2. Literature Review
Sulfide-alkaline wastes (SAW) represent one of the most challenging categories of industrial effluents to treat, generated at facilities within the petroleum refining and petrochemical industries. Their sources lie in technological processes such as the pyrolysis of hydrocarbon feedstocks, catalytic cracking, hydrotreating, and alkylation—processes in which alkaline scrubbing is employed to remove sulfur compounds from gaseous and liquid hydrocarbon fractions [
1,
2]. As a result of the reaction between alkaline reagents—typically sodium hydroxide—and hydrogen sulfide, mercaptans, and other organosulfur compounds, water-soluble sulfides, hydrosulfides, and mercaptides are formed; these substances pass into the aqueous phase, thereby defining the specific composition of the SAW (
Table 1).
The challenge of treating such wastewater has remained a pressing issue for many years, driven both by the consistent volumes generated at typical oil refineries and by increasingly stringent environmental regulations regarding the discharge of industrial effluents [
2]. The composition of sulfide-alkaline wastewaters is highly variable, depending on the type of feedstock being processed, the specific technological processes employed, and equipment operating parameters. A detailed analysis of the component makeup of these effluents reveals the presence of a wide spectrum of reduced sulfur compounds—including sulfides, hydrosulfides, polysulfides, thiosulfates, and sulfites—as well as organic sulfur-containing compounds such as mercaptans, organic sulfides, and disulfides [
1]. Additional constituents include alkaline agents, carbonates, and bicarbonates formed through the absorption of carbon dioxide from the air, as well as a hydrocarbon phase comprising dissolved and emulsified petroleum products, naphthenic acids, phenols, and other organic compounds [
1,
2]. The high Chemical Oxygen Demand (COD) values—reaching tens of thousands of milligrams per liter—and extreme alkalinity levels (pH 12–14) render SAW toxic to aquatic organisms and to the microorganisms within the activated sludge systems of biological wastewater treatment plants. The discharge of even minute quantities of untreated or inadequately treated SAW into water bodies triggers a sharp rise in both COD and Biochemical Oxygen Demand, inhibits natural self-purification processes, and results in the mortality of aquatic organisms due to the acute toxic effects of sulfides and the oxygen depletion caused by their oxidation [
2]. Sulfide ions possess the ability to bind with the metal ions situated within the active sites of respiratory chain enzymes, a mechanism that accounts for their high toxicity to aerobic organisms.
Traditional approaches to the detoxification of sulfide-alkaline effluents can be classified into reagent-based, thermal, and physicochemical methods. Among the reagent-based methods, the most widely adopted are neutralization with acids, oxidation using atmospheric oxygen (both at atmospheric pressure and in autoclaves), ozonation, chlorination, and peroxide oxidation [
2,
3].
Neutralization of sulfide-alkaline effluents with mineral acids—typically sulfuric or hydrochloric acid—results in the conversion of sulfides and sulfide ions into molecular hydrogen sulfide, which subsequently evolves into the gas phase. This method is characterized by the simplicity of its equipment design and relatively low capital costs; however, it suffers from a number of serious drawbacks, the most significant of which is the generation of highly toxic hydrogen sulfide, which requires capture and subsequent recovery or detoxification [
2]. Furthermore, the neutralization process is accompanied by uncontrolled foaming of the solution—caused by the evolution of hydrogen sulfide and carbon dioxide (derived from carbonates)—which creates operational difficulties. The neutral salts formed during neutralization increase the total dissolved solids content of the treated water, thereby complicating or precluding its reuse within the industrial process cycle. Consequently, this necessitates either an additional desalination stage or discharge into natural water bodies—an action associated with the salinization of those aquatic environments. Depending on the specific acid employed and the process conditions, the composition of the resulting salts may vary: the use of sulfuric acid yields sodium sulfate, while the use of hydrochloric acid yields sodium chloride. Sodium sulfates can be recovered and utilized in various industrial applications; however, their recovery entails additional energy consumption. Sodium chlorides are in lower demand as marketable products, and their discharge leads to the salinization of water bodies—a consequence that is particularly critical in regions with limited water resources. The hydrogen sulfide released during neutralization requires mandatory capture and processing—for instance, into elemental sulfur via the Claus process—which significantly complicates the process scheme and increases its cost. Furthermore, hydrogen sulfide exhibits high corrosivity, necessitating the use of specialized corrosion-resistant materials for equipment and pipelines.
The oxidation of alkaline sulfide-containing wastewaters using atmospheric oxygen is implemented in two primary variants: atmospheric oxidation at normal pressure and autoclave oxidation at elevated temperatures and pressures. Atmospheric oxidation is characterized by a low process rate—particularly at high initial pollutant concentrations—and fails to ensure the required degree of purification, as primarily sulfides undergo oxidation, while more stable organic sulfur-containing compounds remain in solution [
2]. The mechanism of atmospheric sulfide oxidation by atmospheric oxygen in an alkaline medium is quite complex and involves stages of intermediate product formation—specifically, polysulfides and thiosulfates. The process rate is limited by the mass transfer step of oxygen from the gaseous phase into the liquid phase and depends on mixing intensity, temperature, and the presence of catalysts. To intensify the process, various catalytic systems—such as salts of variable-valence metals—are employed; however, their use leads to additional contamination of the wastewater and necessitates the subsequent removal of the catalysts. Autoclave oxidation, conducted at temperatures of 150–200 °C and pressures of 1.5–2.5 MPa, allows for process intensification and the achievement of a higher degree of oxidation; however, it entails significant energy costs for heating and maintaining high pressure, requires the use of specialized corrosion-resistant equipment, and presents the challenge of managing salt and elemental sulfur deposits on the internal surfaces of the reaction vessels [
2]. During autoclave oxidation, it is possible to achieve virtually complete oxidation of sulfides to sulfates; however, this precludes the opportunity to recover valuable elemental sulfur. Furthermore, high temperatures and pressures accelerate corrosion processes, thereby necessitating the use of expensive materials, such as titanium or high-alloy steels. The salt and sulfur deposits that form on equipment walls during autoclave oxidation necessitate periodic equipment shutdowns for cleaning, thereby reducing productivity and increasing operating costs.
