Submitted:
18 March 2026
Posted:
23 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
3.1. Synthesis and Characterization of Bio-Composites and Polymer
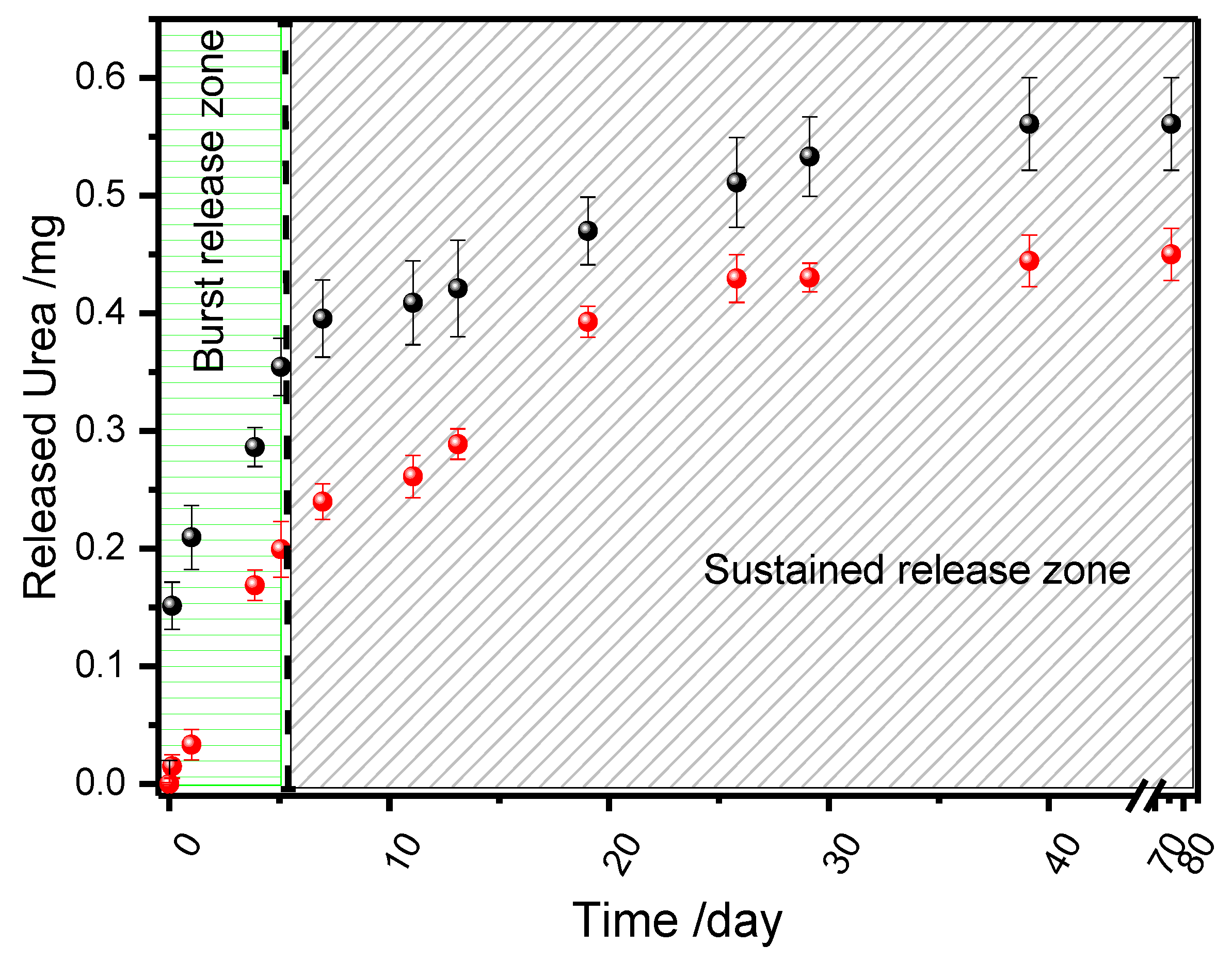
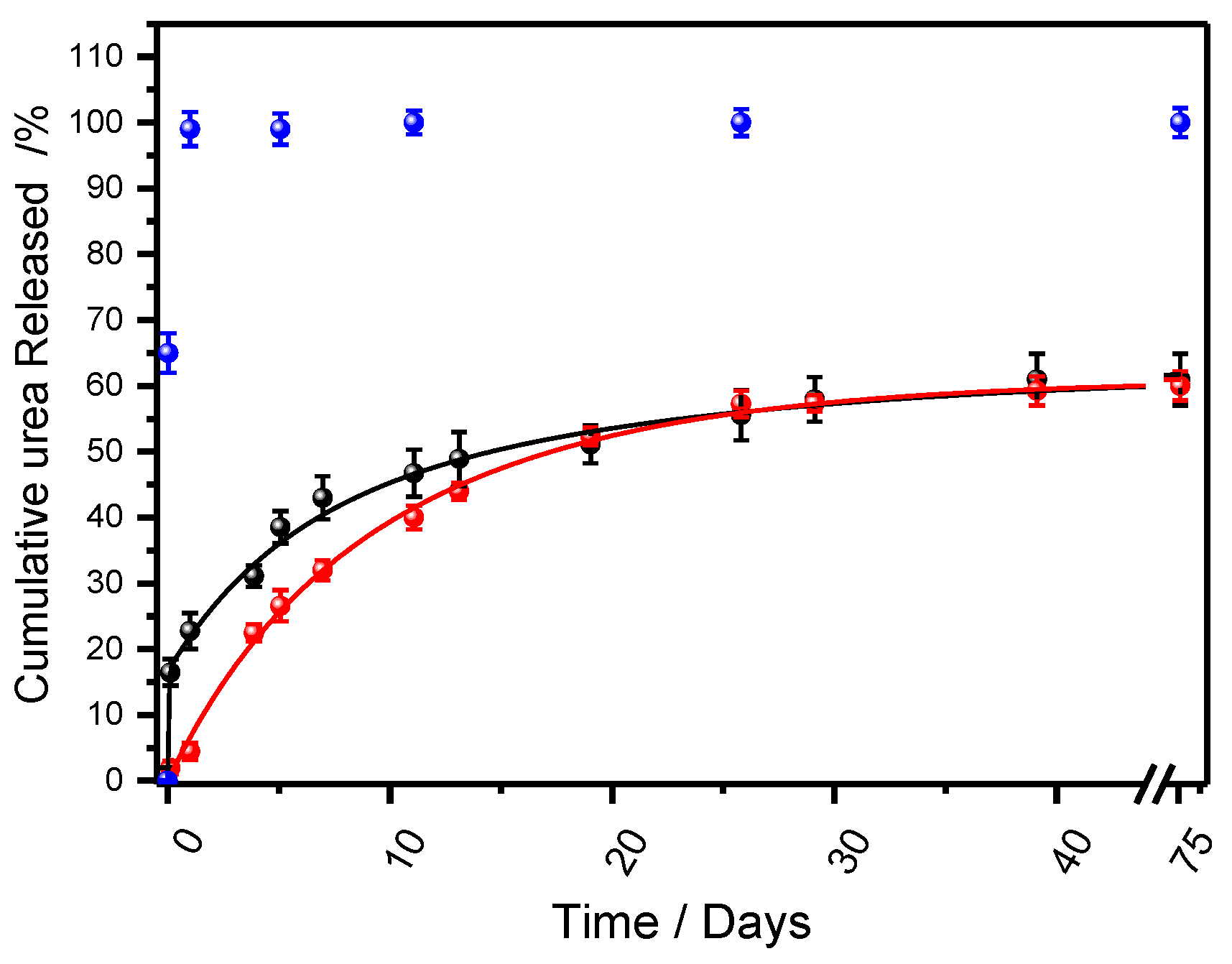
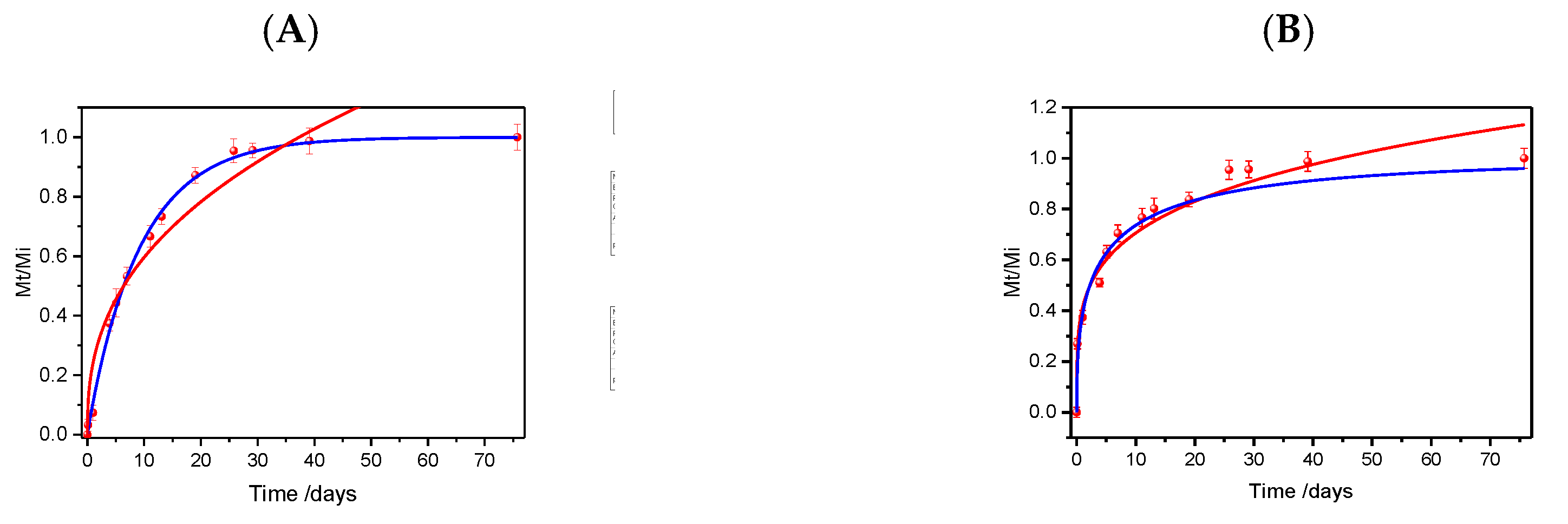
3.2. Urea Release Kinetics from Biocomposites
3.3. Soil Physical Properties
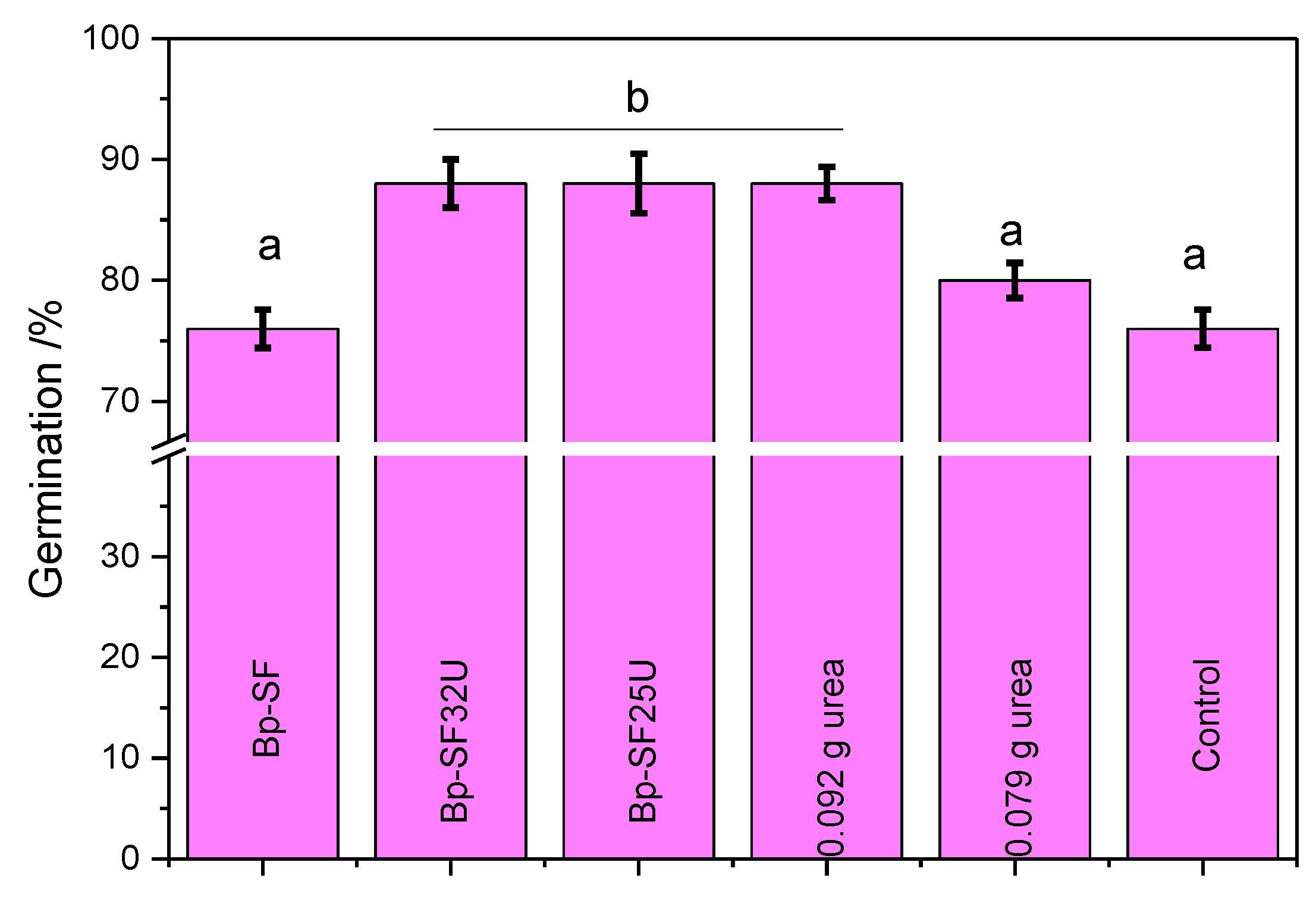
3.4. Germination of Maize Seedlings
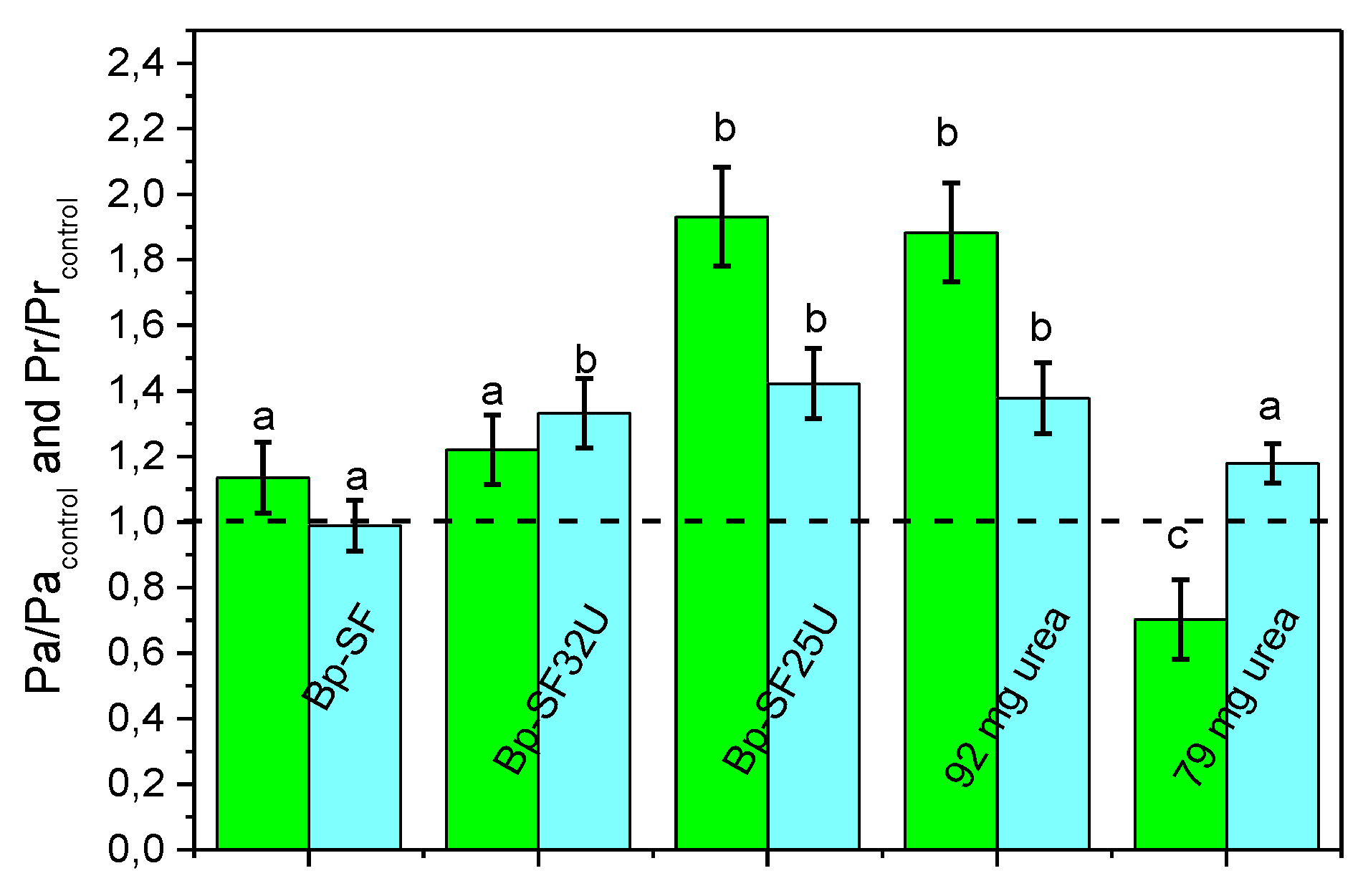
3.5. Effect of Biopolymer on Plant Growth in Pot Condition
3. Materials and Methods
3.1. Materials
3.2. Methods
3.2.1. Synthesis and Characterization Urea Loaded Bio-Composites: Bp-SF-Urea
3.2.2. Contact Angle Measurement
3.2.3. Controlled Release Analysis
3.2.4. Soil Physical Properties
3.2.5. Pot Experiment
3.2.6. Preparation of Maize Seeds
3.2.7. Seed Germination and Early Seedling Growth
3.2.8. Vegetative Growth Measurements
3.2.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CRFs | controlled-release fertilizers |
| Bp-SF | Biopolymer obtained by inverser vulcanization of sunflower oil and S |
| Bp-SF25U | Bio-composite obtained using Bp-SF and 25% of urea |
| Bp-SF32U | Bio-composite obtained using Bp-SF and 32% of urea |
| ESO | Expoxidated sunflower oil |
| SF | sunflower oil |
| Pa | Aerial biomass |
| Pr | Root biomass |
References
- Voran, M.J. Human Population Overshoot: The Disavowed Driver of Ecological Crisis. International Journal of Applied Psychoanalytic Studies 2026, 23, e70028. [Google Scholar] [CrossRef]
