Submitted:
31 March 2026
Posted:
01 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
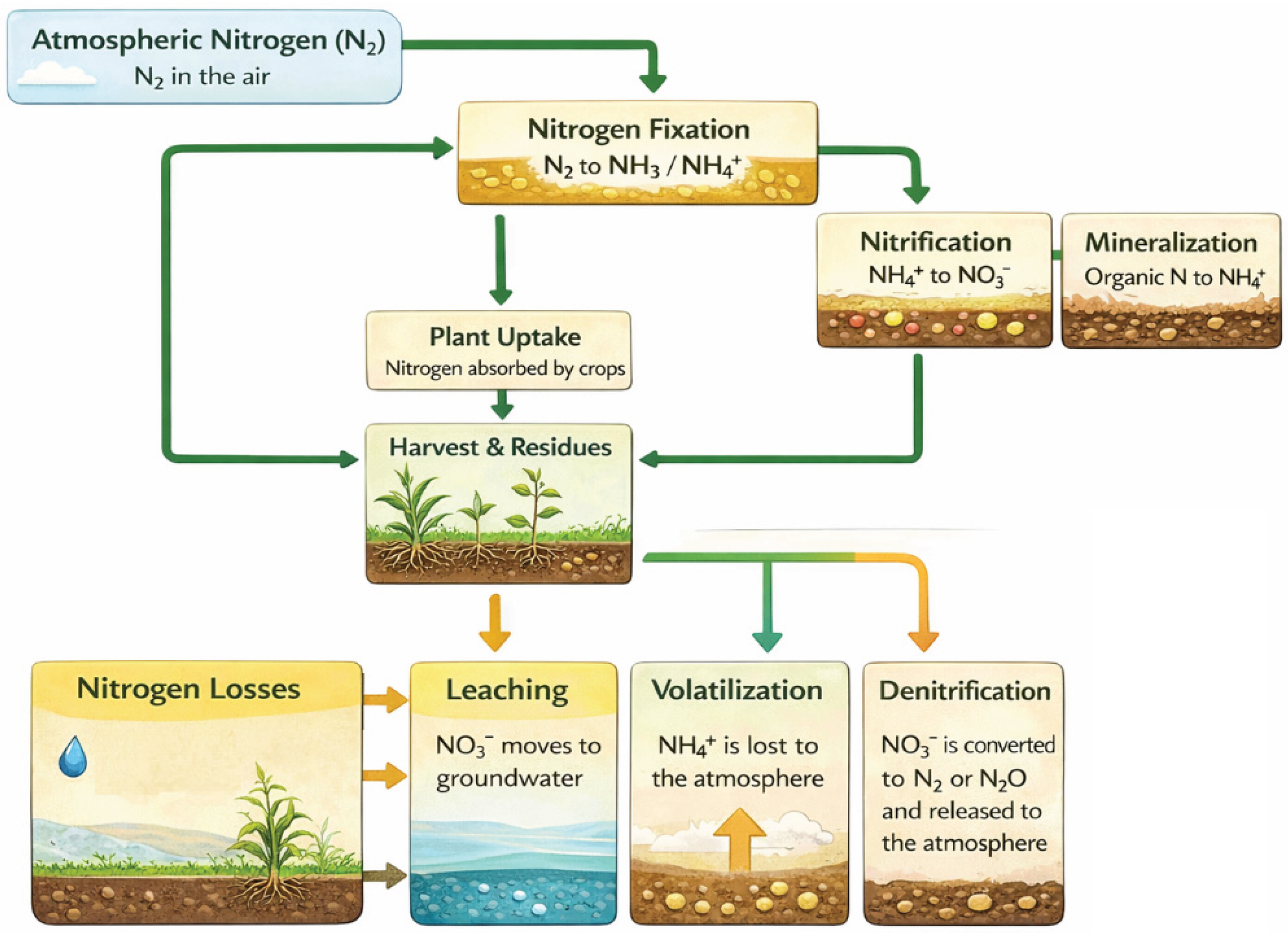
2. Mechanisms Governing NH3 Volatilization in Agricultural Soils
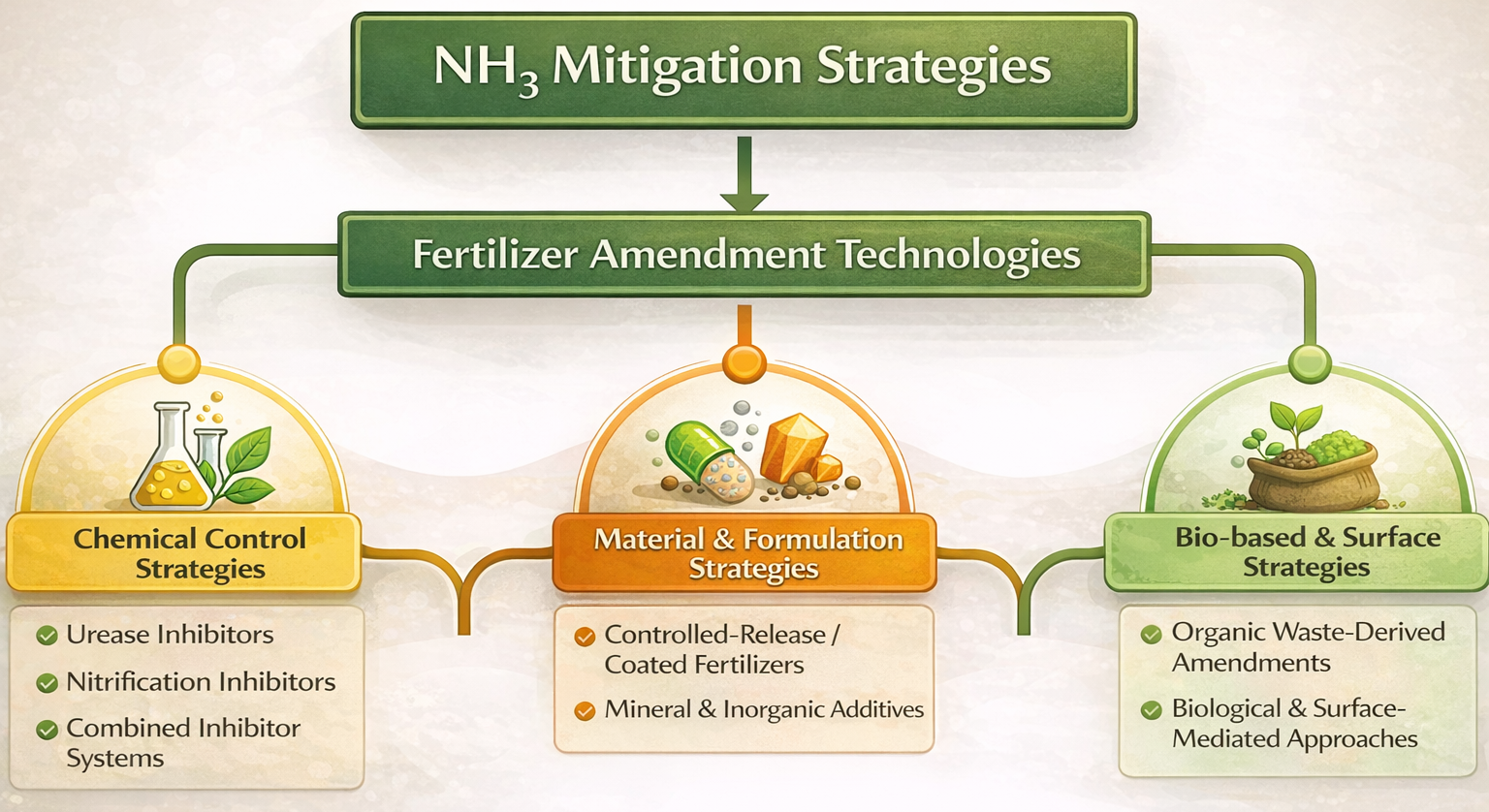
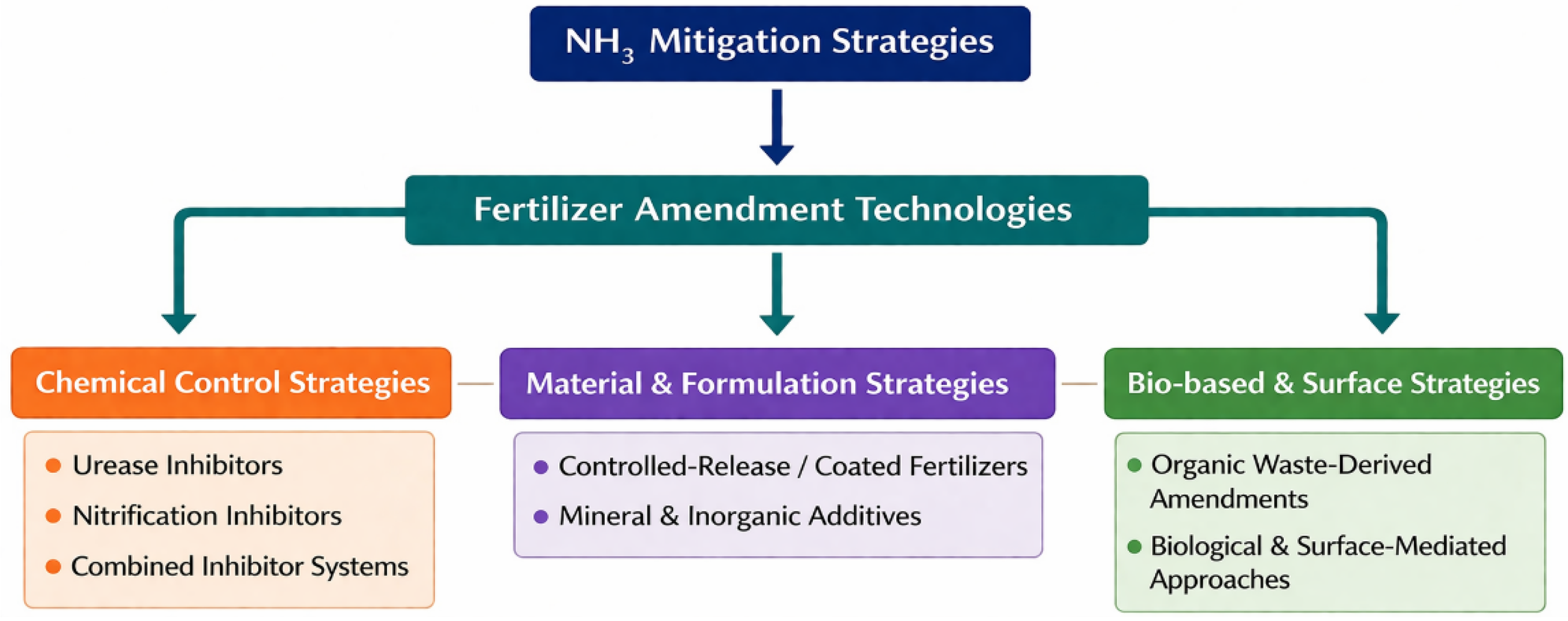
3. Fertilizer-Based Strategies for Reducing NH3 Emissions
3.1. Urease Inhibitors
3.2. Nitrification Inhibitors and Combined Strategies
3.3. Fertilizers Designed for Controlled Nitrogen Release
3.4. Inorganic and Mineral Additives for Ammonia Mitigation
3.5. Waste-Derived Organic Amendments
3.6. Biological and Surface-Mediated Approaches
4. Technology-based Control of the NH4+/NH3 Equilibrium
4.1. Urease Inhibitor Technologies
4.2. Controlled-Release Fertilisers in Practice
4.3. Nitrification Inhibitors and Formulation Effects
4.4. Ammonia Recovery and Nitrogen Conditioning Technologies
5. Variability and Nitrogen Trade-offs
5.1. Main Drivers of Variability
5.2. Trade-offs between Nitrogen Loss Pathways
5.3. High- and Low-Risk Conditions
6. Future Research Directions for Sustainable Ammonia Management
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| 2-NPT | N-(2-nitrophenyl) phosphoric triamide |
| CAN | Calcium ammonium nitrate |
| CEC | Cation Exchange Capacity |
| CRU | Controlled-release urea |
| DCD | Dicyandiamide |
| DMPP | 3,4-dimethylpyrazole phosphate |
| DMPSA | 3,4-dimethylpyrazole succinic acid |
| EEF | Enhanced-efficiency fertilizers |
| MIP | Methyl-phosphoric acid tri-isopropyl ester |
| NBPT | N-(n-butyl) thiophosphoric triamide |
| NPPT | N-(n-propyl) thiophosphoric triamide |
| PM2.5 | Fine particulate matter (particles ≤2.5 µm) |
| UAN | Urea–ammonium nitrate |
References
- Oita, A.; Malik, A.; Kanemoto, K.; Geschke, A.; Nishijima, S.; Lenzen, M. Substantial nitrogen pollution embedded in international trade. Nat. Geosci. 2016, 9, 111–115. [Google Scholar] [CrossRef]
- Statista. World fertilizer demand medium term forecast 2027. Available online: https://www.statista.com/statistics/438964/medium-forecast-fertilizer-demandglobally-by-nutrient/ (accessed on 29 March 2026).
- Tilman, D.; Balzer, C.; Hill, J.; Befort, B.L. Global food demand and the sustainable intensification of agriculture. Proc. Natl. Acad. Sci. USA 2011, 108, 20260–20264. [Google Scholar] [CrossRef] [PubMed]
- Scheer, C.; Rowlings, D.W.; Antille, D.L.; De Antoni Migliorati, M.; Fuchs, K.; Grace, P.R. Improving nitrogen use efficiency in irrigated cotton production. Nutr. Cycl. Agroecosyst 2023, 125, 95–106. [Google Scholar] [CrossRef]
- Zhou, M.; Li, T.; Liu, P.; Zhang, S.; Liu, Y.; An, T.; Zhao, H. Real-time on-site monitoring of soil ammonia emissions using membrane permeation-based sensing probe. Environ. Pollut. 2021, 289, 117850. [Google Scholar] [CrossRef]
