Submitted:
24 March 2026
Posted:
25 March 2026
You are already at the latest version
Abstract
Keywords:
Introduction
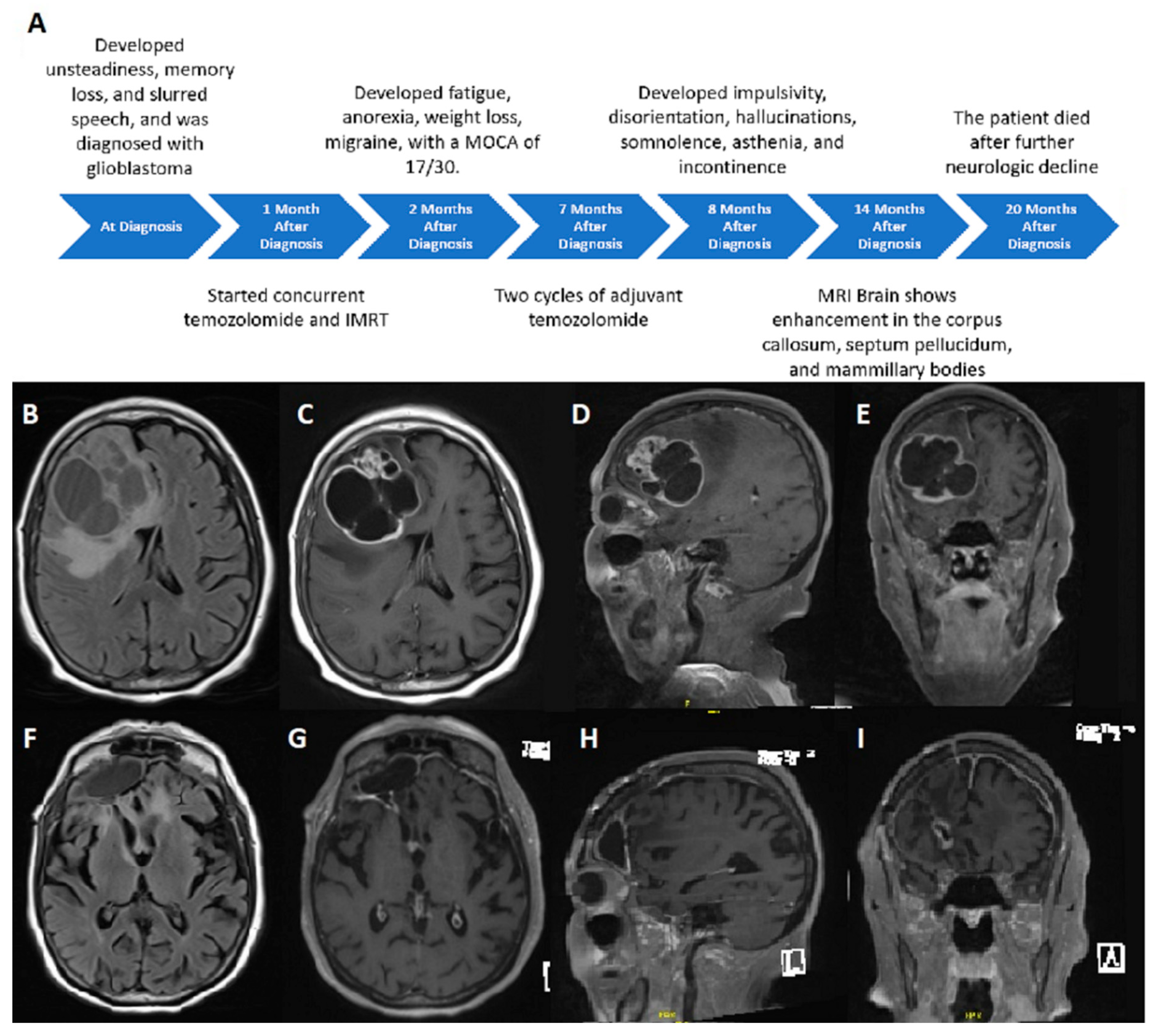
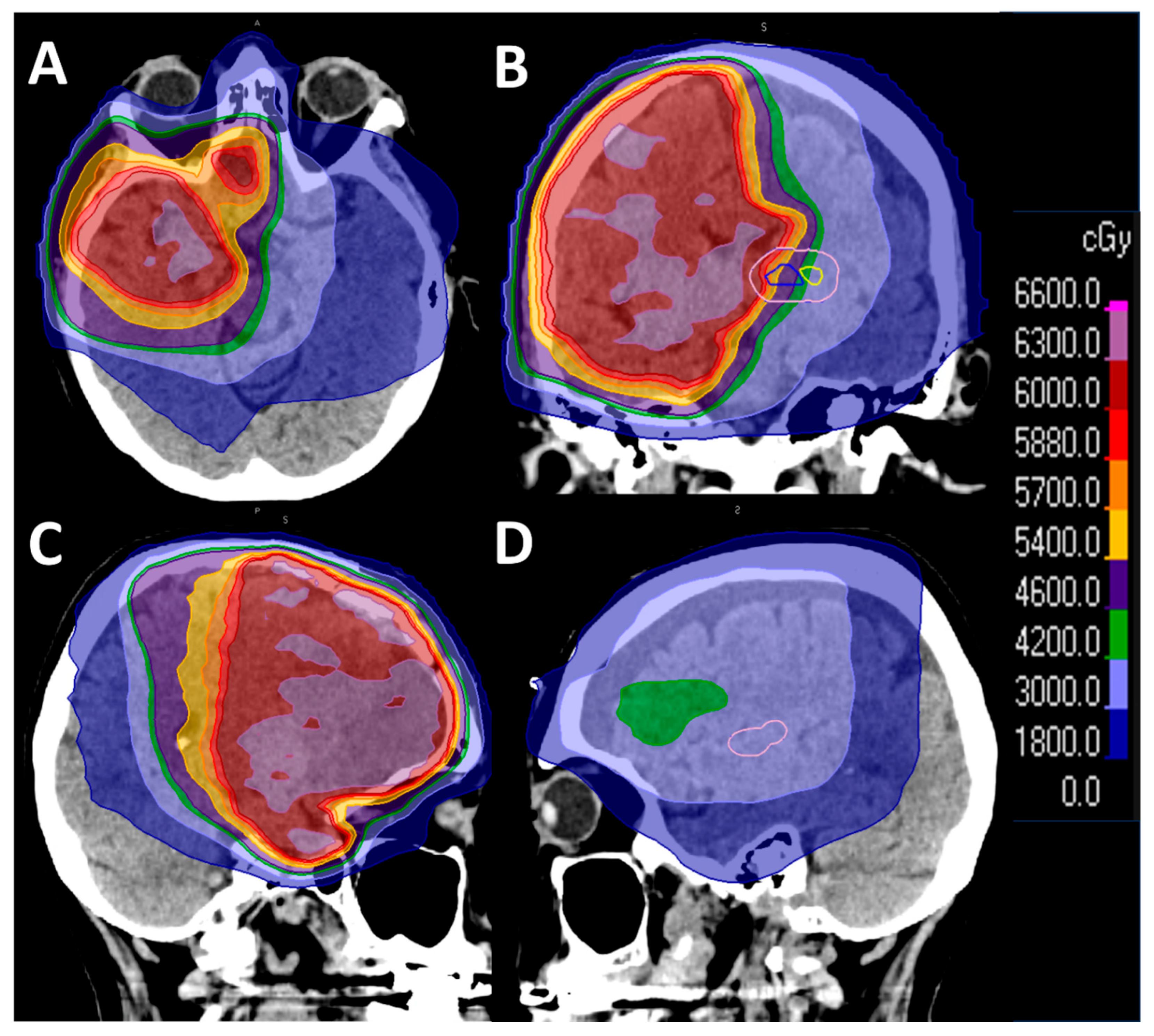
Case Description
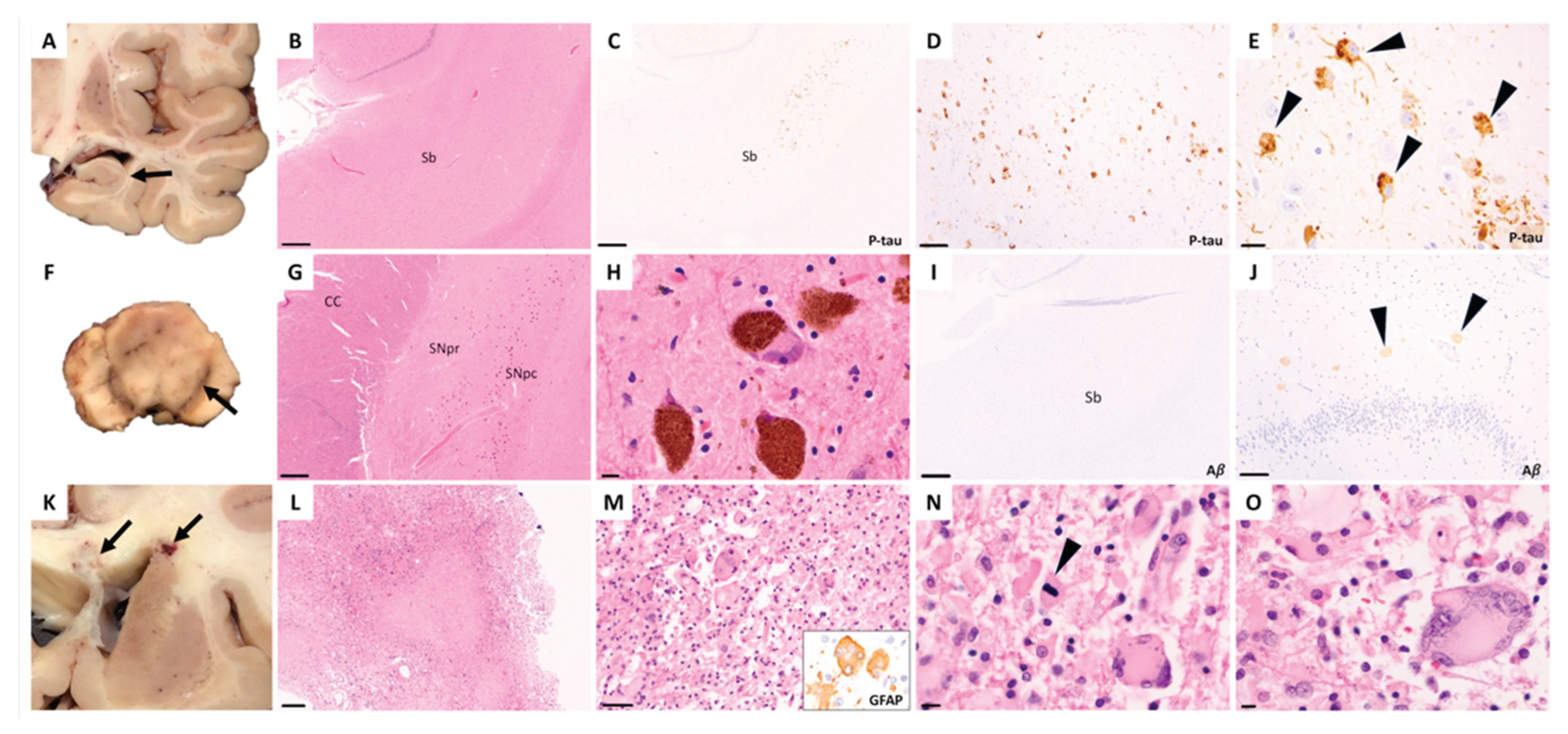
Discussion
Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
List of abbreviations
References
- Stupp, R; Hegi, ME; Mason, WP; et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 2009, 10(5), 459–466. [Google Scholar] [CrossRef] [PubMed]
- Ironside, S; Das, S; Sahgal, A; Moroney, C; Mainprize, T; Perry, JR. Optimal Therapies for Newly Diagnosed Elderly Patients with Glioblastoma. Curr Treat Options Oncol. 2017, 18(11), 66. [Google Scholar] [CrossRef] [PubMed]
- Crary, JF; Trojanowski, JQ; Schneider, JA; et al. Primary age-related tauopathy (PART): a common pathology associated with human aging. Acta Neuropathol. 2014, 128(6), 755–766. [Google Scholar] [CrossRef] [PubMed]
- Teylan, M; Mock, C; Gauthreaux, K; et al. Cognitive trajectory in mild cognitive impairment due to primary age-related tauopathy. Brain 2020, 143(2), 611–621. [Google Scholar] [CrossRef] [PubMed]
- Mondia, MWL; Kritselis, MA; Donahue, JE; et al. Dimorphic glioblastoma with glial and epithelioid phenotypes: Clonal evolution and immune selection. Frontiers in Neurology. 2023, 13. Available online: https://www.frontiersin.org/articles/10.3389/fneur.2022.1017087 (accessed on 30 January 2024).
