Submitted:
30 March 2026
Posted:
31 March 2026
You are already at the latest version
Abstract
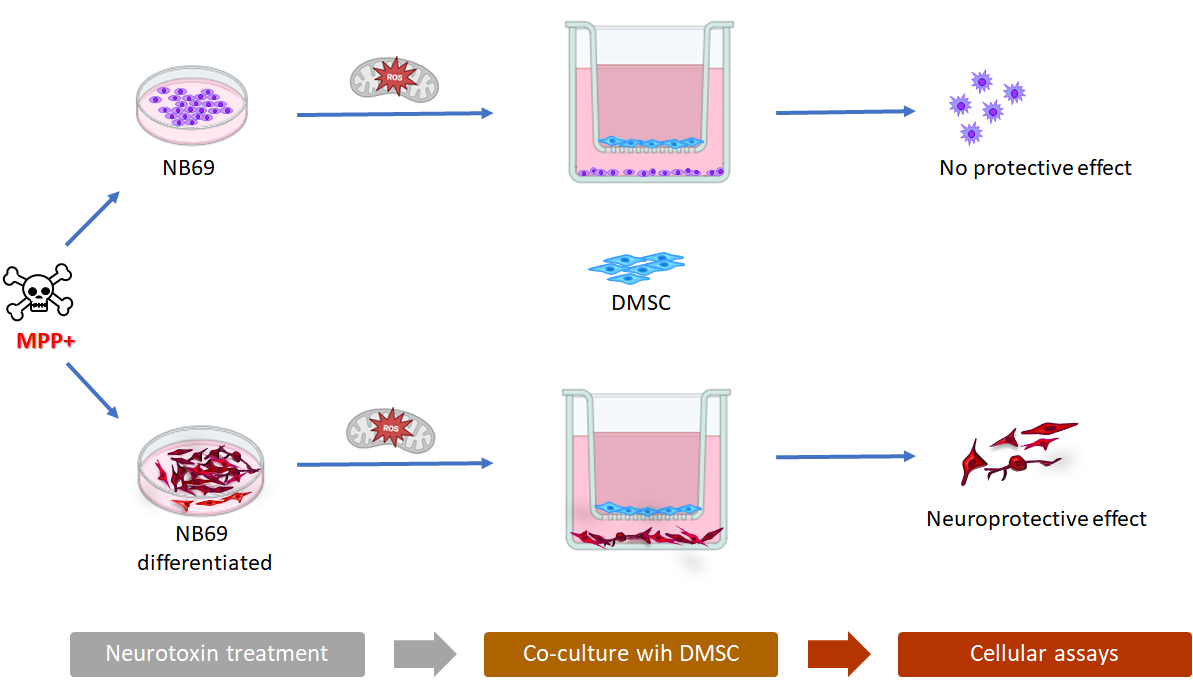
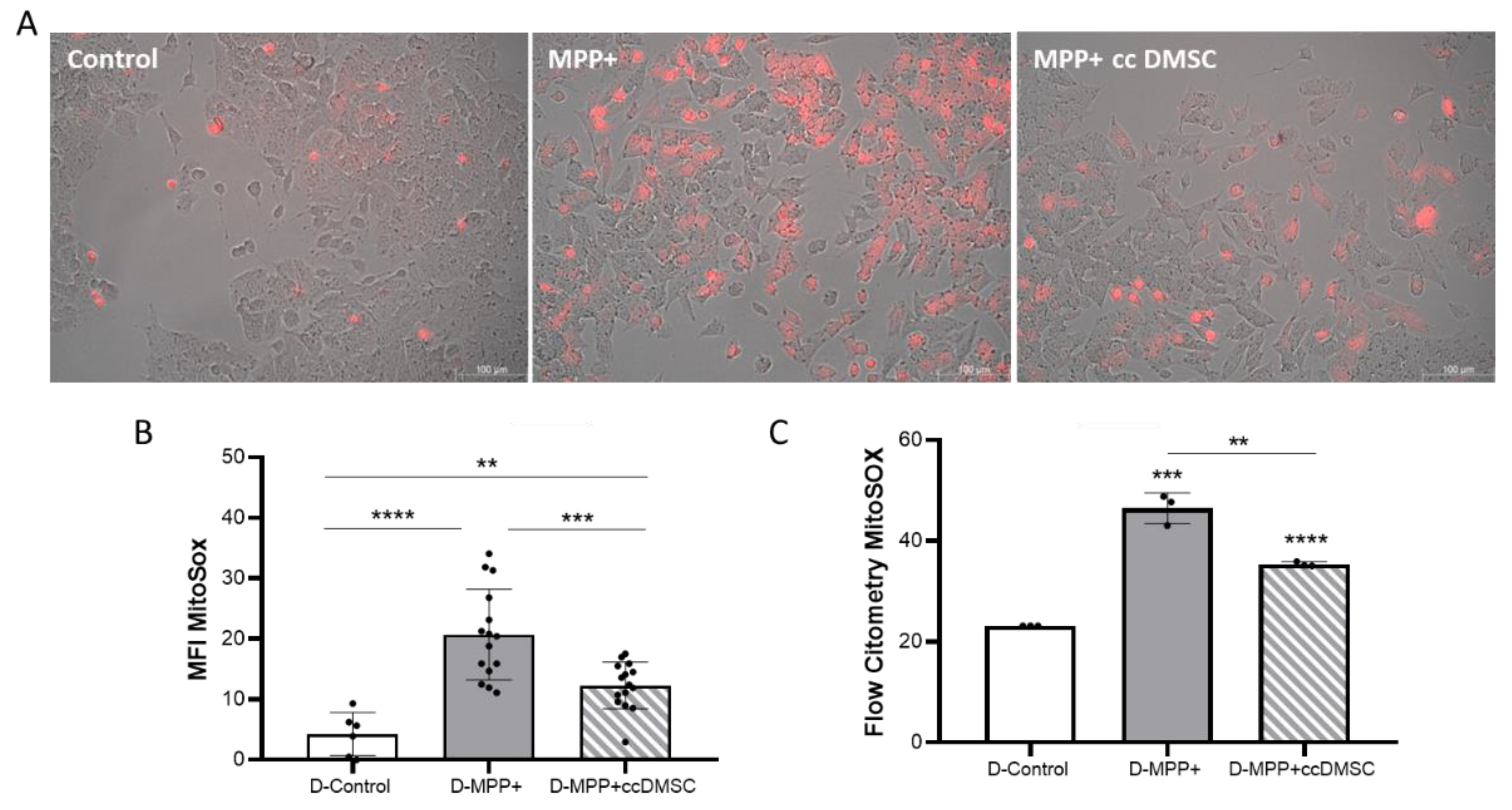
Parkinson’s disease (PD) is the second most prevalent neurodegenerative disorder, characterized by the accumulation of misfolded α-synuclein (α-syn), with the progressive loss of dopaminergic neurons in the substantia nigra. Given the limitations of current therapies, mesenchymal stromal cell (MSC) transplantation has emerged as a promising neuroprotective strategy. This study evaluated the in vitro neuroprotective potential of decidua-derived mesenchymal stem cells (DMSC) using neurotoxin 1-methyl-4-phenylpyridinium (MPP+) neurotoxin-induced damage in a human neuroblastoma cell line (NB69) as a model for PD. NB69 cells were differentiated into a mature dopaminergic phenotype using dibutyryl cyclic adenosine monophosphate (dbcAMP) and subsequently exposed to the MPP+. In proliferative NB69 cells, the effect of DMSC was masked by their inherent anti-tumor activity against the neuroblastoma phenotype. Conversely, in the differentiated NB69 model, DMSC demonstrated a significant protective role against MPP+-induced cytotoxicity. It is interesting that the mechanism by which DMSCs exert a neuroprotective effect against MPP+ damage in differentiated NB69 cells could be through an improvement in mitochondrial function by reducing free radicals. In summary, these findings suggest that DMSC exert a neuroprotective effect in a dopaminergic-like context and highlight the importance of using differentiated cell models to accurately evaluate cell-based therapies for PD in the striatum.
