Submitted:
31 March 2026
Posted:
02 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
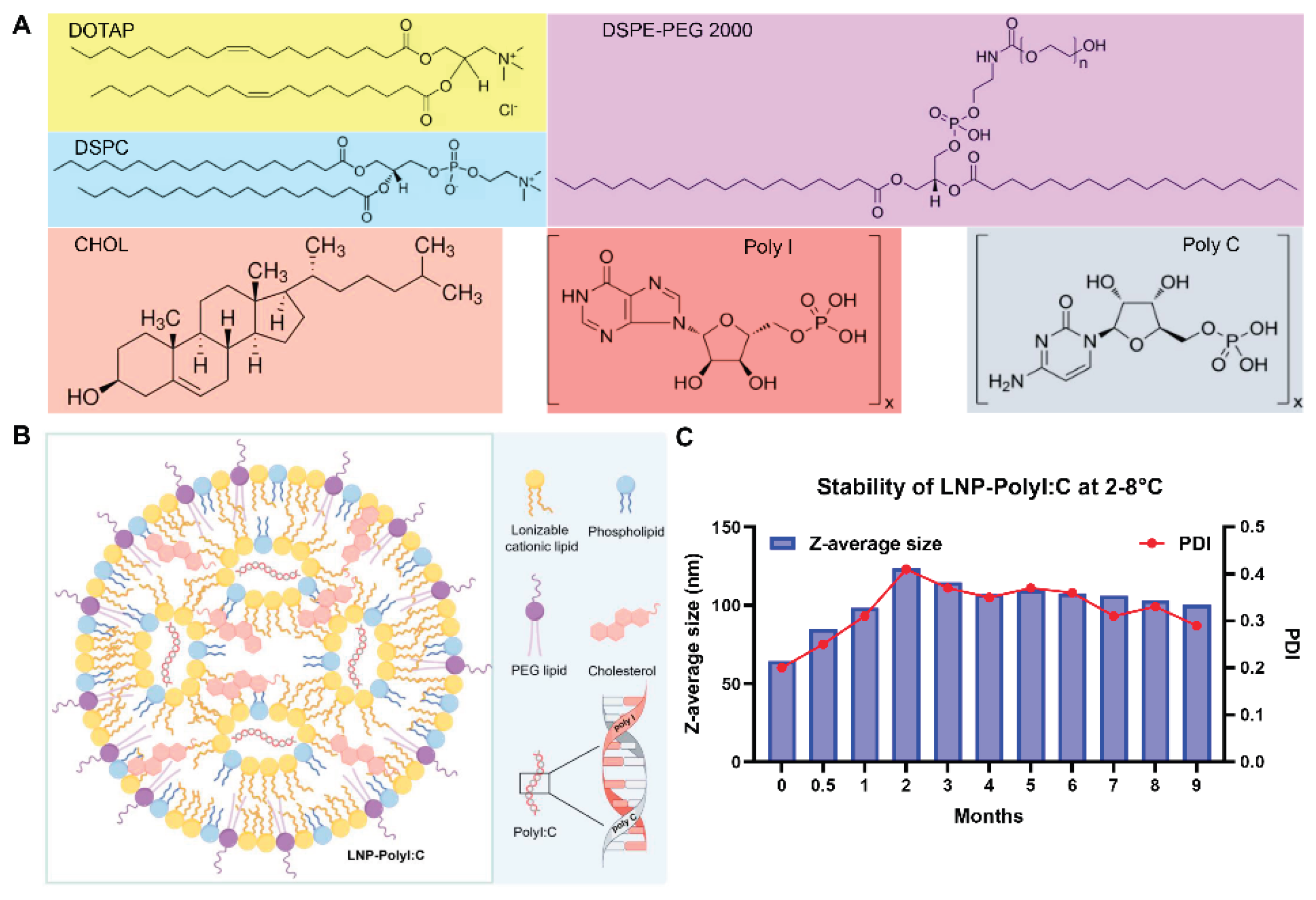
2.1. LNP Encapsulating PolyI:C and Vaccine Preparation
2.2. Animal Experiments
2.3. Enzyme-Linked Immunosorbent Assay
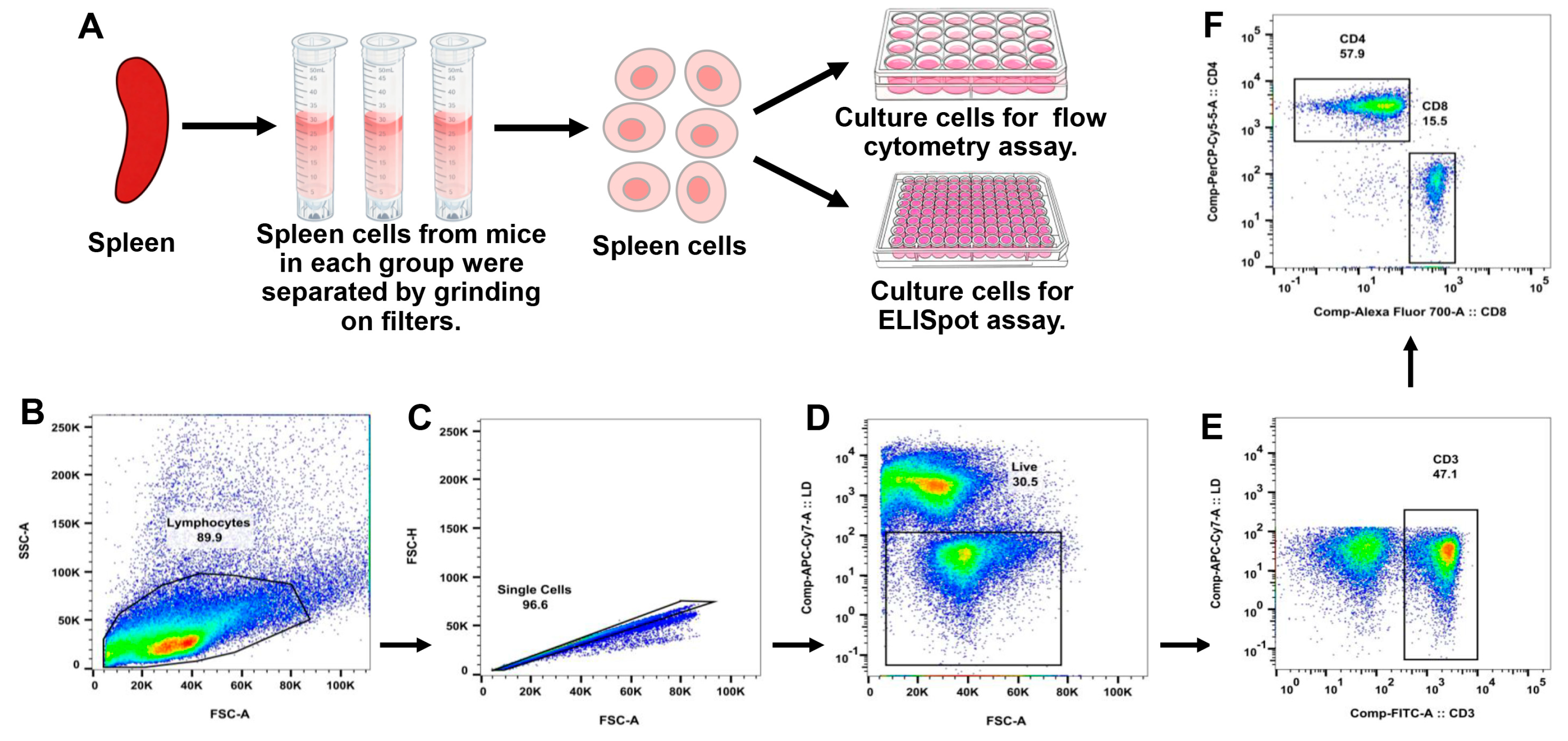
2.4. Isolation of Splenocytes
2.5. Flow Cytometry Analysis
2.6. Enzyme-Linked Immunospot Assay
2.7. Statistical Analysis
3. Results
3.1. Stability Results of LNP-PolyI:C
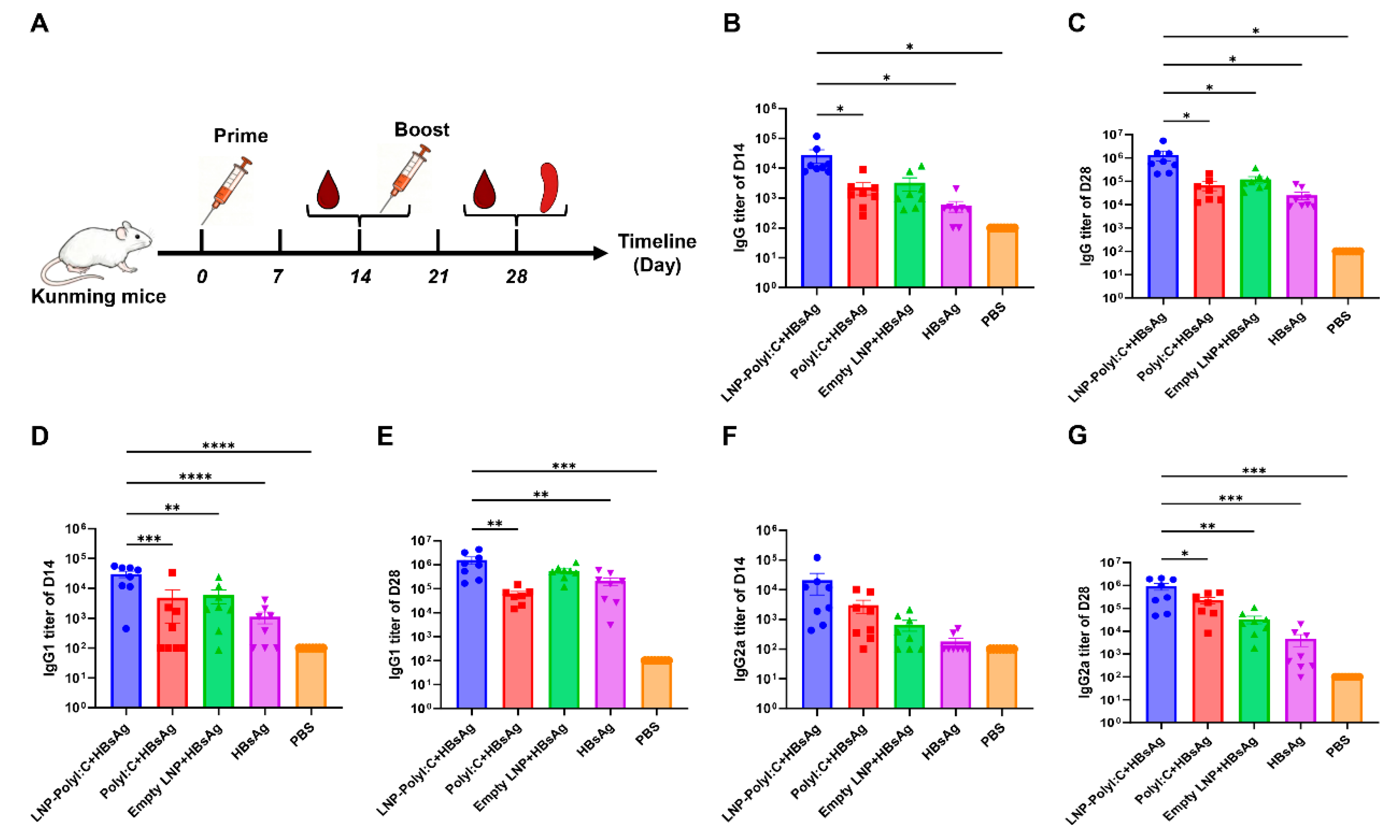
3.2. The LNP-PolyI:C Adjuvant Significantly Enhances Serum Antibody Response Levels
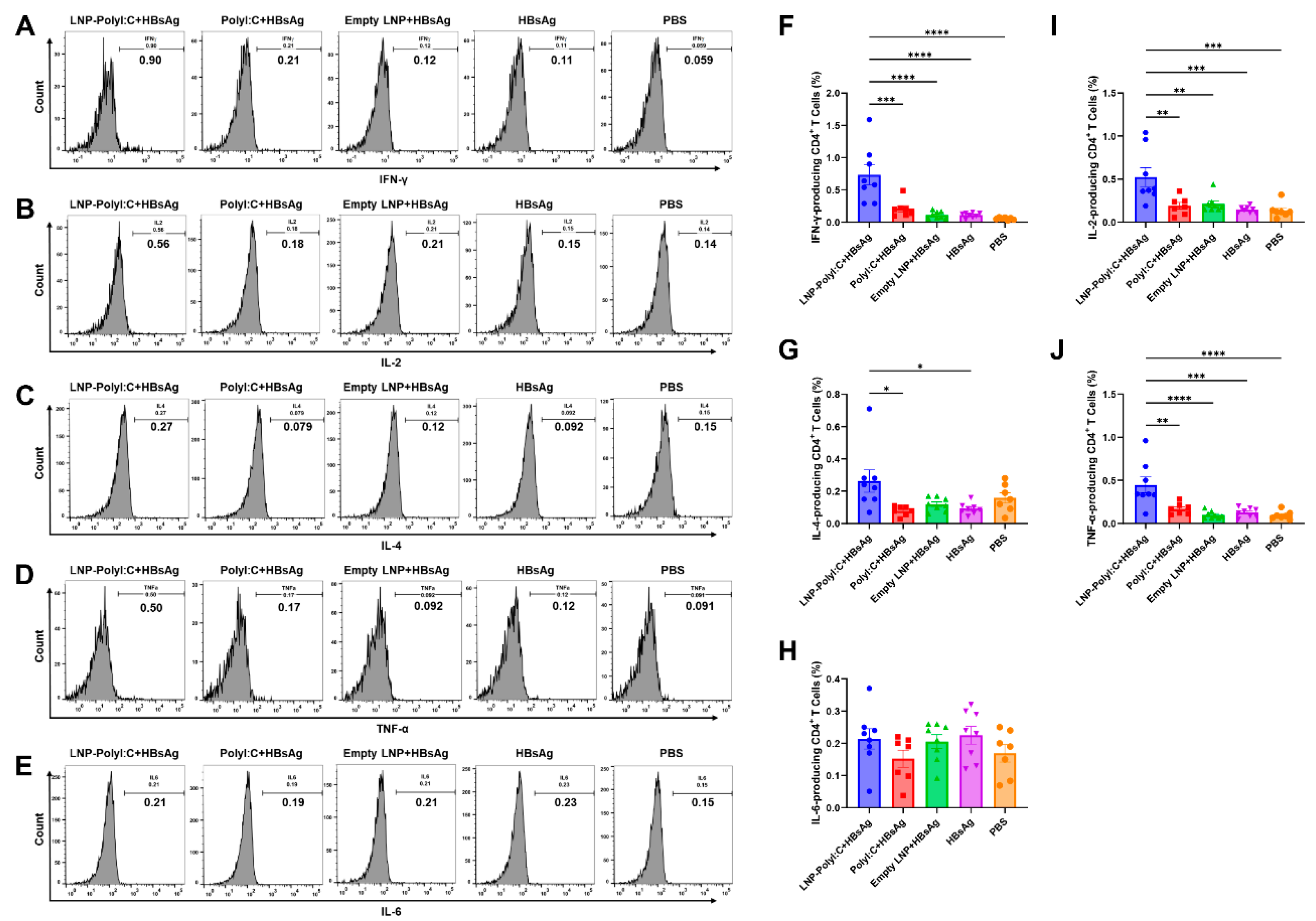
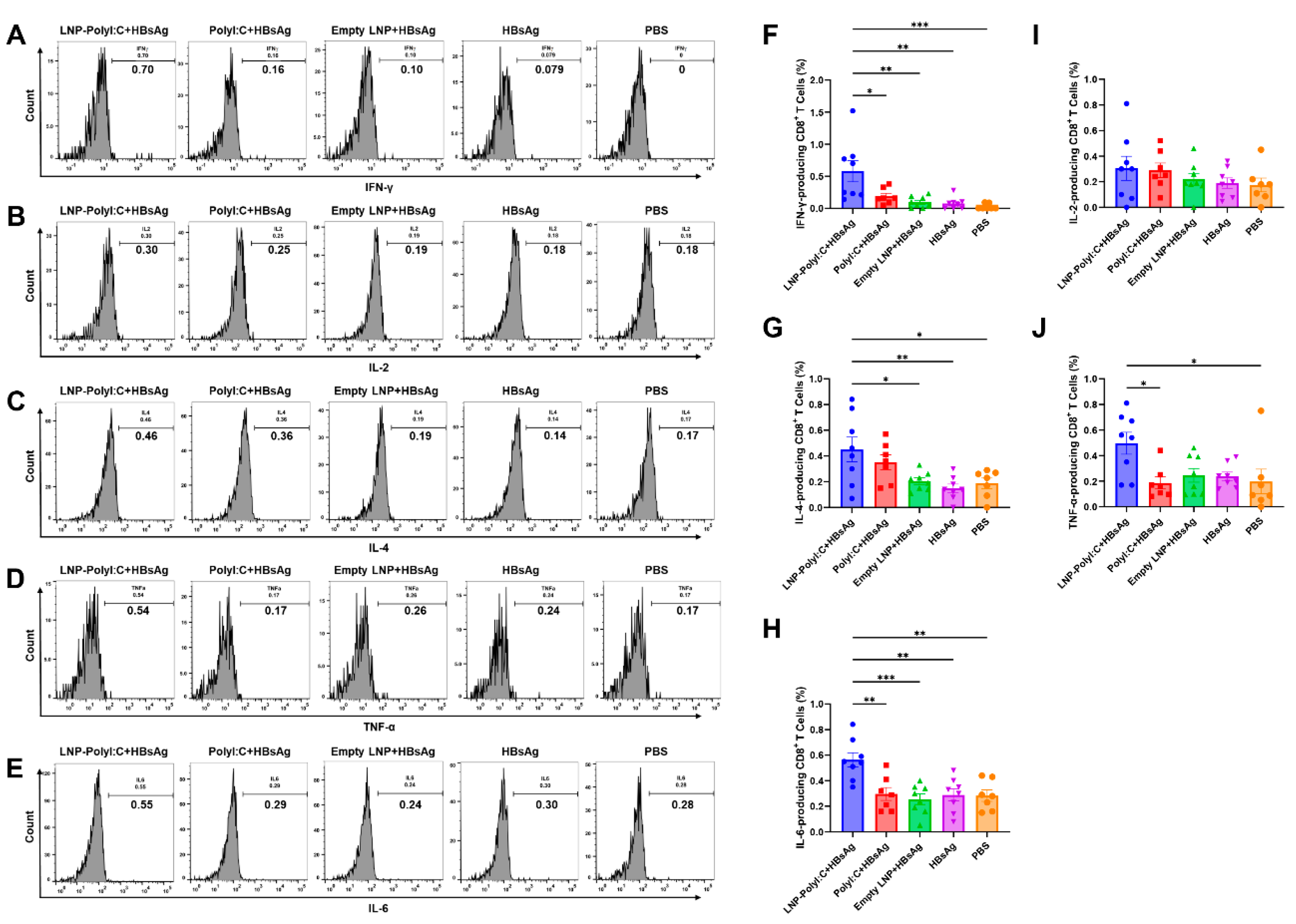
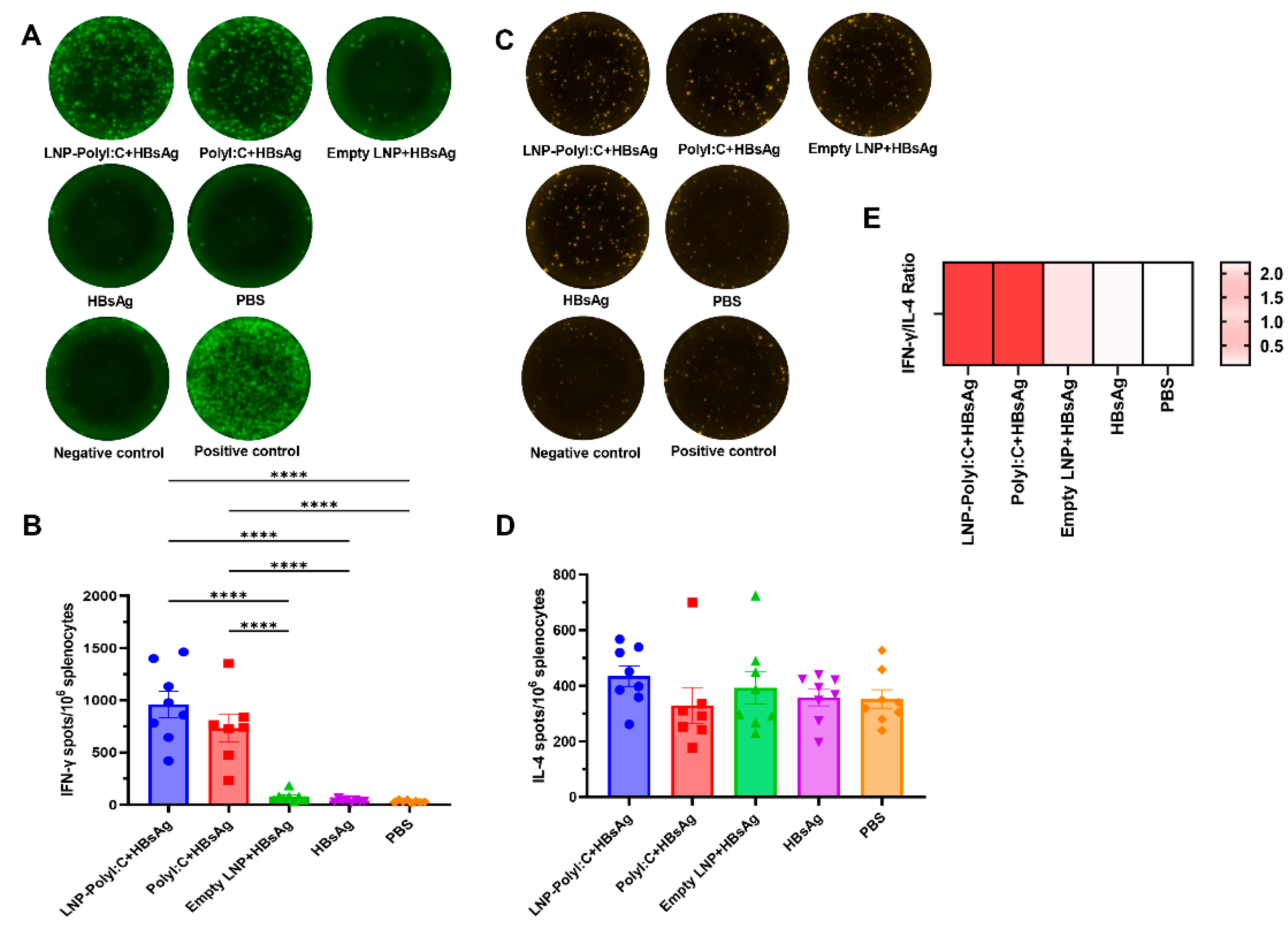
3.3. The LNP-PolyI:C Adjuvant Significantly Enhances Cytokine Expression In Vivo
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PolyI:C | Polyinosinic-polycytidylic acid |
| LNP | Lipid nanoparticle |
| HBsAg | Hepatitis B surface antigen |
| ELISA | Enzyme-linked immunosorbent assay |
| ELISpot | Enzyme-linked immunospot |
| CTL | Cytotoxic T lymphocyte |
| HBV | Hepatitis B virus |
| APC | Antigen-presenting cell |
| TLR9 | Toll-like receptor 9 |
| dsRNA | Double-stranded RNA |
| PAMP | Pathogen-associated molecular pattern |
| MDA-5 | Melanoma differentiation–associated protein 5 |
| IRF3 | Interferon regulatory factor 3 |
| NF-κB | Nuclear factor κB |
| AP-1 | Activator protein-1 |
| CARD | Caspase recruitment domain |
| MAVS | Mitochondrial antiviral signaling |
| PBS | Phosphate-buffered saline |
| PDI | Polydispersity index |
| SPF | Specific pathogen-free |
