Submitted:
31 March 2026
Posted:
01 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
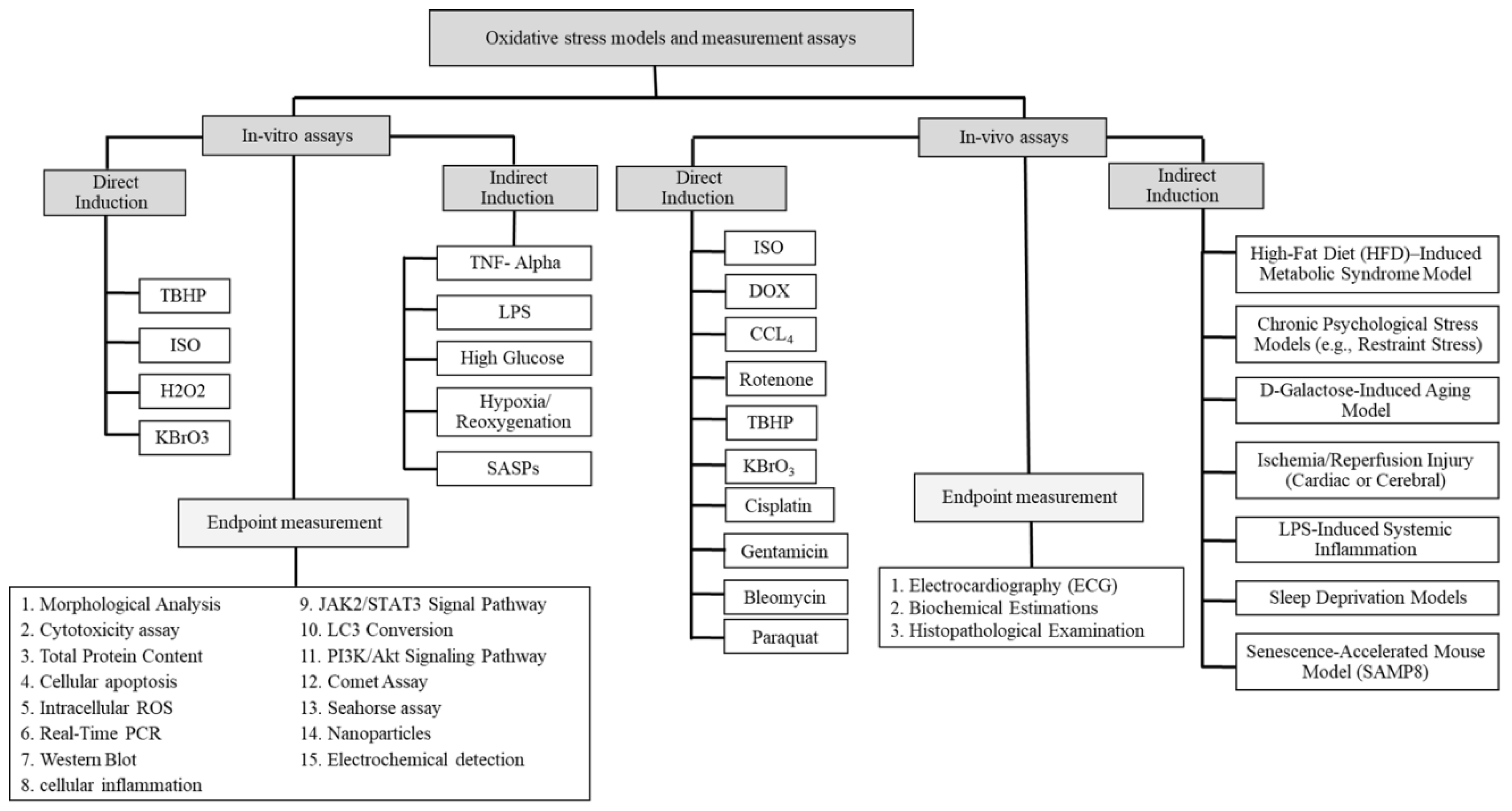
3. Experimental In-Vitro Models
4. Direct Inducers of Oxidative Stress In-Vitro
4.1. Tert-Butyl Hydroperoxide (TBHP)
4.2. Isoproterenol (ISO)
4.3. Hydrogen Peroxide (H2O2)
4.4. Potassium Bromate (KBrO3)
4.5. Indirect Inducers of Oxidative Stress in In-Vitro
4.5.1. Tumor Necrosis Factor-Alpha (TNF-α)
4.5.2. Lipopolysaccharide (LPS)
4.5.3. High Glucose (HG)
4.5.4. Hypoxia/Reoxygenation (H/R)
4.5.5. Senescent Cell Co-Culture
| Inducers | Experimental observation | Molecular Mechanisms | Inducer dose | EC50/appotosis |
|---|---|---|---|---|
| TBHP |
|
|
|
|
| ISO |
|
|
|
|
| H2O2 |
|
|
|
|
| KBrO3 |
|
|
|
|
5. In-Vitro Assays for Oxidative Stress and DNA Damage in Cardiovascular Diseases
Morphological Analysis
6. Cytotoxicity Assay
6.1. Determination of Total Protein Content
6.2. Determination of Cellular Apoptosis
6.3. Determination of Intracellular ROS
7. Quantitative Real-Time PCR
8. Western Blot
9. Measurement of Cellular Inflammation
10. Evaluation of JAK2/STAT3 Signal Pathway
11. Evaluation of LC3 Conversion
11.1. PI3K/Akt Signaling Pathway
11.2. Comet Assay to Evaluate the Oxidative DNA Damage
12. Seahorse Assay
13. Nanoparticles for Detection and Monitoring the Reactive Oxygen Species (ROS)
14. Electrochemical Detection
15. Electron Paramagnetic (Spin) Resonance (EPR/ESR)
16. Extracellular H2O2 Detection by Amplex Red
17. Genetic Sensors for Mitochondrial ROS Measurement
18. In-Vivo Assays
18.1. Experimental In-Vivo Models
18.2. Direct Inducers of Oxidative Stress In-Vivo
18.3. Doxorubicin (DOX)
18.4. Carbon Tetrachloride (CCl4)
18.5. Cisplatin
18.6. Gentamicin
18.7. Bleomycin
18.8. Rotenone
18.9. Paraquat
18.10. Tert-Butyl Hydroperoxide (TBHP)
18.11. Potassium Bromate (KBrO3)
18.12. Indirect Inducers of Oxidative Stress In-Vivo
18.13. High-Fat Diet (HFD)-Induced Metabolic Stress
18.14. Lipopolysaccharide (LPS)-Induced Systemic Inflammation
18.15. D-Galactose-Induced Aging Model
18.16. Hypoxia/Reoxygenation (H/R) Injury Model
18.17. Chronic Restraint Stress Model
18.18. Sleep Deprivation Model
18.19. Senescence-Accelerated Mouse Model (SAMP8)
19. In-Vivo Assays for Oxidative Stress and DNA Damage in Cardiovascular Diseases
19.1. Electrocardiography (ECG)
19.2. Biochemical Estimations
19.3. Histopathological Examination
20. Conclusion and Future Perspective
Author Contributions Statement
Competing Interests
References
- Flora, G.D.; Nayak, M.K. A Brief Review of Cardiovascular Diseases, Associated Risk Factors and Current Treatment Regimes. Current pharmaceutical design 2019, 25, 4063-4084. [CrossRef]
- Dunbar, S.B.; Khavjou, O.A.; Bakas, T.; Hunt, G.; Kirch, R.A.; Leib, A.R.; Morrison, R.S.; Poehler, D.C.; Roger, V.L.; Whitsel, L.P. Projected Costs of Informal Caregiving for Cardiovascular Disease: 2015 to 2035: A Policy Statement From the American Heart Association. Circulation 2018, 137, e558-e577. [CrossRef]
- Ruan, Y.; Guo, Y.; Zheng, Y.; Huang, Z.; Sun, S.; Kowal, P.; Shi, Y.; Wu, F. Cardiovascular disease (CVD) and associated risk factors among older adults in six low-and middle-income countries: results from SAGE Wave 1. BMC public health 2018, 18, 778. [CrossRef]
- Tsermpini, E.E.; Plemenitaš Ilješ, A.; Dolžan, V. Alcohol-Induced Oxidative Stress and the Role of Antioxidants in Alcohol Use Disorder: A Systematic Review. Antioxidants (Basel) 2022, 11. [CrossRef]
- Sambiagio, N.; Berthet, A.; Wild, P.; Sauvain, J.J.; Auer, R.; Schoeni, A.; Rodondi, N.; Feller, M.; Humair, J.P.; Berlin, I.; et al. Associations between urinary biomarkers of oxidative stress and biomarkers of tobacco smoke exposure in smokers. The Science of the total environment 2022, 852, 158361. [CrossRef]
- Pizzino, G.; Irrera, N.; Cucinotta, M.; Pallio, G.; Mannino, F.; Arcoraci, V.; Squadrito, F.; Altavilla, D.; Bitto, A. Oxidative Stress: Harms and Benefits for Human Health. Oxidative medicine and cellular longevity 2017, 2017, 8416763. [CrossRef]
- Dubois-Deruy, E.; Peugnet, V.; Turkieh, A.; Pinet, F. Oxidative Stress in Cardiovascular Diseases. Antioxidants (Basel) 2020, 9. [CrossRef]
- Othman, E.M.; Hintzsche, H.; Stopper, H. Signaling steps in the induction of genomic damage by insulin in colon and kidney cells. Free Radic Biol Med 2014, 68, 247-257. [CrossRef]
- Snezhkina, A.V.; Kudryavtseva, A.V.; Kardymon, O.L.; Savvateeva, M.V.; Melnikova, N.V.; Krasnov, G.S.; Dmitriev, A.A. ROS Generation and Antioxidant Defense Systems in Normal and Malignant Cells. Oxidative medicine and cellular longevity 2019, 2019, 6175804. [CrossRef]
- Othman, E.M.; Oli, R.G.; Arias-Loza, P.A.; Kreissl, M.C.; Stopper, H. Metformin Protects Kidney Cells From Insulin-Mediated Genotoxicity In Vitro and in Male Zucker Diabetic Fatty Rats. Endocrinology 2016, 157, 548-559. [CrossRef]
- D'Oria, R.; Schipani, R.; Leonardini, A.; Natalicchio, A.; Perrini, S.; Cignarelli, A.; Laviola, L.; Giorgino, F. The Role of Oxidative Stress in Cardiac Disease: From Physiological Response to Injury Factor. Oxidative medicine and cellular longevity 2020, 2020, 5732956. [CrossRef]
- Kurian, G.A.; Rajagopal, R.; Vedantham, S.; Rajesh, M. The Role of Oxidative Stress in Myocardial Ischemia and Reperfusion Injury and Remodeling: Revisited. Oxidative medicine and cellular longevity 2016, 2016, 1656450. [CrossRef]
- Antoniades, C.; Tousoulis, D.; Tentolouris, C.; Toutouzas, P.; Stefanadis, C. Oxidative stress, antioxidant vitamins, and atherosclerosis. From basic research to clinical practice. Herz 2003, 28, 628-638. [CrossRef]
- Jové, M.; Mota-Martorell, N.; Pradas, I.; Martín-Gari, M.; Ayala, V.; Pamplona, R. The Advanced Lipoxidation End-Product Malondialdehyde-Lysine in Aging and Longevity. Antioxidants (Basel) 2020, 9. [CrossRef]
- Sanderson, K.J.; van Rij, A.M.; Wade, C.R.; Sutherland, W.H. Lipid peroxidation of circulating low density lipoproteins with age, smoking and in peripheral vascular disease. Atherosclerosis 1995, 118, 45-51. [CrossRef]
