Submitted:
01 May 2026
Posted:
05 May 2026
You are already at the latest version
Abstract
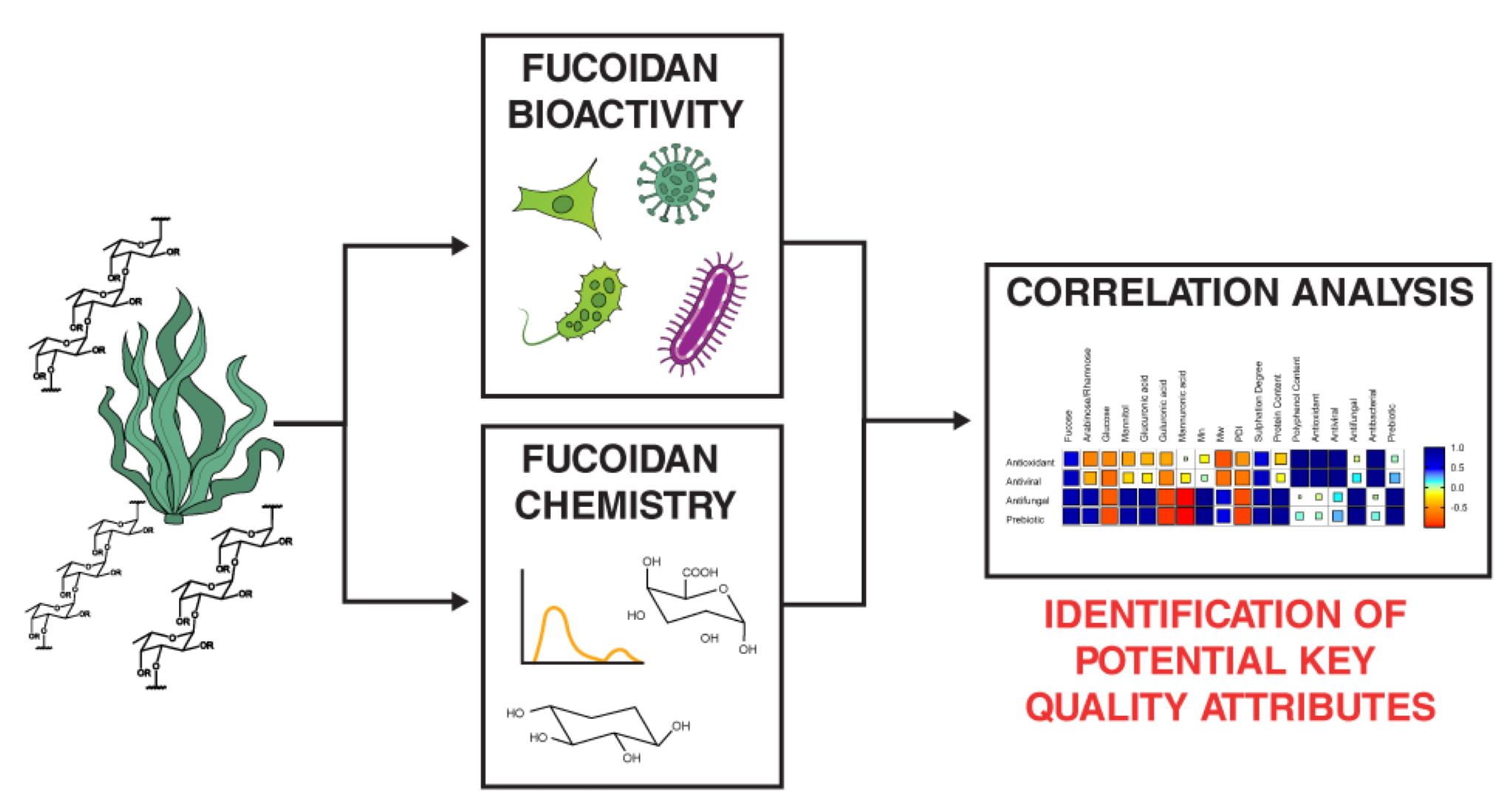
Keywords:
1. Introduction
2. Results
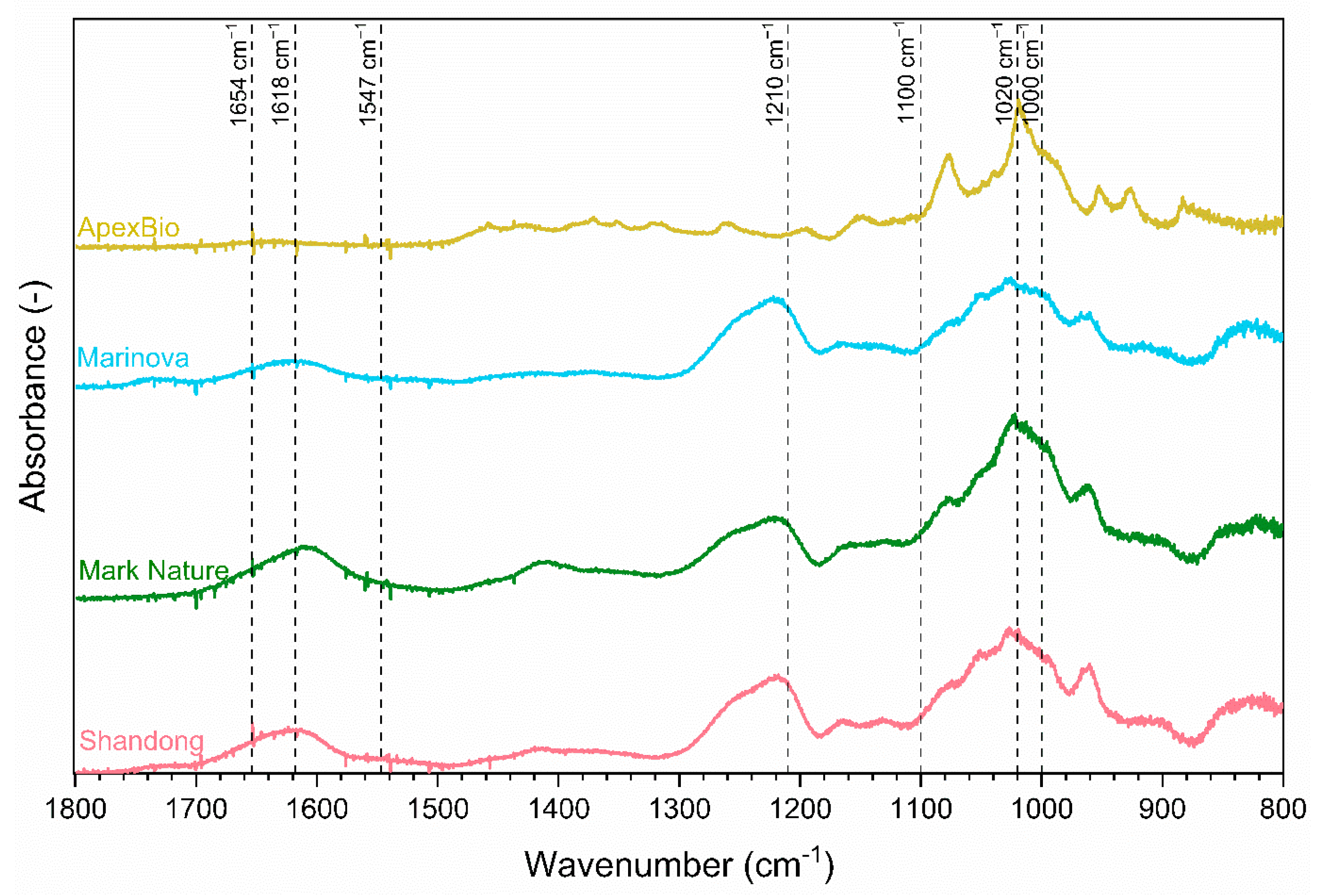
2.1. Structural Characteristics of Extracts
2.2. Chemical Characteristics of Extracts
2.2.1. Monosaccharide and Uronic Acid Content of Extracts
2.2.2. Molecular Weight of Extracts
2.2.3. Elemental Content of Extracts
2.2.4. Phenolic Content of Extracts
2.3. Summary of Extracts Chemical Content and Structural Characterization
2.4. Biological Activity Assessment of Extracts
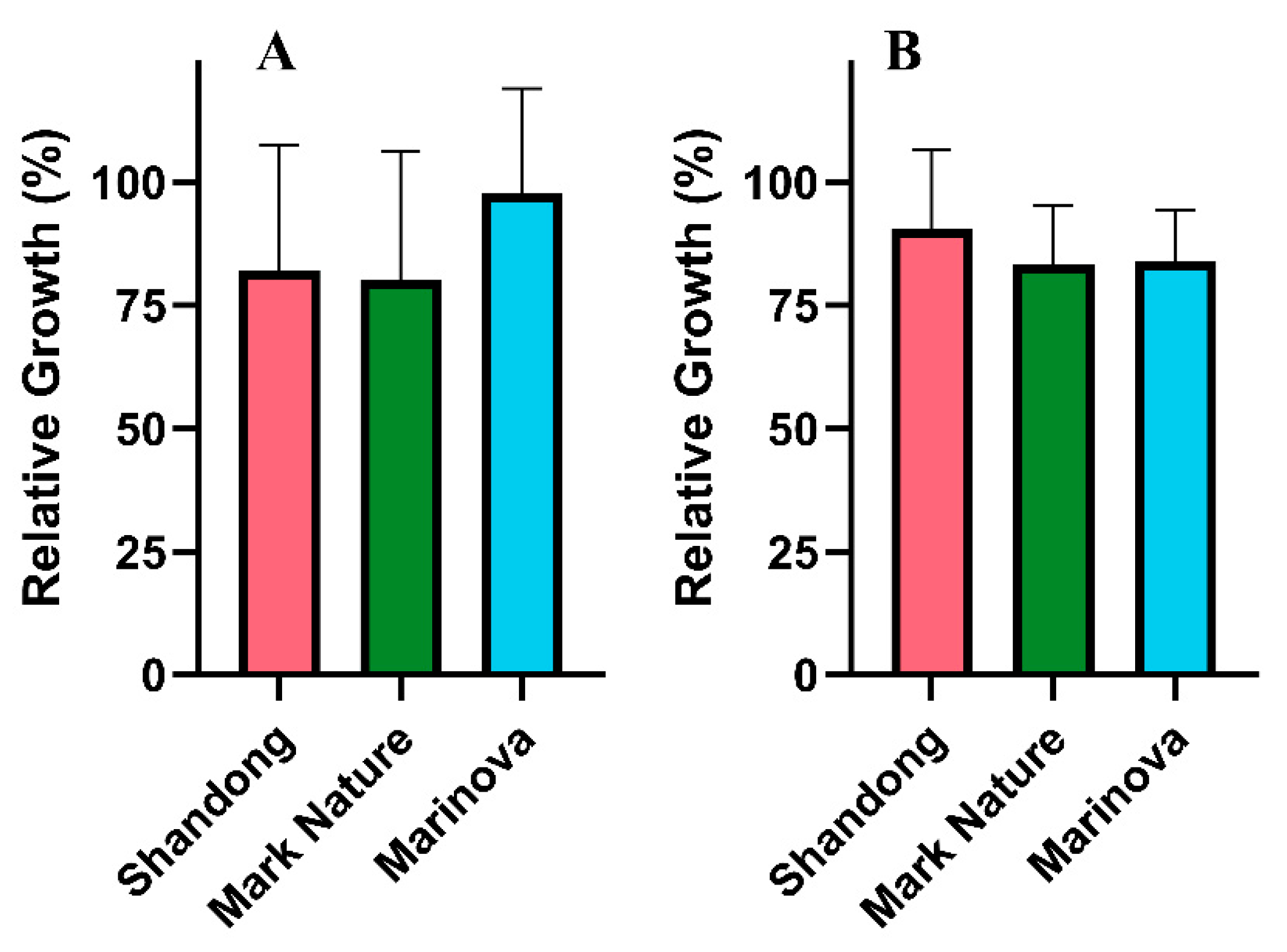
2.4.1. Antioxidant Activity
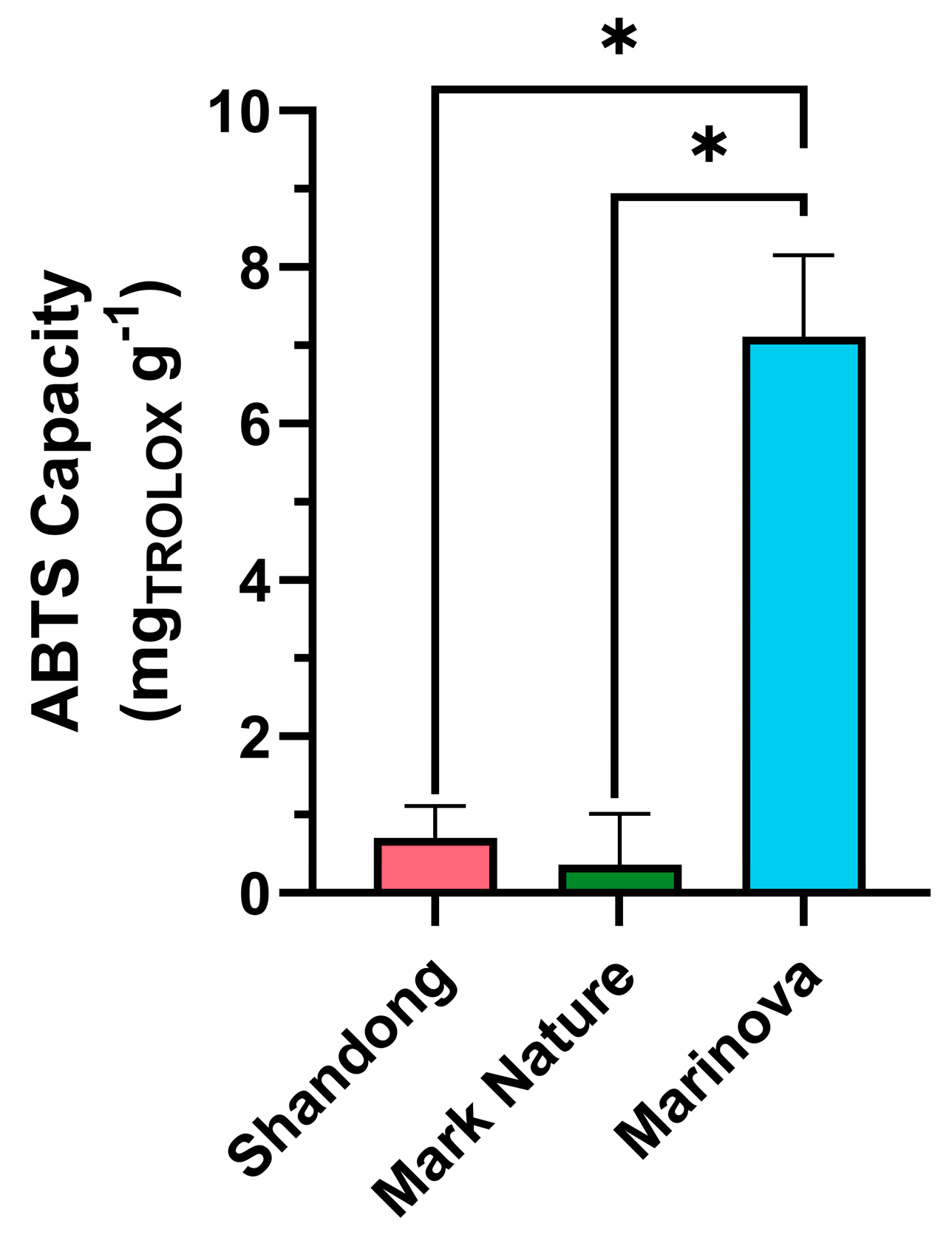
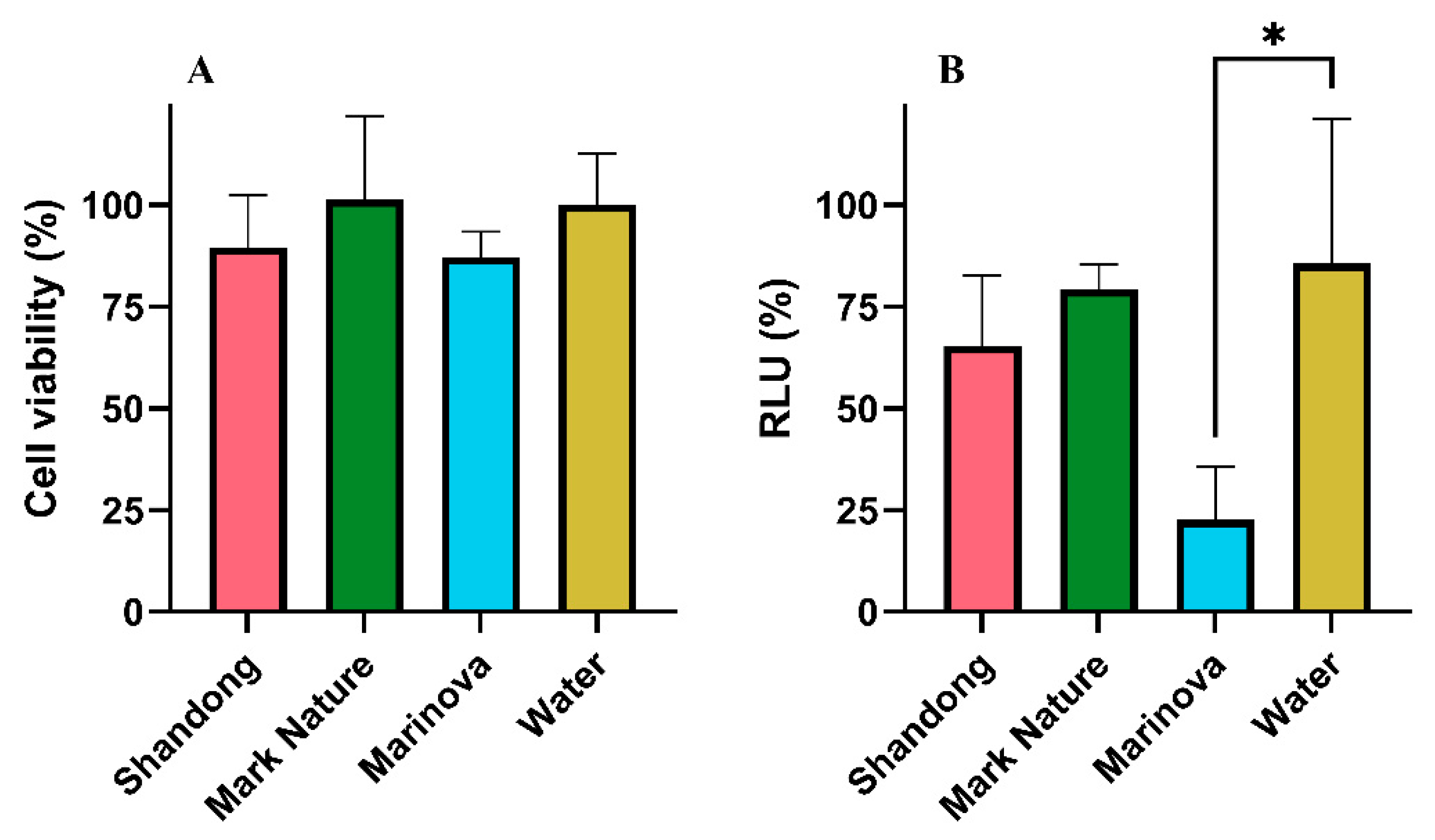
2.4.2. Antiviral Activity
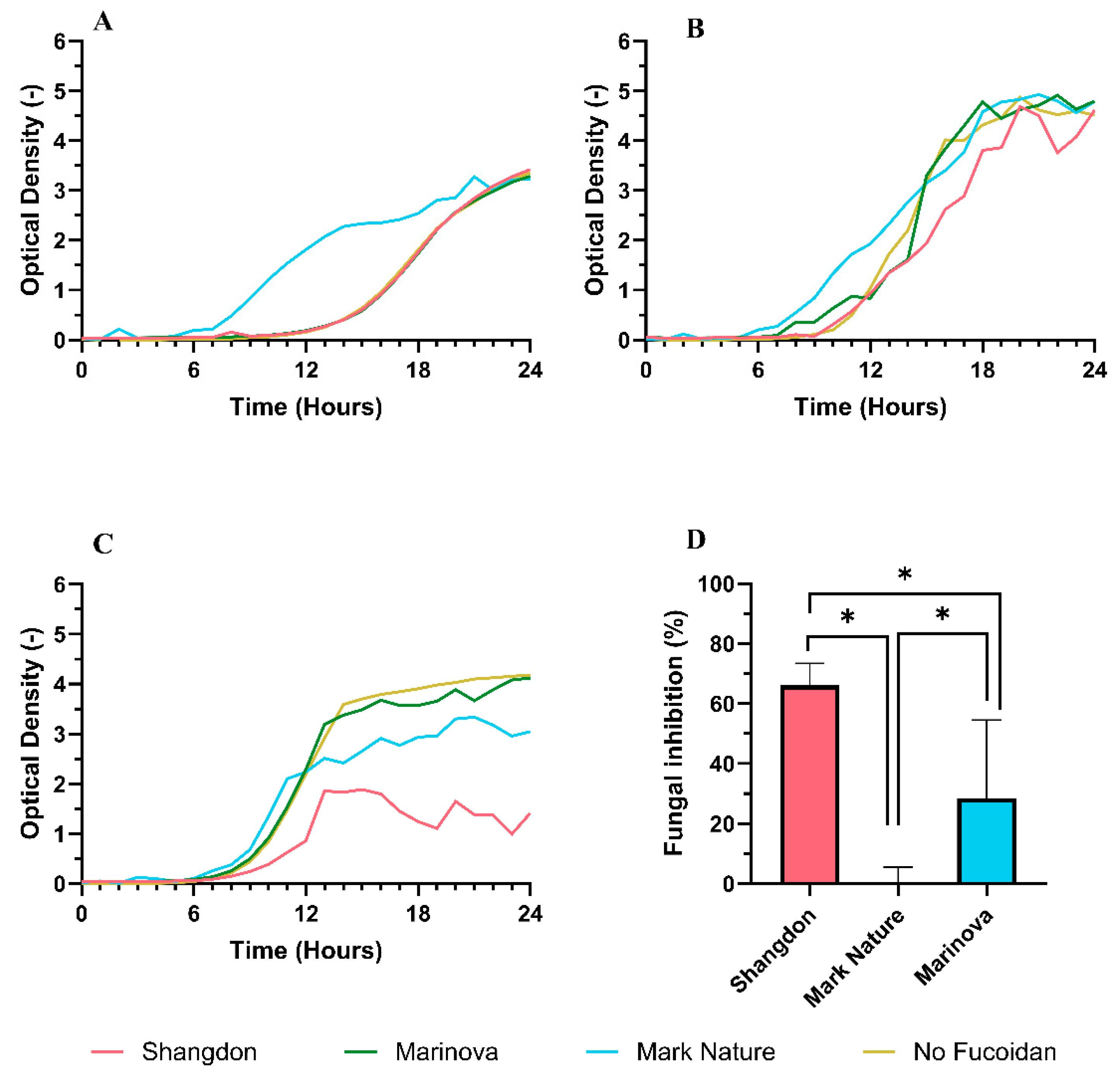
2.4.3. Antifungal Activity
2.4.4. Antibacterial Activity
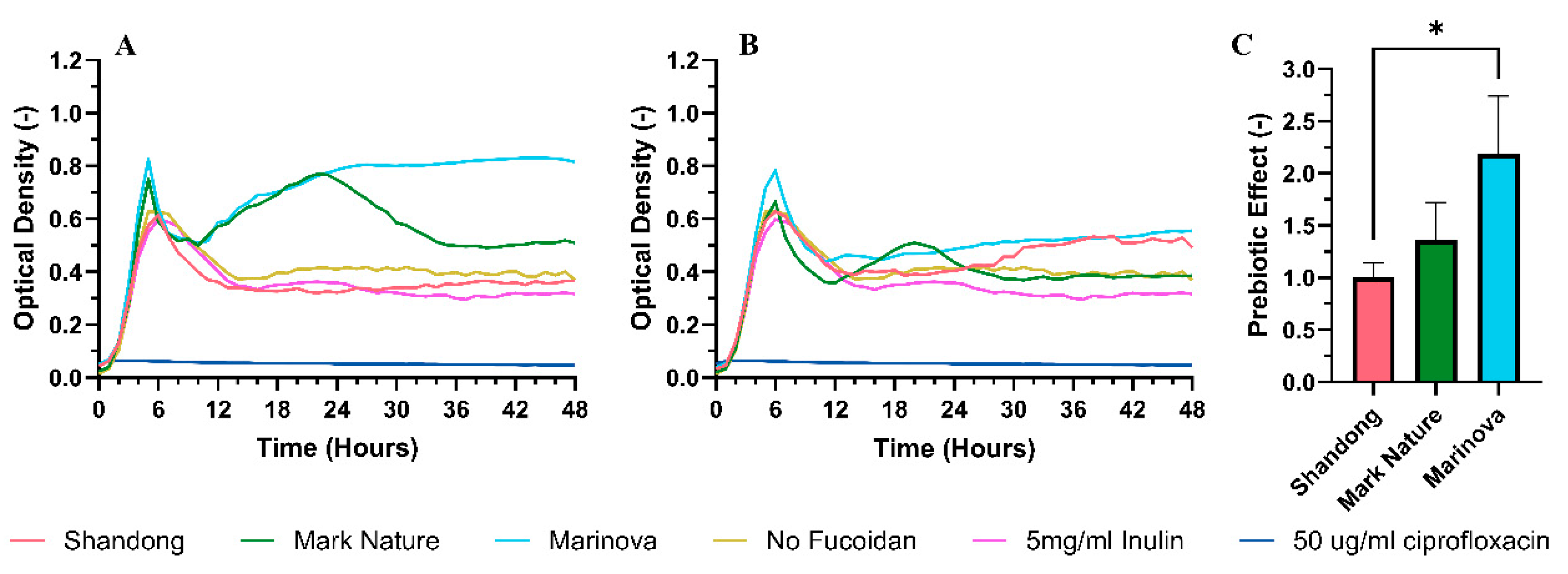
2.4.5. Prebiotic Activity
3. Discussion
3.1. Bioactivity Correlation Matrix
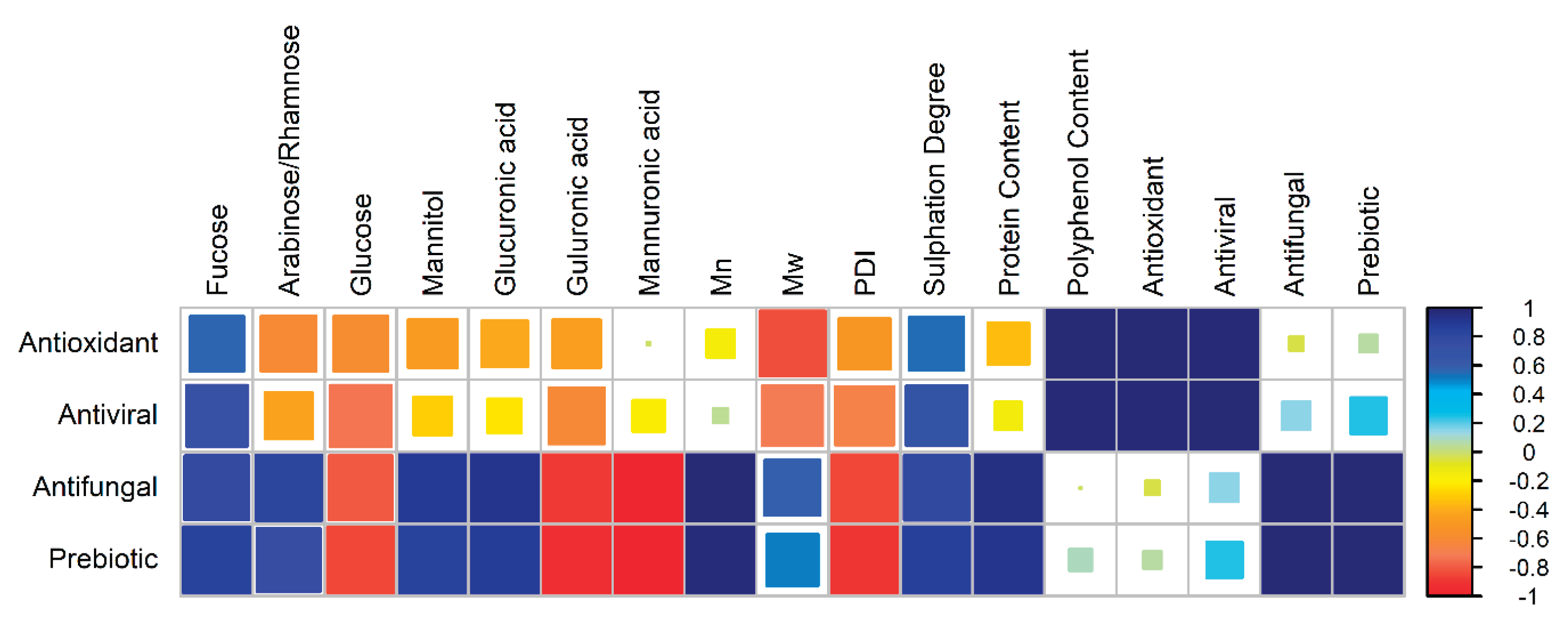
3.2. Key Limitations of This Study
3.3. Further Work
4. Materials and Methods
4.1. Selection of Commercial Fucoidans
4.2. Chemical and Structural Characterisation of Fucoidan Extracts
4.2.1. Fourier Transform Infrared Spectroscopy
4.2.2. Size Exclusion Chromatograpy with Multi-Angled Light Scattering Detector (SEC-MALS)
4.2.3. Elemental Analysis (CHNS)
4.2.4. Inductively Coupled Plasma-Optical Emission Spectrometry (ICP-OES)
