Submitted:
21 March 2026
Posted:
24 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Case Selection
2.2. Data Collection
2.2.1. Preoperative Imaging
2.2.2. MRI Imaging
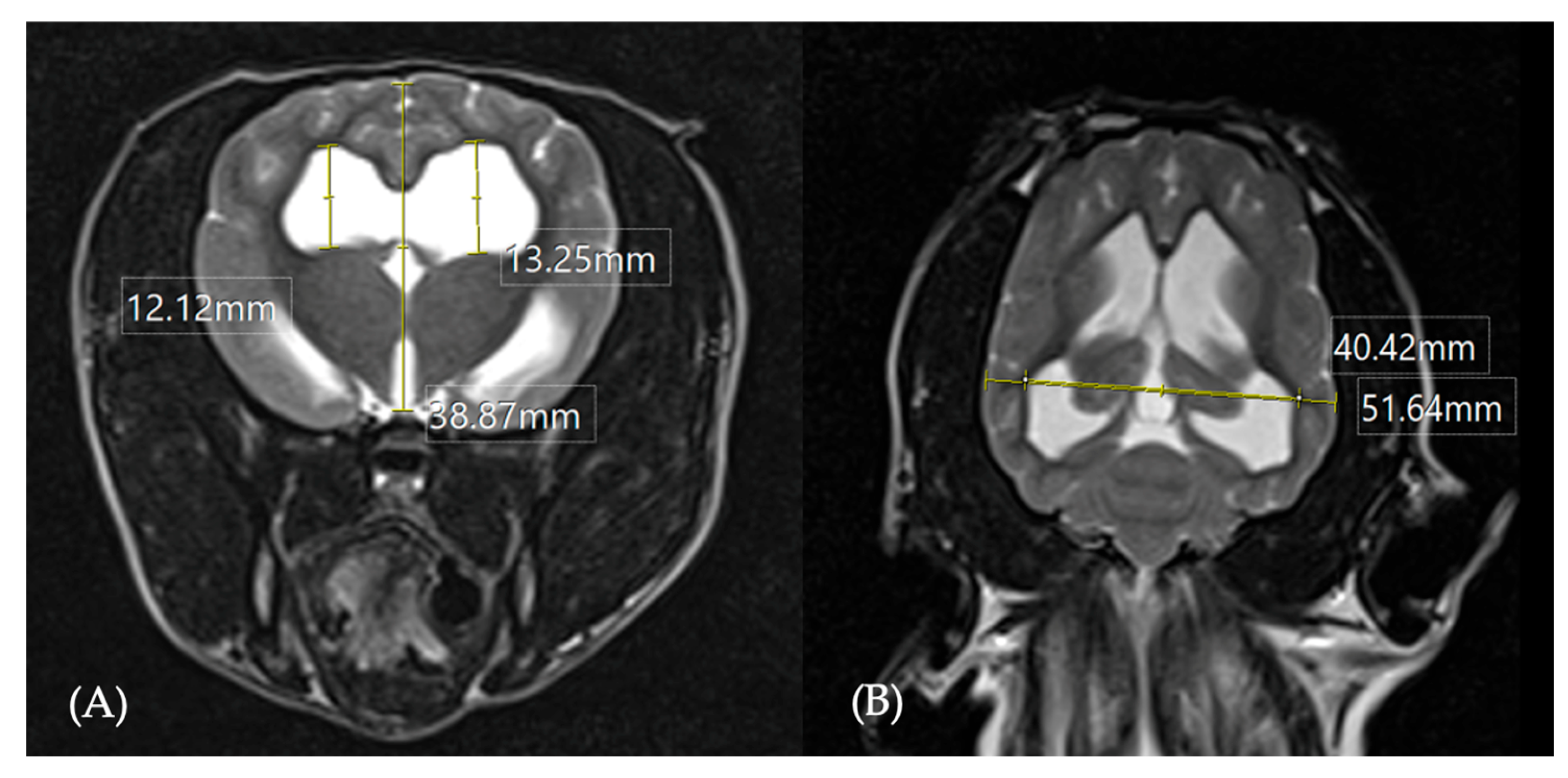
2.2.2.1. The Lateral Ventricular Enlargement
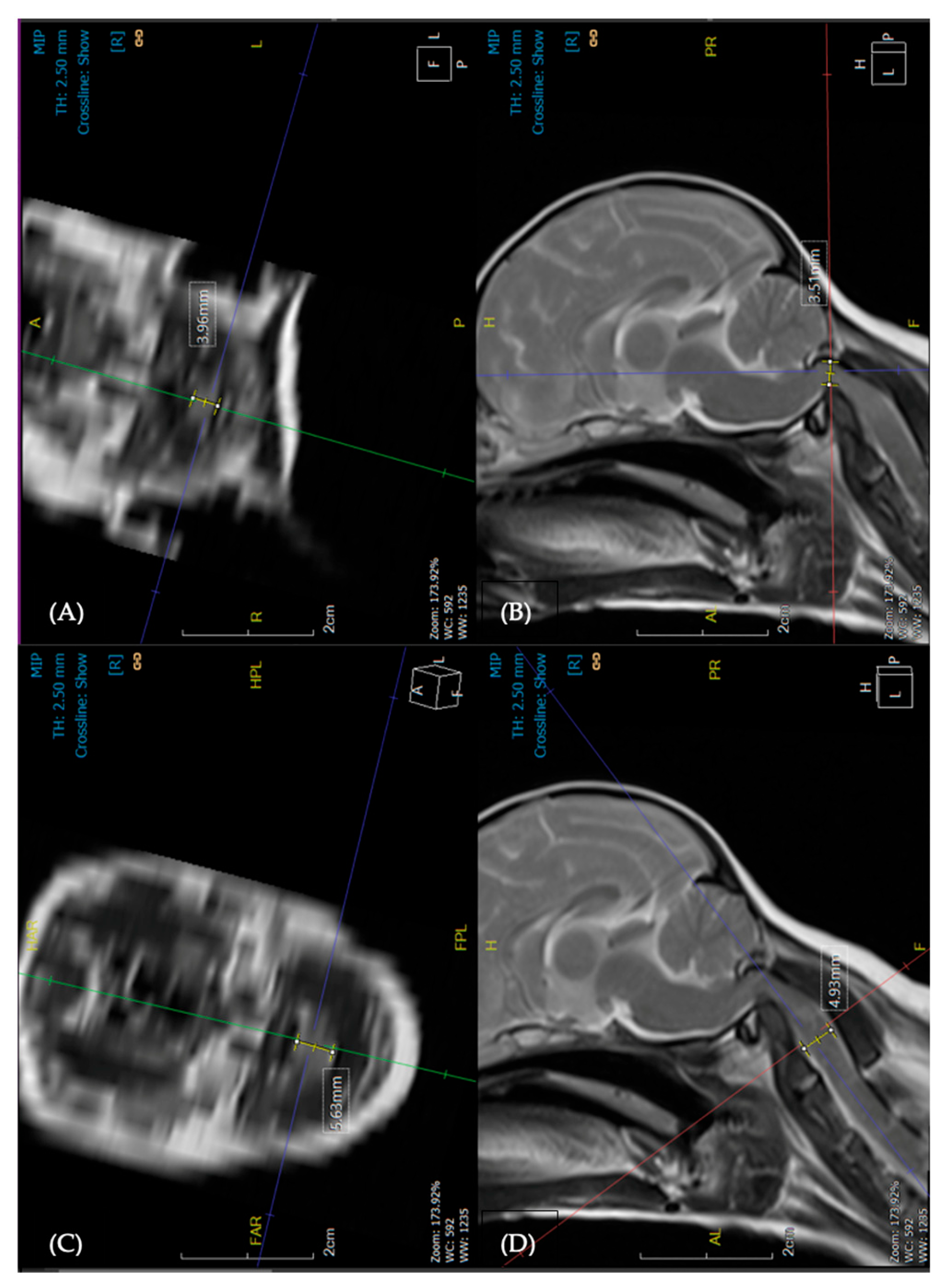
2.2.2.2. Dorsal Compression of the Atlantoaxial Band/Dural Fibrous Band
2.2.2.3. Spinal Cord Compression
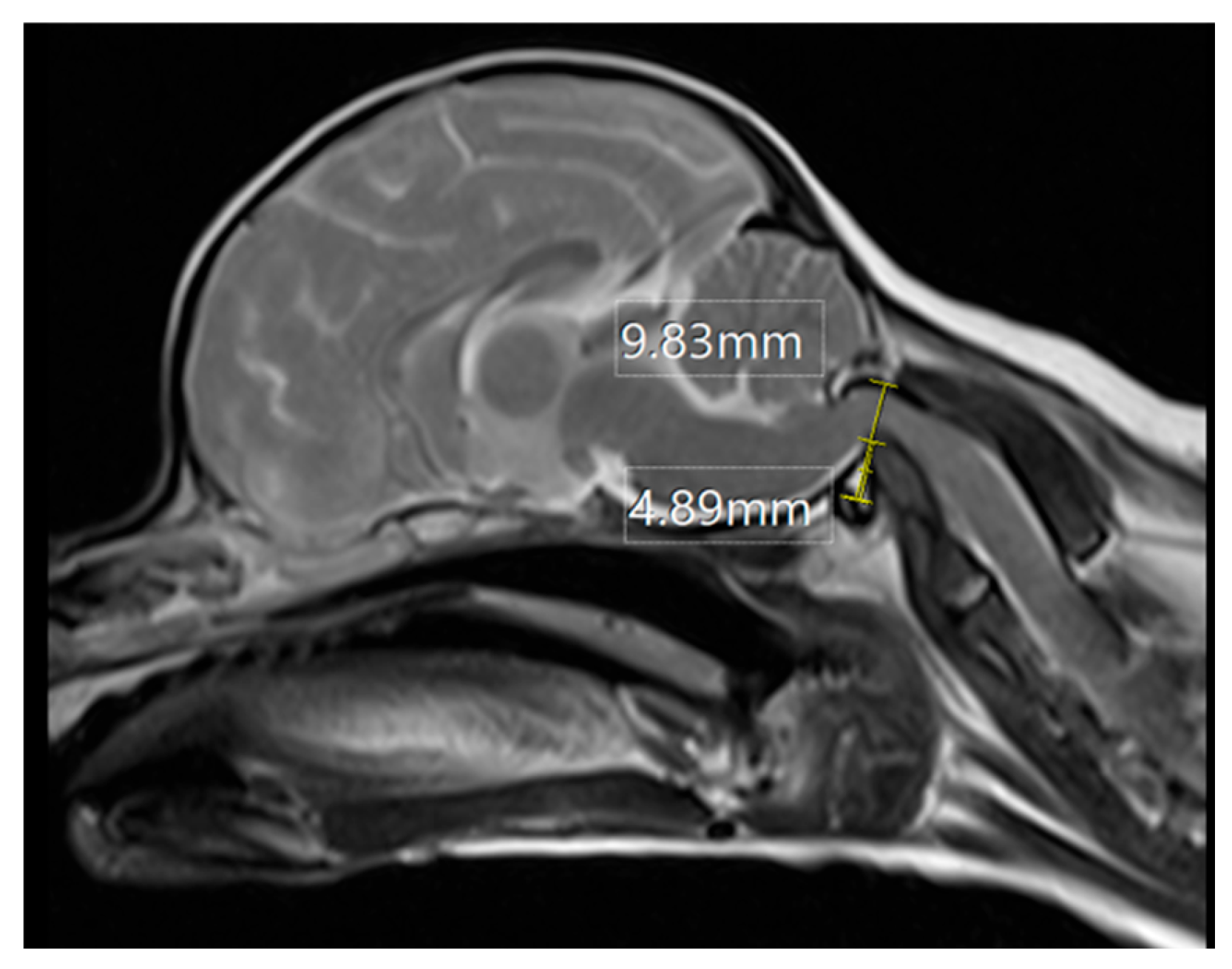
2.2.2.4. Ventral Compression Index
2.2.2.5. Syringomyelia
2.2.2.6. Chiari-like Malformation
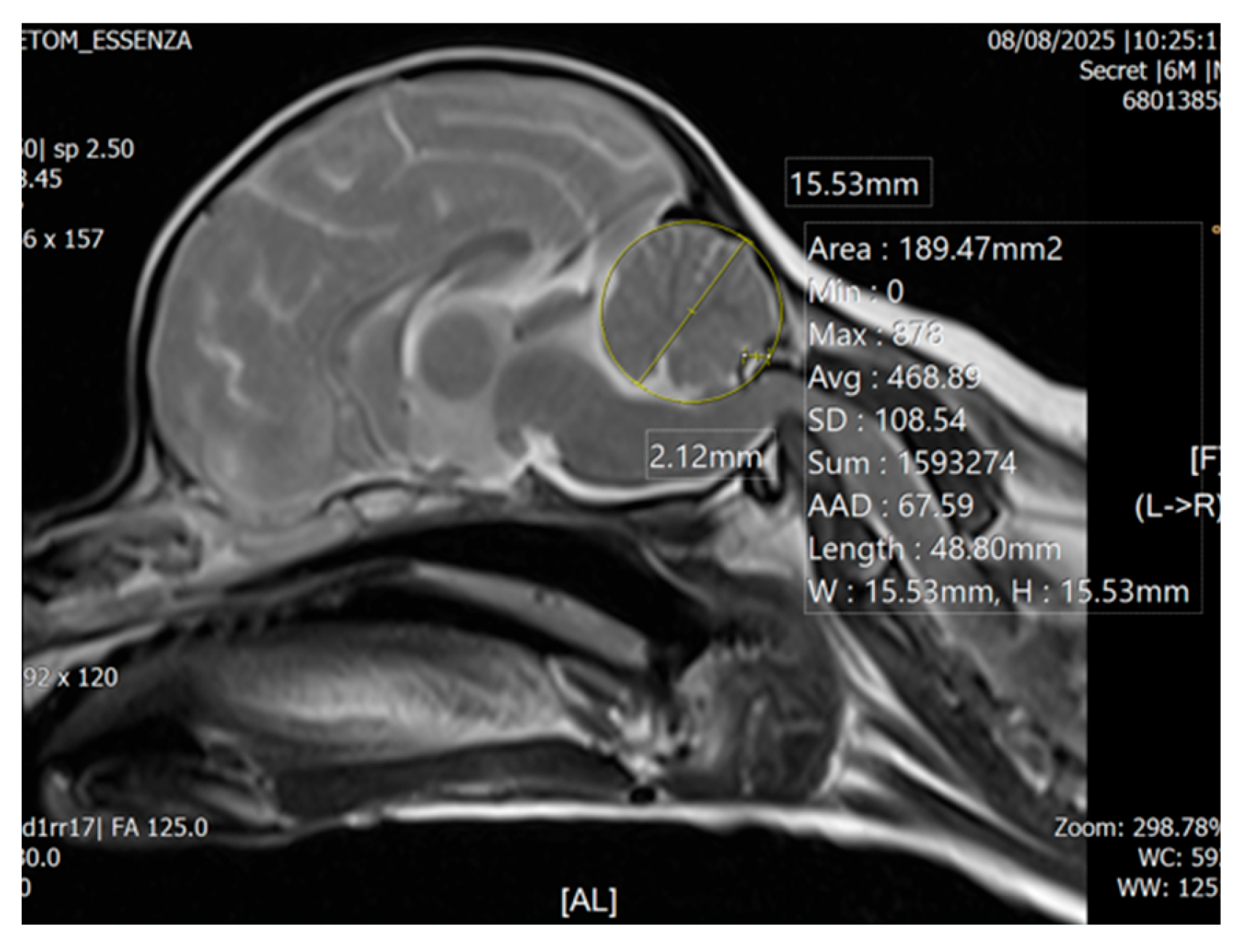
2.2.2.7. Cerebellar Compression
2.2.3. Anesthesia and Surgical Technique
2.2.4. Post-Operative Management and Follow-Up
2.3. Statistical Analysis
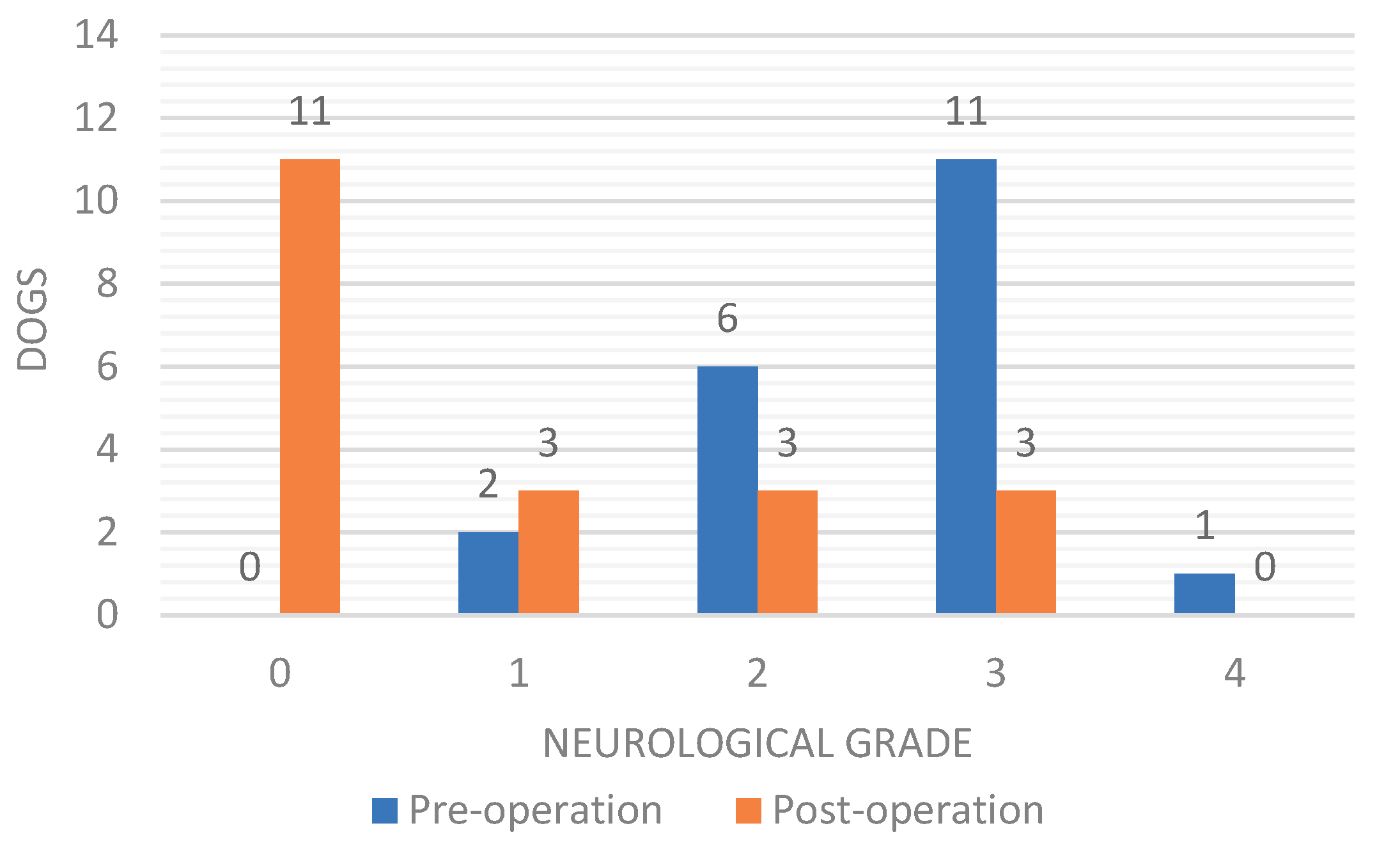
3. Results
3.1. Animals
3.2. MRI Findings
3.2.1. Mechanical Spinal Cord Compression Parameters
3.2.2. Ventricular Parameters
3.2.3. Craniocervical Structural Abnormalities
3.2.4. Intramedullary Pathology
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAI | Atlantoaxial instability |
| CCD | Central canal dilation |
| CC index | Cerebellar compression index |
| CSF | Cerebrospinal fluid |
| DADI | Dorsal atlantodental interval |
