Submitted:
17 March 2026
Posted:
18 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Production and Extraction of Corynebacterial Astaxanthin
2.2. Intracellular In Vivo Antioxidant Assay
2.3. AOP1 Assay with De-Esterified Algal Astaxanthin
2.4. Quantification of Astaxanthin by HPLC
3. Results
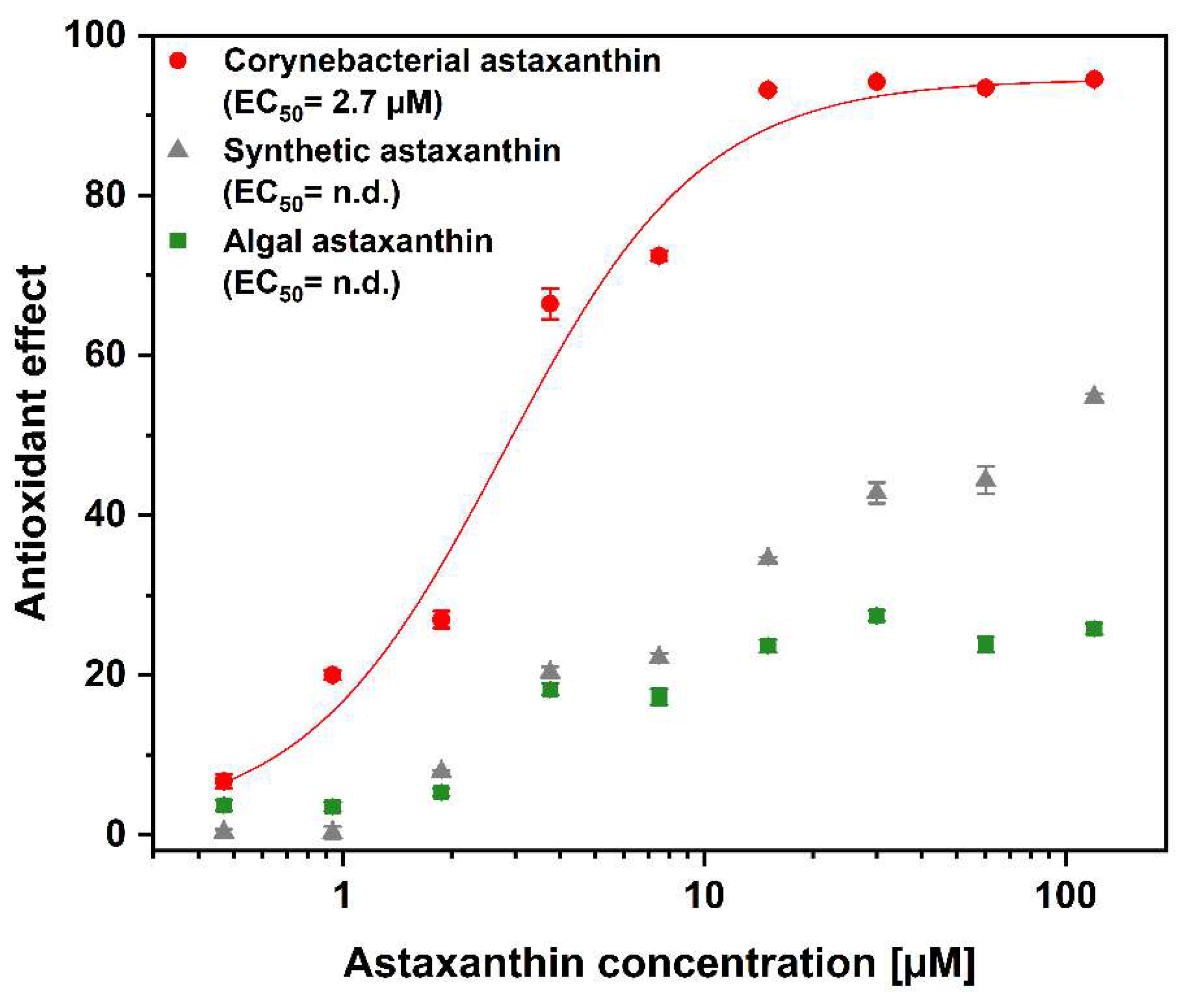
3.1. Intracellular Antioxidant Activity Testing of Different Astaxanthins
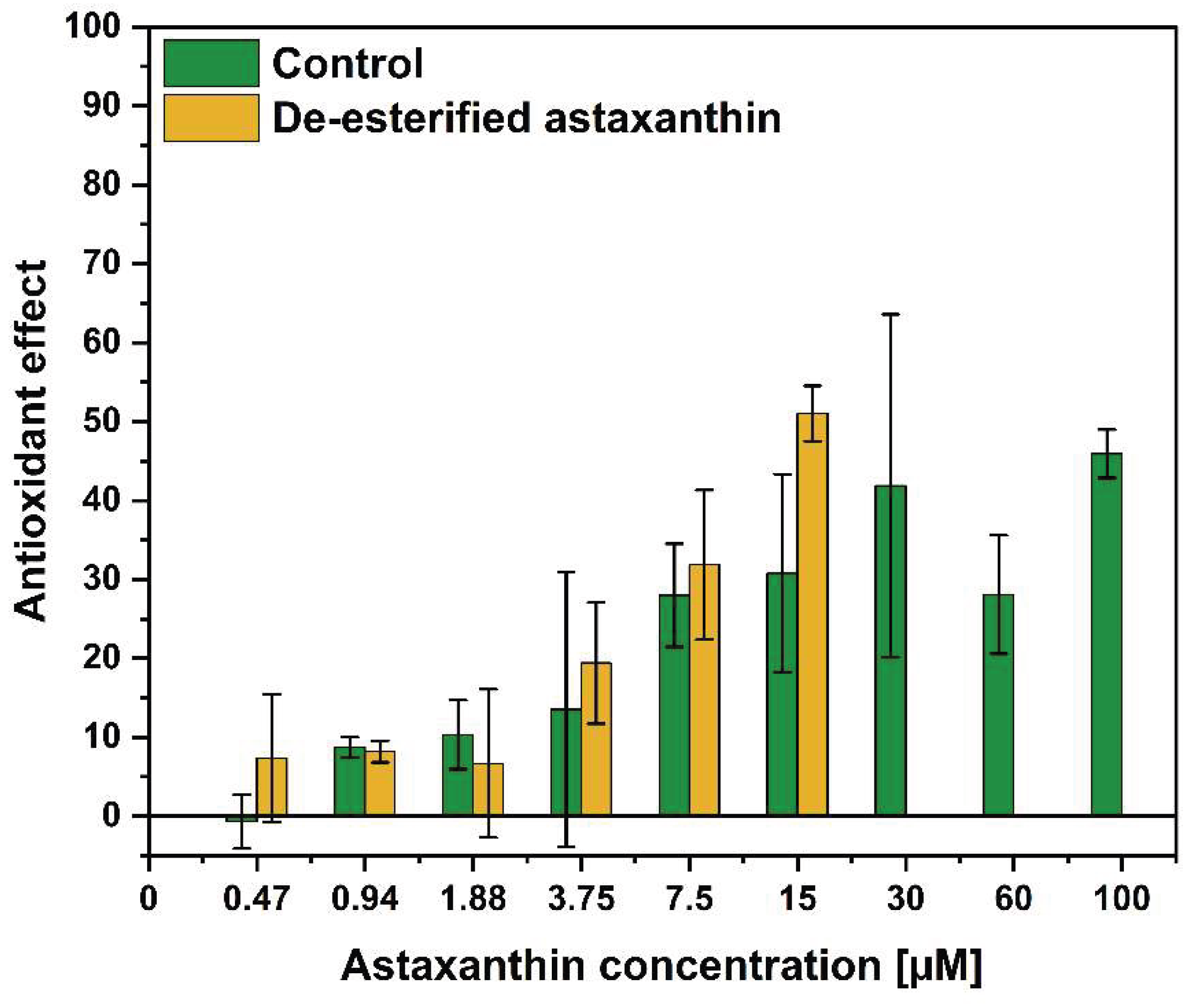
3.2. De-Esterification Algal-Based Astaxanthin and Its Intracellular Activity
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AOP | Antioxidant power test |
| AA | Algal astaxanthin |
| CA | Corynebacterial astaxanthin |
| CDW | Cell dry weight |
| SA | Synthetic astaxanthin |
References
- Zuo, L.; Prather, E.R.; Stetskiv, M.; Garrison, D.E.; Meade, J.R.; Peace, T.I.; Zhou, T. Inflammaging and Oxidative Stress in Human Diseases: From Molecular Mechanisms to Novel Treatments. International Journal of Molecular Sciences 2019, 20, 4472. [Google Scholar] [CrossRef]
- Altanam, S.Y.; Darwish, N.; Bakillah, A. Exploring the Interplay of Antioxidants, Inflammation, and Oxidative Stress: Mechanisms, Therapeutic Potential, and Clinical Implications. Diseases 2025, 13, 309. [Google Scholar] [CrossRef] [PubMed]
- Valaitienė, J.; Laučytė-Cibulskienė, A. Oxidative Stress and Its Biomarkers in Cardiovascular Diseases. Artery Research 2024, 30, 18. [Google Scholar] [CrossRef]