- Nacítarhan, S.; Özben, T.; Tuncer, N.e. Serum and urine malondialdehyde levels in NIDDM patients with and without hyperlipidemia. Free Radical Biology and Medicine 1995, 19, 893-896. [CrossRef]
- Garbern, J.C.; Mummery, C.L.; Lee, R.T. Model systems for cardiovascular regenerative biology. Cold Spring Harbor perspectives in medicine 2013, 3, a014019. [CrossRef]
- Capes-Davis, A.; Bairoch, A.; Barrett, T.; Burnett, E.C.; Dirks, W.G.; Hall, E.M.; Healy, L.; Kniss, D.A.; Korch, C.; Liu, Y.; et al. Cell Lines as Biological Models: Practical Steps for More Reliable Research. Chemical research in toxicology 2019, 32, 1733-1736. [CrossRef]
- Field, L.J. Atrial natriuretic factor-SV40 T antigen transgenes produce tumors and cardiac arrhythmias in mice. Science (New York, N.Y.) 1988, 239, 1029-1033. [CrossRef]
- Claycomb, W.C.; Lanson, N.A., Jr.; Stallworth, B.S.; Egeland, D.B.; Delcarpio, J.B.; Bahinski, A.; Izzo, N.J., Jr. HL-1 cells: a cardiac muscle cell line that contracts and retains phenotypic characteristics of the adult cardiomyocyte. Proceedings of the National Academy of Sciences of the United States of America 1998, 95, 2979-2984. [CrossRef]
- Davidson, M.M.; Nesti, C.; Palenzuela, L.; Walker, W.F.; Hernandez, E.; Protas, L.; Hirano, M.; Isaac, N.D. Novel cell lines derived from adult human ventricular cardiomyocytes. Journal of molecular and cellular cardiology 2005, 39, 133-147. [CrossRef]
- Watkins, S.J.; Borthwick, G.M.; Arthur, H.M. The H9C2 cell line and primary neonatal cardiomyocyte cells show similar hypertrophic responses in vitro. In vitro cellular & developmental biology. Animal 2011, 47, 125-131. [CrossRef]
- Ellingsen, O.; Davidoff, A.J.; Prasad, S.K.; Berger, H.J.; Springhorn, J.P.; Marsh, J.D.; Kelly, R.A.; Smith, T.W. Adult rat ventricular myocytes cultured in defined medium: phenotype and electromechanical function. The American journal of physiology 1993, 265, H747-754. [CrossRef]
- Mitcheson, J.S.; Hancox, J.C.; Levi, A.J. Cultured adult cardiac myocytes: future applications, culture methods, morphological and electrophysiological properties. Cardiovascular research 1998, 39, 280-300. [CrossRef]
- Moretti, A.; Laugwitz, K.L.; Dorn, T.; Sinnecker, D.; Mummery, C. Pluripotent stem cell models of human heart disease. Cold Spring Harbor perspectives in medicine 2013, 3. [CrossRef]
- Witek, P.; Korga, A.; Burdan, F.; Ostrowska, M.; Nosowska, B.; Iwan, M.; Dudka, J. The effect of a number of H9C2 rat cardiomyocytes passage on repeatability of cytotoxicity study results. Cytotechnology 2016, 68, 2407-2415. [CrossRef]
- Zordoky, B.N.; El-Kadi, A.O. H9c2 cell line is a valuable in vitro model to study the drug metabolizing enzymes in the heart. Journal of pharmacological and toxicological methods 2007, 56, 317-322. [CrossRef]
- Kimes, B.W.; Brandt, B.L. Properties of a clonal muscle cell line from rat heart. Experimental cell research 1976, 98, 367-381. [CrossRef]
- Curtis, M.W.; Russell, B. Micromechanical regulation in cardiac myocytes and fibroblasts: implications for tissue remodeling. Pflugers Archiv : European journal of physiology 2011, 462, 105-117. [CrossRef]
- Cerbai, E.; Sartiani, L.; De Paoli, P.; Mugelli, A. Isolated cardiac cells for electropharmacological studies. Pharmacological research 2000, 42, 1-8. [CrossRef]
- Ribeiro, A.J.; Ang, Y.S.; Fu, J.D.; Rivas, R.N.; Mohamed, T.M.; Higgs, G.C.; Srivastava, D.; Pruitt, B.L. Contractility of single cardiomyocytes differentiated from pluripotent stem cells depends on physiological shape and substrate stiffness. Proceedings of the National Academy of Sciences of the United States of America 2015, 112, 12705-12710. [CrossRef]
- von Gise, A.; Lin, Z.; Schlegelmilch, K.; Honor, L.B.; Pan, G.M.; Buck, J.N.; Ma, Q.; Ishiwata, T.; Zhou, B.; Camargo, F.D.; et al. YAP1, the nuclear target of Hippo signaling, stimulates heart growth through cardiomyocyte proliferation but not hypertrophy. Proceedings of the National Academy of Sciences of the United States of America 2012, 109, 2394-2399. [CrossRef]
- Guo, L.; Abrams, R.M.; Babiarz, J.E.; Cohen, J.D.; Kameoka, S.; Sanders, M.J.; Chiao, E.; Kolaja, K.L. Estimating the risk of drug-induced proarrhythmia using human induced pluripotent stem cell-derived cardiomyocytes. Toxicological sciences : an official journal of the Society of Toxicology 2011, 123, 281-289. [CrossRef]
- Lu, J.; Wang, H.Z.; Jia, Z.; Zuckerman, J.; Lu, Z.; Guo, Y.; Boink, G.J.; Brink, P.R.; Robinson, R.B.; Entcheva, E.; et al. Improving cardiac conduction with a skeletal muscle sodium channel by gene and cell therapy. Journal of cardiovascular pharmacology 2012, 60, 88-99. [CrossRef]
- Chaicharoenaudomrung, N.; Kunhorm, P.; Noisa, P. Three-dimensional cell culture systems as an in vitro platform for cancer and stem cell modeling. World journal of stem cells 2019, 11, 1065-1083. [CrossRef]
- Pontes Soares, C.; Midlej, V.; de Oliveira, M.E.; Benchimol, M.; Costa, M.L.; Mermelstein, C. 2D and 3D-organized cardiac cells shows differences in cellular morphology, adhesion junctions, presence of myofibrils and protein expression. PLoS One 2012, 7, e38147. [CrossRef]
- Shimizu, T.; Yamato, M.; Isoi, Y.; Akutsu, T.; Setomaru, T.; Abe, K.; Kikuchi, A.; Umezu, M.; Okano, T. Fabrication of pulsatile cardiac tissue grafts using a novel 3-dimensional cell sheet manipulation technique and temperature-responsive cell culture surfaces. Circulation research 2002, 90, e40. [CrossRef]
- Kirkpatrick, C.J.; Fuchs, S.; Unger, R.E. Co-culture systems for vascularization--learning from nature. Advanced drug delivery reviews 2011, 63, 291-299. [CrossRef]
- Mehling, M.; Tay, S. Microfluidic cell culture. Current Opinion in Biotechnology 2014, 25, 95-102. [CrossRef]
- Duell, B.L.; Cripps, A.W.; Schembri, M.A.; Ulett, G.C. Epithelial cell coculture models for studying infectious diseases: benefits and limitations. Journal of biomedicine & biotechnology 2011, 2011, 852419. [CrossRef]
