Submitted:
23 March 2026
Posted:
25 March 2026
You are already at the latest version
Abstract
Developmental engineering (DE) is a bottom-up strategy for generating functional tissues from modular tissues (MTs), overcoming limitations of conventional top-down approach. This study integrates theoretical simulations with empirical correlations to analyse microcarrier aggregation, oxygen transport, suspension conditions, and cell damage in stirred bioreactors, providing guidance for scalable MT production in DE. Microcarrier aggregates were modelled to evaluate minimum oxygen concentration (Cmin). Results show that larger microcarrier diameters (dmc) increase Cmin because of longer diffusion distances. Aggregate geometry and packing configuration, including hexagonal close packing and the “kissing number,” influence oxygen limitation and explain observed Cmin plateaus. Hydrodynamic behaviour was assessed using Zwietering correlation and Kolmogorov turbulence scaling. Denser microcarrier aggregates require higher minimum stirring speeds (Nmin), while larger dmc increases susceptibility to shear. Aggressive impeller designs and higher revolutions per minute reduce Nmin but increase collision-induced cell damage. In contrast, higher medium density (e.g., 20% FBS) reduces shear stress and energy dissipation. A unified framework is proposed that integrates oxygen diffusion, aggregate geometry, microcarrier properties, and hydrodynamics to predict worst-case oxygen limitation and cell damage. The results clarify trade-offs between impeller design, agitation intensity, and aggregation, supporting scalable MT production using individual or aggregated modular scaffolds for DE-based tissue assembly.
Keywords:
1. Introduction
2. Methodology
2.1. Modeling Oxygen Diffusion in 3D Tissue Constructs
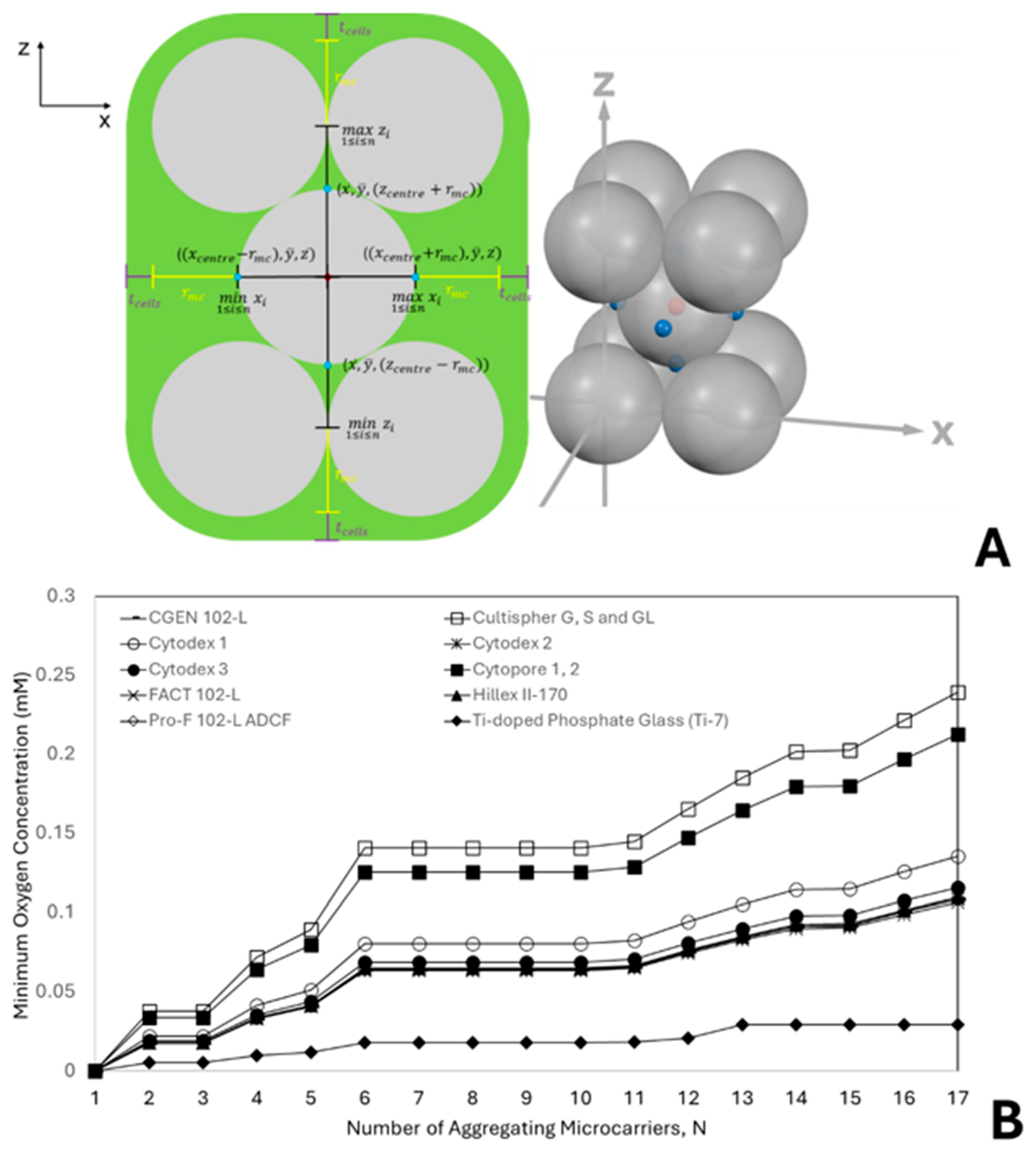
2.2. Geometric Modelling of Microcarrier Aggregation and Diffusion Distance Estimation
2.3. Modeling Hydrodynamics and Cell–Microcarrier Interactions in Stirred Bioreactors
3. Results and Discussions
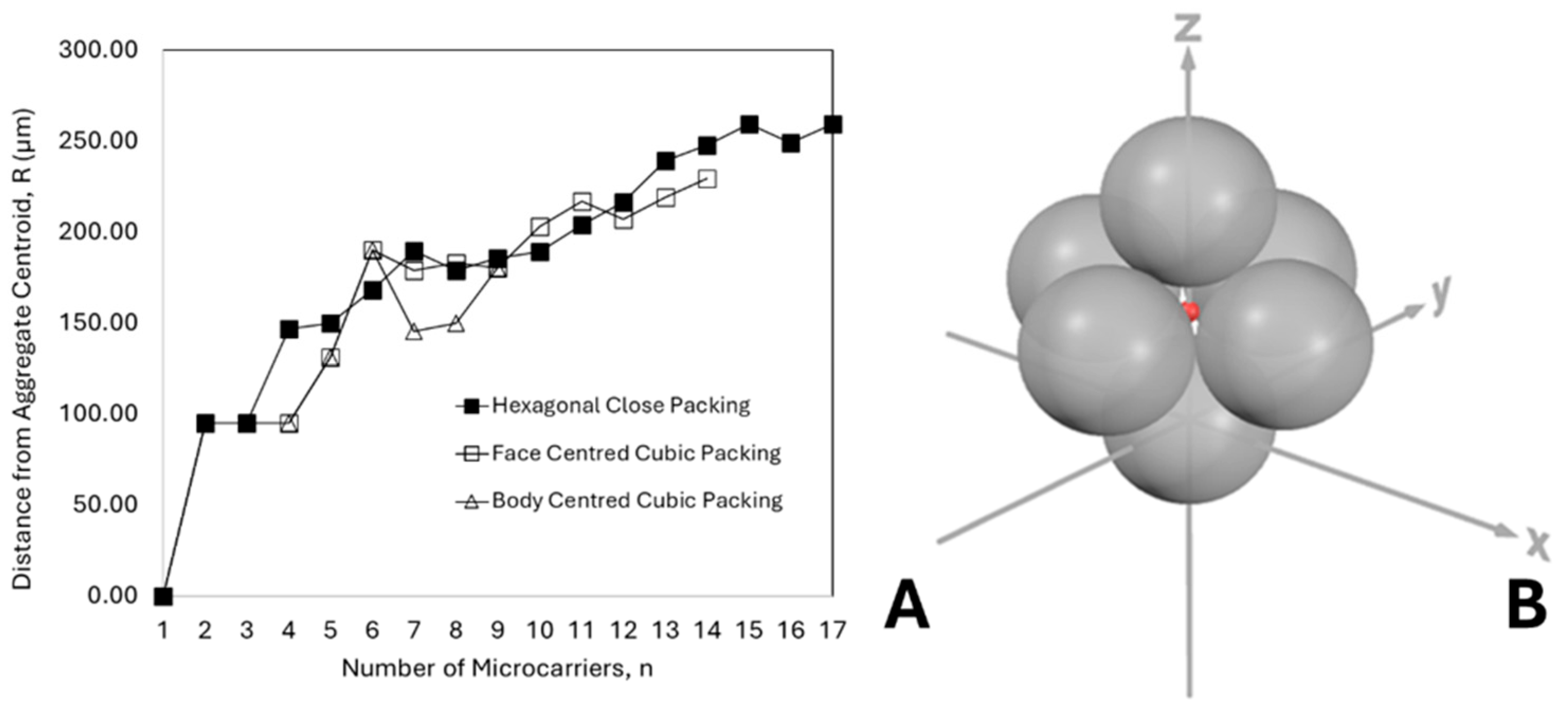
3.1. Influence of Aggregate Geometry and Microcarrier Size on Oxygen Limitation
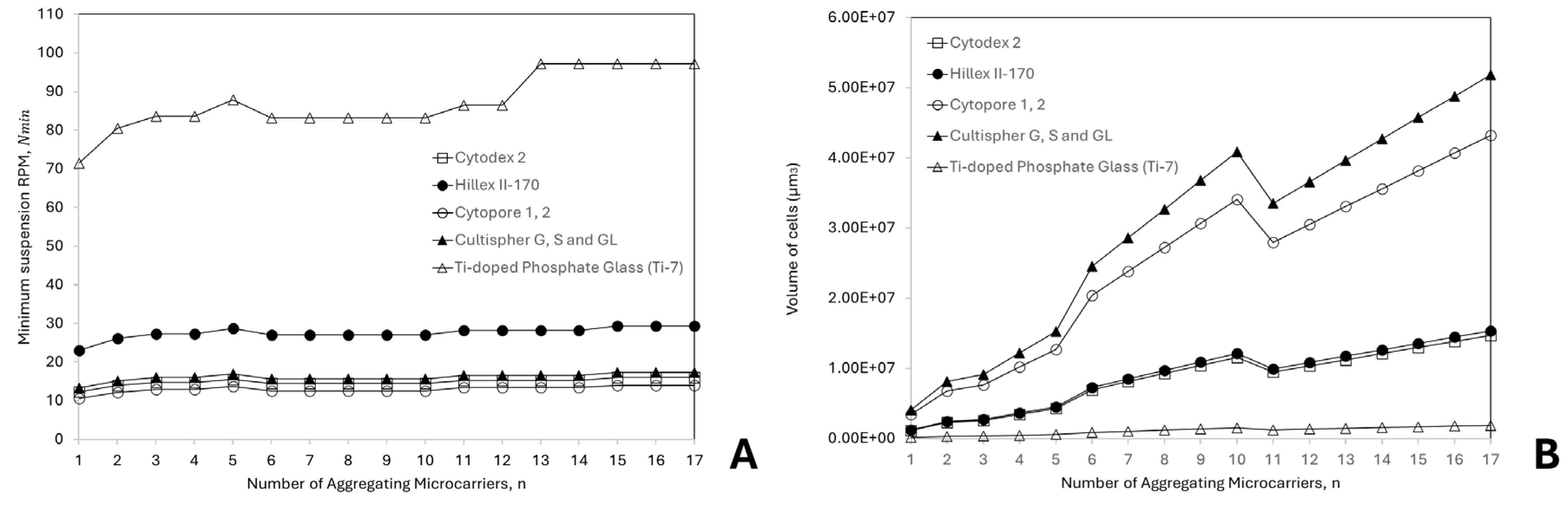
3.2. Effects of Microcarrier Properties and Aggregation on Suspension and Cell Damage
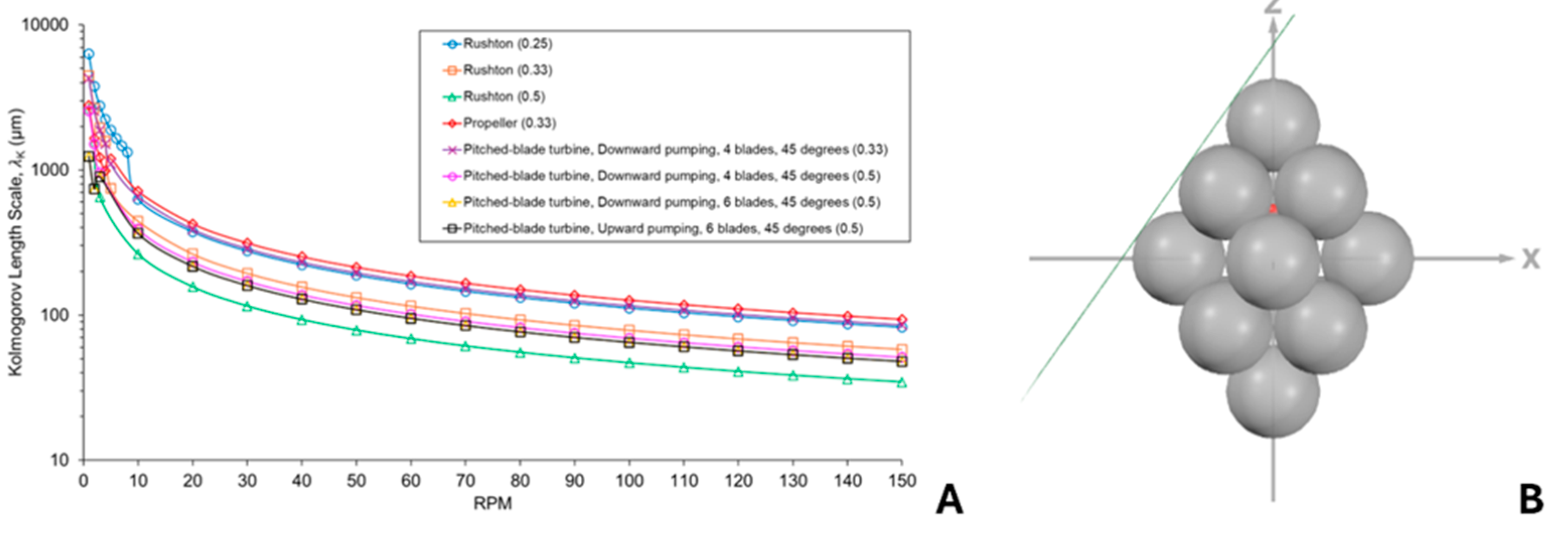
3.3. Effects of Agitation and Impeller Design on Cell Damage Risk
3.4. Model Assumptions, Limitations, and Applicability
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| Symbol | Description | Units |
| Initial oxygen concentration in the media | ||
| Minimum oxygen concentration in the media to move across the diffusion distance to the centroid | or | |