4.2.5. High Performance Anion Exchange Chromatography – Pulsed Amperometric Detection (HPAEC-PAD)
4.2.6. Folin–Ciocâlteu Colorimetric Assay
4.3. Biological Activity Assessment of Extracts
4.3.1. Antioxidant Activity
4.3.2. Antiviral Activity
4.3.3. Antifungal Activity
4.3.4. Antibacterial Activity
4.3.5. Prebiotic Activity
4.3. Statistical Analysis and Pearson’s Correlation Matrix
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- dos Santos, M.A.; Grenha, A. Chapter Seven - Polysaccharide Nanoparticles for Protein and Peptide Delivery: Exploring Less-Known Materials. In Advances in Protein Chemistry and Structural Biology; Protein and Peptide Nanoparticles for Drug Delivery; Donev, R., Ed.; Academic Press, 2015; Vol. 98, pp. 223–261. [Google Scholar]
- Yue, Q.; Liu, Y.; Li, F.; Hong, T.; Guo, S.; Cai, M.; Zhao, L.; Su, L.; Zhang, S.; Zhao, C.; et al. Antioxidant and Anticancer Properties of Fucoidan Isolated from Saccharina Japonica Brown Algae. Sci. Rep. 2025, 15, 8962. [Google Scholar] [CrossRef]
- Dobrinčić, A.; Pedisić, S.; Zorić, Z.; Jurin, M.; Roje, M.; Čož-Rakovac, R.; Dragović-Uzelac, V. Microwave Assisted Extraction and Pressurized Liquid Extraction of Sulfated Polysaccharides from Fucus Virsoides and Cystoseira Barbata. Foods 2021, 10, 1481. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-Y.; Wang, S.-H.; Huang, C.-Y.; Dong, C.-D.; Huang, C.-Y.; Chang, C.