- Halldorsson, S.; Lucumi, E.; Gómez-Sjöberg, R.; Fleming, R.M.T. Advantages and challenges of microfluidic cell culture in polydimethylsiloxane devices. Biosensors and Bioelectronics 2015, 63, 218-231. [CrossRef]
- Bi, Y.M.; Wu, Y.T.; Chen, L.; Tan, Z.B.; Fan, H.J.; Xie, L.P.; Zhang, W.T.; Chen, H.M.; Li, J.; Liu, B.; et al. 3,5-Dicaffeoylquinic acid protects H9C2 cells against oxidative stress-induced apoptosis via activation of the PI3K/Akt signaling pathway. Food & nutrition research 2018, 62. [CrossRef]
- Fan, H.J.; Tan, Z.B.; Wu, Y.T.; Feng, X.R.; Bi, Y.M.; Xie, L.P.; Zhang, W.T.; Ming, Z.; Liu, B.; Zhou, Y.C. The role of ginsenoside Rb1, a potential natural glutathione reductase agonist, in preventing oxidative stress-induced apoptosis of H9C2 cells. Journal of ginseng research 2020, 44, 258-266. [CrossRef]
- Wu, Y.T.; Xie, L.P.; Hua, Y.; Xu, H.L.; Chen, G.H.; Han, X.; Tan, Z.B.; Fan, H.J.; Chen, H.M.; Li, J.; et al. Tanshinone I Inhibits Oxidative Stress-Induced Cardiomyocyte Injury by Modulating Nrf2 Signaling. Frontiers in pharmacology 2021, 12, 644116. [CrossRef]
- T, M.M.; Anand, T.; Khanum, F. Attenuation of cytotoxicity induced by tBHP in H9C2 cells by Bacopa monniera and Bacoside A. Pathophysiology : the official journal of the International Society for Pathophysiology 2018, 25, 143-149. [CrossRef]
- Han, S.; Chen, L.; Zhang, Y.; Xie, S.; Yang, J.; Su, S.; Yao, H.; Shi, P. Lotus Bee Pollen Extract Inhibits Isoproterenol-Induced Hypertrophy via JAK2/STAT3 Signaling Pathway in Rat H9c2 Cells. Antioxidants (Basel) 2022, 12. [CrossRef]
- Han, D.; Wan, C.; Liu, F.; Xu, X.; Jiang, L.; Xu, J. Jujuboside A Protects H9C2 Cells from Isoproterenol-Induced Injury via Activating PI3K/Akt/mTOR Signaling Pathway. Evidence-Based Complementary and Alternative Medicine 2016, 2016, 9593716. [CrossRef]
- Fan, C.; Tang, X.; Ye, M.; Zhu, G.; Dai, Y.; Yao, Z.; Yao, X. Qi-Li-Qiang-Xin Alleviates Isoproterenol-Induced Myocardial Injury by Inhibiting Excessive Autophagy via Activating AKT/mTOR Pathway. Frontiers in pharmacology 2019, 10, 1329. [CrossRef]
- Fan, C.L.; Yao, Z.H.; Ye, M.N.; Fu, L.L.; Zhu, G.N.; Dai, Y.; Yao, X.S. Fuziline alleviates isoproterenol-induced myocardial injury by inhibiting ROS-triggered endoplasmic reticulum stress via PERK/eIF2α/ATF4/Chop pathway. Journal of cellular and molecular medicine 2020, 24, 1332-1344. [CrossRef]
- Ransy, C.; Vaz, C.; Lombès, A.; Bouillaud, F. Use of H(2)O(2) to Cause Oxidative Stress, the Catalase Issue. Int J Mol Sci 2020, 21. [CrossRef]
- Coyle, C.H.; Kader, K.N. Mechanisms of H2O2-induced oxidative stress in endothelial cells exposed to physiologic shear stress. ASAIO journal (American Society for Artificial Internal Organs : 1992) 2007, 53, 17-22. [CrossRef]
- Anestopoulos, I.; Kavo, A.; Tentes, I.; Kortsaris, A.; Panayiotidis, M.; Lazou, A.; Pappa, A. Silibinin protects H9c2 cardiac cells from oxidative stress and inhibits phenylephrine-induced hypertrophy: potential mechanisms. The Journal of nutritional biochemistry 2013, 24, 586-594. [CrossRef]
- Parsons, J.L.; Chipman, J.K. The role of glutathione in DNA damage by potassium bromate in vitro. Mutagenesis 2000, 15, 311-316. [CrossRef]
- Watanabe, S.; Togashi, S.; Fukui, T. Contribution of nitric oxide to potassium bromate-induced elevation of methaemoglobin concentration in mouse blood. Biological & pharmaceutical bulletin 2002, 25, 1315-1319. [CrossRef]
- Priscilla, D.H.; Prince, P.S. Cardioprotective effect of gallic acid on cardiac troponin-T, cardiac marker enzymes, lipid peroxidation products and antioxidants in experimentally induced myocardial infarction in Wistar rats. Chemico-biological interactions 2009, 179, 118-124. [CrossRef]
- Oseni OA; Ogunmoyole T; Idowu KA. Lipid profile and cardio protective effects of aqueous extract of moringa oleifera (lam) leaf on bromate induced cardiotoxicity on Wistar albino rats. European Journal of Advanced Research in Biological and Life Sciences 2015, 3, 52 66.
- Kuo, S.C.; Li, Y.; Cheng, Y.Z.; Lee, W.J.; Cheng, J.T.; Cheng, K.C. Molecular mechanisms regarding potassium bromate-induced cardiac hypertrophy without apoptosis in H9c2 cells. Molecular medicine reports 2018, 18, 4700-4708. [CrossRef]
- Aggarwal, B.B. Signalling pathways of the TNF superfamily: a double-edged sword. Nature reviews. Immunology 2003, 3, 745-756. [CrossRef]
- Yang, Y.; Bazhin, A.V.; Werner, J.; Karakhanova, S. Reactive oxygen species in the immune system. International reviews of immunology 2013, 32, 249-270. [CrossRef]
- Nishikawa, T.; Edelstein, D.; Du, X.L.; Yamagishi, S.; Matsumura, T.; Kaneda, Y.; Yorek, M.A.; Beebe, D.; Oates, P.J.; Hammes, H.P.; et al. Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature 2000, 404, 787-790. [CrossRef]
- Zorov, D.B.; Juhaszova, M.; Sollott, S.J. Mitochondrial ROS-induced ROS release: an update and review. Biochimica et biophysica acta 2006, 1757, 509-517. [CrossRef]
- Coppé, J.P.; Desprez, P.Y.; Krtolica, A.; Campisi, J. The senescence-associated secretory phenotype: the dark side of tumor suppression. Annual review of pathology 2010, 5, 99-118. [CrossRef]
- Davies, M.J. Detection of peroxyl and alkoxyl radicals produced by reaction of hydroperoxides with rat liver microsomal fractions. The Biochemical journal 1989, 257, 603-606. [CrossRef]
- Crane, D.; Häussinger, D.; Graf, P.; Sies, H. Decreased flux through pyruvate dehydrogenase by thiol oxidation during t-butyl hydroperoxide metabolism in perfused rat liver. Hoppe-Seyler's Zeitschrift fur physiologische Chemie 1983, 364, 977-987. [CrossRef]
- Park, J.; Park, E.; Ahn, B.H.; Kim, H.J.; Park, J.H.; Koo, S.Y.; Kwak, H.S.; Park, H.S.; Kim, D.W.; Song, M.; et al. NecroX-7 prevents oxidative stress-induced cardiomyopathy by inhibition of NADPH oxidase activity in rats. Toxicology and applied pharmacology 2012, 263, 1-6. [CrossRef]
- He, J.; Huang, L.; Sun, K.; Li, J.; Han, S.; Gao, X.; Wang, Q.-Q.; Yang, S.; Sun, W.; Gao, H. Oleuropein alleviates myocardial ischemia–reperfusion injury by suppressing oxidative stress and excessive autophagy via TLR4/MAPK signaling pathway.