Keywords:
1. Introduction
2. Results
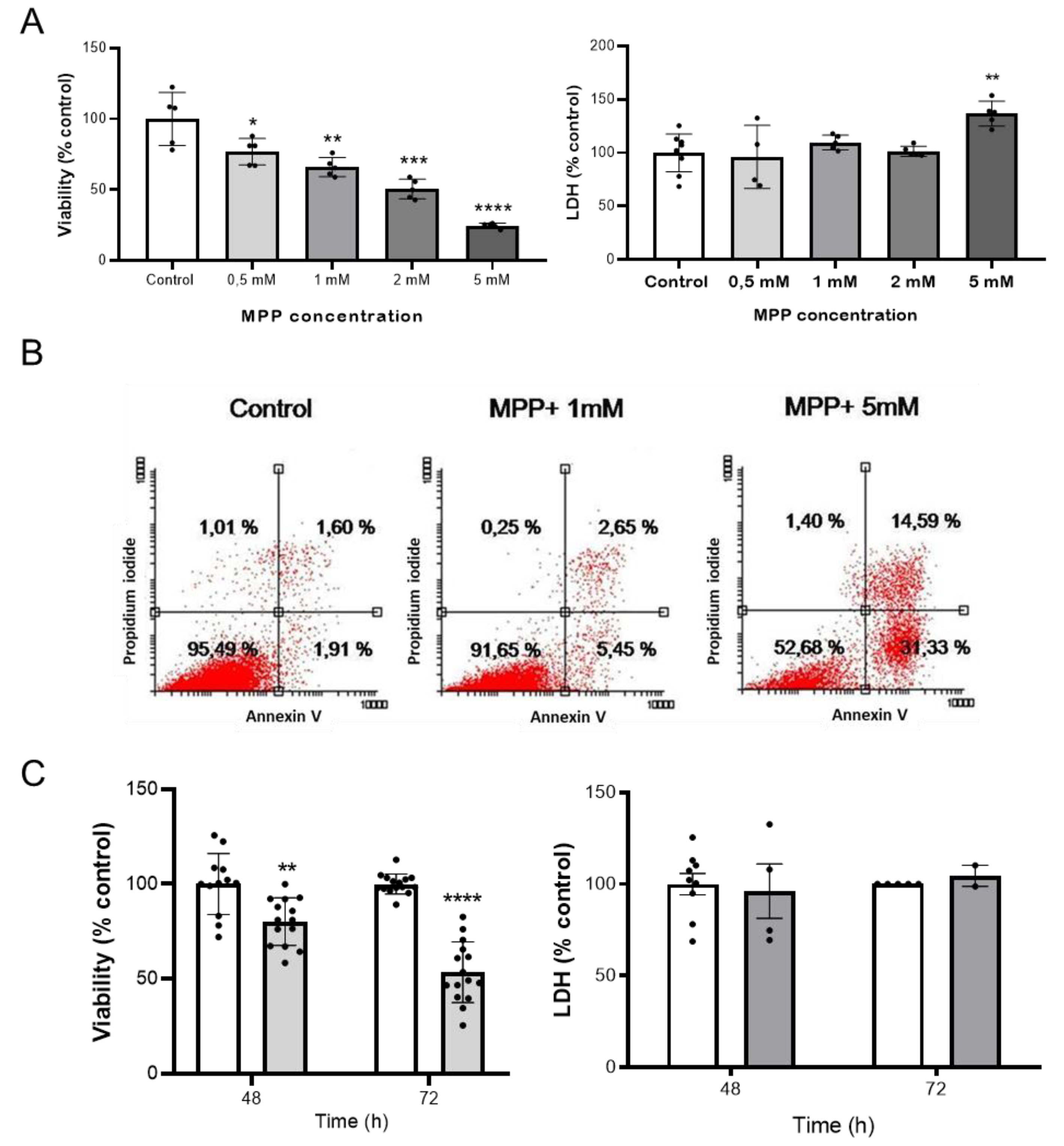
2.1. MPP+ Induces Cell Death by Apoptosis in NB69 Neuroblastoma Cells
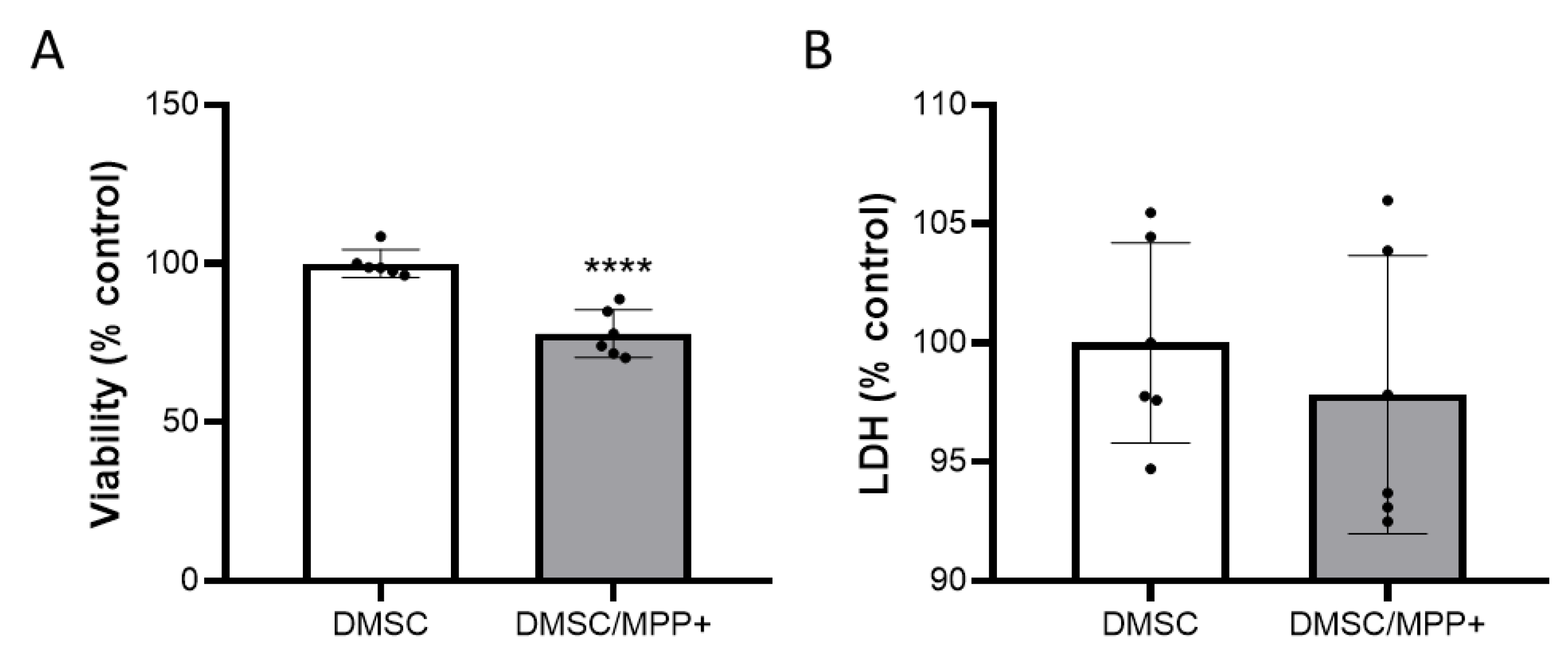
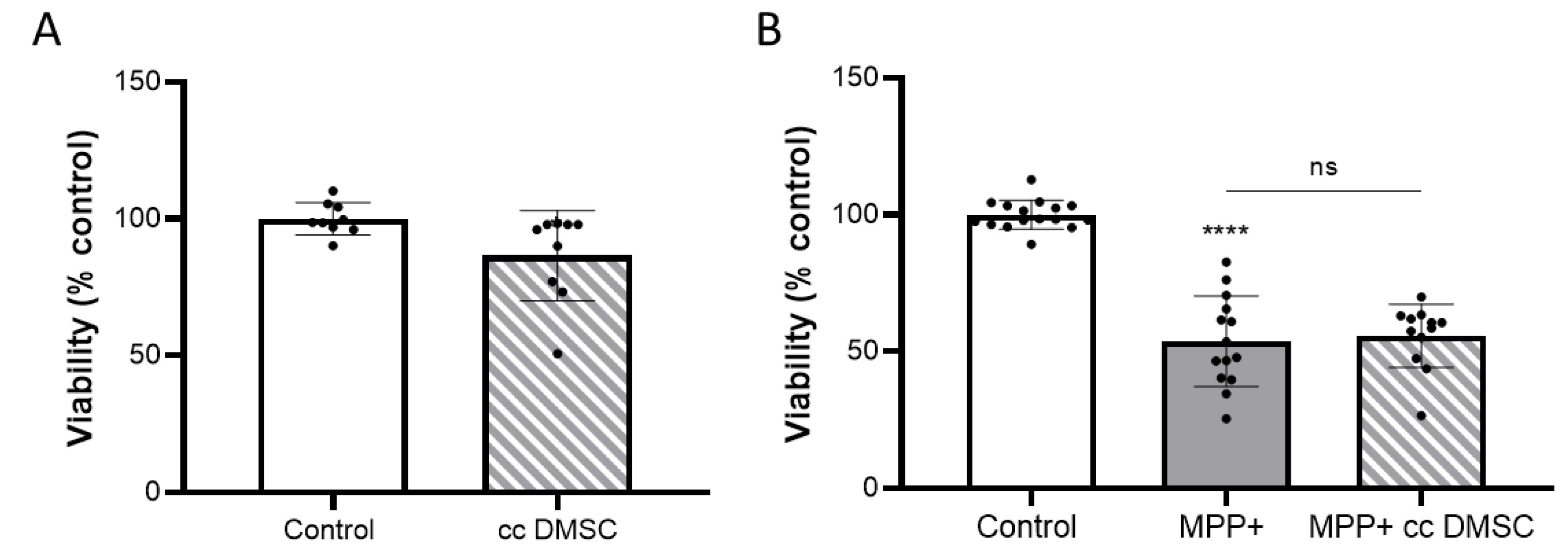
2.2. Analysis of the Possible Anti-Tumor Effect of DMSCs on NB69 Neuroblastoma Cells
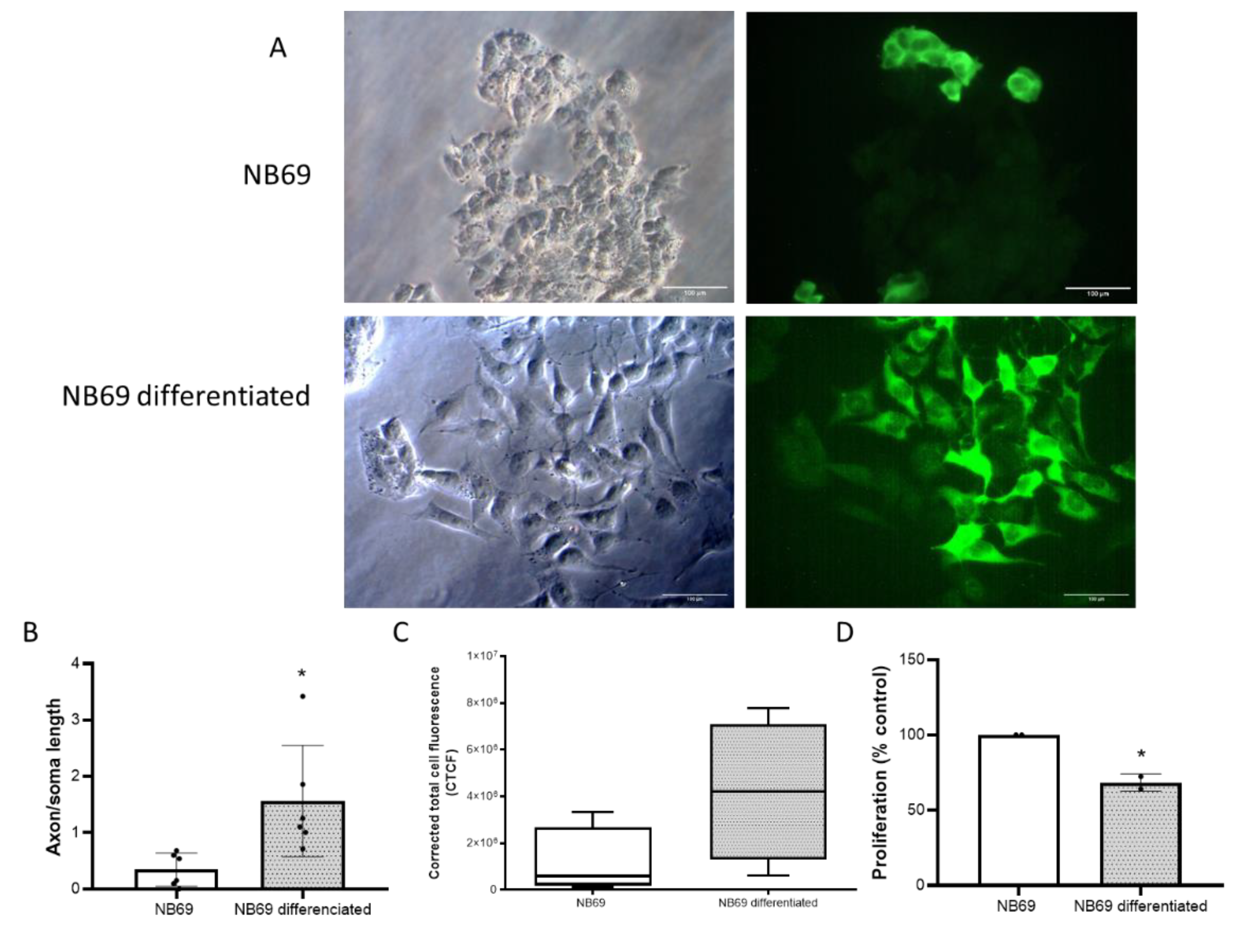
2.3. Differentiation of the Human Neuroblastoma Cell Line NB69 into Dopaminergic Neurons by dbcAMP Treatment
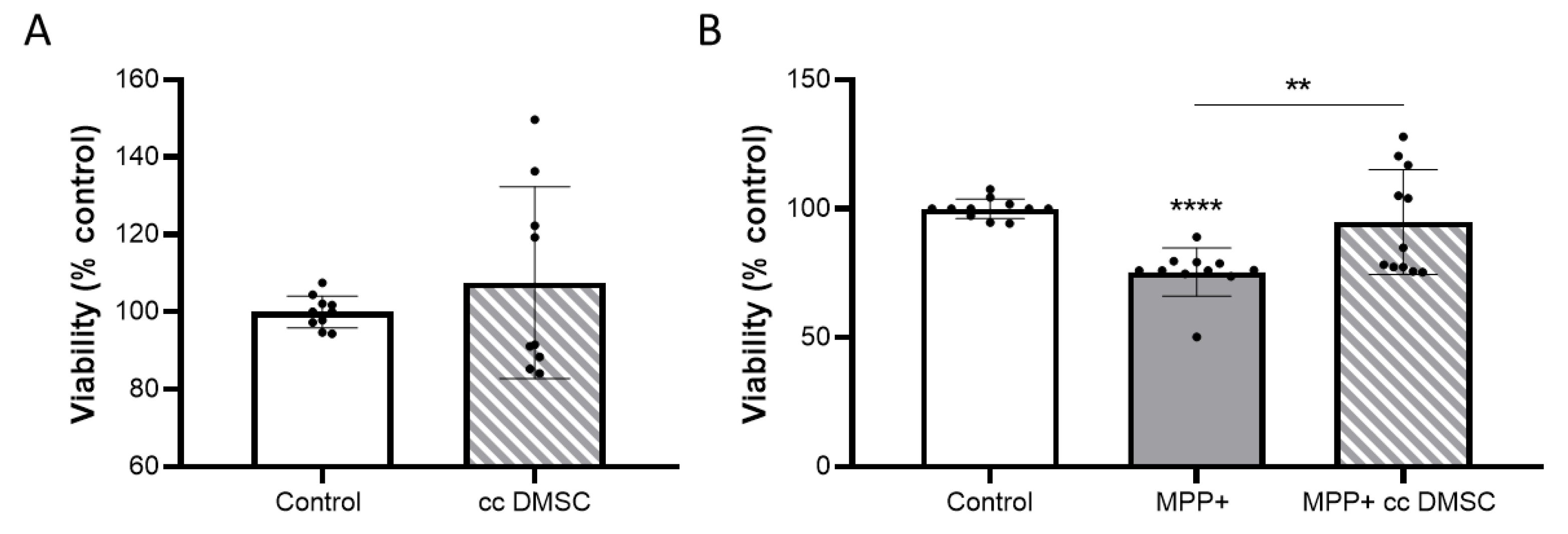
2.4. Effect of Neurotoxin MPP+ on Differentiated NB69 in the Presence or Absence of DMSC
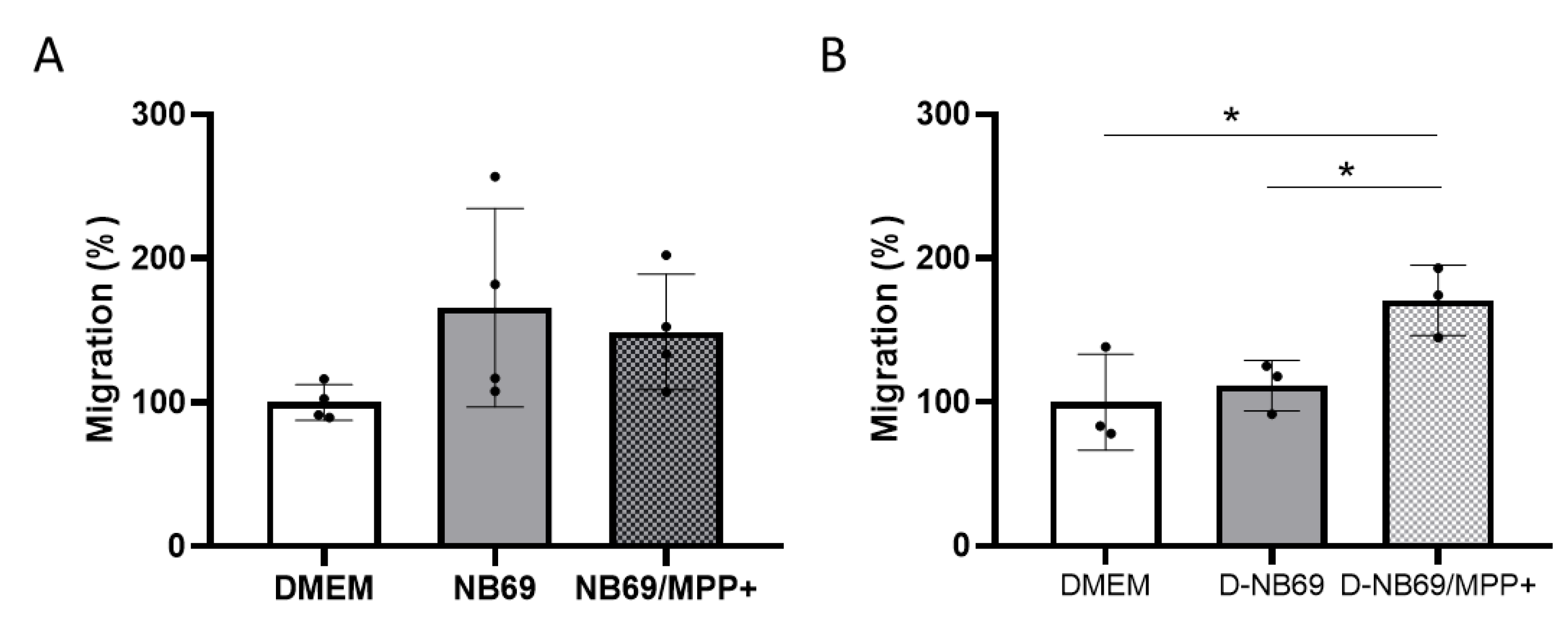
2.5. Study of the In Vitro Migratory Capacity of DMSC in the Presence of NB69 Cells and Differentiated NB69 Cells
2.6. Study of the Mechanism by which DMSC Protect NB69 Cells Damaged by the Toxic MPP+
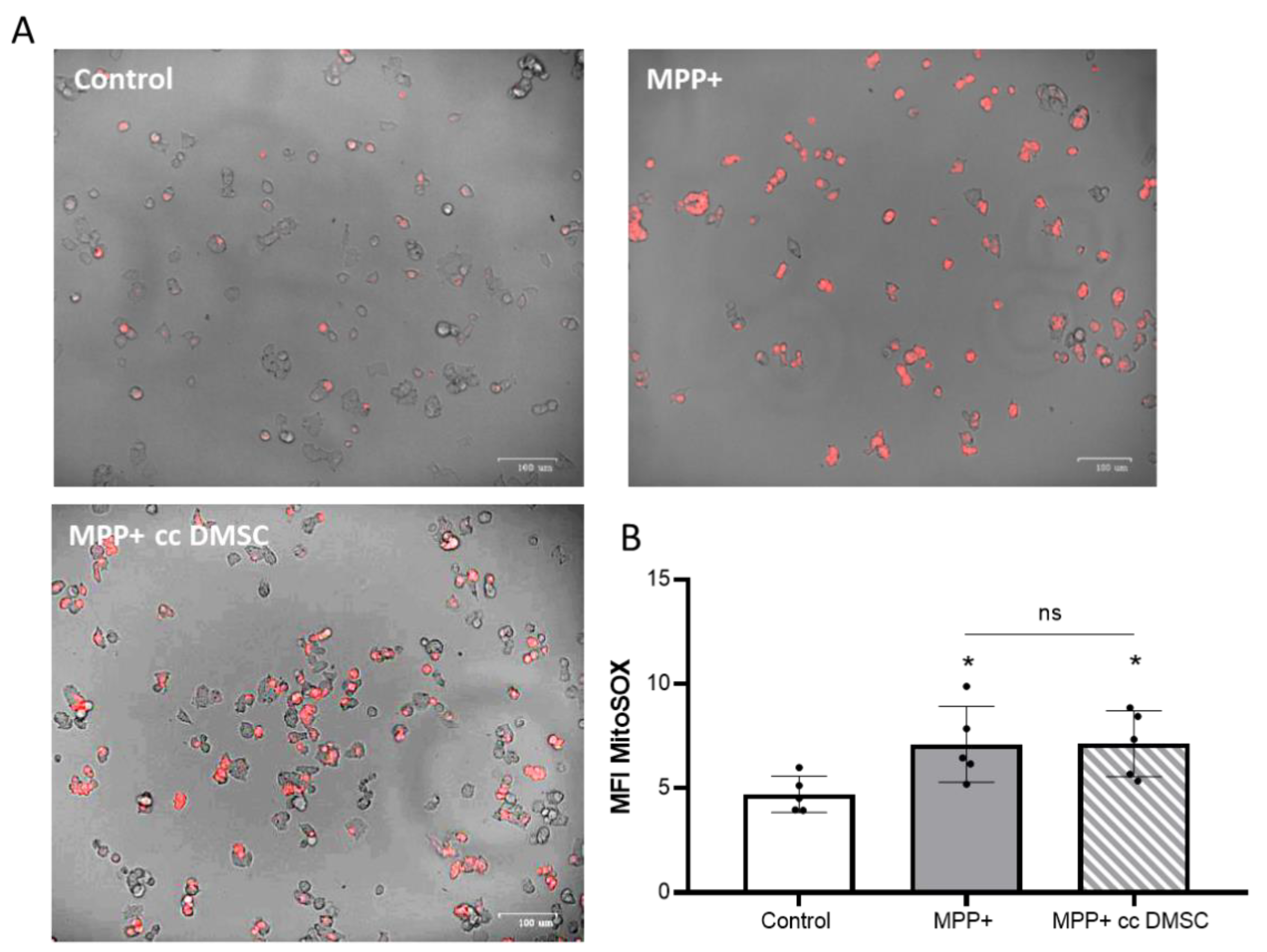
3. Discussion
4. Materials and Methods
4.1. Isolation and Culture of Decidua Mesenchymal Stromal Cells (DMSC)
4.2. Cell Culture and Differentiation of Neuroblastoma Cell Line NB69
4.3. Preparation of MPP+ Toxin and Treatment of NB69 Cells
4.4. Cell Viability Assay
4.5. Cell Death Assay
4.6. Cell Proliferation Assay
4.7. Indirect Co-Culture Assay of NB69 and DMSC Cells
4.8. DMSC Migration Assay
4.9. Immunofluorescence
4.10. Measurement of Neurite Length
4.11. Detection of Mitochondrial of Reactive Oxygen Species (ROS) Levels
4.12. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Connolly, B.S.; Lang, A.E. Pharmacological treatment of Parkinson disease: a review. JAMA 2014, 311, 1670–1683. [Google Scholar] [CrossRef]
- Wirdefeldt, K.; Adami, H.O.; Cole, P.; Trichopoulos, D.; Mandel, J. Epidemiology and etiology of Parkinson's disease: a review of the evidence. Eur J Epidemiol 2011, 26, S1–58. [Google Scholar] [CrossRef] [PubMed]
- Marti, M.J.; Tolosa, E.; Campdelacreu, J. Clinical overview of the synucleinopathies. Mov Disord 2003, 18 Suppl 6, S21–7. [Google Scholar] [CrossRef] [PubMed]
- Rcom-H'cheo-Gauthier, A.; Goodwin, J.; Pountney, D.L. Interactions between calcium and alpha-synuclein in neurodegeneration. Biomolecules 2014, 4, 795–811. [Google Scholar] [CrossRef] [PubMed]
- Chaudhuri, K.R.; Schapira, A.H. Non-motor symptoms of Parkinson's disease: dopaminergic pathophysiology and treatment. Lancet Neurol 2009, 8, 464–474. [Google Scholar] [CrossRef]
- Dexter, D.T.; Jenner, P. Parkinson disease: from pathology to molecular disease mechanisms. Free Radic Biol Med 2013, 62, 132–144. [Google Scholar] [CrossRef]
- Mensikova, K.; Matej, R.; Colosimo, C.; Rosales, R.; Tuckova, L.; Ehrmann, J.; Hrabos, D.; Kolarikova, K.; Vodicka, R.; Vrtel, R.; Prochazka, M.; Nevrly, M.; Kaiserova, M.; Kurcova, S.; Otruba, P.; Kanovsky, P. Lewy body disease or diseases with Lewy bodies? NPJ Parkinsons Dis 2022, 8, 3. [Google Scholar] [CrossRef]