- Sandhu, N.; Sethi, M.; Kumar, A.; Dang, D.; Singh, J.; Chhuneja, P. Biochemical and Genetic Approaches Improving Nitrogen Use Efficiency in Cereal Crops: A Review. Front. Plant Sci. 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Ghumman, A.S.M.; et al. A Degradable Inverse Vulcanized Copolymer as a Coating Material for Urea Produced under Optimized Conditions. Polymers 2021, 13, 4040. [Google Scholar] [CrossRef] [PubMed]
- Cantarella, H.; Otto, R.; Soares, J.R.; de B, A.G. Agronomic efficiency of NBPT as a urease inhibitor: A review. J. Adv. Res. 2018, 13, 19–27. [Google Scholar] [CrossRef]
- Liang, Y.; et al. Optimizing Nitrogen Input Increased Yield and Efficiency in Maize-Soybean Strip Intercropping System. Agronomy 2024, 14, 2472. [Google Scholar] [CrossRef]
- Kifle, D. Soil Fertility Management and Cropping System Function in Ameliorating Maize Productivity in Ethiopia. World Journal of Agriculture and Soil Science 2020, 4. [Google Scholar] [CrossRef]
- Wani, S.H.; Dar, Z.A. Maize improvement: current advances in yield, quality, and stress tolerance under changing climatic scenarios 2023, 333.
- Dar, F.A.S.Z.A.; Lone, P.A.S.A.A. Recent Advances in Breeding for Abiotic Stress (Drought) Tolerance in Maize. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 2226–2243. [Google Scholar] [CrossRef]
- Suman, J.; et al. Enhanced Efficiency N Fertilizers: an Effective Strategy to Improve Use Efficiency and Ecological Sustainability. Journal of Soil Science and Plant Nutrition 2023, 23, 1472–1488. [Google Scholar] [CrossRef]
