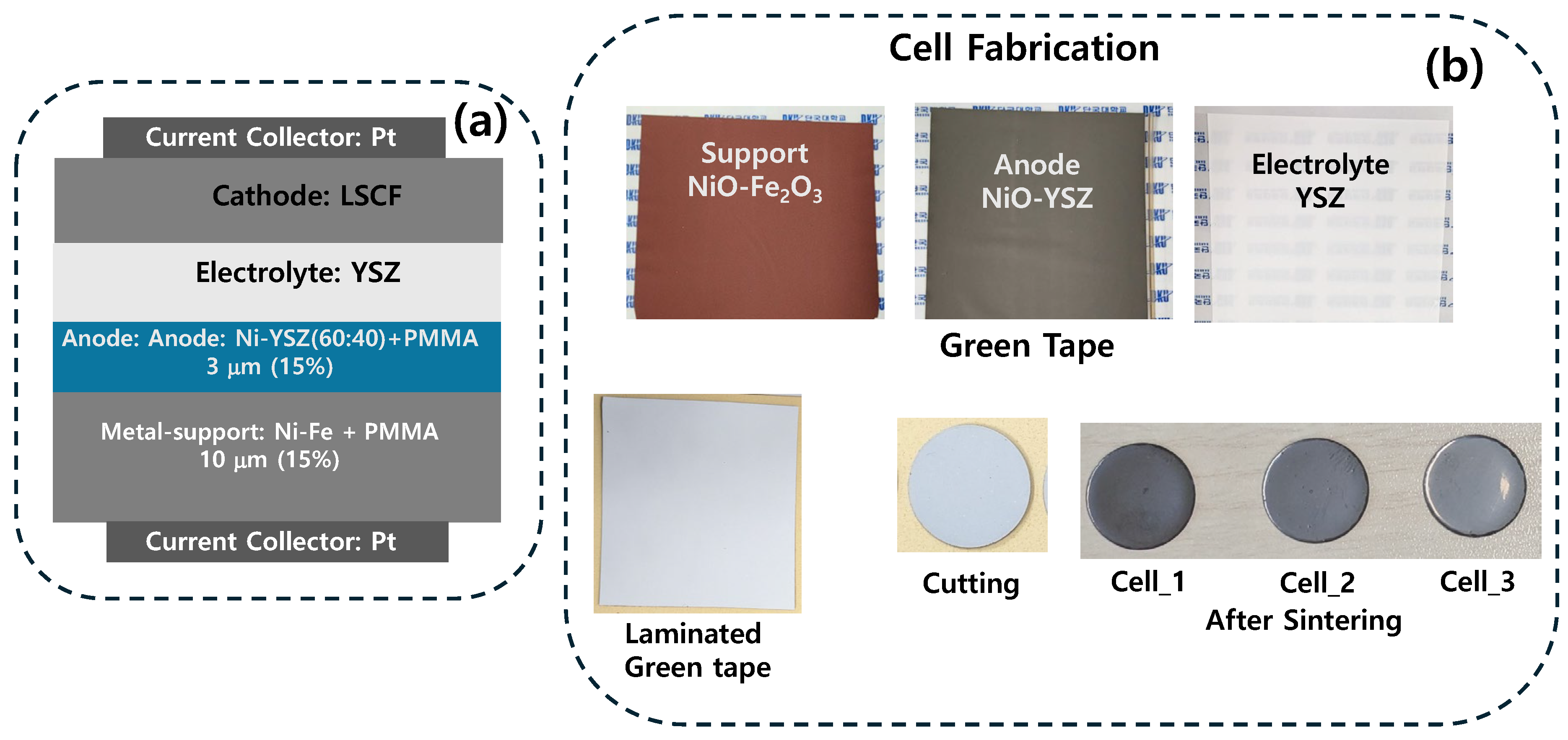
3. Results and Discussions
Figure 2 presents cross-sectional SEM micrographs of the three fabricated NiFe-supported SOFC cells, demonstrating successful co-sintering and consistent microstructure across different electrolyte thicknesses. All three cells exhibit well-defined, distinct layers with clear interfaces between the support, anode, electrolyte, and cathode components. The layered architecture is maintained consistently across all samples, confirming the effectiveness of the tape casting and co-sintering approach for producing high-quality MS-SOFC structures.
The NiFe support shows uniform porosity with interconnected pore networks essential for gas transport. The pore structure appears consistent across all cells, with pore sizes ranging from 8 to 10 microns, created by the burnout of PMMA pore former during sintering. The support thickness is maintained at 280 μm for all samples.
