Submitted:
17 March 2026
Posted:
17 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Adrenal Gland Collection and Sampling
2.3. Histochemical STUDY
2.4. Morphometric Analysis
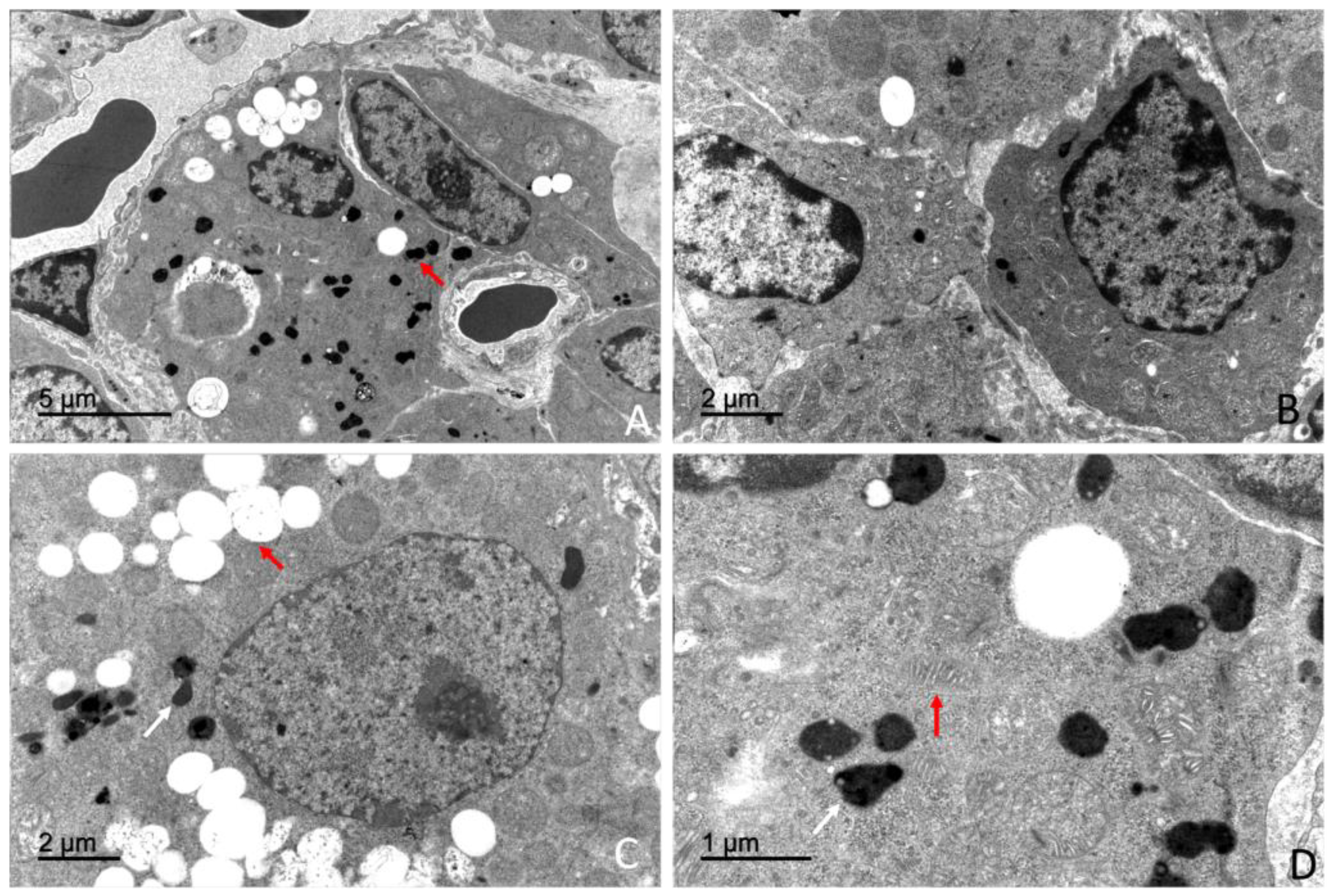
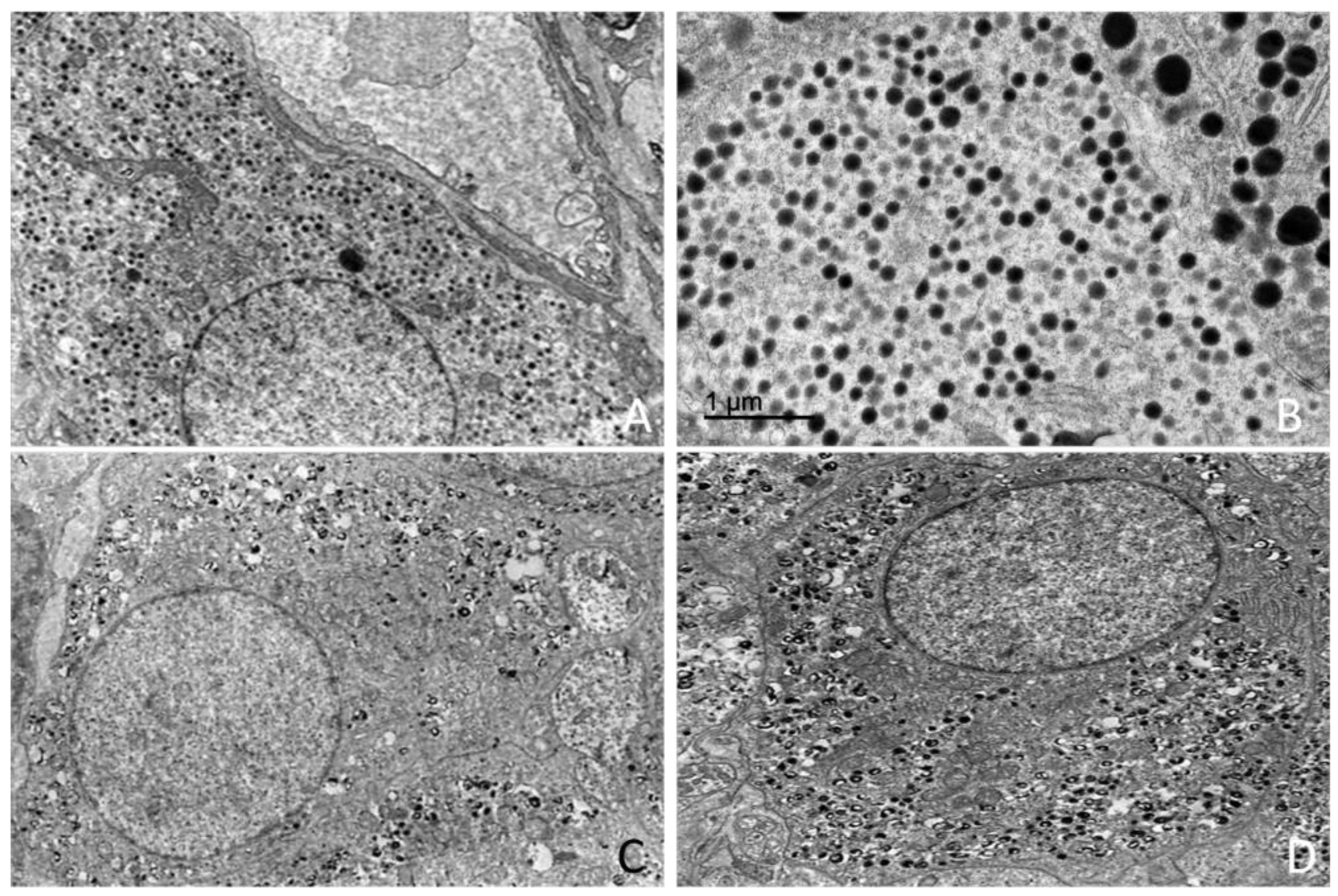
2.5. Ultrastructural Study (Transmission Electron Microscopy)
3. Results
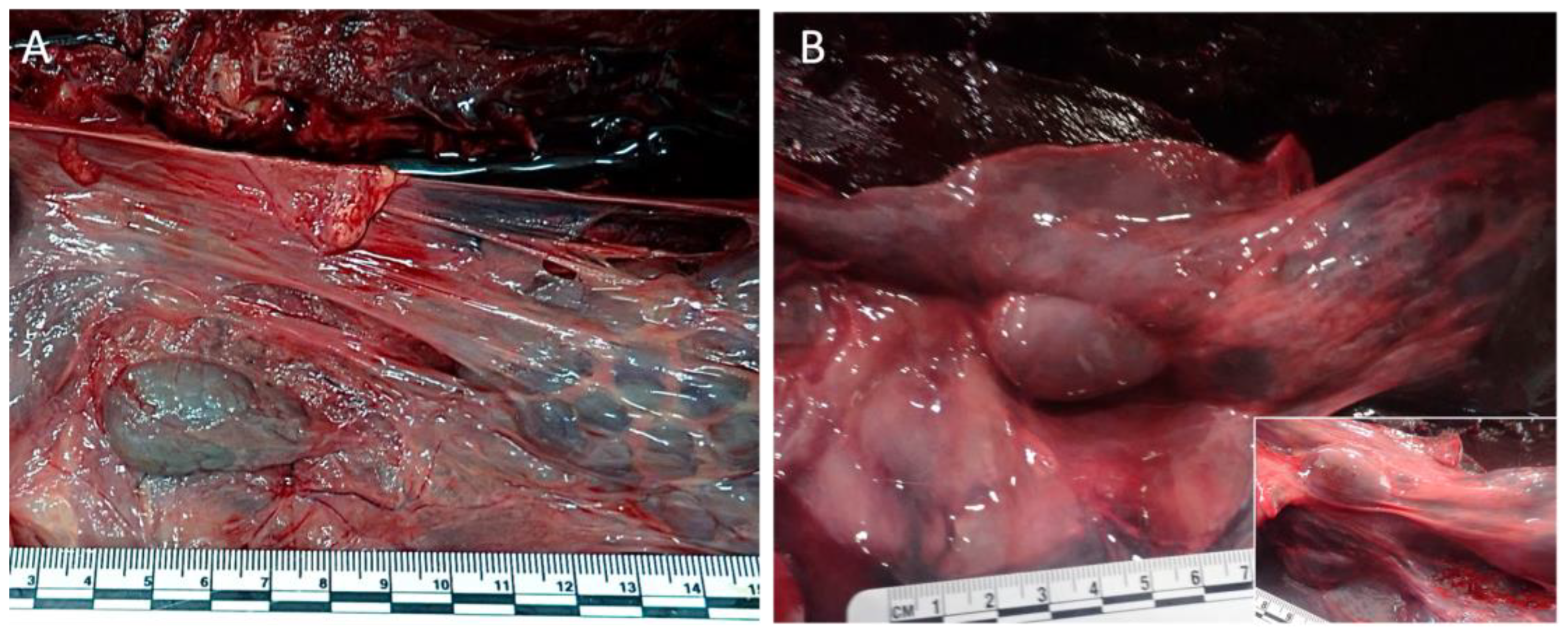
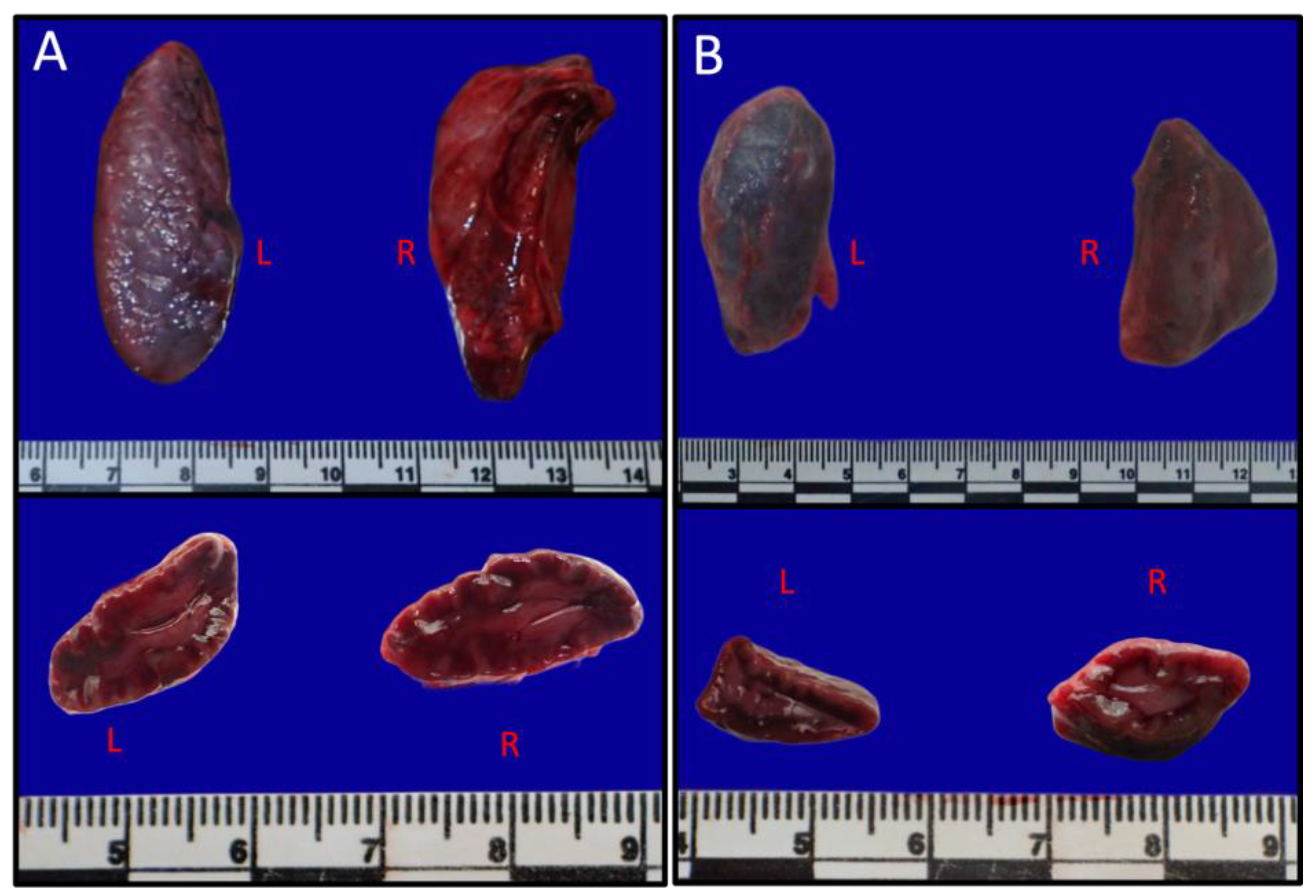
3.1. Gross anatomy and Morphology
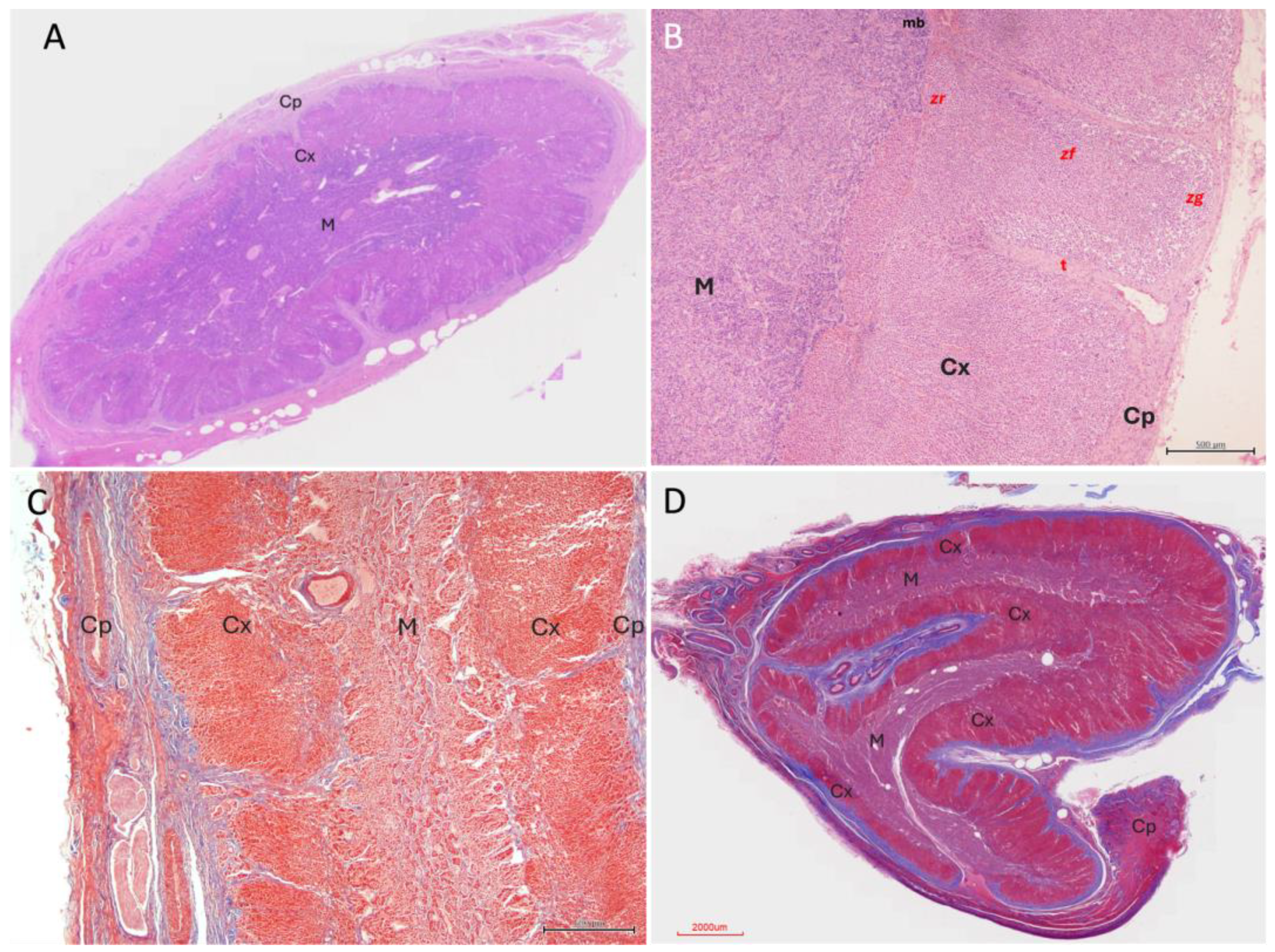
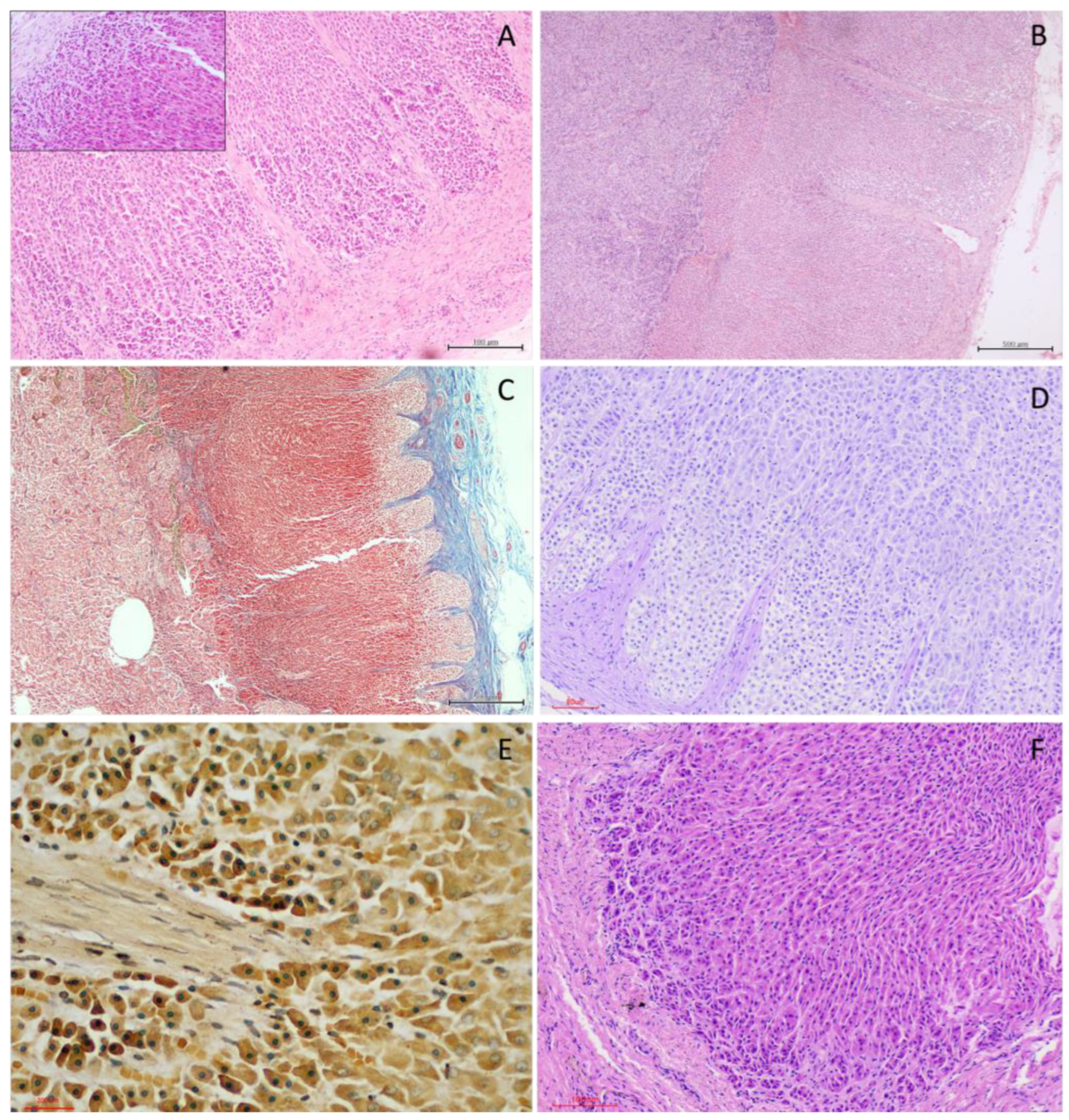
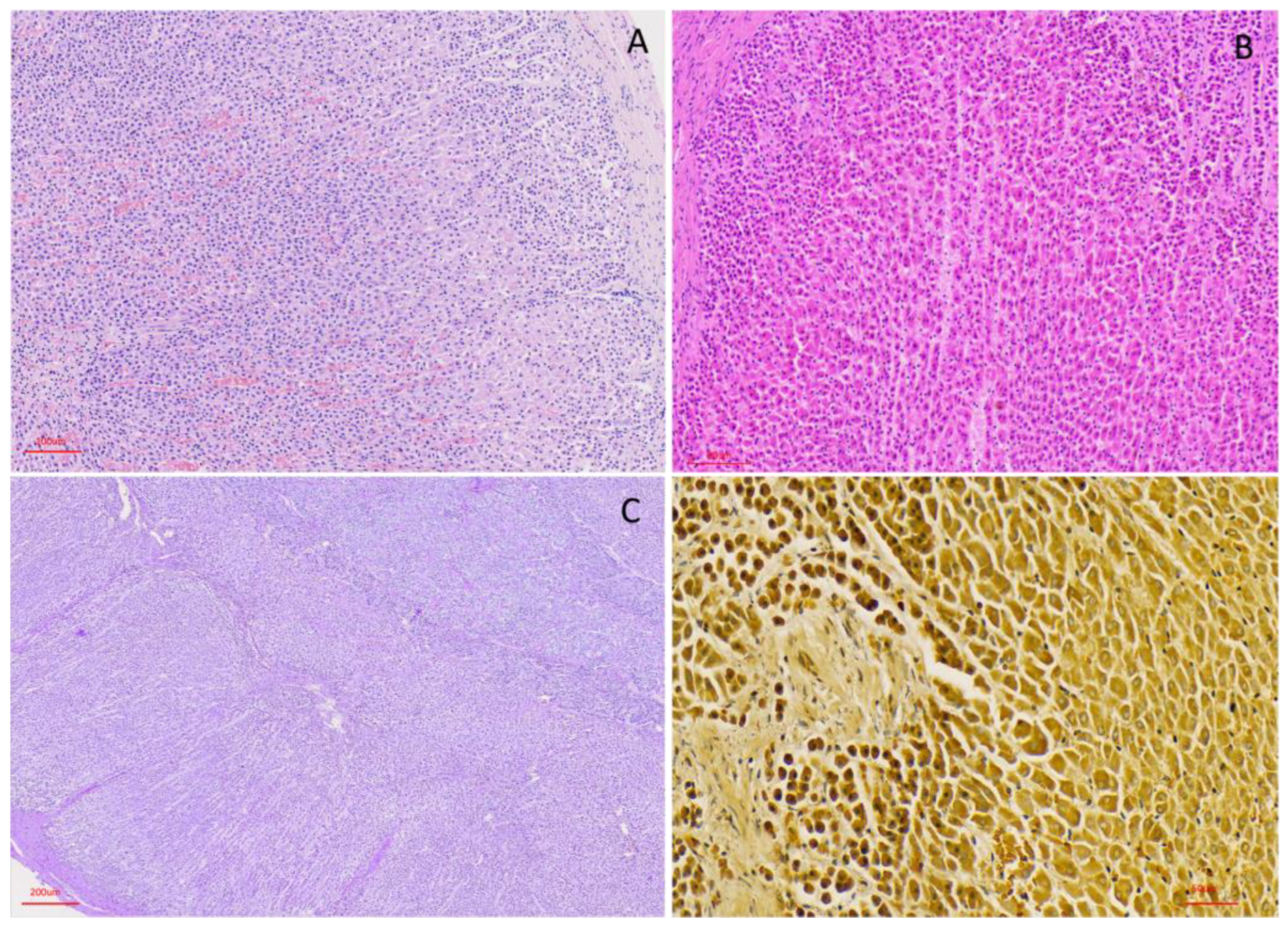
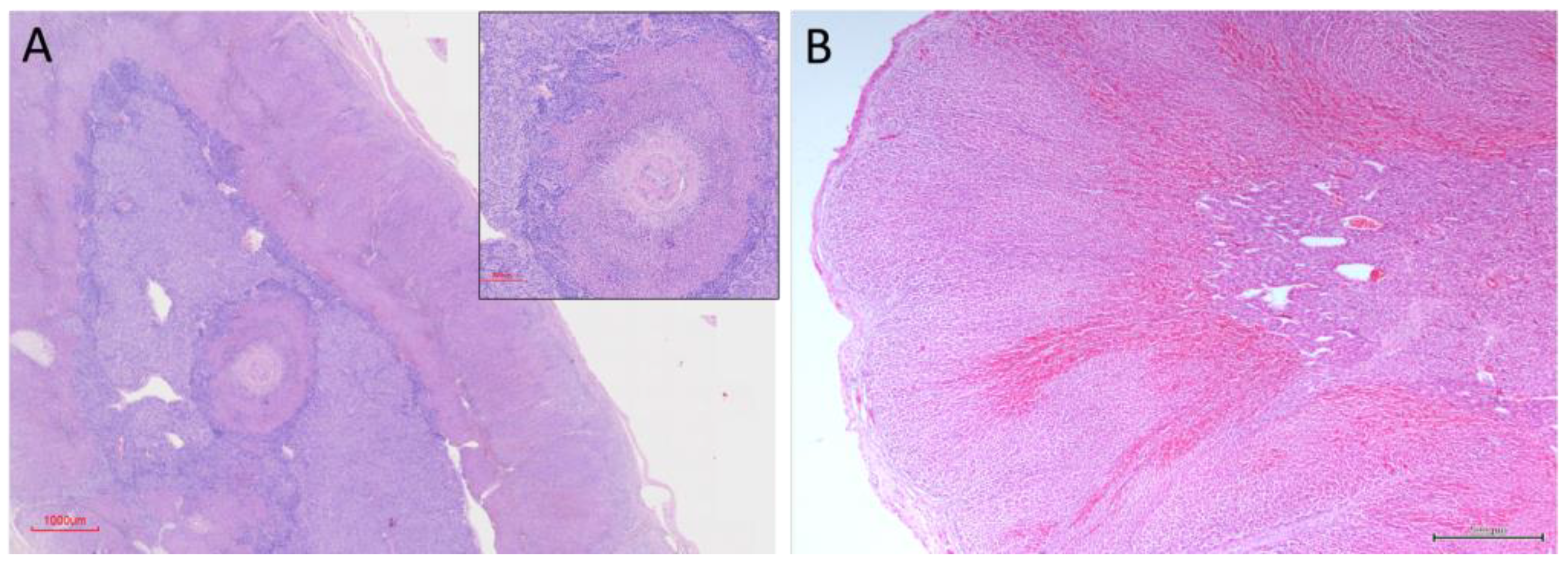
