Submitted:
31 March 2026
Posted:
01 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. SARS-CoV-2 Purified Recombinant Protein Panel
2.2. SARS-CoV-2 Viral Culture Panel
2.3. Residual Clinical Specimen Evaluation Panels
2.4. Precision Analysis
2.5. Statistical Analysis
2.6. Operational Assessment
3. Results
3.1. Performance Using Reference Materials
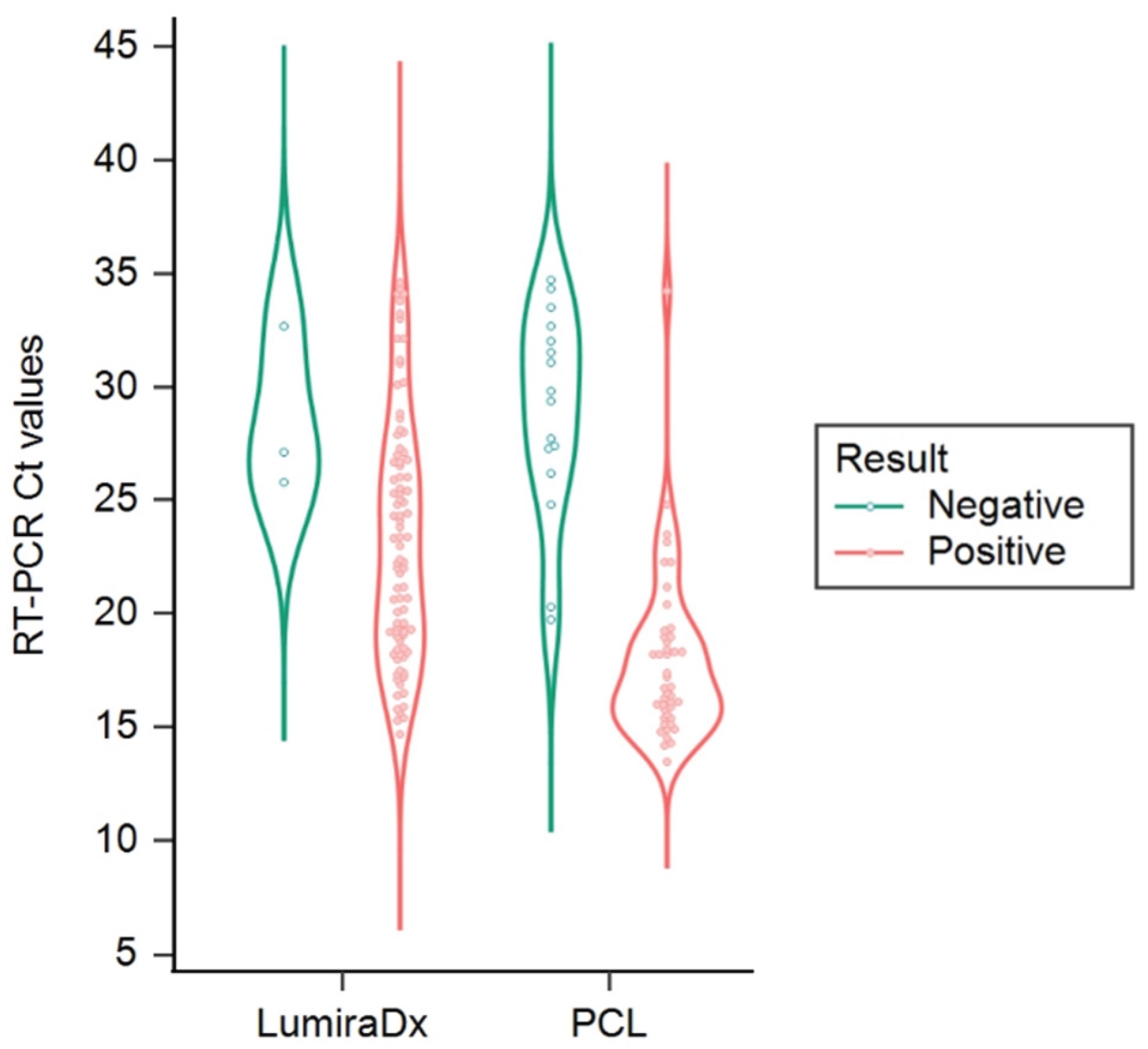
3.2. Performance Using Residual Clinical Specimens
3.3. Precision
3.4. Operational Performance
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. WHO Coronavirus (COVID-19) Dashboard 2023 [updated 28 June 2023. Available online: https://covid19.who.int/.