- Timilsena, Y.P.; Adhikari, R.; Casey, P.; Muster, T.; Gill, H.; Adhikari, B. Enhanced efficiency fertilisers: a review of formulation and nutrient release patterns. J. Sci. Food Agric. 2015, 95, 1131–1142. [Google Scholar] [CrossRef]
- Gil-Ortiz, R.; et al. Agronomic Assessment of a Controlled-Release Polymer-Coated Urea-Based Fertilizer in Maize. Plants 2021, 10, 594. [Google Scholar] [CrossRef] [PubMed]
- Uzoh, C.F.; Onukwuli, O.D.; Ozofor, I.H.; Odera, R.S. Encapsulation of urea with alkyd resin-starch membranes for controlled N2 release: Synthesis, characterization, morphology and optimum N2 release. Process Safety and Environmental Protection 2019, 121, 133–142. [Google Scholar] [CrossRef]
- Chiaregato, C.G.; França, D.; Messa, L.L.; Pereira, T.D.S.; Faez, R. A review of advances over 20 years on polysaccharide-based polymers applied as enhanced efficiency fertilizers. Carbohydr. Polym. 2022, 279, 119014. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; et al. Improvement of bio-based polyurethane and its optimal application in controlled release fertilizer. Journal of Controlled Release 2022, 350, 748–760. [Google Scholar] [CrossRef]
- Mohammadbagheri, Z.; Rahmati, A.; Hoshyarmanesh, P. Synthesis of a novel superabsorbent with slow-release urea fertilizer using modified cellulose as a grafting agent and flexible copolymer. Int. J. Biol. Macromol. 2021, 182, 1893–1905. [Google Scholar] [CrossRef]
- Vejan, P.; Khadiran, T.; Abdullah, R.; Ahmad, N. Controlled release fertilizer: A review on developments, applications and potential in agriculture. Journal of Controlled Release 2021, 339, 321–334. [Google Scholar] [CrossRef]