-C.; Chang, J.-S. Effect of Molecular Mass and Sulfate Content of Fucoidan from Sargassum Siliquosum on Antioxidant, Anti-Lipogenesis, and Anti-Inflammatory Activity. J. Biosci. Bioeng. 2021, 132, 359–364. [Google Scholar] [CrossRef]
- Pradhan, B.; Nayak, R.; Patra, S.; Bhuyan, P.P.; Behera, P.K.; Mandal, A.K.; Behera, C.; Ki, J.-S.; Adhikary, S.P.; MubarakAli, D.; et al. A State-of-the-Art Review on Fucoidan as an Antiviral Agent to Combat Viral Infections. Carbohydr. Polym. 2022, 291, 119551. [Google Scholar] [CrossRef]
- Nosik, M.N.; Krylova, N.V.; Usoltseva, R.V.; Surits, V.V.; Kireev, D.E.; Shchelkanov, M.Yu.; Svitich, O.A.; Ermakova, S.P. In Vitro Anti-HIV-1 Activity of Fucoidans from Brown Algae. Mar. Drugs 2024, 22, 355. [Google Scholar] [CrossRef]
- Thuy, T.T.T.; Ly, B.M.; Van, T.T.T.; Van Quang, N.; Tu, H.C.; Zheng, Y.; Seguin-Devaux, C.; Mi, B.; Ai, U. Anti-HIV Activity of Fucoidans from Three Brown Seaweed Species. Carbohydr. Polym. 2015, 115, 122–128. [Google Scholar] [CrossRef]
- Tabassum, N.; Khan, F.; Kang, M.-G.; Jo, D.-M.; Cho, K.-J.; Kim, Y.-M. Inhibition of Polymicrobial Biofilms of Candida Albicans–Staphylococcus Aureus/Streptococcus Mutans by Fucoidan–Gold Nanoparticles. Mar. Drugs 2023, 21, 123. [Google Scholar] [CrossRef]
- Akl, F.M.A.; El-Sheekh, M.M.; Makhlof, M.E.M.; Ahmed, S.I. Antimicrobial, Antidiabetic, Antiviral, and Antioxidant Activities of Fucoidan Extracted from the Brown Seaweed Padina Pavonica. BMC Biotechnol. 2025, 25, 70. [Google Scholar] [CrossRef]