| Minimum stirring speed for suspension | ||
| Minimum agitation speed for turbulence | ||
| Power number | ||
| The maximum tissue depth | ||
| Overall volume of aggregate | ||
| Volume of cells per aggregate | ||
| Distance of impeller from tank bottom | ||
| Aggregate diameter (Feret’s diameter) | ||
| Impeller diameter | ||
| Microcarrier diameter | ||
| Microcarrier diameter | ||
| Diameter of stirred tank | ||
| Mass transfer coefficient | ||
| Number of blades | ||
| Radius of microcarrier | ||
| Cell thickness | ||
| Impeller width | ||
| Centroid X-coordinate in aggregation space | ||
| Centroid relocated to microcarrier surface X-coordinate in aggregation space | ||
| Centroid Y-coordinate in aggregation space | ||
| Centroid relocated to microcarrier surface Y-coordinate in aggregation space | ||
| Centroid Z-coordinate in aggregation space | ||
| Centroid relocated to microcarrier surface Z-coordinate in aggregation space | ||
| Shear rate | ||
| Total energy dissipation per unit mass of fluid | ||
| Agitator efficiency | ||
| Smallest characteristic eddy size | ||
| Dynamic viscosity of media | ||
| Kinematic viscosity of media | ||
| Aggregate density | ||
| Cell density | ||
| Microcarrier density | ||
| Density of media | ||
| Azimuthal diffusion distance | ||
| Oxygen concentration in the media | ||
| Diffusion coefficient | ||
| Euclidean distance | ||
| Stirring speed | ||
| Power | ||
| Diffusion distance to the centroid | ||
| Reynolds number | ||
| Empirically derived constant based on impeller design and reactor geometry | ||
| Tissue thickness | ||
| Mass fraction of microcarriers | ||
| Gravitational acceleration | ||
| Index of summation for microcarrier / Microcarrier sequence number | ||
| Microcarrier sequence number | ||
| Shear rate constant | ||
| Number of microcarriers within an aggregate | ||
| Radial diffusion distance | ||
| Single cell oxygen consumption rate | ||
| Time | ||
| X-coordinate in aggregation space | ||
| Y-coordinate in aggregation space | ||
| Z-coordinate in aggregation space | ||
| Packing fraction | ||
| Polar diffusion distance | ||
| Shear stress | ||
| Metabolic consumption of oxygen |
Appendix A
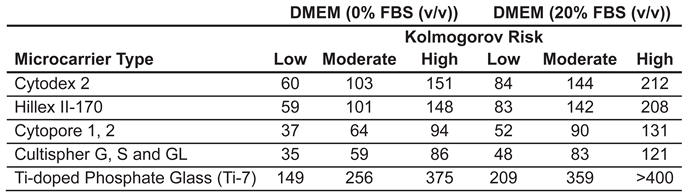
Appendix A.1–Summary of Model Microcarrier Types and Corresponding Parameters Available Within the Simulation
| Microcarrier Type | Matrix | Reference | ||
| Cytodex 1 | 190 ± 58 | 1.03 ∗ 103 | Dextran (positively charged) | [19,62,6]] |
| Cytodex 2 | 167.5 ± 32.5 | 1.04 ∗ 103 | Cotton cellulose | [19,62,66] |
| Cytodex 3 | 175 ± 36 | 1.04 ∗ 103 | Dextran (collagen coated) | [19,62,66] |
| Hillex II-170 | 170 ± 10 | 1.12 ∗ 103 | Polystyrene (cationic trimethyl ammonium coated) | [64] |
