Submitted:
27 March 2026
Posted:
31 March 2026
You are already at the latest version
Abstract
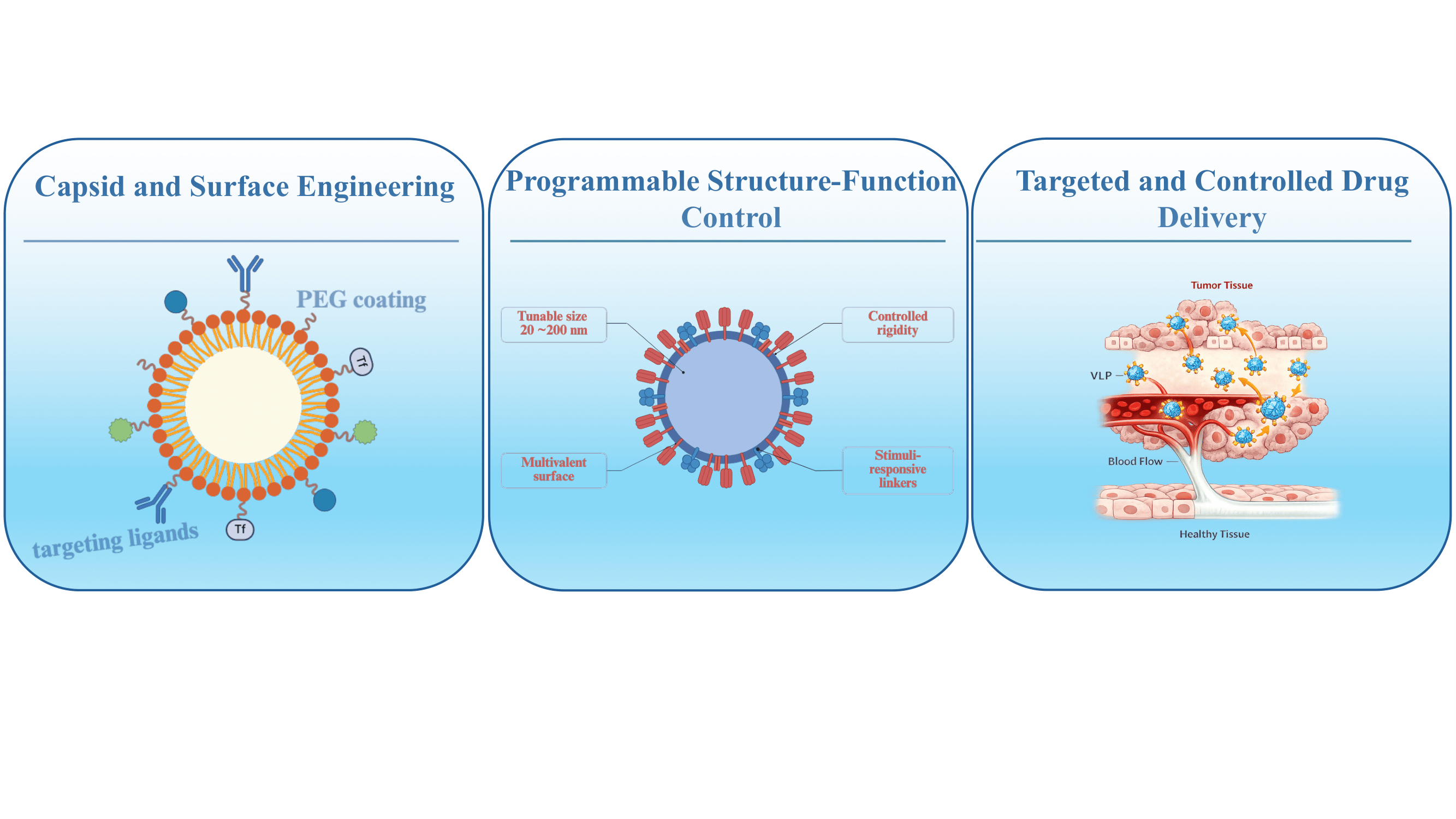
Keywords:
1. Context and Unmet Needs in Cancer Treatment
2. Architectural and Functional Basis
3. Engineering VLPs for Drug Delivery
3.1. Assembly-Driven Modifications and Cargo Loading
3.2. Surface Functionalization Strategies
4. Current Progress in Chemotherapy Delivery
5. Challenges and Future Directions
6. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CCMV | Cowpea chlorotic mottle virus |
| CED | Convection-enhanced delivery |
| CP | Capsid protein |
| CPMV | Cowpea mosaic virus |
| CPP | Cell penetrating peptide |
| CuAAC | Copper-catalyzed azide–alkyne cycloaddition |
| DDS | Drug delivery system |
| Dec | Decoration protein |
| DOX | Doxorubicin |
| EDC | 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide |
| EGFR | Epithelial growth factor receptor |
| EMCH | 6-maleimidocaproyl-hydrazone |
| EPI | Epirubicin |
| EPT | Ellipticin |
| FA | Folic acid |
| FHV | Flock house virus |
| GFP | Green fluorescent protein |
| HBcAg | Hepatitis B virus core antigen |
| HER2 | Human epidermal growth factor 2 |
| HIV-1 | Human immunodeficiency virus 1 |
| HPV | Human papillomavirus |
| HSPG | Heparan sulfate proteoglycan |
| ICD | Immunogenic cell death |
| MMAE | Monomethyl auristatin |
| NHS | N-hydroxysulfosuccinimide |
| NIR | Near-infrared light |
| PEG | Polyethylene glycol |
| PhMV | Physalis mottle virus |
| PVX | Potato virus X |
| RGD | Arginylglycylaspartic acid |
| scFv | Single-chain variable fragment |
| siRNA | Small interfering RNA |
| SV40 | Simian virus 40 |
| TMV | Tobacco mosaic virus |
| UAA | Unnatural amino acid |
| VLP | Virus-like particle |
References
- Bray, F.; Laversanne, M.; Sung, H.; Ferlay, J.; Siegel, R.L.; Soerjomataram, I.; Jemal, A. Global Cancer Statistics 2022: GLOBOCAN.
- Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2024, 74, 229–263.
- OECD and European Commission EU country cancer profiles synthesis report 2025. OECD Publishing; Paris, France, 2025. [Google Scholar]
- Global cancer burden growing, amidst mounting need for services. Available online: https://www.who.int/news/item/01-02-2024-global-cancer-burden-growing--amidst-mounting-need-for-services (accessed on 3rd of March 2026).
- Shahrivarkevishahi, A.; Hagge, L.M.; Brohlin, O.R.; Kumari, S.; Ehrman, R.; Benjamin, C.; Gassensmith, J.J. Virus-like particles: a self-assembled toolbox for cancer therapy. Mater. Today Chem. 2022, 24, 100808. [Google Scholar]
- Chung, Y.H.; Cai, H.; Steinmetz, N. F. Viral nanoparticles for drug delivery, imaging, immunotherapy, and theranostic applications. Adv. Drug Deliv. Rev. 2020, 156, 214–235. [Google Scholar] [CrossRef]
- Kim, K.R.; Lee, A.S.; Kim, S.M.; Heo, H.R.; Kim, C.S. Virus-like nanoparticles as a theranostic platform for cancer. Front. Bioeng. Biotechnol. 2023, 10, 1106767. [Google Scholar] [CrossRef]
- Nooraei, S.; Bahrulolum, H.; Hoseini, Z.S.; Katalani, C.; Hajizade, A.; Easton, A.J.; Ahmadian, G. Virus-like particles: preparation, immunogenicity and their roles as nanovaccines and drug nanocarriers. J. Nanobiotechnology 2021, 19, 59. [Google Scholar] [CrossRef]
- Kim, K.R.; Lee, A.S.; Kim, S.M.; Heo, H.R.; Kim, C.S. Virus-like nanoparticles as a theranostic platform for cancer. Front. Bioeng. Biotechnol. 2023, 10, 1106767. [Google Scholar] [CrossRef]
- Yuan, B.; Liu, Y.; Lv, M.; Sui, Y.; Hou, S.; Yang, T.; Belhadj, Z.; Zhou, Y.; Chang, N.; Ren, Y.; Sun, C. Virus-like particle-based nanocarriers as an emerging platform for drug delivery. J. Drug Target 2023, 31, 433–455. [Google Scholar] [PubMed]
- Le, D.T.; Müller, K.M. In Vitro Assembly of Virus-Like Particles and Their Applications. Life (Basel) 2021, 11 11, 334. [Google Scholar]
- Alvandi, N.; Rajabnejad, M.; Taghvaei, Z.; Esfandiari, N. New generation of viral nanoparticles for targeted drug delivery in cancer therapy. J. Drug Target 2022, 30, 151–165. [Google Scholar] [PubMed]
- Harrison, S.C. Viral membrane fusion. Virology 2015, 13 479–480, 498–507. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Kreutzberger, A. J. B.; Odongo, L.; Nelson, E. A.; Nyenhuis, D. A.; Kiessling, V.; Liang, B.; Cafiso, D. S.; White, J. M.; Tamm, L. K. Ebola virus glycoprotein interacts with cholesterol to enhance membrane fusion and cell entry. Nat. Struct. Mol. Biol. 2021, 28, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Nkanga, C.I.; Steinmetz, N.F. The pharmacology of plant virus nanoparticles. Virology 2021, 15 556, 39–61. [Google Scholar] [CrossRef]
- Chariou, P. L.; Lee, K. L.; Pokorski, J. K.; Saidel, G. M.; Steinmetz, N. F. Diffusion and Uptake of Tobacco Mosaic Virus as Therapeutic Carrier in Tumor Tissue: Effect of Nanoparticle Aspect Ratio. J. Phys. Chem. B. 2016, 120, 6120–6129. [Google Scholar]
- Ikwuagwu, B.; Tullman-Ercek, D. Virus-like particles for drug delivery: a review of methods and applications. Curr. Opin. Biotechnol. 2022, 78, 102785. [Google Scholar] [CrossRef]
- Berardi, A.; Bombelli, F.B.; Thuenemann, E.C.; Lomonossoff, G.P. Viral nanoparticles can elude protein barriers: exploiting rather than imitating nature. Nanoscale 2019, 11, 2306–2316. [Google Scholar] [CrossRef]
- Zackova Suchanova, J.; Hejtmankova, A.; Neburkova, J.; Cigler, P.; Forstova, J.; Spanielova, H. The Protein Corona Does Not Influence Receptor-Mediated Targeting of Virus-like Particles. Bioconjug. Chem. 2020, 31, 1575–1585. [Google Scholar] [CrossRef]
- Duan, X.; Li, Y. Physicochemical characteristics of nanoparticles affect circulation, biodistribution, cellular internalization, and trafficking. Small 2013, 9, 1521–1532. [Google Scholar] [CrossRef] [PubMed]
- McNeale, D.; Dashti, N.; Cheah, L. C.; Sainsbury, F. Protein cargo encapsulation by virus-like particles: Strategies and applications. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol 2023, 15, e1869. [Google Scholar] [CrossRef] [PubMed]
- Pistono, P.E.; Xu, J.; Huang, P.; Fetzer, J.L.; Francis, M.B. Exploring the Effects of Intersubunit Interface Mutations on Virus-Like Particle Structure and Stability. Biochemistry 2024, 63, 1913–1924. [Google Scholar] [CrossRef]