- Matse, D.T.; Krol, D.J.; Richards, K.G.; Danaher, M.; Cummins, E.; Wang, X.; Forrestal, P.J. Field efficacy of urease inhibitors for mitigation of ammonia emissions in agricultural field settings: A systematic review. Front. Environ. Sci. 2024, 12, 1462098. [Google Scholar]
- van Wijk, D.; Raben, C.R.; Erbrink, H.J.; Heederik, D.J.; Dohmen, W. Effects of different ammonia emission reduction strategies from livestock farming on ambient ammonia concentrations in nature areas: A series of scenario analyses. Atmos. Environ. X 2025, 28, 100370. [Google Scholar] [CrossRef]
- Fan, D.; He, W.; Smith, W.N.; Drury, C.F.; Jiang, R.; Grant, B.B.; Zou, G. Global evaluation of inhibitor impacts on ammonia and nitrous oxide emissions from agricultural soils: A meta-analysis. Glob. Change Biol. 2022, 28(17), 5121–5141. [Google Scholar] [CrossRef]
- Lelieveld, J.; Evans, J.S.; Fnais, M.; Giannadaki, D.; Pozzer, A. The contribution of outdoor air pollution sources to premature mortality on a global scale. Nature 2015, 525, 367–371. [Google Scholar] [CrossRef]
- Pozzer, A.; Tsimpidi, A.P.; Karydis, V.A.; de Meij, A.; Lelieveld, J. Impact of agricultural emission reductions on fine-particulate matter and public health. Atmos. Chem. Phys. 2017, 17, 12813–12826. [Google Scholar] [CrossRef]
- Melara, F.; da Silva, L.K.; Martins Sanderi, D.; Dal Castel Krein, D.; Strieder Machado, T.; Dettmer, A.; Steffanello Piccin, J. Enhanced efficiency fertilizer: A review on technologies, perspectives, and research strategies. Environ. Dev. Sustain. 2024, 1–27. [Google Scholar] [CrossRef]
- Liu, C.; Li, B.; Wu, Q.; Chen, D.; Zhou, W.; Ao, J. The effects and mechanism of urease inhibitor and its combination with nitrification inhibitor on nitrous oxide emission across four soil types. Front. Plant Sci. 2025, 16, 1663261. [Google Scholar] [CrossRef]
- Li, T.; Wang, Z.; Wang, C.; Huang, J.; Feng, Y.; Shen, W.; Zhou, M.; Yang, L. Ammonia volatilization mitigation in crop farming: A review of fertilizer amendment technologies and mechanisms. Chemosphere 2022, 303, 134944. [Google Scholar] [CrossRef] [PubMed]
- Guo, T.; Bai, S.H.; Omidvar, N.; Wang, Y.; Chen, F.; Zhang, M. Insight into the functional mechanisms of nitrogen-cycling inhibitors in decreasing yield-scaled ammonia volatilization and nitrous oxide emission: A global meta-analysis. Chemosphere 2023, 338, 139611. [Google Scholar] [CrossRef] [PubMed]
- Sokal, K.; Kachel, M. Impact of agriculture on greenhouse gas emissions—A review. Energies 2025, 18, 2272. [Google Scholar] [CrossRef]
- Liu, Z.; Zhu, P.; Han, W.; Su, W.; Ye, X.; Zhang, Y.; Jing, J. Unraveling the mechanism of straw-derived humic acid in mitigating urea nitrogen volatilization loss in four Chinese soils. Ind. Crops Prod. 2025, 223, 120188. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, L.; Bai, Z.; Xu, W.; Zhang, F.; Zhang, X.; Xie, Y. Comprehensive quantification of global cropland ammonia emissions and potential abatement. Sci. Total Environ. 2022, 812, 151450. [Google Scholar] [CrossRef]
