Submitted:
18 March 2026
Posted:
23 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
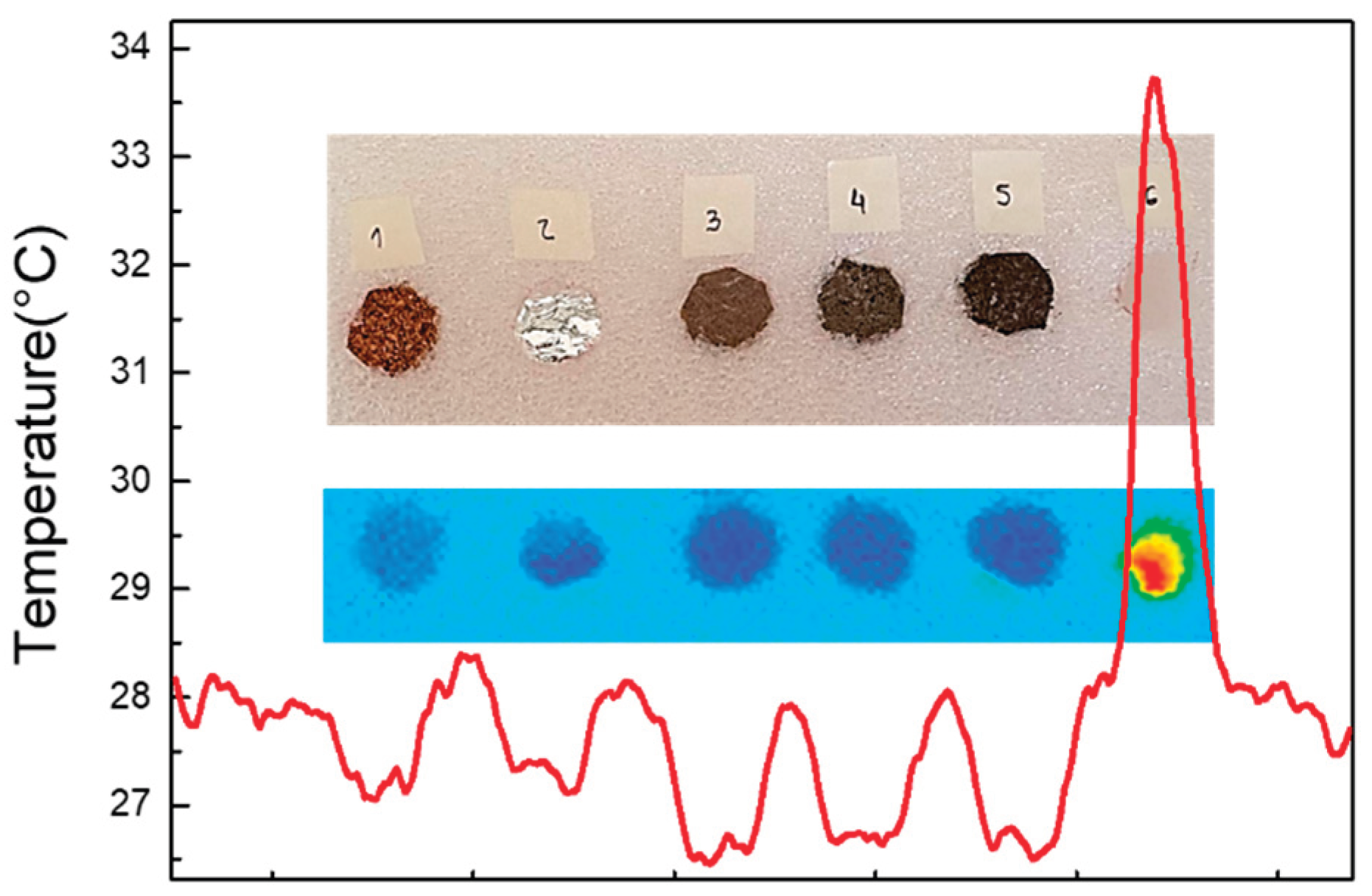
2.1. Characterization of Materials
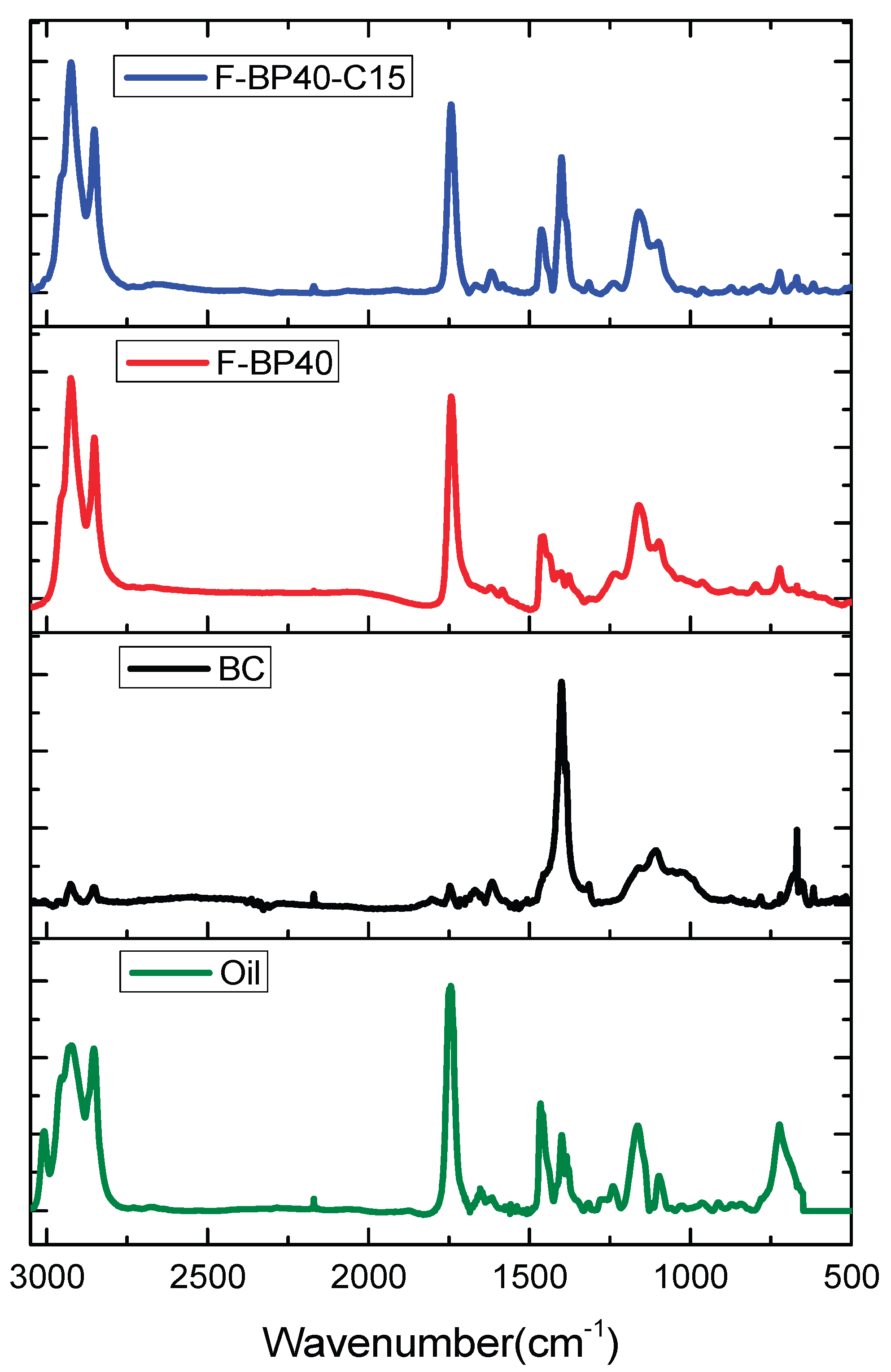
2.1.1. FT-IR
2.1.2. SEM Images
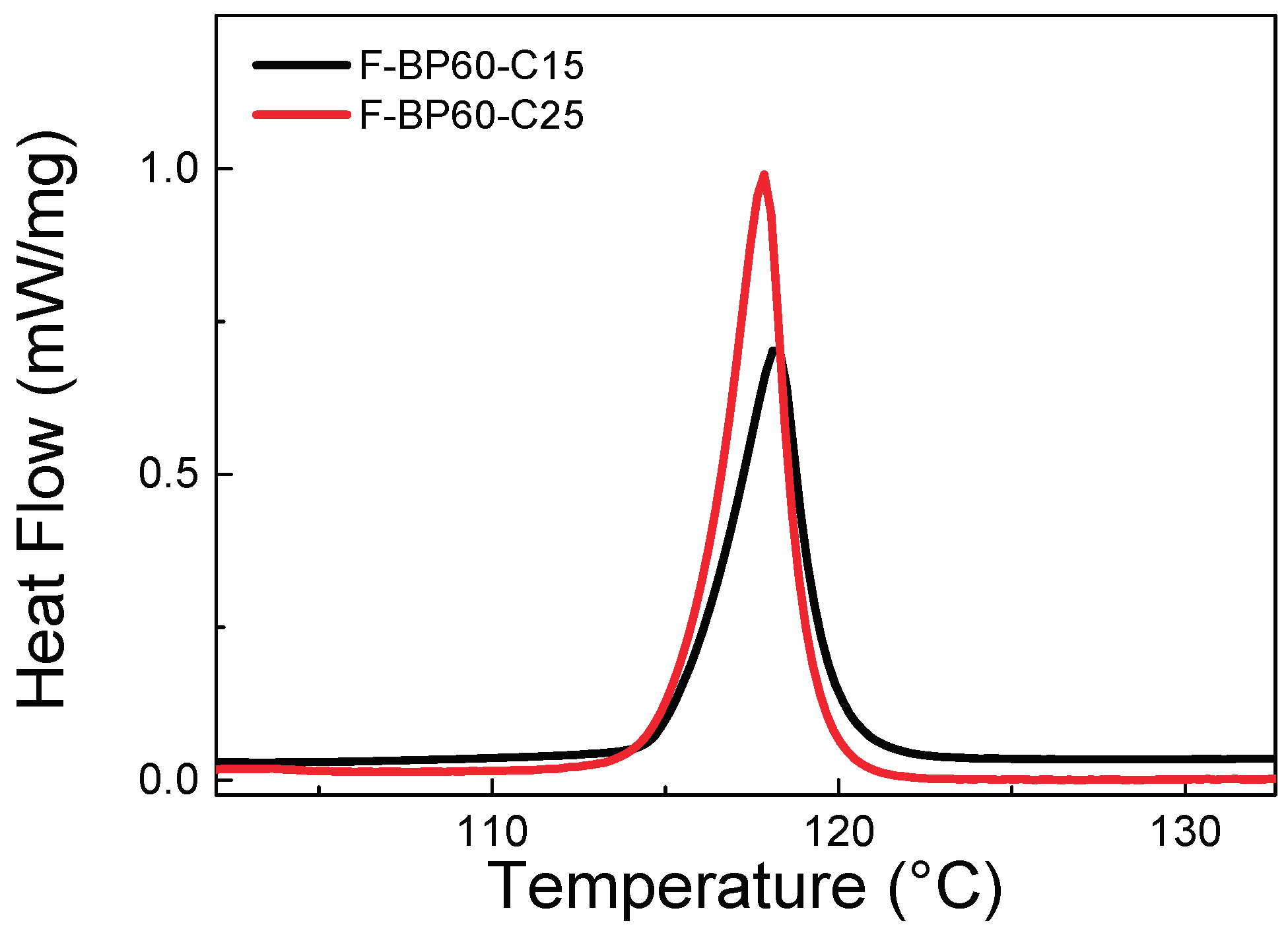
2.1.2. Thermal Properties
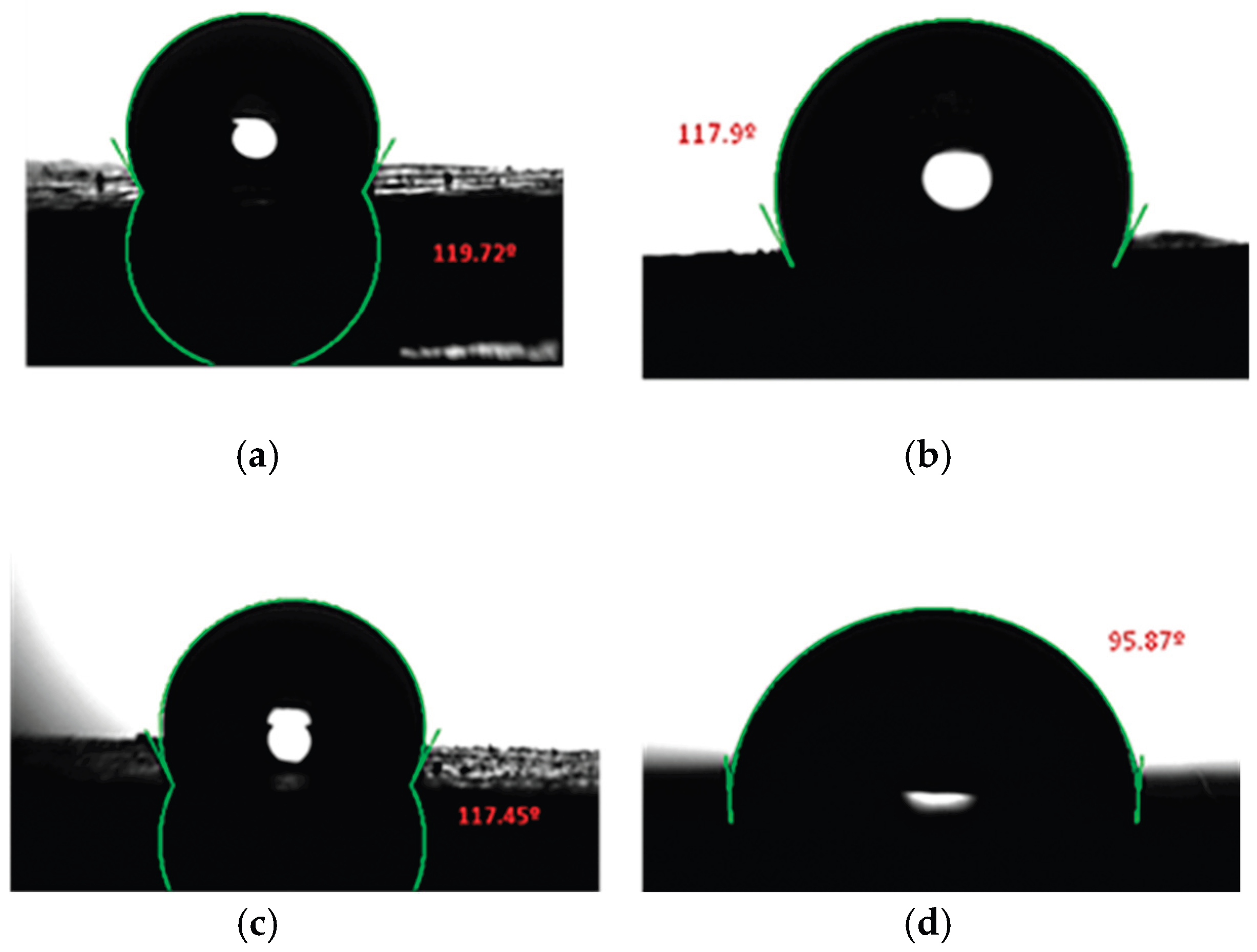
2.1.3. Contact Angle
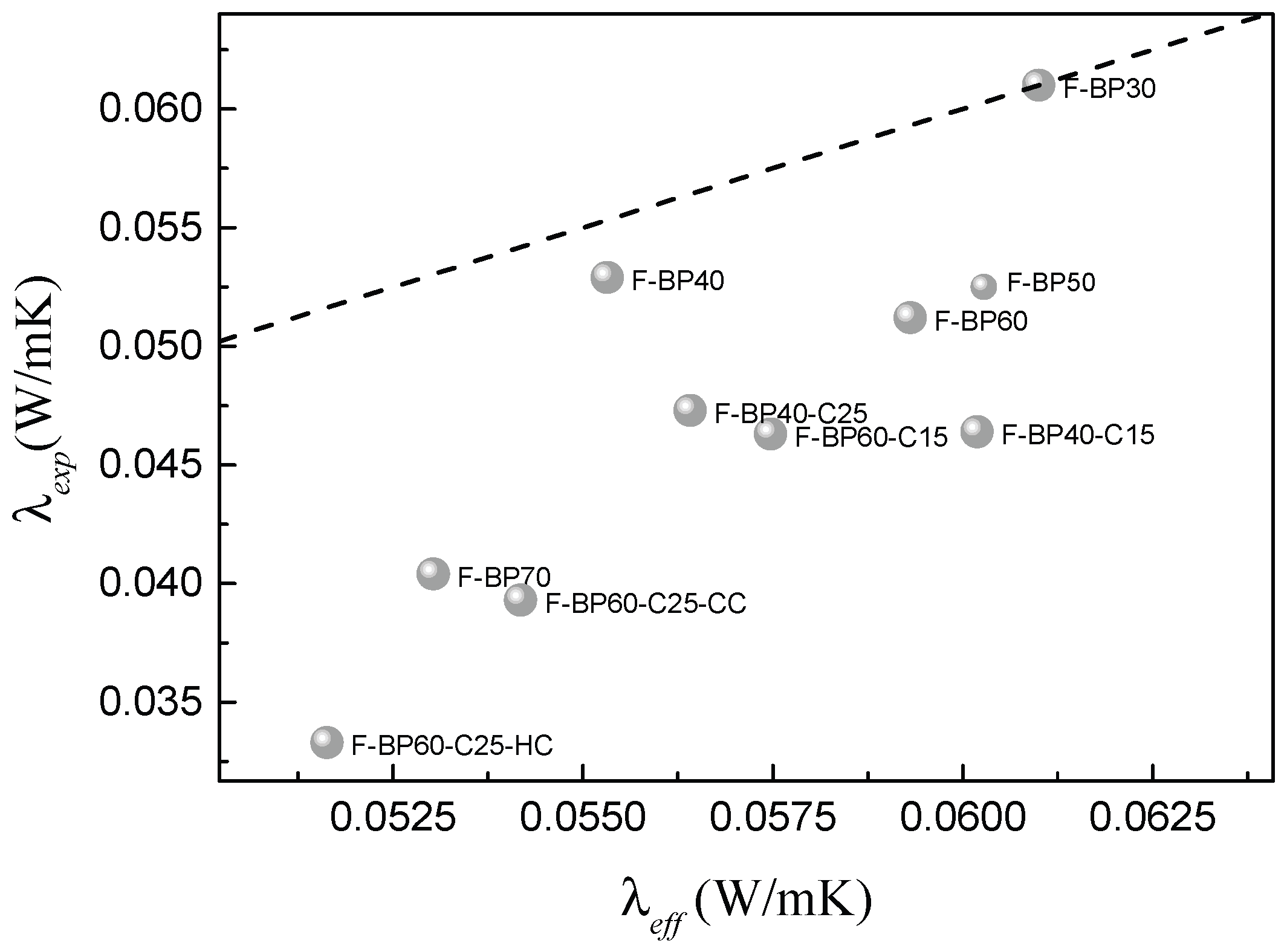
2.2. Thermal Conductivity
2.2.1. Effect of Sulfur Content and Porosity
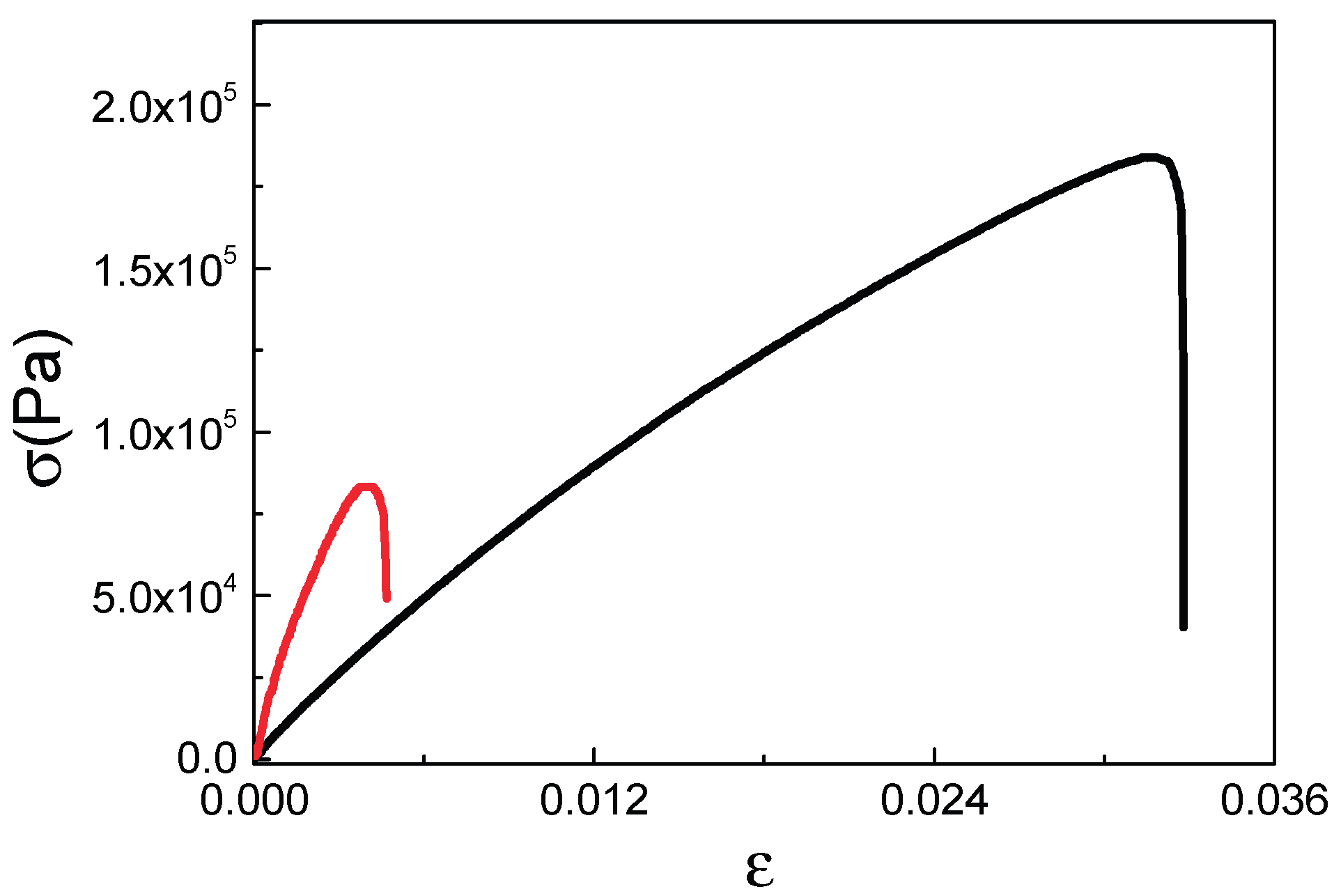
| Sample | Young modulus (MPa) | Tensile strength σmax (MPa) | Elongation at break (%) |
|---|---|---|---|
| F-BP60-25-CC | 19.15 ± 1.25 | 0.088 ± 0.004 | 0.46 ± 0.05 |
| F-BP60-25-HC | 5.83 ± 0.73 | 0.185 ± 0.022 | 3.17 ± 0.02 |
3. Materials and Methods
3.1. Experimental
3.1.1. Synthesis of Sulfur-Soybean Oil Biopolymers
3.1.2. Biomass Filler Preparation
3.1.3. Carbonization Process
3.1.4. Biobased Film Fabrication
3.2. Materials Characterization
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BC | Biochar |
| F-BP | Sulfur–soybean oil biopolymer films |
| F-BP-C | Biochar-filled sulfur–soybean oil biocomposites films |
| DSC | Differential scanning calorimetry |
| SEM | Scanning Electron Microscopy |
| FT-IR | Fourier Transform Infrared Spectroscopy |
| wt% | Weight percentage |
| CA | Contact angle |
| λₜ | Thermal conductivity |
| EPS | Expanded polystyrene |
| φ | Porosity |
| PLA | Polylactic acid |
| CP | Cellulose palmitate |
| DVB | Divinylbenzene |
Appendix
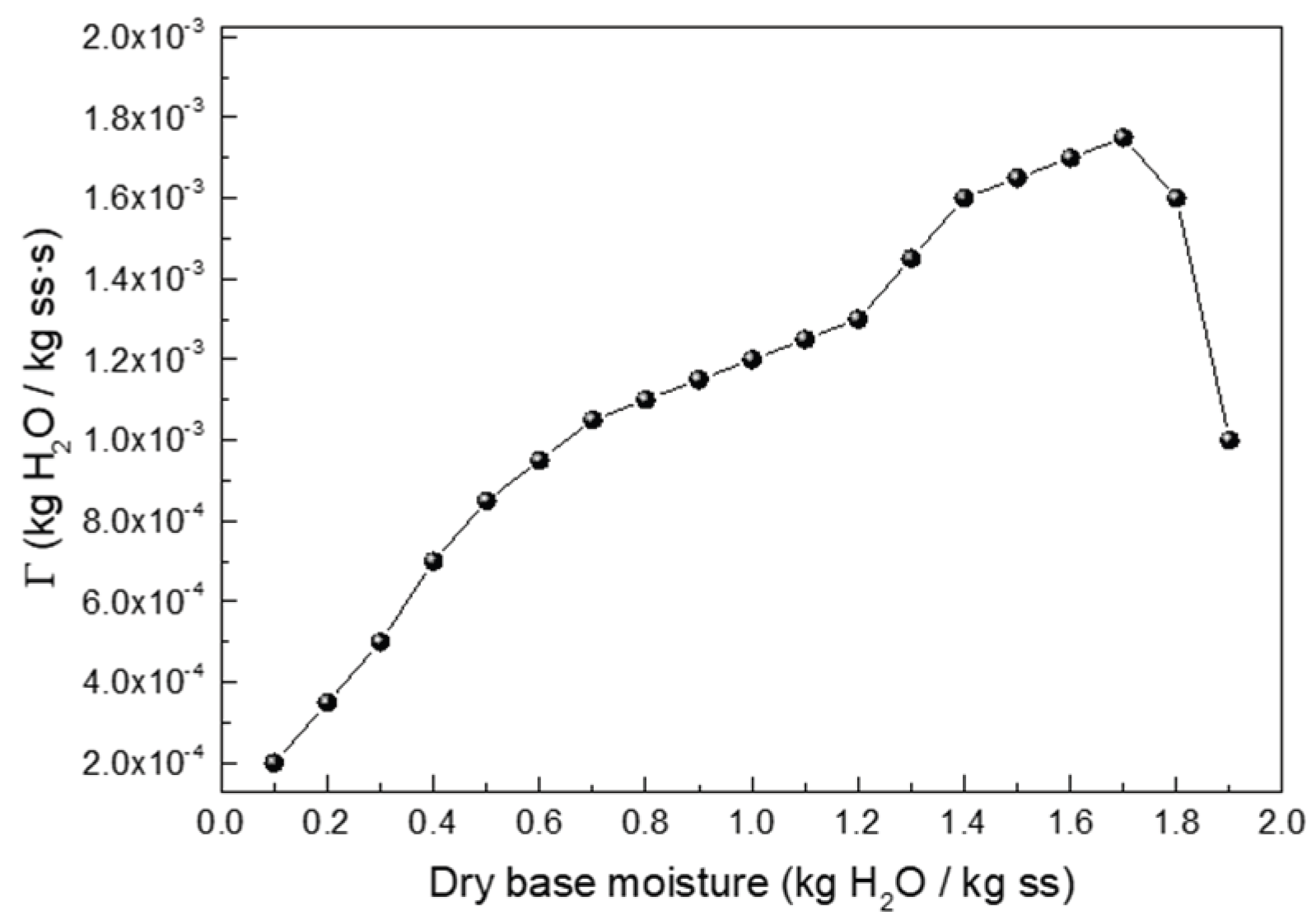
Appendix A1. Additional Drying Data
References
- Zhang, X.; Yin, Z.; Xiang, S.; Yan, H.; Tian, H. Degradation of Polymer Materials in the Environment and Its Impact on the Health of Experimental Animals: A Review. Polymers 2024, 16, 2807. [Google Scholar] [CrossRef]
- Farioli, A.S.; Martinez, M.V.; Barbero, C.; Yslas, E.; Acevedo, D. The Effect of Oil Raw Material Composition in the Synthesis of Bio-Sorbents Based on Inverse Vulcanization on the Ability to Remediate Hydrocarbon-Contaminated Water. A Novel Method for Decontaminating Water/Fuel Emulsions. J. Appl. Polym. Sci. 2024, 141, e54914. [Google Scholar] [CrossRef]
- Rovatta, L.M.; de Prada, R.E.; Bruno, M.M.; Acevedo, D.F.; Monti, G.A. Structure–Property Relationships and Mechanical Performance of Inverse Vulcanized Sulfur-Soybean Oil Polymer Films and Their Biochar-Reinforced Biocomposites. ACS Appl. Polym. Mater. 2025, 7, 15316–15328. [Google Scholar] [CrossRef]
- Dutta, D.; Sit, N. A Comprehensive Review on Types and Properties of Biopolymers as Sustainable Bio-based Alternatives for Packaging. Food Biomacromolecules 2024, 1, 58–87. [Google Scholar] [CrossRef]