| Pro-F 102-L ADCF | 169 ± 44 | 1.02 ∗ 103 | Plastic (ProNectin F® coated) | [66] |
| FACT 102-L | 169 ± 44 | 1.02 ∗ 103 | Polystyrene (collagen coated) | [66] |
| CGEN 102-L | 169 ± 44 | 1.02 ∗ 103 | Polystyrene (collagen coated) | [66] |
| Cytopore 1, 2 | 240 ± 40 | 1.03 ∗ 103 | Cellulose | [19,62,66] |
| Cultispher G, S and GL | 255 ± 125 | 1.04 ∗ 103 | Gelatin | [17,19,62] |
| Ti-doped Phosphate Glass (Ti-7) | 84.5 ± 21.5 | 2.75 ∗ 103 | Phosphate Glass and Titanium | [63] |
Appendix A.2–Summary of Model Medium and Corresponding Parameters Simulated [65]
| Media | |||
| 9.93 ∗ 102 | 6.91 ∗ 10−4 | 6.96 ∗ 10−7 | |
| 9.98 ∗ 102 | 1.00 ∗ 10−3 | 1.00 ∗ 10−6 | |
| DMEM (high glucose & 0% FBS (v/v)) | 1.00 ∗ 103 | 7.31 ∗ 10−4 | 7.31 ∗ 10−7 |
| DMEM (high glucose & 5% FBS (v/v)) | 1.00 ∗ 103 | 8.62 ∗ 10−4 | 8.60 ∗ 10−7 |
| DMEM (high glucose & 10% FBS (v/v)) | 1.01 ∗ 103 | 9.30 ∗ 10−4 | 9.22 ∗ 10−7 |
| DMEM (high glucose & 20% FBS (v/v)) | 1.02 ∗ 103 | 1.05 ∗ 10−3 | 1.03 ∗ 10−6 |
Appendix B–Full Derivation for Oxygen Diffusion Through Tissues
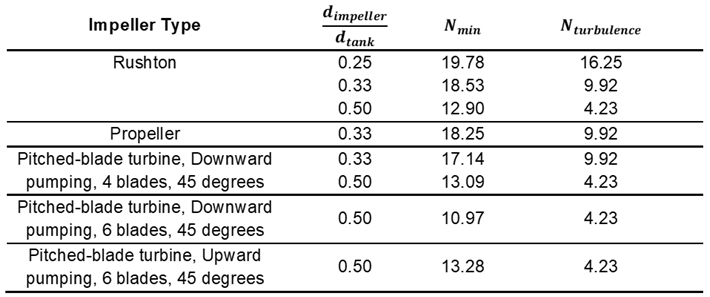
Appendix C–Geometric Parameters for Flat-Bottom, Cylindrical Vessel with Four Baffles of Width 1/10 the Tank Diameter and a Liquid Height Equal to the Tank Diameter Bioreactors Used for in Equation 14 [34,35]
| Impeller Type | S | at Turbulent Flow | ||
| Rushton | 0.25 | 0.25 | 12.00 | 6.00 |
| 0.33 | 0.17 | 5.80 | 6.00 | |
| 0.33 | 0.25 | 6.70 | 6.00 | |
| 0.33 | 0.50 | 8.00 | 6.00 | |
| 0.50 | 0.25 | 4.25 | 6.00 | |
| 0.50 | 0.17 | 3.90 | 6.00 | |
| Propeller | 0.33 | 0.25 | 6.60 | 0.90 |
| Pitched-blade turbine, Downward pumping, 4 blades, 45 degrees | 0.33 | 0.20 | 5.70 | 1.27 |
| 0.33 | 0.25 | 6.20 | 1.27 | |
| 0.33 | 0.33 | 6.80 | 1.27 | |
| 0.33 | 0.50 | 11.50 | 1.27 | |
| 0.50 | 0.25 | 5.80 | 1.27 | |
| Pitched-blade turbine, Downward pumping, 6 blades, 45 degrees | 0.50 | 0.25 | 5.70 | 1.64 |
| Pitched-blade turbine, Upward pumping, 6 blades, 45 degrees | 0.50 | 0.25 | 6.90 | 1.64 |
Appendix D–Power Requirements Based on Agitation Speed for Transient Regions
References
- Blatchley, M.R.; Anseth, K.S. Middle-out methods for spatiotemporal tissue engineering of organoids. Nat. Rev. Bioeng. 2023, 1, 329–345. [Google Scholar] [CrossRef]
- Almeida, A.M.; Mazeda, J.; Pinho, A.R.; Gomes, M.C.; Mano, J.F. The future of automated tissue engineering: Robotic-assisted strategies for complex 3D tissue bottom-up assembly. Adv. Mater. Technol. 2025, 10, 70000. [Google Scholar] [CrossRef]
- Cho, S.; Discher, D.E.; Leong, K.W.; Vunjak-Novakovic, G.; Wu, J.C. Challenges and opportunities for the next generation of cardiovascular tissue engineering. Nat. Methods 2022, 19, 1064–1071. [Google Scholar] [CrossRef] [PubMed]
