Submitted:
20 March 2026
Posted:
23 March 2026
You are already at the latest version
Abstract
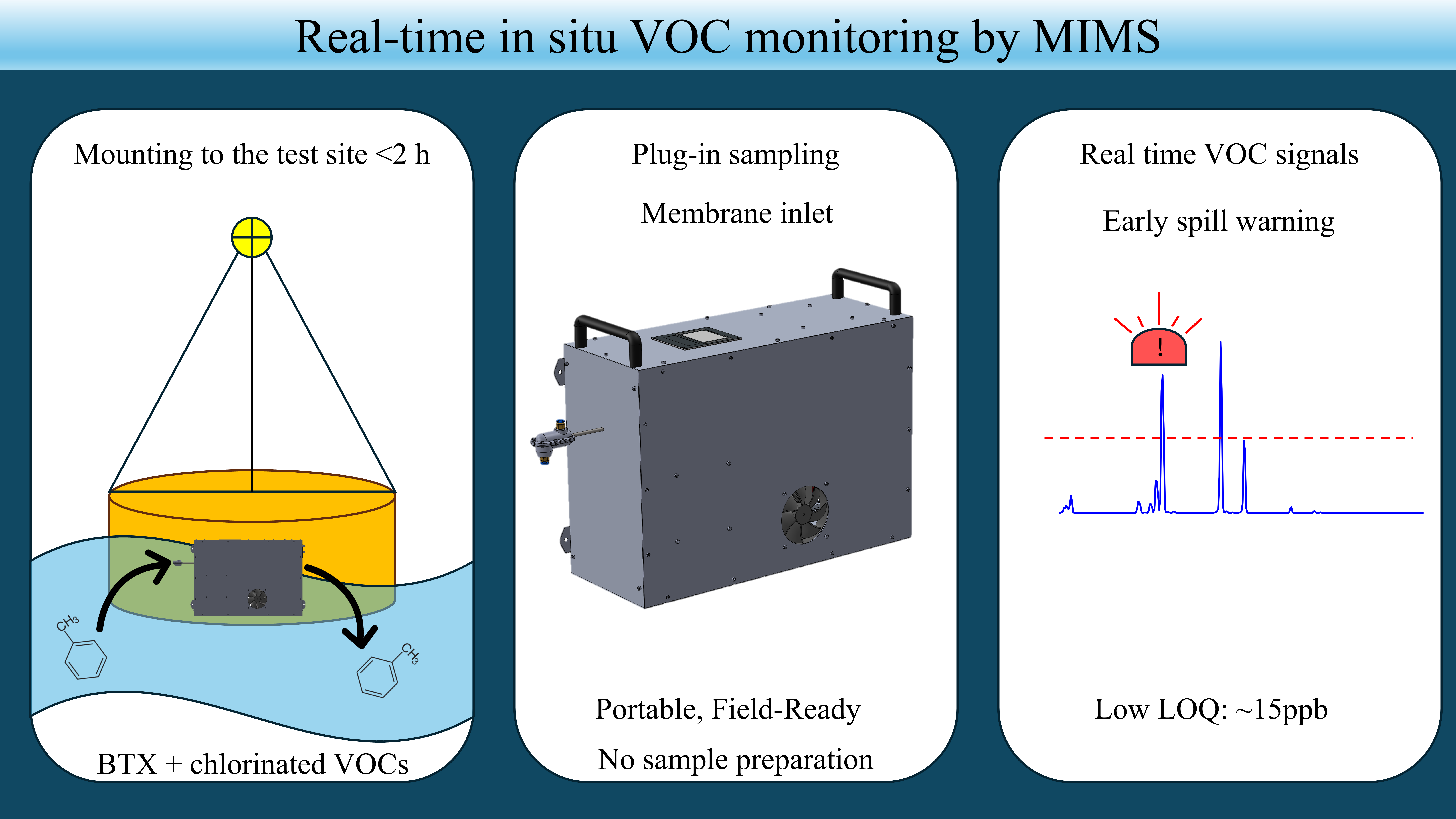
Keywords:
1. Introduction
2. Results
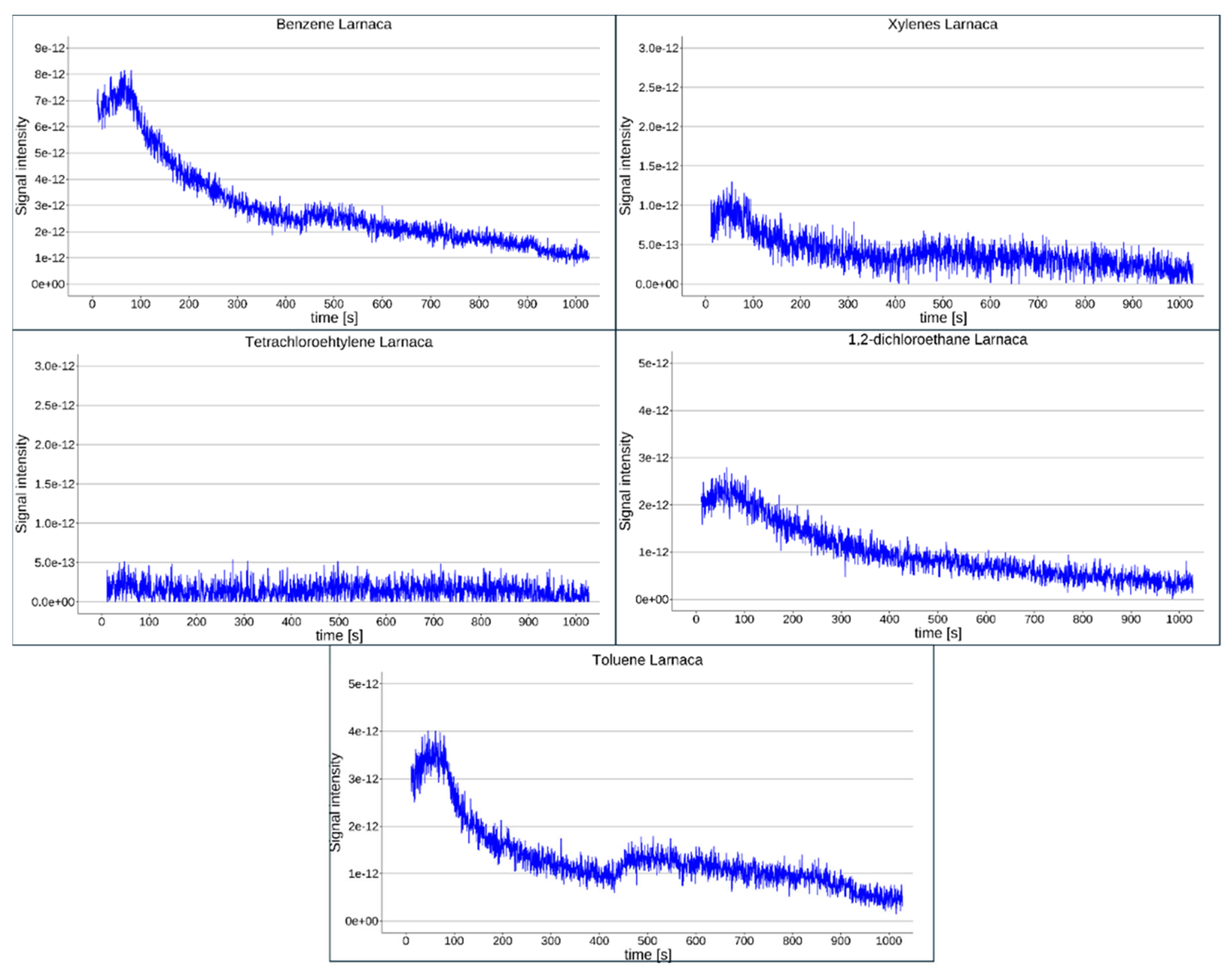
2.1. Results from the Pilot Locations
2.2. Field Evaluation of Detection Performance