- Song, L.; Srilakshmi, M.; Wu, Y.; Saleem, T.S.M. Sulforaphane Attenuates Isoproterenol-Induced Myocardial Injury in Mice. BioMed research international 2020, 2020, 3610285. [CrossRef]
- Zhang, H.; Chen, H.; Li, J.; Bian, Y.; Song, Y.; Li, Z.; He, F.; Liu, S.; Tsai, Y. Hirudin protects against isoproternol-induced myocardial infraction by alleviating oxidative via an Nrf2 dependent manner. International journal of biological macromolecules 2020, 162, 425-435. [CrossRef]
- Liu, F.; Su, H.; Liu, B.; Mei, Y.; Ke, Q.; Sun, X.; Tan, W. STVNa Attenuates Isoproterenol-Induced Cardiac Hypertrophy Response through the HDAC4 and Prdx2/ROS/Trx1 Pathways. Int J Mol Sci 2020, 21. [CrossRef]
- Sabeena Farvin, K.H.; Anandan, R.; Kumar, S.H.; Shiny, K.S.; Sankar, T.V.; Thankappan, T.K. Effect of squalene on tissue defense system in isoproterenol-induced myocardial infarction in rats. Pharmacological research 2004, 50, 231-236. [CrossRef]
- Shao, Y.; Redfors, B.; Scharin Täng, M.; Möllmann, H.; Troidl, C.; Szardien, S.; Hamm, C.; Nef, H.; Borén, J.; Omerovic, E. Novel rat model reveals important roles of β-adrenoreceptors in stress-induced cardiomyopathy. International journal of cardiology 2013, 168, 1943-1950. [CrossRef]
- Gavanji, S.; Bakhtari, A.; Famurewa, A.C.; Othman, E.M. Cytotoxic Activity of Herbal Medicines as Assessed in Vitro: A review. Chemistry & biodiversity 2023, 10.1002/cbdv.202201098. [CrossRef]
- Mu, R.; Ye, S.; Lin, R.; Li, Y.; Guo, X.; An, L. Effects of Peroxiredoxin 6 and Its Mutants on the Isoproterenol Induced Myocardial Injury in H9C2 Cells and Rats. Oxidative medicine and cellular longevity 2022, 2022, 2576310. [CrossRef]
- Cinar, I.; Yayla, M.; Tavaci, T.; Toktay, E.; Ugan, R.A.; Bayram, P.; Halici, H. In Vivo and In Vitro Cardioprotective Effect of Gossypin Against Isoproterenol-Induced Myocardial Infarction Injury. Cardiovascular toxicology 2022, 22, 52-62. [CrossRef]
- Tan, M.; Yin, Y.; Ma, X.; Zhang, J.; Pan, W.; Tan, M.; Zhao, Y.; Yang, T.; Jiang, T.; Li, H. Glutathione system enhancement for cardiac protection: pharmacological options against oxidative stress and ferroptosis. Cell death & disease 2023, 14, 131. [CrossRef]
- Piekarska, J.; Szczypka, M.; Obmińska-Mrukowicz, B.; Gorczykowski, M. Effect of phytohaemagglutinin-P on apoptosis and necrosis in Trichinella spiralis infected mice. Veterinary parasitology 2009, 159, 240-244. [CrossRef]
- Jun, H.O.; Kim, D.H.; Lee, S.W.; Lee, H.S.; Seo, J.H.; Kim, J.H.; Kim, J.H.; Yu, Y.S.; Min, B.H.; Kim, K.W. Clusterin protects H9c2 cardiomyocytes from oxidative stress-induced apoptosis via Akt/GSK-3β signaling pathway. Experimental & molecular medicine 2011, 43, 53-61. [CrossRef]
- Wan, C.R.; Han, D.D.; Xu, J.Q.; Yin, P.; Xu, X.L.; Mei, C.; Liu, F.H.; Xia, Z.F. Jujuboside A attenuates norepinephrine-induced apoptosis of H9c2 cardiomyocytes by modulating MAPK and AKT signaling pathways. Molecular medicine reports 2018, 17, 1132-1140. [CrossRef]
- Chang, H.; Li, C.; Huo, K.; Wang, Q.; Lu, L.; Zhang, Q.; Wang, Y.; Wang, W. Luteolin Prevents H2O2-Induced Apoptosis in H9C2 Cells through Modulating Akt-P53/Mdm2 Signaling Pathway. BioMed research international 2016, 2016, 5125836. [CrossRef]
- Li, H.; Niu, N.; Yang, J.; Dong, F.; Zhang, T.; Li, S.; Zhao, W. Nuclear respiratory factor 1 protects H9C2 cells against hypoxia-induced apoptosis via the death receptor pathway and mitochondrial pathway. Cell biology international 2021, 45, 1784-1796. [CrossRef]
- de Lima-Seolin, B.G.; Nemec-Bakk, A.; Forsyth, H.; Kirk, S.; da Rosa Araujo, A.S.; Schenkel, P.C.; Belló-Klein, A.; Khaper, N. Bucindolol Modulates Cardiac Remodeling by Attenuating Oxidative Stress in H9c2 Cardiac Cells Exposed to Norepinephrine. Oxidative medicine and cellular longevity 2019, 2019, 6325424. [CrossRef]
- Li, M.; Ye, J.; Zhao, G.; Hong, G.; Hu, X.; Cao, K.; Wu, Y.; Lu, Z. Gas6 attenuates lipopolysaccharide-induced TNF-α expression and apoptosis in H9C2 cells through NF-κB and MAPK inhibition via the Axl/PI3K/Akt pathway. International journal of molecular medicine 2019, 44, 982-994. [CrossRef]
- Li, F.; Liu, J.; Tang, S.; Yan, J.; Chen, H.; Li, D.; Yan, X. Quercetin regulates inflammation, oxidative stress, apoptosis, and mitochondrial structure and function in H9C2 cells by promoting PVT1 expression. Acta histochemica 2021, 123, 151819. [CrossRef]
- Luo, Q.; Yang, A.; Cao, Q.; Guan, H. 3,3'-Diindolylmethane protects cardiomyocytes from LPS-induced inflammatory response and apoptosis. BMC pharmacology & toxicology 2018, 19, 71. [CrossRef]
- Zhang, Y.; Zheng, L.M.; Wang, C.X.; Gu, J.M.; Xue, S. SENP3 protects H9C2 cells from apoptosis triggered by H/R via STAT3 pathway. European review for medical and pharmacological sciences 2018, 22, 2778-2786. [CrossRef]
- Huang, G.; Huang, X.; Liu, M.; Hua, Y.; Deng, B.; Jin, W.; Yan, W.; Tan, Z.; Wu, Y.; Liu, B.; et al. Secoisolariciresinol diglucoside prevents the oxidative stress-induced apoptosis of myocardial cells through activation of the JAK2/STAT3 signaling pathway. International journal of molecular medicine 2018, 41, 3570-3576. [CrossRef]
- Han, X.; Qi, J.; Yang, Y.; Zheng, B.; Liu, M.; Liu, Y.; Li, L.; Guan, S.; Jia, Q.; Chu, L. Protective mechanisms of 10-gingerol against myocardial ischemia may involve activation of JAK2/STAT3 pathway and regulation of Ca(2+) homeostasis. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie 2022, 151, 113082. [CrossRef]
- Zhao, L.; Cheng, L.; Wu, Y. Ambra1 Alleviates Hypoxia/Reoxygenation Injury in H9C2 Cells by Regulating Autophagy and Reactive Oxygen Species. BioMed research international 2020, 2020, 3062689. [CrossRef]
- Zhang, Q.; Fu, H.; Gong, W.; Cao, F.; Wu, T.; Hu, F. Plumbagin protects H9c2 cardiomyocytes against TBHP-induced cytotoxicity by alleviating ROS-induced apoptosis and modulating autophagy. Experimental and therapeutic medicine 2022, 24, 501. [CrossRef]
- Ma, L.Q.; Yu, Y.; Chen, H.; Li, M.; Ihsan, A.; Tong, H.Y.; Huang, X.J.; Gao, Y. Sweroside Alleviated Aconitine-Induced Cardiac Toxicity in H9c2 Cardiomyoblast Cell Line. Frontiers in pharmacology 2018, 9, 1138. [CrossRef]
- Zheng, B.; Qi, J.; Yang, Y.; Li, L.; Liu, Y.; Han, X.; Qu, W.; Chu, L. Mechanisms of cinnamic aldehyde against myocardial ischemia/hypoxia injury in vivo and in vitro: Involvement of regulating PI3K/AKT signaling pathway. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie 2022, 147, 112674. [CrossRef]
- Mao, S.; Luo, X.; Li, Y.; He, C.; Huang, F.; Su, C. Role of PI3K/AKT/mTOR Pathway Associated Oxidative Stress and Cardiac Dysfunction in Takotsubo Syndrome. Current neurovascular research 2020, 17, 35-43. [CrossRef]
- Othman, E.M.; Naseem, M.; Awad, E.; Dandekar, T.; Stopper, H. The Plant Hormone Cytokinin Confers Protection against Oxidative Stress in Mammalian Cells. PLoS One 2016, 11, e0168386. [CrossRef]
- Divakaruni, A.S.; Paradyse, A.; Ferrick, D.A.; Murphy, A.N.; Jastroch, M. Chapter Sixteen - Analysis and Interpretation of Microplate-Based Oxygen Consumption and pH Data. In Methods in enzymology, Murphy, A.N., Chan, D.C., Eds. Academic Press: 2014; Vol. 547, pp. 309-354.