| HRP | Horseradish peroxidase |
| BSA | Bovine serum albumin |
| TMB | Tetramethylbenzidine |
| RBC | Red blood cell |
| SFC | Spots forming cell |
| ANOVA | One-way analysis of variance |
| HSD | Honestly significant difference |
| HDF | Hydrodynamic focusing |
| DLS | Dynamic light scattering |
| DOTAP | 1,2-stearoyl-3-trimethylammonium-propane |
| DSPC | 1,2-distearoyl-sn-glycero-3-phosphocholine |
| DSPE-PEG 2000 | 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[methoxy(polyethylene glycol)-2000] |
References
- Debbag, R.; Ávila-Agüero, M.L.; Brea, J.; Espinal, C.; Romero-Feregrino, R.; Torres, J.R.; Vázquez, H.; Cuadros, R.; Lazo-Páez, G.; Schilling, A.; et al. Impact of Vaccines Across the Lifespan: A New Perspective in Public Health—Conclusions of an Expert Panel—Part 2. Vaccines 2026, 14, 204. [Google Scholar] [CrossRef]
- Qiu, J.; Zhang, S.; Feng, Y.; Su, X.; Cai, J.; Chen, S.; Liu, J.; Huang, S.; Huang, H.; Zhu, S.; et al. Efficacy and safety of hepatitis B vaccine: an umbrella review of meta-analyses. Expert Rev Vaccines 2024, 23, 69–81. [Google Scholar] [CrossRef] [PubMed]
- Romano, L.; Paladini, S.; Galli, C.; Raimondo, G.; Pollicino, T.; Zanetti, A.R. Hepatitis B vaccination. Hum Vaccin Immunother 2015, 11, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Reed, S.G.; Orr, M.T.; Fox, C.B. Key roles of adjuvants in modern vaccines. Nat Med 2013, 19, 1597–1608. [Google Scholar] [CrossRef] [PubMed]
- D’Oro, U.; O’Hagan, D.T. The scientific journey of a novel adjuvant (AS37) from bench to bedside. NPJ Vaccines 2024, 9, 26. [Google Scholar] [CrossRef]