- Horváth, L.; Szabó, A.; Weidinger, T. Conception and parameterization of field-scale models for simulating ammonia loss from fertilized lands: A review. Model. Earth Syst. Environ. 2024, 10, 3079–3100. [Google Scholar] [CrossRef]
- Sommer, S.G.; Bittman, S.; Pacholski, A. Expert opinion: Improving process knowledge about ammonia emission from field applied liquid manure. Biosyst. Eng. 2025, 260, 104304. [Google Scholar] [CrossRef]
- Lee, Y.J.; Im, E.C.; Lee, J.I.; Lee, C.G.; Park, S.J. Comparison of ammonia volatilization and crop growth in upland fields fertilized with urea and latex-coated fertilizer. J. Soils Sediments 2025, 25, 2347–2359. [Google Scholar] [CrossRef]
- Woodley, A.L.; Drury, C.F.; Yang, X.M.; Phillips, L.A.; Reynolds, W.D.; Calder, W.; Oloya, T.O. Ammonia volatilization, N2O emissions and corn yields as influenced by nitrogen placement and enhanced efficiency fertilizers. Soil Sci. Soc. Am. J. 2020, 84, 1327–1341. [Google Scholar] [CrossRef]
- Pan, B.; Lam, S.K.; Mosier, A.; Luo, Y.; Chen, D. Ammonia volatilization from synthetic fertilizers and its mitigation strategies: A global synthesis. Agric. Ecosyst. Environ. 2016, 232, 283–289. [Google Scholar] [CrossRef]
- Sigurdarson, J.J.; Svane, S.; Karring, H. The molecular processes of urea hydrolysis in relation to ammonia emissions from agriculture. Rev. Environ. Sci. Biotechnol. 2018, 17, 241–258. [Google Scholar] [CrossRef]
- Vidal, A.; Nguyen, C.; Janot, N.; Eon, P.; Coriou, C.; Cornu, J.Y. Effect of sulfur fertilizers applied at agronomic rates on cadmium availability in agricultural soils: Insights from a batch experiment. Pedosphere 2024, 35, 995–1004. [Google Scholar] [CrossRef]
- Legese, W.; Taddesse, A.M.; Kibret, K.; Wogi, L. Effects of natural and modified zeolite based composite fertilizers on slow release and nutrient use efficiency. Heliyon 2024, 10, e25524. [Google Scholar] [CrossRef]
- Hearn, L.O.; Barton, L.; Schwenke, G.D.; Murphy, D.V. Ammonia volatilisation losses from urea applied to acidic cropping soils is regulated by pH buffering capacity. Soil Res. 2023, 61, 685–696. [Google Scholar] [CrossRef]
- Moradi, S.; Babapoor, A.; Ghanbarlou, S.; Kalashgarani, M.Y.; Salahshoori, I.; Seyfaee, A. Toward a new generation of fertilizers with the approach of controlled-release fertilizers: A review. J. Coat. Technol. Res. 2024, 21, 31–54. [Google Scholar] [CrossRef]
- Sahu, B.; Dash, B.; Pradhan, S.N.; Nalia, A.; Singh, P. Fertilizer management in dryland cultivation for stable crop yields. In Enhancing Resilience of Dryland Agriculture under Changing Climate: Interdisciplinary and Convergence Approaches; Springer Nature: Singapore, 2023; pp. 305–322. [Google Scholar]