- Majeed, Z.; Ramli, N.K.; Mansor, N.; Man, Z. A comprehensive review on biodegradable polymers and their blends used in controlled-release fertilizer processes. Reviews in Chemical Engineering 2015, 31, 69–95. [Google Scholar] [CrossRef]
- Ramli, R.A. Slow release fertilizer hydrogels: a review. Polym. Chem. 2019, 10, 6073–6090. [Google Scholar] [CrossRef]
- Getahun, M.J.; Kassie, B.B.; Alemu, T.S. Recent advances in biopolymer synthesis, properties, & commercial applications: a review. Process Biochemistry 2024, 145, 261–287. [Google Scholar] [CrossRef]
- Farioli, A.S.; Barbero, C.A.; Yslas, E.I.; Acevedo, D.; Cavallo, P.C. Eco-Friendly Polymer Materials Based on ESO and Citric Acid for Controlled-Release Urea and Improved Nutrient Use. ACS Appl. Polym. Mater. 2025, 7, 14108–14119. [Google Scholar] [CrossRef]
- Farioli, A.S.; Martinez, M.V.; Barbero, C.; Yslas, E.; Acevedo, D. The effect of oil raw material composition in the synthesis of bio-sorbents based on inverse vulcanization on the ability to remediate hydrocarbon-contaminated water. A novel method for decontaminating water/fuel emulsions. J. Appl. Polym. Sci. 2024, 141. [Google Scholar] [CrossRef]
- Farioli, A.S.; Martinez, M.V.; Barbero, C.A.; Acevedo, D.F.; Yslas, E.I. [22] Synthesized through Inverse Vulcanization from Soybean and Sunflower Oils. 2024. [Google Scholar] [CrossRef]
