Submitted:
26 March 2026
Posted:
26 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
- (i)
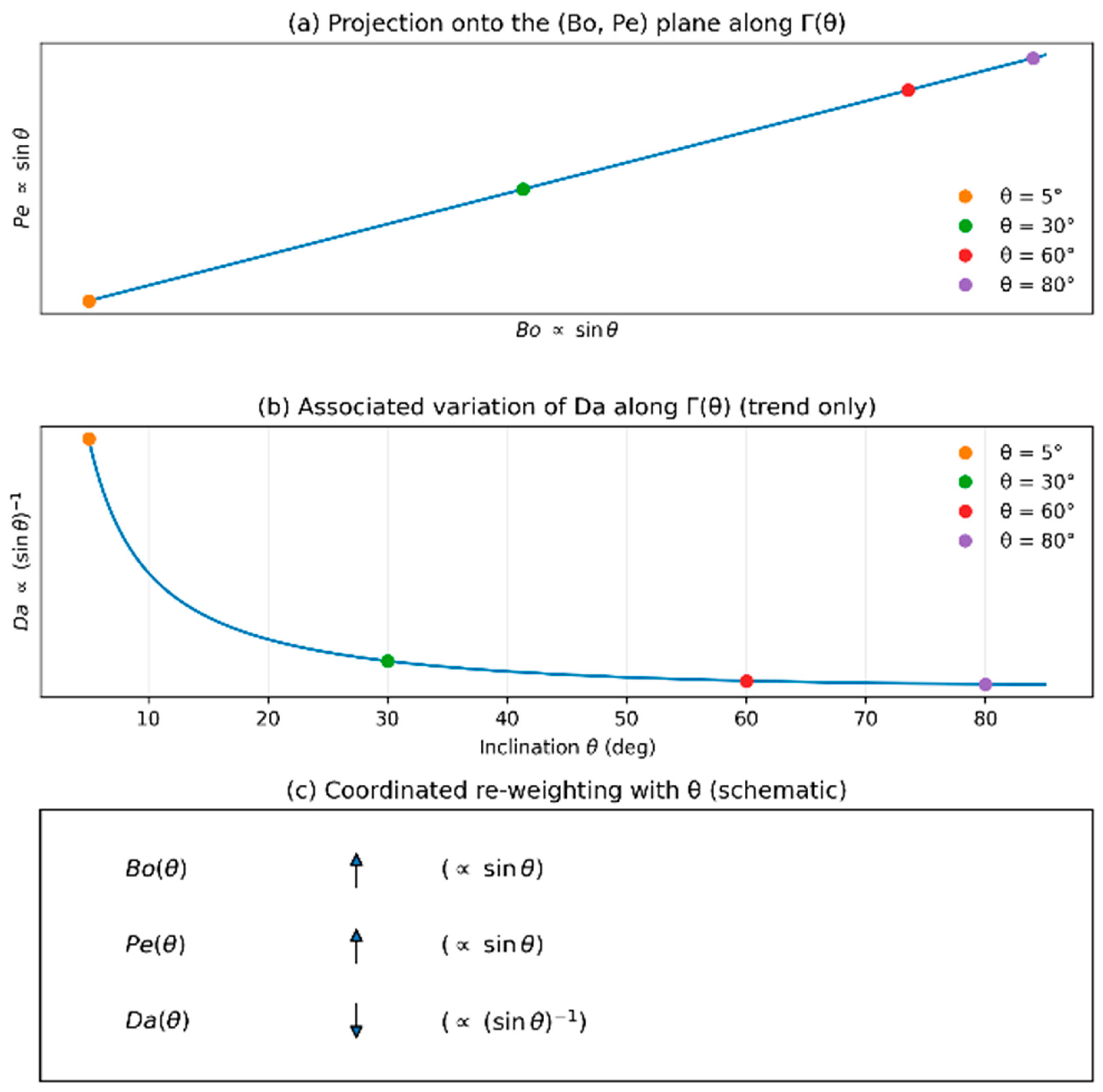
- Inclination enforces a coordinated re-weighting of the control groups such that (Bo, Pe, Da) evolve along a single geometry-imposed path Γ(θ) rather than varying independently.