- An, Z.; Huang, R.J.; Zhang, R.; Tie, X.; Li, G.; Cao, J.; Zhou, W.; Shi, Z.; Han, Y.; Gu, Z.; Ji, Y. Severe haze in northern China: A synergy of anthropogenic emissions and atmospheric processes. Proc. Natl. Acad. Sci. USA 2019, 116, 8657–8666. [Google Scholar] [CrossRef]
- Sabina, R.; Paul, J.; Sharma, S.; Hussain, N. Synthetic nitrogen fertilizer pollution: Global concerns and sustainable mitigating approaches. In Agricultural Nutrient Pollution and Climate Change: Challenges and Opportunities; Springer Nature Switzerland: Cham, Switzerland, 2025; pp. 57–101. [Google Scholar]
- Cantarella, H.; Otto, R.; Soares, J.R.; Silva, A.G.B. Agronomic efficiency of NBPT as a urease inhibitor: A review. J. Adv. Res. 2018, 13, 19–27. [Google Scholar] [CrossRef]
- Martins, M.R.; Sant’Anna, S.A.; Zaman, M.; Santos, R.C.; Monteiro, R.C.; Alves, B.J.; Jantalia, C.P.; Boddey, R.M.; Urquiaga, S. Strategies for the use of urease and nitrification inhibitors with urea: Impact on N2O and NH3 emissions, fertilizer-15N recovery and maize yield in a tropical soil. Agric. Ecosyst. Environ. 2017, 247, 54–62. [Google Scholar] [CrossRef]
- Awale, R.; Chatterjee, A. Enhanced efficiency nitrogen products influence ammonia volatilization and nitrous oxide emission from two contrasting soils. Agron. J. 2017, 109, 47–57. [Google Scholar] [CrossRef]
- Ahmed, M.Y.W.; Lei, M.; Raza, S.; Zhou, J. Mitigation of ammonia volatilization with application of urease and nitrification inhibitors from summer maize at the Loess Plateau. Plant Soil Environ. 2018, 64, 164–172. [Google Scholar] [CrossRef]
- Barberena, I.M. Use of urease inhibitors to reduce ammonia volatilization in Amazonian soils. Pesqui. Agropecu. Bras. 2019, 54, e00253. [Google Scholar] [CrossRef]
- Liang, Y.; Zhang, Y.; Liu, T.; Si, Z.; Gao, Y. Efficiency of 3,4-dimethylpyrazole phosphate in mitigating N2O emission varied with irrigation regime in drip-irrigated wheat field. Agronomy 2024, 14, 3052. [Google Scholar] [CrossRef]
- Tufail, M.A.; Naeem, A.; Arif, M.S.; Farooq, T.H.; Shahzad, S.M.; Dar, A.A.; Shakoor, A. Unraveling the efficacy of nitrification inhibitors (DCD and DMPP) in reducing nitrogen gases emissions across agroecosystems: A three-decade global data synthesis (1993–2021). Fuel 2022, 324, 124725. [Google Scholar] [CrossRef]
- Cui, L.; Li, D.; Wu, Z.; Xue, Y.; Xiao, F.; Zhang, L.; Cui, Y. Effects of nitrification inhibitors on soil nitrification and ammonia volatilization in three soils with different pH. Agronomy 2021, 11, 1674. [Google Scholar] [CrossRef]