- Benalaya, I.; Alves, G.; Lopes, J.; Silva, L.R. A Review of Natural Polysaccharides: Sources, Characteristics, Properties, Food, and Pharmaceutical Applications. International Journal of Molecular Sciences 2024, 25(2024), 1322. [Google Scholar] [CrossRef]
- Lundquist, N.A.; Tikoalu, A.D.; Worthington, M.J.H.; Shapter, R.; Tonkin, S.J.; Stojcevski, F.; Mann, M.; Gibson, C.T.; Gascooke, J.R.; Karton, A.; et al. Reactive Compression Molding Post-Inverse Vulcanization: A Method to Assemble, Recycle, and Repurpose Sulfur Polymers and Composites. Chemistry—A European Journal 2020, 26, 10035–10044. [Google Scholar] [CrossRef]
- Ammar, S.; Iling, A.W.M.; Ramesh, K.; Ramesh, S. Development of Fully Organic Coating System Modified with Epoxidized Soybean Oil with Superior Corrosion Protection Performance. Prog. Org. Coat. 2020, 140, 105523. [Google Scholar] [CrossRef]
- Wagenfeld, J.G.; Al-Ali, K.; Almheiri, S.; Slavens, A.F.; Calvet, N. Sustainable Applications Utilizing Sulfur, a by-Product from Oil and Gas Industry: A State-of-the-Art Review. Waste Management 2019, 95, 78–89. [Google Scholar] [CrossRef]
- Chung, W.J.; Griebel, J.J.; Kim, E.T.; Yoon, H.; Simmonds, A.G.; Ji, H.J.; Dirlam, P.T.; Glass, R.S.; Wie, J.J.; Nguyen, N.A.; et al. The Use of Elemental Sulfur as an Alternative Feedstock for Polymeric Materials. Nat. Chem. 2013, 5, 518–524. [Google Scholar] [CrossRef] [PubMed]
- Chalker, J.M.; Worthington, M.J.H.; Lundquist, N.A.; Esdaile, L.J. Synthesis and Applications of Polymers Made by Inverse Vulcanization. Top. Curr. Chem. 2019, 377, 16. [Google Scholar] [CrossRef] [PubMed]
- Simmonds, A.G.; Griebel, J.J.; Park, J.; Kim, K.R.; Chung, W.J.; Oleshko, V.P.; Kim, J.; Kim, E.T.; Glass, R.S.; Soles, C.L.; et al. Inverse Vulcanization of Elemental Sulfur to Prepare Polymeric Electrode Materials for Li–S Batteries. ACS Macro Lett. 2014, 3, 229–232. [Google Scholar] [CrossRef] [PubMed]
- Tominaga, H.; Tokomoto, J.; Onimura, K.; Yamabuki, K. Synthesis of High-Sulfur-Content Resins via Inverse Vulcanization Using Dithiols and Their Application as Cathode Materials for Lithium–Sulfur Rechargeable Batteries. Electrochem 2025, 6(2025), 8. [Google Scholar] [CrossRef]
- Dodd, L.J. Inverse Vulcanisation: A New Starter’s Guide to an Emerging Field. RSC Applied Polymers 2025, 3, 10–42. [Google Scholar] [CrossRef]
- Chung, W.J.; Griebel, J.J.; Kim, E.T.; Yoon, H.; Simmonds, A.G.; Ji, H.J.; Dirlam, P.T.; Glass, R.S.; Wie, J.J.; Nguyen, N.A.; et al. The Use of Elemental Sulfur as an Alternative Feedstock for Polymeric Materials. Nature Chemistry 2013, 5 5, 518–524. [Google Scholar] [CrossRef]