| MRI | Magnetic resonance imaging |
| SM | Syringomyelia |
| VADI | Ventral atlantodental interval |
| VCI | Ventral compression index |
| V/B ratio | Ventricle/brain ratio |
References
- Havig, M.E.; Cornell, K.K.; Hawthorne, J.C.; et al. Evaluation of Nonsurgical Treatment of Atlantoaxial Subluxation in Dogs- 19 Cases (1992–2001). JAVMA 2005, 227. [Google Scholar] [CrossRef] [PubMed]
- Itoh, H.; Itamoto, T.; Tani, K.; Sunahara, H.; Nemoto, Y.; Nakaichi, M.; Iseri, T.; Horikirizono, H.; Itamoto, K. Relationship between Clinical Parameters and Malformations in Dogs Diagnosed with Atlanto-Axial Instability. PLoS One 2024, 19. [Google Scholar] [CrossRef]
- Cummings, K. R.; Vilaplana Grosso, F.; Moore, G. E.; Rochat, M.; Thomovsky, S. A.; Bentley, R. T. Objective Measurements of the Atlantoaxial Joint on Radiographs Performed without Flexion Can Increase the Confidence of Diagnosis of Atlantoaxial Instability in Toy Breed Dogs. Veterinary Radiology and Ultrasound 2018, 59(6), 667–676. [Google Scholar] [CrossRef] [PubMed]