- Sanniyasi, E.; Gopal, R.K.; Damodharan, R.; Arumugam, A.; Sampath Kumar, M.; Senthilkumar, N.; Anbalagan, M. In Vitro Anticancer Potential of Laminarin and Fucoidan from Brown Seaweeds. Sci. Rep. 2023, 13, 14452. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Yan, C.; Fu, Y.; Ai, C.; Bi, J.; Lin, W.; Song, S. Orally Administrated Fucoidan and Its Low-Molecular-Weight Derivatives Are Absorbed Differentially to Alleviate Coagulation and Thrombosis. Int. J. Biol. Macromol. 2024, 255, 128092. [Google Scholar] [CrossRef] [PubMed]
- Ayrapetyan, O.N.; Obluchinskaya, E.D.; Zhurishkina, E.V.; Skorik, Y.A.; Lebedev, D.V.; Kulminskaya, A.A.; Lapina, I.M. Antibacterial Properties of Fucoidans from the Brown Algae Fucus Vesiculosus L. of the Barents Sea. Biology 2021, 10, 67. [Google Scholar] [CrossRef]
- Alboofetileh, M.; Rezaei, M.; Tabarsa, M.; Rittà, M.; Donalisio, M.; Mariatti, F.; You, S.; Lembo, D.; Cravotto, G. Effect of Different Non-Conventional Extraction Methods on the Antibacterial and Antiviral Activity of Fucoidans Extracted from Nizamuddinia Zanardinii. Int. J. Biol. Macromol. 2019, 124, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Microbiome-Metabolomics Reveals Prebiotic Benefits of Fucoidan Supplementation in Mice. Available online: https://www.mdpi.com/2077-1312/9/5/505 (accessed on 16 June 2025).
