Submitted:
14 March 2026
Posted:
17 March 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Outlining the System Being Studied
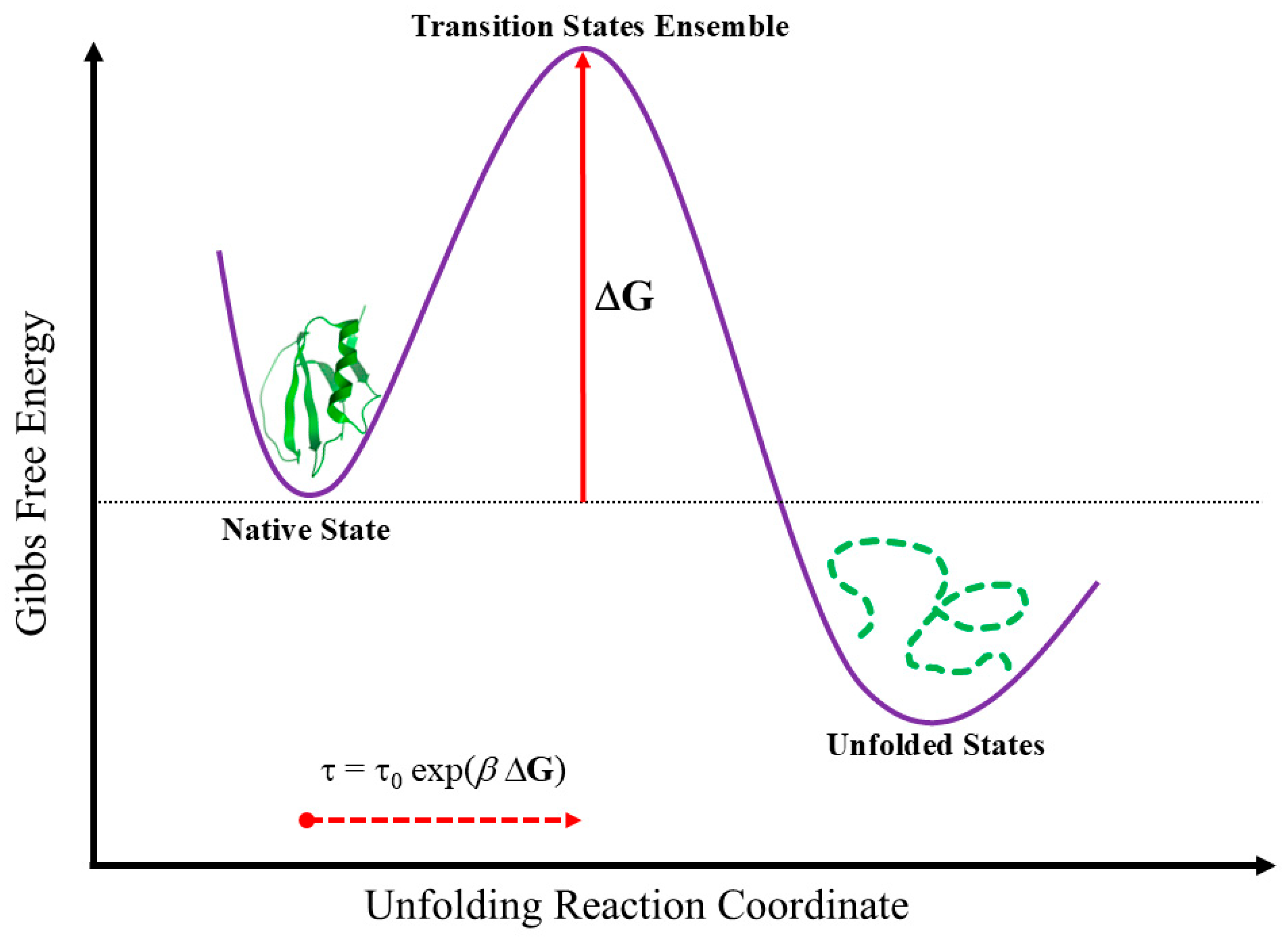
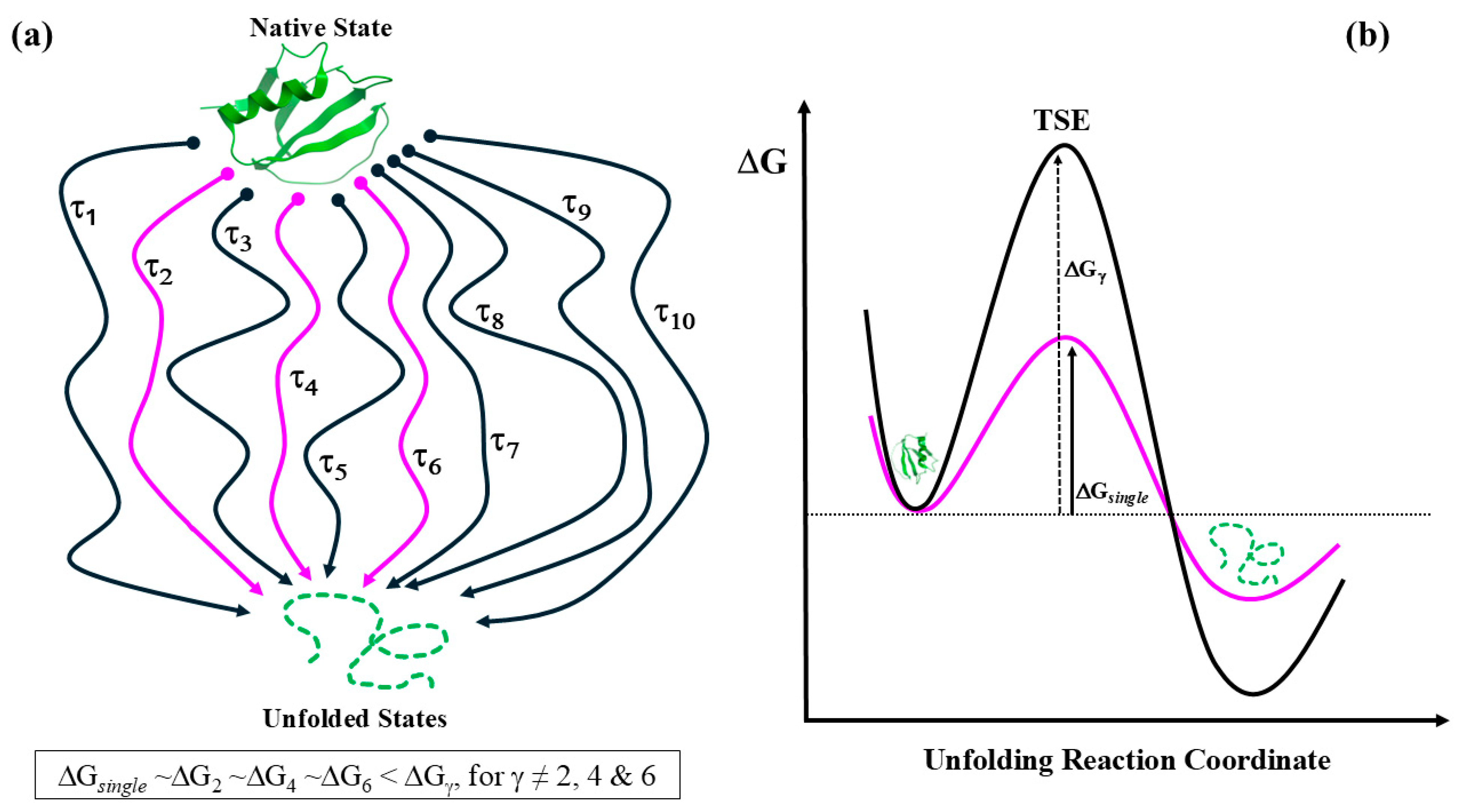
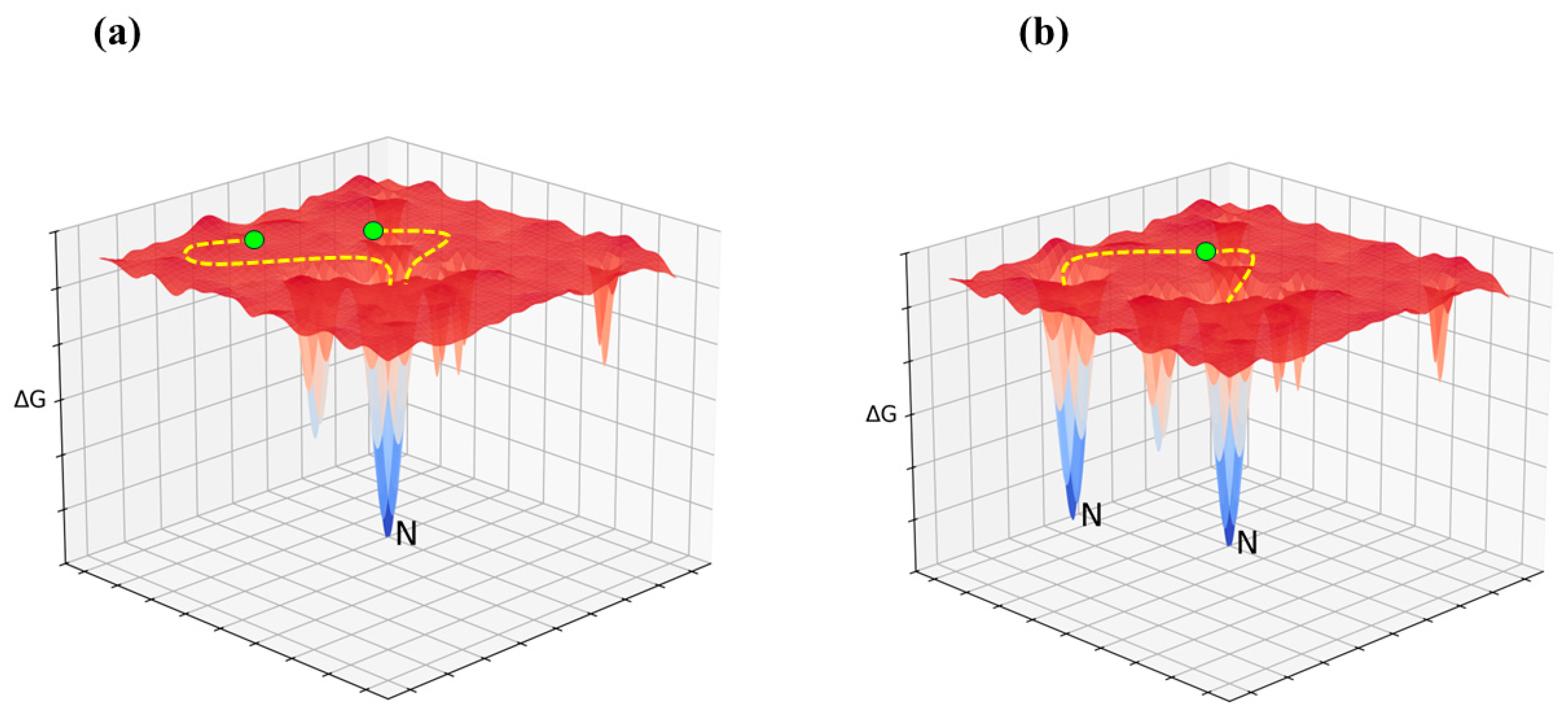
The Least Action Principle and the Effective-Trajectory Conjecture
Avenues for Future Research
Conclusions
Founding
Ethical approval
Acknowledgments
Conflicts of Interest
References
- Akmal, A; Muñoz, V. The nature of the free energy barriers to two-state folding. Proteins 2004, 57, 142–52. [Google Scholar] [CrossRef]
- Anfinsen, CB; Scheraga, HA. Experimental and theoretical aspects of protein folding. Adv Protein Chem 1975, 29, 205–300. [Google Scholar]
- Anfinsen, CB. Principles that govern the folding of protein chains. Science 1973, 181, 223–230. [Google Scholar] [CrossRef]
- Arnautova, YA; Jagielska, A; Scheraga, HA. A new force field (ECEPP-05) for peptides, proteins, and organic molecules. J Phys Chem B 2006, 110(10), 5025–5044. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y; Sosnick, TR; Mayne, L; Englander, SW. Protein folding intermediates: native-state hydrogen exchange. Science 1995, 269(5221), 192–197. [Google Scholar] [CrossRef]