- Beaver, D. P.; Ellison, G. W.; Lewis, D. D.; Goring, R. L.; Kubilis, P. S.; Barchard, C. Risk Factors Affecting the Outcome of Surgery for Atlantoaxial Subluxation in Dogs: 46 Cases (1978-1998). J. Am. Vet. Med. Assoc. 2000, 216(7), 1104–1109. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, F.; Kouno, S.; Yamaguchi, S.; Hara, Y. Evaluation of Atlantooccipital Overlapping and Cerebral Ventricle Size in Dogs with Atlantoaxial Instability. Journal of Veterinary Medical Science 2019, 81(2), 229–236. [Google Scholar] [CrossRef] [PubMed]
- Marino, D.J.; Loughin, C.A.; Dewey, C.W.; Marino, L.J.; Sackman, J.J.; Lesser, M.L.; Akerman, M.B. Morphometric Features of the Craniocervical Region in Dogs with Suspected Chiari-Like Malformation Determined by Combined Use of Magnetic Resonance Imaging and Computed Tomography. Am. J. Vet. Res. 2012, 73, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Slanina, M. C. Atlantoaxial Instability. In Veterinary Clinics of North America—Small Animal Practice; W.B. Saunders March 1, 2016; pp. pp 265–275. [Google Scholar] [CrossRef]
- Tobias, K.M.; Johnston, S.A. Veterinary Surgery: Small Animal, 2nd ed.; Elsevier: St. Louis, MO, USA, 2017. [Google Scholar]
- Olby, N. J..; Platt, S. R. BSAVA Manual of Canine and Feline Neurology; British Small Animal Veterinary Association, 2014. [Google Scholar]