- Liu, X.; Min, Q.; Song, H.; Yue, A.; Li, Q.; Zhou, Q.; Han, W. Potentiating humoral and cellular immunity using a novel hybrid polymer-lipid nanoparticle adjuvant for HBsAg-VLP vaccine. J Nanobiotechnology 2023, 21, 441. [Google Scholar] [CrossRef]
- Liao, X.; Liang, Z. Strategy vaccination against Hepatitis B in China. Hum Vaccin Immunother 2015, 11, 1534–1539. [Google Scholar] [CrossRef]
- Dolan, S.A. Vaccines for hepatitis A and B. The latest recommendations on safe and extended protection. Postgrad Med 1997, 102, 74–80. [Google Scholar] [CrossRef]
- Sinani, G.; Senel, S. Advances in vaccine adjuvant development and future perspectives. Drug Deliv 2025, 32, 2517137. [Google Scholar] [CrossRef]
- Hogenesch, H. Mechanism of immunopotentiation and safety of aluminum adjuvants. Front Immunol 2012, 3, 406. [Google Scholar] [CrossRef]
- Champion, C.R. Heplisav-B: A Hepatitis B Vaccine With a Novel Adjuvant. Ann Pharmacother 2021, 55, 783–791. [Google Scholar] [CrossRef] [PubMed]
- Alexopoulou, L.; Holt, A.C.; Medzhitov, R.; Flavell, R.A. Recognition of double-stranded RNA and activation of NF-kappaB by Toll-like receptor 3. Nature 2001, 413, 732–738. [Google Scholar] [CrossRef] [PubMed]
- Rehwinkel, J.; Gack, M.U. RIG-I-like receptors: their regulation and roles in RNA sensing. Nat Rev Immunol 2020, 20, 537–551. [Google Scholar] [CrossRef] [PubMed]
- Chin, A.I.; Miyahira, A.K.; Covarrubias, A.; Teague, J.; Guo, B.; Dempsey, P.W.; Cheng, G. Toll-like receptor 3-mediated suppression of TRAMP prostate cancer shows the critical role of type I interferons in tumor immune surveillance. Cancer Res 2010, 70, 2595–2603. [Google Scholar] [CrossRef]