- Mohammed, M.; Oleiwi, J.K.; Mohammed, A.M.; Jawad, A.J.M.; Osman, A.F.; Adam, T.; Betar, B.O.; Gopinath, S.C.B. A Review on the Advancement of Renewable Natural Fiber Hybrid Composites: Prospects, Challenges, and Industrial Applications. J. Renew. Mater. 2024, 12, 1237–1290. [Google Scholar] [CrossRef]
- Tengku Yasim-Anuar, T.A.; Yee-Foong, L.N.; Lawal, A.A.; Ahmad Farid, M.A.; Mohd Yusuf, M.Z.; Hassan, M.A.; Ariffin, H. Emerging Application of Biochar as a Renewable and Superior Filler in Polymer Composites. RSC Adv. 2022, 12, 13938–13949. [Google Scholar] [CrossRef]
- Sadok, I.; Krzyszczak-Turczyn, A.; Czech, B.; Parlakidis, P.; Vryzas, Z. Advancements in Biochar-Based Materials for Decontamination and Analytical Detection of Pesticides and Mycotoxins in Food. Food Chem. 2025, 492, 145467. [Google Scholar] [CrossRef]
- Laishram, D.; Kim, S. Bin; Lee, S.Y.; Park, S.J. Advancements in Biochar as a Sustainable Adsorbent for Water Pollution Mitigation. Advanced Science 2025, 12, 2410383. [Google Scholar] [CrossRef]
- Giorcelli, M.; Bartoli, M.; Sanginario, A.; Padovano, E.; Rosso, C.; Rovere, M.; Tagliaferro, A. High-Temperature Annealed Biochar as a Conductive Filler for the Production of Piezoresistive Materials for Energy Conversion Application. ACS Appl. Electron. Mater. 2021, 3, 838–844. [Google Scholar] [CrossRef]
- Zhang, Q.; Xu, H.; Lu, W.; Zhang, D.; Ren, X.; Yu, W.; Wu, J.; Zhou, L.; Han, X.; Yi, W.; et al. Properties Evaluation of Biochar/High-Density Polyethylene Composites: Emphasizing the Porous Structure of Biochar by Activation. Science of The Total Environment 2020, 737, 139770. [Google Scholar] [CrossRef] [PubMed]
- Hoque, M.M.; Saha, B.K.; Scopa, A.; Drosos, M. Biochar in Agriculture: A Review on Sources, Production, and Composites Related to Soil Fertility, Crop Productivity, and Environmental Sustainability. C 2025, 11, 50. [Google Scholar] [CrossRef]
- Waheed, A.; Xu, H.; Qiao, X.; Aili, A.; Yiremaikebayi, Y.; Haitao, D.; Muhammad, M. Biochar in Sustainable Agriculture and Climate Mitigation: Mechanisms, Challenges, and Applications in the Circular Bioeconomy. Biomass Bioenergy 2025, 193, 107531. [Google Scholar] [CrossRef]
- Ali, A.; Issa, A.; Elshaer, A. A Comprehensive Review and Recent Trends in Thermal Insulation Materials for Energy Conservation in Buildings. Sustainability 2024, 16(2024), 8782. [Google Scholar] [CrossRef]
- Wadi, V.S.; Jena, K.K.; Khawaja, S.Z.; Ranagraj, V.M.; Alhassan, S.M. Preparation and Processing of Porous Sulfur Foams Having Low Thermal Conductivity. RSC Adv. 2019, 9, 4397–4403. [Google Scholar] [CrossRef]