- Bergeri, I.; Whelan, M.G.; Ware, H.; Subissi, L.; Nardone, A.; Lewis, H.C.; Li, Z.; Ma, X.; Valenciano, M.; Cheng, B. Global SARS-CoV-2 seroprevalence from January 2020 to April 2022: A systematic review and meta-analysis of standardized population-based studies. PLoS medicine 2022, 19(11), e1004107. [Google Scholar] [CrossRef] [PubMed]
- Rostami, A.; Sepidarkish, M.; Leeflang, M.M.; Riahi, S.M.; Shiadeh, M.N.; Esfandyari, S.; Mokdad, A.H.; Hotez, P.J.; Gasser, R.B. SARS-CoV-2 seroprevalence worldwide: a systematic review and meta-analysis. Clinical Microbiology and Infection 2021, 27(3), 331–40. [Google Scholar] [CrossRef] [PubMed]
- Duroseau, B.; Kipshidze, N.; Limaye, R.J. The impact of delayed access to COVID-19 vaccines in low-and lower-middle-income countries. Frontiers in public health 2023, 10, 1087138. [Google Scholar] [CrossRef] [PubMed]
- Patwary, M.M.; Alam, M.A.; Bardhan, M.; Disha, A.S.; Haque, M.Z.; Billah, S.M.; Kabir, M.P.; Browning, M.H.; Rahman, M.M.; Parsa, A.D. COVID-19 vaccine acceptance among low-and lower-middle-income countries: a rapid systematic review and meta-analysis. Vaccines 2022, 10(3), 427. [Google Scholar] [CrossRef] [PubMed]
- Dutta, N.K.; Mazumdar, K.; Gordy, J.T. The nucleocapsid protein of SARS–CoV-2: a target for vaccine development. Journal of virology 2020, 94(13), e00647-20. [Google Scholar] [CrossRef] [PubMed]
- Zeng, W.; Liu, G.; Ma, H.; Zhao, D.; Yang, Y.; Liu, M.; Mohammed, A.; Zhao, C.; Yang, Y.; Xie, J. Biochemical characterization of SARS-CoV-2 nucleocapsid protein. Biochemical and biophysical research communications 2020, 527(3), 618–23. [Google Scholar] [CrossRef] [PubMed]
- FIND COVID-19 Test Directory [Internet]. 2022. Available online: https://www.finddx.org/tools-and-resources/dxconnect/test-directories/covid-19-test-directory/.
- Africa DoHRoS. Guide to Antigen Testing for SARS-CoV-2 in South Africa 2021.
- World Health Organization. SARS-CoV-2 antigen-detecting rapid diagnostic tests: an implementation guide; World Health Organization, 2021. [Google Scholar]
- Centers for Disease Control and Prevention. Guidance for Antigen Testing for SARS-CoV-2 for Healthcare Providers Testing Individuals in the Community [Internet]. 2022. Available online: https://www.cdc.gov/coronavirus/2019-ncov/lab/resources/antigen-tests-guidelines.html.
- In Vitro Diagnostics EUAs - Antigen Diagnostic Tests for SARS-CoV-2 [Internet]. 2023 [cited 12 January 2023]. Available online: https://www.fda.gov/medical-devices/coronavirus-disease-2019-covid-19-emergency-use-authorizations-medical-devices/in-vitro-diagnostics-euas-antigen-diagnostic-tests-sars-cov-2.