- Akazawa, T.; Ebihara, T.; Okuno, M.; Okuda, Y.; Shingai, M.; Tsujimura, K.; Takahashi, T.; Ikawa, M.; Okabe, M.; Inoue, N.; et al. Antitumor NK activation induced by the Toll-like receptor 3-TICAM-1 (TRIF) pathway in myeloid dendritic cells. Proc Natl Acad Sci U S A 2007, 104, 252–257. [Google Scholar] [CrossRef]
- Cheng, Y.S.; Xu, F. Anticancer function of polyinosinic-polycytidylic acid. Cancer Biol Ther 2010, 10, 1219–1223. [Google Scholar] [CrossRef]
- Lamoot, A.; Jangra, S.; Laghlali, G.; Warang, P.; Singh, G.; Chang, L.A.; Park, S.C.; Singh, G.; De Swarte, K.; Zhong, Z.; et al. Lipid Nanoparticle Encapsulation Empowers Poly(I:C) to Activate Cytoplasmic RLRs and Thereby Increases Its Adjuvanticity. Small 2024, 20, e2306892. [Google Scholar] [CrossRef]
- Lang, K.S.; Recher, M.; Junt, T.; Navarini, A.A.; Harris, N.L.; Freigang, S.; Odermatt, B.; Conrad, C.; Ittner, L.M.; Bauer, S.; et al. Toll-like receptor engagement converts T-cell autoreactivity into overt autoimmune disease. Nat Med 2005, 11, 138–145. [Google Scholar] [CrossRef]
- Walters, A.A.; Qin, Y.; Saleh, A.F.; Cheung, C.C.L.; Lyu, Q.; Zhu, Z.; Gafar, H.A.M.; Tzu-Wen Wang, J.; Al-Jamal, K.T. Triplet RNA Lipid Nanoparticles for Locoregional Cancer Immunotherapy. Small Sci 2026, 6, e202500506. [Google Scholar] [CrossRef]
- DeMuth, P.C.; Min, Y.; Huang, B.; Kramer, J.A.; Miller, A.D.; Barouch, D.H.; Hammond, P.T.; Irvine, D.J. Polymer multilayer tattooing for enhanced DNA vaccination. Nat Mater 2013, 12, 367–376. [Google Scholar] [CrossRef]