- Bolloju, S.; Chang, Y.L.; Sharma, S.U.; Hsu, M.F.; Lee, J.T. Vulcanized Polyisoprene-Graft-Maleic Anhydride as an Efficient Binder for Silicon Anodes in Lithium-Ion Batteries. Electrochim. Acta 2022, 419, 140390. [Google Scholar] [CrossRef]
- Meyer, B. Elemental Sulfur. Chem. Rev. 1976, 76, 367–388. [Google Scholar] [CrossRef]
- van de Voort, F.R.; Ismail, A.A.; Sedman, J.; Dubois, J.; Nicodemo, T. The Determination of Peroxide Value by Fourier Transform Infrared Spectroscopy. J. Am. Oil Chem. Soc. 1994, 71, 921–926. [Google Scholar] [CrossRef]
- Ahmad, M.; Rajapaksha, A.U.; Lim, J.E.; Zhang, M.; Bolan, N.; Mohan, D.; Vithanage, M.; Lee, S.S.; Ok, Y.S. Biochar as a Sorbent for Contaminant Management in Soil and Water: A Review. Chemosphere 2014, 99, 19–33. [Google Scholar] [CrossRef]
- Armynah, B.; Atika; Djafar, Z.; Piarah, W.H.; Tahir, D. Analysis of Chemical and Physical Properties of Biochar from Rice Husk Biomass. J. Phys. Conf. Ser. 2018, 979, 012038. [Google Scholar] [CrossRef]
- Bartoli, M.; Arrigo, R.; Malucelli, G.; Tagliaferro, A.; Duraccio, D. Recent Advances in Biochar Polymer Composites. Polymers (Basel). 2022, 14, 2506. [Google Scholar] [CrossRef]
- Salman, M.K.; Karabay, B.; Karabay, L.C.; Cihaner, A. Elemental Sulfur-based Polymeric Materials: Synthesis and Characterization. J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Kritikaki, A.; Karmali, V.; Vathi, D.; Bartzas, G.; Komnitsas, K. Advanced Characterization of Biochars Produced from Three Different Organic-Based Feedstocks and Their Potential Applications. Circular Economy and Sustainability 2025, 5, 7465–7488. [Google Scholar] [CrossRef]
- Shainova, R.; Muradyan, N.; Arzumanyan, A.; Kalantaryan, M.; Sukiasyan, R.; Yeranosyan, M.; Melikyan, Y.; Simonyan, A.; Laroze, D.; Zendri, E.; et al. Effect of Pyrolysis Temperature on the Performance of Malt Biochar in Cement Mortars. Materials 2025, 18, 5105. [Google Scholar] [CrossRef] [PubMed]
- Reza, M.S.; Taweekun, J.; Afroze, S.; Siddique, S.A.; Islam, M.S.; Wang, C.; Azad, A.K. Investigation of Thermochemical Properties and Pyrolysis of Barley Waste as a Source for Renewable Energy. Sustainability 2023, 15, 1643. [Google Scholar] [CrossRef]
- Aboughaly, M.; Babaei-Ghazvini, A.; Dhar, P.; Patel, R.; Acharya, B. Enhancing the Potential of Polymer Composites Using Biochar as a Filler: A Review. Polymers 2023, 15, 3981. [Google Scholar] [CrossRef]
- Vengadesan, E.; Morakul, S.; Muralidharan, S.; Pullela, P.K.; Alarifi, A.; Arunkumar, T. Enhancement of Polylactic Acid (PLA) with Hybrid Biomass-Derived Rice Husk and Biocarbon Fillers: A Comprehensive Experimental Study. Discover Applied Sciences 2025, 7. [Google Scholar] [CrossRef]