- Perez, R.G.; Hastings, T.G. Could a loss of alpha-synuclein function put dopaminergic neurons at risk? J Neurochem 2004, 89, 1318–1324. [Google Scholar] [CrossRef]
- Franco-Iborra, S.; Vila, M.; Perier, C. The Parkinson Disease Mitochondrial Hypothesis: Where Are We at? Neuroscientist 2016, 22, 266–277. [Google Scholar] [CrossRef]
- Stolzenberg, E.; Berry, D.; Yang, D.; Lee, E.Y.; Kroemer, A.; Kaufman, S.; Wong, G.C.L.; Oppenheim, J.J.; Sen, S.; Fishbein, T.; Bax, A.; Harris, B.; Barbut, D.; Zasloff, M.A. A Role for Neuronal Alpha-Synuclein in Gastrointestinal Immunity. J Innate Immun 2017, 9, 456–463. [Google Scholar] [CrossRef]
- Pajares, M.; I.R., A.; Manda, G.; Bosca, L.; Cuadrado, A. Inflammation in Parkinson's Disease: Mechanisms and Therapeutic Implications. Cells 2020, 9, 7. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.L.; Li, C.; Nabeka, H.; Shimokawa, T.; Saito, S.; Wang, Z.Y.; Cao, Y.M.; Matsuda, S. Attenuation of MPTP/MPP(+) toxicity in vivo and in vitro by an 18-mer peptide derived from prosaposin. Neuroscience 2013, 236, 373–393. [Google Scholar] [CrossRef] [PubMed]
- Singer, T.P.; Ramsay, R.R. Mechanism of the neurotoxicity of MPTP. An update. FEBS Lett 1990, 274(1–2), 1–8. [Google Scholar] [PubMed]
- Liu, M.; Zuo, S.; Guo, X.; Peng, J.; Xing, Y.; Guo, Y.; Li, C.; Xing, H. The Study of Overexpression of Peroxiredoxin-2 Reduces MPP(+)-Induced Toxicity in the Cell Model of Parkinson's Disease. Neurochem Res 2023, 48, 2129–2137. [Google Scholar] [CrossRef]
- Risiglione, P.; Leggio, L.; Cubisino, S.A.M.; Reina, S.; Paterno, G.; Marchetti, B.; Magri, A.; Iraci, N.; Messina, A. High-Resolution Respirometry Reveals MPP(+) Mitochondrial Toxicity Mechanism in a Cellular Model of Parkinson's Disease. Int J Mol Sci 2020, 21, 21. [Google Scholar] [CrossRef]
- Dong, G.; Fan, L.; Li, C.; Jiao, Y.; Li, X.; Li, H.; Liang, Y.; Ren, Y.; Wang, L.; Xiao, D.; Xiao, J.; Wu, T.; Li, L. Schisanhenol Inhibits MPTP/MPP(+)-Induced Ferroptosis in Dopaminergic Neurons Via Nrf2/TrxR1/GPX4 Pathway against Parkinson's Disease. Neurochem Res 2025, 51, 26. [Google Scholar] [CrossRef]
- Svay, T.; Pariyar, R.; Bastola, T.; Yoon, C.S.; Lee, E.S.; Oh, H. Seo, J., 1beta,6alpha-Dihydroxyeudesm-4(15)-ene Protects Against MPP(+)-Induced Cytotoxicity in SH-SY5Y Cells: An In Vitro Model of Parkinson's Disease. J Med Food 2026, 29, 71–81. [Google Scholar] [CrossRef]
- Nakamura, K.; Bindokas, V.P.; Marks, J.D.; Wright, D.A.; Frim, D.M.; Miller, R.J.; Kang, U.J. The selective toxicity of 1-methyl-4-phenylpyridinium to dopaminergic neurons: the role of mitochondrial complex I and reactive oxygen species revisited. Mol Pharmacol 2000, 58, 271–278. [Google Scholar] [CrossRef]
- Perfeito, R.; Cunha-Oliveira, T.; Rego, A.C. Reprint of: revisiting oxidative stress and mitochondrial dysfunction in the pathogenesis of Parkinson disease-resemblance to the effect of amphetamine drugs of abuse. Free Radic Biol Med 2013, 62, 186–201. [Google Scholar] [CrossRef]