Ozonation is regarded as an effective method for the oxidation of sulfides and organic compounds present in alkaline sulfide-bearing wastewaters. Experimental studies demonstrate a high degree of pollutant degradation during ozone treatment; however, the method entails significant energy costs for ozone generation, requires the use of complex equipment, and necessitates addressing the issue of residual ozone disposal in off-gases [
3]. Ozone is a powerful oxidant with a high redox potential of 2.07 V, enabling it to oxidize the majority of organic and inorganic pollutants (
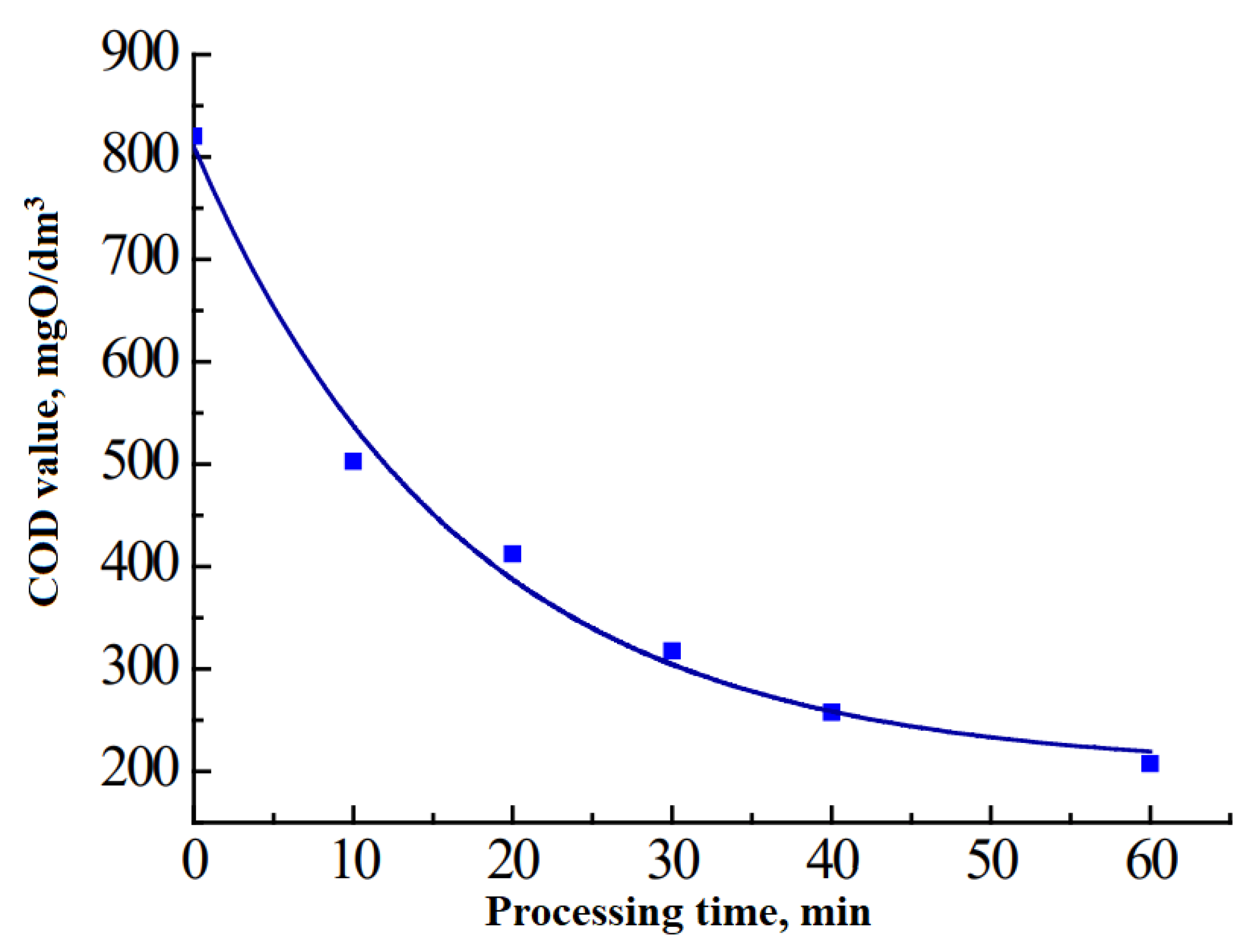
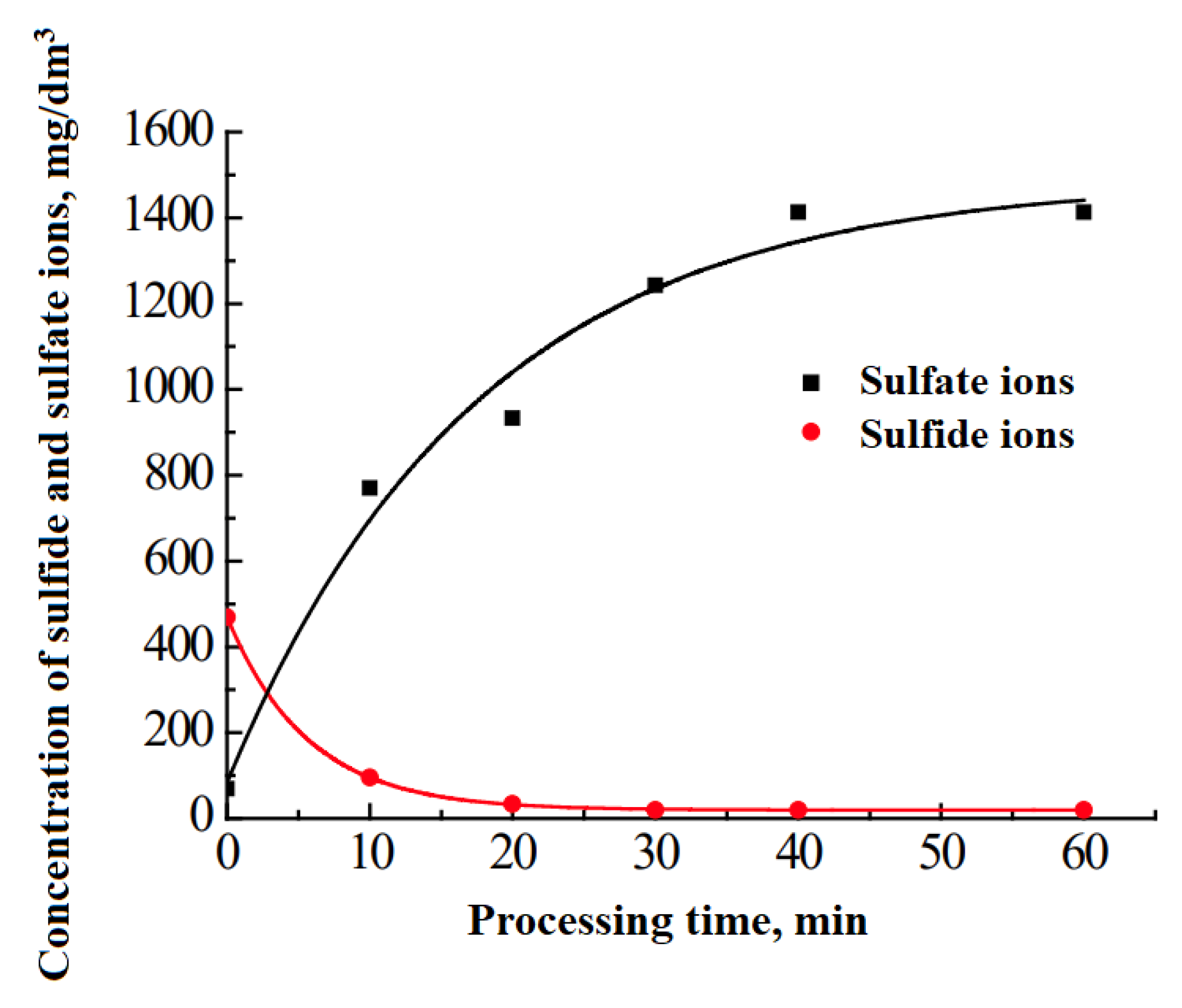
Figure 1 and
Figure 2).
In an alkaline medium, ozone decomposes to form hydroxyl radicals, which possess an even higher oxidation potential of 2.80 V; this further intensifies the oxidation process. However, ozone exhibits selectivity in its reactions with various contaminants; consequently, achieving complete purification may require significant treatment time and a high ozone dosage. Furthermore, ozonation often necessitates preliminary wastewater treatment—specifically, the removal of suspended solids and petroleum products—as these substances reduce the efficiency of ozone mass transfer into the liquid phase. The presence of suspended solids and emulsified petroleum products leads to the shielding of the phase interface and a reduction in the ozone mass transfer coefficient. To ensure effective ozonation, it is essential to achieve proper dispersion of the ozone-air mixture within the liquid being treated; this is accomplished through the use of specialized dispersers—such as porous ceramic or cermet elements, or various types of spargers. However, when contaminant concentrations are high, the pores of these dispersers rapidly become clogged with oxidation byproducts, thereby necessitating their periodic cleaning or replacement. The gaseous emissions generated during ozonation contain residual ozone, which is toxic and must be decomposed prior to discharge into the atmosphere. Thermal, catalytic, or adsorption methods are employed to decompose this ozone, a requirement that further complicates the process flowsheet and increases its overall cost.
Chlorination and peroxide oxidation are also employed for the treatment of sulfide-alkaline wastewaters; however, the use of chlorine carries the risk of forming toxic organochlorine compounds, while hydrogen peroxide—although an environmentally safe oxidant—requires the use of catalysts to activate the process and is characterized by a relatively high reagent cost [
2]. Chlorine and its compounds (sodium hypochlorite, chlorinated lime) have traditionally been used for the oxidation of sulfides and the disinfection of wastewaters. Chlorine oxidizes sulfides to sulfates or elemental sulfur, depending on the process conditions and reagent ratios. However, in the presence of organic compounds—particularly aromatic hydrocarbons and phenols—organochlorine compounds may form, many of which exhibit high toxicity, carcinogenicity, and mutagenicity. The formation of organochlorine compounds during wastewater chlorination poses a serious environmental problem and necessitates additional treatment steps for their removal. Hydrogen peroxide is an environmentally safe oxidant, as its decomposition