The Ni-YSZ anode layer shows appropriate porosity for fuel transport while maintaining good connectivity with both the support and electrolyte layers. The anode thickness remains consistent at 21 μm across all samples, ensuring comparable electrochemical behavior. The microstructure reveals well-distributed Ni and YSZ phases with sufficient percolation for both electronic and ionic conduction.
The YSZ electrolyte layers exhibit dense, crack-free microstructures with excellent gas-tightness across all thickness variations. Cell_1 (7.05 μm) displays a uniform thin electrolyte with high density, Cell_2 (14.1 μm) maintains intermediate thickness while preserving structural integrity, and Cell_3 (21.2 μm) shows the thickest electrolyte configuration with maintained density. The absence of cracks or delamination confirms the effectiveness of the co-sintering process in achieving strong interfacial bonding.
The screen-printed LSCF cathode (22 μm) is visible as the topmost layer in all cells, showing a porous microstructure necessary for oxygen reduction and gas transport. The cathode appears well adhered to the electrolyte surface with good interfacial contact. The SEM analysis confirms successful fabrication of complete cells with varying electrolyte thicknesses while maintaining consistent other layer thicknesses, enabling a systematic study of electrolyte thickness effects on electrochemical performance.
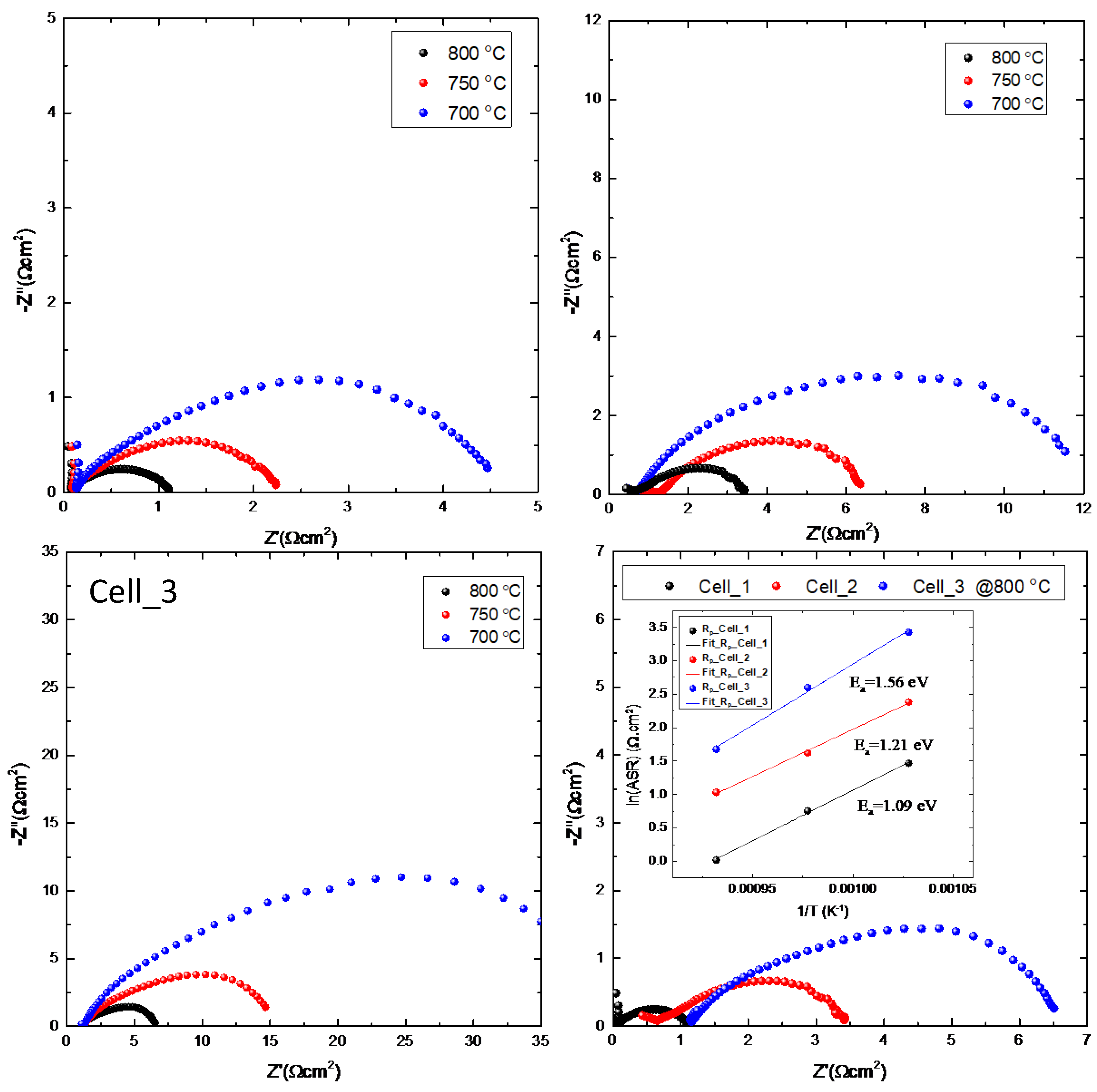
Figure 3 presents the electrochemical impedance spectroscopy (EIS) results for all three NiFe-supported SOFC cells measured at operating temperatures of 700°C, 750°C, and 800°C. The Nyquist plots provide detailed insights into the individual resistance contributions and reveal the dominant factors affecting cell performance as a function of electrolyte thickness. The most striking observation from the EIS data is the dramatic scaling of total cell impedance with electrolyte thickness. Cell_1, with the thinnest electrolyte (7.05 μm electrolyte), exhibits the lowest overall impedance, with the real impedance (Z') extending to approximately 4.5 Ωcm² and imaginary impedance (Z'') reaching a maximum of ~1.2 Ωcm² at 700°C. In contrast, Cell_2 (14.1 μm electrolyte) shows intermediate impedance values with Z' ~ 12 Ωcm² and Z'' up to ~3 Ωcm², while Cell_3 (21.2 μm electrolyte) demonstrates the highest impedance with Z' reaching ~35 Ωcm² and Z'' up to ~12 Ωcm² under the same conditions.
