Submitted:
26 March 2026
Posted:
27 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Participants
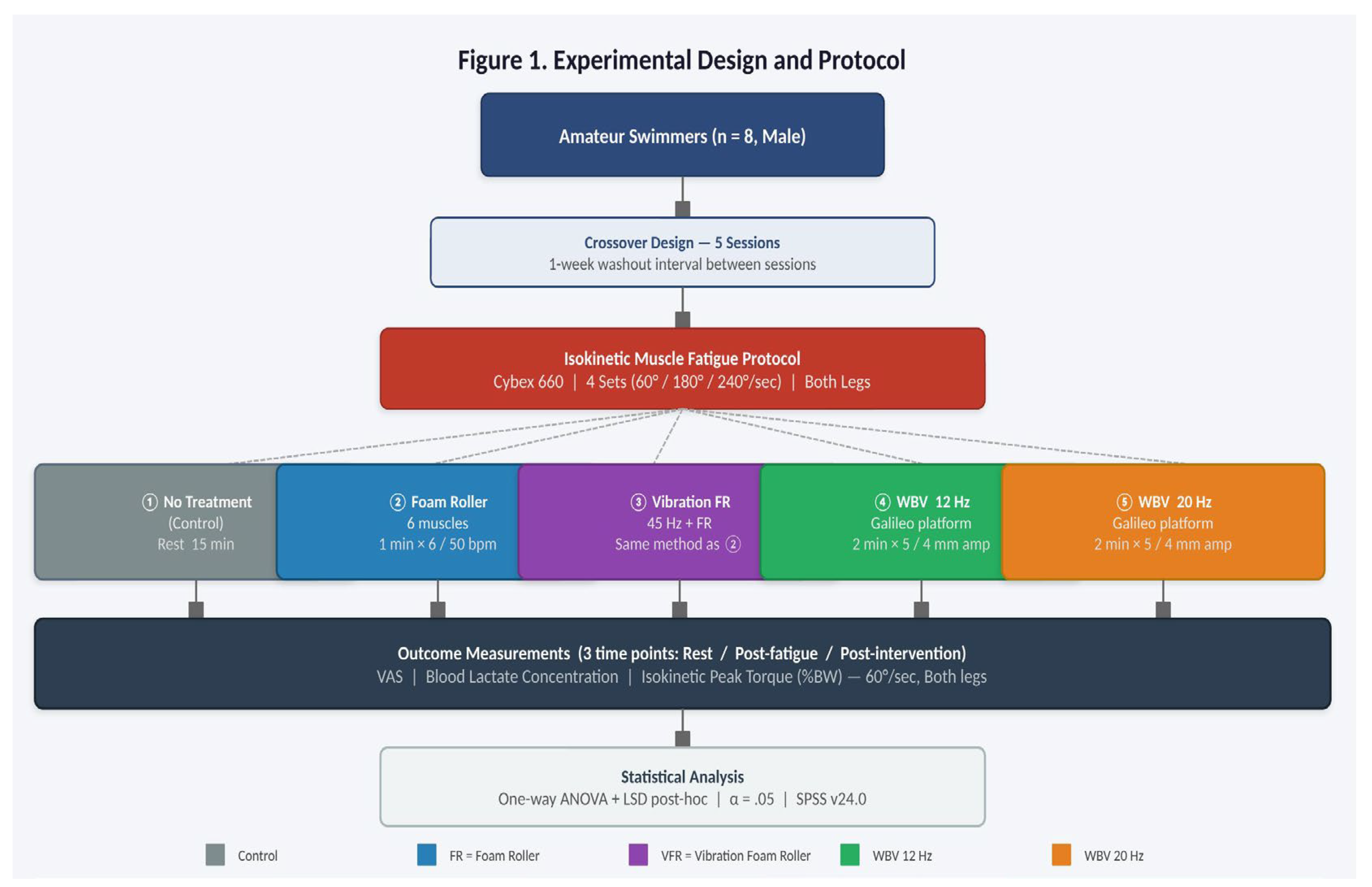
2.2. Exercise-Induced Muscle Fatigue
2.3. Recovery Interventions
2.4. Outcome Measures
2.5. Statistical Analysis
3. Results
3.1. Changes in VAS Scores After Recovery Interventions
3.2. Changes in Blood Lactate Concentration After Recovery Interventions
3.3. Changes in Blood Lactate Recovery Rate After Recovery Interventions
3.4. Changes in Isokinetic Knee Extensor Variables After Recovery Interventions
3.5. Changes in Isokinetic Knee Flexor Variables After Recovery Interventions
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Abbiss, C.R.; Laursen, P.B.; Peiffer, J.J.; Meeusen, R. Role of ratings of perceived exertion during self-paced exercise: What are we actually measuring? Sports Med. 2013, 43, 623–633. [Google Scholar] [CrossRef] [PubMed]
- Allen, D.G.; Lamb, G.D.; Westerblad, H. Skeletal muscle fatigue: Cellular mechanisms. Physiol. Rev. 2008, 88, 287–332. [Google Scholar] [CrossRef]
- Alghadir, A.H.; Zafar, H.; Anwer, S.; Iqbal, Z. Effect of localised vibration on muscle strength in healthy adults: A systematic review. Physiotherapy 2018, 104, 18–24. [Google Scholar] [CrossRef]
- Behm, D.G.; Wilke, J.; Doan, J.B. Foam rolling as a recovery tool after an intense bout of physical activity. Med. Sci. Sports Exerc. 2015, 47, 136–143. [Google Scholar]
- Bosco, C.; Iacovelli, M.; Tsarpela, O.; Cardinale, M.; Bonifazi, M.; Tihanyi, J.; Viru, A. Adaptive response of human skeletal muscle to vibration exposure. Clin. Physiol. 1999, 19, 183–187. [Google Scholar] [CrossRef]
- Byrne, C.; Eston, R.; Edwards, R.H. Characteristics of isometric and dynamic strength loss following eccentric exercise-induced muscle damage. Scand. J. Med. Sci. Sports 2004, 14, 101–106. [Google Scholar]
- Cardinale, M.; Bosco, C. The use of vibration as an exercise intervention. Exerc. Sport Sci. Rev. 2003, 31, 3–7. [Google Scholar] [CrossRef]
- Cardinale, M.; Wakeling, J. Whole body vibration exercise: Are vibrations good for you? Br. J. Sports Med. 2005, 39, 585–589. [Google Scholar] [CrossRef]
- Cheatham, S.W.; Kolber, M.J.; Cain, M.; Lee, M. The effects of self-myofascial release using a foam roll or roller massager on joint range of motion, muscle recovery, and performance: A systematic review. Int. J. Sports Phys. Ther. 2015, 10, 827–838. [Google Scholar] [PubMed]