- Nasreddine, ZS; Phillips, NA; Bédirian, V; et al. The Montreal Cognitive Assessment, MoCA: A brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005, 53(4), 695–699. [Google Scholar] [CrossRef] [PubMed]
- Duyckaerts, C; Braak, H; Brion, JP; et al. PART is part of Alzheimer disease. Acta Neuropathol. 2015, 129(5), 749–756. [Google Scholar] [CrossRef] [PubMed]
- Wong, ET; Rosenberg, H; Dawood, O; et al. Lewy body disease as a potential negative outcome modifier of glioblastoma treatment: a case report. BMC Neurol. 2023, 23(1), 257. [Google Scholar] [CrossRef] [PubMed]
- Ossenkoppele, R; Rabinovici, GD; Smith, R; et al. Discriminative Accuracy of [18F] flortaucipir Positron Emission Tomography for Alzheimer Disease vs Other Neurodegenerative Disorders. JAMA 2018, 320(11), 1151–1162. [Google Scholar] [CrossRef] [PubMed]
- Klunk, WE; Engler, H; Nordberg, A; et al. Imaging brain amyloid in Alzheimer’s disease with Pittsburgh Compound-B. Ann Neurol. 2004, 55(3), 306–319. [Google Scholar] [CrossRef] [PubMed]
- Chen, X; Cassady, KE; Adams, JN; Harrison, TM; Baker, SL; Jagust, WJ. Regional Tau Effects on Prospective Cognitive Change in Cognitively Normal Older Adults. Journal of Neuroscience 2021, 41(2), 366–375. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z; Li, YQ; Wong, CS. Effects of Aging on Hippocampal Neurogenesis After Irradiation. International Journal of Radiation Oncology*Biology*Physics 2016, 94(5), 1181–1189. [Google Scholar] [CrossRef] [PubMed]
- Lensu, S; Waselius, T; Mäkinen, E; et al. Irradiation of the head reduces adult hippocampal neurogenesis and impairs spatial memory, but leaves overall health intact in rats. Eur J Neurosci. 2021, 53(6), 1885–1904. [Google Scholar] [CrossRef] [PubMed]
- Leskinen, S; Shah, HA; Yaffe, B; et al. Hippocampal avoidance in whole brain radiotherapy and prophylactic cranial irradiation: a systematic review and meta-analysis. J Neurooncol 2023, 163(3), 515–527. [Google Scholar] [CrossRef] [PubMed]
- Gondi, V; Pugh, SL; Tome, WA; et al. Preservation of memory with conformal avoidance of the hippocampal neural stem-cell compartment during whole-brain radiotherapy for brain metastases (RTOG 0933): a phase II multi-institutional trial. J Clin Oncol. 2014, 32(34), 3810–3816. [Google Scholar] [CrossRef] [PubMed]
- Thippu Jayaprakash, K; Wildschut, K; Jena, R. Feasibility of hippocampal avoidance radiotherapy for glioblastoma. Clin Oncol (R Coll Radiol) 2017, 29(11), 748–752. [Google Scholar] [CrossRef] [PubMed]
- Gibbons, CH; Levine, T; Adler, C; et al. Skin biopsy detection of phosphorylated α-synuclein in patients with synucleinopathies. JAMA 2024, 331(15), 1298–1306. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