- Kasoju, N.; Sunilkumar, A. Convergence of tissue engineering and sustainable development goals. Biotechnol. Sustain. Mater. 2024, 1, 20. [Google Scholar] [CrossRef]
- Gomes, V.; Salgueiro, S.P. From small to large-scale: A review of recombinant spider silk and collagen bioproduction. Discov. Mater. 2022, 2, 3. [Google Scholar] [CrossRef]
- Chen, L.; Zhang, Y.; Chen, Z.; Dong, Y.; Jiang, Y.; Hua, J.; Liu, Y.; Osman, A.I.; Farghali, M.; Huang, L.; Rooney, D.W. Biomaterials technology and policies in the building sector: A review. Environ. Chem. Lett. 2024, 22, 715–750. [Google Scholar] [CrossRef]
- Major, G.S.; Doan, V.K.; Longoni, A.; Bilek, M.M.; Wise, S.G.; Rnjak-Kovacina, J.; Yeo, G.C.; Lim, K.S. Mapping the microcarrier design pathway to modernise clinical mesenchymal stromal cell expansion. Trends Biotechnol. 2024, 42, 859–876. [Google Scholar] [CrossRef]
- Li, F.; Vijayasankaran, N.; Shen, A.; Kiss, R.; Amanullah, A. Cell culture processes for monoclonal antibody production. mAbs 2010, 2, 466–479. [Google Scholar] [CrossRef]
- Wu, C.Y.; Stoecklein, D.; Kommajosula, A.; Lin, J.; Owsley, K.; Ganapathysubramanian, B.; Di Carlo, D. Shaped 3D microcarriers for adherent cell culture and analysis. Microsyst. Nanoeng. 2018, 4, 21. [Google Scholar] [CrossRef]
- Dong, B.; Wang, R.; Guan, Y.; Zhao, X.; Li, R.; Xu, Q.; Tang, N. Engineering AQP1-deficient DF-1 suspension cells for high-yield IBDV production and vaccine scale-up. Vaccines 2025, 14, 52. [Google Scholar] [CrossRef]
- Zhou, X.; Zheng, H.; Wu, Y.; Yin, H.; Mao, X.; Li, N.; Guo, H.; Chang, Y.; Jiang, X.; Ai, Q.; Xue, C. Scalable production of muscle and adipose cell-laden microtissues using edible macroporous microcarriers for 3D printing of cultured fish fillets. Nat. Commun. 2025, 16, 1740. [Google Scholar] [CrossRef]
- Maillot, C.; De Isla, N.; Loubiere, C.; Toye, D.; Olmos, E. Impact of microcarrier concentration on mesenchymal stem cell growth and death: Experiments and modeling. Biotechnol. Bioeng. 2022, 119, 3537–3548. [Google Scholar] [CrossRef]
- Daňková, V.; Exnerová, A.; Vágnerová, H.; Pavlík, V.; Nešporová, K. Generation of bioactive stem cell-derived secretome in 3D bioreactor system: Towards cell-free therapy in veterinary medicine. Biomolecules 2025, 16, 2. [Google Scholar] [CrossRef] [PubMed]
- Rouwkema, J.; Koopman, B.F.J.M.; Van Blitterswijk, C.A.; Dhert, W.J.A.; Malda, J. Supply of nutrients to cells in engineered tissues. Biotechnol. Genet. Eng. Rev. 2009, 26, 163–178. [Google Scholar] [CrossRef]