- Biela, A. P.; Naskalska, A.; Fatehi, F.; Twarock, R.; Heddle, J. G. Programmable polymorphism of a virus-like particle. Commun. Mater. 2022, 3, 7. [Google Scholar] [CrossRef]
- Schumacher, J.; Bacic, T.; Staritzbichler, R.; Daneschdar, M.; Klamp, T.; Arnold, P.; Jägle, S.; Türeci, Ö.; Markl, J.; Sahin, U. Enhanced stability of a chimeric hepatitis B core antigen virus-like-particle (HBcAg-VLP) by a C-terminal linker-hexahistidine-peptide. J. Nanobiotechnology 2018, 16, 39. [Google Scholar] [CrossRef]
- Xu, C.; Zhu, W.; Mao, H.; Zhang, W.; Yin, G. Q.; Zhang, X. E.; Li, F. Switch from Polymorphic to Homogenous Self-Assembly of Virus-Like Particles of Simian Virus 40 through Double-Cysteine Substitution. Small 2020, 16, e2004484. [Google Scholar] [CrossRef]
- de Ruiter, M. V.; van der Hee, R. M.; Driessen, A. J. M.; Keurhorst, E. D.; Hamid, M.; Cornelissen, J. J. L. M. Polymorphic assembly of virus-capsid proteins around DNA and the cellular uptake of the resulting particles. J. Control. Release 2019, 307, 342–354. [Google Scholar] [CrossRef]
- Shukla, S.; Eber, F. J.; Nagarajan, A. S.; DiFranco, N. A.; Schmidt, N.; Wen, A. M.; Eiben, S.; Twyman, R. M.; Wege, C.; Steinmetz, N. F. The Impact of Aspect Ratio on the Biodistribution and Tumor Homing of Rigid Soft-Matter Nanorods. Adv. Healthc. Mater. 2015, 4, 874–882. [Google Scholar] [CrossRef]
- Bruckman, M. A.; Randolph, L. N.; VanMeter, A.; Hern, S.; Shoffstall, A. J.; Taurog, R. E.; Steinmetz, N. F. Biodistribution, pharmacokinetics, and blood compatibility of native and PEGylated tobacco mosaic virus nano-rods and -spheres in mice. Virology 2014, 449, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.; Ablack, A. L.; Wen, A. M.; Lee, K. L.; Lewis, J. D.; Steinmetz, N. F. Increased tumor homing and tissue penetration of the filamentous plant viral nanoparticle Potato virus X. Mol. Pharm. 2013, 10, 33–42. [Google Scholar] [CrossRef]
- Finbloom, J. A.; Aanei, I. L.; Bernard, J. M.; Klass, S. H.; Elledge, S. K.; Han, K.; Ozawa, T.; Nicolaides, T. P.; Berger, M. S.; Francis, M. B. Evaluation of Three Morphologically Distinct Virus-Like Particles as Nanocarriers for Convection-Enhanced Drug Delivery to Glioblastoma. Nanomaterials (Basel) 2018, 8, 1007. [Google Scholar] [CrossRef] [PubMed]
- Raguram, A.; An, M.; Chen, P. Z.; Liu, D. R. Directed evolution of engineered virus-like particles with improved production and transduction efficiencies. Nat. Biotechnol. 2025, 43, 1635–1647. [Google Scholar] [CrossRef]
- Strugała, A.; Jagielski, J.; Kamel, K.; Nowaczyk, G.; Radom, M.; Figlerowicz, M.; Urbanowicz, A. Virus-Like Particles Produced Using the Brome Mosaic Virus Recombinant Capsid Protein Expressed in a Bacterial System. Int. J. Mol. Sci. 2021, 22, 3098. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Guenther, R. H.; Sit, T. L.; Opperman, C. H.; Lommel, S. A.; Willoughby, J. A. Loading and release mechanism of red clover necrotic mosaic virus derived plant viral nanoparticles for drug delivery of doxorubicin. Small 2014, 10, 5126–5136. [Google Scholar] [CrossRef]
- Wong, S. M.; Ren, Y. In Vitro-Reassembled Plant Virus-Like Particles of Hibiscus Chlorotic Ringspot Virus (HCRSV) as Nano-Protein Cages for Drugs. Methods Mol. Bio 2018, 1776, 229–236. [Google Scholar]
- Hu, L.; Salmen, W.; Chen, R.; Zhou, Y.; Neill, F.; Crowe, J. E., Jr.; Atmar, R. L.; Estes, M. K.; Prasad, B. V. V. Atomic structure of the predominant GII.4 human norovirus capsid reveals novel stability and plasticity. Nat. Commun. 2022, 13, 1241. [Google Scholar] [CrossRef] [PubMed]
- Huynh, N. T.; Hesketh, E. L.; Saxena, P.; Meshcheriakova, Y.; Ku, Y. C.; Hoang, L. T.; Johnson, J. E.; Ranson, N. A.; Lomonossoff, G. P.; Reddy, V. S. Crystal Structure and Proteomics Analysis of Empty Virus-like Particles of Cowpea Mosaic Virus. Structure 2016, 24, 567–575. [Google Scholar] [CrossRef]
- Collett, S.; Torresi, J.; Earnest-Silveira, L.; Christiansen, D.; Elbourne, A.; Ramsland, P. A. Probing and pressing surfaces of hepatitis C virus-like particles. J. Colloid Interface Sci. 2019, 545, 259–268. [Google Scholar] [CrossRef]
- McCormick, R. A.; Ralbovsky, N. M.; Gilbraith, W.; Smith, J. P.; Booksh, K. S. Analyzing atomic force microscopy images of virus-like particles by expectation-maximization. NPJ Vaccines 2014, 9, 112. [Google Scholar]