- Antimicrobial, E.; Farioli, A.S.; Martinez, M. V; Barbero, C.A.; Acevedo, D.F.; Yslas, E.I. Antimicrobial Activity of Gentamicin-Loaded Biocomposites Synthesized through Inverse Vulcanization from Soybean and Sunflower Oils. Sustainable Chemistry 2024, 5, 229–243. [Google Scholar] [CrossRef]
- Chalker, J.M.; Worthington, M.J.H.; Lundquist, N.A.; Esdaile, L.J. Synthesis and Applications of Polymers Made by Inverse Vulcanization. 2019, 125–151. [Google Scholar] [CrossRef]
- He, X.M.; Wang, L.; Pu, W.H.; Ren, J.G.; Wu, W.; Jiang, C.Y.; Wan, C.R. Thermal Analysis of Sulfurization of Polyacrylonitrile with Elemental Sulfur. J. Therm. Anal. Calorim. 2008, 94, 151–155. [Google Scholar] [CrossRef]
- Ficara, M.L.G.; Moreira, E. Utilization of Differential Scanning Calorimetry Technique for Determination of High Sulfur Content. Thermochim. Acta 1988, 134, 435–440. [Google Scholar] [CrossRef]
- Farioli, A.S.; Martinez, M.V.; Barbero, C.; Yslas, E.; Acevedo, D. The Effect of Oil Raw Material Composition in the Synthesis of Bio-sorbents Based on Inverse Vulcanization on the Ability to Remediate Hydrocarbon-contaminated Water. A Novel Method for Decontaminating Water/Fuel Emulsions. J. Appl. Polym. Sci. 2023, 141. [Google Scholar] [CrossRef]
- Worthington, M.J.H.; Kucera, R.L.; Chalker, J.M. Green Chemistry and Polymers Made from Sulfur. Green Chemistry 2017, 19, 2748–2761. [Google Scholar] [CrossRef]
- Abbasi, A.; Yahya, W.Z.N.; Nasef, M.M.; Moniruzzaman, M.; Ghumman, A.S.M. Copolymerization of Palm Oil with Sulfur Using Inverse Vulcanization to Boost the Palm Oil Industry. Polymers and Polymer Composites 2021, 29, S1446–S1456. [Google Scholar] [CrossRef]
- Chung, W.J.; Griebel, J.J.; Kim, E.T.; Yoon, H.; Simmonds, A.G.; Ji, H.J.; Dirlam, P.T.; Glass, R.S.; Wie, J.J.; Nguyen, N.A.; et al. The Use of Elemental Sulfur as an Alternative Feedstock for Polymeric Materials. Nat. Chem. 2013, 5, 518–524. [Google Scholar] [CrossRef]
- Diez, S.; Hoefling, A.; Theato, P.; Pauer, W. Mechanical and Electrical Properties of Sulfur-Containing Polymeric Materials Prepared via Inverse Vulcanization. Polymers (Basel). 2017, 9, 59. [Google Scholar] [CrossRef]
- Dahal, R.K.; Acharya, B.; Dutta, A. Thermal Response of Biocarbon-Filled Hemp Fiber-Reinforced Bioepoxy Composites. ACS Omega 2023, 8, 15422–15440. [Google Scholar] [CrossRef] [PubMed]
- Minugu, O.P.; Gujjala, R.; Shakuntala, O.; Manoj, P.; Chowdary, M.S. Effect of Biomass Derived Biochar Materials on Mechanical Properties of Biochar Epoxy Composites. Proc. Inst. Mech. Eng. C J. Mech. Eng. Sci. 2021, 235, 5626–5638. [Google Scholar] [CrossRef]