- Cheatham, S.W.; Stull, K.R.; Kolber, M.J. Roller massage: Comparison of three different surface-type pattern foam rollers on passive knee range of motion and pain perception. J. Bodyw. Mov. Ther. 2019, 23, 555–560. [Google Scholar] [CrossRef] [PubMed]
- Cheung, K.; Hume, P.; Maxwell, L. Delayed onset muscle soreness: Treatment strategies and performance factors. Sports Med. 2003, 33, 145–164. [Google Scholar] [CrossRef] [PubMed]
- Delecluse, C.; Roelants, M.; Verschueren, S. Strength increase after whole-body vibration compared with resistance training. Med. Sci. Sports Exerc. 2003, 35, 1033–1041. [Google Scholar] [CrossRef] [PubMed]
- Gandevia, S.C. Spinal and supraspinal factors in human muscle fatigue. Physiol. Rev. 2001, 81, 1725–1789. [Google Scholar] [CrossRef]
- Gerdle, B.; Karlsson, S.; Crenshaw, A.G.; Elert, J.; Fridén, J. The influences of muscle fibre proportions and areas upon EMG during maximal dynamic knee extensions. Eur. J. Appl. Physiol. 2000, 81, 2–10. [Google Scholar] [CrossRef]
- Han, S.W.; Lee, Y.S.; Lee, D.J. The influence of the vibration foam roller exercise on the pains in the muscles around the hip joint and the joint performance. J. Phys. Ther. Sci. 2017, 29, 1844–1847. [Google Scholar] [CrossRef]
- Hill, A.V.; Long, C.N.H.; Lupton, H. Muscular exercise, lactic acid, and the supply and utilization of oxygen. Q. J. Exp. Physiol. 1924, 86, 8–29. [Google Scholar] [CrossRef]
- Kang, C.W.; Kim, D.J.; Kim, J.H. The effect of whole-body vibration training on recovery from high-intensity exercise. J. Phys. Ther. Sci. 2017, 29, 129–133. [Google Scholar]
- Kang, S.R.; Min, J.Y.; Yu, C.; Kwon, T.K. Effect of whole body vibration on lactate level recovery and heart rate recovery in rest after intense exercise. Technol. Health Care 2017, 25, 115–123. [Google Scholar] [CrossRef]
- Kim, J.-S.; Moon, D.-C.; Chang, K.-S. Changes of flexibility and plasma catecholamine by myofascial release approach. Korea Contents Assoc. J. 2012, 12, 214–221. [Google Scholar] [CrossRef]
- Kosar, A.C.; Candow, D.G.; Putland, J.T. Potential beneficial effects of whole-body vibration for muscle recovery after exercise. J. Strength Cond. Res. 2012, 26, 2907–2911. [Google Scholar] [CrossRef]