- Kim, T.Y.; Lee, B.D. Current therapeutic strategies in Parkinson's disease: Future perspectives. Mol Cells 2025, 48, 100274. [Google Scholar] [CrossRef]
- Seppi, K.; Ray Chaudhuri, K.; Coelho, M.; Fox, S.H.; Katzenschlager, R.; Perez Lloret, S.; Weintraub, D.; Sampaio, C. the collaborators of the Parkinson's Disease Update on Non-Motor Symptoms Study Group on behalf of the Movement Disorders Society Evidence-Based Medicine, C., Update on treatments for nonmotor symptoms of Parkinson's disease-an evidence-based medicine review. Mov Disord 2019, 34, 180–198. [Google Scholar]
- Zahoor, I.; Shafi, A.; Haq, E. Pharmacological Treatment of Parkinson's Disease. In Parkinson's Disease: Pathogenesis and Clinical Aspects; Stoker, T.B., Greenland, J.C., Eds.; Brisbane (AU), 2018. [Google Scholar]
- Warren Olanow, C.; Kieburtz, K.; Rascol, O.; Poewe, W.; Schapira, A.H.; Emre, M.; Nissinen, H.; Leinonen, M.; Stocchi, F. Stalevo Reduction in Dyskinesia Evaluation in Parkinson's Disease, I., Factors predictive of the development of Levodopa-induced dyskinesia and wearing-off in Parkinson's disease. Mov Disord 2013, 28, 1064–1071. [Google Scholar] [CrossRef]
- Liu, Z.; Cheung, H.H. Stem Cell-Based Therapies for Parkinson Disease. Int J Mol Sci 2020, 21, 21. [Google Scholar] [CrossRef]
- Zhang, A.L.; Wen, L. Synergetic pathways for Parkinson's disease therapy: The intersection of exercise and stem cell science. World J Stem Cells 2026, 18, 113930. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.E.; Qiu, L.; Chan, L.L.; Tan, E.K.; Zeng, L. Current Status of Stem Cell-Derived Therapies for Parkinson's Disease: From Cell Assessment and Imaging Modalities to Clinical Trials. Front Neurosci 2020, 14, 558532. [Google Scholar] [CrossRef] [PubMed]
- Lindvall, O. Clinical translation of stem cell transplantation in Parkinson's disease. J Intern Med 2016, 279, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Piccini, P.; Pavese, N.; Hagell, P.; Reimer, J.; Bjorklund, A.; Oertel, W.H.; Quinn, N.P.; Brooks, D.J.; Lindvall, O. Factors affecting the clinical outcome after neural transplantation in Parkinson's disease. Brain 2005, 128 (Pt 12), 2977–2986. [Google Scholar] [CrossRef]
- Clark, B.J.; Lelos, M.J.; Loring, J.F. Advancing Parkinson's disease treatment: cell replacement therapy with neurons derived from pluripotent stem cells. Stem Cells 2024, 42, 781–790. [Google Scholar] [CrossRef]
- Politis, M.; Lindvall, O. Clinical application of stem cell therapy in Parkinson's disease. BMC Med 2012, 10, 1. [Google Scholar] [CrossRef]
- Thanaskody, K.; Jusop, A.S.; Tye, G.J.; Wan Kamarul Zaman, W.S.; Dass, S.A.; Nordin, F. MSCs vs. iPSCs: Potential in therapeutic applications. Front Cell Dev Biol 2022, 10, 1005926. [Google Scholar] [CrossRef]