- Chalker, J.M.; Worthington, M.J.H.; Lundquist, N.A.; Esdaile, L.J. Synthesis and Applications of Polymers Made by Inverse Vulcanization. Top. Curr. Chem. 2019, 377, 16. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, C.W.; et al. A critical review on the sustainability of inverse vulcanised polymers. RSC Sustainability 2025, 3, 4190–4227. [Google Scholar] [CrossRef]
- Korsmeyer, R.W.; Gurny, R.; Doelker, E.; Buri, P.; Peppas, N.A. Mechanisms of solute release from porous hydrophilic polymers. Int. J. Pharm. 1983, 15, 25–35. [Google Scholar] [CrossRef]
- Ritger, P.L.; Peppas, N.A. A simple equation for description of solute release I. Fickian and non-fickian release from non-swellable devices in the form of slabs, spheres, cylinders or discs. Journal of Controlled Release 1987, 5, 23–36. [Google Scholar] [CrossRef]
- Langenbucher, F. Letters to the Editor: Linearization of dissolution rate curves by the Weibull distribution. 1972. [Google Scholar] [CrossRef]
- Manoharan, G.; Nithyanandam, R.; Antony, J.S.; Rajendran, S. Investigating the Effects of Urea-Zinc Sulfate-L Phenylalanine on the Corrosion Inhibition of Mild Steel Exposed to pH-4 Sulfuric Acid. Curr. Anal. Chem. 2025, 21, 68–78. [Google Scholar] [CrossRef]
- Paśko, P.; et al. Molecular Profiling and FTIR Characterization of Wheat Germ Oil, Supported by the Screening of Its Anti-Inflammatory and Cytotoxic Properties. Biomolecules 2025, 15, 464. [Google Scholar] [CrossRef]
- Worthington, M.J.H.; Kucera, R.L.; Chalker, J.M. Green chemistry and polymers made from sulfur. Green Chemistry 2017, 19, 2748–2761. [Google Scholar] [CrossRef]
- Trofimov, B.A.; Sinegovskaya, L.M.; Gusarova, N.K. Vibrations of the S–S bond in elemental sulfur and organic polysulfides: a structural guide. Journal of Sulfur Chemistry 2009, 30, 518–554. [Google Scholar] [CrossRef]