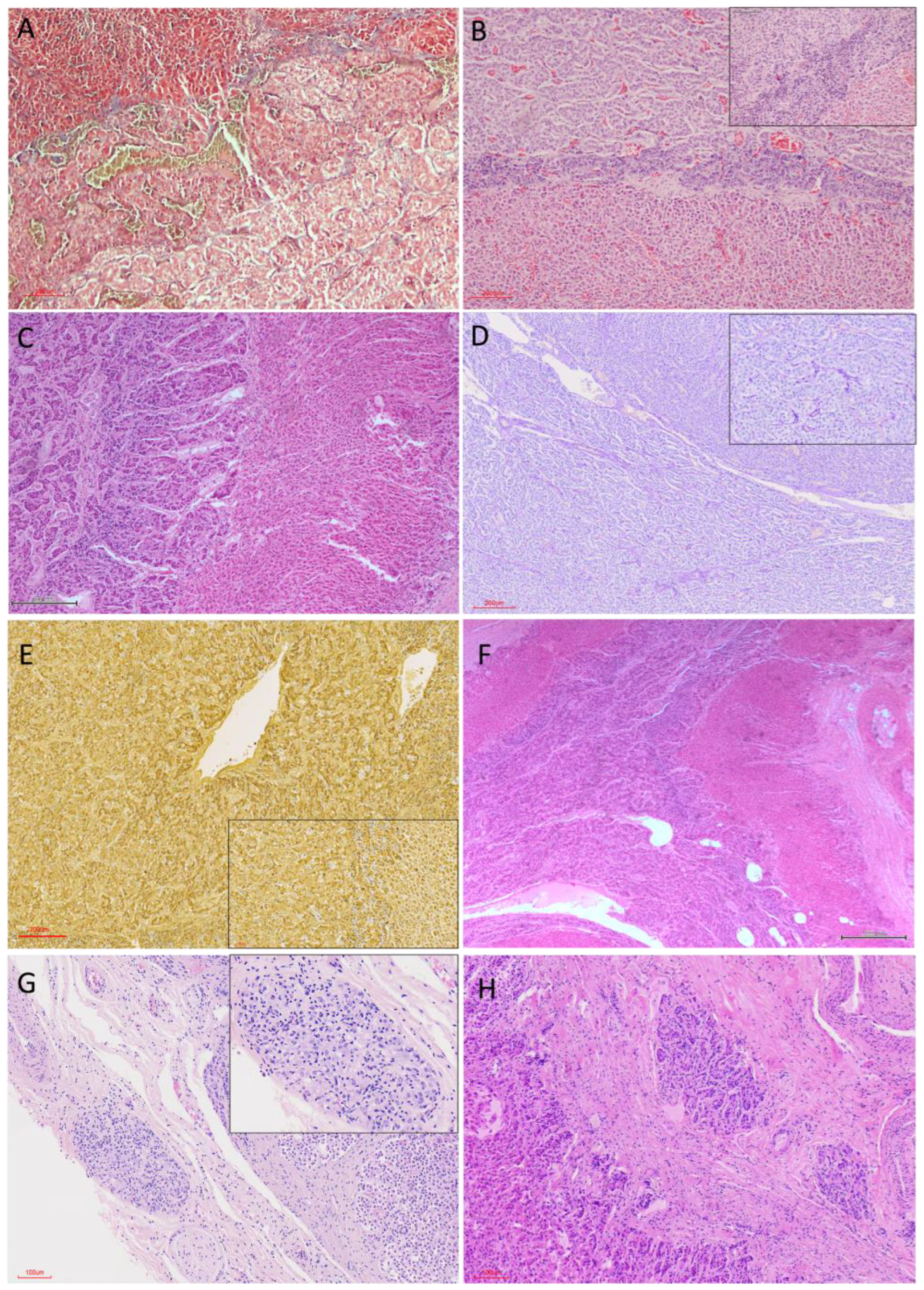
3.2. Histological Organization
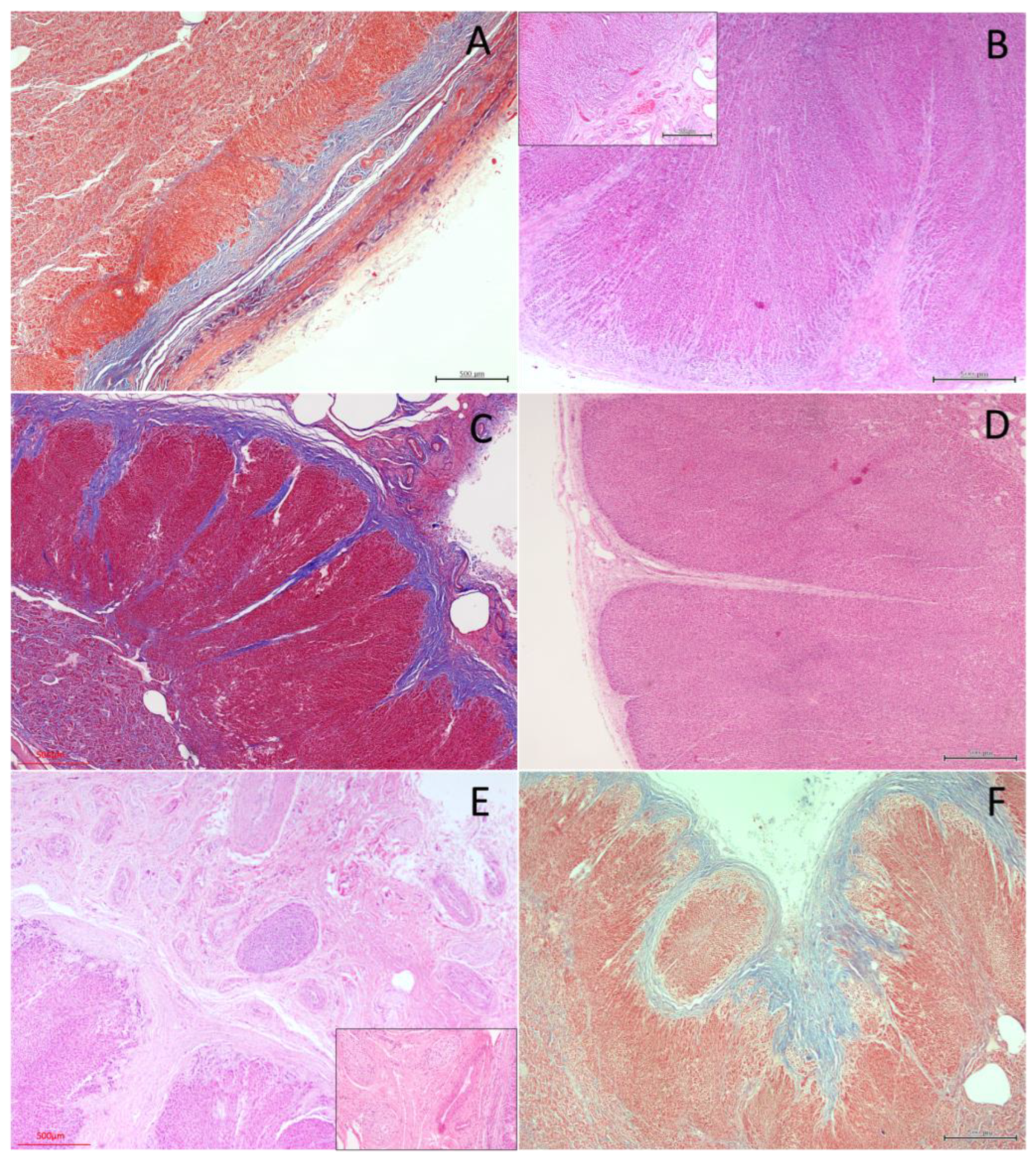
3.2.1. Capsule and Stromal Organisation
3.2.2. Cortical Architecture
3.2.3. Medullary Organisation
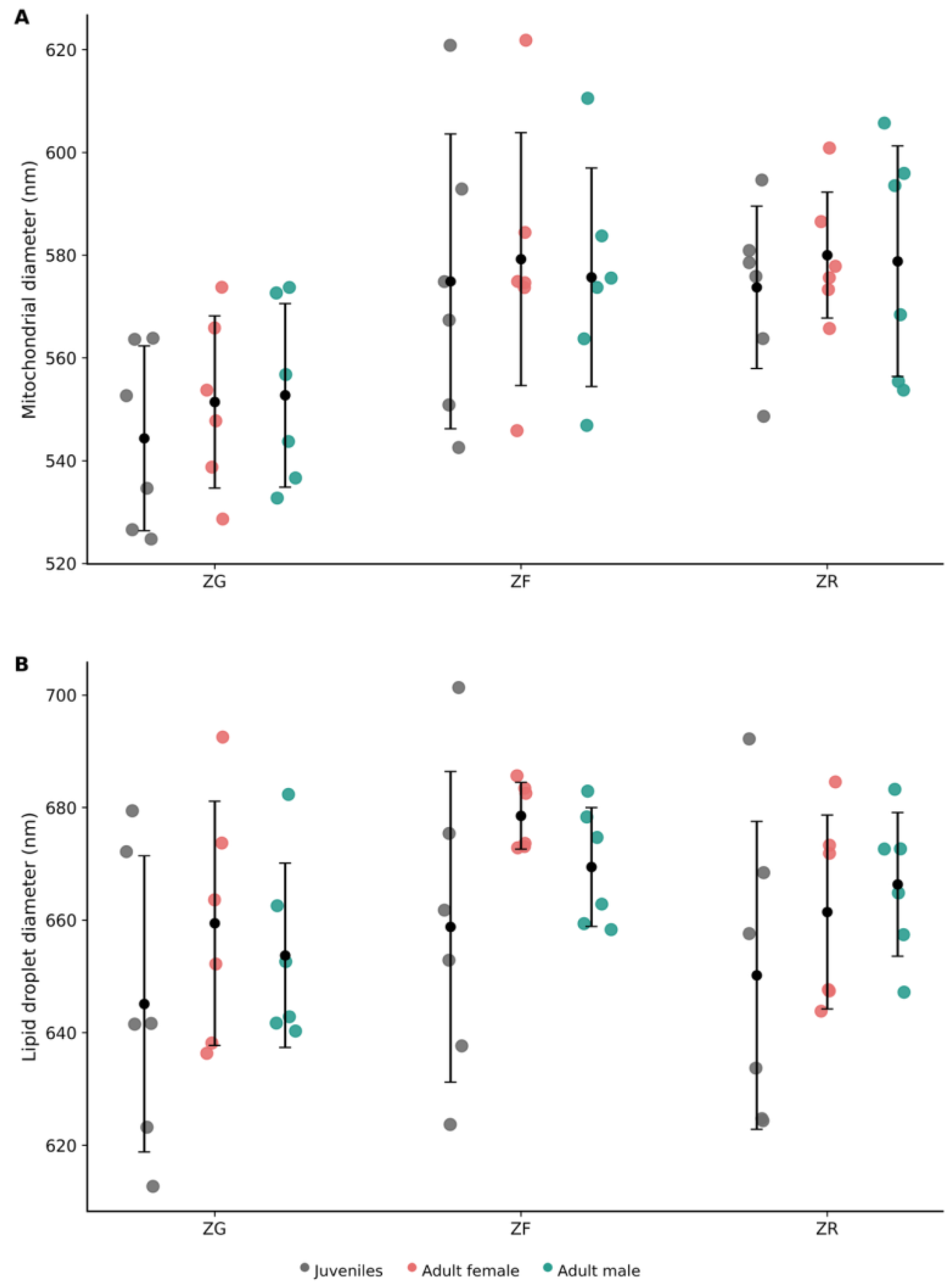
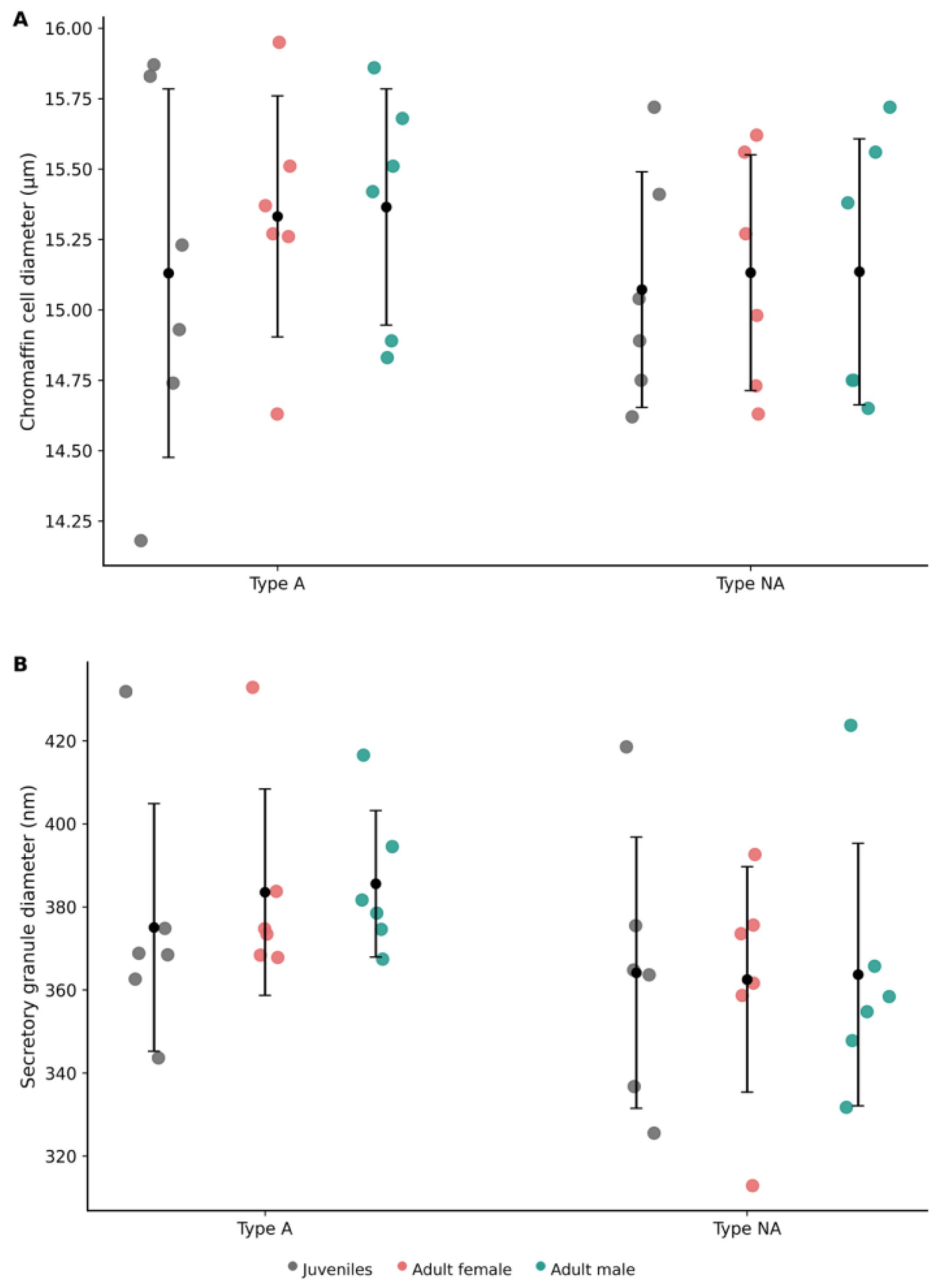
3.3. Adrenal morphometry.
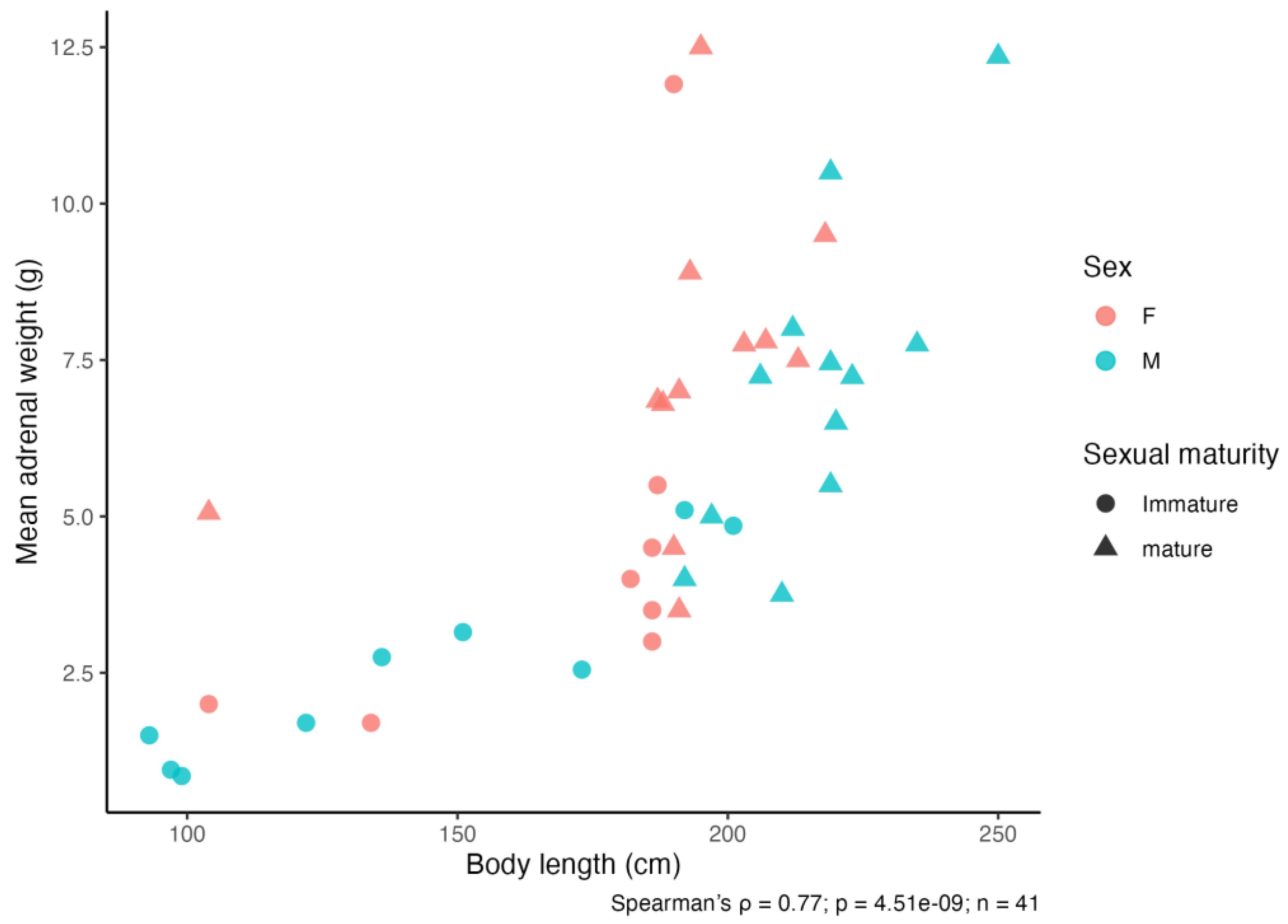
3.3.1. Adrenal Weight and Length in Sexually Mature Individuals
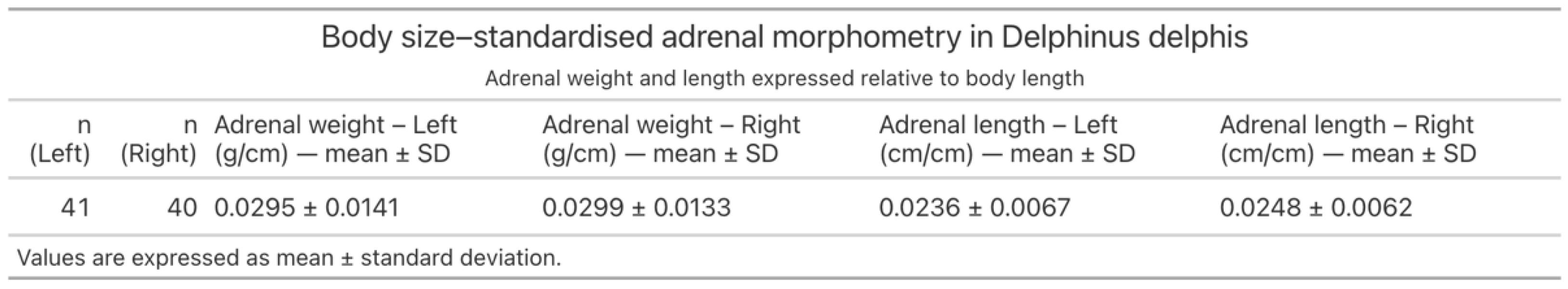
3.3.2. Body Size-Standardised Adrenal Morphometry Across Developmental Stages
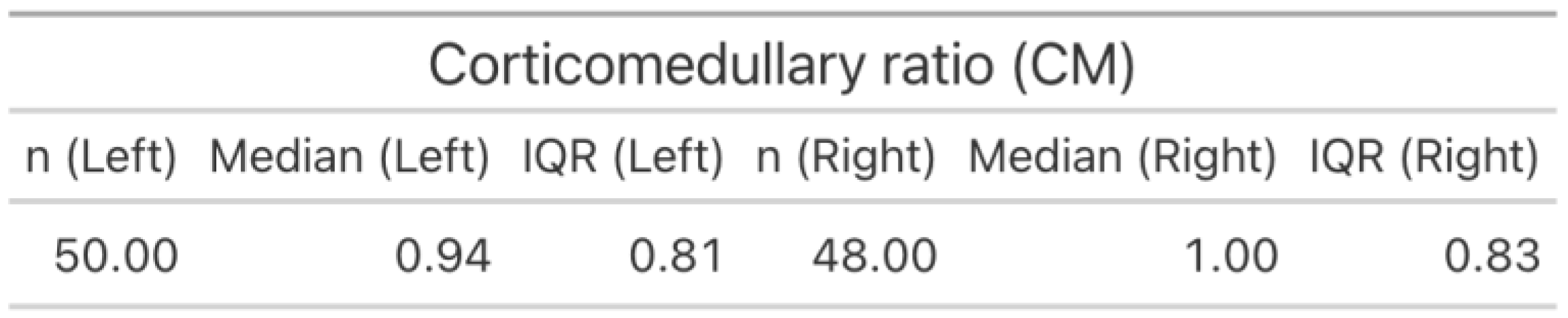
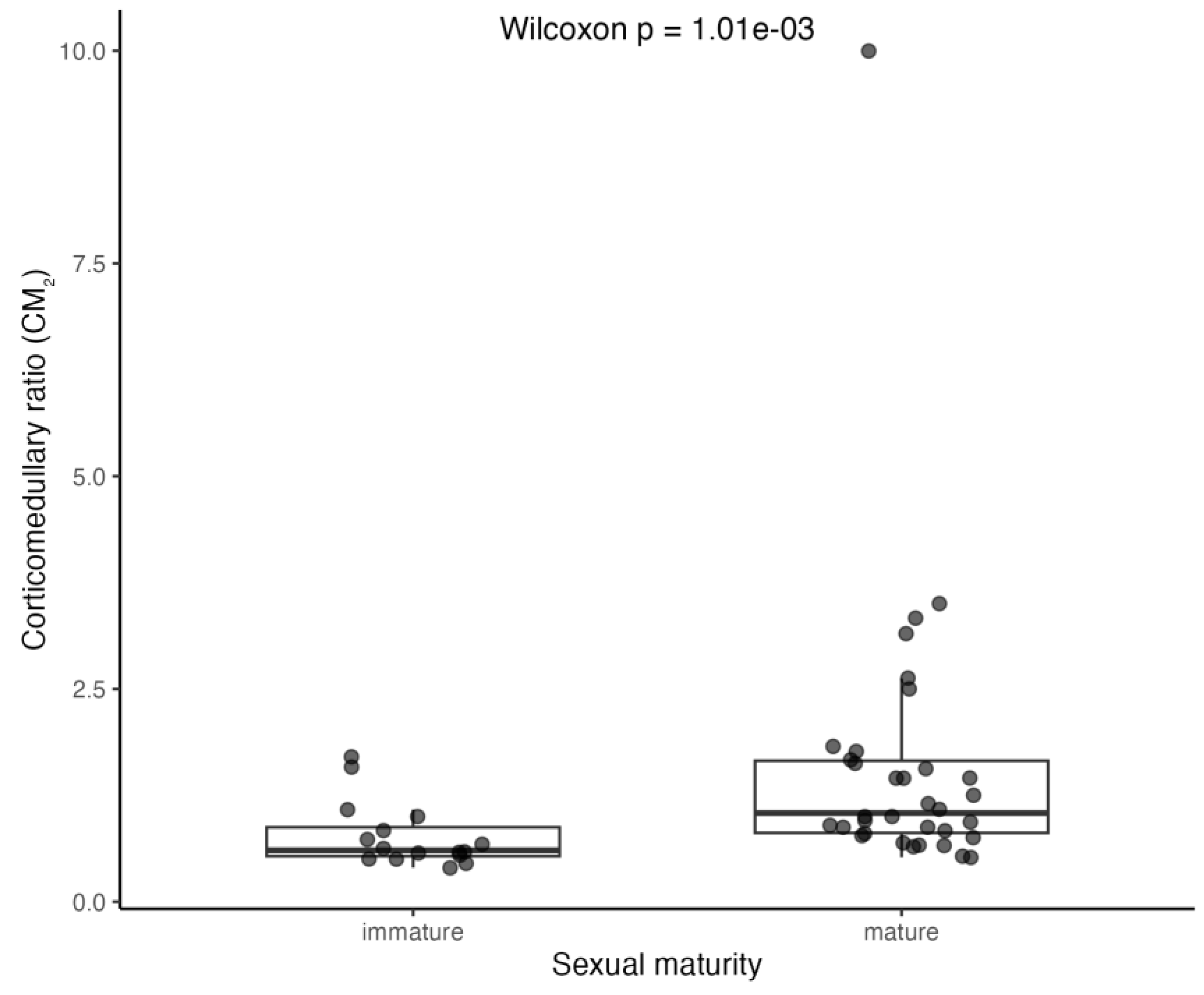