- Organization WH. Antigen-detection in the diagnosis of SARS-CoV-2 infection using rapid immunoassays. 2020. [Google Scholar]
- Kumar, V.; Ghosh, K.; Chandran, A.; Panwar, S.; Bhat, A.; Konaje, S.; Das, S.; Srikanta, S.; Jaganathan, L.; Prasad, S. Multicentric evaluation of a novel point of care electrochemical ELISA platform for SARS-CoV-2 specific IgG and IgM antibody assay. Journal of Virological Methods 2021, 298, 114275. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.; Tse, H. Lateral flow immunoassay; Springer Science & Business Media, 2008. [Google Scholar]
- Kinnamon, D.S.; Heggestad, J.T.; Liu, J.; Chilkoti, A. Technologies for Frugal and Sensitive Point-of-Care Immunoassays. Annual Review of Analytical Chemistry 2022, 15, 123–149. [Google Scholar] [CrossRef] [PubMed]
- Keshav, V.; Scott, L.; David, A.; Noble, L.; Mayne, E.; Stevens, W. Antigen-based point of care testing (poct) for diagnosing sars-cov-2: assessing performance. In Sars-cov-2: Methods and Protocols; Springer, 2022; pp. 45–62. [Google Scholar]
- Puhach, O.; Meyer, B.; Eckerle, I. SARS-CoV-2 viral load and shedding kinetics. Nature Reviews Microbiology 2023, 21(3), 147–61. [Google Scholar] [CrossRef] [PubMed]
- Linares, M.; Pérez-Tanoira, R.; Carrero, A.; Romanyk, J.; Pérez-García, F.; Gómez-Herruz, P.; Arroyo, T.; Cuadros, J. Panbio antigen rapid test is reliable to diagnose SARS-CoV-2 infection in the first 7 days after the onset of symptoms. Journal of Clinical Virology 2020, 133, 104659. [Google Scholar] [CrossRef] [PubMed]
- Nordgren, J.; Sharma, S.; Olsson, H.; Jämtberg, M.; Falkeborn, T.; Svensson, L.; Hagbom, M. SARS-CoV-2 rapid antigen test: High sensitivity to detect infectious virus. Journal of clinical virology 2021, 140, 104846. [Google Scholar] [CrossRef] [PubMed]
- Rabaan, A.A.; Tirupathi, R.; Sule, A.A.; Aldali, J.; Mutair, A.A.; Alhumaid, S.; Muzaheed Gupta, N.; Koritala, T.; Adhikari, R. Viral dynamics and real-time RT-PCR Ct values correlation with disease severity in COVID-19. Diagnostics 2021, 11(6), 1091. [Google Scholar] [CrossRef] [PubMed]
- Calistri, P.; Danzetta, M.; Amato, L. Epidemiological significance of SARS-CoV-2 RNA dynamic in naso-pharyngeal swabs. Microorganisms 2021, 9(6), 1264. [Google Scholar] [CrossRef] [PubMed]
- Mayfield, J.; Hesse, P.; Ledden, D. The Impact of Universal Transport Media and Viral Transport Media Liquid Samples on a SARS-CoV-2 Rapid Antigen Test. medRxiv. 2021:2021.05. 12.21257107.