- Wu, C. Y.; Yeh, Y. C.; Yang, Y. C.; Chou, C.; Liu, M. T.; Wu, H. S.; Chan, J. T.; Hsiao, P. W. Mammalian Expression of Virus-Like Particles for Advanced Mimicry of Authentic Influenza Virus. PLoS One 2010, 5, e9784. [Google Scholar] [CrossRef]
- Steppert, P.; Burgstaller, D.; Klausberger, M.; Tover, A.; Berger, E.; Jungbauer, A. Quantification and characterization of virus-like particles by size-exclusion chromatography and nanoparticle tracking analysis. J. Chromatogr. A. 2017, 1487, 89–99. [Google Scholar] [PubMed]
- Murali, S.; Rustandi, R. R.; Zheng, X.; Payne, A.; Shang, L. Applications of Surface Plasmon Resonance and Biolayer Interferometry for Virus–Ligand Binding. Viruses 2022, 14, 717. [Google Scholar] [CrossRef]
- Zhang, L.; Lua, L.H.L.; Middelberg, A.P.J.; Sun, Y.; Connors, N.K. Biomolecular engineering of virus-like particles aided by computational chemistry methods. Chem. Soc. Rev. 2015, 44, 8608–8618. [Google Scholar] [CrossRef]
- Elrad, O. M.; Hagan, M. F. Mechanisms of Size Control and Polymorphism in Viral Capsid Assembly. Nano Lett. 2008, 8, 3850–3857. [Google Scholar] [CrossRef]
- Luo, H.; Ma, Y.; Ren, Y.; Li, Z.; Sheng, Y.; Wang, Y.; Su, Z.; Bi, J.; Zhang, S. Study of self-assembling properties of HBc-VLP derivatives aided by molecular dynamic simulations from a thermodynamic perspective. J. Biomol. Struct. Dyn. 2024, 42, 12822–12835. [Google Scholar] [CrossRef] [PubMed]
- Wijesundara, Y. H.; Herbert, F. C.; Kumari, S.; Howlett, T.; Koirala, S.; Trashi, O.; Trashi, I.; Al-Kharji, N. M.; Gassensmith, J. J. Rip it, stitch it, click it: A chemist’s guide to VLP manipulation. Virology 2022, 577, 105–123. [Google Scholar] [CrossRef]
- Shukla, S.; Roe, A. J.; Liu, R.; Veliz, F. A.; Commandeur, U.; Wald, D. N.; Steinmetz, N. F. Affinity of plant viral nanoparticle potato virus x (PVX) towards malignant b cells enables cancer drug delivery. Biomater. Sci. 2020, 8, 3935–3943. [Google Scholar] [CrossRef]
- Li, M.; Liu, Z.; Wang, D.; Ye, J.; Shi, Z.; Pan, C.; Zhang, Q.; Ju, R.; Zheng, Y.; Liu, Y. Intraocular mRNA delivery with endogenous MmPEG10-based virus-like particles. Exp. Eye Res. 2024, 243, 109899. [Google Scholar] [CrossRef]
- Gan, B. K.; Rullah, K.; Yong, C. Y.; Ho, K. L.; Omar, A. R.; Alitheen, N. B.; Tan, W. S. Targeted delivery of 5-fluorouracil-1-acetic acid (5-FA) to cancer cells overexpressing epithelial growth factor receptor (EGFR) using virus-like nanoparticles. Sci. Rep. 2020, 10, 16867. [Google Scholar] [CrossRef]
- Thong, Q. X.; Biabanikhankahdani, R.; Ho, K. L.; Alitheen, N. B.; Tan, W. S. Thermally-responsive virus-like particle for targeted delivery of cancer drug. Sci. Rep. 2019, 9, 3945. [Google Scholar] [CrossRef] [PubMed]
- Hovlid, M. L.; Steinmetz, N. F.; Laufer, B.; Lau, J. L.; Kuzelka, J.; Wang, Q.; Hyypiä, T.; Nemerow, G. R.; Kessler, H.; Manchester, M.; Finn, M. G. Guiding Plant Virus Particles to Integrin-Displaying Cells. Nanoscale 2012, 4, 3698–3705. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, D.; Liu, A. P.; Voss, N. R.; Schmid, S. L.; Finn, M. G. Multivalent Display and Receptor-Mediated Endocytosis of Transferrin on Virus-Like Particles. Chembiochem 2010, 11, 1273–1279. [Google Scholar] [CrossRef]
- Charoenphol, P.; Bermudez, H. Aptamer-targeted DNA nanostructures for therapeutic delivery. Mol. Pharm. 2014, 11, 1721–1725. [Google Scholar] [CrossRef]
- Martins, S. A.; Santos, J.; Cabo Verde, S.; Correia, J. D. G.; Melo, R. Construction of HER2-specific HIV-1-based VLPs. Bioengineering (Basel) 2022, 9, 713. [Google Scholar] [CrossRef]
- Wang, G.; Jia, T.; Xu, X.; Chang, L.; Zhang, R.; Fu, Y.; Li, Y.; Yang, X.; Zhang, K.; Lin, G.; Han, Y.; Li, J. Novel miR-122 delivery system based on MS2 virus like particle surface displaying cell-penetrating peptide TAT for hepatocellular carcinoma. Oncotarget 2016, 7, 59402–59416. [Google Scholar] [CrossRef]
- Muñoz-Morris, M. A.; Heitz, F.; Divita, G.; Morris, M. C. The peptide carrier Pep-1 forms biologically efficient nanoparticle complexes. Biochem. Biophys. Res. Commun. 2007, 355, 877–882. [Google Scholar] [CrossRef]
- Liu, B. R.; Lo, S. Y.; Liu, C. C.; Chyan, C. L.; Huang, Y. W.; Aronstam, R. S.; Lee, H. J. Endocytic Trafficking of Nanoparticles Delivered by Cell-penetrating Peptides Comprised of Nona-arginine and a Penetration Accelerating Sequence. PLoS One 2013, 8, e67100. [Google Scholar] [CrossRef]