- Reddy, V.P. Oxidative Stress in Health and Disease. Biomedicines 2023, 11, 2925. [Google Scholar] [CrossRef]
- Petruk, G.; Del Giudice, R.; Rigano, M.M.; Monti, D.M. Antioxidants from Plants Protect against Skin Photoaging. Oxid Med Cell Longev 2018, 2018, 1454936. [Google Scholar] [CrossRef]
- Hooda, R.; Madke, B.; Choudhary, A. Photoaging: Reversal of the Oxidative Stress Through Dietary Changes and Plant-Based Products. Cureus 2023, 15, e37321. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Omaye, S.T. Air pollutants, oxidative stress and human health. Mutat Res 2009, 674, 45–54. [Google Scholar] [CrossRef]
- Kammeyer, A.; Luiten, R.M. Oxidation events and skin aging. Ageing Res Rev 2015, 21, 16–29. [Google Scholar] [CrossRef]
- Yin, H.; Xu, L.; Porter, N.A. Free Radical Lipid Peroxidation: Mechanisms and Analysis. Chem. Rev. 2011, 111, 5944–5972. [Google Scholar] [CrossRef]
- Cadet, J.; Davies, K.J.A. Oxidative DNA damage & repair: An introduction. Free Radic Biol Med 2017, 107, 2–12. [Google Scholar] [CrossRef]
- Sies, H.; Berndt, C.; Jones, D.P. Oxidative Stress. Annual Review of Biochemistry 2017, 86, 715–748. [Google Scholar] [CrossRef]
- Sies, H.; Jones, D.P. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nature Reviews Molecular Cell Biology 2020, 21, 363–383. [Google Scholar] [CrossRef]