- Jääskeläinen, A.; Ahava, M.J.; Jokela, P.; Szirovicza, L.; Pohjala, S.; Vapalahti, O.; Lappalainen, M.; Hepojoki, J.; Kurkela, S. Evaluation of three rapid lateral flow antigen detection tests for the diagnosis of SARS-CoV-2 infection. Journal of Clinical Virology 2021, 137, 104785. [Google Scholar] [CrossRef] [PubMed]
- Diao, B.; Wen, K.; Zhang, J.; Chen, J.; Han, C.; Chen, Y.; Wang, S.; Deng, G.; Zhou, H.; Wu, Y. Accuracy of a nucleocapsid protein antigen rapid test in the diagnosis of SARS-CoV-2 infection. Clinical Microbiology and Infection 2021, 27(2), 289.e1–e4. [Google Scholar] [CrossRef] [PubMed]
- Kohmer, N.; Toptan, T.; Pallas, C.; Karaca, O.; Pfeiffer, A.; Westhaus, S.; Widera, M.; Berger, A.; Hoehl, S.; Kammel, M. The comparative clinical performance of four SARS-CoV-2 rapid antigen tests and their correlation to infectivity in vitro. Journal of clinical medicine 2021, 10(2), 328. [Google Scholar] [CrossRef] [PubMed]
- Brümmer, L.E.; Katzenschlager, S.; Gaeddert, M.; Erdmann, C.; Schmitz, S.; Bota, M.; Grilli, M.; Larmann, J.; Weigand, M.A.; Pollock, N.R. Accuracy of novel antigen rapid diagnostics for SARS-CoV-2: A living systematic review and meta-analysis. PLoS medicine 2021, 18(8), e1003735. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | PCL COVID19 Ag Rapid FIA | LumiraDx SARS-CoV-2 Ag Test |
| Regulatory certification (at time of evaluation) | SAHPRA (08/01/2021), TGA (30/07/2021) | FDA EUA (18/12/2020) |
| Target population | Symptomatic individuals | Symptomatic and asymptomatic individuals |
| Specimen type(s) | Nasopharyngeal/Oropharyngeal | Nasal/Nasopharyngeal |
| Format/design | Immunosandwich lateral flow assay | Immunosandwich microfluidic assay |
| Target SARS-CoV-2 protein | Undisclosed | Nucleocapsid protein |
| Sample volume applied to test cassette | 4 drops | 1 drop (~20 µL) |
| Result interpretation | PCLOK EZ instrument | LumiraDx instrument |
| Time to result | Standard mode: 10 min on-board incubation; Quick mode: 10 min bench incubation + instrument read | 12 minutes after test strip insertion |
| Additional features | On-board printer; barcode scanning; USB data export | Automated quality checks; RFID calibration; cloud-based connectivity; LIS integration |
| Manufacturer performance data* | 89% sensitivity and 99% specificity | 98% sensitivity and 97% specificity |
| Material | Description | |
|---|---|---|
| SARS-CoV-2 purified recombinant N protein | 10 nM, 2.5 nM, 0.625 nM, 0.039 nM, 9.75 pM and 2.4 pM concentrations tested in duplicate | |
| SARS-CoV-2 viral cultures | 1x103 and 1x104 dilutions tested in triplicate and compared to RT-PCR | |
| SARS-CoV-2 residual clinical specimens | PCL COVID19 Ag Rapid FIA | LumiraDx SARS-CoV-2 Ag Test |
| Collected: Oct - Dec 2020 | Collected: April - June 2021 | |
| N=110 | N=110 | |
| n=54 HVL, n=10 MVL, n=17 LVL, n=30 Neg | n=42 HVL, n=18 MVL, n=20 LVL, n=30 neg | |
| Panel comprised of SA wave 1* specimens | Panel comprised of SA wave 1 and wave 2* specimens | |
| Methods applied | Simulated swab into kit buffer (A); 1:1 dilution of specimen with kit buffer (B) | Simulated swab into kit buffer (A); 1:7 dilution of specimen with kit buffer (C) |