- Bakk, A; Høye, JS; Hansen, A; Sneppen, K; Jensen, MH. Pathways in two-state protein folding. Biophys J 2000, 79(5), 2722–2727. [Google Scholar] [CrossRef]
- Baldwin, RL. Protein folding. Matching speed and stability. Nature 1994, 369(6477), 183–184. [Google Scholar] [CrossRef]
- Baldwin, RL. The nature of protein folding pathways: the classical versus the new view. J Biomol NMR 1995, 5(2), 103–109. [Google Scholar] [CrossRef] [PubMed]
- Ben-Naim, A. Levinthal’s Paradox Revisited and Dismissed. Open Journal of Biophysics 2012, 2, 23–32. [Google Scholar] [CrossRef]
- Bogatyreva, NS; Finkelstein, AV. Cunning simplicity of protein folding landscapes. Protein Eng 2001, 14(8), 521–523. [Google Scholar] [CrossRef]
- Chang, L; Perez, A. Rapid estimation of protein folding pathways from sequence alone using AlphaFold2. Nat Commun 2025. [Google Scholar] [CrossRef]
- Coopersmith, J. The Lazy Universe: An Introduction to the Principle of Least Action; Oxford University Press, 2017. [Google Scholar] [CrossRef]
- Creighton, TE. The problem of how and why proteins adopt folded conformations. The Journal of Physical Chemistry 1985, 89(12), 2452–2459. [Google Scholar] [CrossRef]
- Dill, KA; Chan, HS. From Levinthal to pathways to funnels. Nat Struct Biol 1997, 4(1), 10–19. [Google Scholar] [CrossRef] [PubMed]
- Ding, F; Dokholyan, NV; Buldyrev, SV; Stanley, HE; Shakhnovich, EI. Direct molecular dynamics observation of protein folding transition state ensemble. Biophys J 2002, 83(6), 3525–3532. [Google Scholar] [CrossRef]
- Dishman, AF; Volkman, BF. Unfolding the Mysteries of Protein Metamorphosis. ACS Chemical Biology 2018, 13(6), 1438–1446. [Google Scholar] [CrossRef]
- Dobson, CM; Šali, A; Karplus, M. Protein Folding: A Perspective from Theory and Experiment. Angew Chem Int Ed Engl 1998, 37(7), 868–893. [Google Scholar] [CrossRef]
- Eaton, WA; Wolynes, PG. Theory, simulations, and experiments show that proteins fold by multiple pathways. Proc Natl Acad Sci USA 2017, 114(46), E9759–E9760. [Google Scholar] [CrossRef]