- (ii)
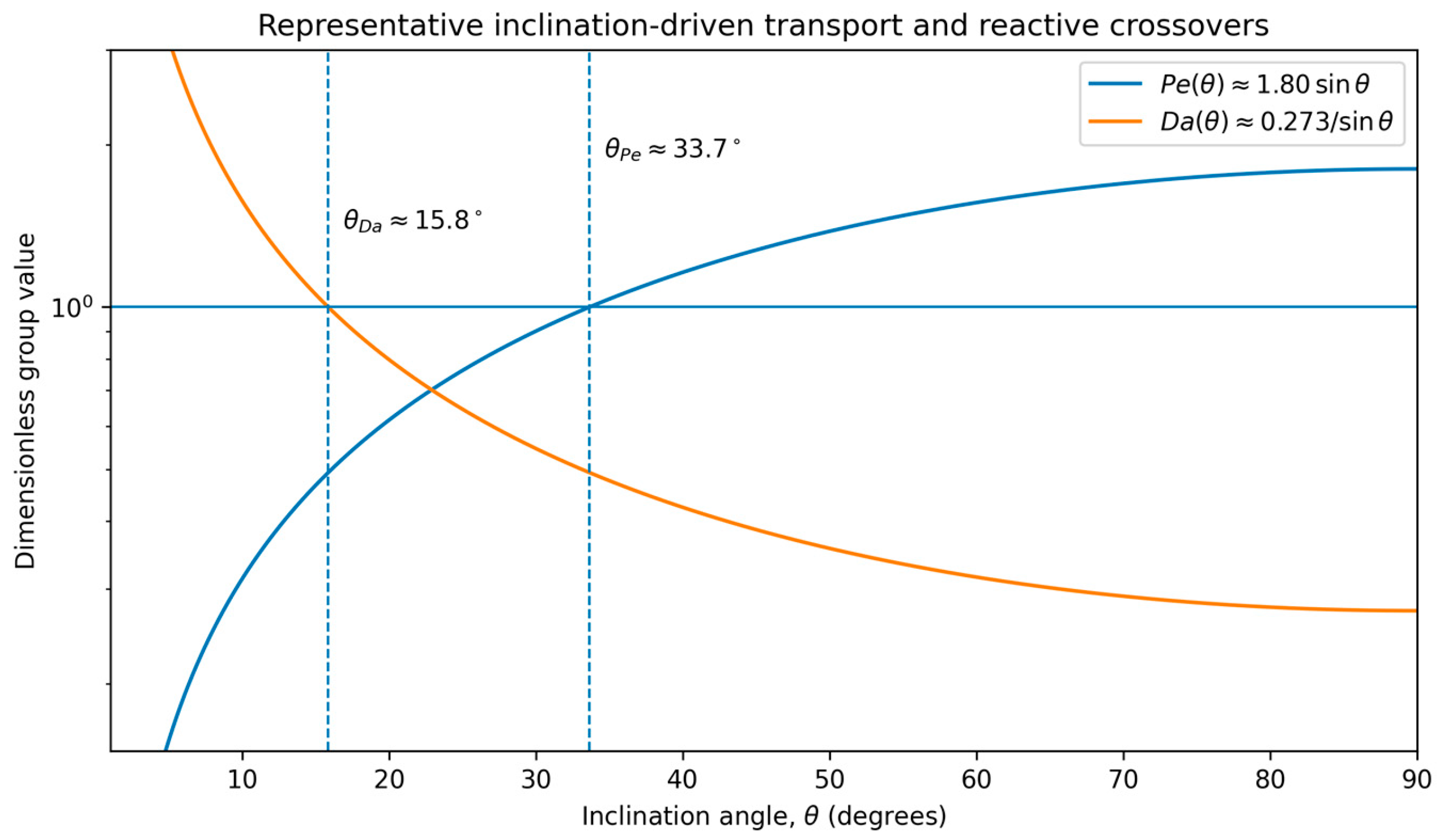
- This re-weighting produces an ordered sequence of transport and confinement crossovers (Pe ≃ 1; Da ≃ 1) that can be evaluated directly from material and environmental parameters.