- Chadwick, M.; Carvalho, L.G.; Vanegas, C.; Dimartino, S. A Comparative Review of Alternative Fucoidan Extraction Techniques from Seaweed. Mar. Drugs 2025, 23, 27. [Google Scholar] [CrossRef]
- Zayed, A.; El-Aasr, M.; Ibrahim, A.-R.S.; Ulber, R. Fucoidan Characterization: Determination of Purity and Physicochemical and Chemical Properties. Mar. Drugs 2020, 18, 571. [Google Scholar] [CrossRef] [PubMed]
- Cabral, E.M.; Mondala, J.R.M.; Oliveira, M.; Przyborska, J.; Fitzpatrick, S.; Rai, D.K.; Sivagnanam, S.P.; Garcia-Vaquero, M.; O’Shea, D.; Devereux, M.; et al. Influence of Molecular Weight Fractionation on the Antimicrobial and Anticancer Properties of a Fucoidan Rich-Extract from the Macroalgae Fucus Vesiculosus. Int. J. Biol. Macromol. 2021, 186, 994–1002. [Google Scholar] [CrossRef]
- Wang, Y.; Xing, M.; Cao, Q.; Ji, A.; Liang, H.; Song, S. Biological Activities of Fucoidan and the Factors Mediating Its Therapeutic Effects: A Review of Recent Studies. Mar. Drugs 2019, 17, 183. [Google Scholar] [CrossRef]
- Wang, L.; Oliveira, C.; Li, Q.; Ferreira, A.S.; Nunes, C.; Coimbra, M.A.; Reis, R.L.; Martins, A.; Wang, C.; Silva, T.H.; et al. Fucoidan from Fucus Vesiculosus Inhibits Inflammatory Response, Both In Vitro and In Vivo. Mar. Drugs 2023, 21, 302. [Google Scholar] [CrossRef] [PubMed]
- Mabate, B.; Daub, C.D.; Malgas, S.; Edkins, A.L.; Pletschke, B.I. Fucoidan Structure and Its Impact on Glucose Metabolism: Implications for Diabetes and Cancer Therapy. Mar. Drugs 2021, 19, 30. [Google Scholar] [CrossRef]
- Yuan, Y.; Macquarrie, D. Microwave Assisted Extraction of Sulfated Polysaccharides (Fucoidan) from Ascophyllum Nodosum and Its Antioxidant Activity. Carbohydr. Polym. 2015, 129, 101–107. [Google Scholar] [CrossRef]
- Okolie, C.L.; Mason, B.; Mohan, A.; Pitts, N.; Udenigwe, C.C. The Comparative Influence of Novel Extraction Technologies on in Vitro Prebiotic-Inducing Chemical Properties of Fucoidan Extracts from Ascophyllum Nodosum. Food Hydrocoll. 2019, 90, 462–471. [Google Scholar] [CrossRef]
- McGurrin, A.; Suchintita Das, R.; Soro, A.B.; Maguire, J.; Flórez Fernández, N.; Dominguez, H.; Torres, M.D.; Tiwari, B.K.; Garcia-Vaquero, M. Antimicrobial Activities of Polysaccharide-Rich Extracts from the Irish Seaweed Alaria Esculenta, Generated Using Green and Conventional Extraction Technologies, Against Foodborne Pathogens. Mar. Drugs 2025, 23, 46. [Google Scholar] [CrossRef]
- Mensah, E.O.; Kanwugu, O.N.; Panda, P.K.; Adadi, P. Marine Fucoidans: Structural, Extraction, Biological Activities and Their Applications in the Food Industry. Food Hydrocoll. 2023, 142, 108784. [Google Scholar] [CrossRef]
- Dimartino, S.; Savory, D.M.; Fraser-Miller, S.J.; Gordon, K.C.; McQuillan, A.J. Microscopic and Infrared Spectroscopic Comparison of the Underwater Adhesives Produced by Germlings of the Brown Seaweed Species Durvillaea Antarctica and Hormosira Banksii. J. R. Soc. Interface 2016, 13, 20151083. [Google Scholar] [CrossRef]
- Barth, A. Infrared Spectroscopy of Proteins. Biochim Biophys. Acta 2007, 1767, 1073–1101. [Google Scholar] [CrossRef] [PubMed]
- Balboa, E.M.; Rivas, S.; Moure, A.; Domínguez, H.; Parajó, J.C. Simultaneous Extraction and Depolymerization of Fucoidan from Sargassum Muticum in Aqueous Media. Mar. Drugs 2013, 11, 4612–4627. [Google Scholar] [CrossRef] [PubMed]
- Ganapathy, S.; Lingappa, S.; Naidu, K.; Selvaraj, U.; Ramachandiran, S.; Ponnusamy, S.; Somasundaram, S.T. Isolation and Bioactive Potential of Fucoidan from Marine Macroalgae Turbinaria Conoides. ChemistrySelect 2019, 4, 14114–14119. [Google Scholar] [CrossRef]
- Soto-Vásquez, M.R.; Alvarado-García, P.A.A.; Youssef, F.S.; Ashour, M.L.; Bogari, H.A.; Elhady, S.S. FTIR Characterization of Sulfated Polysaccharides Obtained from Macrocystis Integrifolia Algae and Verification of Their Antiangiogenic and Immunomodulatory Potency In Vitro and In Vivo. Mar. Drugs 2022, 21, 36. [Google Scholar] [CrossRef]
- Lesco, K.C.; Williams, S.K.R.; Laurens, L.M.L. Marine Algae Polysaccharides: An Overview of Characterization Techniques for Structural and Molecular Elucidation. Mar. Drugs 2025, 23, 105. [Google Scholar] [CrossRef]
- Rhein-Knudsen, N.; Reyes-Weiss, D.; Horn, S.J. Extraction of High Purity Fucoidans from Brown Seaweeds Using Cellulases and Alginate Lyases. Int. J. Biol. Macromol. 2023, 229, 199–209. [Google Scholar] [CrossRef]
- Amin, M.N.G.; Rosenau, T.; Böhmdorfer, S. The Structure of Fucoidan by Linkage Analysis Tailored for Fucose in Four Algae Species: Fucus Serratus, Fucus Evanescens, Fucus Vesiculosus and Laminaria Hyperborea. Carbohydr. Polym. Technol. Appl. 2024, 7, 100455. [Google Scholar] [CrossRef]
- Li, B.; Lu, F.; Wei, X.; Zhao, R. Fucoidan: Structure and Bioactivity. Molecules 2008, 13, 1671–1695. [Google Scholar] [CrossRef] [PubMed]
- Tagliapietra, B.L.; Clerici, M.T.P.S. Brown Algae and Their Multiple Applications as Functional Ingredient in Food Production. Food Res. Int. 2023, 167, 112655. [Google Scholar] [CrossRef]
- Song, S.H.; Vieille, C. Recent Advances in the Biological Production of Mannitol. Appl. Microbiol. Biotechnol. 2009, 84, 55–62. [Google Scholar] [CrossRef]
- Madsen, M.; Westh, P.; Khan, S.; Ipsen, R.; Almdal, K.; Aachmann, F.L.; Svensson, B. Impact of Alginate Mannuronic-Guluronic Acid Contents and PH on Protein Binding Capacity and Complex Size. Biomacromolecules 2021, 22, 649–660. [Google Scholar] [CrossRef]
- Neupane, S.; Bittkau, K.S.; Alban, S. Size Distribution and Chain Conformation of Six Different Fucoidans Using Size-Exclusion Chromatography with Multiple Detection. J. Chromatogr. A 2020, 1612, 460658. [Google Scholar] [CrossRef]
- Dörschmann, P.; Kopplin, G.; Thalenhorst, T.; Seeba, C.; Ullah, S.F.; Srivastava, V.; Roider, J.; Klettner, A. Influence of a Very High-Molecular Weight Fucoidan from Laminaria Hyperborea on Age-Related Macular Degeneration-Relevant Pathomechanisms in Ocular Cell Models. Mar. Drugs 2025, 23, 101. [Google Scholar] [CrossRef]
- Kopplin, G.; Rokstad, A.M.; Mélida, H.; Bulone, V.; Skjåk-Bræk, G.; Aachmann, F.L. Structural Characterization of Fucoidan from Laminaria Hyperborea: Assessment of Coagulation and Inflammatory Properties and Their Structure–Function Relationship. ACS Appl. Bio Mater. 2018, 1, 1880–1892. [Google Scholar] [CrossRef]
- Atashrazm, F.; Lowenthal, R.; Woods, G.; Holloway, A.; Dickinson, J. Fucoidan and Cancer: A Multifunctional Molecule with Anti-Tumor Potential. Mar. Drugs 2015, 13, 2327–2346. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, C.; Ferreira, A.S.; Novoa-Carballal, R.; Nunes, C.; Pashkuleva, I.; Neves, N.M.; Coimbra, M.A.; Reis, R.L.; Martins, A.; Silva, T.H. The Key Role of Sulfation and Branching on Fucoidan Antitumor Activity. Macromol. Biosci. 2017, 17, 1600340. [Google Scholar] [CrossRef] [PubMed]
- Cotas, J.; Leandro, A.; Monteiro, P.; Pacheco, D.; Figueirinha, A.; Gonçalves, A.M.M.; Da Silva, G.J.; Pereira, L. Seaweed Phenolics: From Extraction to Applications. Mar. Drugs 2020, 18, 384. [Google Scholar] [CrossRef]
- Audibert, L.; Fauchon, M.; Blanc, N.; Hauchard, D.; Ar Gall, E. Phenolic Compounds in the Brown Seaweed Ascophyllum Nodosum: Distribution and Radical-Scavenging Activities. Phytochem. Anal. 2010, 21, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Jayawardena, T.U.; Nagahawatta, D.P.; Fernando, I.P.S.; Kim, Y.-T.; Kim, J.-S.; Kim, W.-S.; Lee, J.S.; Jeon, Y.-J. A Review on Fucoidan Structure, Extraction Techniques, and Its Role as an Immunomodulatory Agent. Mar. Drugs 2022, 20, 755. [Google Scholar] [CrossRef]
- Pozharitskaya, O.N.; Obluchinskaya, E.D.; Shikov, A.N. Mechanisms of Bioactivities of Fucoidan from the Brown Seaweed Fucus Vesiculosus L. of the Barents Sea. Mar. Drugs 2020, 18, 275. [Google Scholar] [CrossRef]
- Lee, Z.J.; Xie, C.; Ng, K.; Suleria, H.A.R. Study of Phenolic-Polysaccharide Interactions in Brown Seaweed. Food Chem. 2025, 477, 143494. [Google Scholar] [CrossRef] [PubMed]
- Zayed, A.; Hahn, T.; Finkelmeier, D.; Burger-Kentischer, A.; Rupp, S.; Krämer, R.; Ulber, R. Phenomenological Investigation of the Cytotoxic Activity of Fucoidan Isolated from Fucus Vesiculosus. Process Biochem. 2019, 81, 182–187. [Google Scholar] [CrossRef]
- Obluchinskaya, E.D.; Pozharitskaya, O.N.; Shikov, A.N. In Vitro Anti-Inflammatory Activities of Fucoidans from Five Species of Brown Seaweeds. Mar. Drugs 2022, 20, 606. [Google Scholar] [CrossRef] [PubMed]
- Meng, W.; Sun, H.; Mu, T.; Garcia-Vaquero, M. Spray-Drying and Rehydration on β-Carotene Encapsulated Pickering Emulsion with Chitosan and Seaweed Polyphenol. Int. J. Biol. Macromol. 2024, 268, 131654. [Google Scholar] [CrossRef]
- Song, S.; Peng, H.; Wang, Q.; Liu, Z.; Dong, X.; Wen, C.; Ai, C.; Zhang, Y.; Wang, Z.; Zhu, B. Inhibitory Activities of Marine Sulfated Polysaccharides against SARS-CoV-2. Food Funct. 2020, 11, 7415–7420. [Google Scholar] [CrossRef]
- Dinesh, S.; Menon, T.; Hanna, L.E.; Suresh, V.; Sathuvan, M.; Manikannan, M. In Vitro Anti-HIV-1 Activity of Fucoidan from Sargassum Swartzii. Int. J. Biol. Macromol. 2016, 82, 83–88. [Google Scholar] [CrossRef]
- Elizondo-Gonzalez, R.; Cruz-Suarez, L.E.; Ricque-Marie, D.; Mendoza-Gamboa, E.; Rodriguez-Padilla, C.; Trejo-Avila, L.M. In Vitro Characterization of the Antiviral Activity of Fucoidan from Cladosiphon Okamuranus against Newcastle Disease Virus. Virol. J. 2012, 9, 307. [Google Scholar] [CrossRef]
- Sun, T.; Zhang, X.; Miao, Y.; Zhou, Y.; Shi, J.; Yan, M.; Chen, A. Studies on Antiviral and Immuno-Regulation Activity of Low Molecular Weight Fucoidan from Laminaria Japonica. J. Ocean Univ. China 2018, 17, 705–711. [Google Scholar] [CrossRef]
- Taoda, N.; Shinji, E.; Nishii, K.; Nishioka, S.; Yonezawa, Y.; Uematsu, J.; Hattori, E.; Yamamoto, H.; Kawano, M.; Tsurudome, M.; et al. Fucoidan Inhibits Parainfluenza Virus Type 2 Infection to LLCMK2 Cells. Biomed. Res. 2008, 29, 331–334. [Google Scholar] [CrossRef]
- Epstein, J.H.; Field, H.E.; Luby, S.; Pulliam, J.R.C.; Daszak, P. Nipah Virus: Impact, Origins, and Causes of Emergence. Curr. Infect. Dis. Rep. 2006, 8, 59–65. [Google Scholar] [CrossRef] [PubMed]
- WHO Prioritizing Diseases for Research and Development in Emergency Contexts. Available online: https://www.who.int/activities/prioritizing-diseases-for-research-and-development-in-emergency-contexts (accessed on 8 April 2025).