- Geary, J. C.; Oliver, J. E.; Hoerleins, B. F. Atlanto Axial Subluxation in the Canine*; Pergamon Press Ltd., 1967; Vol. 8. [Google Scholar] [CrossRef]
- Bray, K. Y.; Platt, S. R.; Kent, M.; Olby, N. J.; Early, P. J.; Mariani, C. L.; Muñana, K. R.; Holmes, S. P. Magnetic Resonance Imaging Characteristics of Atlanto-Axial Subluxation in 42 Dogs: Analysis of Joint Cavity Size, Subluxation Distance, and Craniocervical Junction Anomalies. Open Vet. J. 2023, 13(9), 1091–1098. [Google Scholar] [CrossRef] [PubMed]
- Vilkovysky, I. F.; Rusnak, I. A.; Yagnikov, S. A.; Sakhno, N. V.; Seleznev, S. B. Analysis of Surgical Correction of Atlantoaxial Instability in Dogs. RUDN Journal of Agronomy and Animal Industries 2023, 18(2), 241–249. [Google Scholar] [CrossRef]
- Barillaro, G.; Tabbì, M.; Minniti, S.; Iannelli, N. M.; Macrì, F.; Interlandi, C. Treatment of Canine Atlantoaxial Subluxation with a Modified Cervical Distraction–Stabilization Technique and Clinical Outcomes. Animals (Basel). 2025, 15(5), 716. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.; Liebel, F. X.; Fadda, A.; Lazzerini, K.; Harcourt-Brown, T. Same-Day Surgery May Reduce the Risk of Losing Pain Perception in Dogs with Thoracolumbar Disc Extrusion. Journal of Small Animal Practice 2020, 61(7), 442–448. [Google Scholar] [CrossRef] [PubMed]