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
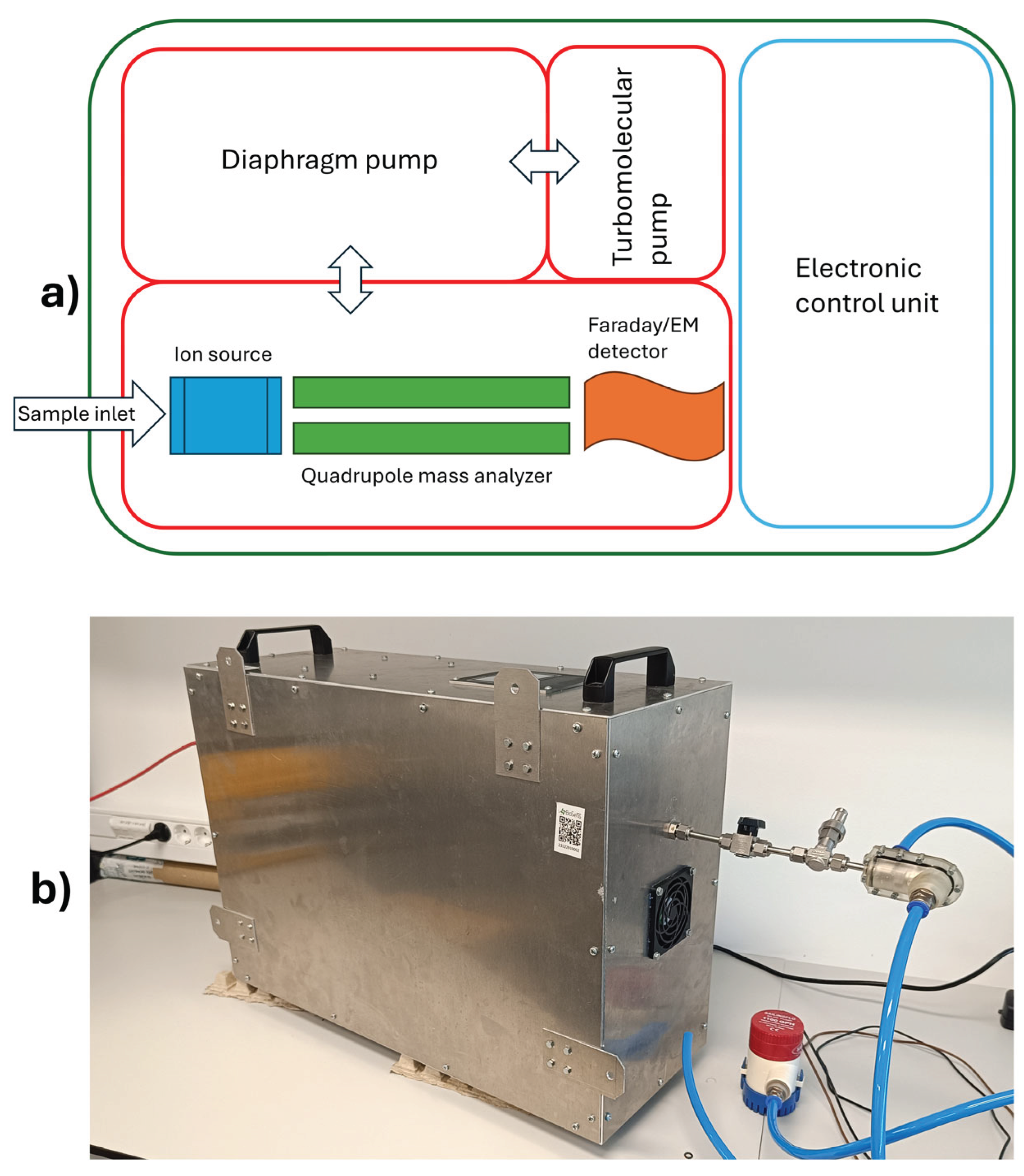
4.2. Instrumentation
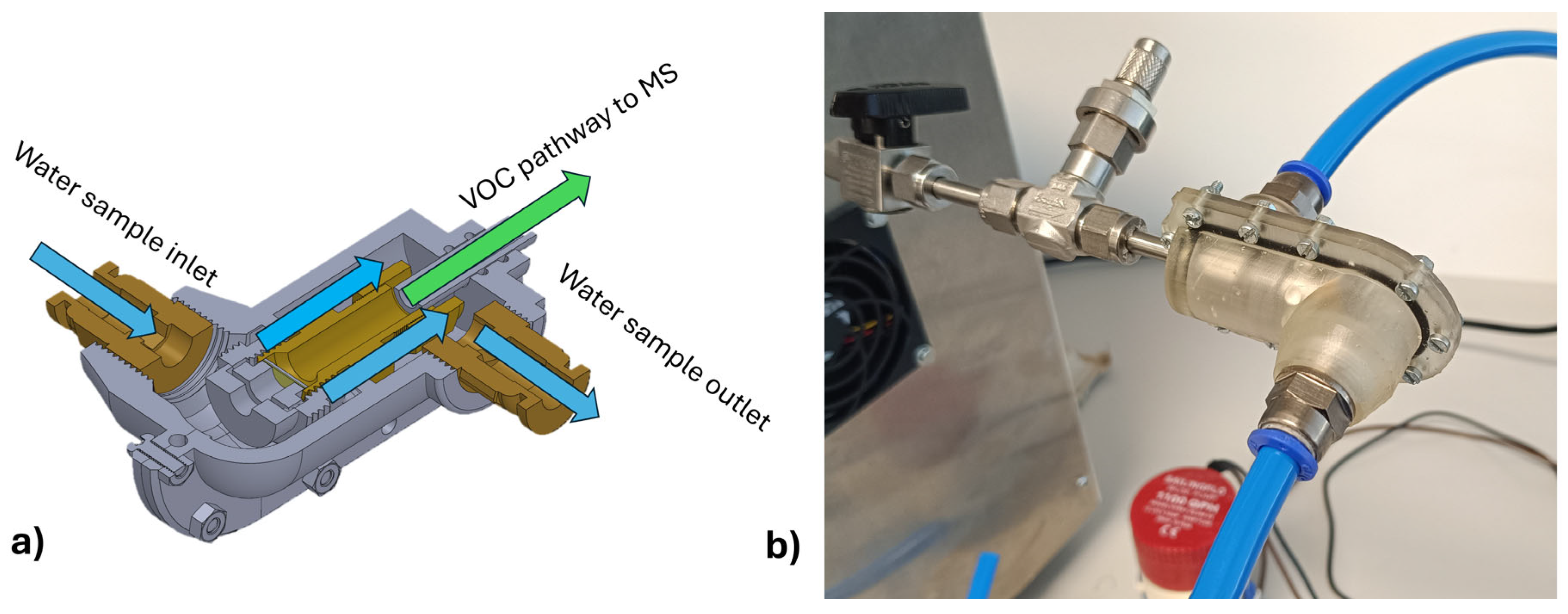
4.3. Membrane Inlet and Sampling Interface
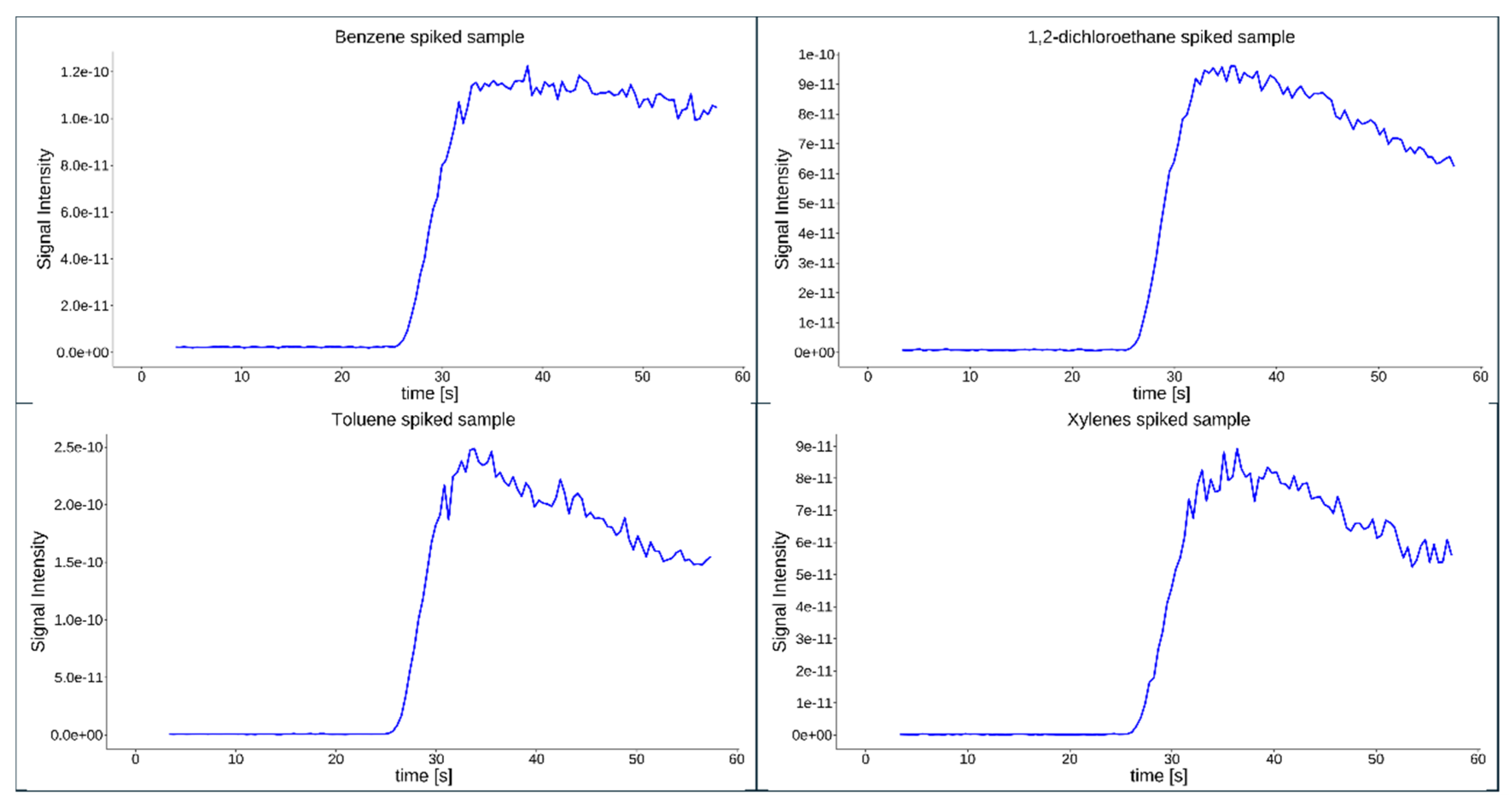
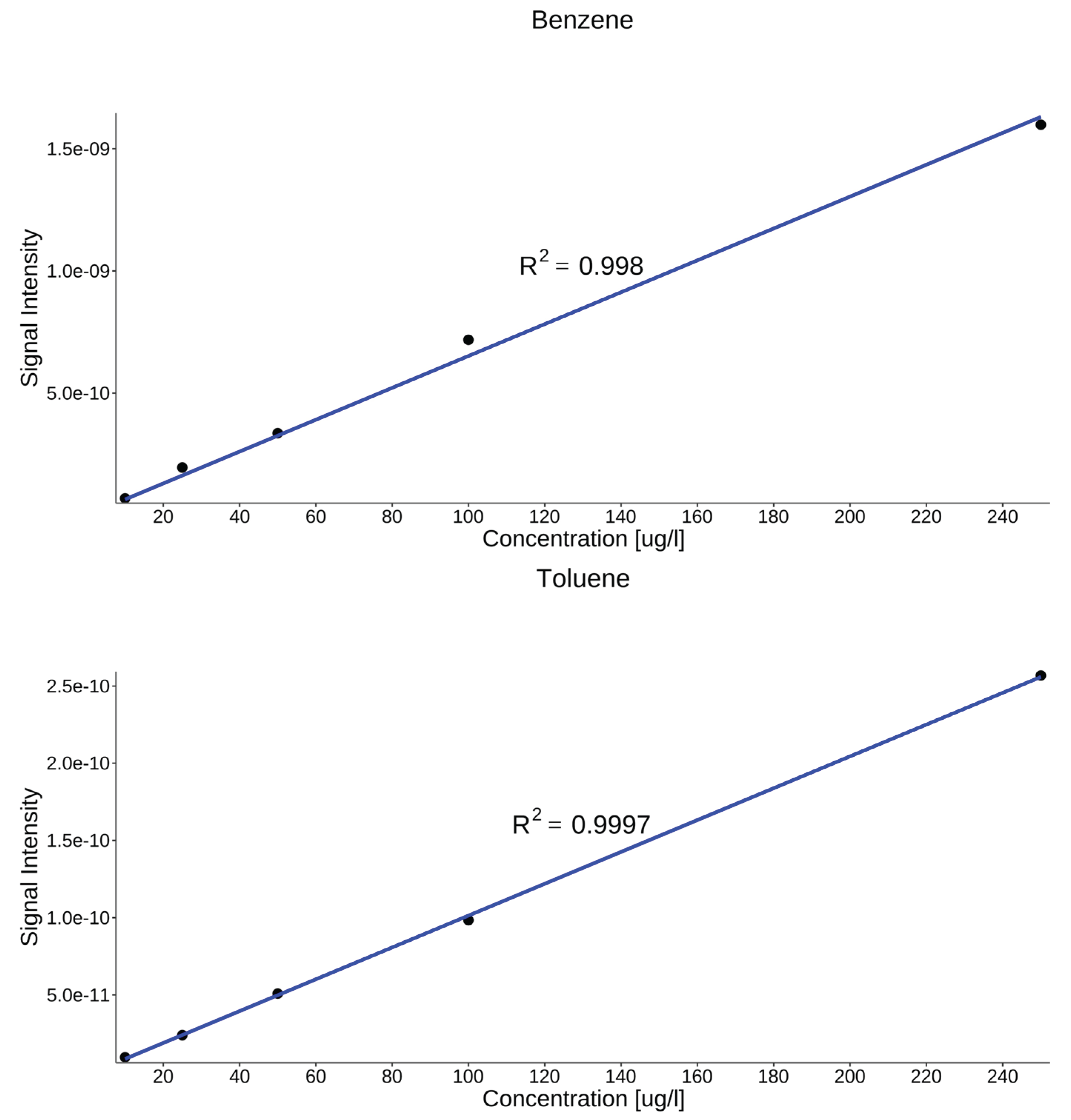