products are water and oxygen. However, the rate at which hydrogen peroxide oxidizes pollutants is low in the absence of catalysts, particularly at low temperatures. To activate hydrogen peroxide, catalysts are employed, including variable-valence metal ions, solid catalysts based on metal oxides, and UV irradiation. Fenton’s reagent—a mixture of hydrogen peroxide and iron(II) salts—enables the effective oxidation of a wide spectrum of organic pollutants through the generation of hydroxyl radicals. However, the use of iron salts results in the formation of iron-containing precipitates that require disposal. Furthermore, for the Fenton reaction to proceed effectively, a specific pH range—typically 2–4—must be maintained; for highly alkaline sulfide-alkaline wastewaters, this necessitates preliminary neutralization followed by subsequent alkalization prior to discharge or reuse.
Thermal methods for the detoxification of spent alkaline waste (SAW)—which involve concentration via evaporation followed by the incineration of the organic concentrate, or the direct incineration of the wastewater in specialized furnaces—enable the achievement of virtually complete destruction of organic pollutants and the recovery of mineral salts. However, the high energy intensity of evaporation and incineration processes—driven by the necessity to vaporize large volumes of water—as well as issues regarding equipment corrosion and the removal of sulfur compounds from flue gases, limit the practical application of thermal methods [
2]. The evaporation of SAW can be carried out in various types of evaporators, including thin-film, rotary, and forced-circulation units. To minimize energy consumption, multi-stage evaporation systems utilizing secondary steam, as well as heat pumps, are employed. Nevertheless, the highly corrosive nature of concentrated alkaline solutions at elevated temperatures necessitates the use of expensive corrosion-resistant materials—such as nickel, titanium, and specialized steels. The concentrate generated during evaporation—which contains sulfides, organic compounds, and mineral salts—is subsequently directed for incineration. During incineration, organic compounds undergo oxidation to form carbon dioxide and water, while sulfur-containing compounds are oxidized into sulfur oxides. These sulfur oxides must be captured from the flue gases—for instance, through absorption using milk of lime, resulting in the formation of calcium sulfite and calcium sulfate. The mineral salts generated during incineration—specifically sodium sulfates and carbonates—can be recovered and utilized; however, they frequently contain impurities and therefore require further purification. The direct incineration of SAW is performed in specialized furnaces, often utilizing nozzles to atomize the liquid stream within the combustion flame. This process entails rapid heating and vaporization of the water, followed by the combustion of the organic compounds. To ensure complete combustion and prevent the formation of toxic products of incomplete combustion, it is necessary to maintain a high temperature—900–1200 °C—and ensure a sufficient residence time for the gases within the high-temperature zone. This entails significant fuel consumption and the generation of large volumes of flue gases that require treatment.