- Eaton, WA. Modern Kinetics and Mechanism of Protein Folding: A Retrospective. J Phys Chem B 2021, 19 125, 3452–3467. [Google Scholar] [CrossRef]
- Englander, SW; Mayne, L; Krishna, MM. Protein folding and misfolding: mechanism and principles. Q Rev Biophys 2007, 40(4), 287–326. [Google Scholar] [CrossRef]
- Englander, SW; Mayne, L. Reply to Eaton and Wolynes: How do proteins fold? Proc Natl Acad Sci USA 2017, 114(46), E9761–E9762. [Google Scholar] [CrossRef] [PubMed]
- Englander, SW; Mayne, L. The nature of protein folding pathways. Proc Natl Acad Sci USA 2014, 111(45), 15873–15880. [Google Scholar] [CrossRef] [PubMed]
- Englander, SW. Protein folding intermediates and pathways studied by hydrogen exchange. Annu Rev Biophys Biomol Struct 2000, 29, 213–38. [Google Scholar] [CrossRef]
- Fakhoury, Z; Sosso, GC; Habershon, S. Generating Protein Folding Trajectories Using Contact-Map-Driven Directed Walks. J Chem Inf Model 2023, 63(7), 2181–2195. [Google Scholar] [CrossRef]
- Fee, J. Maupertuis, and the Principle of Least Action. The Scientific Monthly, JSTOR, 1941; 52, pp. 496–503. Available online: http://www.jstor.org/stable/17253.
