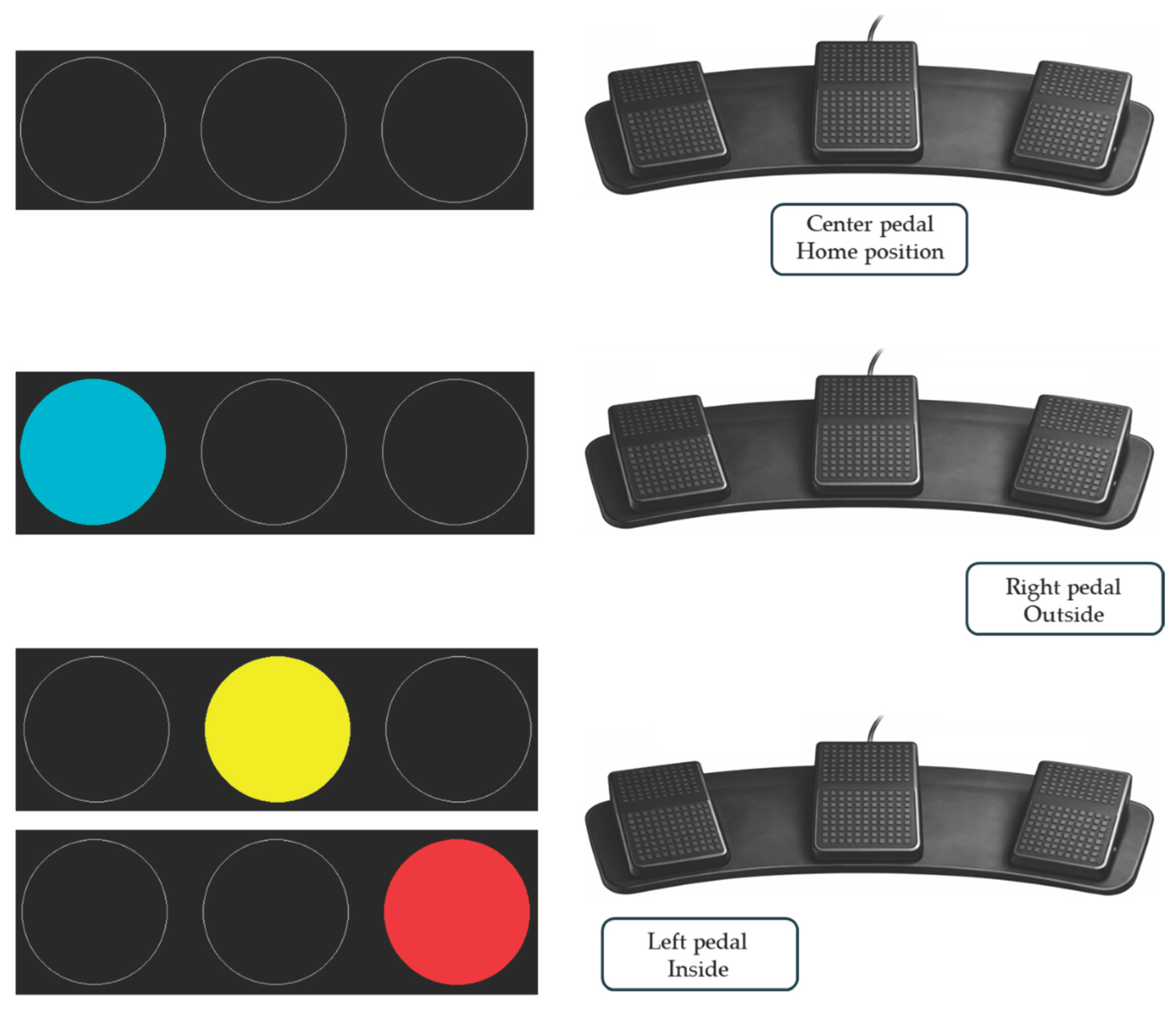
2.2. Foot PVT Procedure
2.3. Sleep Assessment
2.4. Calculation of PAL
2.5. Statistical Analysis
2.5.1. Within-Group Analysis
2.5.2. Between-Group Comparison
2.5.3. Comparison of Participant Characteristics
2.5.4. Correlation Analysis
2.5.5. Sample Size Sensitivity Analysis
3. Results
3.1. Descriptive Statistics of RT in Younger and Older Adults
3.2. Within-Group Analysis (Wilcoxon Signed-Rank Test)
3.3. Between-Group Comparison of dRT
3.4. Between-Group Comparison of Physical Characteristics
3.5. Associations Between dRT and Physical Characteristics and Sleep Measures
4. Discussion
4.1. Main Findings
4.2. Age-Related Slowing of RT
4.3. Dissociation Between Speed and Response Structure
4.4. Stability of Inside–Outside Asymmetry
4.5. Implications for Driving and Pedal Control
4.6. Preservation of Relative Response Relationships
4.7. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| RT | Reaction time |
| PVT | Psychomotor Vigilance Test |
| Foot PVT | Foot-response version of the PVT |
| dRT | difference between inside and outside RT |
References
- Hardwick, R.M.; Forrence, A.D.; Costello, M.G.; Zackowski, K.; Haith, A.M. Age-related increases in reaction time result from slower preparation, not delayed initiation. J. Neurophysiol. 2022, 128, 582–592. [Google Scholar] [CrossRef]
- Woods, D.L.; Wyma, J.M.; Yund, E.W.; Herron, T.J.; Reed, B. Factors influencing the latency of simple reaction time. Front. Hum. Neurosci. 2015, 9, 131. [Google Scholar] [CrossRef]
- Der, G.; Deary, I.J. Age and sex differences in reaction time in adulthood: Results from the United Kingdom Health and Lifestyle Survey. Psychol. Aging 2006, 21, 62–73. [Google Scholar] [CrossRef]
- Woods, D.L.; Wyma, J.M.; Yund, E.W.; Herron, T.J.; Reed, B. Age-related slowing of response selection and production in a visual choice reaction time task. Front. Hum. Neurosci. 2015, 9, 193. [Google Scholar]
- Salthouse, T.A. The processing-speed theory of adult age differences in cognition. Psychol. Rev. 1996, 103, 403–428. [Google Scholar] [CrossRef]
- Alves, I.I.P.S.; Bueno, G.A.S.; Elmescany, R.B.; Borges, L.A.; Pinto, D.K.; Martins, A.C.; Menezes, R.L. Motor reaction time, sarco penia and functional skills in elderly women: A cross-sectional study. J. Nutr. Health Aging 2023, 27, 878–884. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.N.; Xu, S.; Chai, Y.; Basner, M.; Dinges, D.F.; Rao, H. Sleep deprivation enhances inter-stimulus interval effect on vigilant attention performance. Sleep 2018, 41, zsy189. [Google Scholar] [CrossRef] [PubMed]