The high-frequency intercept of each Nyquist plot corresponds to the ohmic resistance (RO) of the cell, which is primarily determined by the ionic conductivity of the YSZ electrolyte. A clear correlation between electrolyte thickness and ohmic resistance is observed across all operating temperatures. At 800°C, the ohmic resistance increases from approximately 0.08 Ωcm² for Cell_1 to ~0.65 Ωcm² for Cell_2 and ~1.15 Ωcm² for Cell_3. This scaling is consistent with the expected linear relationship between electrolyte thickness and ionic resistance (R = ρl/A, where ρ is resistivity, l is thickness, and A is area), confirming that the YSZ electrolyte represents the primary source of ohmic losses in these cells. The direct comparison of all three cells at 800°C (bottom-right panel) clearly illustrates the benefits of optimizing electrolyte thickness. Cell_1 demonstrates a total area-specific resistance (ASR) of approximately 1.1 Ωcm², compared to 3.4 Ωcm² for Cell_2 and 6.5 Ωcm² for Cell_3.
Arrhenius analysis of the total resistance (inset of the bottom-right panel) yields activation energies of 1.09, 1.21, and 1.56 eV for cells with electrolyte thicknesses of 7.05, 14.2, and 21.2 μm, respectively. For polycrystalline YSZ, the bulk (grain interior) activation energy is typically 0.85-0.90 eV, while grain boundary contributions add ~1.0-1.3 eV [
11,
33]. For the thinnest electrolyte (7.05 μm), the activation energy of 1.09 eV suggests predominantly bulk conduction with minimal grain boundary contribution. In contrast, Cell_3 (21.2 μm) exhibits 1.56 eV, indicating significant grain boundary resistance combined with enhanced polarization resistance due to current constriction effects at the electrode/electrolyte interface [
10]. These interfacial effects particularly impact oxygen-ion incorporation kinetics [
1], one of the rate-limiting elementary steps in the oxygen reduction reaction and gas diffusion. Thus, the systematic increase in total activation energy with electrolyte thickness can be attributed to two primary factors: (1) increased grain boundary resistance as thicker electrolytes contain more grain boundaries along the conduction path, and (2) enhanced polarization resistance, particularly affecting oxygen-ion incorporation kinetics at the electrode/electrolyte interface.
The semicircular features in the Nyquist plots represent the combined electrode polarization resistances from both anode and cathode processes. Remarkably, the size and shape of these semicircles remain relatively consistent across all three cells when normalized for the different impedance scales. This observation indicates that the co-sintering fabrication process successfully maintained consistent electrode microstructures and electrochemical activity despite variations in electrolyte thickness.