- Rwandamuriye, F.X.; Evans, C.W.; Wylie, B.; Norret, M.; Vitali, B.; Ho, D.; Nguyen, D.; Roper, E.A.; Wang, T.; Hepburn, M.S.; et al. A surgically optimized intraoperative poly(I:C)-releasing hydrogel prevents cancer recurrence. Cell Rep Med 2023, 4, 101113. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Huang, P.; Niu, J.; Shi, G.; Zhang, C.; Kong, D.; Wang, W. Injectable polypeptide hydrogel for dual-delivery of antigen and TLR3 agonist to modulate dendritic cells in vivo and enhance potent cytotoxic T-lymphocyte response against melanoma. Biomaterials 2018, 159, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Jewell, C.M.; Lopez, S.C.; Irvine, D.J. In situ engineering of the lymph node microenvironment via intranodal injection of adjuvant-releasing polymer particles. Proc Natl Acad Sci U S A 2011, 108, 15745–15750. [Google Scholar] [CrossRef] [PubMed]
- Koerner, J.; Horvath, D.; Herrmann, V.L.; MacKerracher, A.; Gander, B.; Yagita, H.; Rohayem, J.; Groettrup, M. PLGA-particle vaccine carrying TLR3/RIG-I ligand Riboxxim synergizes with immune checkpoint blockade for effective anti-cancer immunotherapy. Nat Commun 2021, 12, 2935. [Google Scholar] [CrossRef]
- Ko, K.H.; Lee, S.H.; Choi, Y.H.; Kang, S.M.; Yang, H.S.; Lee, S.M.; Jo, E.B.; Bae, H.S.; Hong, S.B.; Kim, D.H.; et al. Lipid nanoparticle encapsulated TLR3 agonist adjuvant elicits potent T cell immunity against cancer and viruses. NPJ Vaccines 2025, 11, 26. [Google Scholar] [CrossRef]