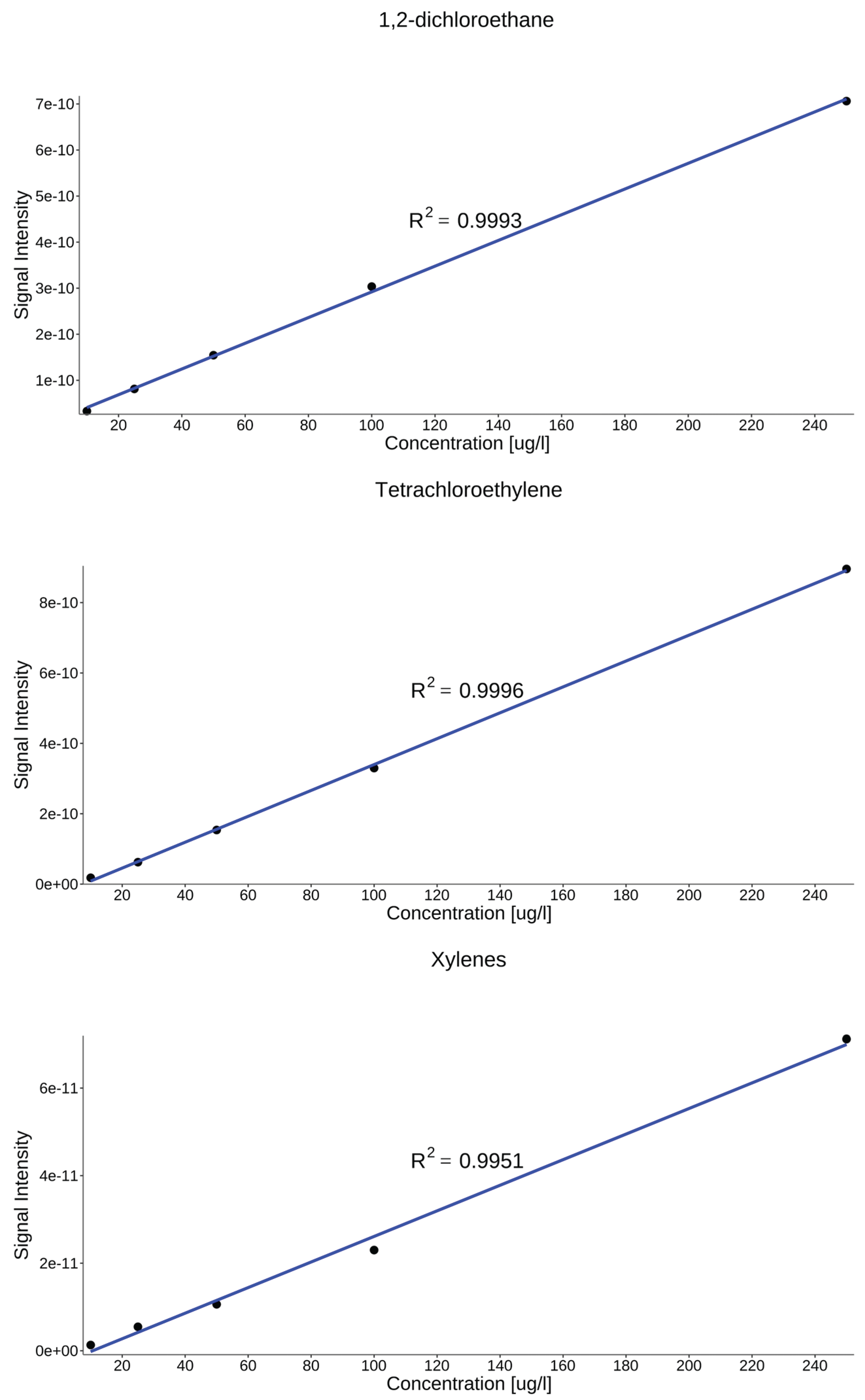
4.4. Calibration and Analytical Performance Evaluation
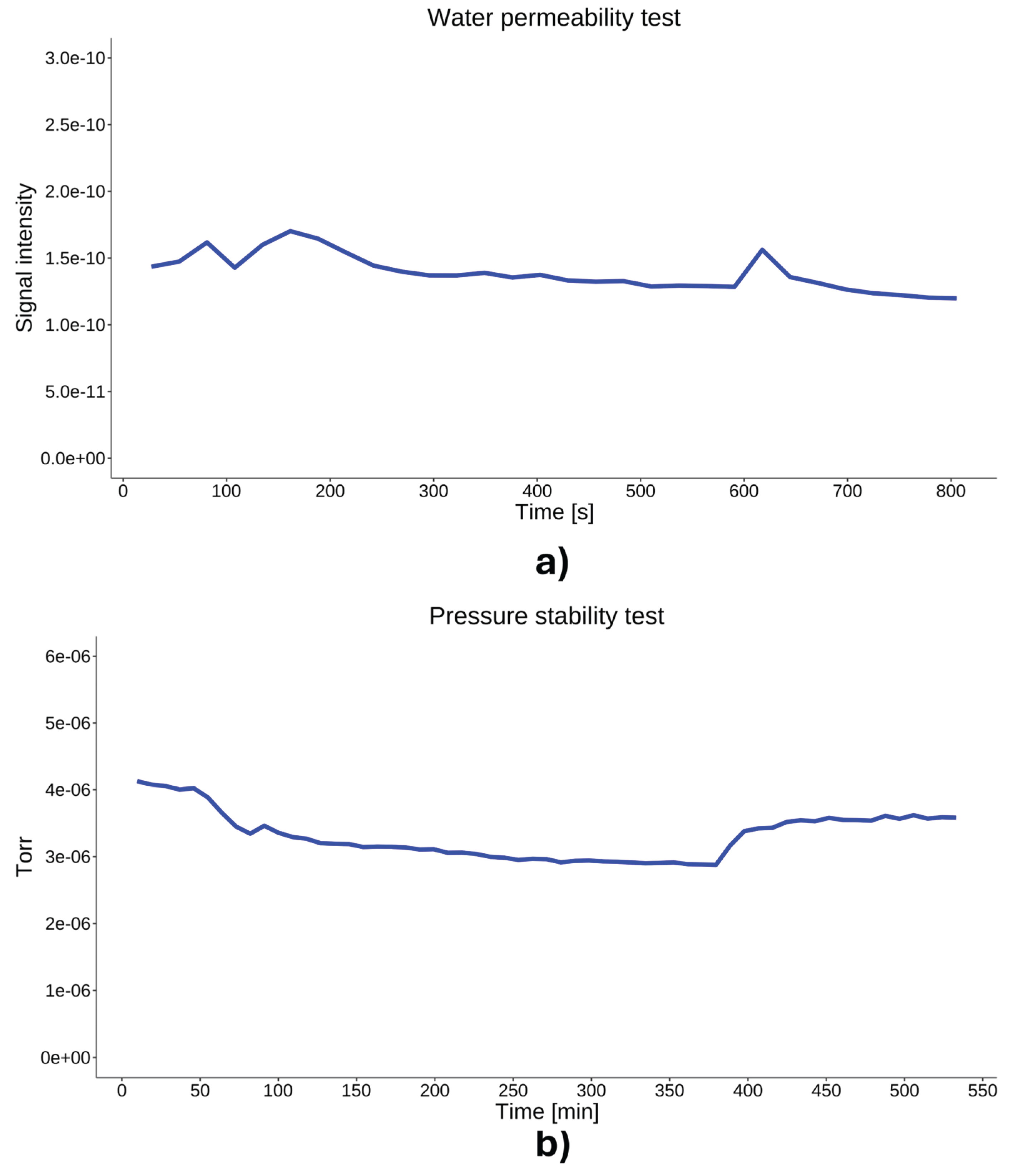
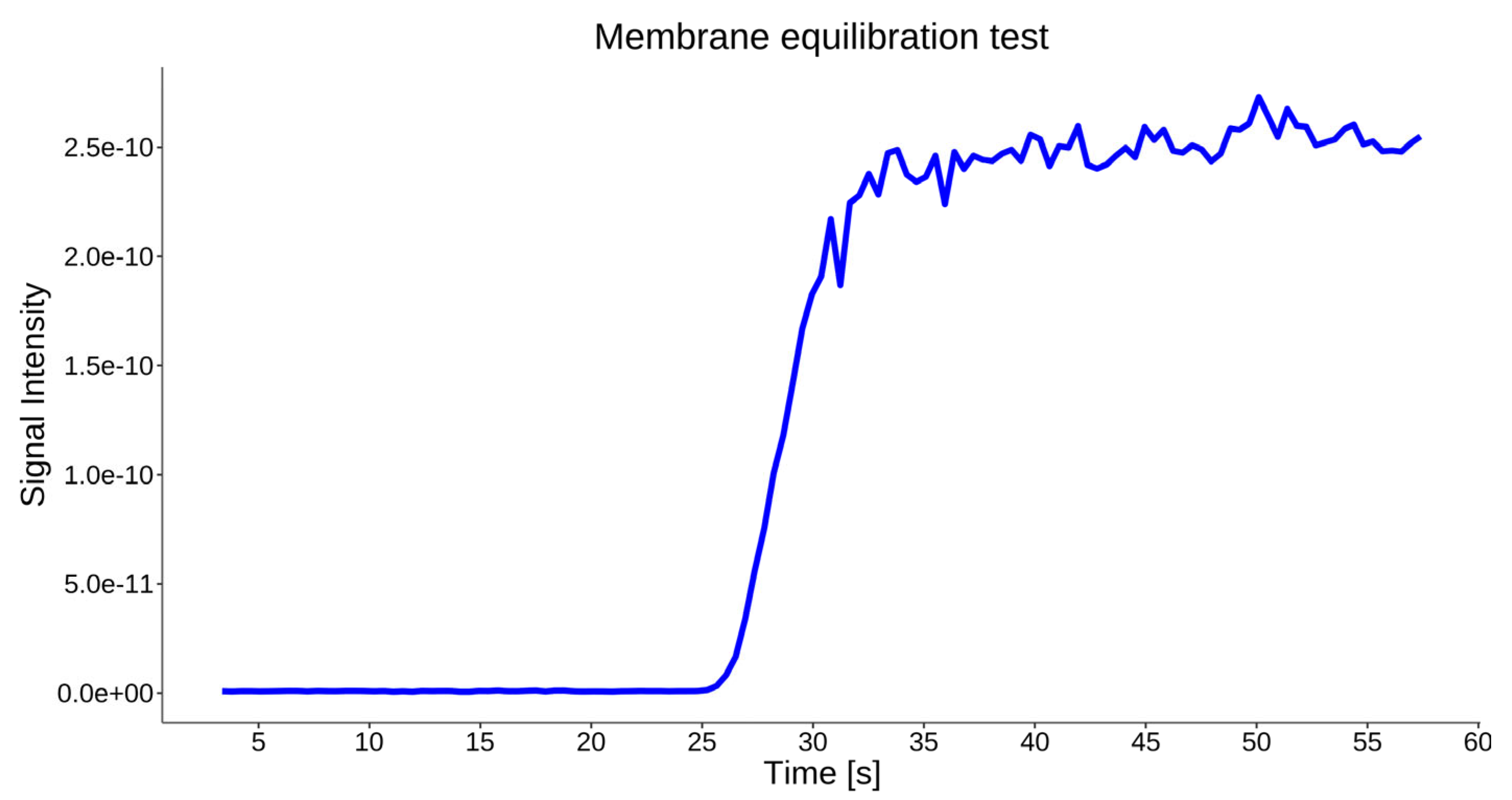
4.5. System Performance and Signal Stability
4.5. Field Deployment—Pilot Experiments
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BTX | Benzene, Toluene, Xylene |
| CVD | Cardiovascular disease |
| EI | Electron ionization |
| GC-MS | Gass chromatography—mass spectrometry |
| HPLC | High performance liquid chromatography |
| LOD | Limit of detection |
| LOQ | Limit of quantification |
| MIMS | Membrane Inlet Mass Spectrometry |
| PDMS | Polydimethylsiloxane |
| PTR-MS | Proton-transfer-reaction mass spectrometry |
| PTFE | Polytetrafluoroethylene |
| SIM | Selected ion monitoring |
| S/N | Signal-to-noise |
| VOC | Volatile organic compounds |
References
- Mediterranean Region. In Climate Change 2022—Impacts, Adaptation and Vulnerability: Working Group II Contribution to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Intergovernmental Panel on Climate Change (IPCC), Ed.; Cambridge University Press: Cambridge, 2023; pp. 2233–2272 ISBN 978-1-009-32583-7.