- Planchamp, B.; Forterre, F.; Vidondo, B.; Hernandez-Guerra, A. M.; Plessas, I. N.; Schmidt, M. J.; Waschk, M. A.; Precht, C. Determination of Cutoff Values on Computed Tomography and Magnetic Resonance Images for the Diagnosis of Atlantoaxial Instability in Small-Breed Dogs. Veterinary Surgery 2022, 51(4), 620–630. [Google Scholar] [CrossRef] [PubMed]
- Laubner, S.; Ondreka, N.; Failing, K.; Kramer, M.; Schmidt, M. J. Magnetic Resonance Imaging Signs of High Intraventricular Pressure—Comparison of Findings in Dogs with Clinically Relevant Internal Hydrocephalus and Asymptomatic Dogs with Ventriculomegaly. BMC Vet. Res. 2015, 11(1). [Google Scholar] [CrossRef] [PubMed]
- Pivetta, M.; De Risio, L.; Newton, R.; Dennis, R. Prevalence of Lateral Ventricle Asymmetry in Brain Mri Studies of Neurologically Normal Dogs and Dogs with Idiopathic Epilepsy. Veterinary Radiology and Ultrasound 2013, 54(5), 516–521. [Google Scholar] [CrossRef] [PubMed]
- Mai, W. Diagnostic MRI in Dogs and Cats; Mai, W., Ed.; CRC Press: Boca Raton; CRC Press, 2018. [Google Scholar] [CrossRef]