Physicochemical methods—such as coagulation, flocculation, sorption, ion exchange, and membrane separation—can be employed either independently or in combination with other techniques as part of integrated process schemes. The efficacy of individual physicochemical methods—including sorption—for treating multicomponent industrial wastewaters is limited; this has spurred the development of hybrid technologies that integrate various processes [
4]. Sorption-based methods, which utilize natural and synthetic sorbents, are effective for the removal of petroleum products and certain organic compounds; however, they are insufficiently effective against low-molecular-weight polar compounds and sulfides. Recently, studies have emerged focusing on the development of modified sorbents derived from power-generation waste for the removal of phenols and other organic pollutants from wastewater, thereby demonstrating the advancement of sorption technologies for the treatment of complex effluents [
8]. Research indicates that natural sorbents, such as bentonite, can be optimized for the removal of pollutants from petrochemical wastewaters, with treatment efficiency being contingent upon process conditions and sorbent modification [
9]. Common sorbents include activated carbons, zeolites, and clay minerals, as well as industrial byproducts such as fly ash, slag, and sawdust. Surface modification of sorbents enables the enhancement of their selectivity and sorption capacity toward specific classes of pollutants. For instance, modifying carbon sorbents with metal compounds enhances their efficiency in extracting sulfur-containing compounds; similarly, the use of composite materials based on manganese and cerium yields promising results in treating wastewater to remove specific organic pollutants—such as tetramethylammonium—found in effluents from optoelectronic manufacturing facilities, a technique that may also prove beneficial for petrochemical wastewater [
10]. However, sorption-based methods inherently result in the generation of spent sorbents saturated with pollutants, which subsequently require either regeneration or disposal. Sorbent regeneration often entails additional costs and leads to the generation of secondary waste products, specifically spent regeneration solutions. Landfilling spent sorbents fails to resolve the fundamental issue of permanently neutralizing pollutants and poses a long-term environmental hazard. In this context, biosorption is regarded as a promising technology that utilizes non-living biomass to extract pollutants; nevertheless, the efficacy of biosorbents in removing ammonia and other specific compounds warrants further investigation [
11].
Ion exchange enables the selective extraction of heavy metal ions and certain anions; however, it necessitates the thorough pretreatment of wastewater to remove organic contaminants that poison the ion-exchange resins [
4]. Organic compounds—particularly high-molecular-weight substances and surfactants—adsorb onto the surface of ion-exchange resins, thereby blocking active sites and reducing the resins’ exchange capacity. The regeneration of poisoned resins is complex and not always effective; consequently, protecting the resins requires the preliminary removal of organic pollutants using alternative methods. Furthermore, ion-exchange resins are sensitive to the high pH levels characteristic of sulfide-alkaline wastewater (SAW), which can lead to their degradation. Membrane-based methods—including microfiltration, ultrafiltration, nanofiltration, and reverse osmosis—are capable of achieving a high degree of purification; however, their application for treating SAW is limited due to the high osmotic activity of the wastewater, the membranes’ susceptibility to fouling by organic substances and sulfides, and the challenges associated with the disposal of the resulting concentrates. The high ionic strength of these solutions generates significant osmotic pressure, necessitating the application of high operating pressures to drive water through the membrane, which, in turn, increases energy consumption. Organic substances—particularly petroleum products—and sulfides form deposits on the membrane surfaces; these deposits drastically reduce throughput and necessitate frequent chemical cleaning of the membranes. The concentrates generated during membrane separation contain all the pollutants in a concentrated form and require further processing or disposal—a significant challenge.
The problem of treating industrial wastewater to remove various contaminants—including sulfides, organic compounds, phenols, phosphates, and other constituents—is a subject of active global research. The application of biological treatment methods, particularly the activated sludge process, is widespread at the wastewater treatment facilities of petrochemical enterprises; however, life cycle assessments and carbon footprint analyses of such systems indicate a need for their improvement and integration with other technologies [
12]. Combined natural wastewater treatment systems have demonstrated effectiveness in removing organic matter and phosphorus from contaminated watercourses, a capability that holds promise for the development of multi-stage treatment schemes [
13]. Research into the use of ochre—a by-product of water treatment processes—for controlling phosphates and sulfides in anaerobic systems highlights the potential for utilizing waste materials to address environmental challenges [
14]. The application of mineral coagulants for treating dye-contaminated wastewater has demonstrated the efficacy of such methods; however, the complex, multi-component effluents characteristic of the petrochemical industry necessitate the development of specialized reagents and treatment protocols [
15]. Chemical and biological treatment processes applied to wastewater generated during the manufacture of specific chemical compounds—such as N,N’-dinitrosopentamethylenetetramine—underscore the necessity of combining diverse approaches to achieve regulatory compliance [
16].
In recent decades, electrochemical methods for wastewater treatment have attracted increasing attention from researchers, as they enable the oxidation of pollutants directly at the electrodes—either without the introduction of additional chemical reagents or with their minimal consumption. Electrochemical methods offer a number of advantages, including the ability to conduct the process at ambient temperatures and pressures, high selectivity, ease of automation, and the capacity to control the process by adjusting electrical parameters [
6,
7].