- (iii)
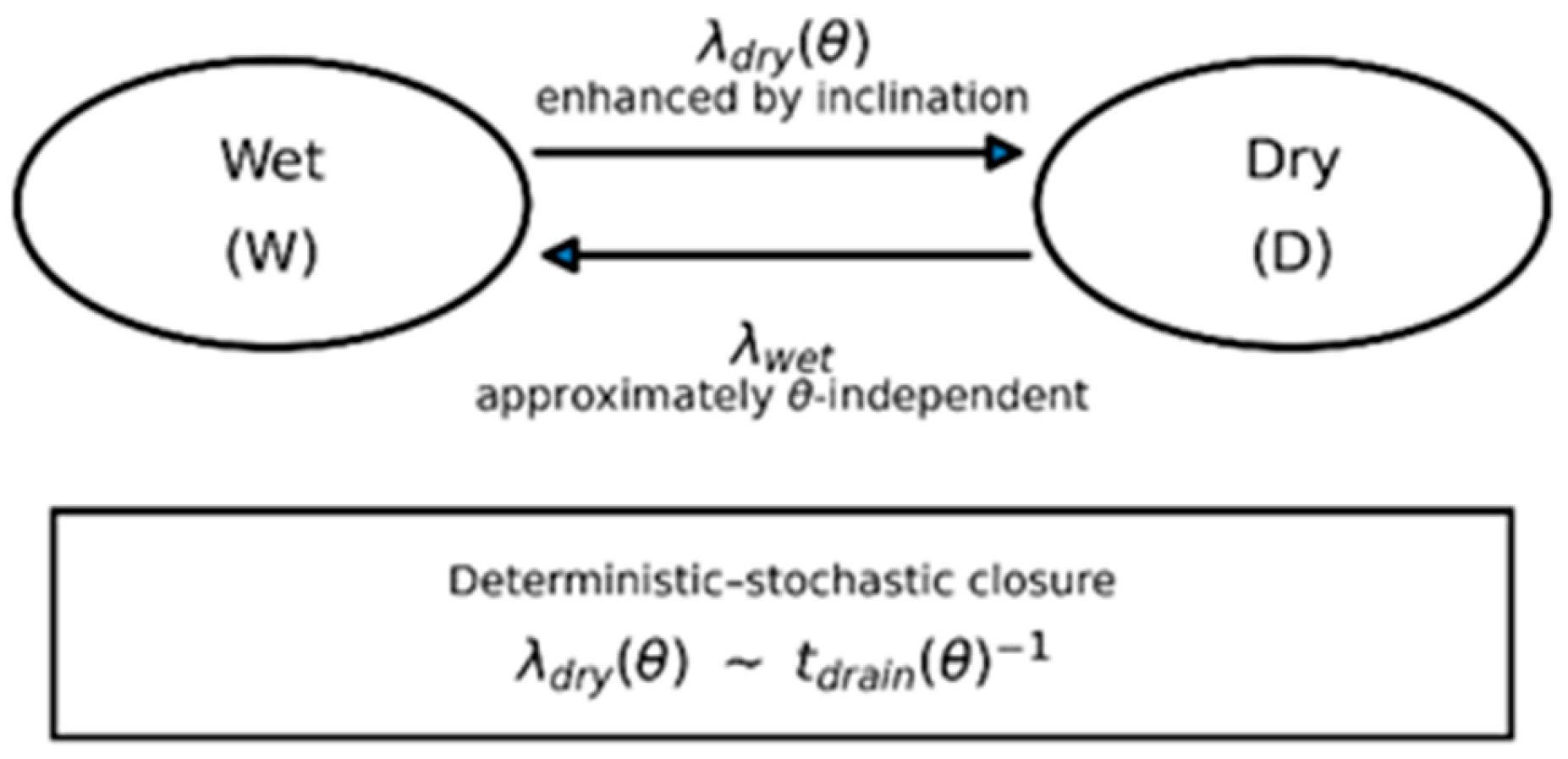
- The same structure provides closure to a minimal stochastic wet–dry model by linking the drying rate
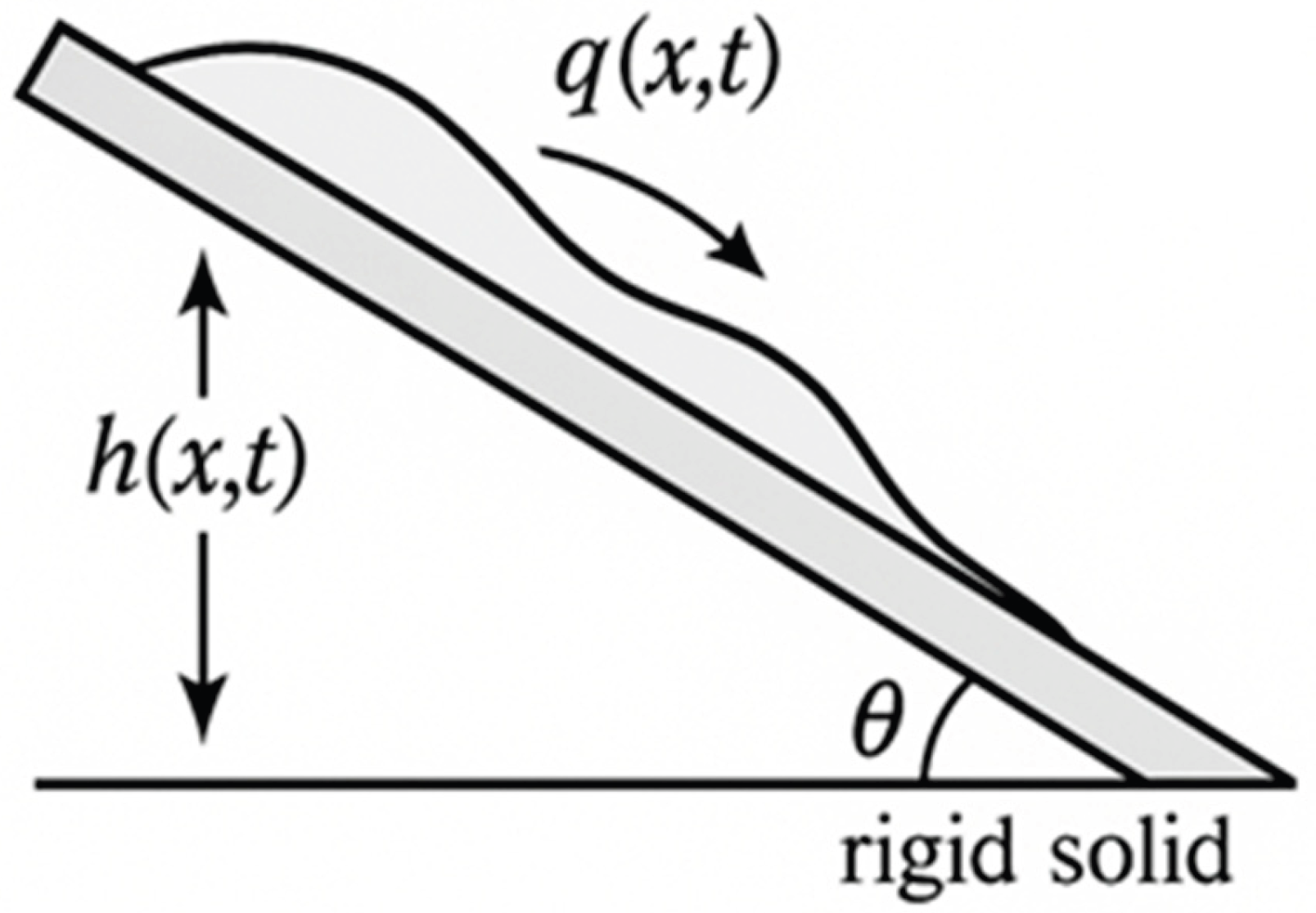
2. Governing Equations and Coupled Film Dynamics
2.1. Lubrication Framework and Gravitational Symmetry Breaking
- is viscosity,
- is surface tension,
- is the disjoining pressure,
- is the tangential gravitational forcing.
2.2. Capillary Curvature and Intermolecular Forces
2.3. Source–Sink Terms and Environmental Coupling
2.4. Confined Electrostatics in Ultrathin Films
- solid–liquid interface:
- liquid–air interface:
2.5. Ionic Advection–Diffusion and Reactive Transport
- is the depth-averaged lateral velocity,
- is the diffusivity,
- represents reactive or electrochemical processes.
2.6. Structure of the Coupled Thin-Film Fields
3. Dimensionless Formulation and the Inclination-Driven Trajectory
3.1. Characteristic Scales and Nondimensional Variables
3.2. Balance Between Capillarity, Intermolecular Forces and Gravity
- (capillarity),
- (intermolecular forces),
- (gravity),
- the gravitational contribution strengthens monotonically,
- the effective stabilizing influence of capillarity weakens in relative terms,
- the film becomes increasingly sensitive to intermolecular attraction at small thickness.