- Cassim, B.M.A.R.; Lisboa, I.P.; Besen, M.R.; Otto, R.; Cantarella, H.; Inoue, T.T.; Batista, M.A. Nitrogen: From discovery, plant assimilation, sustainable usage to current enhanced efficiency fertilizers technologies—A review. Rev. Bras. Cienc. Solo 2024, 48, e0230037. [Google Scholar] [CrossRef]
- Tufail, M.A.; Irfan, M.; Umar, W.; Wakeel, A.; Schmitz, R.A. Mediation of gaseous emissions and improving plant productivity by DCD and DMPP nitrification inhibitors: Meta-analysis of last three decades. Environ. Sci. Pollut. Res. 2023, 30, 64719–64735. [Google Scholar] [CrossRef]
- Guo, X.; Ren, H.; Ren, B.; Zhang, J.; Liu, P.; Shah, S.; Zhao, B. Long-term application of controlled-release urea reduced ammonia volatilization, raising the risk of N2O emissions and improved summer maize yield. Field Crops Res. 2024, 306, 109227. [Google Scholar] [CrossRef]
- Tang, J.; Xu, J.; Fan, H.; Li, R.; Shi, M.; Liu, Y.; Liu, P. Preparation of hydroxy-terminated polydimethylsiloxane and nano-SiO2 hydrophobic polyurethane coated urea and investigation of controlled nitrogen release. Chem. Eng. Sci. 2025, 302, 120728. [Google Scholar] [CrossRef]
- Hassan, M.U.; Guoqin, H.; Arif, M.S.; Mubarik, M.S.; Tang, H.; Xu, H.; Shakoor, A. Can urea-coated fertilizers be an effective means of reducing greenhouse gas emissions and improving crop productivity? J. Environ. Manage. 2024, 367, 121927. [Google Scholar] [CrossRef]
- Li, J.; Wang, D.; Chen, T.; Zhou, W.; Zhan, X. Risks of microplastics from polyurethane and polyethylene-polycarbonate coated fertilizers to soil–crop system. J. Hazard. Mater. 2025, 140181. [Google Scholar] [CrossRef] [PubMed]
- Isakov, V.; Vlasova, E.; Forer, V.; Kenny, J.; Lyulin, S. Analysis of slow-released fertilisers as a source of microplastics. Land 2024, 14, 38. [Google Scholar] [CrossRef]
- Sun, Y.; Wu, Q.; Chen, H.; Jia, X.; Gong, F.; Liu, X.; Chi, D. N-loaded clinoptilolite under water-saving irrigation mitigates ammonia volatilization while increasing grain yield and water–nitrogen use efficiency. Field Crops Res. 2023, 300, 109000. [Google Scholar] [CrossRef]
- Sun, Y.; Zhou, H.; Sun, Y.; Wu, Q.; Chen, H.; Chi, D. The application of nano-clinoptilolite based nitrogen fertilizer mixed with urea promotes nitrogen balance and enhances economic and ecological benefits in paddy fields. J. Clean. Prod. 2024, 453, 142257. [Google Scholar] [CrossRef]
- Dawar, K.; Dawar, A.; Tariq, M.; Mian, I.A.; Muhammad, A.; Farid, L.; Tahzeeb-ul-Hassan, M. Enhancing nitrogen use efficiency and yield of maize through ammonia volatilization mitigation and nitrogen management approaches. BMC Plant Biol. 2024, 24, 74. [Google Scholar] [CrossRef]
- Chen, H.; Zhou, H.; Sun, Y.; Liu, X.; Wu, Q.; Chi, D. Nitrogen-loaded biochar for environmental management: Enhancing nitrogen utilization balance in farmland, mitigating ammonia volatilization, and improving fertilizer efficiency. Environ. Technol. Innov. 2025, 37, 104006. [Google Scholar] [CrossRef]