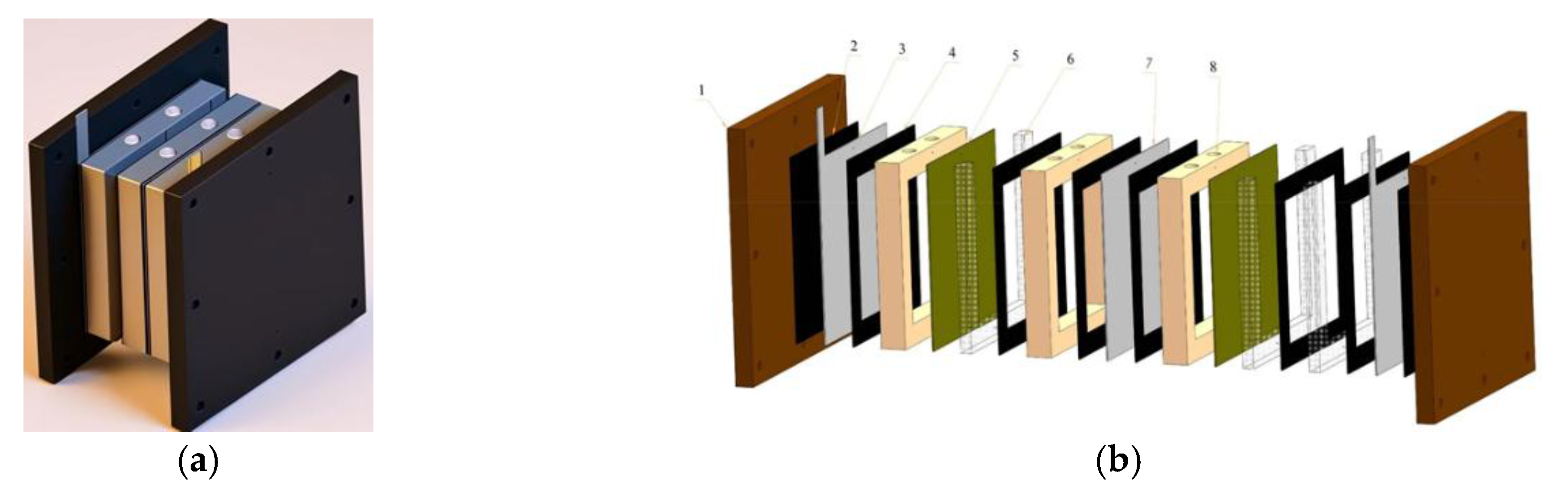
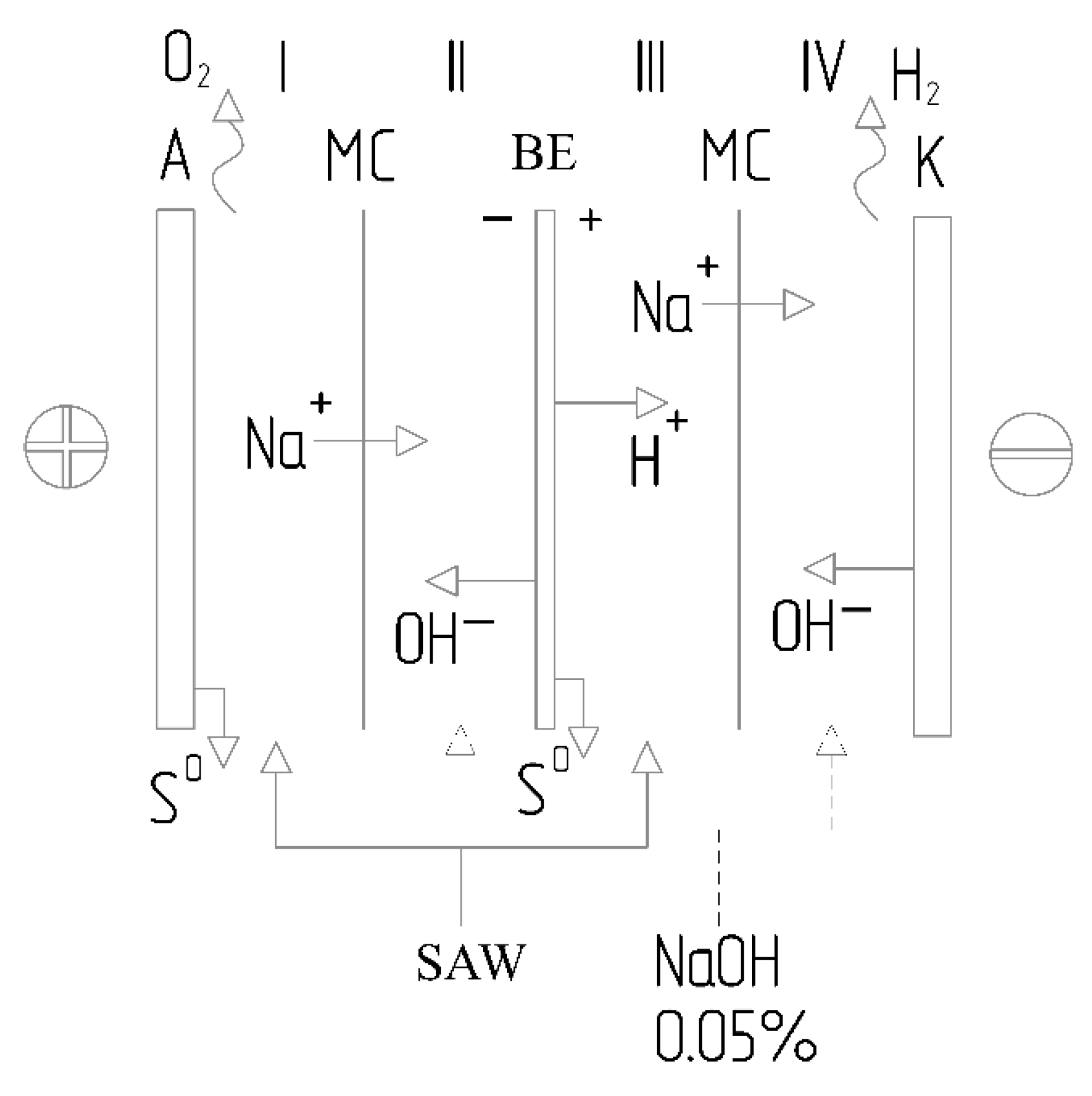
Figure 3 illustrates a system of electromembrane units designed for drinking water purification. It is worth noting that these units are compact, automated, suitable for industrial application, feature an attractive design, and can be supplied in a containerized configuration.