- Sugeta, H.; Go, A.; Miyazawa, T. S–S and C–S stretching vibrations and molecular conformations of dialkyl disulfides and cystine. Chem. Lett. 1972, 1, 83–86. [Google Scholar] [CrossRef]
- Etminani-Esfahani, N.; Rahmati, A.; Mansouri, O. Preparation of green hydrogel for slow-release urea and its positive effect on improving wheat yield under deficit irrigation conditions and remediation of soil. Ind. Crops Prod. 2025, 231, 121168. [Google Scholar] [CrossRef]
- Salimi, M.; Motamedi, E.; Motesharezedeh, B.; Hosseini, H.M.; Alikhani, H.A. Starch-g-poly(acrylic acid-co-acrylamide) composites reinforced with natural char nanoparticles toward environmentally benign slow-release urea fertilizers. J. Environ. Chem. Eng. 2020, 8, 103765. [Google Scholar] [CrossRef]
- Yadav, P.; Mehata, D.K.; Yadav, B.; Adhikari, R.; Lahutiya, V. Varietal trial of maize (Zea mays L.) for evaluation of growth and yield parameters in Terai region of Nepal. Journal of Agriculture 2024, 7, 221–232. [Google Scholar] [CrossRef]
- T. Tricahyati, … S. S.-S. N., and undefined 2024, “Inventarisasi Penyakit Pada Jagung (Zea mays L.) di Desa Bakung Kecamatan Indralaya Utara Kabupaten Ogan Ilir Sumatera Selatan. conference.unsri.ac.idT Tricahyati, SHK Suparman, H Hamidson, A Arsi, V Agustin, M Taqiyyuddin, YP SariSeminar Nasional Lahan Suboptimal, 2024•conference.unsri.ac.id, Accessed: Feb. 21, 2026. [Online]. Available: https://conference.unsri.ac.id/index.php/lahansuboptimal/article/view/2998.