- Liu, L.; Eckert, M.A.; Riazifar, H.; Kang, D.K.; Agalliu, D.; Zhao, W. From blood to the brain: can systemically transplanted mesenchymal stem cells cross the blood-brain barrier? Stem Cells Int 2013, 2013, 435093. [Google Scholar] [CrossRef]
- Vegh, I.; Grau, M.; Gracia, M.; Grande, J.; de la Torre, P.; Flores, A.I. Decidua mesenchymal stem cells migrated toward mammary tumors in vitro and in vivo affecting tumor growth and tumor development. Cancer Gene Ther 2013, 20, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Volkman, R.; Offen, D. Concise Review: Mesenchymal Stem Cells in Neurodegenerative Diseases. Stem Cells 2017, 35, 1867–1880. [Google Scholar] [CrossRef] [PubMed]
- Torre, P.; Flores, A.I. Current Status and Future Prospects of Perinatal Stem Cells. Genes (Basel) 2020, 12((1)). [Google Scholar] [CrossRef] [PubMed]
- Macias, M.I.; Grande, J.; Moreno, A.; Dominguez, I.; Bornstein, R.; Flores, A.I. Isolation and characterization of true mesenchymal stem cells derived from human term decidua capable of multilineage differentiation into all 3 embryonic layers. Am J Obstet Gynecol 2010, 203, 495 e9–495 e23. [Google Scholar] [CrossRef]
- Bravo, B.; Gallego, M.I.; Flores, A.I.; Bornstein, R.; Puente-Bedia, A.; Hernandez, J.; de la Torre, P.; Garcia-Zaragoza, E.; Perez-Tavarez, R.; Grande, J.; Ballester, A.; Ballester, S. Restrained Th17 response and myeloid cell infiltration into the central nervous system by human decidua-derived mesenchymal stem cells during experimental autoimmune encephalomyelitis. Stem Cell Res Ther 2016, 7, 43. [Google Scholar] [CrossRef]
- de la Torre, P.; Paris, J.L.; Fernandez-de la Torre, M.; Vallet-Regi, M.; Flores, A.I. Endostatin Genetically Engineered Placental Mesenchymal Stromal Cells Carrying Doxorubicin-Loaded Mesoporous Silica Nanoparticles for Combined Chemo- and Antiangiogenic Therapy. Pharmaceutics 2021, 13((2)). [Google Scholar] [CrossRef]
- De La Torre, P.; Perez-Lorenzo, M.J.; Alcazar-Garrido, A.; Collado, J.; Martinez-Lopez, M.; Forcen, L.; Masero-Casasola, A.R.; Garcia, A.; Gutierrez-Velez, M.C.; Medina-Polo, J.; Munoz, E.; Flores, A.I. Perinatal mesenchymal stromal cells of the human decidua restore continence in rats with stress urinary incontinence induced by simulated birth trauma and regulate senescence of fibroblasts from women with stress urinary incontinence. Front Cell Dev Biol 2022, 10, 1033080. [Google Scholar] [CrossRef]
- Paris, J.L.; de la Torre, P.; Cabanas, M.V.; Manzano, M.; Flores, A.I.; Vallet-Regi, M. Suicide-gene transfection of tumor-tropic placental stem cells employing ultrasound-responsive nanoparticles. Acta Biomater 2019, 83, 372–378. [Google Scholar] [CrossRef]
- Paris, J.L.; de la Torre, P.; Flores, A.I. New Therapeutic Approaches for Allergy: A Review of Cell Therapy and Bio- or Nano-Material-Based Strategies. Pharmaceutics 2021, 13, 12. [Google Scholar] [CrossRef]