Among electrochemical methods, anodic oxidation is of particular interest—a process in which active oxygen species (such as hydroxyl radicals and atomic oxygen) are generated on the anode surface, or in which direct electron transfer occurs from the substance undergoing reduction to the electrode, leading to the destruction of organic and inorganic pollutants [
6,
7]. The electrochemical degradation of persistent organic pollutants—such as poly- and perfluoroalkyl substances—present in industrial wastewater has demonstrated high efficiency and can be implemented on an industrial scale under appropriate electrolysis conditions [
17]. Bioelectrochemical methods, which combine biological and electrochemical treatment, enable not only wastewater purification but also the recovery of metals, accompanied by the simultaneous generation of electricity; this opens up new prospects for the development of energy-efficient waste treatment technologies [
18].
Depending on the anode material and electrolysis conditions, the mechanism of anodic oxidation may vary. Anodes made of inert materials—such as platinum or metal oxides—facilitate the generation of hydroxyl radicals via water discharge; these radicals act as potent, non-selective oxidants capable of oxidizing virtually any organic compound into carbon dioxide and water. Conversely, anodes made of active materials—such as nickel, cobalt, or their oxides—can participate directly in the oxidation process through the formation of higher-valence oxides or adsorbed oxygen-containing species, thereby enabling more selective oxidation. A significant advantage of electrochemical methods is the ability to control the process by adjusting parameters such as current density, electrode potential, and electrolyte composition, as well as the potential to integrate wastewater treatment with the recovery of valuable products—for instance, the generation of alkali and hydrogen at the cathode, or the extraction of elemental sulfur from sulfide-containing solutions [
7].
The fundamental feasibility of electrochemically oxidizing sulfide ions in an alkaline medium to form elemental sulfur has been demonstrated in a number of studies. In an alkaline environment, sulfide ions can be oxidized at the anode to various products, depending on the electrode potential and the process conditions. At low anodic potentials, elemental sulfur is predominantly formed, whereas at higher potentials, further oxidation of the sulfur to thiosulfate, sulfite, and sulfate ions is possible. The recovery of elemental sulfur in the solid phase is of significant interest, as it allows not only for the removal of a toxic pollutant from wastewater but also for the production of a valuable commercial product used in various industrial sectors—such as sulfuric acid manufacturing, rubber vulcanization, fertilizer production, and others [
2]. The sulfur generated during electrochemical oxidation can be separated from the solution using simple mechanical methods: settling, filtration, or centrifugation. The purity of the recovered sulfur depends on the electrolysis conditions and the composition of the influent wastewater; however, in many cases, it is sufficiently high for industrial application. Simultaneously, during the electrolysis of aqueous solutions, water reduction occurs at the cathode, resulting in the evolution of gaseous hydrogen and the formation of hydroxide ions. This enables the production of an alkaline solution and hydrogen—valuable products suitable for reuse within the technological cycle [
7]. High-purity hydrogen can be utilized as a fuel, for hydrogenation processes in petroleum refining, or in chemical manufacturing. The alkaline solution can be recycled back into the hydrocarbon alkaline washing process, thereby reducing the need to purchase fresh alkali and minimizing the volume of waste requiring disposal. However, the practical implementation of the electrochemical purification of spent alkaline solutions—coupled with the simultaneous recovery of valuable components—requires the optimization of the electrolyzer design to ensure the separation of the anodic and cathodic compartments, thereby preventing product recombination and secondary contamination [
2]. The use of ion-exchange membranes to separate the electrode compartments resolves the issue of product mixing and creates conditions conducive to the selective isolation of target components. In recent years, the advancement of electrochemical technologies has focused on the development of energy-efficient electrolyzers equipped with membranes that enable the separation of anodic and cathodic products and enhance the purity of the resulting substances [
5,
19].
The fundamental operating principles of proton exchange membrane water electrolyzers (PEMWE) are detailed in a comprehensive review that examines materials for membranes and electrodes, the kinetics of electrode reactions, as well as key challenges and prospects for the commercialization of this technology for hydrogen production [
5]. PEMWE systems utilize proton exchange membranes that conduct protons and ensure the high purity of the generated gases. However, the operation of PEMWE requires the use of noble metals—specifically platinum and iridium—as catalysts, which increases equipment costs. Alkaline water electrolysis technology utilizing anion exchange membranes (AEMWE) is regarded as a more economical alternative, as it permits the use of electrocatalysts based on non-noble metals—such as nickel, cobalt, iron, and their alloys—thereby allowing for a substantial reduction in both equipment and operating costs [
6,
7]. AEMWE systems employ anion exchange membranes that conduct hydroxide ions, enabling the process to be conducted in an alkaline environment where non-noble metals can function effectively.
Research studies on alkaline water electrolysis with anion exchange membranes involve the experimental evaluation of various electrode types, including those based on nickel and its alloys [
6]. The results demonstrate the feasibility of achieving high current densities and efficiencies in an alkaline medium, thereby opening up prospects for the industrial application of this technology—not only for hydrogen production but also for addressing environmental challenges. A comprehensive overview of the current state of alkaline water electrolysis with anion exchange membranes is provided by review articles that summarize the latest advancements in the development of membranes, catalysts, and electrodes [
7,
20]. Particular attention is focused on the development of membranes exhibiting high ionic conductivity and chemical stability in alkaline environments, as well as on the creation of catalysts characterized by high activity and stability. It has been demonstrated that catalysts based on nickel and its alloys with iron, cobalt, and molybdenum can achieve activity comparable to that of noble-metal catalysts, at a significantly lower cost.
Research results regarding the catalytic activity of electrode materials are of significant interest for the development of processes for the anodic oxidation of sulfur-containing compounds. Studies investigating nickel and nickel-iron catalysts for alkaline electrolysis have demonstrated their high activity in oxygen evolution reactions, indicating the fundamental feasibility of utilizing these materials for the anodic oxidation of various compounds in alkaline media [
21]. Experimental studies indicate that the Ni
90Fe
10 catalyst is the most active among non-noble metal catalysts; furthermore, in a practical electrolytic cell utilizing this catalyst, operating voltages are achieved that are comparable to those obtained with a benchmark iridium catalyst [
21]. This opens up promising avenues for the development of cost-effective and efficient anodes for the electrochemical treatment of sulfur-containing wastewater. This line of research has been further advanced in studies presenting improved nickel gas-diffusion anodes, which demonstrate high stability under alkaline electrolysis conditions [
22]. Gas-diffusion electrodes facilitate the efficient supply of reactants and removal of reaction products, thereby enabling the achievement of high current densities at low overpotentials. Contemporary publications propose fundamentally new approaches to catalyst design for operation in media with reduced alkalinity—a particularly relevant consideration for the treatment of actual wastewater streams, where the chemical composition may vary and alkalinity levels may decrease during the treatment process [
23]. For instance, the creation of catalysts with hierarchical structures, the deposition of active components onto high-surface-area substrates, and doping with various elements serve to enhance catalyst activity and stability across a wide pH range.