- Begum, T.; Follett, P.A.; Sharaby, M.R.; Shankar, S.; Salmieri, S.; Lacroix, M. Development of Optimized Bioactive Nanocomposite Films Using a Central Composite Design for Control of Microbial Contamination in Stored Rice. J. Food Sci. 2025, 90, e70760. [Google Scholar] [CrossRef]
- Broukanlou, M.; Hosseini, S.A.; Pazhang, Y. ZSM-5/PEG/DOX nanocarrier for pH-responsive doxorubicin release: Kinetic, isothermal, and cytotoxic studies. Microporous and Mesoporous Materials 2025, 387, 113531. [Google Scholar] [CrossRef]
- Wambsganss, J.; Huys, R.; Hättenschwiler, S.; Poirier, V.; Munson, A.D.; Freschet, G.T. The afterlife effects of leaf and root litter traits on soil N cycling. Journal of Ecology 2025, 113, 913–930. [Google Scholar] [CrossRef]
- Mandal, M.; Lodhi, R.S.; Chourasia, S.; Das, S.; Das, P. A Review on Sustainable Slow-Release N, P, K Fertilizer Hydrogels for Smart Agriculture; Wiley Online Library, 2025; p. 90. [Google Scholar] [CrossRef]
- S. Phang, L. Sin, S. Bee, T. T.-A. C. Proceedings, and undefined 2020, “Release kinetic model of nitrogen released encapsulated in starch-alginate controlled released urea: Diffusion and its decay release. pubs.aip.org, 2020. [CrossRef]
- Lan, R.; Wang, G.; Yang, L.; Wang, T.J.; Kan, C.; Jin, Y. Prediction of Release Characteristics of Film-Coated Urea from Structure Characterization Data of the Film. Chem. Eng. Technol. 2013, 36. [Google Scholar] [CrossRef]
- Babadi, F.E.; Yunus, R.; Soltani, S.M.; Shotipruk, A. Release Mechanisms and Kinetic Models of Gypsum-Sulfur-Zeolite-Coated Urea Sealed with Microcrystalline Wax for Regulated Dissolution. ACS Omega 2021, 6. [Google Scholar] [CrossRef]
- Nanganoa, L.T.; Ngome, F.A.; Suh, C.; Basga, S.D. Assessing Soil Nutrients Variability and Adequacy for the Cultivation of Maize, Cassava, and Sorghum in Selected Agroecological Zones of Cameroon. International Journal of Agronomy 2020, 2020, 8887318. [Google Scholar] [CrossRef]
- Fadl, M.E.; et al. Irrigation Practices and Their Effects on Soil Quality and Soil Characteristics in Arid Lands: A Comprehensive Geomatic Analysis. Soil Systems 2024, 8, 52. [Google Scholar] [CrossRef]