- Basner, M.; Dinges, D.F. Maximizing sensitivity of the psychomotor vigilance test (PVT) to sleep loss. Sleep 2011, 34, 581–591. [Google Scholar] [CrossRef]
- Basner, M.; Moore, T.M.; Nasrini, J.; Gur, R.C.; Dinges, D.F. Response speed measurements on the psychomotor vigilance test: How precise is precise enough? Sleep 2021, 44, zsaa121. [Google Scholar] [CrossRef] [PubMed]
- Chaisilprungraung, T.; Stekl, E.K.; Thomas, C.L.; Blanchard, M.E.; Hughes, J.D.; Balkin, T.J.; Doty, T.J. Quantifying the effects of sleep loss: Relative effect sizes of the psychomotor vigilance test, multiple sleep latency test, and maintenance of wakefulness test. Sleep Adv. 2022, 3, zpac034. [Google Scholar] [CrossRef]
- Jones, M.J.; Dunican, I.C.; Murray, K.; Peeling, P.; Dawson, B.; Halson, S.; Miller, J.; Eastwood, P.R. The psychomotor vigilance test: A comparison of different test durations in elite athletes. J. Sports Sci. 2018, 36, 2033–2037. [Google Scholar] [CrossRef]
- Yoshida, Y.; Yuda, E.; Yokoyama, K. Design of the New Foot Psychomotor Vigilance Test (PVT) for Screening Driving Ability. Hardware 2025, 3, 3. [Google Scholar] [CrossRef]
- Yoshida, Y.; Yokoyama, K. Comparison of Foot-Response Reaction Time Between Younger and Older Adults Using the Foot Psychomotor Vigilance Test. J. Ageing Longev. 2026, 6, 17. [Google Scholar] [CrossRef]
- Vallesi, A.; Tronelli, V.; Lomi, F.; Pezzetta, R. Age differences in sustained attention tasks: A meta-analysis. Psychon. Bull. Rev. 2021, 28, 1755–1775. [Google Scholar] [CrossRef]
- Lim, J.; Dinges, D.F. Sleep deprivation and vigilant attention. Ann. N. Y. Acad. Sci. 2008, 1129, 305–322. [Google Scholar] [CrossRef]
- Blomstrand, P.; Tesan, D.; Nylander, E.M.; Ramstrand, N. Mind body exercise improves cognitive function more than aerobic- and resistance exercise in healthy adults aged 55 years and older—An umbrella review. Eur. Rev. Aging Phys. Act. 2023, 20, 15. [Google Scholar] [CrossRef] [PubMed]
- Japan Organization of Better Sleep (JOBS). OSA Sleep Inventory MA Version. Available online: https://www.jobs.gr.jp/osa_ma.html (accessed on 1 March 2026).