- Xue, Y.; Georgakopoulou, T.; Van der Wijk, A.E.; Józsa, T.I.; Van Bavel, E.; Payne, S.J. Quantification of hypoxic regions distant from occlusions in cerebral penetrating arteriole trees. PLoS Comput. Biol. 2022, 18, e1010166. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Guertin, P.; Jia, G.; Lv, Z.; Yang, H.; Ju, D. Large-scale microcarrier culture of HEK293T cells and Vero cells in single-use bioreactors. AMB Express 2019, 9. [Google Scholar] [CrossRef]
- Zhang, B.; Lu, Q.; Dai, G.; Zhou, Y.; Ye, Q.; Zhou, Y.; Tan, W.S. Enhancing mesenchymal stem cells cultivated on microcarriers in spinner flasks via impeller design optimization for aggregated suspensions. Bioresour. Bioprocess. 2023, 10. [Google Scholar] [CrossRef] [PubMed]
- Koh, B.; Sulaiman, N.; Fauzi, M.B.; Law, J.X.; Ng, M.H.; Idrus, R.B.H.; Yazid, M.D. Three dimensional microcarrier system in mesenchymal stem cell culture: A systematic review. Cell Biosci. 2020, 10, 75. [Google Scholar] [CrossRef]
- McMurtrey, R.J. Analytic models of oxygen and nutrient diffusion, metabolism dynamics, and architecture optimization in three-dimensional tissue constructs with applications and insights in cerebral organoids. Tissue Eng. Part C Methods 2016, 22, 221–249. [Google Scholar] [CrossRef]
- Brown, D.A.; MacLellan, W.R.; Laks, H.; Dunn, J.C.Y.; Wu, B.M.; Beygui, R.E. Analysis of oxygen transport in a diffusion-limited model of engineered heart tissue. Biotechnol. Bioeng. 2007, 97, 962–975. [Google Scholar] [CrossRef]
- Napitupulu, G.; Nagi, A.; Putri, M.R.; Radjawane, I.M. The one-dimensional numerical model: An application to oxygen diffusion in mitochondria cell. ComTech Comput. Math. Eng. Appl. 2023, 14, 101–118. [Google Scholar] [CrossRef]
- Krogh, A. The number and distribution of capillaries in muscles with calculations of the oxygen pressure head necessary for supplying the tissue. J. Physiol. 1919, 52, 409. [Google Scholar] [CrossRef]
- Ferrari, C.; Balandras, F.; Guedon, E.; Olmos, E.; Chevalot, I.; Marc, A. Limiting cell aggregation during mesenchymal stem cell expansion on microcarriers. Biotechnol. Prog. 2012, 28, 780–787. [Google Scholar] [CrossRef]
- Botte, E.; Mancini, P.; Magliaro, C.; Ahluwalia, A. A sense of proximity: Cell packing modulates oxygen consumption. APL Bioeng. 2023, 7. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, I.; Okada, S. Stationary phase of cultured mammalian cells (L5178Y). J. Cell Biol. 1967, 35, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Alva, R.; Mirza, M.; Baiton, A.; Lazuran, L.; Samokysh, L.; Bobinski, A.; Stuart, J.A. Oxygen toxicity: Cellular mechanisms in normobaric hyperoxia. Cell Biol. Toxicol. 2023, 39, 111–143. [Google Scholar] [CrossRef] [PubMed]
- Balestra, C.; Mrakic-Sposta, S.; Virgili, F. Oxygen variations—insights into hypoxia, hyperoxia and hyperbaric hyperoxia—is the dose the clue? Int. J. Mol. Sci. 2023, 24, 13472. [Google Scholar] [CrossRef]