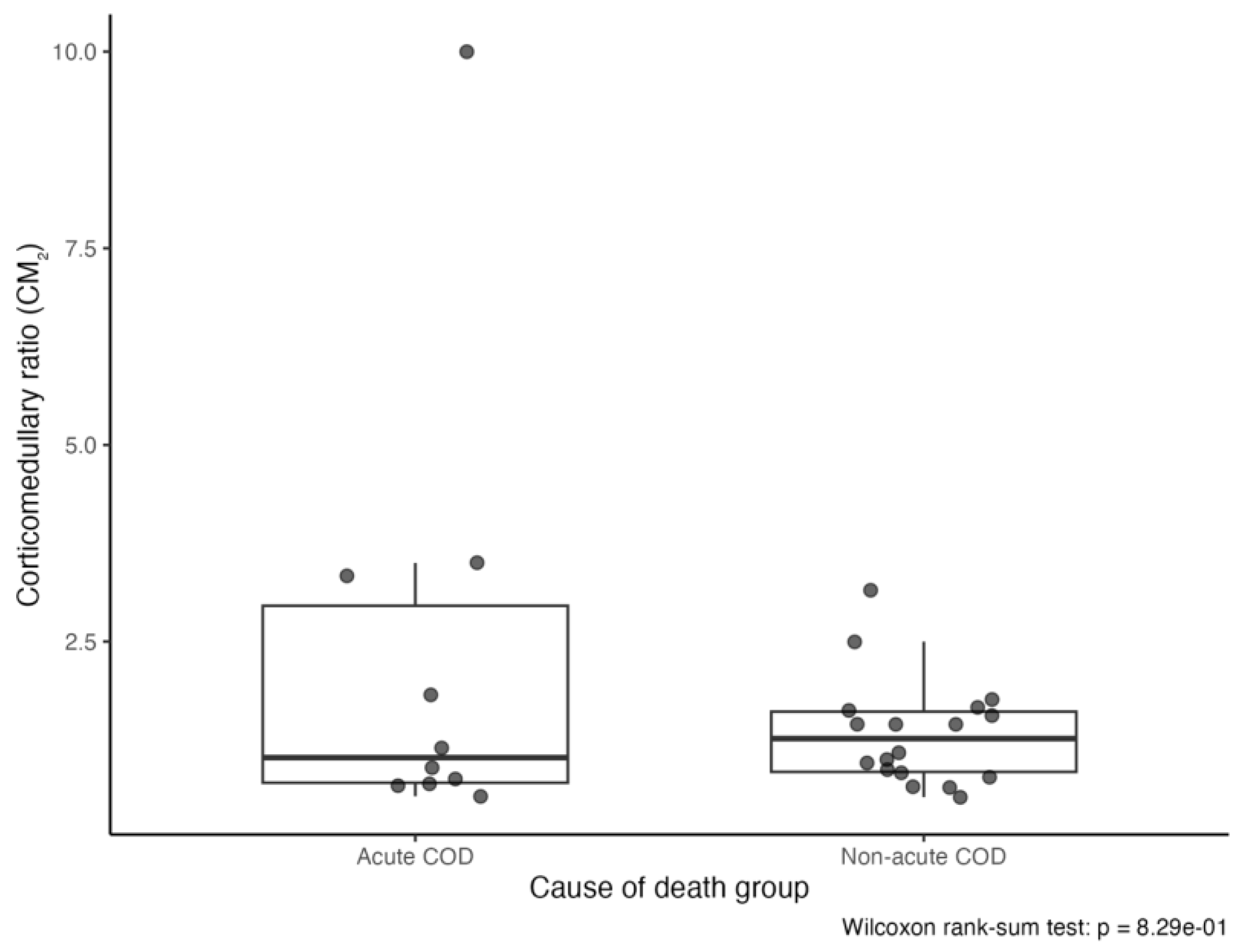
3.3.3. Corticomedullary Ratio (CM)
3.4.2. Adrenal Medulla
4. Discussion
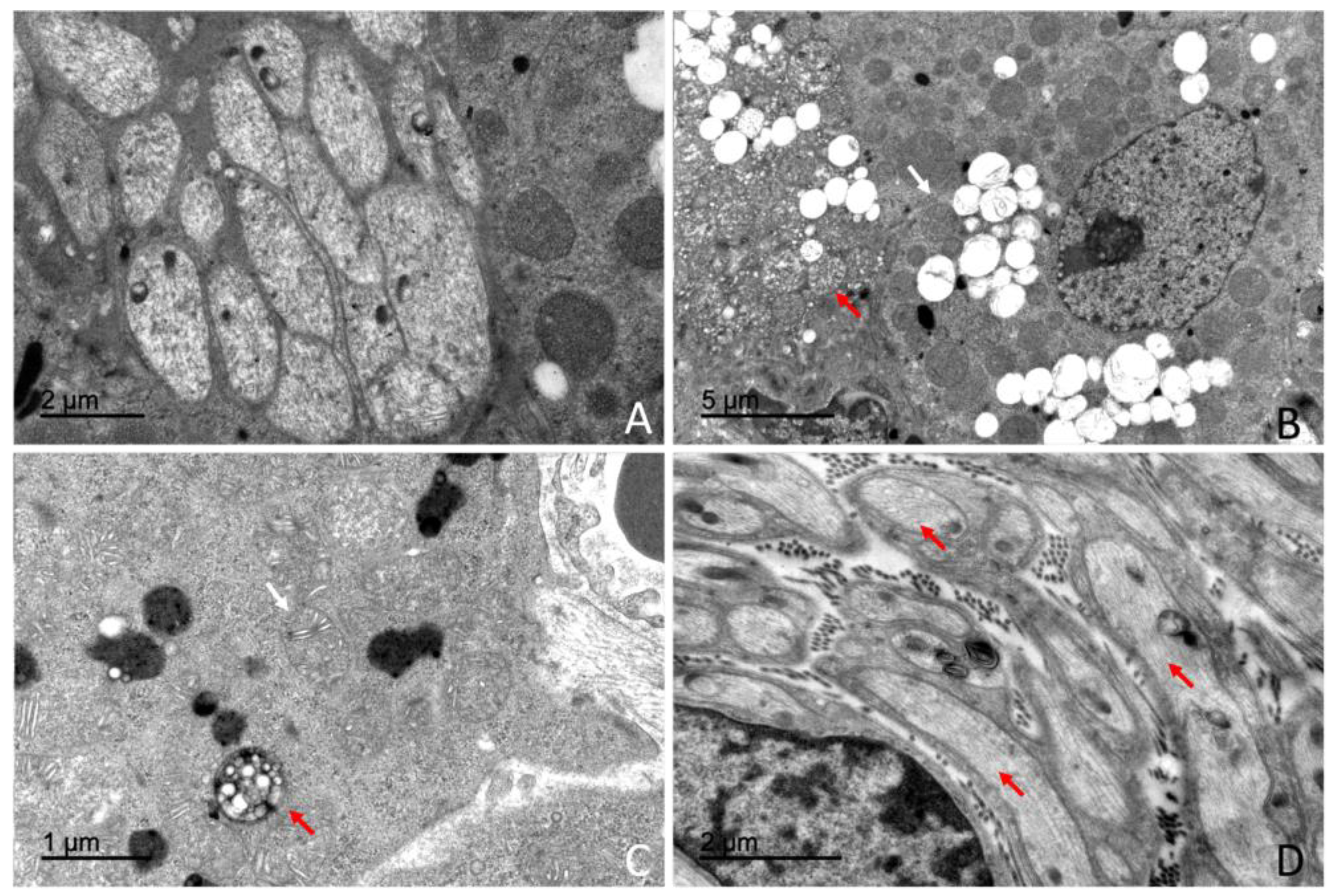
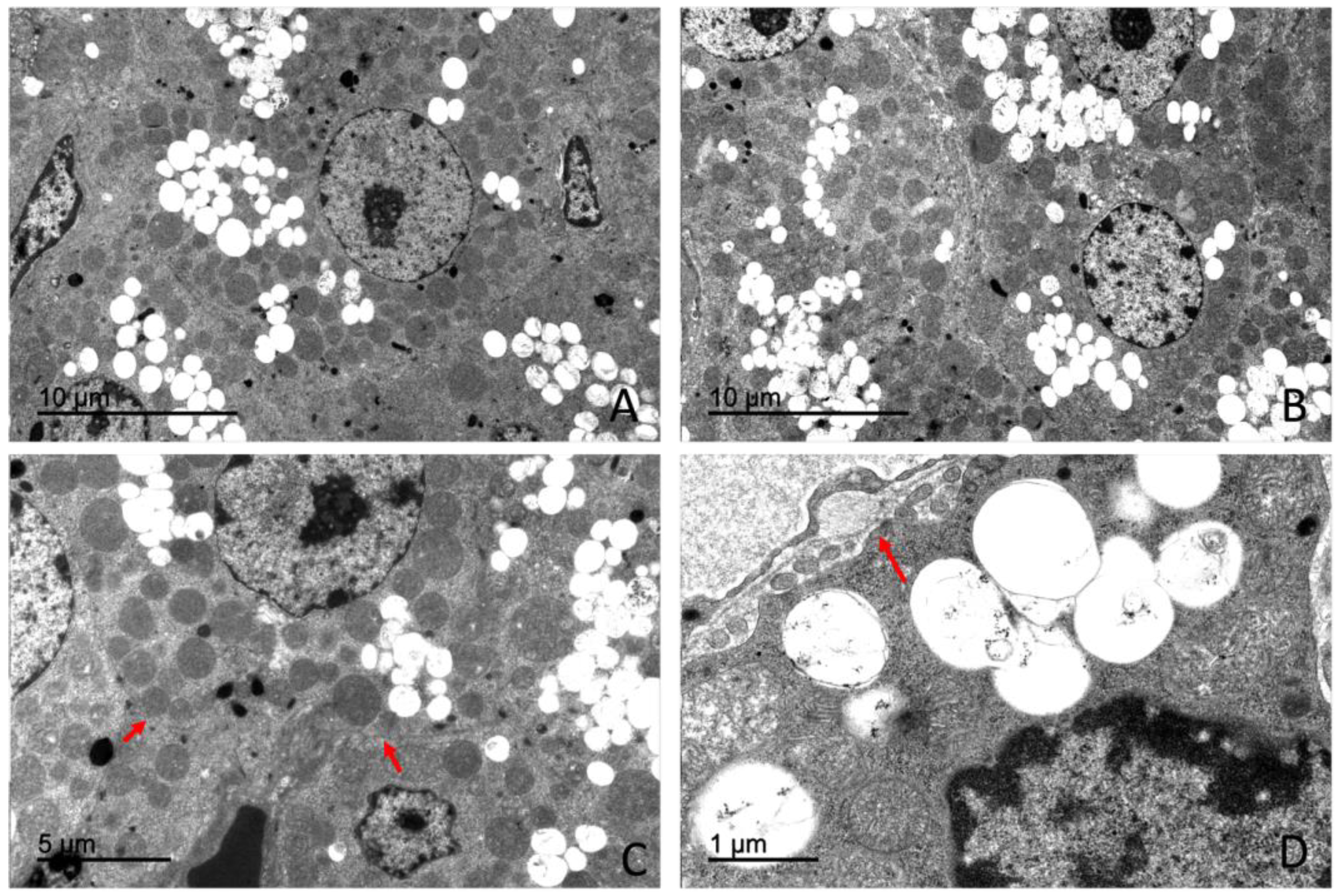
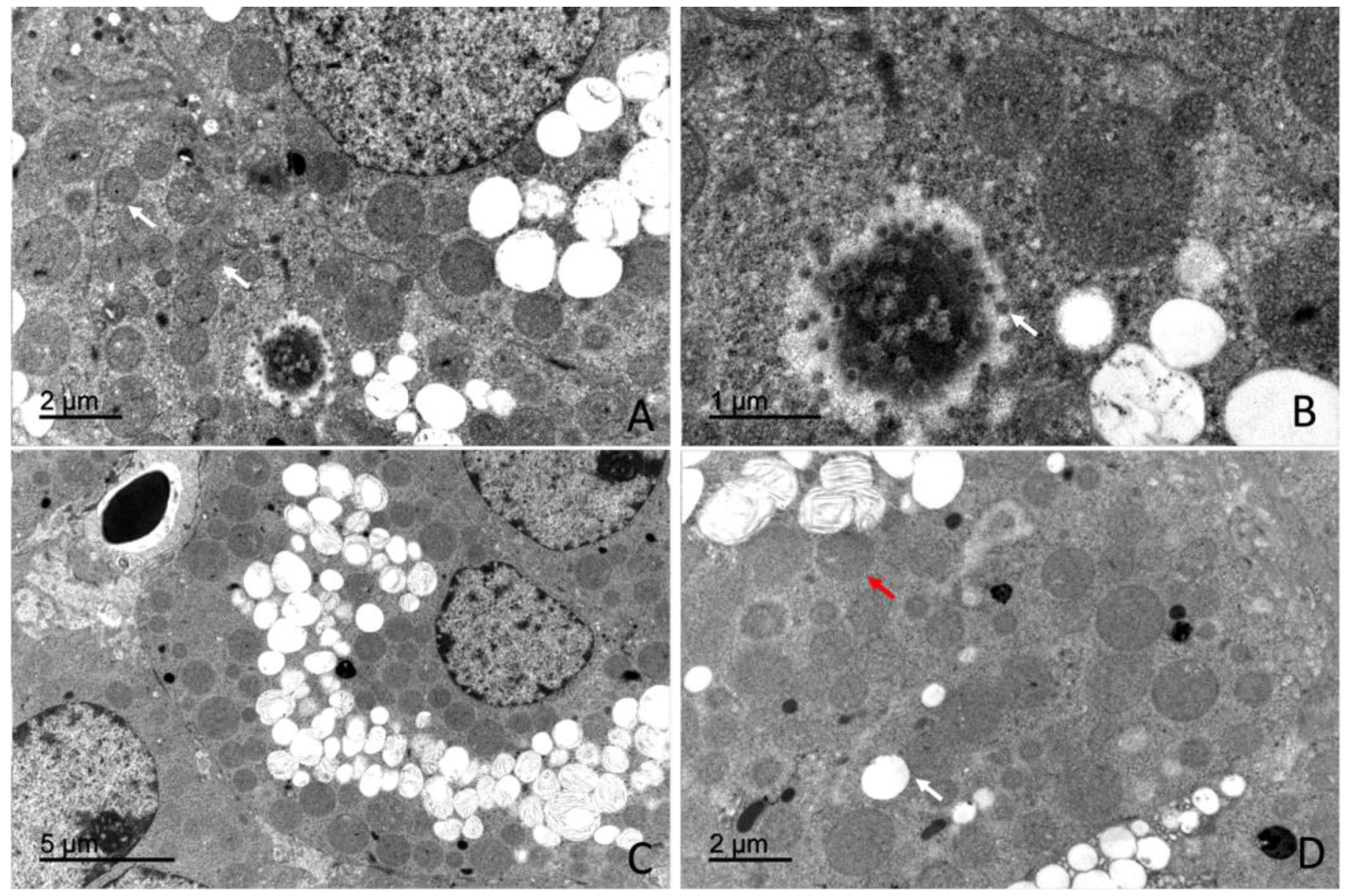
4.3. Ultrastructural Organization of the Adrenal Gland
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HPA | Hypothalamic-pituitary-adrenal |
| CM | Corticomedullary |
| COD | Cause of death |
| TEM | Transmission electron microscopy |
| IUSA | Instituto Universitario de Sanidad Animal y Seguridad Alimentaria |
| ULPGC | Universidad de Las Palmas de Gran Canaria |