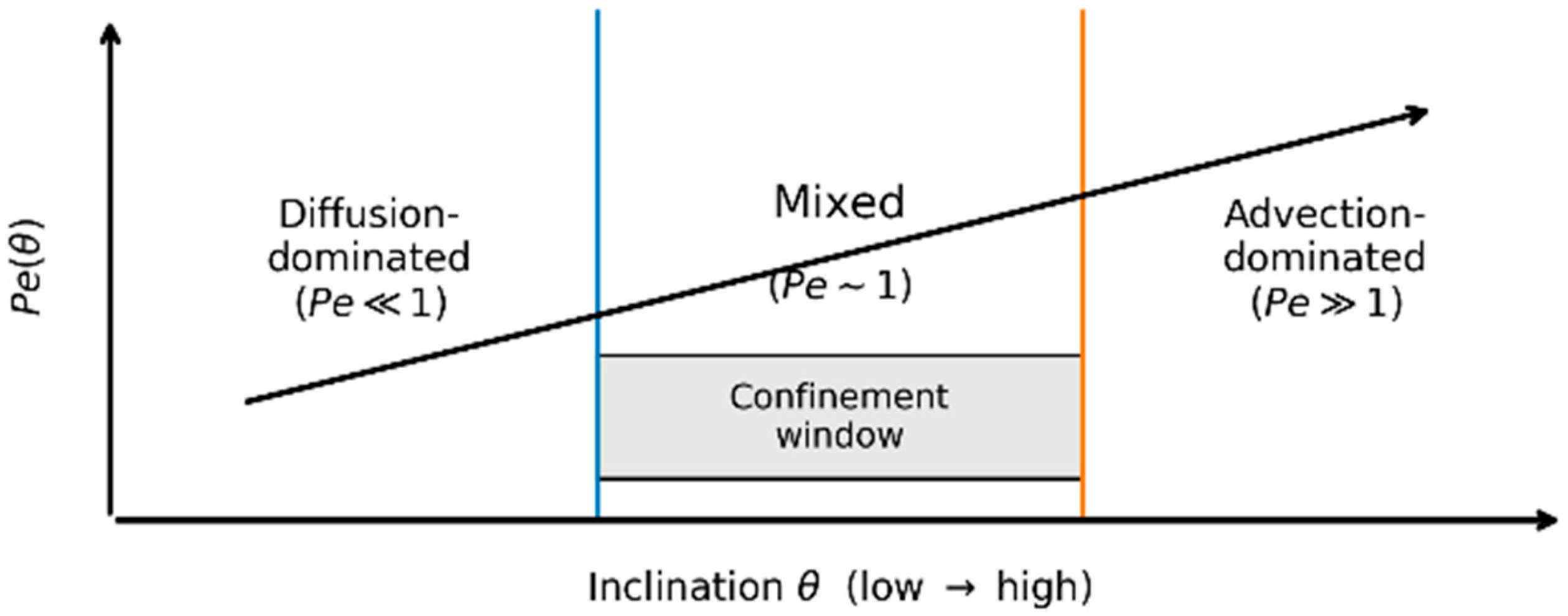
3.3. Solute Transport and the Péclet Number
-
Diffusion-dominated regime ():concentration gradients relax efficiently, and transport is primarily controlled by confinement through .
-
Intermediate regime ():advection and diffusion compete, generating longitudinal concentration gradients and coupling transport to film morphology.
-
Advection-dominated regime ():solutes are rapidly displaced downslope, residence times shorten, and the system becomes sensitive to even small variations in inclination.
3.4. Reactive Confinement and the Damköhler Number
-
Reaction-dominated regime ():solute residence times exceed reactive timescales; reactivity is governed primarily by film thickness and intermolecular interactions.
-
Transport-limited regime ():advection removes solute faster than it can react; reaction becomes constrained by supply rather than kinetics.
-
Coupled regime ():comparable timescales lead to strong sensitivity to local variations in , and confined electrostatics.