- Jin, P.; Chen, Z.; Wang, H.; Lv, R.; Hu, T.; Zhou, R.; Xie, Z. 12-year continuous biochar application: Mitigating reactive nitrogen loss in paddy fields but without rice yield enhancement. Agric. Ecosyst. Environ. 2024, 375, 109223. [Google Scholar] [CrossRef]
- Selvarajh, G.; Ch’ng, H.Y.; Zain, N.M.; Wei, L.S.; Liew, J.Y.; Azmin, S.N.H.M.; Damrongrak, I. Enriched rice husk biochar superior to commercial biochar in ameliorating ammonia loss from urea fertilizer and improving plant uptake. Heliyon 2024, 10, e32080. [Google Scholar] [CrossRef]
- Ma, Y.; Xie, W.; Yao, R.; Feng, Y.; Wang, X.; Xie, H.; Yang, J. Biochar and hydrochar application influence soil ammonia volatilization and the dissolved organic matter in salt-affected soils. Sci. Total Environ. 2024, 926, 171845. [Google Scholar] [CrossRef]
- He, Z.; Lue, H.; Wang, Y.; Yuan, H.; Liu, Y.; Li, N.; He, L. Effects of biochar combined with nitrification inhibitors on NH3 and N2O emission under different water conditions from vegetable soils. Front. Microbiol. 2025, 16, 1547979. [Google Scholar] [CrossRef]
- Xue, L.; Sun, B.; Yang, Y.; Jin, B.; Zhuang, G.; Bai, Z.; Zhuang, X. Efficiency and mechanism of reducing ammonia volatilization in alkaline farmland soil using Bacillus amyloliquefaciens biofertilizer. Environ. Res. 2021, 202, 111672. [Google Scholar] [CrossRef]
- Maihaiti, M.; Ma, S.; Wang, X.; Sun, X.; Xia, B.; Yang, H.; Zhuang, X. A study of controlling of soil ammonia volatilization by Bacillus amyloliquefaciens and its mechanism. Water Air Soil Pollut. 2023, 234, 3. [Google Scholar] [CrossRef]
- Li, H.; Liang, X.; Lian, Y.; Xu, L.; Chen, Y. Reduction of ammonia volatilization from urea by a floating duckweed in flooded rice fields. Soil Sci. Soc. Am. J. 2009, 73, 1890–1895. [Google Scholar] [CrossRef]
- Yang, G.; Ji, H.; Liu, H.; Feng, Y.; Zhang, Y.; Chen, L.; Guo, Z. Nitrogen fertilizer reduction in combination with Azolla cover for reducing ammonia volatilization and improving nitrogen use efficiency of rice. PeerJ 2021, 9, e11077. [Google Scholar] [CrossRef] [PubMed]
- Schoof, J.; Fuß, R.; Wulf, S.; Pacholski, A. Regionalized ammonia emission abatement by urease inhibitor treatment of urea for agro-environmental conditions of Western Central Europe. Geoderma 2025, 457, 117285. [Google Scholar] [CrossRef]
- Cancian, A.; Viero, F.; Fontoura, S.M.V.; Soares, J.R.; Bayer, C. Ammonia volatilization from NBPT-treated urea in no-till maize at different soil textures in South Brazil. Geoderma Reg. 2025, 40, e00946. [Google Scholar] [CrossRef]
- Teagasc. Protected Urea (NBPT). Available online: https://teagasc.ie/crops/soil--soil-fertility/protected-urea-nbpt/ (accessed on 20 March 2026).