| HE | Hematoxylin-eosin |
| MT | Massons’s trichrome |
| PAS | Periodic acid–Schiff |
| PAS-D | Periodic acid–Schiff-Diastase |
| IQR | Interquartile range |
| SD | Standard deviation |
| PBS | Phosphate-buffered saline |
| ZG | Zona glomerulosa |
| ZF | Zona fasciculata |
| ZR | Zona reticularis |
| A | Adrenaline |
| NA | Noradrenaline |
References
- Vogt, M. THE ROLE OF THE ADRENAL GLAND IN HOMEOSTASIS. Exp. Physiol. 1954, 39, 245–252. [Google Scholar] [CrossRef]
- De Silva, D.; Journal, B.W.-C.M. undefined The Adrenal Glands and Their Functions. cmj.sljol.info 2009. [Google Scholar]
- Peters, A.; Conrad, M.; Hubold, C.; Schweiger, U.; Fischer, B.; Fehm, H.L. The principle of homeostasis in the hypothalamus-pituitary-adrenal system: new insight from positive feedback. Am. J. Physiol. Integr. Comp. Physiol. 2007, 293, R83–R98. [Google Scholar] [CrossRef]
- Armario, A. The Hypothalamic-Pituitary-Adrenal Axis: What can it Tell us About Stressors? CNS Neurol. Disord. - Drug Targets 2006, 5, 485–501. [Google Scholar] [CrossRef] [PubMed]
- Papadimitriou, A.; Priftis, K.N. Regulation of the Hypothalamic-Pituitary-Adrenal Axis. Neuroimmunomodulation 2009, 16, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Harvey, S.; Phillips, J.G.; Rees, A.; Hall, T.R. Stress and adrenal function. J. Exp. Zoöl. 1984, 232, 633–645. [Google Scholar] [CrossRef] [PubMed]
- Möstl, E.; Palme, R. Hormones as Indicators of Stress. Domest. Anim. Endocrinol. 2002, 23, 67–74. [Google Scholar] [CrossRef]
- Ehrhart-Bornstein, M.; Bornstein, S.R. Cross-talk between Adrenal Medulla and Adrenal Cortex in Stress. Ann. New York Acad. Sci. 2008, 1148, 112–117. [Google Scholar] [CrossRef]
- Tsigos, C.; research, G.C.-J. of psychosomatic; 2002, undefined Hypothalamic–Pituitary–Adrenal Axis, Neuroendocrine Factors and Stress; Elsevier.