- Laimi, K.; Mäkilä, A.; Bärlund, E.; Katajapuu, N.; Oksanen, A.; Seikkula, V.; Karppinen, J.; Saltychev, M. Effectiveness of myofascial release in treatment of chronic musculoskeletal pain: A systematic review. J. Rehabil. Med. 2017, 49, 546–554. [Google Scholar] [CrossRef]
- Macdonald, G.Z.; Button, D.C.; Drinkwater, E.J.; Behm, D.G. Foam rolling as a recovery tool after an intense bout of physical activity. Med. Sci. Sports Exerc. 2014, 46, 131–142. [Google Scholar] [CrossRef]
- Mawhinney, C.; Jones, H.; Joo, C.H.; Low, D.A.; Green, D.J.; Gregson, W. Influence of cold-water immersion on limb and cutaneous blood flow after exercise. Med. Sci. Sports Exerc. 2013, 45, 2277–2285. [Google Scholar] [CrossRef] [PubMed]
- Meeusen, R.; Duclos, M.; Gleeson, M.; Rietjens, G.; Steinacker, J.; Urhausen, A. Prevention, diagnosis and treatment of the overtraining syndrome. Eur. J. Sport Sci. 2007, 7, 1–14. [Google Scholar] [CrossRef]
- Osawa, Y.; Oguma, Y.; Ishii, N. The effects of whole-body vibration on muscle strength and power: A meta-analysis. J. Musculoskelet. Neuronal Interact. 2013, 13, 380–390. [Google Scholar]
- Pearcey, G.E.P.; Bradbury-Squires, D.J.; Kawamoto, J.E.; Drinkwater, E.J.; Behm, D.G.; Button, D.C. Foam rolling for delayed-onset muscle soreness and recovery of dynamic performance measures. J. Athl. Train. 2015, 50, 5–13. [Google Scholar] [CrossRef]
- Pojednic, R.M.; Clark, D.J.; Fielding, R.A. Vibration exercise in rehabilitation and sports: Mechanisms and applications. Phys. Ther. Sport 2021, 49, 108–117. [Google Scholar]
- Ramos-Campo, D.J.; Llorente-Cantarero, F.J.; Clément-Suárez, V.J.; Rubio-Arias, J.Á. A meta-analysis of foam roller effects on performance and recovery. Front. Physiol. 2019, 10, 376. [Google Scholar]
- Romero-Moraleda, B.; La Torre, A.; Juárez, D.; García-López, D.; Maestro, A.; Morencos, E. Effects of vibration and non-vibration foam rolling on recovery. J. Sports Sci. Med. 2019, 18, 172–180. [Google Scholar] [PubMed]
- Rittweger, J. Vibration as an exercise modality: How it may work, and what its potential might be. Eur. J. Appl. Physiol. 2010, 108, 877–904. [Google Scholar] [CrossRef] [PubMed]
- Simao, R.; Santos, L.; Ferreira, J.V.; Prestes, J. Vibration training and recovery: A review. Sports Med. Open 2022, 8, 65. [Google Scholar]
- Song, C.H. The effects of whole-body vibration exercise on body composition, flexibility, and muscular strength. J. Phys. Ther. Sci. 2010, 22, 7–11. [Google Scholar]