- Zhao, L.; Kopylov, M.; Potter, C.S.; Carragher, B.; Finn, M.G. Engineering the pp7 virus capsid as a peptide display platform. ACS Nano 2019, 13, 4443–4454. [Google Scholar] [CrossRef]
- Dang, M.; Wu, L. J.; Zhang, S. R.; Zhu, J. R.; Hu, Y. Z.; Yang, C. X.; Zhang, X. Y. Ms2 virus-like particles as a versatile peptide presentation platform: Insights into the deterministic abilities for accommodating heterologous peptide lengths. ACS Synth. Biol. 2023, 12, 3704–3715. [Google Scholar] [CrossRef]
- Essus, V. A.; Souza Júnior, G. S. E.; Nunes, G. H. P.; Oliveira, J. D. S.; de Faria, B. M.; Romão, L. F.; Cortines, J. R. Bacteriophage P22 capsid as a pluripotent nanotechnology tool. Viruses 2023, 15, 516. [Google Scholar] [CrossRef] [PubMed]
- Zepeda-Cervantes, J.; Ramírez-Jarquín, J. O.; Vaca, L. Interaction between virus-like particles (VLPs) and pattern recognition receptors (PRRs) from dendritic cells (DCs): Toward better engineering of VLPs. Front Immunol. 2020, 11, 1100. [Google Scholar] [CrossRef]
- Smith, M. L.; Lindbo, J. A.; Dillard-Telm, S.; Brosio, P. M.; Lasnik, A. B.; McCormick, A. A.; Nguyen, L. V.; Palmer, K. E. Modified tobacco mosaic virus particles as scaffolds for display of protein antigens for vaccine applications. Virology 2006, 348, 475–488. [Google Scholar] [CrossRef] [PubMed]
- Peyret, H.; Gehin, A.; Thuenemann, E. C.; Blond, D.; El Turabi, A.; Beales, L.; Clarke, D.; Gilbert, R. J.; Fry, E. E.; Stuart, D. I.; Holmes, K.; Stonehouse, N. J.; Whelan, M.; Rosenberg, W.; Lomonossoff, G. P.; Rowlands, D. J. Tandem fusion of hepatitis B core antigen allows assembly of virus-like particles in bacteria and plants with enhanced capacity to accommodate foreign proteins. PLoS One 2015, 10, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Lino, C. A.; Caldeira, J. C.; Peabody, D. S. Display of single-chain variable fragments on bacteriophage MS2 virus-like particles. J. Nanobiotechnology 2017, 15, 13. [Google Scholar] [CrossRef]
- Grataitong, K.; Huault, S.; Chotwiwatthanakun, C.; Jariyapong, P.; Thongsum, O.; Chawiwithaya, C.; Chakrabandhu, K.; Hueber, A. O.; Weerachatyanukul, W. Chimeric virus-like particles (VLPs) designed from shrimp nodavirus (MrNV) capsid protein specifically target EGFR-positive human colorectal cancer cells. Sci. Rep. 2021, 11, 16579. [Google Scholar]
- Martino, M. L.; Crooke, S. N.; Manchester, M.; Finn, M. G. Single-point mutations in Qβ virus-like particles change binding to cells. Biomacromolecules 2021, 22, 3332–3341. [Google Scholar] [CrossRef]
- Servid, A.; Jordan, P.; O’Neil, A.; Prevelige, P.; Douglas, T. Location of the bacteriophage P22 coat protein C-terminus provides opportunities for the design of capsid-based materials. Biomacromolecules 2013, 14, 2989–2995. [Google Scholar] [CrossRef]
- Pokorski, J. K.; Steinmetz, N. F. The art of engineering viral nanoparticles. Mol. Pharm. 2011, 8, 29–43. [Google Scholar]
- Biabanikhankahdani, R.; Ho, K. L.; Alitheen, N. B.; Tan, W. S. A dual bioconjugated virus-like nanoparticle as a drug delivery system and comparison with a pH-responsive delivery system. Nanomaterials (Basel) 2018, 8, 236. [Google Scholar] [CrossRef]
- Edwardson, T. G. W.; Levasseur, M. D.; Tetter, S.; Steinauer, A.; Hori, M.; Hilvert, D. Protein cages: From fundamentals to advanced applications. Chem. Rev. 2022, 122, 9145–9197. [Google Scholar] [CrossRef] [PubMed]
- Chen, P. L.; Wang, M.; Ou, W. C.; Lii, C. K.; Chen, L. S.; Chang, D. Disulfide bonds stabilize JC virus capsid-like structure by protecting calcium ions from chelation. FEBS Lett. 2001, 500, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Finbloom, J. A.; Han, K.; Aanei, I. L.; Hartman, E. C.; Finley, D. T.; Dedeo, M. T.; Fishman, M.; Downing, K. H.; Francis, M. B. Stable disk assemblies of a tobacco mosaic virus mutant as nanoscale scaffolds for applications in drug delivery. Bioconjug. Chem. 2016, 27, 2480–2485. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Chariou, P. L.; Steinmetz, N. F. Site-specific antibody conjugation strategy to functionalize virus-based nanoparticles. Bioconjug. Chem. 2020, 31, 1408–1416. [Google Scholar] [CrossRef]
- Strable, E.; Prasuhn, D. E., Jr.; Udit, A. K.; Brown, S.; Link, A. J.; Ngo, J. T.; Lander, G.; Quispe, J.; Potter, C. S.; Carragher, B.; Tirrell, D. A.; Finn, M. G. Unnatural amino acid incorporation into virus-like particles. Bioconjug. Chem. 2009, 19, 866–875. [Google Scholar] [CrossRef]