- Burgess, E.A.; Hunt, K.E.; Kraus, S.D.; Rolland, R.M. Adrenal responses of large whales: Integrating fecal aldosterone as a complementary biomarker to glucocorticoids. Gen. Comp. Endocrinol. 2017, 252, 103–110. [Google Scholar] [CrossRef]
- Pujade, L. Development of a Biomarker Panel for Identifying Stressed Marine Mammals; 2019. [Google Scholar]
- Santana, C.M.; Slattery, O.; O’donovan, J.; Murphy, S. Histological and Proteomic Approaches to Assessing the Adrenal Stress Response in Common Dolphins (Delphinus delphis). Animals 2025, 15, 2924. [Google Scholar] [CrossRef]
- Burgess, E.A.; Hunt, K.E.; Kraus, S.D.; Rolland, R.M. Adrenal responses of large whales: Integrating fecal aldosterone as a complementary biomarker to glucocorticoids. Gen. Comp. Endocrinol. 2017, 252, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Kuiken, T.; Höfle, U.; Bennett, P.; Allchin, C.; Kirkwood, J.; Baker, J.; Appleby, E.; Lockyer, C.; Walton, M.; Sheldrick, M. Adrenocortical hyperplasia, disease and chlorinated hydrocarbons in the harbour porpoise (Phocoena phocoena). Mar. Pollut. Bull. 1993, 26, 440–446. [Google Scholar] [CrossRef]
- Aubin, S.; J., D.; Ridgway, S.H.; Wells, R.S.; Rhinehart, H. DOLPHIN THYROID AND ADRENAL HORMONES: CIRCULATING LEVELS IN WILD AND SEMIDOMESTICATED TURSIOPS TRUNCATUS, AND INFLUENCE OF SEX, AGE, AND SEASON. Mar. Mammal Sci. 1996, 12, 1–13. [Google Scholar] [CrossRef]
- Avisse, C.; Marcus, C.; Patey, M.; Ladam-Marcus, V.; Delattre, J.-F.; Flament, J.-B. SURGICAL ANATOMY AND EMBRYOLOGY OF THE ADRENAL GLANDS. Surg. Clin. North Am. 2000, 80, 403–415. [Google Scholar] [CrossRef]
- Zwemer, R.L. A STUDY OF ADRENAL CORTEX MORPHOLOGY. 1936, 84, 79–U36. [Google Scholar] [CrossRef]
- Neville, A.M.; O'HAre, M. Histopathology of the human adrenal cortex. Clin. Endocrinol. Metab. 1985, 14, 791–820. [Google Scholar] [CrossRef]
- Gorgas, K.; Böck, P. Morphology and Histochemistry of the Adrenal Medulla: I. Various Types of Primary Catecholamine-Storing Cells in the Mouse Adrenal Medulla. Histochemistry 1976, 50, 17–31. [Google Scholar] [CrossRef]
- Siasios, A.; Delis, G.; Tsingotjidou, A.; Pourlis, A.; Grivas, I. Adrenal glands of mice and rats: A comparative morphometric study. Lab. Anim. 2021, 56, 247–258. [Google Scholar] [CrossRef]
- Milano, E.G.; Basari, F.; Chimenti, C. Adrenocortical and Adrenomedullary Homologs in Eight Species of Adult and Developing Teleosts: Morphology, Histology, and Immunohistochemistry. Gen. Comp. Endocrinol. 1997, 108, 483–496. [Google Scholar] [CrossRef]
- Barszcz, K.; Przespolewska, H.; Olbrych, K.; Czopowicz, M.; Klećkowska-Nawrot, J.; Goździewska-Harłajczuk, K.; Kupczyńska, M. The morphology of the adrenal gland in the European bison (Bison bonasus). BMC Veter- Res. 2016, 12, 1–11. [Google Scholar] [CrossRef]
- Zhongjie, L. The Adrenal Gland of Chinese River Dolphin (Lipotes Vexillifer). Acta Hydrobiol. Sinica 1988, 12, 59–64. [Google Scholar]
- Bourne, G.H. Some observations on the mammalian adrenal gland. 1949, 83, 70. [Google Scholar]
- Jacobsen, A.P. Endocrinological Studies in the Blue Whale:(Balaenoptera Musculus L.); I kommisjon hos Jacob Dybwad; 1941. [Google Scholar]
- Vuković, S.; Lucić, H.; Živković, A.; Gomerčić, M.Đ.; Gomerčić, T.; Galov, A. Histological Structure of the Adrenal Gland of the Bottlenose Dolphin (Tursiops truncatus) and the Striped Dolphin (Stenella coeruleoalba) from the Adriatic Sea. Anat. Histol. Embryol. 2010, 39, 59–66. [Google Scholar] [CrossRef]
- Clark, L.S.; Pfeiffer, D.C.; Cowan, D.F. Morphology and Histology of the Atlantic Bottlenose Dolphin (Tursiops truncatus) Adrenal Gland with Emphasis on the Medulla. Anat. Histol. Embryol. 2005, 34, 132–140. [Google Scholar] [CrossRef]
- Clark, L.S.; Cowan, D.F.; Pfeiffer, D.C. A Morphological and Histological Examination of the Pan-tropical Spotted Dolphin (Stenella attenuata) and the Spinner Dolphin (Stenella longirostris) Adrenal Gland. Anat. Histol. Embryol. 2007, 37, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Carballeira, A.; Brown, J.W.; Fishman, L.M.; Trujillo, D.; Odell, D.K. The adrenal gland of stranded whales (Kogia breviceps and Mesoplodon europaeus): Morphology, hormonal contents, and biosynthesis of corticosteroids. Gen. Comp. Endocrinol. 1987, 68, 293–303. [Google Scholar] [CrossRef] [PubMed]
- Simpson, J.G.; Gardner, M.B. Comparative Microscopic Anatomy of Selected Marine Mammals; 1972. [Google Scholar]
- Clark, L.; Cowan, D.; Pfeiffer, D. Morphological Changes in the Atlantic Bottlenose Dolphin (Tursiops truncatus) Adrenal Gland Associated with Chronic Stress. J. Comp. Pathol. 2006, 135, 208–216. [Google Scholar] [CrossRef]
- Jefferson, T.A.; Fertl, D.; Bolaños-Jiménez, J.; Zerbini, A.N. Distribution of common dolphins (Delphinus spp.) in the western Atlantic Ocean: a critical re-examination. Mar. Biol. 2009, 156, 1109–1124. [Google Scholar] [CrossRef]
- Selzer, L.A.; Payne, P.M. THE DISTRIBUTION OF WHITE-SIDED (LAGENORHYNCHUS ACUTUS) AND COMMON DOLPHINS (DELPHINUS DELPHIS) VS. ENVIRONMENTAL FEATURES OF THE CONTINENTAL SHELF OF THE NORTHEASTERN UNITED STATES. Mar. Mammal Sci. 1988, 4, 141–153. [Google Scholar] [CrossRef]
- Vuković, S.; Lucić, H.; Đuras Gomerčić, M.; Galov, A.; Gomerčić, T.; Ćurković, S.; Škrtić, D.; Domitran, G.; Gomerčić, H. Anatomical and Histological Characteristics of the Pituitary Gland in the Bottlenose Dolphin ( Tursiops Truncatus ) from the Adriatic Sea. the Adriatic Sea. Vet. arhiv 2011, 81, 143–151. [Google Scholar]
- Alonso-Almorox, P.; Blanco, A.; Fiorito, C.; Gómez-Villamandos, J.C.; Risalde, M.A.; Almunia, J.; Fernández, A. The orca (Orcinus orca) pituitary gland: an anatomical, immunohistochemical and ultrastructural analysis. Front. Neuroanat. 2025, 19, 1626079. [Google Scholar] [CrossRef]
- Alonso-Almorox, P.; Blanco, A.; Fiorito, C.; Sierra, E.; Suárez-Santana, C.; Consolli, F.; Arbelo, M.; Guzmán, R.G.; Molpeceres-Diego, I.; Gómez, A.F.; et al. Dolphin Pituitary Gland: Immunohistochemistry and Ultrastructural Cell Characterization Following a Novel Anatomical Dissection Protocol and Non-Invasive Imaging (MRI). Animals 2025, 15, 735. [Google Scholar] [CrossRef]
- Kuiken, T.; García-Hartmann, M. Proceedings of the First European Cetacean Society Workshop on Cetacean Pathology: Dissection Techniques and Tissue Sampling. In Proceedings of the ECS Workshop on Cetacean Pathology, 1991; p. 20. [Google Scholar]
- Reynolds, J.E.; Rommel.