- Spencer, M.; Bishop, D.; Dawson, B.; Goodman, C.; Duffield, R. Performance and metabolism in repeated sprint exercise: Effect of recovery intensity. Eur. J. Appl. Physiol. 2008, 103, 545–552. [Google Scholar] [CrossRef]
- Takahashi, J.; Ishihara, K.; Aoki, J. Effect of aqua exercise on recovery of lower limb muscles after downhill running. J. Sports Sci. 2006, 24, 835–842. [Google Scholar] [CrossRef] [PubMed]
- Weerapong, P.; Hume, P.A.; Kolt, G.S. The mechanisms of massage and effects on performance, muscle recovery and injury prevention. Sports Med. 2005, 35, 235–256. [Google Scholar] [CrossRef]
- Watanabe, Y.; Madarame, H.; Ogasawara, R.; Nakazato, K.; Ishii, N. Effect of very low-intensity resistance training with slow movement on muscle size and strength in healthy older adults. Clin. Physiol. Funct. Imaging 2019, 51, 725–731. [Google Scholar] [CrossRef]
- Wiewelhove, T.; Schneider, C.; Döweling, A.; Hanakam, F.; Rasche, C.; Meyer, T.; Kellmann, M.; Pfeiffer, M.; Ferrauti, A. Effects of different recovery strategies following a half-marathon on fatigue markers in recreational runners. PLOS ONE 2018, 13, e0207313. [Google Scholar] [CrossRef]
- Zainuddin, Z.; Newton, M.; Sacco, P.; Nosaka, K. Effects of massage on delayed-onset muscle soreness, swelling, and recovery of muscle function. J. Athl. Train. 2005, 40, 174–180. [Google Scholar]
| Variables | Age (years) | Height (cm) | Weight (kg) | BMI (kg/m2) | Fat (%) |
Career (years) |
|---|---|---|---|---|---|---|
| Subjects (n = 8) | 25.1 ± 2.7 | 175.8 ± 5.8 | 75.7 ± 8.1 | 24.5 ± 2.7 | 14.4 ± 6.5 | 6.1 ± 3.0 |
| Item | Angular Velocity (°/sec) | Repetitions (REP) | Recovery Time | Sets |
|---|---|---|---|---|
| Right lower extremity | 60 | 5 | 1 min 30 sec | 4 |
| Right lower extremity | 180 | 10 | ||
| Right lower extremity | 240 | 30 | ||
| Left lower extremity | 60 | 5 | ||
| Left lower extremity | 180 | 10 | ||
| Left lower extremity | 240 | 30 |
| Time Point | a (Control) |
b (FR) |
c (VFR) |
d (WBV-12) |
e (WBV-20) |
F | p | η2 | Post-hoc |
|---|---|---|---|---|---|---|---|---|---|
| Rest | 1.38 ± 0.92 | 1.63 ± 0.92 | 1.75 ± 1.39 | 2.13 ± 1.55 | 2.13 ± 1.36 | 0.522 | .720 | 0.056 | NS |
| PF | 7.25 ± 1.16 | 7.50 ± 1.41 | 7.00 ± 1.60 | 7.25 ± 1.58 | 7.50 ± 1.93 | 0.144 | .964 | 0.016 | NS |