- Abbasi, A.; Taghinezhad, S.F.; Mansourieh, M.; Xu, H.; Nasef, M.M.; Major, I. Inverse Vulcanized Sulfur-Styrene Polymers as Effective Plasticizers for Polystyrene. Polym. Test. 2024, 140, 108625. [Google Scholar] [CrossRef]
- Genzer, J.; Efimenko, K. Creating Long-Lived Superhydrophobic Polymer Surfaces through Mechanically Assembled Monolayers. science.orgJ Genzer, K EfimenkoScience, 2000•science.org 2000, 290, 2130–2133. [Google Scholar] [CrossRef]
- Kota, A.; Kwon, G.; Materials, A.T.-N.A. 2014, undefined The Design and Applications of Superomniphobic Surfaces. nature.comAK Kota, G Kwon, A TutejaNPG Asia Materials, 2014•nature.com 2014, 6, 109. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, T.; Pan, H.; Yuan, Y.; Chen, L.; Liu, M.; Zhang, K.; Zhang, S.; Wu, P.; Xu, J. Photoemission Mechanism of Water-Soluble Silver Nanoclusters: Ligand-to-Metal–Metal Charge Transfer vs. Strong Coupling between Surface Plasmon and Emitters. J. Am. Chem. Soc. 2014, 136, 1686–1689. [Google Scholar] [CrossRef]
- Aboughaly, M.; Babaei-Ghazvini, A.; Dhar, P.; Patel, R.; Acharya, B. Enhancing the Potential of Polymer Composites Using Biochar as a Filler: A Review. Polymers 2023, 15, 3981. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Özgün, I.; Erci, V.; Madenci, E.; Erci, F. The Effect of Nano-Biochar Derived from Olive Waste on the Thermal and Mechanical Properties of Epoxy Composites. Polymers 2025, 17, 1337. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, A. Thermal Conductivity of Sulfur Accompanying Crystal Transition and Phase Change. J. Appl. Phys. 1965, 36, 2375–2377. [Google Scholar] [CrossRef]
- Daniels, A.U.; Chang, M.K. 0; Andriano, K.P.; Heller, J. Mechanical Properties of Biodegradable Polymers and Composites Proposed for Internal Fixation of Bone. [CrossRef]
- Cahill, D.G.; Ford, W.K.; Goodson, K.E.; Mahan, G.D.; Majumdar, A.; Maris, H.J.; Merlin, R.; Phillpot, S.R. Nanoscale Thermal Transport. J. Appl. Phys. 2003, 93, 793–818. [Google Scholar] [CrossRef]
- Henry, A.; Chen, G. High Thermal Conductivity of Single Polyethylene Chains Using Molecular Dynamics Simulations. Phys. Rev. Lett. 2008, 101, 235502. [Google Scholar] [CrossRef]
- Kannuluik, W.G.; Carman, E.H. The Temperature Dependence of the Thermal Conductivity of Air. Australian Journal of Scientific Research Series A: Physical Sciences 1951, 4, 305–314. [Google Scholar] [CrossRef]
- Islam, S.; Bhat, G. A Model for Predicting Thermal Conductivity of Porous Composite Materials. Heat and Mass Transfer 2023 2023, 59, 2023–2034. [Google Scholar] [CrossRef]