- Okamura, H.; Mihara, K.; Tsuda, A.; Morisaki, T.; Tanaka, Y.; Shoji, Y. Subjective Happiness Is Associated with Objectively Eval uated Sleep Efficiency and Heart Rate during Sleep: An Exploratory Study Using Non-Contact Sheet Sensors. Sustainability 2020, 12, 4630. [Google Scholar] [CrossRef]
- Ainsworth, B.E.; Haskell, W.L.; Herrmann, S.D.; Meckes, N.; Bassett, D.R., Jr.; Tudor-Locke, C.; Greer, J.L.; Vezina, J.; Whitt-Glover, M.C.; Leon, A.S. 2011 Compendium of Physical Activities: A second update of codes and MET values. Med. Sci. Sports Exerc. 2011, 43, 1575–1581. [Google Scholar] [CrossRef]
- National Institutes of Biomedical Innovation; Health and Nutrition. Revised “List of Physical Activity METs (METs Table)”. Available online: https://www.nibn.go.jp/eiken/programs/2011mets.pdf (accessed on 1 March 2026).
- Ketcham, C.; Stelmach, G. Age-related declines in motor control. In Handbook of the Psychology of Aging; Birren, J.E., Schaie, K.W., Eds.; Academic Press: San Diego, CA, USA, 2001; pp. 313–348. [Google Scholar]
- Hunter, S.K.; Pereira, H.M.; Keenan, K.G. The aging neuromuscular system and motor performance. J. Appl. Physiol. 2016, 121, 982–995. [Google Scholar] [CrossRef]
- Levin, O.; Fujiyama, H.; Boisgontier, M.P.; Swinnen, S.P.; Summers, J.J. Aging and motor inhibition: A converging perspective provided by brain stimulation and imaging approaches. Neurosci. Biobehav. Rev. 2014, 43, 100–117. [Google Scholar] [CrossRef] [PubMed]
- Heuninckx, S.; Wenderoth, N.; Swinnen, S.P. Systems neuroplasticity in the aging brain: Recruiting additional neural resources for successful motor performance in elderly persons. J. Neurosci. 2008, 28, 91–99. [Google Scholar] [CrossRef]
- Seidler, R.D.; Bernard, J.A.; Burutolu, T.B.; Fling, B.W.; Gordon, M.T.; Gwin, J.T.; Kwak, Y.; Lipps, D.B. Motor control and aging: Links to age-related brain structural, functional, and biochemical effects. Neurosci. Biobehav. Rev. 2010, 34, 721–733. [Google Scholar] [CrossRef]
- Sleimen-Malkoun, R.; Temprado, J.J.; Berton, E. Age-related changes of movement patterns in discrete Fitts' task. BMC Neurosci. 2013, 14, 145. [Google Scholar] [CrossRef] [PubMed]
- Swinnen, S.P.; Wenderoth, N. Two hands, one brain: Cognitive neuroscience of bimanual skill. Trends Cogn. Sci. 2004, 8, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Carson, R.G. Neural pathways mediating bilateral interactions between the upper limbs. Brain Res. Brain Res. Rev. 2005, 49, 641–662. [Google Scholar] [CrossRef]
- van Opstal, A.J. Dynamic patterns: The self-organization of brain and behavior. J. Cogn. Neurosci. 1996, 8, 385–386. [Google Scholar] [CrossRef]
- Lee, H.C.; Cameron, D.; Lee, A.H. Assessing the driving performance of older adult drivers: On-road versus simulated driving. Accid. Anal. Prev. 2003, 35, 797–803. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Mizuno, K.; Nitta, Y.; Kovaceva, J.; Thomson, R. Comparing the responses and visual behaviors of older and younger drivers in car-to-cyclist collisions using a driving simulator. J. Saf. Res. 2025, 94, 254–264. [Google Scholar] [CrossRef]