- Forrester, S.J.; Kikuchi, D.S.; Hernandes, M.S.; Xu, Q.; Griendling, K.K. Reactive Oxygen Species in Metabolic and Inflammatory Signaling. Circulation Research 2018, 122, 877–902. [Google Scholar] [CrossRef]
- Gulcin, İ. Antioxidants: a comprehensive review. Arch Toxicol 2025, 99, 1893–1997. [Google Scholar] [CrossRef]
- Forman, H.J.; Zhang, H. Targeting oxidative stress in disease: promise and limitations of antioxidant therapy. Nat Rev Drug Discov 2021, 20, 689–709. [Google Scholar] [CrossRef] [PubMed]
- Dini, I.; Laneri, S. Nutricosmetics: A brief overview. Phytother Res 2019, 33, 3054–3063. [Google Scholar] [CrossRef] [PubMed]
- Budzianowska, A.; Banaś, K.; Budzianowski, J.; Kikowska, M. Antioxidants to Defend Healthy and Youthful Skin—Current Trends and Future Directions in Cosmetology. Applied Sciences 2025, 15, 2571. [Google Scholar] [CrossRef]
- Dutta, S.; Kumar, S.P.J.; Banerjee, R. A comprehensive review on astaxanthin sources, structure, biochemistry and applications in the cosmetic industry. Algal Research 2023, 74, 103168. [Google Scholar] [CrossRef]
- Brotosudarmo, T.H.P.; Limantara, L.; Setiyono, E.; Heriyanto. Structures of Astaxanthin and Their Consequences for Therapeutic Application. Int J Food Sci 2020, 2020, 2156582. [Google Scholar] [CrossRef] [PubMed]
- Insights, G.M. Astaxanthin Market Size & Share 2025 - 2034: Market Size by Source, by Application, Industry Analysis, Growth Forecast April 2025 2025.