- Paris, J.L.; de la Torre, P.; Manzano, M.; Cabanas, M.V.; Flores, A.I.; Vallet-Regi, M. Decidua-derived mesenchymal stem cells as carriers of mesoporous silica nanoparticles. In vitro and in vivo evaluation on mammary tumors. Acta Biomater 2016, 33, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Paris, J.L.; de la Torre, P.; Victoria Cabanas, M.; Manzano, M.; Grau, M.; Flores, A.I.; Vallet-Regi, M. Vectorization of ultrasound-responsive nanoparticles in placental mesenchymal stem cells for cancer therapy. Nanoscale 2017, 9, 5528–5537. [Google Scholar] [CrossRef] [PubMed]
- Mena, M.A.; Casarejos, M.J.; Bonin, A.; Ramos, J.A.; Garcia Yebenes, J. Effects of dibutyryl cyclic AMP and retinoic acid on the differentiation of dopamine neurons: prevention of cell death by dibutyryl cyclic AMP. J Neurochem 1995, 65, 2612–2620. [Google Scholar] [CrossRef] [PubMed]
- Lopes, F.M.; Bristot, I.J.; da Motta, L.L.; Parsons, R.B.; Klamt, F. Mimicking Parkinson's Disease in a Dish: Merits and Pitfalls of the Most Commonly used Dopaminergic In Vitro Models. Neuromolecular Med 2017, 19((2–3)), 241–255. [Google Scholar] [CrossRef]
- Ferrari, E.; Cardinale, A.; Picconi, B.; Gardoni, F. From cell lines to pluripotent stem cells for modelling Parkinson's Disease. J Neurosci Methods 2020, 340, 108741. [Google Scholar] [CrossRef]
- Mena, M.A.; Pardo, B.; Casarejos, M.J.; Fahn, S.; Garcia de Yebenes, J. Neurotoxicity of levodopa on catecholamine-rich neurons. Mov Disord 1992, 7, 23–31. [Google Scholar] [CrossRef]
- Rodriguez-Martin, E.; Canals, S.; Casarejos, M.J.; de Bernardo, S.; Handler, A.; Mena, M.A. L-DOPA and glia-conditioned medium have additive effects on tyrosine hydroxylase expression in human catecholamine-rich neuroblastoma NB69 cells. J Neurochem 2001, 78, 535–545. [Google Scholar] [CrossRef]
- Harberts, J.; Siegmund, M.; Schnelle, M.; Zhang, T.; Lei, Y.; Yu, L.; Zierold, R.; Blick, R.H. Robust neuronal differentiation of human iPSC-derived neural progenitor cells cultured on densely-spaced spiky silicon nanowire arrays. Sci Rep 2021, 11, 18819. [Google Scholar] [CrossRef]
- Kim, H.; Zahir, T.; Tator, C.H.; Shoichet, M.S. Effects of dibutyryl cyclic-AMP on survival and neuronal differentiation of neural stem/progenitor cells transplanted into spinal cord injured rats. PLoS One 2011, 6, e21744. [Google Scholar] [CrossRef]
- Khwanraj, K.; Phruksaniyom, C.; Madlah, S.; Dharmasaroja, P. Differential Expression of Tyrosine Hydroxylase Protein and Apoptosis-Related Genes in Differentiated and Undifferentiated SH-SY5Y Neuroblastoma Cells Treated with MPP(.). Neurol Res Int 2015, 2015, 734703. [Google Scholar] [CrossRef]
- Tambe, P.; Undale, V.; Sanap, A.; Bhonde, R.; Mante, N. The prospective role of mesenchymal stem cells in Parkinson's disease. Parkinsonism Relat Disord 2024, 127, 107087. [Google Scholar] [CrossRef]
