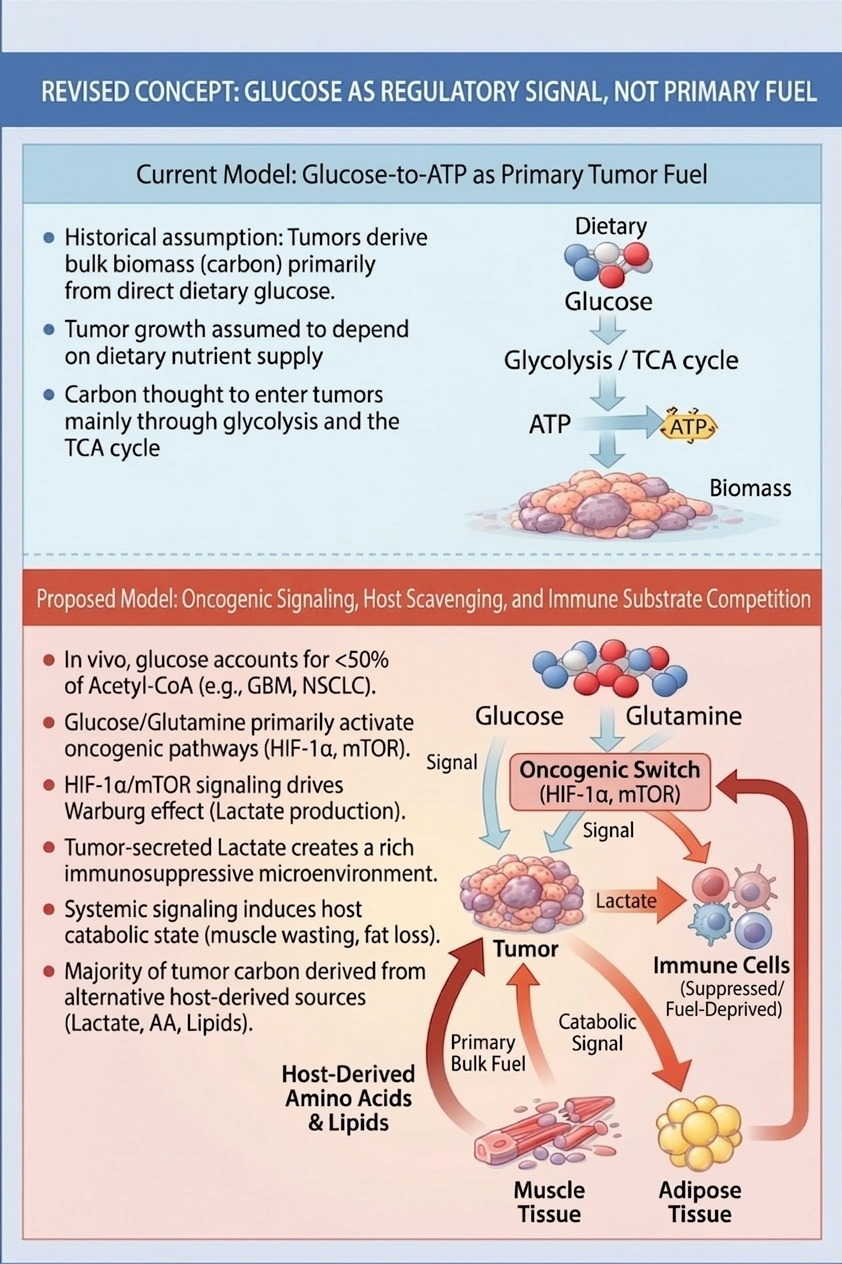
After decades of in vivo isotope tracing, human solid tumors have not been shown to derive the majority of their carbon from circulating glucose. Despite this, glucose uptake by tumors continues to be widely interpreted as evidence of glucose dependence for growth. In contrast, mounting clinical and metabolic evidence indicates that glucose and glutamine are consumed primarily as regulatory and competitive substrates rather than as dominant carbon sources, with tumor biomass supplied largely by lactate, glutamine, and host-derived amino acids and lipids.Cachexia is commonly described as a secondary complication of advanced cancer, yet this metabolic behavior suggests it functions instead as a tumor-maintained systemic state that favors malignant survival at the expense of host tissues. By consuming glucose and glutamine at high rates, tumors restructure host metabolism, suppress immune function through substrate deprivation, and induce a catabolic shift that mobilizes host tissues as the tumor’s true nutrient reservoir. Dietary deprivation strategies therefore fail in solid tumors not because tumors adapt to starvation, but because restriction accelerates host metabolic collapse rather than depriving the tumor.Central to this argument is a newly proposed construct: homeostatic deception via dissociated catabolic ketosis, a tumor-orchestrated state in which physiological ketogenesis is genuinely present but decoupled from its normal protein-sparing function. Circulating ketones satisfy central energy-sensing mechanisms, silencing counter-regulatory alarms while unrestrained muscle proteolysis and lipolysis proceed. The resulting catabolic loop supplies tumors with substrates released from host tissues while the host’s regulatory systems interpret the state as normal adaptive fasting. Cachexia persists as long as the tumor driver remains active and reverses primarily when tumor burden and inflammatory signaling are controlled. A case of metastatic NSCLC, with photographic documentation, serves as the observational origin of this framework (Johnson CL, 2026, https://doi.org/10.5281/zenodo.18988466). This manuscript integrates metabolic tracing, immunometabolism, and clinical observation to propose a mechanistic hypothesis reframing cachexia as a tumor-maintained state. The framework identifies multiple targets for companion therapeutic intervention and explains the failure of diet-based strategies.