- Sun, J.; Zhang, Z.; Gao, L.; Yang, F. Advances and trends for astaxanthin synthesis in Phaffia rhodozyma. Microbial cell factories 2025, 24, 100. [Google Scholar] [CrossRef]
- Ji, W.; Wang, W.; Chen, X.; Sun, M.; Zhang, J.; Nan, B.; Li, X.; Wang, Y.; Wang, Y.; Piao, C. Stress-induced astaxanthin biosynthesis in Phaffia rhodozyma: Bridging mechanistic understanding to industrial Feasibility. Bioresource technology 2025, 435, 132957. [Google Scholar] [CrossRef]
- Luna-Flores, C.H.; Wang, A.; von Hellens, J.; Speight, R.E. Towards commercial levels of astaxanthin production in Phaffia rhodozyma. Journal of Biotechnology 2022, 350, 42–54. [Google Scholar] [CrossRef]
- Mussagy, C.U.; Pereira, J.F.B.; Dufossé, L. Astaxanthin production using Paracoccus carotinifaciens: a way forward? Trends in biotechnology 2023, 41, 996–999. [Google Scholar] [CrossRef] [PubMed]
- Ariyadasa, T.U.; Thevarajah, B.; Anthonio, R.A.D.P.; Nimarshana, P.H.V.; Wasath, W.A.J. From present to prosperity: assessing the current status and envisioning opportunities in the industrial-scale cultivation of Haematococcus pluvialis for astaxanthin production. Phytochem Rev 2024, 23, 749–779. [Google Scholar] [CrossRef]
- Wang, C.; Hong, Z.; Song, M.; Zheng, H.; Zhou, Q.; Yang, H.; Li, H.; Huang, D. Production of astaxanthin with high purity and activity based on engineering improvement strategies. Journal of Biotechnology 2025, 405, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Bu, Y.-F.; Liu, J.-Z. Metabolic Engineering of Escherichia coli for Producing Astaxanthin as the Predominant Carotenoid. Marine Drugs 2017, 15, 296. [Google Scholar] [CrossRef]
- Li, N.; Han, Z.; O’Donnell, T.J.; Kurasaki, R.; Kajihara, L.; Williams, P.G.; Tang, Y.; Su, W.W. Production and excretion of astaxanthin by engineered Yarrowia lipolytica using plant oil as both the carbon source and the biocompatible extractant. Applied microbiology and biotechnology 2020, 104, 6977–6989. [Google Scholar] [CrossRef]
- Tramontin, L.R.R.; Kildegaard, K.R.; Sudarsan, S.; Borodina, I. Enhancement of Astaxanthin Biosynthesis in Oleaginous Yeast Yarrowia lipolytica via Microalgal Pathway. Microorganisms 2019, 7, 472. [Google Scholar] [CrossRef]
- Meyer, F.; Schmitt, I.; Schäffer, T.; Wendisch, V.F.; Henke, N.A. Design-of-Experiment-Guided Establishment of a Fermentative Bioprocess for Biomass-Bound Astaxanthin with Corynebacterium glutamicum. Fermentation 2023, 9, 969. [Google Scholar] [CrossRef]
- Meyer, F.; Schmitt, I.; Wendisch, V.F.; Henke, N.A. Response surface-based media optimization for astaxanthin production in Corynebacterium glutamicum. Frontiers in bioengineering and biotechnology 2025, 13–2025. [Google Scholar] [CrossRef] [PubMed]
- Göttl, V.L.; Meyer, F.; Schmitt, I.; Persicke, M.; Peters-Wendisch, P.; Wendisch, V.F.; Henke, N.A. Enhancing astaxanthin biosynthesis and pathway expansion towards glycosylated C40 carotenoids by Corynebacterium glutamicum. Scientific reports 2024, 14, 8081. [Google Scholar] [CrossRef] [PubMed]
- Seeger, J.; Wendisch, V.F.; Henke, N.A. Extraction and Purification of Highly Active Astaxanthin from Corynebacterium glutamicum Fermentation Broth. Marine Drugs 2023, 21, 530. [Google Scholar] [CrossRef]
- Seeger, J.; Zäh, M.; Wendisch, V.F.; Brandenbusch, C.; Henke, N.A. Supercritical carbon dioxide extraction of astaxanthin from Corynebacterium glutamicum. Bioresources and Bioprocessing 2025, 12, 46. [Google Scholar] [CrossRef]