- Sun, Y.-Q.; Zhang, Y.-Y.; Liu, M.-C.; Chen, J.-J.; Li, T.-T.; Liu, Y.-N.; Zhang, L.-Y.; Wang, T.; Yu, L.-J.; Che, T.-L.; et al. Mapping the Distribution of Nipah Virus Infections: A Geospatial Modelling Analysis. Lancet Planet. Health 2024, 8, e463–e475. [Google Scholar] [CrossRef]
- Vasudevan, S.S.; Subash, A.; Mehta, F.; Kandrikar, T.Y.; Desai, R.; Khan, K.; Khanduja, S.; Pitliya, A.; Raavi, L.; Kanagala, S.G.; et al. Global and Regional Mortality Statistics of Nipah Virus from 1994 to 2023: A Comprehensive Systematic Review and Meta-Analysis. Pathog. Glob. Health 2024, 118, 471–480. [Google Scholar] [CrossRef]
- Aguilar, H.C.; Matreyek, K.A.; Filone, C.M.; Hashimi, S.T.; Levroney, E.L.; Negrete, O.A.; Bertolotti-Ciarlet, A.; Choi, D.Y.; McHardy, I.; Fulcher, J.A.; et al. N-Glycans on Nipah Virus Fusion Protein Protect against Neutralization but Reduce Membrane Fusion and Viral Entry. J. Virol. 2006, 80, 4878–4889. [Google Scholar] [CrossRef]
- Cagno, V.; Tseligka, E.D.; Jones, S.T.; Tapparel, C. Heparan Sulfate Proteoglycans and Viral Attachment: True Receptors or Adaptation Bias? Viruses 2019, 11, 596. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Kar, S. Are We Ready to Fight the Nipah Virus Pandemic? An Overview of Drug Targets, Current Medications, and Potential Leads. Struct. Chem. 2023, 34, 2119–2137. [Google Scholar] [CrossRef]
- Mak, N.S.C.; Liu, J.; Zhang, D.; Taylor, J.; Li, X.; Rahman, K.; Chen, F.; Datta, S.A.K.; Lai, K.K.; Shi, Z.; et al. Alternative Splicing Expands the Antiviral IFITM Repertoire in Chinese Rufous Horseshoe Bats. PLoS Pathog. 2024, 20, e1012763. [Google Scholar] [CrossRef] [PubMed]
- Cantoni, D.; Wilkie, C.; Bentley, E.M.; Mayora-Neto, M.; Wright, E.; Scott, S.; Ray, S.; Castillo-Olivares, J.; Heeney, J.L.; Mattiuzzo, G.; et al. Correlation between Pseudotyped Virus and Authentic Virus Neutralisation Assays, a Systematic Review and Meta-Analysis of the Literature. Front. Immunol. 2023, 14. [Google Scholar] [CrossRef]
- Rizatdinova, S.N.; Ershova, A.E.; Astrakhantseva, I.V. Pseudotyped Viruses: A Useful Platform for Pre-Clinical Studies Conducted in a BSL-2 Laboratory Setting. Biomolecules 2025, 15, 135. [Google Scholar] [CrossRef] [PubMed]
- Bentley, E.M.; Mather, S.T.; Temperton, N.J. The Use of Pseudotypes to Study Viruses, Virus Sero-Epidemiology and Vaccination. Vaccine 2015, 33, 2955–2962. [Google Scholar] [CrossRef] [PubMed]
- Andreu, S.; von Kobbe, C.; Delgado, P.; Ripa, I.; Buzón, M.J.; Genescà, M.; Gironès, N.; del Moral-Salmoral, J.; Ramírez, G.A.; Zúñiga, S.; et al. Dextran Sulfate from Leuconostoc Mesenteroides B512F Exerts Potent Antiviral Activity against SARS-CoV-2 in Vitro and in Vivo. Front. Microbiol. 2023, 14. [Google Scholar] [CrossRef]
- Yang, C.-W.; Hsu, H.-Y.; Lee, Y.-Z.; Jan, J.-T.; Chang, S.-Y.; Lin, Y.-L.; Yang, R.-B.; Chao, T.-L.; Liang, J.-J.; Lin, S.-J.; et al. Natural Fucoidans Inhibit Coronaviruses by Targeting Viral Spike Protein and Host Cell Furin. Biochem. Pharmacol. 2023, 215, 115688. [Google Scholar] [CrossRef]
- Essalmani, R.; Jain, J.; Susan-Resiga, D.; Andréo, U.; Evagelidis, A.; Derbali, R.M.; Huynh, D.N.; Dallaire, F.; Laporte, M.; Delpal, A.; et al. Distinctive Roles of Furin and TMPRSS2 in SARS-CoV-2 Infectivity. J. Virol. 2022, 96, e0012822. [Google Scholar] [CrossRef]
- Kwon, P.S.; Oh, H.; Kwon, S.-J.; Jin, W.; Zhang, F.; Fraser, K.; Hong, J.J.; Linhardt, R.J.; Dordick, J.S. Sulfated Polysaccharides Effectively Inhibit SARS-CoV-2 in Vitro. Cell Discov. 2020, 6, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Sloan, R.D.; Kuhl, B.D.; Mesplède, T.; Münch, J.; Donahue, D.A.; Wainberg, M.A. Productive Entry of HIV-1 during Cell-to-Cell Transmission via Dynamin-Dependent Endocytosis. J. Virol. 2013, 87, 8110–8123. [Google Scholar] [CrossRef]
- Denning, D.W. Global Incidence and Mortality of Severe Fungal Disease. Lancet Infect. Dis. 2024, 24, e428–e438. [Google Scholar] [CrossRef]
- Fisher, M.C.; Alastruey-Izquierdo, A.; Berman, J.; Bicanic, T.; Bignell, E.M.; Bowyer, P.; Bromley, M.; Brüggemann, R.; Garber, G.; Cornely, O.A.; et al. Tackling the Emerging Threat of Antifungal Resistance to Human Health. Nat. Rev. Microbiol. 2022, 20, 557–571. [Google Scholar] [CrossRef]
- Jackson, K.M.; Ding, M.; Nielsen, K. Importance of Clinical Isolates in Cryptococcus Neoformans Research. J. Fungi 2023, 9, 364. [Google Scholar] [CrossRef]
- Rajasingham, R.; Smith, R.M.; Park, B.J.; Jarvis, J.N.; Govender, N.P.; Chiller, T.M.; Denning, D.W.; Loyse, A.; Boulware, D.R. Global Burden of Disease of HIV-Associated Cryptococcal Meningitis: An Updated Analysis. Lancet Infect. Dis. 2017, 17, 873–881. [Google Scholar] [CrossRef]
- Turner, S.A.; Butler, G. The Candida Pathogenic Species Complex. Cold Spring Harb. Perspect. Med. 2014, 4, a019778. [Google Scholar] [CrossRef]
- Soriano, A.; Honore, P.M.; Puerta-Alcalde, P.; Garcia-Vidal, C.; Pagotto, A.; Gonçalves-Bradley, D.C.; Verweij, P.E. Invasive Candidiasis: Current Clinical Challenges and Unmet Needs in Adult Populations. J. Antimicrob. Chemother. 2023, 78, 1569–1585. [Google Scholar] [CrossRef]
- Oka, S.; Okabe, M.; Tsubura, S.; Mikami, M.; Imai, A. Properties of Fucoidans Beneficial to Oral Healthcare. Odontology 2020, 108, 34–42. [Google Scholar] [CrossRef]
- Ventola, C.L. The Antibiotic Resistance Crisis. P T 2015, 40, 277–283. [Google Scholar] [PubMed]
- Murray, C.J.L.; Ikuta, K.S.; Sharara, F.; Swetschinski, L.; Aguilar, G.R.; Gray, A.; Han, C.; Bisignano, C.; Rao, P.; Wool, E.; et al. Global Burden of Bacterial Antimicrobial Resistance in 2019: A Systematic Analysis. The Lancet 2022, 399, 629–655. [Google Scholar] [CrossRef]
- Lam, M.M.C.; Wick, R.R.; Watts, S.C.; Cerdeira, L.T.; Wyres, K.L.; Holt, K.E. A Genomic Surveillance Framework and Genotyping Tool for Klebsiella Pneumoniae and Its Related Species Complex. Nat. Commun. 2021, 12, 4188. [Google Scholar] [CrossRef] [PubMed]
- WHO Joint FAO/WHO Expert Consultation on Evaluation of Health and Nutritional Properties of Probiotics in Food Including Powder Milk with Live Lactic Acid Bacteria. Available online: https://openknowledge.fao.org/items/db384295-64d9-47e2-b65b-3c918efc5140 (accessed on 15 April 2025).