- Da Fonseca, A.B.; Santos, C.; Nunes, A.P.P.; Oliveira, D.P.; De Melo, M.E.A.; Takayama, T.; Guelfi, D. Urease inhibitors technologies as strategy to mitigate agricultural ammonia emissions and enhance the use efficiency of urea-based fertilizers. Sci. Rep. 2023, 13, 22739. [Google Scholar] [CrossRef]
- Qiao, Y.; Yue, G.; Mo, X.; Zhang, L.; Sun, S. Controlled-release urea derived from various coating materials on the impacts of maize production: A meta-analysis. Ind. Crops Prod. 2025, 225, 120485. [Google Scholar] [CrossRef]
- Lü, H.D. Assessment of the crucial factors influencing the responses of ammonia and nitrous oxide emissions to controlled release nitrogen fertilizer: A meta-analysis. J. Integr. Agric. 2023, 22, 3549–3559. [Google Scholar] [CrossRef]
- Nitrogen, Smart. Recommended ESN blends produce maximum profits. Available online: https://smartnitrogen.com/facts-from-the-field/recommended-esn-blends-produce-maximum-profits/ (accessed on 20 March 2026).
- Wyngaard, N.; Crespo, C.; Iglesias, M.P.; Sainz Rozas, H.R.; Aparicio, V.; Toribio, M.; Reussi Calvo, N.I. Impact of a nitrification inhibitor on nitrogen losses in contrasting soils and water regimes. J. Soil Sci. Plant Nutr. 2026, 1–12. [Google Scholar] [CrossRef]
- Torralbo, F.; Boardman, D.; Houx, J.H., III; Fritschi, F.B. Distinct enhanced efficiency urea fertilizers differentially influence ammonia volatilization losses and maize yield. Plant Soil 2022, 475, 551–563. [Google Scholar] [CrossRef]
- European Environment Agency. EMEP/EEA Air Pollutant Emission Inventory Guidebook 2023. Part B: Sectoral Guidance Chapters. 3. Agriculture. 3.D Agricultural Soils 2023. Available online: https://www.eea.europa.eu/en/analysis/publications/emep-eea-guidebook-2023 (accessed on 20 March 2026).
- Hurtado, C.; Doltra, J.; Villar-Mir, J.M.; Cabrera, M.L.; Quemada, M. Ammonia emissions from fertilized Mediterranean soils: Drivers, variability and mitigation potential. Agric. Ecosyst. Environ. 2024, 356, 108630. [Google Scholar]
- Gurung, R.; Bhandari, H.; Brinton, S.; Mulla, D.J. Soil buffering capacity and ammonia volatilization: Interactions with soil properties and management. Soil Sci. Soc. Am. J. 2021, 85, 1502–1514. [Google Scholar]
- Sarfraz, U.; Qian, Y.; Yu, Q.; Cao, Y.; Jiang, X.; Mahreen, N.; Tao, R.; Ma, Q.; Zhu, M.; Ding, J.; Li, C.; Guo, W.; Zhu, X. Microplastic effects on soil nitrogen storage, nitrogen emissions, and ammonia volatilization in relation to soil health and crop productivity: Mechanisms and future considerations. Front. Plant Sci. 2025, 16, 1621542. [Google Scholar] [CrossRef] [PubMed]
- Wester-Larsen, L.; Elsgaard, L.; Søgaard, H.T.; Petersen, S.O. Soil buffering and ammonia volatilization in sandy soils: Role of organic matter and pH interactions. Biogeochemistry 2022, 160, 45–60. [Google Scholar]
- Abeed, A.A.; Abd El-Mageed, T.A.; Rady, M.M.; Semida, W.M. Climate and management interactions controlling ammonia volatilization in Mediterranean cropping systems. Agronomy 2023, 13, 1890. [Google Scholar]
- Das, S.; Saha, A.; Adhya, T.K. Temperature and moisture controls on ammonia volatilization from fertilized soils: Implications for management. J. Environ. Manage. 2024, 350, 119600. [Google Scholar]
- Jiménez-de-Santiago, D.; Guardia, G.; Sanz-Cobena, A.; Vallejo, A. Drivers of spatial variability in ammonia volatilization from fertilized soils across cropping systems. Agric. Ecosyst. Environ. 2024, 349, 108564. [Google Scholar]
- Wang, Y.; Guo, T.; Bai, S.H.; Chen, F.; Zhang, M. Interactions between nitrogen forms and ammonia volatilization under different soil conditions. Sci. Total Environ. 2025, 926, 172245. [Google Scholar]
- Wu, D.; Cardenas, L.M.; Calvet, S.; Brüggemann, N.; Loick, N.; Liu, S.; Bol, R. The effect of nitrification inhibitors on ammonia volatilization: A review. Agric. Ecosyst. Environ. 2021, 318, 107512. [Google Scholar]
- Wang, Z.; Li, T.; Shen, W.; Huang, J.; Feng, Y.; Yang, L. Global assessment of enhanced-efficiency fertilizers on nitrogen losses and crop productivity. Chemosphere 2025, 345, 140123. [Google Scholar]
- Kohira, M.; Saito, T.; Yamamoto, A.; Nakajima, Y. Challenges and future directions in nitrogen management for sustainable agriculture. Agric. Syst. 2024, 220, 103873. [Google Scholar]
- Sha, Z.; Li, X.; Wang, Y.; Chen, D.; Zhang, J. Improving prediction of ammonia volatilization from agricultural soils: Model limitations and future perspectives. Sci. Total Environ. 2023, 875, 162487. [Google Scholar]
- Wang, J.; Liu, X.; Zhang, Y.; Li, Q.; Chen, W. Biological nitrification inhibition in agroecosystems: Mechanisms and applications. Plant Soil 2021, 466, 1–18. [Google Scholar]
- Chen, H.; Zhang, S.; Liu, Y.; Wang, Q.; Zhao, L. Integrated nitrogen management strategies for improving nitrogen use efficiency and reducing emissions in cropping systems. Agric. Ecosyst. Environ. 2024, 352, 108612. [Google Scholar]
- FertiCovery Project. Nutrient recovery from secondary raw materials for sustainable fertilization. Horizon Europe Project 2025. Available online: https://ferticovery.eu (accessed on 30 March 2026).