- Tew, V.K.; Barathan, M.; Nordin, F.; Law, J.X.; Ng, M.H. Emerging Role of Mesenchymal Stromal Cell and Exosome Therapies in Treating Cognitive Impairment. Pharmaceutics 2025, 17((3)). [Google Scholar] [CrossRef] [PubMed]
- Lee, W.K.; Wolff, N.A.; Thevenod, F. Organic cation transporters: physiology, toxicology and special focus on ethidium as a novel substrate. Curr Drug Metab 2009, 10, 617–631. [Google Scholar] [CrossRef]
- Uhlen, M.; Fagerberg, L.; Hallstrom, B.M.; Lindskog, C.; Oksvold, P.; Mardinoglu, A.; Sivertsson, A.; Kampf, C.; Sjostedt, E.; Asplund, A.; Olsson, I.; Edlund, K.; Lundberg, E.; Navani, S.; Szigyarto, C.A.; Odeberg, J.; Djureinovic, D.; Takanen, J.O.; Hober, S.; Alm, T.; Edqvist, P.H.; Berling, H.; Tegel, H.; Mulder, J.; Rockberg, J.; Nilsson, P.; Schwenk, J.M.; Hamsten, M.; von Feilitzen, K.; Forsberg, M.; Persson, L.; Johansson, F.; Zwahlen, M.; von Heijne, G.; Nielsen, J.; Ponten, F. Proteomics. Tissue-based map of the human proteome. Science 2015, 347, 1260419. [Google Scholar] [CrossRef] [PubMed]
- Sata, R.; Ohtani, H.; Tsujimoto, M.; Murakami, H.; Koyabu, N.; Nakamura, T.; Uchiumi, T.; Kuwano, M.; Nagata, H.; Tsukimori, K.; Nakano, H.; Sawada, Y. Functional analysis of organic cation transporter 3 expressed in human placenta. J Pharmacol Exp Ther 2005, 315, 888–895. [Google Scholar] [CrossRef] [PubMed]
- Yanagawa, T.; Kishimoto, Y.; Tada, K.; Arai, F.; Kondo, Y.; Kudo, T. Presence of dopamine DA-1 receptors in human decidua. Placenta 1997, 18((2–3)), 169–172. [Google Scholar] [CrossRef]
- Khatlani, T.; Algudiri, D.; Alenzi, R.; Al Subayyil, A.M.; Abomaray, F.M.; Bahattab, E.; AlAskar, A.S.; Kalionis, B.; El-Muzaini, M.F.; Abumaree, M.H. Preconditioning by Hydrogen Peroxide Enhances Multiple Properties of Human Decidua Basalis Mesenchymal Stem/Multipotent Stromal Cells. Stem Cells Int 2018, 2018, 6480793. [Google Scholar] [CrossRef]
- Carvajal-Oliveros, A.; Roman-Martinez, C.; Reynaud, E.; Martinez-Martinez, E. The BE (2)-M17 neuroblastoma cell line: revealing its potential as a cellular model for Parkinson's disease. Front Cell Neurosci 2024, 18, 1485414. [Google Scholar] [CrossRef]
- Yari, H.; Mikhailova, M.V.; Mardasi, M.; Jafarzadehgharehziaaddin, M.; Shahrokh, S.; Thangavelu, L.; Ahmadi, H.; Shomali, N.; Yaghoubi, Y.; Zamani, M.; Akbari, M.; Alesaeidi, S. Emerging role of mesenchymal stromal cells (MSCs)-derived exosome in neurodegeneration-associated conditions: a groundbreaking cell-free approach. Stem Cell Res Ther 2022, 13, 423. [Google Scholar] [CrossRef]
- Abomaray, F.M.; Al Jumah, M.A.; Alsaad, K.O.; Jawdat, D.; Al Khaldi, A.; AlAskar, A.S.; Al Harthy, S.; Al Subayyil, A.M.; Khatlani, T.; Alawad, A.O.; Alkushi, A.; Kalionis, B.; Abumaree, M.H. Phenotypic and Functional Characterization of Mesenchymal Stem/Multipotent Stromal Cells from Decidua Basalis of Human Term Placenta. Stem Cells Int 2016, 2016, 5184601. [Google Scholar] [CrossRef]