3.5. Inclination-Driven Trends in Space
3.6. Representative Parameterization and Crossover Angles
3.7. Implications for Film Stability, Transport and Rupture
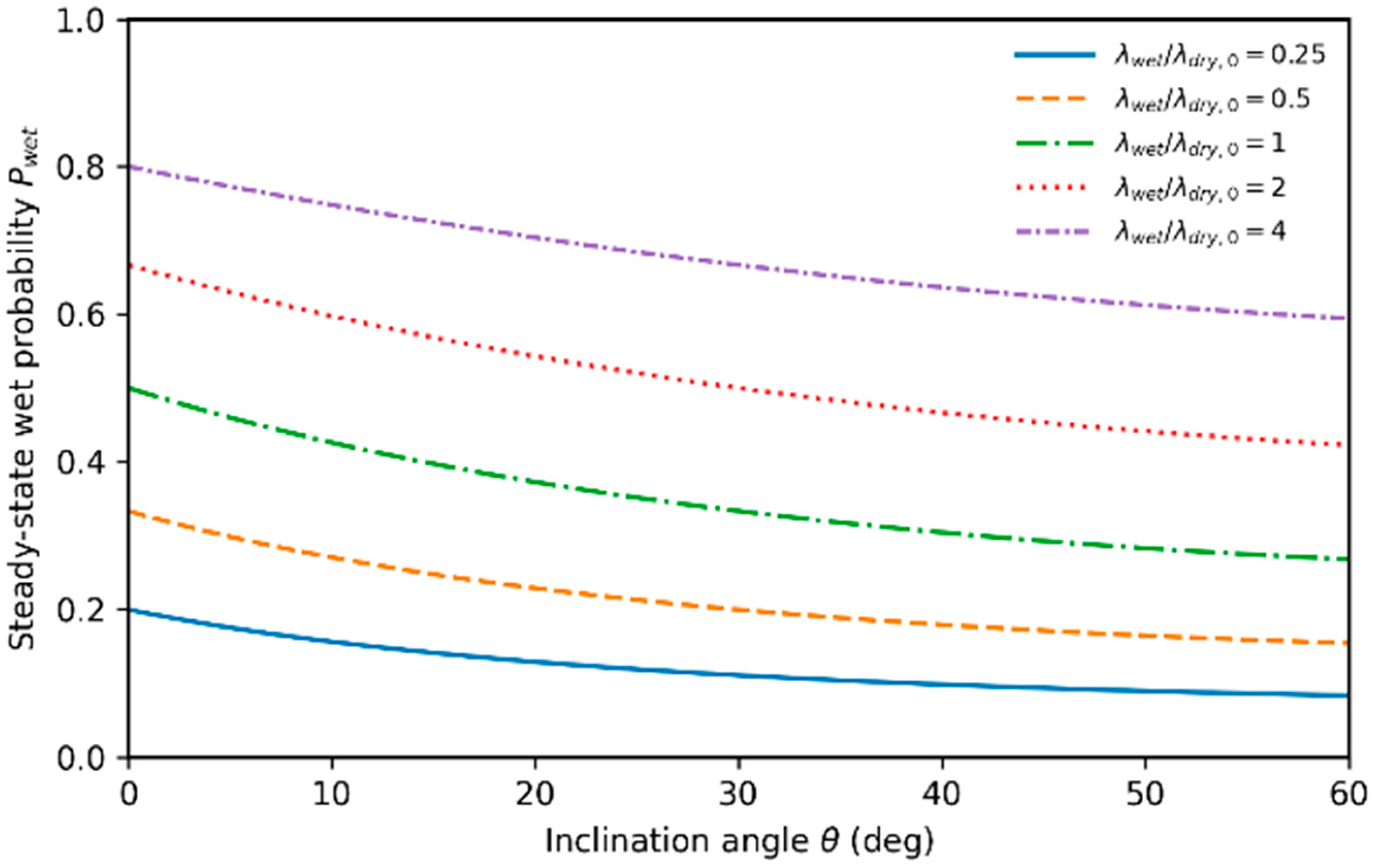
4. Stochastic Wetting and Time-of-Wetness Dynamics
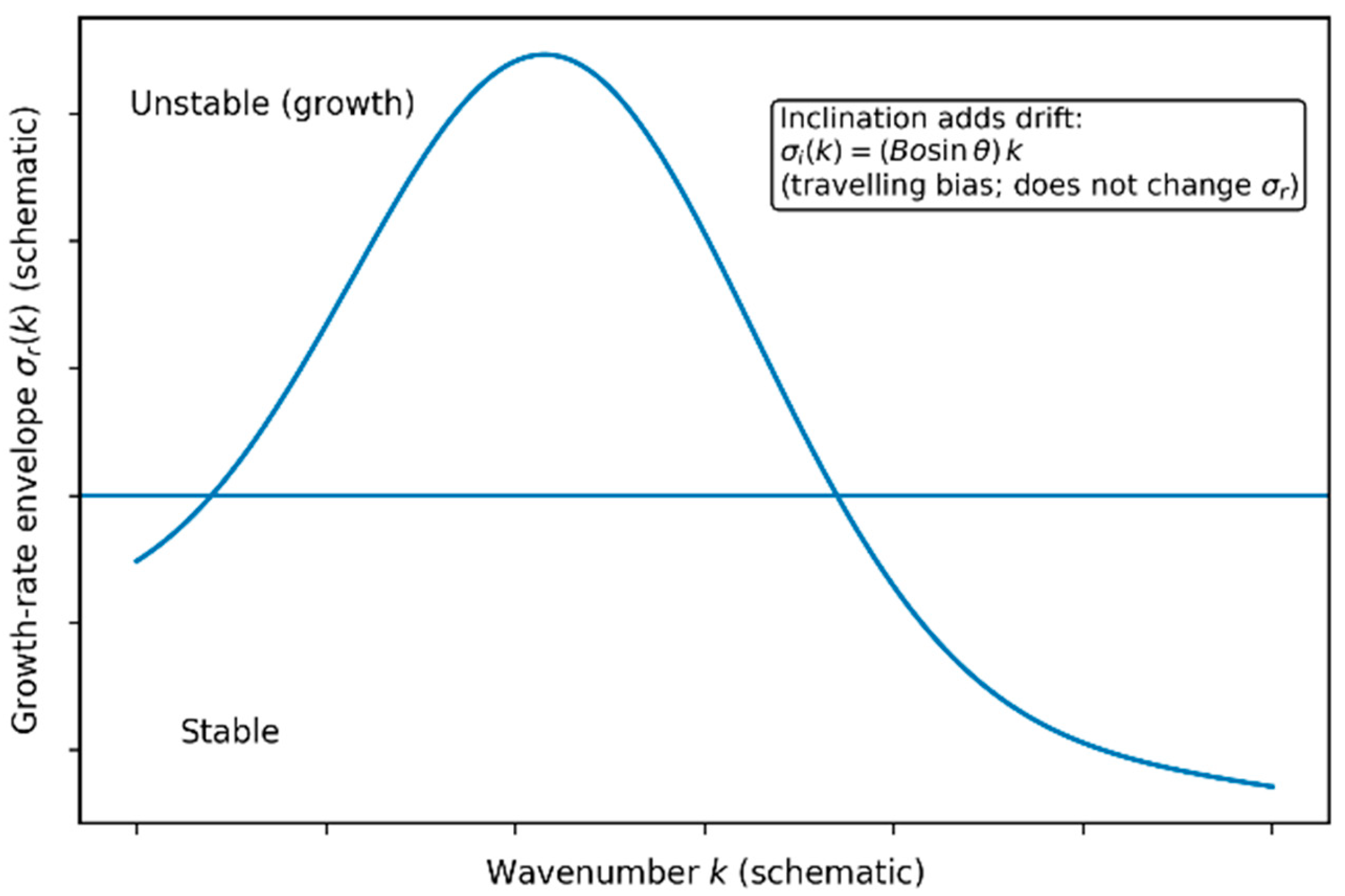
4.1. Deterministic Thin-Film Dynamics and Rupture Thresholds
4.2. Stochastic Transition Between Wet and Dry States
- a wet state (W), where a continuous or metastable film exists, and
- a dry state (D), where the film has ruptured or is absent.
4.3. Wet-State Persistence and Time-Of-Wetness
4.4. Coupling Deterministic Dynamics to Stochastic Transitions
4.5. Implications for Interfacial Reactivity and Transport
5. Physical Consequences of the Inclination-Driven Trajectory
5.1. Stability Regimes Under Inclination
5.2. Drainage and Thinning Under Inclination
5.3. Transport Regimes: Advection, Diffusion, and Confinement
5.4. Inclination Effects on Reactive Confinement
5.5. Long-Time Intermittency and Wet-State Persistence
6. Discussion
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| Bo | Bond number |
| Pe | Péclet number |
| Da | Damköhler number |
| EDL | Electric double layer |
| PDE | Partial differential equation |
References
- de Gennes, P.-G. Wetting: Statics and dynamics. Reviews of Modern Physics 1985, 57(3), 827–863. [Google Scholar] [CrossRef]
- Snoeijer, J. H.; Andreotti, B. Moving contact lines: Scales, regimes, and dynamical transitions. Annual Review of Fluid Mechanics 2013, 45(1), 269–292. [Google Scholar] [CrossRef]
- Oron, A.; Davis, S. H.; Bankoff, S. G. Long-scale evolution of thin liquid films. Reviews of Modern Physics 1997, 69(3), 931–980. [Google Scholar] [CrossRef]
- Craster, R. V.; Matar, O. K. Dynamics and stability of thin liquid films. Reviews of Modern Physics 2009, 81(3), 1131–1198. [Google Scholar] [CrossRef]