- Higuera-Ciapara, I.; Félix-Valenzuela, L.; Goycoolea, F.M. Astaxanthin: a review of its chemistry and applications. Crit Rev Food Sci Nutr 2006, 46, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Yao, W.; Wen, D.; Li, X.; Wu, S.; Leng, X. Dietary Adonis. aestivalis extract improved the flesh pigmentation, antioxidative status and shelf-life of rainbow trout (Oncorhynchus mykiss). Aquaculture Nutrition 2020, 26, 2032–2042. [Google Scholar] [CrossRef]
- Renstrøm, B.; Borch, G.; Skulberg, O.M.; Liaaen-Jensen, S. Optical purity of (3S,3’S)-astaxanthin from Haematococcus pluvialis. Phytochemistry 1981, 20, 2561–2564. [Google Scholar] [CrossRef]
- Andrewes, A.G.; Starr, M.P. (3R,3′R)-astaxanthin from the yeast Phaffia rhodozyma. Phytochemistry 1976, 15, 1009–1011. [Google Scholar] [CrossRef]
- Holtin, K.; Kuehnle, M.; Rehbein, J.; Schuler, P.; Nicholson, G.; Albert, K. Determination of astaxanthin and astaxanthin esters in the microalgae Haematococcus pluvialis by LC-(APCI)MS and characterization of predominant carotenoid isomers by NMR spectroscopy. Analytical and Bioanalytical Chemistry 2009, 395, 1613–1622. [Google Scholar] [CrossRef]
- Miao, F.; Lu, D.; Li, Y.; Zeng, M. Characterization of astaxanthin esters in Haematococcus pluvialis by liquid chromatography–atmospheric pressure chemical ionization mass spectrometry Anal. Analytical Biochemistry 2006, 352, 176–181. [Google Scholar] [CrossRef]
- Blois, M.S. Antioxidant Determinations by the Use of a Stable Free Radical. Nature 1958, 181, 1199–1200. [Google Scholar] [CrossRef]
- Stielow, M.; Witczyńska, A.; Kubryń, N.; Fijałkowski, Ł.; Nowaczyk, J.; Nowaczyk, A. The Bioavailability of Drugs-The Current State of Knowledge. Molecules (Basel, Switzerland) 2023, 28. [Google Scholar] [CrossRef]
- Chen, M.L.; Shah, V.; Patnaik, R.; Adams, W.; Hussain, A.; Conner, D.; Mehta, M.; Malinowski, H.; Lazor, J.; Huang, S.M.; et al. Bioavailability and bioequivalence: an FDA regulatory overview. Pharm Res 2001, 18, 1645–1650. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Cui, Y.; Liu, H.; Wang, Y.; Nan, B.; Li, X.; Wang, Y. Progress in the bioavailability of natural astaxanthin: Influencing factors, enhancement strategies, evaluation methods, and limitations of current research. Trends in Food Science & Technology 2025, 160, 104998. [Google Scholar] [CrossRef]
- Tominaga, K.; Hongo, N.; Karato, M.; Yamashita, E. Cosmetic benefits of astaxanthin on humans subjects. Acta biochimica Polonica 2012, 59 1, 43–47. [Google Scholar] [CrossRef]
- Meephansan, J.; Rungjang, A.; Yingmema, W.; Deenonpoe, R.; Ponnikorn, S. Effect of astaxanthin on cutaneous wound healing. Clin Cosmet Investig Dermatol 2017, 10, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Hama, S.; Takahashi, K.; Inai, Y.; Shiota, K.; Sakamoto, R.; Yamada, A.; Tsuchiya, H.; Kanamura, K.; Yamashita, E.; Kogure, K. Protective effects of topical application of a poorly soluble antioxidant astaxanthin liposomal formulation on ultraviolet-induced skin damage. Journal of Pharmaceutical Sciences 2012, 101, 2909–2916. [Google Scholar] [CrossRef]
- Gironde, C.; Rigal, M.; Dufour, C.; Furger, C. AOP1, a New Live Cell Assay for the Direct and Quantitative Measure of Intracellular Antioxidant Effects. Antioxidants 2020, 9, 471. [Google Scholar] [CrossRef]
- Gómez, P.I.; Inostroza, I.; Pizarro, M.; Pérez, J. From genetic improvement to commercial-scale mass culture of a Chilean strain of the green microalga Haematococcus pluvialis with enhanced productivity of the red ketocarotenoid astaxanthin. AoB PLANTS 2013, 5. [Google Scholar] [CrossRef]
- Blanchard, G.; Pich, C.; Hohl, D. HaCaT cells as a model system to study primary cilia in keratinocytes. Experimental Dermatology 2022, 31, 1276–1280. [Google Scholar] [CrossRef]