- Dunlap, R.A. The symmetry and packing fraction of the body centered tetragonal structure. Eur. J. Phys. Educ. 2012, 3, 19–24. [Google Scholar]
- Mukherji, D.; Strunz, P.; Piegert, S.; Gilles, R.; Hofmann, M.; Hölzel, M.; Rösler, J. The hexagonal close-packed (hcp)⇆ face-centered cubic (fcc) transition in Co-Re-based experimental alloys investigated by neutron scattering. Metall. Mater. Trans. A 2012, 43, 1834–1844. [Google Scholar] [CrossRef]
- Merten, O.W. Advances in cell culture: Anchorage dependence. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370. [Google Scholar] [CrossRef]
- Liste-Calleja, L.; Lecina, M.; Cairó, J.J. HEK293 cell culture media study: Increasing cell density for different bioprocess applications. In Proceedings of the BMC Proceedings Conference, London, UK, December 2013. [Google Scholar]
- Aida, M.; Murakami, Y.; Shono, A. Estimation of the just suspension speed of mixed particles in a solid-liquid stirred tank. J. Chem. Eng. Jpn. 2023, 56, 2197949. [Google Scholar] [CrossRef]
- Zwietering, T.N. Suspending of solid particles in liquid by agitators. Chem. Eng. Sci. 1958, 8, 244–253. [Google Scholar] [CrossRef]
- Doran, P.M. Bioprocess Engineering Principles, 2nd ed.; Elsevier: Waltham, MA, USA, 2013. [Google Scholar]
- Hall, S. Blending and agitation. In Branan’s Rules of Thumb for Chemical Engineers; Elsevier: Amsterdam, Netherlands, 2012; pp. 257–279. [Google Scholar]
- Van Hemert, P.; Kilburn, D.G.; Van Wezel, A.L. Homogeneous cultivation of animal cells for the production of virus and virus products. Biotechnol. Bioeng. 1969, 11, 875–885. [Google Scholar] [CrossRef]
- Nienow, A.W. Hydrodynamics of stirred bioreactors. Appl. Mech. Rev. 1998, 51, 3–32. [Google Scholar] [CrossRef]
- Croughan, M.S.; Hamel, J.F.; Wang, D.I.C. Hydrodynamic effects on animal cells grown in microcarrier cultures. Biotechnol. Bioeng. 1987, 29, 130–141. [Google Scholar] [CrossRef] [PubMed]
- Croughan, M.S.; Sayre, E.S.; Wang, D.I.C. Viscous reduction of turbulent damage in animal cell culture. Biotechnol. Bioeng. 1989, 33, 862–872. [Google Scholar] [CrossRef]
- Preissmann, A.; Wiesmann, R.; Buchholz, R.; Werner, R.G.; Noé, W. Investigations on oxygen limitations of adherent cells growing on macroporous microcarriers. Cytotechnology 1997, 24, 121–134. [Google Scholar] [CrossRef]
- Rafiq, Q.A.; Coopman, K.; Nienow, A.W.; Hewitt, C.J. Systematic microcarrier screening and agitated culture conditions improves human mesenchymal stem cell yield in bioreactors. Biotechnol. J. 2016, 11, 473–486. [Google Scholar] [CrossRef]
- Huang, L.; Abdalla, A.M.; Xiao, L.; Yang, G. Biopolymer-based microcarriers for three-dimensional cell culture and engineered tissue formation. Int. J. Mol. Sci. 2020, 21, 1895. [Google Scholar] [CrossRef]