- Liang, J.Z.; Liu, G.S. A New Heat Transfer Model of Inorganic Particulate-Filled Polymer Composites. Journal of Materials Science 2009, 44, 4715–4720. [Google Scholar] [CrossRef]
- Giuma, A.; Khalil, H.P.S.A.; Yahya, E.B.; Sukeksi, L.; Alfatah, T.; Nurazzi, N.M.; Jaber, M.; Surya, I. Green Thermal Insulators: A Review into the Role of Biopolymer-Based Aerogels in Thermal Insulation Applications. Polym. Eng. Sci. 2024, 64, 4611–4629. [Google Scholar] [CrossRef]
- Zaidi, M.; Baillis, D.; Naouar, N.; Depriester, M.; Delattre, F. Thermal Conductivity and Microstructure of Novel Flaxseed-Gum-Filled Epoxy Resin Biocomposite: Analytical Models and X-Ray Computed Tomography. Materials 2023, 16, 6318. [Google Scholar] [CrossRef]
- He, B.; Mortazavi, B.; Zhuang, X.; Rabczuk, T. Modeling Kapitza Resistance of Two-Phase Composite Material. Compos. Struct. 2016, 152, 939–946. [Google Scholar] [CrossRef]
- Anokhina, T.S.; Ilyin, S.O.; Ignatenko, V.Y.; Bakhtin, D.S.; Kostyuk, A. V.; Antonov, S. V.; Volkov, A. V. Formation of Porous Films with Hydrophobic Surface from a Blend of Polymers. Polymer Science, Series A 2019, 61, 619–626. [Google Scholar] [CrossRef]
- Matias, L.; Santos, C.; Reis, M.; Gil, L. Declared Value for the Thermal Conductivity Coefficient of Insulation Corkboard. Wood Sci. Technol. 1997, 31, 355–365. [Google Scholar] [CrossRef]
- Ter-Zakaryan, K.A.; Zhukov, A.D.; Bobrova, E.Y.; Bessonov, I. V.; Mednikova, E.A. Foam Polymers in Multifunctional Insulating Coatings. Polymers (Basel). 2021, 13, 3698. [Google Scholar] [CrossRef]
- Anderson, D.R. Thermal Conductivity of Polymers. Chem. Rev. 1966, 66, 677–690. [Google Scholar] [CrossRef]
- Abraham, A.M.; Kumar, S.V.; Alhassan, S.M. Porous Sulphur Copolymer for Gas-Phase Mercury Removal and Thermal Insulation. Chemical Engineering Journal 2018, 332, 1–7. [Google Scholar] [CrossRef]
- Boussatour, G.; Cresson, P.Y.; Genestie, B.; Joly, N.; Brun, J.F.; Lasri, T. Measurement of the Thermal Conductivity of Flexible Biosourced Polymers Using the 3-Omega Method. Polym. Test. 2018, 70, 503–510. [Google Scholar] [CrossRef]
- Wadi, V.S.; Jena, K.K.; Khawaja, S.Z.; Ranagraj, V.M.; Alhassan, S.M. Preparation and Processing of Porous Sulfur Foams Having Low Thermal Conductivity. RSC Adv. 2019, 9, 4397–4403. [Google Scholar] [CrossRef]
- Lujan, L.; Goñi, M.L.; Martini, R.E. Cellulose–Chitosan Biodegradable Materials for Insulating Applications. ACS Sustain. Chem. Eng. 2022, 10, 12000–12008. [Google Scholar] [CrossRef]