- Feynman, RP; Leighton, RB; Sands, M. The Feynman Lectures on Physics; Addison-Wesley: Reading, MA, 1963; Vol. II, p. Chap. 19. [Google Scholar]
- Feynman, RP. The Principle of Least Action in Quantum Mechanics. Feynman’s Thesis—A New Approach to Quantum Theory, World Scientific, 2005; pp. pp 1–69. [Google Scholar]
- Finkelstein, AV; Badretdinov, AYa. Rate of protein folding near the point of thermodynamic equilibrium between the coil and the most stable chain fold. Fold Des 1997, 2(2), 115–121. [Google Scholar] [CrossRef]
- Finkelstein, AV; Garbuzynskiy, SO. Levinthal’s question answered again? J Biomol Struct Dyn. 2013, 31(9), 1013–1015. [Google Scholar] [CrossRef]
- Garbuzynskiy, SO; Ivankov, DN; Bogatyreva, NS; Finkelstein, AV. Golden triangle for folding rates of globular proteins. Proc Natl Acad Sci USA 2013, 110(1), 147–150. [Google Scholar] [CrossRef]
- Glyakina, AV; Galzitskaya, OV. How Quickly Do Proteins Fold and Unfold, and What Structural Parameters Correlate with These Values? Biomolecules 2020, 10, 197. [Google Scholar] [CrossRef] [PubMed]
- Gromiha, MM. A statistical model for predicting protein folding rates from amino acid sequence with structural class information. J Chem Inf Model 2005, 45(2), 494–501. [Google Scholar] [CrossRef] [PubMed]
- Hanc, J; Taylor, EF. From conservation of energy to the principle of least action: a story line. Am J Phys 2004, 72, 514–521. [Google Scholar] [CrossRef]
- Harrington, WF; Schellman, JA. Evidence for the instability of hydrogen-bonded peptide structures in water, based on studies of ribonuclease and oxidized ribonuclease. C R Trav Lab Carlsberg Chim 1956, 30(3), 21–43. [Google Scholar]
- Honig, B. Protein folding: from the Levinthal paradox to structure prediction. J Mol Biol 1999, 293(2), 283–293. [Google Scholar] [CrossRef]
- Huang, LT; Gromiha, MM. Analysis and prediction of protein folding rates using quadratic response surface models. J Comput Chem 2008, 29(10), 1675–1683. [Google Scholar] [CrossRef]
- Itzhaki, LS; Otzen, DE; Fersht, AR. The structure of the transition state for folding of chymotrypsin inhibitor 2 analyzed by protein engineering methods: evidence for a nucleation-condensation mechanism for protein folding. J Mol Biol 1995, 254, 260–288. [Google Scholar] [CrossRef]
- Ivankov, DN; Finkelstein, AV. Solution of Levinthal’s Paradox and a Physical Theory of Protein Folding Times. Biomolecules 2020, 10(2), 250. [Google Scholar] [CrossRef]
- Jackson, SE. How do small single-domain proteins fold? Fold Des 1998, 3(4), R81–R91. [Google Scholar] [CrossRef]
- Kaila, VRI; Annila, A. Natural selection for least action. Proc R Soc A 2008, 464, 3055–3070. [Google Scholar] [CrossRef]
- Karplus, M. Behind the folding funnel diagram. Nat Chem Biol 2011, 7(7), 401–404. [Google Scholar] [CrossRef] [PubMed]
- Karplus, M. The Levinthal paradox: yesterday and today. Fold Des 1997, 2(4), S69–S75. [Google Scholar] [CrossRef]
- Kikuchi, T. Decoding an Amino Acid Sequence to Extract Information on Protein Folding. Molecules 2022, 27(9), 3020. [Google Scholar] [CrossRef]