- Luo, Y.; Li, Q.; Zhou, S.; Oh, H.; Jablonski, J.; Song, Y.; Su, Y.; Wu, Y.; Zhu, H.; Ortega, J.; et al. Lipid nanoparticles that co-deliver poly(I:C) and short peptide antigens elicit anti-tumor responses with vaccination. Biomaterials 2026, 327, 123754. [Google Scholar] [CrossRef]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Perez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N Engl J Med 2020, 383, 2603–2615. [Google Scholar] [CrossRef]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N Engl J Med 2021, 384, 403–416. [Google Scholar] [CrossRef]
- Hald Albertsen, C.; Kulkarni, J.A.; Witzigmann, D.; Lind, M.; Petersson, K.; Simonsen, J.B. The role of lipid components in lipid nanoparticles for vaccines and gene therapy. Adv Drug Deliv Rev 2022, 188, 114416. [Google Scholar] [CrossRef]
- Chen, S.P.; Blakney, A.K. Immune response to the components of lipid nanoparticles for ribonucleic acid therapeutics. Curr Opin Biotechnol 2024, 85, 103049. [Google Scholar] [CrossRef]
- Hou, X.; Zaks, T.; Langer, R.; Dong, Y. Lipid nanoparticles for mRNA delivery. Nat Rev Mater 2021, 6, 1078–1094. [Google Scholar] [CrossRef]
- Aldosari, B.N.; Alfagih, I.M.; Almurshedi, A.S. Lipid Nanoparticles as Delivery Systems for RNA-Based Vaccines. Pharmaceutics 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.; Chernatynskaya, A.; Qi, L.; Chuang, H.Y.; Cole, T.; Jeyalatha, V.M.; Bhargava, L.; Yeudall, W.A.; Farkas, L.; Yang, H. Liposomes-Encapsulating Double-Stranded Nucleic Acid (Poly I:C) for Head and Neck Cancer Treatment. ACS Pharmacol Transl Sci 2024, 7, 1612–1623. [Google Scholar] [CrossRef] [PubMed]