| (a) SARS-CoV-2 viral cultures (SA wild-type) | ||||||||
| Assay | Dilution factor | FIA result | N | S | ORF1ab | |||
| PCL COVID19 Ag Rapid FlA | 1x103 | Positive | 24,1 | 24,6 | 23,9 | |||
| 1x104 | Negative | 27,2 | 27,9 | 27,1 | ||||
| LumiraDx SARS-CoV-2 Ag Test | 1x103 | Positive | 24,59 | 24,09 | 23,90 | |||
| 1x104 | Negative | 27,86 | 27,23 | 27,13 | ||||
| (b) SARS-CoV-2 purified recombinant nucleocapsid proteins (SA wild-type) | ||||||||
| Protein Concentration | 10 nM | 2.5 nM | 0.625 nM | 0.039 nM | 9.75 pM | 2.4 pM | 0.6 pM | 0.15 pM |
| PCL COVID19 Ag Rapid FlA | Positive | Positive | Positive | Positive | Negative | Negative | Negative | Negative |
| LumiraDx SARS-CoV-2 Ag Test | Positive | Positive | Positive | Positive | Positive | Positive | Positive | Negative |
| Assay | Method | Ct range | n | Sensitivity (95% CI) | Specificity (95% CI) | PPV (95% CI) | NPV (95% CI) | Cohen Kappa (95% CI) | Agreement score |
| PCL COVID19 Ag Rapid FIA | A and B | Overall performance | 110 | 63% (51-73) | 100% (88-100) | 100% (93-100) | 50% (37-63) | 0.48 (0.34-0.61) | Moderate |
| A | Ct ≤ 25 | 53 pos, 30 neg | 91% (79-97) | 100% (88-100) | 100% (93-100) | 86% (70-95) | 0.87 (0.77-0.98) | Very Good | |
| B | Ct ≤ 25 | 53 pos, 30 neg | 93% (82-98) | 100% (88-100) | 100% (93-100) | 88% (73-97) | 0.89 (0.80-1.00) | Very Good | |
| A, B | Ct 25-30 | 7 pos, 30 neg | No positives detected (0%)/ All negatives correctly identified (100%) | ||||||
| A, B | Ct 30-35 | 20 pos, 30 neg | |||||||
| LumiraDx SARS-CoV-2 Ag Test | A and C | Overall performance | 110 | 95% (88-99) | 97% (83-100) | 99% (93-100) | 88% (72-97) | 0.89 (0.79-0.98) | Very Good |
| A | Ct ≤ 25 | 42 pos, 30 neg | 100% (92-100) | 97% (83-100) | 98% (88-100) | 100% (88-100) | 0.97 (0.92-1.02) | Very Good | |
| A | Ct 25-30 | 18 pos, 30 neg | 83% (59-96) | 97% (83-100) | 94% (70-100) | 91% (75-98) | 0.97 (0.92-1.03) | Very Good | |
| A | Ct 30-35 | 20 pos, 30 neg | 65% (41-85) | 97% (83-100) | 93% (66-100) | 81% (64-92) | 0.82 (0.65-0.99) | Very Good | |
| C | Ct ≤ 25 | 42 pos, 30 neg | 100% (92-100) | 97% (83-100) | 98% (88-100) | 100% (88-100) | 0.91 (0.80-1.03) | Very Good | |
| C | Ct 25-30 | 18 pos, 30 neg | 94% (73-100) | 97% (83-100) | 94% (73-100) | 97% (83-100) | 0.65 (0.43-0.87) | Good | |
| C | Ct 30-35 | 20 pos, 30 neg | 85% (62-97) | 97% (83-100) | 94% (73-100) | 91% (75-98) | 0.83 (0.67-0.99) | Very Good | |
| Characteristic | PCL COVID19 Ag Rapid FlA | LumiraDx SARS-CoV-2 Ag Test |
| Kit contents ready for implementation | 4 | 4 |
| Need for biosafety | 4 | 4 |
| Training required | 5 | 5 |
| Ease of use from specimen collection to result interpretation | 4* | 4ǂ |
| Time to result | 5 | 5 |
| Invalid (error rate) | 5 | 4* |
| Comments | * Testing time increased with single-read mode | ǂTesting time increased with single-read cartridge *Analyser displayed “sample error - insufficient volume” despite adequate well coverage |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