- CM/SM Screening Scheme; The Kennel Club: London, UK; Available online: https://www.thekennelclub.org.uk/health-and-dog-care/health/getting-started-with-health-testing-and-screening/cmsm-screening-scheme/ (accessed on 18 March 2026).
- Spitzbarth, I.; Moore, S. A.; Stein, V. M.; Levine, J. M.; Olby, N. J.; Gjessing, K. M.; Davidson, R. M.; Lewis, M. J.; Jeffery, N. D.; da Costa, R. C.; Nout-Lomas, Y. S.; Fenn, J.; Granger, N.; Tipold, A.; Lim, J. H.; Volk, H. Current Insights Into the Pathology of Canine Intervertebral Disc Extrusion-Induced Spinal Cord Injury. In Frontiers in Veterinary Science; Frontiers Media S.A, 27 October 2020. [Google Scholar] [CrossRef]
- Jones, R.; Cirovic, S.; Rusbridge, C. A Review of Cerebrospinal Fluid Circulation with Respect to Chiari-like Malformation and Syringomyelia in Brachycephalic Dogs. In Fluids and Barriers of the CNS; BioMed Central Ltd, 1 March 2025. [Google Scholar] [CrossRef]
- Bach, F. S.; Mai, W.; Weber, L. F. S.; Villanova Junior, J. A.; Bianchi de Oliveira, L.; Montiani-Ferreira, F. Association between Spinal Cord Compression Ratio in Magnetic Resonance Imaging, Initial Neurological Status, and Recovery after Ventral Slot in 57 Dogs with Cervical Disc Extrusion. Front. Vet. Sci. 2023, 9. [Google Scholar] [CrossRef] [PubMed]