- Ruterbusch, M.; Pruner, K.B.; Shehata, L.; Pepper, M. In Vivo CD4(+) T Cell Differentiation and Function: Revisiting the Th1/Th2 Paradigm. Annu Rev Immunol 2020, 38, 705–725. [Google Scholar] [CrossRef] [PubMed]
- Chin’ombe, N.; Bourn, W.R.; Williamson, A.-L.; Shephard, E.G. Oral vaccination with a recombinant Salmonella vaccine vector provokes systemic HIV-1 subtype C Gag-specific CD4+ Th1 and Th2 cell immune responses in mice. Virology Journal 2009, 6, 87. [Google Scholar] [CrossRef]
- Chung, N.H.; Chen, Y.C.; Yang, S.J.; Lin, Y.C.; Dou, H.Y.; Hui-Ching Wang, L.; Liao, C.L.; Chow, Y.H. Induction of Th1 and Th2 in the protection against SARS-CoV-2 through mucosal delivery of an adenovirus vaccine expressing an engineered spike protein. Vaccine 2022, 40, 574–586. [Google Scholar] [CrossRef]
- Paludan, S.R. Interleukin-4 and interferon-gamma: the quintessence of a mutual antagonistic relationship. Scand J Immunol 1998, 48, 459–468. [Google Scholar] [CrossRef]
- Kedmi, R.; Ben-Arie, N.; Peer, D. The systemic toxicity of positively charged lipid nanoparticles and the role of Toll-like receptor 4 in immune activation. Biomaterials 2010, 31, 6867–6875. [Google Scholar] [CrossRef]
- Gao, S.; Yang, D.; Fang, Y.; Lin, X.; Jin, X.; Wang, Q.; Wang, X.; Ke, L.; Shi, K. Engineering Nanoparticles for Targeted Remodeling of the Tumor Microenvironment to Improve Cancer Immunotherapy. Theranostics 2019, 9, 126–151. [Google Scholar] [CrossRef]
- Liu, X.; Min, Q.; Li, Y.; Chen, S. Enhanced Cellular Immunity for Hepatitis B Virus Vaccine: A Novel Polyinosinic-Polycytidylic Acid-Incorporated Adjuvant Leveraging Cytoplasmic Retinoic Acid-Inducible Gene-Like Receptor Activation and Increased Antigen Uptake. Biomaterials Research 2024, 28, 0096. [Google Scholar] [CrossRef]