The EIS analysis conclusively demonstrates that electrolyte thickness is the dominant factor determining overall cell resistance in this thickness range. The linear scaling of ohmic resistance with thickness, combined with minimal changes in electrode kinetics, indicates that further reduction of the electrolyte thickness below 7.05 μm could yield additional performance improvements, provided gas-tightness and mechanical integrity are maintained. These results establish clear design guidelines for optimizing MS-SOFC performance by systematically reducing electrolyte thickness while maintaining robust co-sintered interfaces.
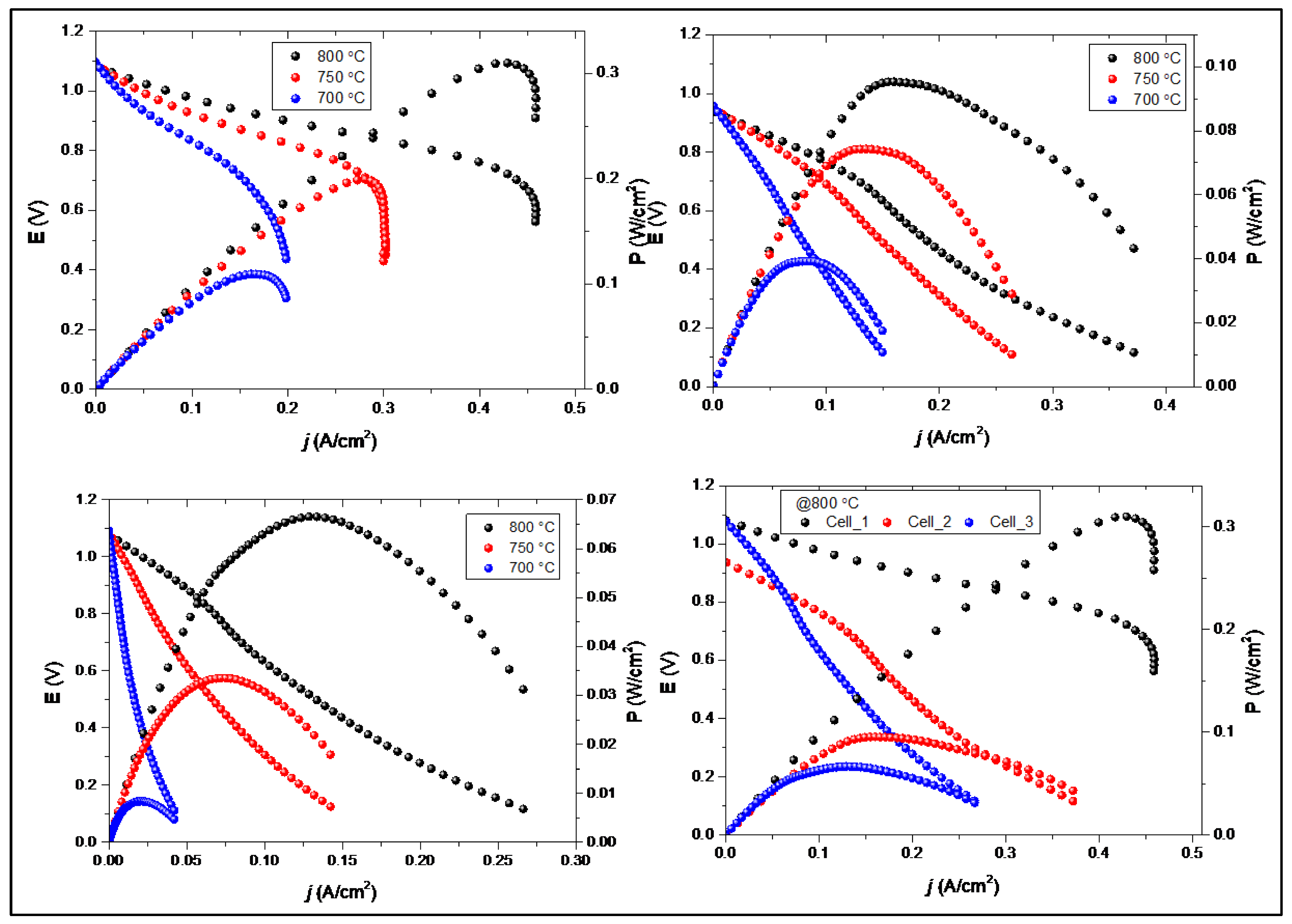
Figure 4 presents the electrochemical performance characteristics of the three Ni-Fe supported SOFC cells, showing both current-voltage (I-V) curves and corresponding power density plots as a function of operating temperature. The results demonstrate a clear correlation between electrolyte thickness and cell performance across the entire temperature range investigated.