| PR | 5.75 ± 0.89 | 3.88 ± 1.81 | 3.38 ± 1.73 | 3.88 ± 1.73 | 3.88 ± 1.64 | 2.892 | .036 * | 0.248 | a > b, c, d, e |
| Time Point | a (Control) |
b (FR) |
c (VFR) |
d (WBV-12) |
e (WBV-20) |
F | p | η2 | Post-hoc |
|---|---|---|---|---|---|---|---|---|---|
| Rest | 1.74 ± 0.44 | 1.80 ± 1.26 | 1.59 ± 0.36 | 1.47 ± 0.43 | 1.44 ± 0.57 | 0.419 | .794 | 0.046 | NS |
| PF | 10.15 ± 1.95 | 10.00 ± 1.88 | 10.07 ± 2.37 | 10.41 ± 2.18 | 9.95 ± 1.36 | 0.066 | .992 | 0.007 | NS |
| PR | 5.72 ± 1.79 | 4.70 ± 1.47 | 3.72 ± 1.01 | 5.16 ± 1.51 | 3.92 ± 0.95 | 2.937 | .034* | 0.251 | a > b, c, e |
| Item | a (Control) |
b (FR) |
c (VFR) |
d (WBV-12) |
e (WBV-20) |
F | p | η2 | Post-hoc |
|---|---|---|---|---|---|---|---|---|---|
| PR | 52.18 ± 16.82 | 67.45 ± 15.30 | 75.10 ± 8.76 | 59.73 ± 8.02 | 70.82 ± 8.02 | 4.625 | .004 ** | 0.346 | a < b, c, e |
| Item | a (Control) |
B (FR) |
c (VFR) |
d (WBV-12) |
e (WBV-20) |
F | p | η2 | Post-hoc |
|---|---|---|---|---|---|---|---|---|---|
| PF | 186.75 ± 32.94 | 192.75 ± 33.25 | 190.13 ± 25.88 | 202.50 ± 24.88 | 212.25 ± 50.77 | 0.965 | .439 | 0.099 | NS |
| PR | 176.63 ± 41.78 | 201.25 ± 41.47 | 216.13 ± 38.92 | 228.88 ± 50.94 | 252.25 ± 50.77 | 2.975 | .032 * | 0.254 | a < d, e; b < e |
| Item | a (Control) |
b (FR) |
c (VFR) |
d (WBV-12) |
e (WBV-20) |
F | p | η2 | Post-hoc |
|---|---|---|---|---|---|---|---|---|---|
| PF | 192.88 ± 25.55 | 181.50 ± 31.86 | 197.25 ± 24.92 | 205.00 ± 24.84 | 202.88 ± 30.64 | 0.910 | .469 | 0.094 | NS |
| PR | 180.75 ± 27.54 | 185.25 ± 31.21 | 212.00 ± 49.42 | 216.63 ± 47.43 | 227.63 ± 47.69 | 4.006 | .034 * | 0.314 | a, b < e |
| Item | a (Control) |
b (FR) |
c (VFR) |
d (WBV-12) |
e (WBV-20) |
F | p | η2 | Post-hoc |
|---|---|---|---|---|---|---|---|---|---|
| PF | 99.75 ± 28.84 | 102.50 ± 28.25 | 108.50 ± 14.78 | 106.25 ± 17.33 | 118.63 ± 22.30 | 0.796 | .536 | 0.083 | NS |
| PR | 99.38 ± 27.25 | 101.50 ± 22.70 | 114.63 ± 21.84 | 119.38 ± 26.17 | 129.50 ± 25.66 | 3.540 | .047 * | 0.288 | a, b < e |
| Item |
a (Control) |
b (FR) |
c (VFR) |
d (WBV-12) |
e (WBV-20) |
F | p | η2 | Post-hoc |
| PF | 100.75 ± 23.19 | 94.25 ± 24.42 | 102.63 ± 22.93 | 103.63 ± 27.42 | 101.13 ± 22.07 | 0.186 | .944 | 0.021 | NS |
| PR | 93.38 ± 25.63 | 93.38 ± 21.80 | 106.75 ± 26.05 | 109.75 ± 31.85 | 120.13 ± 32.51 | 1.350 | .271 | 0.134 | NS |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