- Reddy, M.S.; Ponnamma, D.; Choudhary, R.; Sadasivuni, K.K. A comparative review of natural and synthetic biopolymer composite scaffolds. Polymers 2021, 13, 1105. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Xiang, Y.; Turner, F.; Bao, X. Integrated experimental and mathematical exploration of modular tissue cultures for developmental engineering. Int. J. Mol. Sci. 2024, 25, 2987. [Google Scholar] [CrossRef] [PubMed]
- Ng, Y.C.; Berry, J.M.; Butler, M. Optimization of physical parameters for cell attachment and growth on macroporous microcarriers. Biotechnol. Bioeng. 1996, 50, 627–635. [Google Scholar] [CrossRef]
- Grein, T.A.; Loewe, D.; Dieken, H.; Weidner, T.; Salzig, D.; Czermak, P. Aeration and shear stress are critical process parameters for the production of oncolytic measles virus. Front. Bioeng. Biotechnol. 2019, 7, 78. [Google Scholar] [CrossRef]
- Ebrahimian, A.; Schalk, M.; Dürkop, M.; Maurer, M.; Bliem, R.; Kühnel, H. Seed train optimization in microcarrier-based cell culture post in situ cell detachment through scale-down hybrid modeling. Bioengineering 2024, 11, 268. [Google Scholar] [CrossRef]
- Cherry, R.S.; Papoutsakis, E.T. Shear and mixing effects on cells in agitated microcarrier tissue culture reactors. In Proceedings of the Space Bioreactor Science Workshop, NASA Lyndon B. Johnson Space Center, Houston, TX, USA, 1 December 1987. [Google Scholar]
- Jenish, I.; Appadurai, M.; Raj, E.F. CFD analysis of modified Rushton turbine impeller. Int. J. Sci. Manag. Stud. 2021, 4, 8–13. [Google Scholar] [CrossRef]
- Cherry, R.S.; Papoutsakis, E.T. Hydrodynamic effects on cells in agitated tissue culture reactors. Bioprocess Eng. 1986, 1, 29–41. [Google Scholar] [CrossRef]
- Ibrahim, S.; Nienow, A.W. Suspension of microcarriers for cell culture with axial flow impellers. Chem. Eng. Res. Des. 2004, 82, 1082–1088. [Google Scholar] [CrossRef]
- Bernauer, S.; Eibl, P.; Witz, C.; Khinast, J.; Hardiman, T. Analyzing the effect of using axial impellers in large-scale bioreactors. Biotechnol. Bioeng. 2022, 119, 2494–2504. [Google Scholar] [CrossRef]
- Ramírez, L.A.; Pérez, E.L.; García Díaz, C.; Camacho Luengas, D.A.; Ratkovich, N.; Reyes, L.H. CFD and experimental characterization of a bioreactor: Analysis via power curve, flow patterns and kLa. Processes 2020, 8, 878. [Google Scholar] [CrossRef]
- Kaiser, S.C.; Werner, S.; Jossen, V.; Kraume, M.; Eibl, D. Development of a method for reliable power input measurements in conventional and single-use stirred bioreactors at laboratory scale. Eng. Life Sci. 2017, 17, 500–511. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Wu, W.; Fang, J.; Yin, J. Polymer-based porous microcarriers as cell delivery systems for applications in bone and cartilage tissue engineering. Int. Mater. Rev. 2021, 66, 77–113. [Google Scholar] [CrossRef]