- Angelova, P.R.; Barilani, M.; Lovejoy, C.; Dossena, M.; Vigano, M.; Seresini, A.; Piga, D.; Gandhi, S.; Pezzoli, G.; Abramov, A.Y.; Lazzari, L. Mitochondrial dysfunction in Parkinsonian mesenchymal stem cells impairs differentiation. Redox Biol 2018, 14, 474–484. [Google Scholar] [CrossRef]
- Mukhopadhyay, P.; Rajesh, M.; Yoshihiro, K.; Hasko, G.; Pacher, P. Simple quantitative detection of mitochondrial superoxide production in live cells. Biochem Biophys Res Commun 2007, 358, 203–208. [Google Scholar] [CrossRef]
- Dikalov, S.I.; Harrison, D.G. Methods for detection of mitochondrial and cellular reactive oxygen species. Antioxid Redox Signal 2014, 20, 372–382. [Google Scholar] [CrossRef]
- Chen, H.; Chen, X.; Zhou, Z.H.; Zheng, J.R.; Lu, Y.; Lin, P.; Lin, Y.F.; Zheng, Y.C.; Xiong, B.; Xu, R.W.; Cui, L.; Zhao, X.Y. Mesenchymal stromal cell-mediated mitochondrial transfer unveils new frontiers in disease therapy. Stem Cell Res Ther 2025, 16, 546. [Google Scholar] [CrossRef] [PubMed]
- Prasertsuksri, P.; Kraokaew, P.; Pranweerapaiboon, K.; Sobhon, P.; Chaithirayanon, K. Neuroprotection of Andrographolide against Neurotoxin MPP(+)-Induced Apoptosis in SH-SY5Y Cells via Activating Mitophagy, Autophagy, and Antioxidant Activities. Int J Mol Sci 2023, 24((10)). [Google Scholar] [CrossRef] [PubMed]
- Mukkala, A.N.; Jerkic, M.; Khan, Z.; Szaszi, K.; Kapus, A.; Rotstein, O. Therapeutic Effects of Mesenchymal Stromal Cells Require Mitochondrial Transfer and Quality Control. Int J Mol Sci 2023, 24, 21. [Google Scholar] [CrossRef] [PubMed]
- Eo, H.; Yu, S.H.; Choi, Y.; Kim, Y.; Kang, Y.C.; Lee, H.; Kim, J.H.; Han, K.; Lee, H.K.; Chang, M.Y.; Oh, M.S.; Kim, C.H. Mitochondrial transplantation exhibits neuroprotective effects and improves behavioral deficits in an animal model of Parkinson's disease. Neurotherapeutics 2024, 21, e00355. [Google Scholar] [CrossRef]
- Thakur, P.; Nehru, B. Inhibition of neuroinflammation and mitochondrial dysfunctions by carbenoxolone in the rotenone model of Parkinson's disease. Mol Neurobiol 2015, 51, 209–219. [Google Scholar] [CrossRef]
- Cheng, X.Y.; Biswas, S.; Li, J.; Mao, C.J.; Chechneva, O.; Chen, J.; Li, K.; Li, J.; Zhang, J.R.; Liu, C.F.; Deng, W.B. Human iPSCs derived astrocytes rescue rotenone-induced mitochondrial dysfunction and dopaminergic neurodegeneration in vitro by donating functional mitochondria. Transl Neurodegener 2020, 9, 13. [Google Scholar] [CrossRef]
- Tran, C.M.; Nguyen, T.T. Mesenchymal stem cells and the rescue of mitochondrial damage in different disease models (Review). World Acad Sci J 2026, 8, 4. [Google Scholar] [CrossRef]
- Mena, M.A.; Garcia de Yebenes, J.; Dwork, A.; Fahn, S.; Latov, N.; Herbert, J.; Flaster, E.; Slonim, D. Biochemical properties of monoamine-rich human neuroblastoma cells. Brain Res 1989, 486, 286–296. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