- Patel, K. G.; Swartz, J. R. Surface functionalization of virus-like particles by direct conjugation using azide–alkyne click chemistry. Bioconjug. Chem. 2011, 22, 376–387. [Google Scholar]
- Chen, Z.; Li, N.; Li, S.; Dharmarwardana, M.; Schlimme, A.; Gassensmith, J. J. Viral chemistry: the chemical functionalization of viral architectures to create new technology. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2016, 8, 512–534. [Google Scholar] [CrossRef]
- Jewett, J. C.; Bertozzi, C. R. Cu-free click cycloaddition reactions in chemical biology. Chem. Soc. Rev. 2010, 39, 1272–1279. [Google Scholar] [CrossRef]
- Patterson, D.; Schwarz, B.; Avera, J.; Western, B.; Hicks, M.; Krugler, P.; Terra, M.; Uchida, M.; McCoy, K.; Douglas, T. Sortase-Mediated Ligation as a Modular Approach for the Covalent Attachment of Proteins to the Exterior of the Bacteriophage P22 Virus-like Particle. Bioconjug. Chem. 2017, 28, 2114–2124. [Google Scholar] [CrossRef]
- Brune, K. D.; Leneghan, D. B.; Brian, I. J.; Ishizuka, A. S.; Bachmann, M. F.; Draper, S. J.; Biswas, S.; Howarth, M. Plug-and-Display: decoration of Virus-Like Particles via isopeptide bonds for modular immunization. Sci. Rep. 2016, 6, 19234. [Google Scholar] [CrossRef]
- Rohovie, M. J.; Nagasawa, M.; Swartz, J. R. Virus-like particles: Next-generation nanoparticles for targeted therapeutic delivery. Bioeng. Transl. Med. 2017, 2, 43–57. [Google Scholar] [PubMed]
- Schwarz, B.; Madden, P.; Avera, J.; Gordon, B.; Larson, K.; Miettinen, H. M.; Uchida, M.; LaFrance, B.; Basu, G.; Rynda-Apple, A.; Douglas, T. Symmetry Controlled, Genetic Presentation of Bioactive Proteins on the P22 Virus-like Particle Using an External Decoration Protein. ACS Nano 2015, 9, 9134–9147. [Google Scholar] [CrossRef]
- Koho, T.; Ihalainen, T. O.; Stark, M.; Uusi-Kerttula, H.; Wieneke, R.; Rahikainen, R.; Blazevic, V.; Marjomäki, V.; Tampé, R.; Kulomaa, M. S.; Hytönen, V. P. His-tagged norovirus-like particles: A versatile platform for cellular delivery and surface display. Eur. J. Pharm. Biopharm. 2015, 96, 22–31. [Google Scholar]
- Dickens, E.; Ahmed, S. Principles of cancer treatment by chemotherapy. Surgery (Oxford) 2018, 36, 134–138. [Google Scholar] [CrossRef]
- Gala, U. H.; Miller, D. A.; Williams, R. O., 3rd. Harnessing the therapeutic potential of anticancer drugs through amorphous solid dispersions. Biochim. Biophys. Acta Rev. Cancer 2020, 1873, 188319. [Google Scholar] [PubMed]
- Khan, S. U.; Fatima, K.; Aisha, S.; Malik, F. Unveiling the mechanisms and challenges of cancer drug resistance. Cell Comun. Signal 2024, 22, 109. [Google Scholar] [CrossRef]
- Wolfram, J.; Ferrari, M. Clinical cancer nanomedicine. Nano Today 2019, 25, 85–98. [Google Scholar] [CrossRef]
- Ghosh, S.; Banerjee, M. A smart viral vector for targeted delivery of hydrophobic drugs. Sci. Rep. 2021, 11, 7030. [Google Scholar] [CrossRef] [PubMed]
- Odegard, A.; Banerjee, M.; Johnson, J. E. Flock house virus: A model system for understanding nonenveloped virus entry and membrane penetration. Curr. Top. Microbiol. Immunol. 2010, 343, 1–22. [Google Scholar] [PubMed]
- Song, N.; Zhao, L.; Zhu, M.; Zhao, J. Recent progress in LyP-1-based strategies for targeted imaging and therapy. Drug Deliv. 2019, 26, 363–375. [Google Scholar] [CrossRef]
- Hu, H.; Steinmetz, N. F. Doxorubicin-loaded physalis mottle virus particles function as a pH-responsive prodrug enabling cancer therapy. Biotechnol. J. 2020, 15, e2000077. [Google Scholar] [CrossRef]
- Hu, H.; Masarapu, H.; Gu, Y.; Zhang, Y.; Yu, X.; Steinmetz, N. F. Physalis mottle virus-like nanoparticles for targeted cancer imaging. ACS Appl Mater Interfaces 2019, 11, 18213–18223. [Google Scholar] [CrossRef]
- Shukla, S.; Wen, A. M.; Ayat, N. R.; Commandeur, U.; Gopalkrishnan, R.; Broome, A. M.; Lozada, K. W.; Keri, R. A.; Steinmetz, N. F. Biodistribution and clearance of a filamentous plant virus in healthy and tumor-bearing mice. Nanomedicine (Lond.) 2019, 9, 221–235. [Google Scholar] [CrossRef]
- Pang, H. H.; Chen, P. Y.; Wei, K. C.; Huang, C. W.; Shiue, Y. L.; Huang, C. Y.; Yang, H. W. Convection-enhanced delivery of a virus-like nanotherapeutic agent with dual-modal imaging for besiegement and eradication of brain tumors. Theranostics 2019, 9, 752–1763. [Google Scholar] [CrossRef]