The performance follows the expected trend based on electrolyte thickness: Cell_1 > Cell_2 > Cell_3. At the highest operating temperature of 800°C, Cell_1 achieves a peak power density of approximately 0.32 W/cm² at a current density of ~0.4 A/cm², representing superior performance compared to the thicker electrolyte variants. Cell_2 demonstrates an intermediate peak power density of ~0.10 W/cm² at 800°C, while Cell_3 exhibits the lowest performance with a maximum power density of ~0.07 W/cm² under the same conditions. This represents approximately a 4.6-fold improvement in peak power density between the thinnest and thickest electrolyte configurations. The power density achieved by Cell_1 is competitive with MS-SOFC performance reported in the literature [
38].
All cells exhibit the characteristic improvement in performance with increasing temperature, consistent with enhanced ionic conductivity and electrode kinetics at higher temperatures [
22]. However, the magnitude of temperature sensitivity varies significantly with electrolyte thickness. Cell_1 maintains reasonable performance even at 700°C, achieving a peak power density of ~0.12 W/cm², while Cell_3 shows severely degraded performance at this lower temperature with peak power density dropping to ~0.02 W/cm². This temperature-sensitivity difference becomes particularly important for intermediate-temperature SOFC applications, where lower operating temperatures are desired to improve durability and system integration [
31].
The power density curves reveal well-defined maxima for all cells, indicating optimal operating current densities that balance voltage and current to maximize power output. Cell_1 achieves peak power at current densities around 0.45 A/cm², compared to ~0.2 A/cm² for Cell_2 and ~0.12 A/cm² for Cell_3 at 800°C.
The direct comparison at 800°C (bottom right panel) provides a clear visualization of the performance hierarchy. The dramatic separation of the I-V curves demonstrates the substantial impact of electrolyte thickness optimization on practical cell performance. The voltage at any given current density follows the order Cell_1 > Cell_2 > Cell_3, which directly correlates with the impedance results and confirms that ohmic losses dominate the performance differences.
The I-V and power density results establish that electrolyte thickness optimization represents a critical design parameter for achieving high-performance MS-SOFCs. The 4.6-fold improvement in peak power density achieved through thickness reduction from 21.2 μm to 7.05 μm, combined with enhanced current-carrying capability and improved low-temperature performance, demonstrates the significant practical benefits obtainable through systematic electrolyte optimization in co-sintered metal-supported architectures.
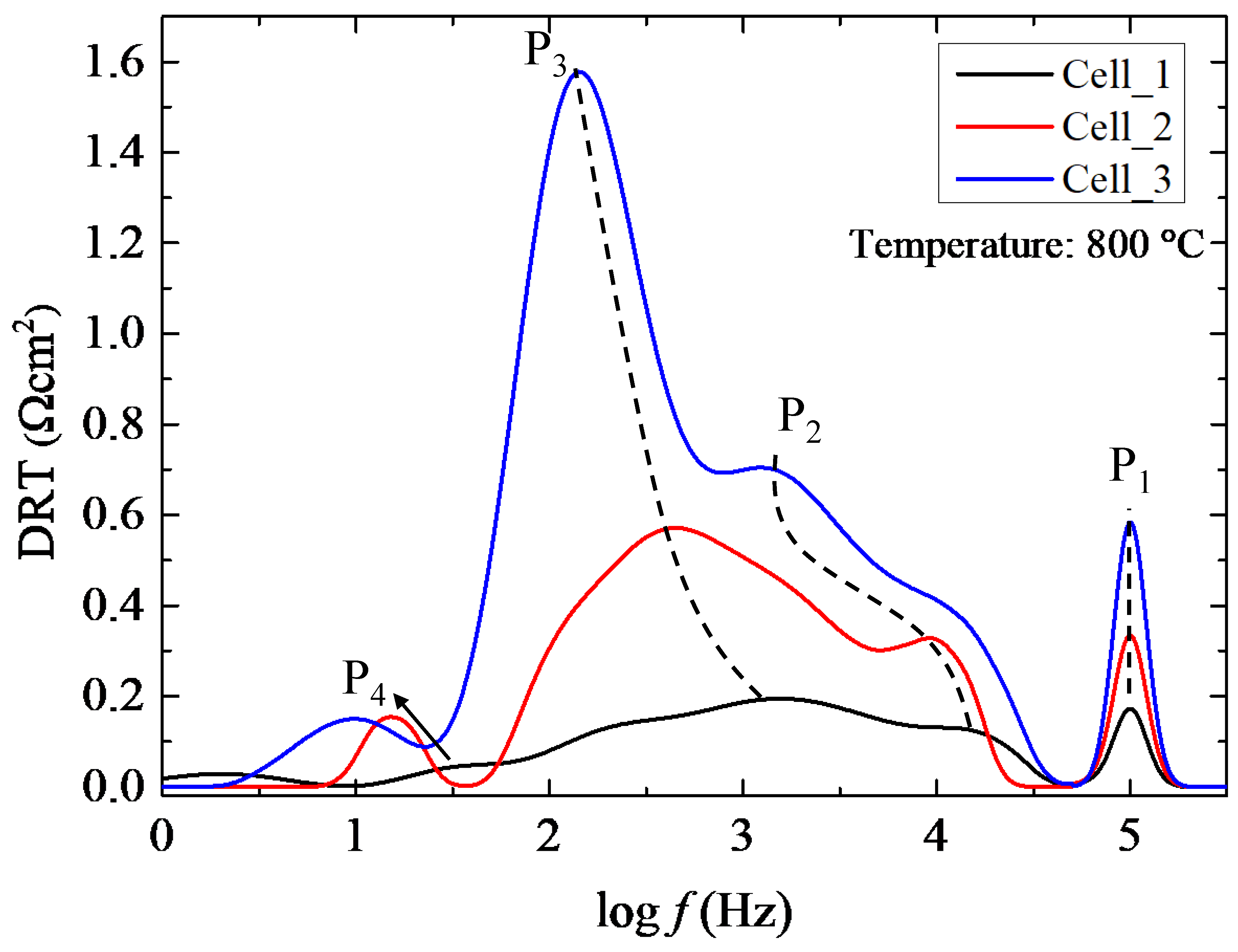