- Rommel, S.A.; L. J. Lowenstine Gross and Microscopic Anatomy. CRC Hanbook of Marine Mammal Medicine 2001. [Google Scholar]
- Jelinek, F.; Konecny, R. Adrenal Glands of Slaughtered Bulls, Heifers and Cows: A Histological Study. Anat. Histol. Embryol. 2010, 40, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Cowan, D.F. Observations on the pilot whale Globicephala melaena: Organweight and growth. Anat. Rec. 1966, 155, 623–628. [Google Scholar] [CrossRef]
- Nicolaides, N.C.; Willenberg, H.S.; Bornstein, S.R.; Chrousos, G.P. Adrenal Cortex: Embryonic Development, Anatomy, Histology and Physiology; Endotext, 2023. [Google Scholar]
- Neville, A.; metabolism, M.O.-C. in endocrinology and; 1985, undefined Histopathology of the Human Adrenal Cortex; Elsevier.
- Anderson, J.; journal, A.R.-P. medical; 1980, undefined Ectopic Adrenal Tissue in Adults. Available online: pmc.ncbi.nlm.nih.govJR.
- Schmidt, M.; Enthoven, L.; van der Mark, M.; Levine, S.; de Kloet, E.; Oitzl, M. The postnatal development of the hypothalamic–pituitary–adrenal axis in the mouse. Int. J. Dev. Neurosci. 2003, 21, 125–132. [Google Scholar] [CrossRef]
- Fenwick, E.M.; Fajdiga, P.B.; Howe, N.B.; Livett, B.G. Functional and morphological characterization of isolated bovine adrenal medullary cells. J. Cell Biol. 1978, 76, 12–30. [Google Scholar] [CrossRef]
- Coupland, R.; Anatomy, B.W.-J. of; 1970, undefined Electron Microscopic Observation on the Adrenal Medulla and Extra-Adrenal Chromaffin Tissue of the Postnatal Rabbit. Available online: pmc.ncbi.nlm.nih.govRE.
- E Coupland, R. ELECTRON MICROSCOPIC OBSERVATIONS ON STRUCTURE OF RAT ADRENAL MEDULLA.I. ULTRASTRUCTURE AND ORGANIZATION OF CHROMAFFIN CELLS IN NORMAL ADRENAL MEDULLA. 1965, 99, 231–+. [Google Scholar]
- Bornstein, S.R.; A Gonzalez-Hernandez, J.; Ehrhart-Bornstein, M.; Adler, G.; A Scherbaum, W. Intimate contact of chromaffin and cortical cells within the human adrenal gland forms the cellular basis for important intraadrenal interactions. J. Clin. Endocrinol. Metab. 1994, 78, 225–232. [Google Scholar] [CrossRef]
- Bornstein, S.R.; Ehrhart-Bornstein, M.; Scherbaum, W.A. Morphological and Functional Studies of the Paracrine Interaction between Cortex and Medulla in the Adrenal Gland. Microsc. Res. Tech. 1997, 36, 520–533. [Google Scholar] [CrossRef]
- Mitani, F.; Suzuki, H.; Hata, J.; Ogishima, T.; Shimada, H.; Ishimura, Y. A novel cell layer without corticosteroid-synthesizing enzymes in rat adrenal cortex: histochemical detection and possible physiological role. Endocrinology 1994, 135, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, B.; Zoology, M.; K.-A. undefined The Adrenal Gland of the African Buffalo, Syncerus Caffer: A Light and Electron Microscopic Study. journals.co.za 1993. [Google Scholar] [CrossRef]
- Olukole, S.G.; Adeagbo, M.A.; Oke, B.O. Histology and Histochemistry of the Adrenal Gland African Giant Rat (Cricetomys gambianus, Waterhouse). Int. J. Morphol. 2016, 34, 1455–1460. [Google Scholar] [CrossRef]
- Neville, A.; O’Hare, M. The Human Adrenal Cortex: Pathology and Biology—an Integrated Approach; 2012. [Google Scholar]
- Weiss, C.; Cahill, A.; Laslop, A.; Fischer-Colbrie, R.; Perlman, R.; Winkler, H. Differences in the composition of chromaffin granules in adrenaline and noradrenaline containing cells of bovine adrenal medulla. Neurosci. Lett. 1996, 211, 29–32. [Google Scholar] [CrossRef]
- Carbone, E.; Borges, R.; Eiden, L.E.; García, A.G.; Hernández-Cruz, A. Chromaffin Cells of the Adrenal Medulla: Physiology, Pharmacology, and Disease. Compr. Physiol. 2019, 9, 1443–1502. [Google Scholar] [CrossRef]
- Paul, B.; I.M.N., S.S.; D.R. Morphological and Histological Investigations on the Adrenal Glands in Black Bengal Goat (Capra Hircus). Journal of the Sylhet Agricultural University 2016, 3, 181–187. [Google Scholar]
- Nicolaides, N.C.; Willenberg, H.S.; Bornstein, S.R.; Chrousos, G.P. Adrenal Cortex: Embryonic Development, Anatomy, Histology and Physiology; Endotext, 2023. [Google Scholar]
- Abdellatif, A.B.; Fernandes-Rosa, F.L.; Boulkroun, S.; Zennaro, M.-C. Vascular and hormonal interactions in the adrenal gland. Front. Endocrinol. 2022, 13, 995228. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Sanchez, C.E. Regulation of Adrenal Arterial Tone by Adrenocorticotropin: The Plot Thickens. Endocrinology 2007, 148, 3566–3568. [Google Scholar] [CrossRef] [PubMed]
- Vinson, G.P.; Pudney, J.A.; Whitehouse, B.J. The mammalian adrenal circulation and the relationship between adrenal blood flow and steroidogenesis. J. Endocrinol. 1985, 105, 285–94. [Google Scholar] [CrossRef]
- Breed, D.; Meyer, L.C.R.; A Steyl, J.C.; Goddard, A.; Burroughs, R.; A Kohn, T. Conserving wildlife in a changing world: Understanding capture myopathy—a malignant outcome of stress during capture and translocation. Conserv. Physiol. 2019, 7, coz027. [Google Scholar] [CrossRef]
- Turner, J.P.; Clark, L.S.; Haubold, E.M.; Worthy, G.A.J.; Cowan, D.F. Organ Weights and Growth Profiles in Bottlenose Dolphins (Tursiops truncatus) from the Northwestern Gulf of Mexico. Aquat. Mamm. 2006, 32, 46–57. [Google Scholar] [CrossRef]
- Venn-Watson, S.; Colegrove, K.M.; Litz, J.; Kinsel, M.; Terio, K.; Saliki, J.; Fire, S.; Carmichael, R.; Chevis, C.; Hatchett, W.; et al. Adrenal Gland and Lung Lesions in Gulf of Mexico Common Bottlenose Dolphins (Tursiops truncatus) Found Dead following the Deepwater Horizon Oil Spill. PLOS ONE 2015, 10, e0126538–e0126538. [Google Scholar] [CrossRef]
- Lair, S.; Béland, P.; De Guise, S.; Martineau, D. ADRENAL HYPERPLASTIC AND DEGENERATIVE CHANGES IN BELUGA WHALES. J. Wildl. Dis. 1997, 33, 430–437. [Google Scholar] [CrossRef]
- Martí, O.; Gavaldà, A.; Jolín, T.; Armario, A. Effect of regularity of exposure to chronic immobilization stress on the circadian pattern of pituitary adrenal hormones, growth hormone, and thyroid stimulating hormone in the adult male rat. Psychoneuroendocrinology 1993, 18, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Ulrich-Lai, Y.M.; Figueiredo, H.F.; Ostrander, M.M.; Choi, D.C.; Engeland, W.C.; Herman, J.P. Chronic stress induces adrenal hyperplasia and hypertrophy in a subregion-specific manner. Am. J. Physiol. Metab. 2006, 291, E965–E973. [Google Scholar] [CrossRef]
- Koko, V.; Djordjeviæ, J.; Cvijiæ, G.; Davidoviæ, V. Effect of acute heat stress on rat adrenal glands: a morphological and stereological study. J. Exp. Biol. 2004, 207, 4225–4230. [Google Scholar] [CrossRef] [PubMed]
- Berger, I.; Werdermann, M.; Bornstein, S.R.; Steenblock, C. The adrenal gland in stress – Adaptation on a cellular level. J. Steroid Biochem. Mol. Biol. 2019, 190, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Spencer, K.A. Developmental stress and social phenotypes: integrating neuroendocrine, behavioural and evolutionary perspectives. Philos. Trans. R. Soc. B: Biol. Sci. 2017, 372, 20160242. [Google Scholar] [CrossRef]
- DeRijk, R.; de Kloet, E.R. Corticosteroid Receptor Genetic Polymorphisms and Stress Responsivity. Endocrine 2005, 28, 263–270. [Google Scholar] [CrossRef]
- Charmandari, E. Primary Generalized Glucocorticoid Resistance and Hypersensitivity: The End-Organ Involvement in the Stress ResponseA Presentation from the European Society for Paediatric Endocrinology (ESPE) New Inroads to Child Health (NICHe) Conference on Stress Response and Child Health in Heraklion, Crete, Greece, 18 to 20 May 2012. Sci. Signal. 2012, 5, pt5. [Google Scholar] [CrossRef]
- Kinlein, S.A.; Karatsoreos, I.N. The hypothalamic-pituitary-adrenal axis as a substrate for stress resilience: Interactions with the circadian clock. Front. Neuroendocr. 2020, 56, 100819–100819. [Google Scholar] [CrossRef]
- Herman, J.P.; McKlveen, J.M.; Ghosal, S.; Kopp, B.; Wulsin, A.; Makinson, R.; Scheimann, J.; Myers, B. Regulation of the Hypothalamic-Pituitary-Adrenocortical Stress Response. Compr. Physiol. 2016, 6, 603–621. [Google Scholar] [CrossRef] [PubMed]
- Yiallouris, A.; Filippou, C.; Themistocleous, S.C.; Menelaou, K.; Kalodimou, V.; Michaeloudes, C.; Johnson.
- Li, N.; Li, Y.; Lu, Y.; Zhang, K.; Wang, S.; Wang, C. The impact of chronic stress on cortical thickness in patients with depression. Front. Psychiatry 2025, 16, 1554476. [Google Scholar] [CrossRef]
- Ishimura, K.; Fujita, H. Light and electron microscopic immunohistochemistry of the localization of adrenal steroidogenic enzymes. Microsc. Res. Tech. 1997, 36, 445–453. [Google Scholar] [CrossRef]
- Kawaoi, A. ULTRASTRUCTURAL ZONATION OF THE HUMAN ADRENAL CORTEX. Acta Pathol. Jpn. 1969, 19, 115–149. [Google Scholar] [CrossRef] [PubMed]
- Pathologie, E.M.-B. zur. undefined Ultrastructure and Function of the Mesenchyme of the Rat Adrenal Cortex; Elsevier, 1974. [Google Scholar]
- Dickens, R. An Ultrastructural and Morphometric Study of the Adrenal Cortex in the Intact and Hypophysectomized Male Rat; 1975. [Google Scholar]
- Pihlajoki, M.; Dã¶Rner, J.; Cochran, R.S.; Heikinheimo, M.; Wilson, D.B. Adrenocortical Zonation, Renewal, and Remodeling. Front. Endocrinol. 2015, 6, 27–27. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, S.; American, H.W.-S. 1985, undefined The Adrenal Chromaffin Cell. In JSTORSW Carmichael, H WinklerScientific American; 1985. [Google Scholar]
- Neurosciences, H.W.-C. in the; 1980, undefined The Composition of Adrenal Chromaffin Granules: An Assessment of Controversial Results; Elsevier.
| Cell type | Juveniles | Adult female | Adult males |
|---|---|---|---|
| Zona glomerulosa | 16.34 ± 0.46 | 16.61 ± 0.98 | 16.48 ± 0.62 |
| Zona fasciculata | 16.66 ± 0.79 | 16.75 ± 0.51 | 16.81 ± 0.78 |
| Zona reticularis | 16.06 ± 0.36 | 16.20 ± 0.62 | 16.16 ± 0.51 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