- Toscano, D. The Impact of Shipping on Air Quality in the Port Cities of the Mediterranean Area: A Review. Atmosphere 2023, 14, 1180. [Google Scholar] [CrossRef]
- European Environment Agency. European Maritime Safety Agency European Maritime Transport Environmental Report 2025; Publications Office of the European Union: Luxembourg, 2025. [Google Scholar]
- Xie, M.; Li, Y.; Zhang, Z.; Fu, Q.; Jiang, H. Remote Sensing of the Oil Spills Caused by Ships: A Review. Marine Pollution Bulletin 2025, 214, 117754. [Google Scholar] [CrossRef]
- Jha, M.N.; Levy, J.; Gao, Y. Advances in Remote Sensing for Oil Spill Disaster Management: State-of-the-Art Sensors Technology for Oil Spill Surveillance. Sensors 2008, 8, 236–255. [Google Scholar] [CrossRef]
- Polinov, S.; Bookman, R.; Levin, N. Spatial and Temporal Assessment of Oil Spills in the Mediterranean Sea. Marine Pollution Bulletin 2021, 167, 112338. [Google Scholar] [CrossRef]
- Boviatsis, M.; Alexopoulos, A.B.; Vlachos, G.P. Evaluation of the Response to Emerging Environmental Threats, Focusing on Carbon Dioxide (CO2), Volatile Organic Compounds (VOCs), and Scrubber Wash Water (SOx). Euro-Mediterranean Journal for Environmental Integration 2022, 7, 391–398. [Google Scholar] [CrossRef]
- Databases & Data Centre—EMSA—European Maritime Safety Agency. Available online: https://www.emsa.europa.eu/we-do/digitalisation/465-databases.html (accessed on 26 February 2026).
- Oil Tanker Spill Statistics 2025—ITOPF. Available online: https://www.itopf.org/knowledge-resources/data-statistics/oil-tanker-spill-statistics-2025/ (accessed on 26 February 2026).
- Hou, Y.; Li, Y.; Liu, B.; Liu, Y.; Wang, T. Design and Implementation of a Coastal-Mounted Sensor for Oil Film Detection on Seawater. Sensors 2017, 18, 70. [Google Scholar] [CrossRef]
- Worton, D.R.; Zhang, H.; Isaacman-VanWertz, G.; Chan, A.W.H.; Wilson, K.R.; Goldstein, A.H. Comprehensive Chemical Characterization of Hydrocarbons in NIST Standard Reference Material 2779 Gulf of Mexico Crude Oil. Environ. Sci. Technol. 2015, 49, 13130–13138. [Google Scholar] [CrossRef] [PubMed]
- Gros, J.; Nabi, D.; Würz, B.; Wick, L.Y.; Brussaard, C.P.D.; Huisman, J.; Van Der Meer, J.R.; Reddy, C.M.; Arey, J.S. First Day of an Oil Spill on the Open Sea: Early Mass Transfers of Hydrocarbons to Air and Water. Environ. Sci. Technol. 2014, 48, 9400–9411. [Google Scholar] [CrossRef]
- Drozd, G.T.; Worton, D.R.; Aeppli, C.; Reddy, C.M.; Zhang, H.; Variano, E.; Goldstein, A.H. Modeling Comprehensive Chemical Composition of Weathered Oil Following a Marine Spill to Predict Ozone and Potential Secondary Aerosol Formation and Constrain Transport Pathways. Journal of Geophysical Research: Oceans 2015, 120, 7300–7315. [Google Scholar] [CrossRef]
- Giannoukos, S.; Agapiou, A.; Brkić, B.; Taylor, S. Volatolomics: A Broad Area of Experimentation. Journal of Chromatography B 2019, 1105, 136–147. [Google Scholar] [CrossRef]
- Li, X.D. and Y. Sources and Fates of BTEX in the General Environment and Its Distribution in Coastal Cities of China. Journal of Environmental Science and Public Health, 2017; 1, pp. 86–106. [CrossRef]
- Shukla, D.; Czech, H.; Kokkola, T.; Bauer, M.; Paul, A.; Etzien, U.; Ihalainen, M.; Fang, Z.; Hartikainen, A.; Gawlitta, N.; et al. Emission Speciation of Volatile and Intermediate Volatility Organic Compounds from a Marine Engine: Effect of Engine Load, Fuel Type and Photochemical Aging. Environmental Science: Atmospheres 2025, 5, 973–986. [Google Scholar] [CrossRef]
- Yuewen, D.; Adzigbli, L. Assessing the Impact of Oil Spills on Marine Organisms. J Oceanogr Mar Res 2018, 6, 179. [Google Scholar] [CrossRef]
- Acharya, S.M.; Chakraborty, R.; Tringe, S.G. Emerging Trends in Biological Treatment of Wastewater From Unconventional Oil and Gas Extraction. Front. Microbiol. 2020, 11, 569019. [Google Scholar] [CrossRef] [PubMed]