Further, to understand the overall effect of electrolyte thicknesses on electrochemical performance, the Distribution of Relaxation Times (DRT) obtained from Electrochemical Impedance Spectroscopy (EIS) for three Ni-Fe supported SOFCs with varying electrolyte thicknesses (7.05, 14.2, and 21.2 μm) at 800 ͦC.
Figure 5 presents the DRT results, revealing four distinct polarization processes (P
1-P
4) whose contributions vary systematically with electrolyte thickness. The DRT provides detailed insights into the distribution of different relaxation processes occurring within the cell, which are related to various resistance contributions, including ohmic resistance and electrode polarization.
High frequency peak P
1, attributed to ionic conduction through the YSZ electrolyte, based on its characteristic frequency range consistent with oxygen ion transport in YSZ [
19,
20]. This peak is weaker in Cell_1 (7.05 μm) but becomes increasingly prominent with thicker electrolytes, reaching maximum intensity in Cell_3 (21.2 μm). The significant growth of P
1 directly visualizes the increased ohmic resistance associated with increased electrolyte thickness, confirming that ionic transport resistance scales proportionally with the conduction path length [
16].
The dominant medium-range frequency peak P
2 corresponds to charge transfer processes at the electrode/electrolyte interfaces, consistent with oxygen reduction reaction kinetics reported for Ni-YSZ/YSZ interfaces [
19]. The systematic increase in P
2 magnitude with increasing electrolyte thickness can be attributed to current-constriction effects, where longer ion-conduction paths lead to a non-uniform current distribution at the interfaces [
24].
Peak P
3 is assigned to oxygen-ion incorporation and exchange at triple-phase boundaries, a process well documented in SOFC electrode systems [
2]. The relatively consistent magnitude of P
3 across all cells indicates that this fundamental electrochemical process is not significantly affected by variations in electrolyte thickness. The low-frequency contribution P
4 represents gas diffusion and conversion processes within the porous electrode structure [
12,
19]. The minimal variation of P
4 across cells confirms that electrode microstructure and gas transport pathways remain unaffected by electrolyte thickness.
Integration of the DRT peaks reveals that the total polarization resistance increase from Cell_1 to Cell_3 is dominated by the growth of P1 (ohmic) and P2 (charge transfer), while electrode-related processes (P3, P4) remain relatively constant. This quantitative deconvolution demonstrates that reducing YSZ thickness to ~7 μm effectively eliminates the high-frequency ohmic contribution while preserving electrode functionality, validating thin-electrolyte design strategies for high-performance MS-SOFCs.