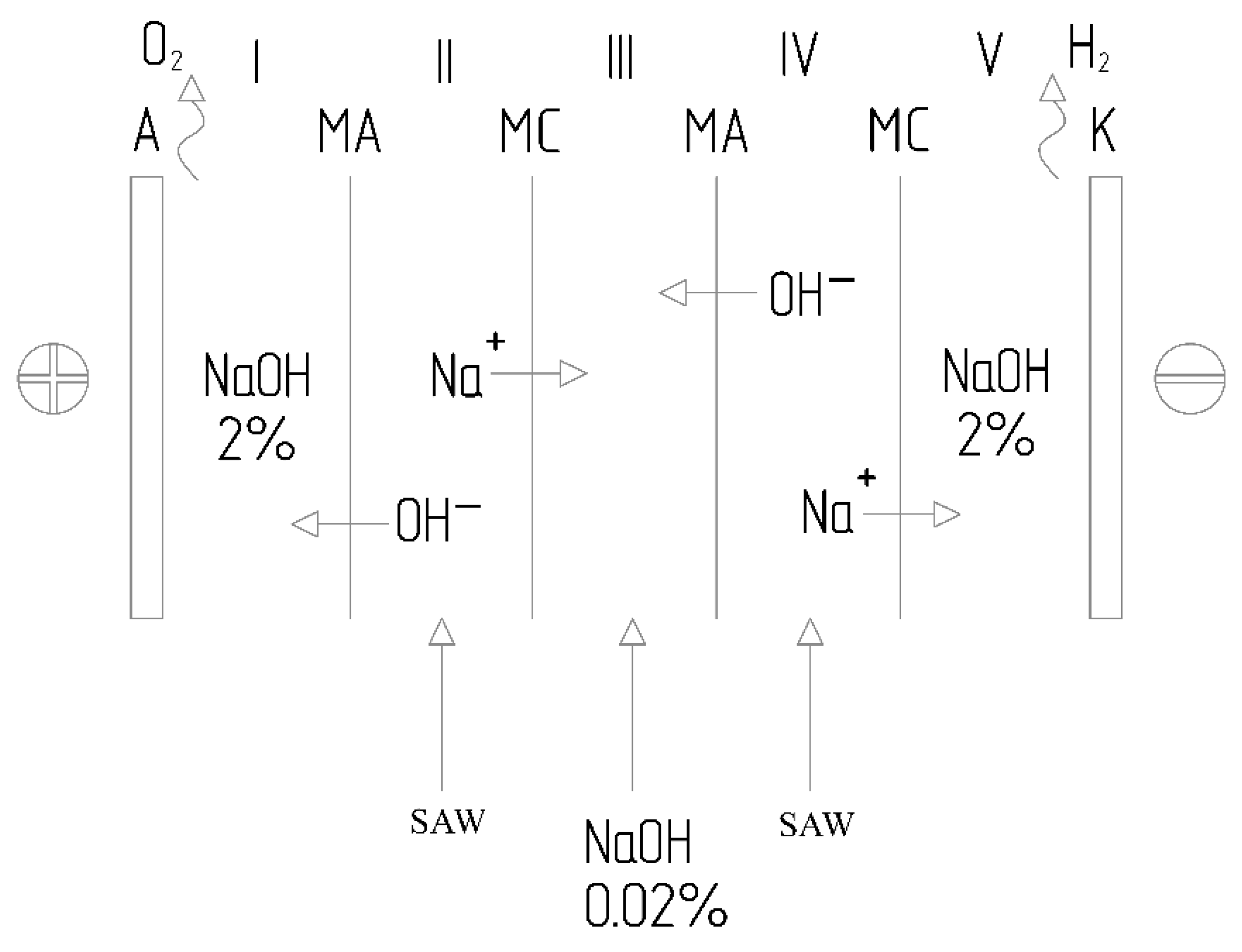
Issues regarding the design of electrochemical devices for processing waste from the industrial-energy complex—including the principles governing directed mass transfer and the selection of membrane materials—have been examined in detail in specialized literature [
24]. The expediency of employing multicompartment bipolar cells for separating liquid media and recovering valuable components has been substantiated, as this approach allows for increased process efficiency and reduced energy consumption [
24]. Multicompartment bipolar electrolyzers consist of several cells connected in series, wherein bipolar electrodes function as the anode for one cell and the cathode for the adjacent one. This configuration makes it possible to achieve a higher overall voltage across the entire electrolyzer—and, consequently, higher productivity—while operating at the same power supply voltage. The use of ion-exchange membranes positioned between the electrodes ensures the separation of process streams and prevents the intermingling of products from different compartments. While the literature provides detailed coverage of electrolyzer designs utilizing proton-exchange and anion-exchange membranes for hydrogen production [
5,
19], the majority of research focuses either on water purification or on electrolysis specifically for hydrogen generation; only a limited number of studies are dedicated to combined processes that simultaneously address the detoxification of toxic effluents and the recovery of valuable components [
23,
24,
25]. In such combined processes, the anode compartment is utilized for the oxidation of contaminants (such as sulfides and organic compounds), while the cathode compartment is employed for the generation of valuable products (such as hydrogen and alkali). The membranes serve to separate the respective media and prevent the back-mixing of the generated products.