- Kechagia, M.; Basoulis, D.; Konstantopoulou, S.; Dimitriadi, D.; Gyftopoulou, K.; Skarmoutsou, N.; Fakiri, E.M. Health Benefits of Probiotics: A Review. ISRN Nutr. 2013, 2013, 481651. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Tran, D.Q.; Rhoads, J.M. Probiotics in Disease Prevention and Treatment. J. Clin. Pharmacol. 2018, 58, S164–S179. [Google Scholar] [CrossRef]
- Cristofori, F.; Dargenio, V.N.; Dargenio, C.; Miniello, V.L.; Barone, M.; Francavilla, R. Anti-Inflammatory and Immunomodulatory Effects of Probiotics in Gut Inflammation: A Door to the Body. Front Immunol. 2021, 12, 578386. [Google Scholar] [CrossRef]
- Davani-Davari, D.; Negahdaripour, M.; Karimzadeh, I.; Seifan, M.; Mohkam, M.; Masoumi, S.J.; Berenjian, A.; Ghasemi, Y. Prebiotics: Definition, Types, Sources, Mechanisms, and Clinical Applications. Foods 2019, 8, 92. [Google Scholar] [CrossRef] [PubMed]
- Habibi, M.; Golmakani, M.-T.; Eskandari, M.H.; Hosseini, S.M.H. Potential Prebiotic and Antibacterial Activities of Fucoidan from Laminaria Japonica. Int. J. Biol. Macromol. 2024, 268, 131776. [Google Scholar] [CrossRef]
- Ricós-Muñoz, N.; Maicas, S.; Pina-Pérez, M.C. Probiotic Lactobacillus Reuteri Growth Improved under Fucoidan Exposure. In Proceedings of the The 1st International Electronic Conference on Food Science and Functional Foods, MDPI, November 10 2020; p. 106. [Google Scholar]
- Zhou, X.; Zhang, Y.; Wei, L.; Yang, Y.; Wang, B.; Liu, C.; Bai, J.; Wang, C. In Vitro Fermentation Characteristics of Fucoidan and Its Regulatory Effects on Human Gut Microbiota and Metabolites. Food Chem. 2025, 465, 141998. [Google Scholar] [CrossRef]
- Jones, R.M. The Use of Lactobacillus Casei and Lactobacillus Paracasei in Clinical Trials for the Improvement of Human Health. In The Microbiota in Gastrointestinal Pathophysiology; Floch, M.H., Ringel, Y., Allan Walker, W., Eds.; Academic Press: Boston, 2017; pp. 99–108. ISBN 978-0-12-804024-9. [Google Scholar]
- Guerin-Danan, C.; Chabanet, C.; Pedone, C.; Popot, F.; Vaissade, P.; Bouley, C.; Szylit, O.; Andrieux, C. Milk Fermented with Yogurt Cultures and Lactobacillus Casei Compared with Yogurt and Gelled Milk: Influence on Intestinal Microflora in Healthy Infants. Am. J. Clin. Nutr. 1998, 67, 111–117. [Google Scholar] [CrossRef]
- Aktas, B.; Wolfe, T.J.D.; Safdar, N.; Darien, B.J.; Steele, J.L. The Impact of Lactobacillus Casei on the Composition of the Cecal Microbiota and Innate Immune System Is Strain Specific. PLoS ONE 2016, 11, e0156374. [Google Scholar] [CrossRef]
- Rizzardini, G.; Eskesen, D.; Calder, P.C.; Capetti, A.; Jespersen, L.; Clerici, M. Evaluation of the Immune Benefits of Two Probiotic Strains Bifidobacterium Animalis Ssp. Lactis, BB-12® and Lactobacillus Paracasei Ssp. Paracasei, L. Casei 431® in an Influenza Vaccination Model: A Randomised, Double-Blind, Placebo-Controlled Study. Br. J. Nutr. 2012, 107, 876–884. [Google Scholar] [CrossRef]
- Villena, J.; Racedo, S.; Agüero, G.; Bru, E.; Medina, M.; Alvarez, S. Lactobacillus Casei Improves Resistance to Pneumococcal Respiratory Infection in Malnourished Mice. J. Nutr. 2005, 135, 1462–1469. [Google Scholar] [CrossRef]
- Yang, C.; Dwan, C.; Wimmer, B.C.; Wilson, R.; Johnson, L.; Caruso, V. Fucoidan from Undaria Pinnatifida Enhances Exercise Performance and Increases the Abundance of Beneficial Gut Bacteria in Mice. Mar. Drugs 2024, 22, 485. [Google Scholar] [CrossRef] [PubMed]
- Tucey, T.M.; Verma, J.; Harrison, P.F.; Snelgrove, S.L.; Lo, T.L.; Scherer, A.K.; Barugahare, A.A.; Powell, D.R.; Wheeler, R.T.; Hickey, M.J.; et al. Glucose Homeostasis Is Important for Immune Cell Viability during Candida Challenge and Host Survival of Systemic Fungal Infection. Cell Metab. 2018, 27, 988–1006.e7. [Google Scholar] [CrossRef]
- Kondybayev, A.; Konuspayeva, G.; Strub, C.; Loiseau, G.; Mestres, C.; Grabulos, J.; Manzano, M.; Akhmetsadykova, S.; Achir, N. Growth and Metabolism of Lacticaseibacillus Casei and Lactobacillus Kefiri Isolated from Qymyz, a Traditional Fermented Central Asian Beverage. Fermentation 2022, 8, 367. [Google Scholar] [CrossRef]
- Geun Lee, H.; Jayawardena, T.U.; Liyanage, N.M.; Song, K.-M.; Choi, Y.-S.; Jeon, Y.-J.; Kang, M.-C. Antioxidant Potential of Low Molecular Weight Fucoidans from Sargassum Autumnale against H2O2-Induced Oxidative Stress in Vitro and in Zebrafish Models Based on Molecular Weight Changes. Food Chem. 2022, 384, 132591. [Google Scholar] [CrossRef]
- Gunarathne, R.; Zhang, C.; Zhou, J.; Xin, X.; Lu, J. Low Molecular Weight Fucoidan: Production Methods and Their Therapeutic Applications. Int. J. Biol. Macromol. 2025, 318, 145309. [Google Scholar] [CrossRef]
- Krylova, N.V.; Silchenko, A.S.; Pott, A.B.; Ermakova, S.P.; Iunikhina, O.V.; Rasin, A.B.; Kompanets, G.G.; Likhatskaya, G.N.; Shchelkanov, M.Y. In Vitro Anti-Orthohantavirus Activity of the High-and Low-Molecular-Weight Fractions of Fucoidan from the Brown Alga Fucus Evanescens. Mar. Drugs 2021, 19, 577. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Ai, C.; Wen, C.; Peng, H.; Yang, J.; Cui, Y.; Song, S. Inhibitory Effects of Fucoidan from Laminaria Japonica against Some Pathogenic Bacteria and SARS-CoV-2 Depend on Its Large Molecular Weight. Int. J. Biol. Macromol. 2023, 229, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Xia, Q.; Li, L.; Shi, Y.; Gao, Y.; Ma, Y.; Liu, S.; He, Y.; Wang, Q.; Ren, D. Absorption Patterns of Fucoidan Oligosaccharides from Kjellmaniella Crassifolia in the Caco-2 Monolayer Cell Model and Their Pharmacokinetics in Mice. Foods 2025, 14, 1486. [Google Scholar] [CrossRef]
- Tan, J.; Song, Y.; Wang, J.; Wu, N.; Yue, Y.; Zhang, Q. Pharmacokinetics of Fucoidan and Low Molecular Weight Fucoidan from Saccharina Japonica after Oral Administration to Mice. J. Ocean. Limnol. 2023, 41, 1900–1909. [Google Scholar] [CrossRef]
- Sun, X.; Yang, Y.; Song, C.; Ai, C.; Yang, J.; Song, S. Degradation of Low-Molecular-Weight Fucoidans by Human Intestinal Microbiota and Their Regulation Effect on Intestinal Microbiota and Metabolites during in Vitro Fermentation. Food Biosci. 2024, 62, 105287. [Google Scholar] [CrossRef]
- Mandal, P.; Mateu, C.G.; Chattopadhyay, K.; Pujol, C.A.; Damonte, E.B.; Ray, B. Structural Features and Antiviral Activity of Sulphated Fucans from the Brown Seaweed Cystoseira Indica. Antivir. Chem. Chemother. 2007, 18, 153–162. [Google Scholar] [CrossRef]
- Wang, W.; Wu, J.; Zhang, X.; Hao, C.; Zhao, X.; Jiao, G.; Shan, X.; Tai, W.; Yu, G. Inhibition of Influenza A Virus Infection by Fucoidan Targeting Viral Neuraminidase and Cellular EGFR Pathway. Sci. Rep. 2017, 7, 40760. [Google Scholar] [CrossRef]
- Wang, L.; Oh, J.-Y.; Yang, H.-W.; Hyun, J.; Ahn, G.; Fu, X.; Xu, J.; Gao, X.; Cha, S.-H.; Jeon, Y.-J. Protective Effect of Sargassum Fusiforme Fucoidan against Ethanol-Induced Oxidative Damage in In Vitro and In Vivo Models. Polymers 2023, 15, 1912. [Google Scholar] [CrossRef]
- Sepúlveda-Crespo, D.; Ceña-Díez, R.; Jiménez, J.L.; Ángeles Muñoz-Fernández, M. Mechanistic Studies of Viral Entry: An Overview of Dendrimer-Based Microbicides As Entry Inhibitors Against Both HIV and HSV-2 Overlapped Infections. Med. Res. Rev. 2017, 37, 149–179. [Google Scholar] [CrossRef] [PubMed]