- LIFE Ammonia Trapping Project. Recovery of ammonia emissions from waste streams using gas-permeable membranes. LIFE Programme 2020. Available online: https://lifeammoniatrapping.eu (accessed on 30 March 2026).
- LIFE Green Ammonia Project. Sustainable ammonia recovery and reuse technologies. LIFE Programme 2026. Available online: https://lifegreenammonia.eu (accessed on 30 March 2026).
- NitroScope Project. Digital tools for monitoring and optimizing nitrogen fertilization in agriculture. 2025. Available online: https://nitroscope.eu (accessed on 30 March 2026).
| Process | Main field drivers | Conditions increasing NH3 emissions | Mitigation approaches |
| Urea hydrolysis and formation of NH4+ hotspots | Urease activity, soil moisture, fertilizer placement | Surface-applied urea, warm conditions, localized concentration | Urease inhibitors, soil incorporation, timely irrigation or rainfall |
| NH4+/NH3 equilibrium (speciation) | Soil pH, temperature, buffering capacity, surface water | High pH soils, elevated temperature, water accumulation | Local pH adjustment (acidifying amendments), avoid surface water accumulation |
| Ammonium retention (adsorption and CEC) | Soil texture, cation exchange capacity, organic matter | Low CEC soils, sandy textures, surface application | Use of adsorbents (e.g., zeolites, clays), improve soil–fertilizer contact |
| Transport of NH3 to the atmosphere | Wind speed, turbulence, soil moisture, boundary-layer resistance | Windy conditions, exposed soil surface, low resistance to diffusion | Soil incorporation, surface cover, application under low-wind conditions |
| Technology | Feedstock | Process description | Output products | Development |
| Rustica biochar pilot | Lignocellulosic fractions from fruit and vegetable residues (stems, cores, peels) | Pyrolysis under oxygen-limited conditions (350–700 °C), followed by cooling, sieving and optional enrichment | Fine or granulated biochar for soil conditioning and microbial carriers | TRL 7 |
| Abfallwirtschaftsbetriebe Münster (AWM) | Green waste from garden and park maintenance | Small-scale pyrolysis under oxygen-free conditions at elevated temperature | Biochar for potential soil amendment use | TRL 7 |
| AgroAmerica | Pig manure and digestate from organic residues | Solid–liquid separation, drying, pyrolysis with heat recovery, ammonia recovery and potassium extraction | Biochar, ammonia solution, potassium concentrate, treated water | TRL 9 |
| CarbonFX (Airex Energy) | Forestry and agricultural biomass residues | Torrefaction in cyclonic reactor with pre-drying and densification steps | Biocoal, biochar, biocoke | TRL 9 |
| TerraNova Ultra | Dewatered sewage sludge (5–30% dry matter) | Hydrothermal carbonization (~200 °C, pressurised), heat recovery and dewatering | Hydrochar, Ca-based P products, ammonium sulphate | Not specified |
| Technology | Feedstock | Process description | Output products | Development |
| REALM | Greenhouse runoff and drainage water | Microalgae cultivation using nutrients (N, P) in water, supported by solar energy and CO2 capture; treated water reused | Algal biomass for biostimulants and aquaculture feed | TRL 7 |
| SABANA | Marine water enriched with nutrients from waste streams | Cultivation in thin-layer cascade and raceway pond systems | Biostimulants, biopesticides, feed additives and biofertilisers | TRL 8 |
| WALNUT pilot system | Industrial wastewater | Hybrid cultivation combining photoautotrophic and heterotrophic growth modes | Microalgal biomass for fertiliser, feed or bio-based applications | TRL 6 |
| LIFE ALGAECAN | Saline wastewater from food processing industries | Closed heterotrophic cultivation, biomass separation and drying, integrated with renewable energy | Treated water and microalgae powder | TRL 8 |
| EXTRAALGAE (NCBR) | Marine and cultivated algae species | Supercritical CO2 extraction followed by formulation into stable emulsions | Algae-based biostimulant formulations | TRL 7 |
| Technology | Feedstock | Process description | Output products | Development |
| 2 | Mixture of manure, organic wastes and/or energy crops | Chemical air scrubber used to capture ammonia from the solid fraction of digestate | Liquid ammonium sulphate (~8% N; ~25% SO4) | TRL 9 |
| BTS Biogas NITROStrip | Animal manure; agricultural residues and agro-industrial by-products | Ammonia stripping followed by acid scrubbing after heating and aeration of digestate | Ammonium sulphate solution | TRL 9 |
| H2-VOLAZ | Pig manure and digestate | Separation (NF/RO) combined with ammonia recovery via scrubbing | Ammonium sulphate; clean water; nutrient concentrates | TRL 9 |
| Detricon | Liquid fraction of manure, digestate or similar waste streams | Stripping and scrubbing system for nitrogen recovery from liquid waste streams | Liquid fertiliser based on ammonium salts | TRL 9 |
| N2Applied | Livestock slurry; liquid digestate fraction | Plasma-based nitrogen fixation converting slurry into enriched fertiliser | Liquid fertiliser based on ammonium nitrate | TRL 9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