- Amstislavski, P.; Pöhler, T.; Valtonen, A.; Wikström, L.; Ali, ·; Satu, H.·; Petri, S.·; Géza, J.·; Szilvay, R.; Amstislavski, P.; et al. Low-Density, Water-Repellent, and Thermally Insulating Cellulose-Mycelium Foams. Cellulose 2024 2024, 31, 8769–8785. [Google Scholar] [CrossRef]
- Mei, X.; Li, S.; Chen, Y.; Huang, X.; Cao, Y.; Guro, V.P.; Li, Y. Silica–Chitosan Composite Aerogels for Thermal Insulation and Adsorption. Crystals 2023, 13, 755. [Google Scholar] [CrossRef]
- Lundquist, N.A.; Tikoalu, A.D.; Worthington, M.J.H.; Shapter, R.; Tonkin, S.J.; Stojcevski, F.; Mann, M.; Gibson, C.T.; Gascooke, J.R.; Karton, A.; et al. Reactive Compression Molding Post-Inverse Vulcanization: A Method to Assemble, Recycle, and Repurpose Sulfur Polymers and Composites. Chemistry—A European Journal 2020, 26, 10035–10044. [Google Scholar] [CrossRef]
- Vitale, J.P.; Francucci, G.; Stocchi, A. Thermal Conductivity of Sandwich Panels Made with Synthetic and Vegetable Fiber Vacuum-Infused Honeycomb Cores. Journal of Sandwich Structures & Materials 2017, 19, 66–82. [Google Scholar] [CrossRef]
- Zhmakin, A.I. Heat Conduction Beyond the Fourier Law. Technical Physics 2021, 66, 1–22. [Google Scholar] [CrossRef]
- Test Method for Tensile Properties of Plastics. 2010. [CrossRef] [PubMed]
- Montes, F.; Valavala, S.; Haselbach, L. A New Test Method for Porosity Measurements of Portland Cement Pervious Concrete. 2005. [Google Scholar] [CrossRef]
- Sang, Q.; Zhang, S.; Li, Y.; Dong, M.; Bryant, S. Determination of Organic and Inorganic Hydrocarbon Saturations and Effective Porosities in Shale Using Vacuum-Imbibition Method. Int. J. Coal Geol. 2018, 200, 123–134. [Google Scholar] [CrossRef]
- Green, T.K.; Kovac, J.; Larsen, J.W. A Rapid and Convenient Method for Measuring the Swelling of Coals by Solvents. Fuel 1984, 63, 935–938. [Google Scholar] [CrossRef]



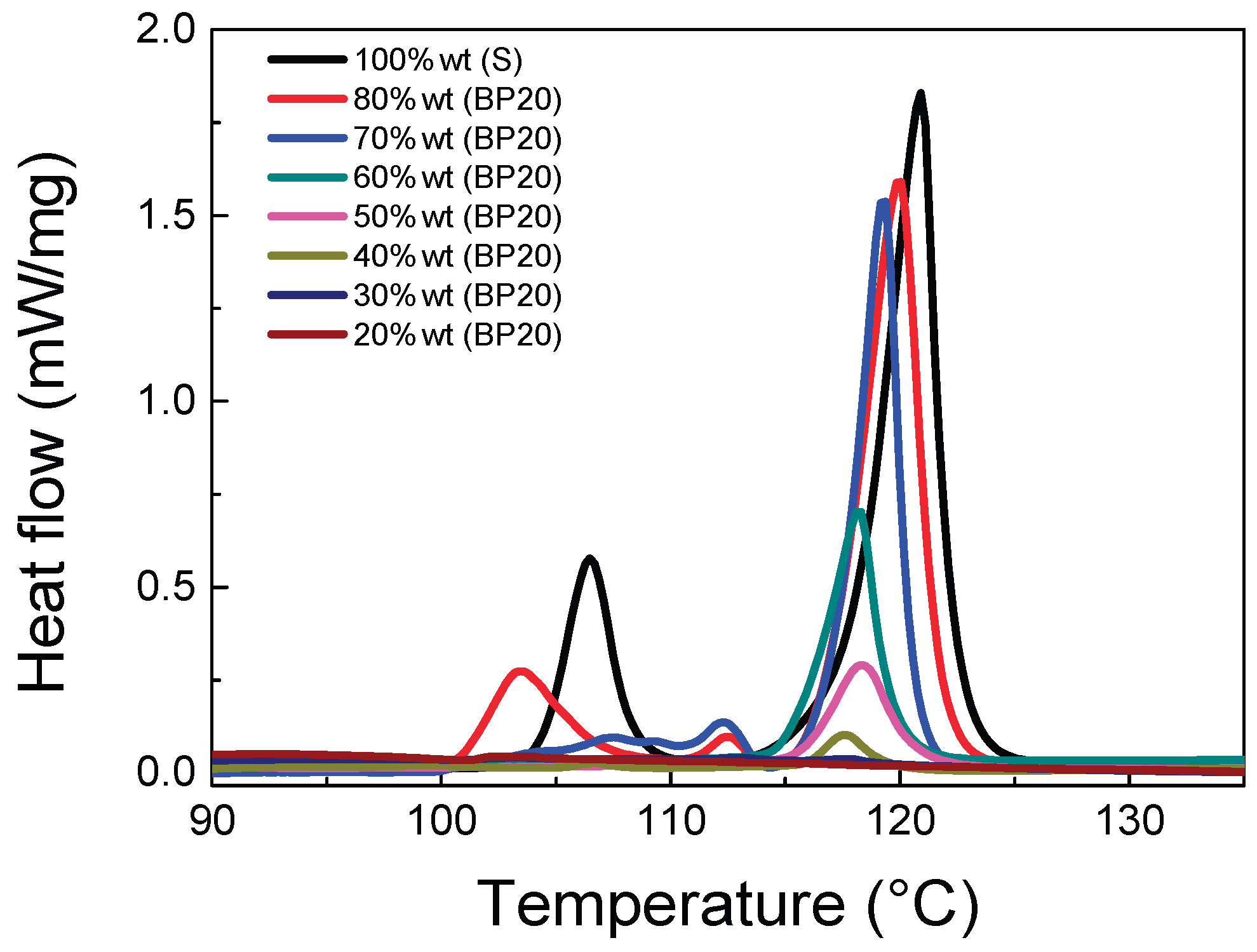


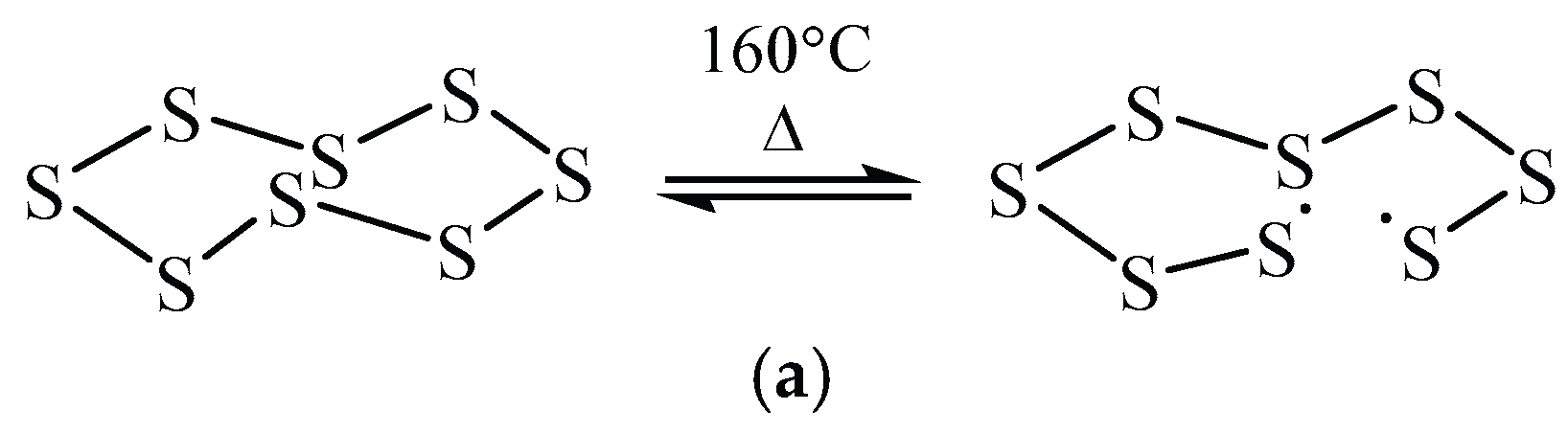
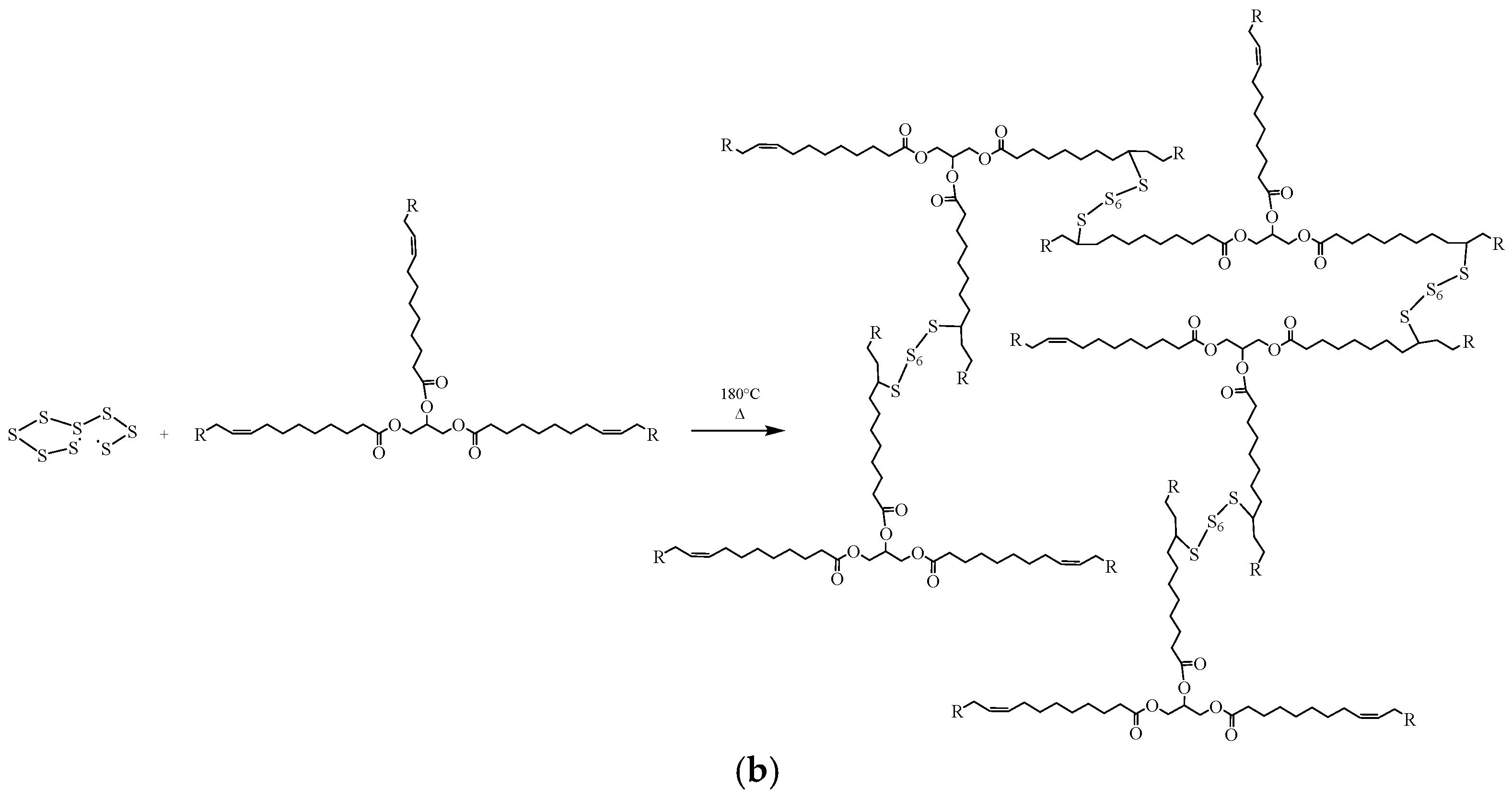
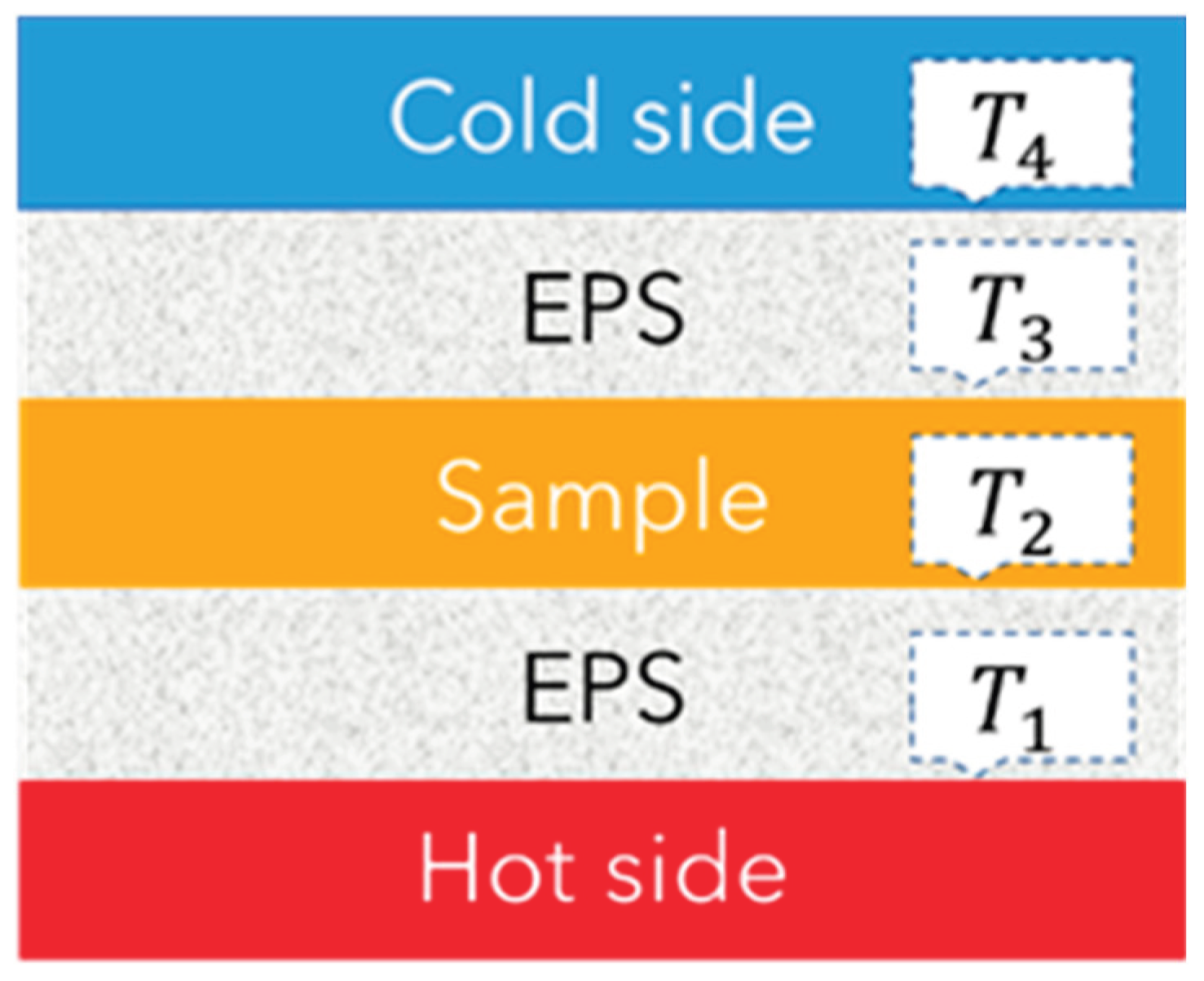
| F-BP | DSC area (J/g) | Free S (%) |
|---|---|---|
| BP20 | 0.12 | 0.21 |
| BP30 | 0.20 | 0.35 |
| BP40 | 1.37 | 2.46 |
| BP50 | 5.74 | 10.31 |
| BP60 | 11.13 | 20.02 |
| BP70 | 27.00 | 48.56 |
| BP80 | 36.92 | 66.40 |
| Pristine Sulfur | 55.60 | 100.00 |
| F-BP | λt(W/m K) | F-BP-C | λt(W/m K) |
|---|---|---|---|
| F-BP70 | 0.0404 ± 0.0013 | F-BP60-C15 | 0.0463 ± 0.0017 |
| F-BP60 | 0.0512 ± 0.0017 | F-BP60-C25-HC | 0.0393 ± 0.0012 |
| F-BP50 | 0.0525 ± 0.0016 | F-BP60-C25-CC | 0.0333 ± 0.0010 |
| F-BP40 | 0.0529 ± 0.0018 | F-BP40-C15 | 0.0464 ± 0.0018 |
| F-BP30 | 0.0610 ± 0.0021 | F-BP40-C25 | 0.0473 ± 0.0019 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