- Penning, V.; Platt, S. R.; Dennis, R.; Cappello, R.; Adams, V. Association of Spinal Cord Compression Seen on Magnetic Resonance Imaging with Clinical Outcome in 67 Dogs with Thoracolumbar Intervertebral Disc Extrusion. J. Small Anim. Pract. 2006, 47(11), 644–650. [Google Scholar] [CrossRef]
- Takahashi, F.; Hakozaki, T.; Kouno, S.; Suzuki, S.; Sato, A.; Kanno, N.; Harada, Y.; Yamaguchi, S.; Hara, Y. Atlantooccipital Overlapping and Its Effect on Outcomes after Ventral Fixation in Dogs with Atlantoaxial Instability. Journal of Veterinary Medical Science. Japanese Society of Veterinary Science 2018, pp 526–531. [Google Scholar] [CrossRef] [PubMed]
- Šulla, I.; Balik, V.; Ledecký, V. SPINAL CORD INJURIES IN DOGS PART I: A REVIEW OF BASIC KNOWLEDGE. [CrossRef]
| Age (years) | 2.8 (6 mth – 10 yr) |
|---|---|
| Bodyweight (kg) | 2.11 ( 1.3 – 4.3 ) |
| Gender | |
| Female | 7 |
| Male | 13 |
| Breed | |
| Chihuahua | 11 (55%) |
| Pomeranian | 7 (35%) |
| Shih tzu | 1 (5%) |
| Maltese | 1 (5%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