- McLean, P.; Marshall, J.; García-Bilbao, A.; Beal, D.; Katsumiti, A.; Carrière, M.; Boyles, M.S.P. A comparison of dermal toxicity models; assessing suitability for safe(r)-by-design decision-making and for screening nanomaterial hazards. Toxicology in Vitro 2024, 97, 105792. [Google Scholar] [CrossRef]
- Lima, S.G.M.; Freire, M.; Oliveira, V.D.S.; Solisio, C.; Converti, A.; de Lima Á, A.N. Astaxanthin Delivery Systems for Skin Application: A Review. Mar Drugs 2021, 19. [Google Scholar] [CrossRef]
- Liu, X.; Luo, Q.; Rakariyatham, K.; Cao, Y.; Goulette, T.; Liu, X.; Xiao, H. Antioxidation and anti-ageing activities of different stereoisomeric astaxanthin in vitro and in vivo. Journal of Functional Foods 2016, 25, 50–61. [Google Scholar] [CrossRef]
- Apak, R. Current Issues in Antioxidant Measurement. J Agric Food Chem 2019, 67, 9187–9202. [Google Scholar] [CrossRef] [PubMed]
- Oninku, B.; Lomas, M.W.; Burr, G.; Aryee, A.N.A. Astaxanthin: An overview of its sources, extraction methods, encapsulation techniques, characterization, and bioavailability. Journal of Agriculture and Food Research 2025, 21, 101869. [Google Scholar] [CrossRef]
- Snell, T.W.; Carberry, J. Astaxanthin Bioactivity Is Determined by Stereoisomer Composition and Extraction Method. Nutrients 2022, 14. [Google Scholar] [CrossRef]
- Yang, L.; Qiao, X.; Gu, J.; Li, X.; Cao, Y.; Xu, J.; Xue, C. Influence of molecular structure of astaxanthin esters on their stability and bioavailability. Food Chemistry 2020, 343, 128497. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Zhong, S.; Kopec, R.E. Carotenoid Bioaccessibility and Caco-2 Cell Uptake Following Novel Encapsulation Using Medium Chain Triglycerides. J Diet Suppl 2024, 21, 756–770. [Google Scholar] [CrossRef]
- Yang, L.; Qiao, X.; Gu, J.; Li, X.; Cao, Y.; Xu, J.; Xue, C. Influence of molecular structure of astaxanthin esters on their stability and bioavailability. Food Chemistry 2021, 343, 128497. [Google Scholar] [CrossRef]
- Zhou, Q.; Xu, J.; Yang, L.; Gu, C.; Xue, C. Thermal stability and oral absorbability of astaxanthin esters from Haematococcus pluvialis in Balb/c mice. Journal of the Science of Food and Agriculture 2019, 99, 3662–3671. [Google Scholar] [CrossRef]
- Ulloth, J.E.; Casiano, C.A.; De Leon, M. Palmitic and stearic fatty acids induce caspase-dependent and -independent cell death in nerve growth factor differentiated PC12 cells. Journal of Neurochemistry 2003, 84, 655–668. [Google Scholar] [CrossRef] [PubMed]
- Alsabeeh, N.; Chausse, B.; Kakimoto, P.A.; Kowaltowski, A.J.; Shirihai, O. Cell culture models of fatty acid overload: Problems and solutions. Biochim Biophys Acta Mol Cell Biol Lipids 2018, 1863, 143–151. [Google Scholar] [CrossRef]
- Yang, L.; Gu, J.; Luan, T.; Qiao, X.; Cao, Y.; Xue, C.; Xu, J. Influence of oil matrixes on stability, antioxidant activity, bioaccessibility and bioavailability of astaxanthin ester. Journal of the Science of Food and Agriculture 2020, 101. [Google Scholar] [CrossRef]
- Yan, X.; Huang, J.; Huang, L.; Luo, C.; Li, Z.; Xu, P.; Tan, K.; Cheong, K.-L.; Tan, K. Effects of dietary lipids on bioaccessibility and bioavailability of natural carotenoids. LWT 2024, 200, 116171. [Google Scholar] [CrossRef]
- Kaur, R.; Arora, S.; Goswami, M. Evaluation of fabricated solid microneedles as smart approach for transdermal drug delivery system of astaxanthin. International Journal of Applied Pharmaceutics 2023. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