- Kines, R. C.; Varsavsky, I.; Choudhary, S.; Bhattacharya, D.; Spring, S.; McLaughlin, R.; Kang, S. J.; Grossniklaus, H. E.; Vavvas, D.; Monks, S.; MacDougall, J. R.; de Los Pinos, E.; Schiller, J. T. An infrared dye–conjugated virus-like particle for the treatment of primary uveal melanoma. Mol. Cancer Ther. 2018, 17, 565–574. [Google Scholar] [CrossRef]
- Kines, R. C.; Cerio, R. J.; Roberts, J. N.; Thompson, C. D.; de Los Pinos, E.; Lowy, D. R.; Schiller, J. T. Human papillomavirus capsids preferentially bind and infect tumor cells. Int. J. Cancer 2016, 138, 901–911. [Google Scholar] [CrossRef]
- Ma, S.; Huis In’t Veld, R. V.; Houy, A.; Stern, M. H.; Rich, C.; Ossendorp, F. A.; Jager, M. J. In vitro testing of the virus-like drug conjugate belzupacap sarotalocan (AU-011) on uveal melanoma suggests BAP1-related immunostimulatory capacity. Invest. Ophthalmol. Vis. Sci. 2023, 64, 10. [Google Scholar] [CrossRef]
- Kines, R. C.; Thompson, C. D.; Spring, S.; Li, Z.; de Los Pinos, E.; Monks, S.; Schiller, J. T. Virus-like particle-drug conjugates induce protective, long-lasting adaptive anti-tumor immunity in the absence of specifically targeted tumor antigens. Cancer Immunol. Res. 2021, 9, 693–706. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, Q.; Liu, Y.; Zhang, X.; Shan, W.; Ye, S.; Zhou, X.; Ge, Y.; Wang, X.; Ren, L. Nanoparticle-based co-delivery of siRNA and paclitaxel for dual-targeting of glioblastoma. Nanomedicine (Lond.) 2020, 15, 1391–1409. [Google Scholar] [CrossRef]
- Deo, V. K.; Kato, T.; Park, E. Y. Virus-like particles displaying recombinant short-chain fragment region and interleukin 2 for targeting colon cancer tumors and attracting macrophages. J. Pharm. Sci. 2016, 105, 1614–1622. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Ganguillet, S.; Turgay, Y.; Keys, T. G.; Causa, E.; Fradique, R.; Lutz-Bueno, V.; Chesnov, S.; Tan-Lin, C. W.; Lentsch, V.; Kotar, J.; Cicuta, P.; Mezzenga, R.; Slack, E.; Radiom, M. Surface cross-linking by macromolecular tethers enhances virus-like particles’ resilience to mucosal stress factors. ACS Nano 2024, 18, 3382–3396. [Google Scholar] [CrossRef] [PubMed]
- Owens, D. E., 3rd; Peppas, N. A. Opsonization, biodistribution, and pharmacokinetics of polymeric nanoparticles. Int. J. Pharm. 2006, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Shi, D.; Beasock, D.; Fessler, A.; Szebeni, J.; Ljubimova, J.Y.; Afonin, K.A.; Dobrovolskaia, M.A. To PEGylate or not to PEGylate: immunological properties of nanomedicine’s most popular component, poly(ethylene) glycol and its alternatives. Adv Drug Deliv. Rev. 2021, 180, 114079. [Google Scholar] [CrossRef]
- Wu, Z.; Bayón, J. L.; Kouznetsova, T. B.; Ouchi, T.; Barkovich, K. J.; Hsu, S. K.; Craig, S. L.; Steinmetz, N. F. Virus-like particles armored by an endoskeleton. Nano Lett. 2024, 24, 2989–2997. [Google Scholar] [CrossRef]
- Lucon, J.; Qazi, S.; Uchida, M.; Bedwell, G. J.; LaFrance, B.; Prevelige, P. E., Jr.; Douglas, T. Use of the interior cavity of the p22 capsid for site-specific initiation of atom-transfer radical polymerization with high-density cargo loading. Nat. Chem. 2012, 4, 781–788. [Google Scholar] [CrossRef]
- Kim, K. J.; Kim, G.; Bae, J. H.; Song, J. J.; Kim, H. S. A pH-responsive virus-like particle as a protein cage for a targeted delivery. Adv. Healthc. Mater. 2024, 13, e2302656. [Google Scholar] [CrossRef]
- Luo, H.; Ma, Y.; Su, Z.; Gu, Y.; Zhang, S.; Gerstweiler, L. Investigating the stability of chimeric murine polyomavirus VP1 Capsomeres via molecular dynamics simulations and experimental analysis. Int. J. Biol. Macromol. 2025, 286, 138372. [Google Scholar] [CrossRef]
- Laxmi, B.; Devi, P. U. M.; Thanjavur, N.; Buddolla, V. The applications of artificial intelligence (AI)-driven tools in virus-like particles (VLPs) research. Curr. Microbiol. 2024, 81, 234. [Google Scholar] [CrossRef]
- Beatty, P. H.; Lewis, J. D. Cowpea mosaic virus nanoparticles for cancer imaging and therapy. Adv. Drug Deliv. Rev. 2019, 145, 130–144. [Google Scholar] [CrossRef]
- Koudelka, K. J.; Destito, G.; Plummer, E. M.; Trauger, S. A.; Siuzdak, G.; Manchester, M. Endothelial targeting of cowpea mosaic virus (CPMV) via surface vimentin. PLoS Pathog. 2009, 5, e1000417. [Google Scholar] [CrossRef] [PubMed]