- Muralimanoharan, S.; Maloyan, A.; Mele, J.; Guo, C.; Myatt, L.G.; Myatt, L. MIR-210 modulates mitochondrial respiration in placenta with preeclampsia. Placenta 2012, 33, 816-823. [CrossRef]
- Wang, M.; Wang, R.; Xie, X.; Sun, G.; Sun, X. Araloside C protects H9c2 cardiomyoblasts against oxidative stress via the modulation of mitochondrial function. Biomedicine & Pharmacotherapy 2019, 117, 109143. [CrossRef]
- Yu, J.; Li, Y.; Liu, X.; Ma, Z.; Michael, S.; Orgah, J.O.; Fan, G.; Zhu, Y. Mitochondrial dynamics modulation as a critical contribution for Shenmai injection in attenuating hypoxia/reoxygenation injury. Journal of Ethnopharmacology 2019, 237, 9-19. [CrossRef]
- Huynh, G.T.; Kesarwani, V.; Walker, J.A.; Frith, J.E.; Meagher, L.; Corrie, S.R. Review: Nanomaterials for Reactive Oxygen Species Detection and Monitoring in Biological Environments. Frontiers in Chemistry 2021, 9.
- Koren, K.; Borisov, S.M.; Klimant, I. Stable optical oxygen sensing materials based on click-coupling of fluorinated platinum(II) and palladium(II) porphyrins—A convenient way to eliminate dye migration and leaching. Sensors and Actuators B: Chemical 2012, 169, 173-181. [CrossRef]
- Lee, Y.E.; Smith, R.; Kopelman, R. Nanoparticle PEBBLE sensors in live cells and in vivo. Annual review of analytical chemistry (Palo Alto, Calif.) 2009, 2, 57-76. [CrossRef]
- Lee, Y.-E.K.; Kopelman, R. Optical nanoparticle sensors for quantitative intracellular imaging. WIREs Nanomedicine and Nanobiotechnology 2009, 1, 98-110. [CrossRef]
- Barone, P.W.; Parker, R.S.; Strano, M.S. In Vivo Fluorescence Detection of Glucose Using a Single-Walled Carbon Nanotube Optical Sensor: Design, Fluorophore Properties, Advantages, and Disadvantages. Analytical Chemistry 2005, 77, 7556-7562. [CrossRef]
- Wang, X.; Li, F.; Guo, Y. Recent Trends in Nanomaterial-Based Biosensors for Point-of-Care Testing. Frontiers in Chemistry 2020, 8.
- Lee, C.H.; Folz, J.; Tan, J.W.Y.; Jo, J.; Wang, X.; Kopelman, R. Chemical Imaging in Vivo: Photoacoustic-Based 4-Dimensional Chemical Analysis. Analytical Chemistry 2019, 91, 2561-2569. [CrossRef]
- Kim, C.; Erpelding, T.N.; Jankovic, L.; Pashley, M.D.; Wang, L.V. Deeply penetrating in vivo photoacoustic imaging using a clinical ultrasound array system. Biomedical optics express 2010, 1, 278-284. [CrossRef]
- Hariri, A.; Zhao, E.; Jeevarathinam, A.S.; Lemaster, J.; Zhang, J.; Jokerst, J.V. Molecular imaging of oxidative stress using an LED-based photoacoustic imaging system. Scientific reports 2019, 9, 11378. [CrossRef]
- Ahn, J.; Baik, J.W.; Kim, D.; Choi, K.; Lee, S.; Park, S.M.; Kim, J.Y.; Nam, S.H.; Kim, C. In vivo photoacoustic monitoring of vasoconstriction induced by acute hyperglycemia. Photoacoustics 2023, 30, 100485. [CrossRef]
- Jung, E.; Kang, C.; Lee, J.; Yoo, D.; Hwang, D.W.; Kim, D.; Park, S.-C.; Lim, S.K.; Song, C.; Lee, D. Molecularly Engineered Theranostic Nanoparticles for Thrombosed Vessels: H2O2-Activatable Contrast-Enhanced Photoacoustic Imaging and Antithrombotic Therapy. ACS Nano 2018, 12, 392-401. [CrossRef]
- Chen, R.; Zhang, L.; Gao, J.; Wu, W.; Hu, Y.; Jiang, X. Chemiluminescent nanomicelles for imaging hydrogen peroxide and self-therapy in photodynamic therapy. Journal of biomedicine & biotechnology 2011, 2011, 679492. [CrossRef]
- Lim, C.-K.; Lee, Y.-D.; Na, J.; Oh, J.M.; Her, S.; Kim, K.; Choi, K.; Kim, S.; Kwon, I.C. Chemiluminescence-Generating Nanoreactor Formulation for Near-Infrared Imaging of Hydrogen Peroxide and Glucose Level in vivo. Advanced Functional Materials 2010, 20, 2644-2648. [CrossRef]
- Dasari, M.; Lee, D.; Erigala, V.R.; Murthy, N. Chemiluminescent PEG-PCL micelles for imaging hydrogen peroxide. Journal of Biomedical Materials Research Part A 2009, 89A, 561-566. [CrossRef]
- Lee, D.; Erigala, V.R.; Dasari, M.; Yu, J.; Dickson, R.M.; Murthy, N. Detection of hydrogen peroxide with chemiluminescent micelles. International journal of nanomedicine 2008, 3, 471-476.