- Starov, V. M.; Velarde, M. G.; Radke, C. J. Wetting and spreading dynamics; CRC Press, 2007. [Google Scholar]
- Derjaguin, B. V.; Churaev, N. V.; Muller, V. M. Surface forces; Springer, 1987. [Google Scholar]
- Seemann, R.; Herminghaus, S.; Jacobs, K. Dewetting patterns and molecular forces: A reconciliation. Physical Review Letters 2001, 86(24), 5534–5537. [Google Scholar] [CrossRef] [PubMed]
- Thiele, U.; Velarde, M. G.; Neuffer, K. Dewetting: Film rupture by nucleation in the spinodal regime. Physical Review Letters 2001, 87(1), 016104. [Google Scholar] [CrossRef] [PubMed]
- Vrij, A. Possible mechanism for the spontaneous rupture of thin, free liquid films. Discussions of the Faraday Society 1966, 42, 23–33. [Google Scholar] [CrossRef]
- Cook, B. P.; Bertozzi, A. L.; Hosoi, A. E. Shock solutions for particle-laden thin films. SIAM Journal on Applied Mathematics 2008, 68(3), 760–783. [Google Scholar] [CrossRef]
- Bonn, D.; Eggers, J.; Indekeu, J.; Meunier, J.; Rolley, E. Wetting and spreading. Reviews of Modern Physics 2009, 81(2), 739–805. [Google Scholar] [CrossRef]
- Santana, J. J.; Cano, V.; Vasconcelos, H. C.; Souto, R. M. The influence of test-panel orientation and exposure angle on the corrosion rate of carbon steel: Mathematical modelling. Metals 2020, 10(2), 196. [Google Scholar] [CrossRef]
- Chatzigiannakis, E.; Jaensson, N.; Vermant, J. Thin liquid films: Where hydrodynamics, capillarity, surface stresses and intermolecular forces meet. Current Opinion in Colloid & Interface Science 2021, 53, 101441. [Google Scholar] [CrossRef]
- Rednikov, A.; Colinet, P. Singularity-free description of moving contact lines for volatile liquids. Physical Review E 2013, 87(1), 010401(R). [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Stone, H. A. Spreading of complex fluids with a soft blade. Physical Review Fluids 2022, 7(8), 084002. [Google Scholar] [CrossRef]