- Verdú-Navarro, F.; Moreno-Cid, J.A.; Weiss, J.; Egea-Cortines, M. Cascade oxygen control enhances growth of Nicotiana benthamiana cell cultures in stirred-tank bioreactors. Plants 2025, 14, 2879. [Google Scholar] [CrossRef]
- Birmingham, J. Impact of Surface Aeration on Scale-Up with Aerobic Bioreactors. Ph.D. Thesis, University of Delaware, Newark, DE, USA, 2014. [Google Scholar]
- Flagiello, D.; Parisi, A.; Lancia, A.; Di Natale, F. A review on gas–liquid mass transfer coefficients in packed-bed columns. ChemEngineering 2021, 5, 43. [Google Scholar] [CrossRef]
- Uyar, B.; Ali, M.D.; Uyar, G.E.O. Design parameters comparison of bubble column, airlift and stirred tank photobioreactors for microalgae production. Bioprocess Biosyst. Eng. 2024, 47, 195–209. [Google Scholar] [CrossRef] [PubMed]
- Walls, P.L.; McRae, O.; Natarajan, V.; Johnson, C.; Antoniou, C.; Bird, J.C. Quantifying the potential for bursting bubbles to damage suspended cells. Sci. Rep. 2017, 7, 15102. [Google Scholar] [CrossRef] [PubMed]
- Mesa, D.; Brito-Parada, P.R. Bubble size distribution in aerated stirred tanks: Quantifying the effect of impeller-stator design. Chem. Eng. Res. Des. 2020, 160, 356–369. [Google Scholar] [CrossRef]
- Chen, X.Y.; Chen, J.Y.; Tong, X.M.; Mei, J.G.; Chen, Y.F.; Mou, X.Z. Recent advances in the use of microcarriers for cell cultures and their ex vivo and in vivo applications. Biotechnol. Lett. 2020, 42, 1–10. [Google Scholar] [CrossRef]
- De Silva Thompson, D.; Peticone, C.; Burova, I.; Shipley, R.J.; Knowles, J.C.; Kim, H.W.; Micheletti, M.; Wall, I.B. Assessing behaviour of osteoblastic cells in dynamic culture conditions using titanium-doped phosphate glass microcarriers. J. Tissue Eng. 2019, 10, 2041731419825772. [Google Scholar] [CrossRef]
- Mel, M.; Karim, M.I.A.; Yusuf, S.A.M.; Hashim, Y.Z.H.Y.; Ahmad Nor, Y. Comparing BRIN-BD11 culture producing insulin using different type of microcarriers. Cytotechnology 2010, 62, 423–430. [Google Scholar] [CrossRef] [PubMed]
- Poon, C. Measuring the density and viscosity of culture media for optimized computational fluid dynamics analysis of in vitro devices. J. Mech. Behav. Biomed. Mater. 2022, 126, 105024. [Google Scholar] [CrossRef]
- Serra, A.T.; Serra, M.; Silva, A.C.; Brckalo, T.; Seshire, A.; Brito, C.; Wolf, M.; Alves, P.M. Scalable culture strategies for the expansion of patient-derived cancer stem cell lines. Stem Cells Int. 2019, 2019, 8347595. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