- De Biase, M.E.M.; Alonso, A.C.; da Silva, R.N.; Soares, S.M.; Canonica, A.C.; Belini, A.P.D.R.; Soares-Junior, J.M.; Baracat, E.C.; Busse, A.L.; Jacob-Filho, W.; Brech, G.C.; Greve, J.M.D. Multifactorial assessment of braking time predictors in a driving simulator among older adults according to gender. Clinics (Sao Paulo) 2024, 79, 100405. [Google Scholar] [CrossRef]
| Group | n | Response type | Color | Mean ± SD (ms) |
Median (IQR) (ms) |
Min–Max (ms) |
|---|---|---|---|---|---|---|
| Younger | 20 | Outside_RT | Blue | 697±74 | 693 (51) | 571–847 |
| Inside_RTr | Red | 673± 72 | 683 (33) | 558–784 | ||
| Inside_RTy | Yellow | 662±81 | 692 (74) | 544–802 | ||
| Older | 24 | Outside_RT | Blue | 818±113 | 786 (60) | 620–1035 |
| Inside_RTr | Red | 798±104 | 775 (51) | 635–1023 | ||
| Inside_RTy | Yellow | 781±106 | 770 (100) | 550–966 |
| Group | Contrast | Median (IQR) (ms) |
W | p-value | Effect size (r) |
|---|---|---|---|---|---|
| Younger (n=20) |
dRT | ー20 (51) | 32 | 0.005 | 0.61 |
| dRTr | ー14 (33) | 35 | 0.007 | 0.58 | |
| dRTy | ー27 (74) | 39 | 0.012 | 0.55 | |
| Older (n=24) |
dRT | ー22 (56) | 82 | 0.053 | 0.40 |
| dRTr | ー7 (51) | 114 | 0.317 | 0.21 | |
| dRTy | ー31 (100) | 73 | 0.025 | 0.45 |
| Contrast | Group | n | Mean ± SD (ms) |
Median (ms) | Test | Statistic | p-value | Cohen’s d |
|---|---|---|---|---|---|---|---|---|
| dRT | Younger | 20 | −30±46 | −20 (51) | S | −0.002 | 0.998 | −0.001 |
| Older | 24 | −30±67 | −22 (60) | |||||
| dRTr | Younger | 20 | −23±38 | −14 (33) | M | 203.5 | 0.396 | −0.053 |
| Older | 24 | −20±66 | −7 (51) | |||||
| dRTy | Younger | 20 | −35±55 | −27 (74) | S | 0.113 | 0.911 | 0.034 |
| Older | 24 | −37±73 | −31 (100) |
| Variable | Group | n | Mean ± SD | Median (IQR) | Test | Statistic | p-value | Cohen's d |
|---|---|---|---|---|---|---|---|---|
| Height (cm) | Younger | 20 | 168± 9 | 171 (13) | S | 2.132 | 0.039 | 0.645 |
| Older | 24 | 162± 9 | 162 (13) | |||||
| PAL (METs·h/week) |
Younger | 20 | 4.90± 7.6 | 0.0 (6.0) | M | 113 | 0.002 | -0.821 |
| Older | 24 | 19.62± 23.2 | 15.0 (13.7) |
| Variable | Group | n | Height | PAL | Sleep1 | Sleep2 | Sleep3 | Sleep4 | Sleep5 |
|---|---|---|---|---|---|---|---|---|---|
| dRT | ALL | 44 | 0.029 | 0.149 | 0.048 | 0.052 | 0.083 | 0.009 | −0.177 |
| Younger | 20 | −0.109 | 0.100 | 0.063 | 0.172 | −0.262 | −0.325 | 0.030 | |
| Older | 24 | 0.183 | 0.196 | 0.070 | −0.014 | 0.387 | 0.251 | −0.293 | |
| dRTr | ALL | 44 | −0.082 | 0.196 | 0.087 | −0.003 | 0.059 | 0.065 | −0.185 |
| Younger | 20 | −0.136 | 0.118 | 0.154 | 0.066 | −0.203 | −0.327 | 0.038 | |
| Older | 24 | 0.019 | 0.120 | 0.027 | −0.011 | 0.266 | 0.252 | −0.321 | |
| dRTy | ALL | 44 | 0.119 | 0.069 | 0.010 | −0.005 | 0.098 | −0.029 | −0.207 |
| Younger | 20 | −0.093 | 0.042 | 0.077 | 0.199 | −0.379 | −0.318 | −0.015 | |
| Older | 24 | 0.322 | 0.146 | 0.002 | −0.100 | 0.416 | 0.192 | −0.313 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