- Danaei, M.; Dehghankhold, M.; Ataei, S.; Hasanzadeh Davarani, F.; Javanmard, R.; Dokhani, A.; Khorasani, S.; Mozafari, M.R. Impact of Particle Size and Polydispersity Index on the Clinical Applications of Lipidic Nanocarrier Systems. Pharmaceutics 2018, 10, 57. [Google Scholar] [CrossRef]
- Muramatsu, H.; Lam, K.; Bajusz, C.; Laczko, D.; Kariko, K.; Schreiner, P.; Martin, A.; Lutwyche, P.; Heyes, J.; Pardi, N. Lyophilization provides long-term stability for a lipid nanoparticle-formulated, nucleoside-modified mRNA vaccine. Mol Ther 2022, 30, 1941–1951. [Google Scholar] [CrossRef]
- Alejo, T.; Toro-Córdova, A.; Fernández, L.; Rivero, A.; Stoian, A.M.; Pérez, L.; Navarro, V.; Martínez-Oliván, J.; de Miguel, D. Comprehensive Optimization of a Freeze-Drying Process Achieving Enhanced Long-Term Stability and In Vivo Performance of Lyophilized mRNA-LNPs. Int J Mol Sci 2024, 25, 10603. [Google Scholar] [CrossRef]
- Khan, M.F.H.; Sudalaiyadum Perumal, A.; Kamen, A.A. Investigation on a Freeze-Drying Process for Long-Term Stability of mRNA-LNPs. Vaccines 2026, 14, 242. [Google Scholar] [CrossRef]
| Vaccine Group | Vaccine Ingredients (per milliliter) | |||
| HBsAg (μg) | LNP-PolyI:C (μg) | PolyI:C (μg) | Empty LNP | |
| LNP-PolyI:C+HBsAg | 40 | 1000 | ||
| PolyI:C+HBsAg | 40 | 1000 | ||
| Empty LNP+HBsAg | 40 | The volume of the empty LNP is the same as that of LNP-PolyI:C. | ||
| HBsAg | 40 | |||
| PBS | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