- Steinmetz, N. F.; Cho, C. F.; Ablack, A.; Lewis, J. D.; Manchester, M. Cowpea Mosaic Virus Nanoparticles Target Surface Vimentin on Cancer Cells. Nanomedicine (Lond.) 2011, 6, 351–364. [Google Scholar] [CrossRef] [PubMed]
- Steinmetz, N. F.; Ablack, A. L.; Hickey, J. L.; Ablack, J.; Manocha, B.; Mymryk, J. S.; Luyt, L. G.; Lewis, J. D. Intravital Imaging of Human Prostate Cancer Using Viral Nanoparticles Targeted to Gastrin-Releasing Peptide Receptors. Small 2011, 7, 1664–1672. [Google Scholar] [CrossRef] [PubMed]
- Herdiana, Y. Bridging the Gap: The Role of Advanced Formulation Strategies in the Clinical Translation of Nanoparticle-Based Drug Delivery Systems. Int. J. Nanomedicine 2025, 20, 13039–13053. [Google Scholar] [CrossRef] [PubMed]
- Metselaar, J. M.; Lammers, T. Challenges in nanomedicine clinical translation. Drug Deliv. Transl. Res. 2020, 10, 721–725. [Google Scholar] [CrossRef]
- Bachmann, M. F.; van Damme, P.; Lienert, F.; Schwarz, T. F. Virus-like particles: a versatile and effective vaccine platform. Expert Rev. Vaccines 2025, 24, 444–456. [Google Scholar] [CrossRef]
- Huynh, E.; Zheng, G. Engineering multifunctional nanoparticles: all-in-one versus one-for-all. WIREs Nanomed. Nanobiotechnol. 2013, 5, 250–265. [Google Scholar] [CrossRef] [PubMed]
- Guidolin, K.; Zheng, G. Nanomedicines Lost in Translation. ACS Nano 2019, 13, 13620–13626. [Google Scholar] [CrossRef] [PubMed]
- Gessner, I. Optimizing nanoparticle design and surface modification toward clinical translation. MRS Bull. 2021, 46, 643–649. [Google Scholar] [CrossRef] [PubMed]
| Functional Group Type | Compounds | Function/Target | Application | Reference |
|---|---|---|---|---|
| Therapeutic Agents | Doxorubicin (DOX) | DNA intercalation; inhibits topoisomerase II | Chemotherapy | 29 |
| Monomethyl auristatin (MMAE) | Human B lymphoma cells targeting | Chemotherapy | 45 | |
| mRNA | Protein expression | Immunotherapy, gene therapy | 46 | |
| 5-fluorouracil-1-acetic acid | Binds epithelial growth factor receptor (EGFR) |
Chemotherapy | 47 | |
| Targeting Ligands | Folic Acid | Binds folate receptor | Targeting folate receptor–positive tumors | 48 |
| RGD peptides | Binds integrin αvβ3 | Anti-angiogenesis, tumor targeting | 49 | |
| Transferrin | Binds transferrin receptor | Targeting rapidly dividing cells | 50 | |
| Aptamer (e.g., AS1411) | Targets nucleolin | Selective tumor targeting | 51 | |
| Anti-HER2 antibody | Binds HER2 receptor | Targeting HER2-positive breast cancer | 52 | |
| Cell-Penetrating Peptides | TAT (HIV-derived) | Facilitates nuclear and cytosolic delivery | Intracellular delivery of drugs or genes | 53 |
| Pep-1 | Non-covalent cargo delivery | Protein or peptide delivery | 54 | |
| R9 (nona-arginine) | Promotes uptake | Enhanced internalization | 55 |
| Design Parameter | Biomaterial Feature | Controls | Design Trade-Off | References |
|---|---|---|---|---|
| Capsid geometry | Size and aspect ratio | Tumor penetration, circulation | Small particles enhance diffusion; elongated forms alter clearance |
[15,26,27,28,29] |
| Capsid stability | Inter-subunit cohesion, crosslinking | Circulatory integrity, endosomal resistance | Excess rigidity may hinder intracellular release | [23,24,98,101] |
| Surface charge | Zeta potential | Cellular uptake, serum interaction |
Positive charge increases uptake but accelerates clearance | [19,66] |
| Surface shielding | P[49,50,63EG or hydrophilic coatings | Immune evasion, reduced opsonization |
High density may mask targeting ligands | [99,100,109] |
| Ligand density | Multivalent display | Receptor engagement, selectivity |
Overcrowding may cause steric hindrance | [49,50,63] |
| Cargo loading strategy |
Encapsulation or conjugation |
Drug ratio, release kinetics | High loading can destabilize capsids |
[32,33,67,102] |
| Stimuli-responsive elements | pH-, redox-, or enzyme-sensitive linkers |
Site-specific drug release |
Premature activation reduces efficacy |
[32,88,103] |
| Native tropism | Intrinsic receptor affinity |
Target specificity | May cause off-target accumulation |
[106,107,108,109] |
| Manufacturing platform |
Expression and purification system |
Scalability, reproducibility |
Complex designs increase cost | [110,111,112] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