- Jiang, C.; et al. Effects of Sulfur Application on the Quality of Fresh Waxy Maize. Plants 2024, 13. [Google Scholar] [CrossRef]
- Wang, H.; Cui, S.; Fu, J.; Gong, H.; Liu, S. Sulfur Application Improves the Nutritional Quality of Maize by Regulating the Amino Acid Balance of Grains. Agronomy 2023, 13. [Google Scholar] [CrossRef]
- Xu, R.; et al. Combination of Polymer-Coated Urea and Rapid-Release Urea Increases Grain Yield and Nitrogen Use Efficiency of Rice by Improving Root and Shoot Activities. Agronomy 2024, 14. [Google Scholar] [CrossRef]
- Ejemplo Ouyang, P.; Narayanan, M.; Shi, X.; Chen, X.; Li, Z.; Luo, Y.; Ma, Y. Integrating biochar and bacteria for sustainable remediation of metal-contaminated soils. Biochar 2023, 5, 63. [Google Scholar] [CrossRef]
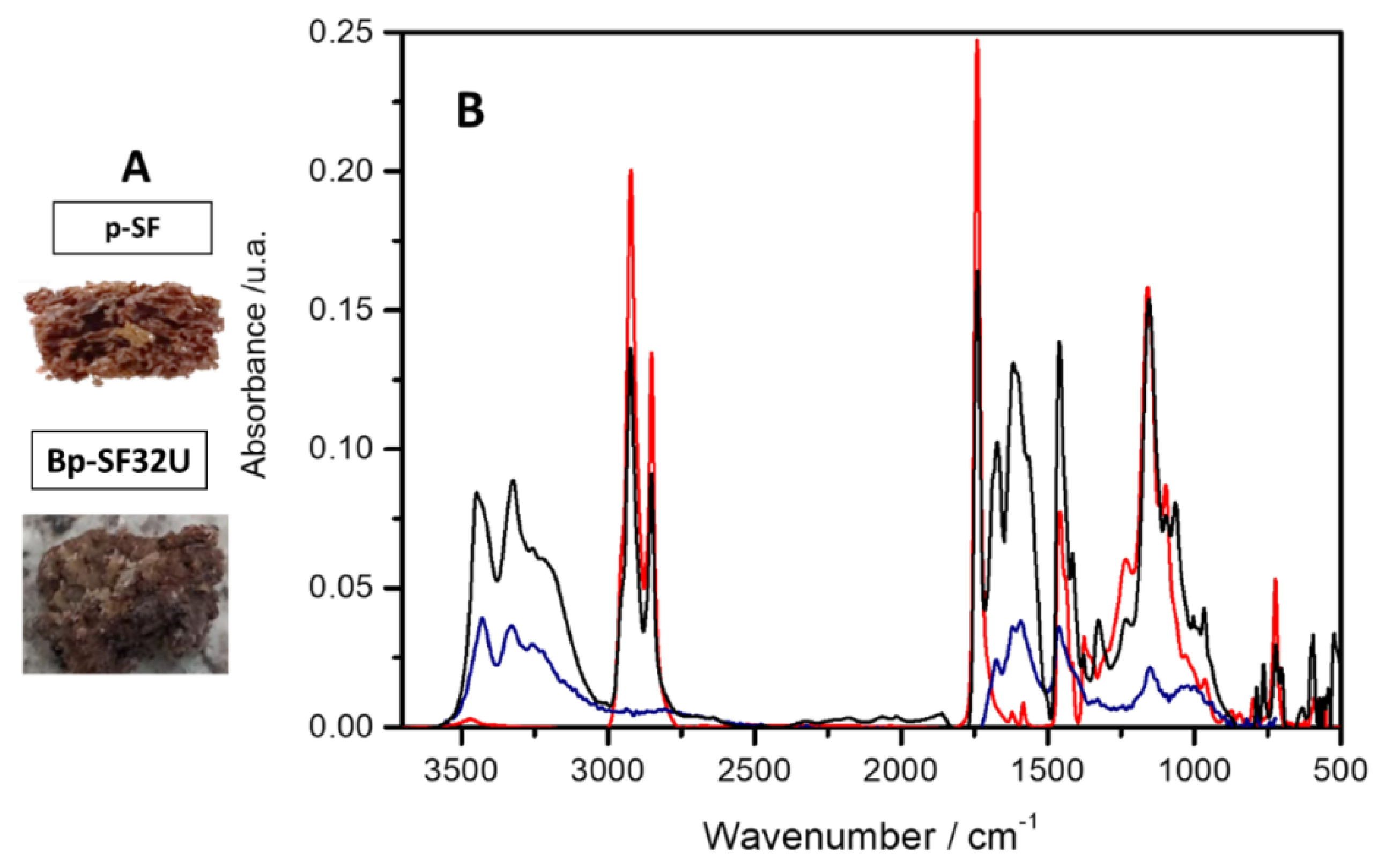
| Wavenumber (cm-1) | Assignation | Reference |
|---|---|---|
| ~ 3455 | -NH stretching | [25] |
| ~2954 | C–H stretching | [26] |
| ~ 1700 | C=O stretching | [22] |
| [25] | C-N deformation | [25] |
| ~ 1457 | C-N stretching | [25] |
| ~1300–1000 | C–O stretching | [27] |
| 930 | S–S stretching | [21,28,29] |
| 790 | C–S stretching | [28,29] |
| ~500–400 | S–S stretching | [28,29] |
| Sample | Mathematical models | Parameters | ||
|---|---|---|---|---|
| Korsmeyer-Peppas* | k | n | R2 | |
| Bp-SF25U | 0.24139 ± 0.03984 | 0.39273 ± 0.05133 | 0.97295 | |
| Bp-SF32U | 0.41256 ± 0.02076 | 0.23330 ± 0.01766 | 0.97458 | |
| Weibull* | α | b | R2 | |
| Bp-SF25U | 9.31504 ± 0.24478 | 0.96778 ± 0.03709 | 0.99898 | |
| Bp-SF32U | 5.17808 ± 0.73226 | 0.43536 ± 0.05148 | 0.96152 | |
| Physical Parameters | |
|---|---|
| Conductivity | 147.8 μs |
| pH | 5.90 |
| Soil color | Dark brown |
| Soil Odor | Odorless |
| Biocomposites | Sulfur (grams) |
SF oil (grams) |
Urea (grams) |
%U |
|---|---|---|---|---|
| Bp-SF | 30 | 30 | 0.00 | 0.00% |
| Bp-SF25U | 30 | 30 | 20.00 | 25.00% |
| Bp-SF32U | 30 | 30 | 28.00 | 31.81% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