- Kumawat, T. C. Thin film flow inside a uniformly heated/cooled inclined rotating cylinder. Physics of Fluids 2024, 36(4), 042103. [Google Scholar] [CrossRef]
- Israelachvili, J. N.; Adams, G. E. Measurement of forces between two mica surfaces in aqueous electrolyte solutions in the range 0–100 nm. Journal of the Chemical Society, Faraday Transactions 1978, 1(74(4)), 975–1001. [Google Scholar] [CrossRef]
- Oron, A.; Bankoff, S. G. Dewetting of a heated surface by an evaporating liquid film under conjoining/disjoining pressures. Journal of Colloid and Interface Science 1999, 218(1), 152–166. [Google Scholar] [CrossRef] [PubMed]
- Ajaev, V. S. Spreading of thin volatile liquid droplets on uniformly heated surfaces. Journal of Fluid Mechanics 2005, 528, 279–296. [Google Scholar] [CrossRef]
- Cazabat, A.-M.; Guéna, G. Evaporation of macroscopic sessile droplets. Soft Matter 2010, 6(12), 2591–2612. [Google Scholar] [CrossRef]
- Harvey, A. H. Improved and always improving—Reference formulations for thermophysical properties of water. Journal of Physical and Chemical Reference Data 2023, 52(1), 011501. [Google Scholar] [CrossRef]
- Spiesz, P.; Ballari, M. M.; Brouwers, H. J. H. RCM: A new model accounting for the non-linear chloride binding isotherm and the non-equilibrium conditions between the free- and bound-chloride concentrations. Construction and Building Materials 2012, 27(1), 293–304. [Google Scholar] [CrossRef]
| Parameter | Value |
|---|---|
| H | 1 μm |
| L | 1 mm |
| ρ | 997 kg m−3 |
| μ | 0.89×10−3 Pa s |
| γ | 71.97×10−3 N m−1 |
| D | 2.03×10−9 m2 s−1 |
| k a | 10−3 s−1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