- Patel, O.P.; Edwards, J.K.; Kucharska-Newton, A.M.; Whitsel, E.A.; Christenbury, K.; Jackson, W.B.; Lawrence, K.G.; Stewart, P.A.; Stenzel, M.R.; Sandler, D.P.; et al. Association of Oil Spill-Related Volatile Organic Compound Exposure with CVD-Related Biomarkers in the Gulf Long-Term Follow-up Study. Ann Work Expo Health 2026, 70, wxaf091. [Google Scholar] [CrossRef]
- Davey, N.G.; Krogh, E.T.; Gill, C.G. Membrane-Introduction Mass Spectrometry (MIMS). TrAC Trends in Analytical Chemistry 2011, 30, 1477–1485. [Google Scholar] [CrossRef]
- Johnson, R.C.; Cooks, R.G.; Allen, T.M.; Cisper, M.E.; Hemberger, P.H. Membrane Introduction Mass Spectrometry: Trends and Applications. Mass Spectrom. Rev. 2000, 19, 1–37. [Google Scholar] [CrossRef]
- Ketola, R.A.; Kotiaho, T.; Cisper, M.E.; Allen, T.M. Environmental Applications of Membrane Introduction Mass Spectrometry. Journal of Mass Spectrometry 2002, 37, 457–476. [Google Scholar] [CrossRef]
- Jakšić, M.; Simeon, A.; Vujić, D.; Giannoukos, S.; Brkić, B. Membrane Inlet Mass Spectrometry Method for Food Intake Impact Assessment on Specific Volatile Organic Compounds in Exhaled Breath. Analytical and Bioanalytical Chemistry 2022, 414, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Giannoukos, S.; Brkić, B.; Taylor, S. Analysis of Chlorinated Hydrocarbons in Gas Phase Using a Portable Membrane Inlet Mass Spectrometer. Anal. Methods 2016, 8, 6607–6615. [Google Scholar] [CrossRef]
- Brkić, B.; France, N.; Taylor, S. Oil-in-Water Monitoring Using Membrane Inlet Mass Spectrometry. Anal. Chem. 2011, 83, 6230–6236. [Google Scholar] [CrossRef] [PubMed]
- Brkić, B.; Giannoukos, S.; Taylor, S.; Lee, D.F. Mobile Mass Spectrometry for Water Quality Monitoring of Organic Species Present in Nuclear Waste Ponds. Anal. Methods 2018, 10, 5827–5833. [Google Scholar] [CrossRef]
- Tortell, P.D. Dissolved Gas Measurements in Oceanic Waters Made by Membrane Inlet Mass Spectrometry. Limnology and Oceanography: Methods 2005, 3, 24–37. [Google Scholar] [CrossRef]
- Chua, E.J.; Savidge, W.; Short, R.T.; Cardenas-Valencia, A.M.; Fulweiler, R.W. A Review of the Emerging Field of Underwater Mass Spectrometry. Front. Mar. Sci. 2016, 3. [Google Scholar] [CrossRef]
- Astrahan, P. Monocyclic Aromatic Hydrocarbons (Phthalates and BTEX) and Aliphatic Components in the SE Mediterranean Costal Sea-Surface Microlayer (SML): Origins and Phase Distribution Analysis. Marine Chemistry 2018, 205, 56–69. [Google Scholar] [CrossRef]
- Rocco, M.; Dunne, E.; Peltola, M.; Barr, N.; Williams, J.; Colomb, A.; Safi, K.; Saint-Macary, A.; Marriner, A.; Deppeler, S.; et al. Oceanic Phytoplankton Are a Potentially Important Source of Benzenoids to the Remote Marine Atmosphere. Commun Earth Environ 2021, 2, 175. [Google Scholar] [CrossRef]
- Chary, N.S.; Fernandez-Alba, A.R. Determination of Volatile Organic Compounds in Drinking and Environmental Waters. TrAC Trends in Analytical Chemistry 2012, 32, 60–75. [Google Scholar] [CrossRef]
- Camilli, R.; Duryea, A.N. Characterizing Spatial and Temporal Variability of Dissolved Gases in Aquatic Environments with in Situ Mass Spectrometry. Environ. Sci. Technol. 2009, 43, 5014–5021. [Google Scholar] [CrossRef] [PubMed]
- Kartalovic, B.; Vujić, D.; Ilić, D.; Brkić, B. Development and Validation of Portable Membrane Inlet Mass Spectrometry Method for Measurements of Monoaromatic Hydrocarbons from the River Canal. Analytical Methods 2024, 16. [Google Scholar] [CrossRef]

| Compound | CAS number | Molecular weight (g mol-1) |
Target ion (m/z) |
|---|---|---|---|
| Benzene | 71-43-2 | 78.11 | 78,77 |
| Toluene | 108-88-3 | 92.14 | 91,92 |
| Xylenes | 1330-20-7 | 106.1 | 105, 106 |
| 1,2-dichloroethane | 107-06-2 | 98.96 | 62, 64 |
| Tetrachloroethylene | 127-18-4 | 165.8 | 166, 164 |
| Compound | LODa | LOQa | Equation | R2 |
|---|---|---|---|---|
| Benzene | 4.90 | 16.3 | 0.998 | |
| Toluene | 7.50 | 24.8 | 0.999 | |
| Xylenes | 7.50 | 24.8 | 0.995 | |
| Tetrachloroethylene | 3.50 | 11.6 | 0.999 | |
| 1,2-dichloroethane | 4.30 | 14.4 | 0.999 | |
| aconcentration in µg L⁻¹ | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