- Lee, D.; Khaja, S.; Velasquez-Castano, J.C.; Dasari, M.; Sun, C.; Petros, J.; Taylor, W.R.; Murthy, N. In vivo imaging of hydrogen peroxide with chemiluminescent nanoparticles. Nature Materials 2007, 6, 765-769. [CrossRef]
- Wen, F.; Dong, Y.; Feng, L.; Wang, S.; Zhang, S.; Zhang, X. Horseradish Peroxidase Functionalized Fluorescent Gold Nanoclusters for Hydrogen Peroxide Sensing. Analytical Chemistry 2011, 83, 1193-1196. [CrossRef]
- Shiang, Y.-C.; Huang, C.-C.; Chang, H.-T. Gold nanodot-based luminescent sensor for the detection of hydrogen peroxide and glucose. Chemical Communications 2009, 10.1039/B901916B, 3437-3439. [CrossRef]
- Li, D.-W.; Qin, L.-X.; Li, Y.; Nia, R.P.; Long, Y.-T.; Chen, H.-Y. CdSe/ZnS quantum dot–Cytochrome c bioconjugates for selective intracellular O2˙− sensing. Chemical Communications 2011, 47, 8539-8541. [CrossRef]
- Wang, S.; Han, M.-Y.; Huang, D. Nitric Oxide Switches on the Photoluminescence of Molecularly Engineered Quantum Dots. Journal of the American Chemical Society 2009, 131, 11692-11694. [CrossRef]
- Casanova, D.; Bouzigues, C.; Nguyên, T.-L.; Ramodiharilafy, R.O.; Bouzhir-Sima, L.; Gacoin, T.; Boilot, J.-P.; Tharaux, P.-L.; Alexandrou, A. Single europium-doped nanoparticles measure temporal pattern of reactive oxygen species production inside cells. Nature Nanotechnology 2009, 4, 581-585. [CrossRef]
- Auchinvole, C.A.R.; Richardson, P.; McGuinnes, C.; Mallikarjun, V.; Donaldson, K.; McNab, H.; Campbell, C.J. Monitoring Intracellular Redox Potential Changes Using SERS Nanosensors. ACS Nano 2012, 6, 888-896. [CrossRef]
- Chaichi, A.; Prasad, A.; Gartia, M.R. Raman Spectroscopy and Microscopy Applications in Cardiovascular Diseases: From Molecules to Organs. Biosensors 2018, 8. [CrossRef]
- Kim, J.-Y.; Choi, W.I.; Kim, Y.H.; Tae, G. Highly selective in-vivo imaging of tumor as an inflammation site by ROS detection using hydrocyanine-conjugated, functional nano-carriers. Journal of Controlled Release 2011, 156, 398-405. [CrossRef]
- Kim, G.; Lee, Y.-E.K.; Xu, H.; Philbert, M.A.; Kopelman, R. Nanoencapsulation Method for High Selectivity Sensing of Hydrogen Peroxide inside Live Cells. Analytical Chemistry 2010, 82, 2165-2169. [CrossRef]
- King, M.; Kopelman, R. Development of a hydroxyl radical ratiometric nanoprobe. Sensors and Actuators B: Chemical 2003, 90, 76-81. [CrossRef]
- Cao, Y.; Koo, Y.-E.L.; Koo, S.M.; Kopelman, R. Ratiometric Singlet Oxygen Nano-optodes and Their Use for Monitoring Photodynamic Therapy Nanoplatforms. Photochemistry and Photobiology 2005, 81, 1489-1498. [CrossRef]
- Hammond, V.J.; Aylott, J.W.; Greenway, G.M.; Watts, P.; Webster, A.; Wiles, C. An optical sensor for reactive oxygen species: encapsulation of functionalised silica nanoparticles into silicate nanoprobes to reduce fluorophore leaching. Analyst 2007, 133, 71-75. [CrossRef]
- Tian, J.; Chen, H.; Zhuo, L.; Xie, Y.; Li, N.; Tang, B. A Highly Selective, Cell-Permeable Fluorescent Nanoprobe for Ratiometric Detection and Imaging of Peroxynitrite in Living Cells. Chemistry – A European Journal 2011, 17, 6626-6634. [CrossRef]
- Kim, S.-H.; Kim, B.; Yadavalli, V.K.; Pishko, M.V. Encapsulation of Enzymes within Polymer Spheres To Create Optical Nanosensors for Oxidative Stress. Analytical Chemistry 2005, 77, 6828-6833. [CrossRef]
- Lee, H.; Lee, K.; Kim, I.-K.; Park, T.G. Fluorescent Gold Nanoprobe Sensitive to Intracellular Reactive Oxygen Species. Advanced Functional Materials 2009, 19, 1884-1890. [CrossRef]
- Guo, C.; Hu, F.; Li, C.M.; Shen, P.K. Direct electrochemistry of hemoglobin on carbonized titania nanotubes and its application in a sensitive reagentless hydrogen peroxide biosensor. Biosensors and Bioelectronics 2008, 24, 819-824. [CrossRef]
- Hrapovic, S.; Liu, Y.; Male, K.B.; Luong, J.H.T. Electrochemical Biosensing Platforms Using Platinum Nanoparticles and Carbon Nanotubes. Analytical Chemistry 2004, 76, 1083-1088. [CrossRef]
- Yu, X.; Chattopadhyay, D.; Galeska, I.; Papadimitrakopoulos, F.; Rusling, J.F. Peroxidase activity of enzymes bound to the ends of single-wall carbon nanotube forest electrodes. Electrochemistry Communications 2003, 5, 408-411. [CrossRef]
- Zeng, X.; Li, X.; Liu, X.; Liu, Y.; Luo, S.; Kong, B.; Yang, S.; Wei, W. A third-generation hydrogen peroxide biosensor based on horseradish peroxidase immobilized on DNA functionalized carbon nanotubes. Biosensors and Bioelectronics 2009, 25, 896-900. [CrossRef]
- Wang, J. Carbon-Nanotube Based Electrochemical Biosensors: A Review. Electroanalysis 2005, 17, 7-14. [CrossRef]
- Balasubramanian, K.; Burghard, M. Biosensors based on carbon nanotubes. Analytical and Bioanalytical Chemistry 2006, 385, 452-468. [CrossRef]
- Besteman, K.; Lee, J.-O.; Wiertz, F.G.M.; Heering, H.A.; Dekker, C. Enzyme-Coated Carbon Nanotubes as Single-Molecule Biosensors. Nano Letters 2003, 3, 727-730. [CrossRef]
- Xu, J.-Z.; Zhu, J.-J.; Wu, Q.; Hu, Z.; Chen, H.-Y. An Amperometric Biosensor Based on the Coimmobilization of Horseradish Peroxidase and Methylene Blue on a Carbon Nanotubes Modified Electrode. Electroanalysis 2003, 15, 219-224. [CrossRef]
- Vaneev, A.N.; Gorelkin, P.V.; Garanina, A.S.; Lopatukhina, H.V.; Vodopyanov, S.S.; Alova, A.V.; Ryabaya, O.O.; Akasov, R.A.; Zhang, Y.; Novak, P.; et al. In Vitro and In Vivo Electrochemical Measurement of Reactive Oxygen Species After Treatment with Anticancer Drugs. Analytical Chemistry 2020, 92, 8010-8014. [CrossRef]
- He, R.; Tang, H.; Jiang, D.; Chen, H.-y. Electrochemical Visualization of Intracellular Hydrogen Peroxide at Single Cells. Analytical Chemistry 2016, 88, 2006-2009. [CrossRef]
- Clausmeyer, J.; Schuhmann, W. Nanoelectrodes: Applications in electrocatalysis, single-cell analysis and high-resolution electrochemical imaging. TrAC Trends in Analytical Chemistry 2016, 79, 46-59. [CrossRef]
- Zhang, X.-W.; Oleinick, A.; Jiang, H.; Liao, Q.-L.; Qiu, Q.-F.; Svir, I.; Liu, Y.-L.; Amatore, C.; Huang, W.-H. Electrochemical Monitoring of ROS/RNS Homeostasis Within Individual Phagolysosomes Inside Single Macrophages. Angewandte Chemie International Edition 2019, 58, 7753-7756. [CrossRef]
- Li, Y.; Hu, K.; Yu, Y.; Rotenberg, S.A.; Amatore, C.; Mirkin, M.V. Direct Electrochemical Measurements of Reactive Oxygen and Nitrogen Species in Nontransformed and Metastatic Human Breast Cells. Journal of the American Chemical Society 2017, 139, 13055-13062. [CrossRef]
- Wang, Y.; Noël, J.-M.; Velmurugan, J.; Nogala, W.; Mirkin, M.V.; Lu, C.; Guille Collignon, M.; Lemaître, F.; Amatore, C. Nanoelectrodes for determination of reactive oxygen and nitrogen species inside murine macrophages. Proceedings of the National Academy of Sciences 2012, 109, 11534-11539. [CrossRef]
- Jiang, H.; Zhang, X.-W.; Liao, Q.-L.; Wu, W.-T.; Liu, Y.-L.; Huang, W.-H. Electrochemical Monitoring of Paclitaxel-Induced ROS Release from Mitochondria inside Single Cells. Small 2019, 15, 1901787. [CrossRef]
- Actis, P.; Tokar, S.; Clausmeyer, J.; Babakinejad, B.; Mikhaleva, S.; Cornut, R.; Takahashi, Y.; López Córdoba, A.; Novak, P.; Shevchuck, A.I.; et al. Electrochemical Nanoprobes for Single-Cell Analysis. ACS Nano 2014, 8, 875-884. [CrossRef]
- Erofeev, A.; Gorelkin, P.; Garanina, A.; Alova, A.; Efremova, M.; Vorobyeva, N.; Edwards, C.; Korchev, Y.; Majouga, A. Novel method for rapid toxicity screening of magnetic nanoparticles. Scientific reports 2018, 8, 7462. [CrossRef]
- Akasov, R.A.; Sholina, N.V.; Khochenkov, D.A.; Alova, A.V.; Gorelkin, P.V.; Erofeev, A.S.; Generalova, A.N.; Khaydukov, E.V. Photodynamic therapy of melanoma by blue-light photoactivation of flavin mononucleotide. Scientific reports 2019, 9, 9679. [CrossRef]
- Dikalov, S.I.; Dikalova, A.E.; Morozov, D.A.; Kirilyuk, I.A. Cellular accumulation and antioxidant activity of acetoxymethoxycarbonyl pyrrolidine nitroxides. Free radical research 2018, 52, 339-350. [CrossRef]
- Dikalov, S.I.; Harrison, D.G. Methods for detection of mitochondrial and cellular reactive oxygen species. Antioxidants & redox signaling 2014, 20, 372-382. [CrossRef]
- Hawkins, C.L.; Davies, M.J. Detection and characterisation of radicals in biological materials using EPR methodology. Biochimica et Biophysica Acta (BBA) - General Subjects 2014, 1840, 708-721. [CrossRef]
- Ouari, O.; Hardy, M.; Karoui, H.; Tordo, P. Recent developments and applications of the coupled EPR/Spin trapping technique (EPR/ST). In Electron Paramagnetic Resonance: Volume 22, The Royal Society of Chemistry: 2011; Vol. 22, pp. 1-40.