Issues regarding the removal of co-contaminants—which may be present in complex industrial wastewaters alongside sulfides—are addressed in reviews dedicated to modern treatment methods. An analysis of the efficacy of technologies such as sorption, ion exchange, and membrane processes for removing various classes of pollutants highlights the importance of combined approaches, as real-world wastewaters often contain multicomponent contaminants requiring sequential or simultaneous treatment using different methods [
4]. In particular, the presence of phenols and other organic compounds—characteristic of refinery wastewaters—can significantly complicate the electrochemical treatment process, necessitating either their preliminary removal or the selection of appropriate operating parameters to ensure their oxidation [
8]. Phenols and their derivatives are highly toxic and resistant to biological oxidation. The electrochemical oxidation of phenols using anodes made of lead dioxide, tin oxides, antimony oxides, and other materials enables the effective degradation of these compounds into low-toxicity products or their complete mineralization. However, this process may result in the formation of polymeric products that precipitate onto the anode surface, thereby passivating it. Consequently, the electrochemical treatment of wastewaters containing phenols requires the careful selection of both the anode material and the electrolysis parameters.
Research in the field of rational water management within the oil industry underscores the necessity of integrating advanced treatment methods into technological cycles; this not only reduces the environmental burden but also enhances production efficiency through the recycling of treated water and the recovery of valuable components for reuse in the production process [
2]. This approach is viewed as a strategic direction for development, enabling a reduction in water consumption and a decrease in the volume of discharged waste. In the context of sulfide-alkaline wastewaters, this translates into the potential for establishing a closed-loop system involving the recycling of alkali back into the hydrocarbon alkaline washing process, as well as the production of marketable commodities—specifically, elemental sulfur and hydrogen. A closed-loop water circulation system enables the repeated reuse of water and its dissolved components, which is particularly relevant for regions facing water scarcity and stringent requirements regarding the quality of discharged water.
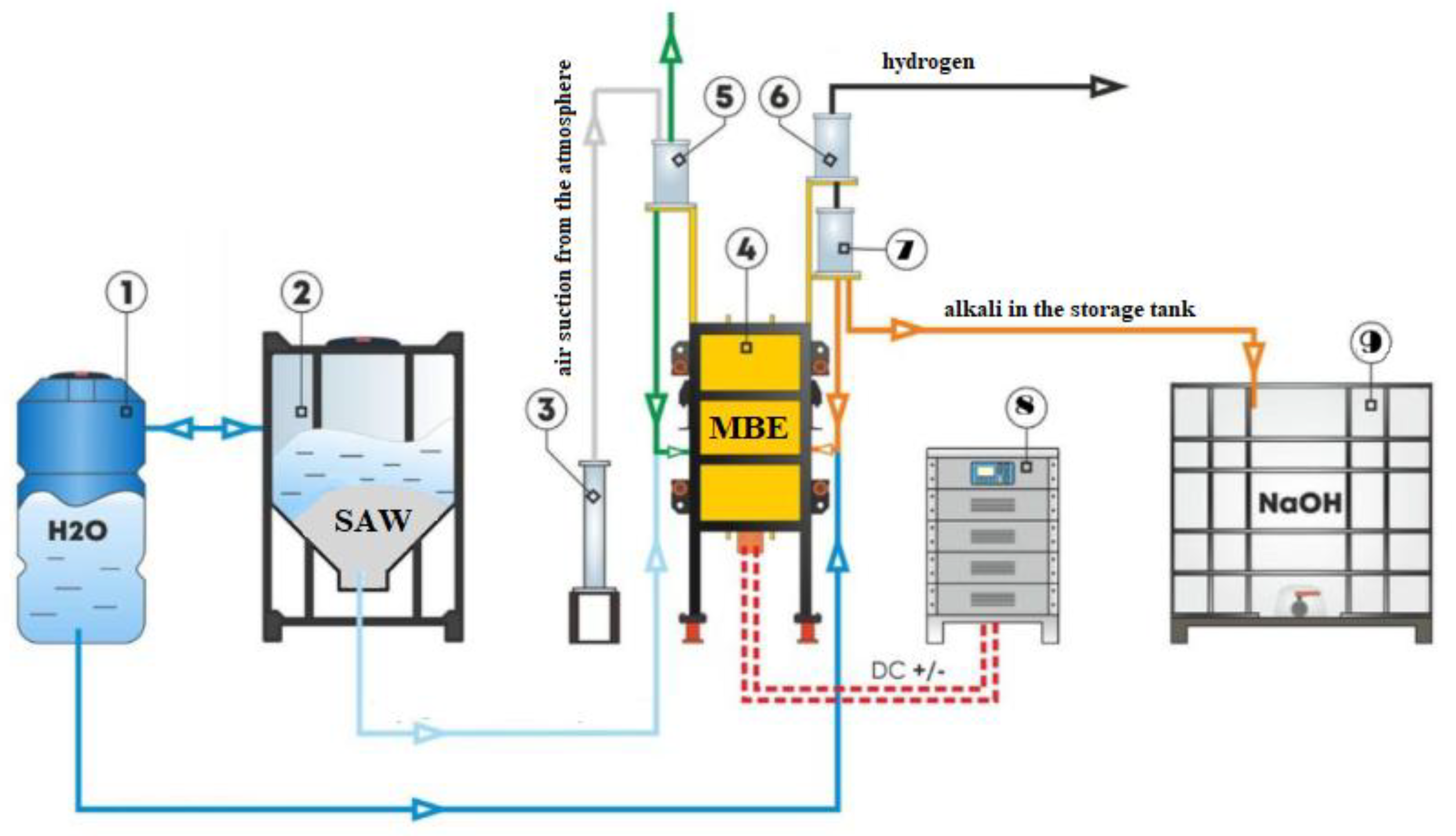
Thus, the development of an electrolyzer design incorporating ion-exchange membranes—capable of facilitating directed mass transfer and media separation to enable the simultaneous oxidation of sulfides in the anode compartment and the recovery of alkaline components in the cathode compartment—constitutes the subject of the present study. The proposed approach builds upon principles established in existing works on the design of electrochemical apparatuses, applying them to a specific target: highly concentrated sulfide-alkaline wastewaters generated by petrochemical industries. Its objective is to create an efficient and economically viable technology that not only detoxifies hazardous waste but also yields valuable products suitable for reuse within the industrial process cycle. Particular attention is devoted to the selection of optimal electrode materials—specifically nickel—and membranes (cation-exchange type), ensuring both high process efficiency and a long equipment service life. The results of this research can be applied to the development of industrial-scale systems for the localized treatment of highly concentrated sulfide-bearing wastewaters from oil refining operations, as well as for the establishment of closed-loop water recirculation systems within the industry.