- Inglese, J.; Johnson, R.L.; Simeonov, A.; Xia, M.; Zheng, W.; Austin, C.P.; Auld, D.S. High-Throughput Screening Assays for the Identification of Chemical Probes. Nat. Chem. Biol. 2007, 3, 466–479. [Google Scholar] [CrossRef]
- Sichert, A.; Le Gall, S.; Klau, L.J.; Laillet, B.; Rogniaux, H.; Aachmann, F.L.; Hehemann, J.-H. Ion-Exchange Purification and Structural Characterization of Five Sulfated Fucoidans from Brown Algae. Glycobiology 2021, 31, 352–357. [Google Scholar] [CrossRef] [PubMed]
- Saravana, P.S.; Karuppusamy, S.; Rai, D.K.; Wanigasekara, J.; Curtin, J.; Tiwari, B.K. Elimination of Ethanol for the Production of Fucoidans from Brown Seaweeds: Characterization and Bioactivities. Mar. Drugs 2024, 22, 493. [Google Scholar] [CrossRef]
- Marinval, N.; Saboural, P.; Haddad, O.; Maire, M.; Bassand, K.; Geinguenaud, F.; Djaker, N.; Ben Akrout, K.; Lamy de la Chapelle, M.; Robert, R.; et al. Identification of a Pro-Angiogenic Potential and Cellular Uptake Mechanism of a LMW Highly Sulfated Fraction of Fucoidan from Ascophyllum Nodosum. Mar. Drugs 2016, 14, 185. [Google Scholar] [CrossRef]
- Colorado-Ríos, J.; Restrepo-Espinosa, D.C.; Restrepo-Moná, Y.; Monsalve, J.D.; Márquez-Fernández, D.M.; Castellanos, L.; Martínez-Martínez, A. Chemical Exploration of Polysaccharides, Fatty Acids, and Antioxidants as Functional Ingredients from Colombian Macroalgae Acanthophora Spicifera, Sargassum Ramifolium, and Sargassum Fluitans. Molecules 2025, 30, 3333. [Google Scholar] [CrossRef]
- Shen, C.; Mackeigan, D.T.; Shoara, A.A.; Xu, R.; Bhoria, P.; Karakas, D.; Ma, W.; Cerenzia, E.; Chen, Z.; Hoard, B.; et al. Dual Roles of Fucoidan-GPIbα Interaction in Thrombosis and Hemostasis: Implications for Drug Development Targeting GPIbα. J. Thromb. Haemost. 2023, 21, 1274–1288. [Google Scholar] [CrossRef]
- Tong, C.; Liang, Y.; Zhang, Z.; Wang, S.; Zheng, X.; Liu, Q.; Song, B. Review of Knockout Technology Approaches in Bacterial Drug Resistance Research. PeerJ 2023, 11, e15790. [Google Scholar] [CrossRef]
- Ponce, N.M.A.; Stortz, C.A. A Comprehensive and Comparative Analysis of the Fucoidan Compositional Data Across the Phaeophyceae. Front. Plant Sci. 2020, 11, 556312. [Google Scholar] [CrossRef] [PubMed]
- Ptak, S.H.; Hjuler, A.L.; Ditlevsen, S.I.; Fretté, X.; Errico, M.; Christensen, K.V. The Effect of Seasonality and Geographic Location on Sulphated Polysaccharides from Brown Algae. Aquac. Res. 2021, 52, 6235–6243. [Google Scholar] [CrossRef]
- APExBIO Technology LLC. Fucoidan. Available online: https://www.apexbt.com/fucoidan.html (accessed on 22 April 2025).
- Shandong Jiejing Group Fucoidan_Seafood_Product_Shandong Jiejing Group. Available online: http://en.china-jiejing.com/index.php?c=show&id=23 (accessed on 22 April 2025).
- MarkNature Marine Sulfated Polysaccharide Fucoidan: Nutraceutical & Cosmeceutical Powerhouse. Available online: https://www.marknature.com/products/fucoidan-sulfated-polysaccharide (accessed on 22 April 2025).
- Marinova Pty Ltd. Maritech® Undaria Pinnatifida Extract | Product Portfolio. Available online: https://maritechfucoidan.com.au/maritech-undaria-pinnatifida-extract/ (accessed on 22 April 2025).
- Rajauria, G.; Ravindran, R.; Garcia-Vaquero, M.; Rai, D.K.; Sweeney, T.; O’Doherty, J. Purification and Molecular Characterization of Fucoidan Isolated from Ascophyllum Nodosum Brown Seaweed Grown in Ireland. Mar. Drugs 2023, 21, 315. [Google Scholar] [CrossRef]
- Vandanjon, L.; Burlot, A.-S.; Zamanileha, E.F.; Douzenel, P.; Ravelonandro, P.H.; Bourgougnon, N.; Bedoux, G. The Use of FTIR Spectroscopy as a Tool for the Seasonal Variation Analysis and for the Quality Control of Polysaccharides from Seaweeds. Mar. Drugs 2023, 21, 482. [Google Scholar] [CrossRef] [PubMed]
- Ptak, S.H.; Sanchez, L.; Fretté, X.; Kurouski, D. Complementarity of Raman and Infrared Spectroscopy for Rapid Characterization of Fucoidan Extracts. Plant Methods 2021, 17, 130. [Google Scholar] [CrossRef] [PubMed]
- Apostolova, E.; Lukova, P.; Baldzhieva, A.; Delattre, C.; Molinié, R.; Petit, E.; Elboutachfaiti, R.; Nikolova, M.; Iliev, I.; Murdjeva, M.; et al. Structural Characterization and In Vivo Anti-Inflammatory Activity of Fucoidan from Cystoseira Crinita (Desf.) Borry. Mar. Drugs 2022, 20, 714. [Google Scholar] [CrossRef]
- de Falco, B.; Fiore, A.; Bochicchio, R.; Amato, M.; Lanzotti, V. Metabolomic Analysis by UAE-GC MS and Antioxidant Activity of Salvia Hispanica (L.) Seeds Grown under Different Irrigation Regimes. Ind. Crops Prod. 2018, 112, 584–592. [Google Scholar] [CrossRef]
- Casper, J.; Schenk, S.H.; Parhizkar, E.; Detampel, P.; Dehshahri, A.; Huwyler, J. Polyethylenimine (PEI) in Gene Therapy: Current Status and Clinical Applications. J. Control. Release 2023, 362, 667–691. [Google Scholar] [CrossRef]
- Kuete, V.; Karaosmanoğlu, O.; Sivas, H. Anticancer Activities of African Medicinal Spices and Vegetables. In Medicinal Spices and Vegetables from Africa; Elsevier, 2017; pp. 271–297. ISBN 978-0-12-809286-6. [Google Scholar]
- Lescat, M.; Poirel, L.; Tinguely, C.; Nordmann, P. A Resazurin Reduction-Based Assay for Rapid Detection of Polymyxin Resistance in Acinetobacter Baumannii and Pseudomonas Aeruginosa. J. Clin. Microbiol. 2019, 57, e01563-18. [Google Scholar] [CrossRef] [PubMed]
- Fookes, M.; Yu, J.; De Majumdar, S.; Thomson, N.; Schneiders, T. Genome Sequence of Klebsiella Pneumoniae Ecl8, a Reference Strain for Targeted Genetic Manipulation. Genome Announc 2013, 1, e00027-12. [Google Scholar] [CrossRef]
- Majumdar, S.D.; Yu, J.; Fookes, M.; McAteer, S.P.; Llobet, E.; Finn, S.; Spence, S.; Monaghan, A.; Kissenpfennig, A.; Ingram, R.J.; et al. Elucidation of the RamA Regulon in Klebsiella Pneumoniae Reveals a Role in LPS Regulation. PLoS Pathog. 2015, 11, e1004627. [Google Scholar] [CrossRef]
| Sample | Shandong | Mark Nature | Marinova | ApexBio | |
|---|---|---|---|---|---|
| Monosaccharide Content | Fucose (%) | 38.04±2.40a | 8.62±2.29b | 39.50±5.88a | 0.00±0.00b |
| Glucose (%) | 5.80±0.41b | 69.00±3.92a | 2.20±0.57bc | 0.00±0.00c | |
| Mannitol (%) | 0.42±0.00b | 0.00±0.00b | 0.00±0.02b | 100.00±2.60a | |
| Arabinose/Rhamnose (%) | 2.80±0.00a | 0.46±0.10b | 0.00±0.50b | 0.00±0.00b | |
| Uronic acid Content |
Glucuronic Acid (%) | 24.08±0.57a | 2.21±0.76b | 3.63±2.74b | 0.00±0.00b |
| Guluronic Acid (%) | 0.00±0.15b | 1.19±0.31a | 0.14±0.00b | 0.00±0.00b | |
| Mannuronic Acid (%) | 1.10±0.46c | 12.92±2.14a | 7.43±0.57b | 0.00±0.00c | |
| Extract Properties | Mn (kDa)† | 22.31±0.08 | 2.30±0.08 | 8.88±0.16 | 1.61±0.05 |
| Mw (kDa)‡ | 49.46±0.33 | 36.54±0.25 | 24.91±0.23 | 9.65±0.09 | |
| Polydispersity (-) | 2.22±0.02 | 15.87±0.55 | 2.81±0.06 | 6.00±0.18 | |
| Sulfation Degree (-) | 0.86±0.16a | 0.13±0.01b | 0.88±0.01a | 0.01±0.00c* | |
| Extract Impurities | Protein Content (%) | 5.10±0.42a | 1.51±0.06c | 3.32±0.03b | 1.10±0.03c |
| Polyphenol Content (mgGAE gsample-1)§ |
0.70±0.41b | 0.36±0.65b | 7.11±1.04a | 1.68±0.10b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