- Dikalov, S.I.; Polienko, Y.F.; Kirilyuk, I. Electron Paramagnetic Resonance Measurements of Reactive Oxygen Species by Cyclic Hydroxylamine Spin Probes. Antioxidants & redox signaling 2018, 28, 1433-1443. [CrossRef]
- Dikalov, S.; Griendling, K.K.; Harrison, D.G. Measurement of Reactive Oxygen Species in Cardiovascular Studies. Hypertension 2007, 49, 717-727. [CrossRef]
- Dikalov, S.I.; Kirilyuk, I.A.; Voinov, M.; Grigor'ev, I.A. EPR detection of cellular and mitochondrial superoxide using cyclic hydroxylamines. Free radical research 2011, 45, 417-430. [CrossRef]
- Kozuleva, M.; Klenina, I.; Mysin, I.; Kirilyuk, I.; Opanasenko, V.; Proskuryakov, I.; Ivanov, B. Quantification of superoxide radical production in thylakoid membrane using cyclic hydroxylamines. Free Radical Biology and Medicine 2015, 89, 1014-1023. [CrossRef]
- Israeli, A.; Patt, M.; Oron, M.; Samuni, A.; Kohen, R.; Goldstein, S. Kinetics and mechanism of the comproportionation reaction between oxoammonium cation and hydroxylamine derived from cyclic nitroxides. Free Radical Biology and Medicine 2005, 38, 317-324. [CrossRef]
- Zhou, M.; Diwu, Z.; Panchuk-Voloshina, N.; Haugland, R.P. A Stable Nonfluorescent Derivative of Resorufin for the Fluorometric Determination of Trace Hydrogen Peroxide: Applications in Detecting the Activity of Phagocyte NADPH Oxidase and Other Oxidases. Analytical Biochemistry 1997, 253, 162-168. [CrossRef]
- Weber, D.S.; Rocic, P.; Mellis, A.M.; Laude, K.; Lyle, A.N.; Harrison, D.G.; Griendling, K.K. Angiotensin II-induced hypertrophy is potentiated in mice overexpressing p22phox in vascular smooth muscle. American Journal of Physiology-Heart and Circulatory Physiology 2005, 288, H37-H42. [CrossRef]
- Belousov, V.V.; Fradkov, A.F.; Lukyanov, K.A.; Staroverov, D.B.; Shakhbazov, K.S.; Terskikh, A.V.; Lukyanov, S. Genetically encoded fluorescent indicator for intracellular hydrogen peroxide. Nature Methods 2006, 3, 281-286. [CrossRef]
- Gutscher, M.; Sobotta, M.C.; Wabnitz, G.H.; Ballikaya, S.; Meyer, A.J.; Samstag, Y.; Dick, T.P. Proximity-based Protein Thiol Oxidation by H2O2-scavenging Peroxidases*♦. Journal of Biological Chemistry 2009, 284, 31532-31540. [CrossRef]
- Ermakova, Y.G.; Bilan, D.S.; Matlashov, M.E.; Mishina, N.M.; Markvicheva, K.N.; Subach, O.M.; Subach, F.V.; Bogeski, I.; Hoth, M.; Enikolopov, G.; et al. Red fluorescent genetically encoded indicator for intracellular hydrogen peroxide. Nature Communications 2014, 5, 5222. [CrossRef]
- Gibhardt, C.S.; Zimmermann, K.M.; Zhang, X.; Belousov, V.V.; Bogeski, I. Imaging calcium and redox signals using genetically encoded fluorescent indicators. Cell Calcium 2016, 60, 55-64. [CrossRef]
- Hernández-Barrera, A.; Quinto, C.; Johnson, E.A.; Wu, H.-M.; Cheung, A.Y.; Cárdenas, L. Chapter Fifteen - Using Hyper as a Molecular Probe to Visualize Hydrogen Peroxide in Living Plant Cells: A Method with Virtually Unlimited Potential in Plant Biology. In Methods in enzymology, Cadenas, E., Packer, L., Eds. Academic Press: 2013; Vol. 527, pp. 275-290.
- Zhuravlev, A.; Ezeriņa, D.; Ivanova, J.; Guriev, N.; Pugovkina, N.; Shatrova, A.; Aksenov, N.; Messens, J.; Lyublinskaya, O. HyPer as a tool to determine the reductive activity in cellular compartments. Redox biology 2024, 70, 103058. [CrossRef]
- Hernández-Barrera, A.; Quinto, C.; Johnson, E.A.; Wu, H.M.; Cheung, A.Y.; Cárdenas, L. Using hyper as a molecular probe to visualize hydrogen peroxide in living plant cells: a method with virtually unlimited potential in plant biology. Methods in enzymology 2013, 527, 275-290. [CrossRef]
- Nietzel, T.; Elsässer, M.; Ruberti, C.; Steinbeck, J.; Ugalde, J.M.; Fuchs, P.; Wagner, S.; Ostermann, L.; Moseler, A.; Lemke, P.; et al. The fluorescent protein sensor roGFP2-Orp1 monitors in vivo H(2) O(2) and thiol redox integration and elucidates intracellular H(2) O(2) dynamics during elicitor-induced oxidative burst in Arabidopsis. The New phytologist 2019, 221, 1649-1664. [CrossRef]
- Arnaud, D.; Deeks, M.J.; Smirnoff, N. Organelle-targeted biosensors reveal distinct oxidative events during pattern-triggered immune responses. Plant physiology 2023, 191, 2551-2569. [CrossRef]
- Gutscher, M.; Sobotta, M.C.; Wabnitz, G.H.; Ballikaya, S.; Meyer, A.J.; Samstag, Y.; Dick, T.P. Proximity-based protein thiol oxidation by H2O2-scavenging peroxidases. The Journal of biological chemistry 2009, 284, 31532-31540. [CrossRef]
- Rampon, C.; Volovitch, M.; Joliot, A.; Vriz, S. Hydrogen Peroxide and Redox Regulation of Developments. Antioxidants (Basel) 2018, 7. [CrossRef]
- Neely, J.R.; Rovetto, M.J.; Whitmer, J.T.; Morgan, H.E. Effects of ischemia on function and metabolism of the isolated working rat heart. The American journal of physiology 1973, 225, 651-658. [CrossRef]
- Vidavalur, R.; Swarnakar, S.; Thirunavukkarasu, M.; Samuel, S.M.; Maulik, N. Ex vivo and in vivo approaches to study mechanisms of cardioprotection targeting ischemia/reperfusion (i/r) injury: useful techniques for cardiovascular drug discovery. Current drug discovery technologies 2008, 5, 269-278. [CrossRef]
- Halapas, A.; Papalois, A.; Stauropoulou, A.; Philippou, A.; Pissimissis, N.; Chatzigeorgiou, A.; Kamper, E.; Koutsilieris, M. In vivo models for heart failure research. In vivo (Athens, Greece) 2008, 22, 767-780.
- Liang, J.; Wu, M.; Chen, C.; Mai, M.; Huang, J.; Zhu, P. Roles of Reactive Oxygen Species in Cardiac Differentiation, Reprogramming, and Regenerative Therapies. Oxidative medicine and cellular longevity 2020, 2020, 2102841. [CrossRef]
- Sajid, A.; Ahmad, T.; Ikram, M.; Khan, T.; Shah, A.J.; Mahnashi, M.H.; Alhasaniah, A.H.; Al Awadh, A.A.; Almazni, I.A.; Alshahrani, M.M. Cardioprotective Potential of Aqueous Extract of Fumaria indica on Isoproterenol-Induced Myocardial Infarction in SD Rats. Oxidative medicine and cellular longevity 2022, 2022, 2112956. [CrossRef]
- Tiwari, R.; Mohan, M.; Kasture, S.; Maxia, A.; Ballero, M. Cardioprotective potential of myricetin in isoproterenol-induced myocardial infarction in Wistar rats. Phytotherapy research : PTR 2009, 23, 1361-1366. [CrossRef]
- Octavia, Y.; Tocchetti, C.G.; Gabrielson, K.L.; Janssens, S.; Crijns, H.J.; Moens, A.L. Doxorubicin-induced cardiomyopathy: from molecular mechanisms to therapeutic strategies. Journal of molecular and cellular cardiology 2012, 52, 1213-1225. [CrossRef]
- Vejpongsa, P.; Yeh, E.T. Prevention of anthracycline-induced cardiotoxicity: challenges and opportunities. Journal of the American College of Cardiology 2014, 64, 938-945. [CrossRef]
- Weber, L.W.; Boll, M.; Stampfl, A. Hepatotoxicity and mechanism of action of haloalkanes: carbon tetrachloride as a toxicological model. Critical reviews in toxicology 2003, 33, 105-136. [CrossRef]
- Rechnagel, R.O.; Glende, E.A., Jr. Carbon tetrachloride hepatotoxicity: an example of lethal cleavage. CRC critical reviews in toxicology 1973, 2, 263-297. [CrossRef]
- Chirino, Y.I.; Pedraza-Chaverri, J. Role of oxidative and nitrosative stress in cisplatin-induced nephrotoxicity. Experimental and toxicologic pathology : official journal of the Gesellschaft fur Toxikologische Pathologie 2009, 61, 223-242. [CrossRef]
- Yao, X.; Panichpisal, K.; Kurtzman, N.; Nugent, K. Cisplatin nephrotoxicity: a review. The American journal of the medical sciences 2007, 334, 115-124. [CrossRef]
- Rodríguez-Barbero, A.; Bosque, E.; Gonzalez-Buitrago, J.M.; Garcia-Bastos, J.L.; López-Novoa, J.M. Gentamicin nephrotoxicity in rats is not modified by verapamil. Archives internationales de physiologie, de biochimie et de biophysique 1993, 101, 395-397. [CrossRef]
- Balakumar, P.; Rohilla, A.; Thangathirupathi, A. Gentamicin-induced nephrotoxicity: Do we have a promising therapeutic approach to blunt it? Pharmacological research 2010, 62, 179-186. [CrossRef]
- Moeller, A.; Ask, K.; Warburton, D.; Gauldie, J.; Kolb, M. The bleomycin animal model: a useful tool to investigate treatment options for idiopathic pulmonary fibrosis? The international journal of biochemistry & cell biology 2008, 40, 362-382. [CrossRef]
- Chaudhary, N.I.; Schnapp, A.; Park, J.E. Pharmacologic differentiation of inflammation and fibrosis in the rat bleomycin model. American journal of respiratory and critical care medicine 2006, 173, 769-776. [CrossRef]
- Cannon, J.R.; Greenamyre, J.T. The role of environmental exposures in neurodegeneration and neurodegenerative diseases. Toxicological sciences : an official journal of the Society of Toxicology 2011, 124, 225-250. [CrossRef]
- Betarbet, R.; Sherer, T.B.; MacKenzie, G.; Garcia-Osuna, M.; Panov, A.V.; Greenamyre, J.T. Chronic systemic pesticide exposure reproduces features of Parkinson's disease. Nature neuroscience 2000, 3, 1301-1306. [CrossRef]
- Bus, J.S.; Gibson, J.E. Paraquat: model for oxidant-initiated toxicity. Environmental health perspectives 1984, 55, 37-46. [CrossRef]
- Dinis-Oliveira, R.J.; Remião, F.; Carmo, H.; Duarte, J.A.; Navarro, A.S.; Bastos, M.L.; Carvalho, F. Paraquat exposure as an etiological factor of Parkinson's disease. Neurotoxicology 2006, 27, 1110-1122. [CrossRef]
- Comporti, M. Lipid peroxidation and cellular damage in toxic liver injury. Laboratory investigation; a journal of technical methods and pathology 1985, 53, 599-623.
- Yagi, K. Lipid peroxides and human diseases. Chemistry and physics of lipids 1987, 45, 337-351. [CrossRef]
- Kurokawa, Y.; Maekawa, A.; Takahashi, M.; Hayashi, Y. Toxicity and carcinogenicity of potassium bromate--a new renal carcinogen. Environmental health perspectives 1990, 87, 309-335. [CrossRef]
- Doridot, L.; Jeljeli, M.; Chêne, C.; Batteux, F. Implication of oxidative stress in the pathogenesis of systemic sclerosis via inflammation, autoimmunity and fibrosis. Redox biology 2019, 25, 101122. [CrossRef]
- Furukawa, S.; Fujita, T.; Shimabukuro, M.; Iwaki, M.; Yamada, Y.; Nakajima, Y.; Nakayama, O.; Makishima, M.; Matsuda, M.; Shimomura, I. Increased oxidative stress in obesity and its impact on metabolic syndrome. The Journal of clinical investigation 2004, 114, 1752-1761. [CrossRef]
- McGregor, R.A.; Kwon, E.Y.; Shin, S.K.; Jung, U.J.; Kim, E.; Park, J.H.; Yu, R.; Yun, J.W.; Choi, M.S. Time-course microarrays reveal modulation of developmental, lipid metabolism and immune gene networks in intrascapular brown adipose tissue during the development of diet-induced obesity. International journal of obesity (2005) 2013, 37, 1524-1531. [CrossRef]
- Park, B.S.; Lee, J.O. Recognition of lipopolysaccharide pattern by TLR4 complexes. Experimental & molecular medicine 2013, 45, e66. [CrossRef]
- Heinen, A.; Nederlof, R.; Panjwani, P.; Spychala, A.; Tschaidse, T.; Reffelt, H.; Boy, J.; Raupach, A.; Gödecke, S.; Petzsch, P.; et al. IGF1 Treatment Improves Cardiac Remodeling after Infarction by Targeting Myeloid Cells. Molecular therapy : the journal of the American Society of Gene Therapy 2019, 27, 46-58. [CrossRef]
- Kalogeris, T.; Baines, C.P.; Krenz, M.; Korthuis, R.J. Cell biology of ischemia/reperfusion injury. International review of cell and molecular biology 2012, 298, 229-317. [CrossRef]
- Madrigal, J.L.; Olivenza, R.; Moro, M.A.; Lizasoain, I.; Lorenzo, P.; Rodrigo, J.; Leza, J.C. Glutathione depletion, lipid peroxidation and mitochondrial dysfunction are induced by chronic stress in rat brain. Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology 2001, 24, 420-429. [CrossRef]
- Everson, C.A.; Laatsch, C.D.; Hogg, N. Antioxidant defense responses to sleep loss and sleep recovery. American journal of physiology. Regulatory, integrative and comparative physiology 2005, 288, R374-383. [CrossRef]
- Butterfield, D.A.; Boyd-Kimball, D. Redox proteomics and amyloid β-peptide: insights into Alzheimer disease. J Neurochem 2019, 151, 459-487. [CrossRef]
- Takeda, T. Senescence-accelerated mouse (SAM): a biogerontological resource in aging research. Neurobiology of aging 1999, 20, 105-110. [CrossRef]
- Mihara, M.; Uchiyama, M. Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal Biochem 1978, 86, 271-278. [CrossRef]
- Karmen, A.; Wroblewski, F.; Ladue, J.S. Transaminase activity in human blood. The Journal of clinical investigation 1955, 34, 126-131. [CrossRef]
- Rajadurai, M.; Stanely Mainzen Prince, P. Preventive effect of naringin on cardiac markers, electrocardiographic patterns and lysosomal hydrolases in normal and isoproterenol-induced myocardial infarction in Wistar rats. Toxicology 2007, 230, 178-188. [CrossRef]
- Macdonald, R.P.; Simpson, J.R.; Nossal, E. Serum lactic dehydrogenase; a diagnostic aid in myocardial infarction. Journal of the American Medical Association 1957, 165, 35-40. [CrossRef]
- Molh, A.K.; Ting, L.C.; Khan, J.; Al-Jashamy, K.; Jaafar, H.; Islam, M.N. Histopathological studies of cardiac lesions after an acute high dose administration of methamphetamine. The Malaysian journal of medical sciences : MJMS 2008, 15, 23-30.
- Liao, W.; Rao, Z.; Wu, L.; Chen, Y.; Li, C. Cariporide Attenuates Doxorubicin-Induced Cardiotoxicity in Rats by Inhibiting Oxidative Stress, Inflammation and Apoptosis Partly Through Regulation of Akt/GSK-3β and Sirt1 Signaling Pathway. Frontiers in pharmacology 2022, 13, 850053. [CrossRef]
- Othmène, Y.B.; Hamdi, H.; Amara, I.; Abid-Essefi, S. Tebuconazole induced oxidative stress and histopathological alterations in adult rat heart. Pesticide biochemistry and physiology 2020, 170, 104671. [CrossRef]
| In vitro models | Advantages | Disadvantages |
|---|---|---|
| Cardiac single cell |
|
|
| Two-dimensional (2D) cell cultures | ||
| Three- dimensional (3D) cell cultures |
|
|
| Coculture |
|
|
| Microfluidic cell culture |
|
| Nano Sensors Nanomicelles/Nanopolymer/Carbon Nanotubes/ Metallic |
Applications | Advantages | Disadvantages | References |
|---|---|---|---|---|
| Luminescence |
|
|
|
[109,110,111,112,113] |
| Fluorescent-quenching |
|
|
|
[114,115,116,117,118] |
| Surface-enhanced raman spectroscopy (SERS) |
|
|
|
[119,120] |
| ROS-dye encapsulation |
|
|
|
[121,122,123,124,125,126,127] |
| Nano surface energy transfer (NSET) |
|
|
|
[128] |
| Electrochemical |
|
|
|
[129,130,131,132,133,134,135,136] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