- Kuwajima, K. The Molten Globule, and Two-State vs. Non-Two-State Folding of Globular Proteins. Biomolecules 2020, 10, 407. [Google Scholar] [CrossRef] [PubMed]
- Landau, LD; Lifshitz, EM. Course of Theoretical Physics. In Mechanics, 3rd ed.; Butterworth-Heinemann: Oxford, 1982; Volume 1. [Google Scholar]
- Landau, LD; Lifshitz, EM. Course of Theoretical Physics. In The Classical Theory of Fields, 4th ed.; Butterworth-Heinemann: Oxford, 1987; Volume 2. [Google Scholar]
- Lange, OF; Lakomek, N-A; Farès, C; Schröder, GF; Walter, KFA; Becker, S; Meiler, J; Grubmüller, H; Griesinger, C; de Groot, BL. Recognition Dynamics Up to Microseconds Revealed from an RDC-Derived Ubiquitin Ensemble in Solution. Science 2008, 13, 1471–1475. [Google Scholar] [CrossRef]
- Lapidus, LJ. The road less traveled in protein folding: evidence for multiple pathways. Curr Opin Struct Biol 2021, 66, 83–88. [Google Scholar] [CrossRef]
- Lazaridis, T; Karplus, M. ‘New view’ of protein folding reconciled with the old through multiple unfolding simulations. Science 1997, 278(5345), 1928–1931. [Google Scholar] [CrossRef]
- Levinthal, C. Are There Pathways for Protein Folding? Journal de Chimie Physique 1968, 65, 44–45. [Google Scholar] [CrossRef]
- Levinthal, C. How to fold graciously. In Mossbauer Spectroscopy in Biological Systems, Proceedings of a Meeting held at Allerton House, Monticello, Illinois; Debrunner, P, Tsibris, JCM, Münck, E, Eds.; University of Illinois Press: Urbana, 1969; p. 22. [Google Scholar]
- Li, Y; Gong, H. Identifying a Feasible Transition Pathway between Two Conformational States for a Protein. J Chem Theory Comput 2022, 18, 4529–4543. [Google Scholar] [CrossRef]
- Martin, AO; Vila, JA. The Marginal Stability of Proteins: How the Jiggling and Wiggling of Atoms is Connected to Neutral Evolution. Journal of Molecular Evolution 2020, 88, 424–426. [Google Scholar] [CrossRef]
- Martin, OA; Vorobjev, Y; Scheraga, HA; Vila, JA. Outline of an experimental design aimed to detect a protein A mirror image in solution. PeerJ Phys Chem 2019, 1, e2. [Google Scholar] [CrossRef] [PubMed]
- Martinez, L. Introducing the Levinthal’s Protein Folding Paradox and Its Solution. J Chem Educ 2014, 91(11), 1918–1923. [Google Scholar] [CrossRef]
- Matouschek, A; Kellis, JT, Jr.; Serrano, L; Fersht, AR. Mapping the transition state and pathway of protein folding by protein engineering. Nature 1989, 340, 122–126. [Google Scholar] [CrossRef] [PubMed]
- Mayor, U; Johnson, CM; Daggett, V; Fersht, AR. Protein folding and unfolding in microseconds to nanoseconds by experiment and simulation. Proc Natl Acad Sci USA 2000, 97, 13518–13522. [Google Scholar] [CrossRef]
- McCammon, JA; Gelin, BR; Karplus, M. Dynamics of folded proteins. Nature 1977, 267, 585–590. [Google Scholar] [CrossRef] [PubMed]
- McCammon, JA. A speed limit for protein folding. Proc Natl Acad Sci USA 1996, 93, 11426–11427. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, V; Cerminara, M. When fast is better: protein folding fundamentals and mechanisms from ultrafast approaches. Biochem J 2016, 473, 2545–2559. [Google Scholar] [CrossRef]
- Murzin, A.G. Biochemistry. Metamorphic proteins. Science 2008, 320, 1725–1726. [Google Scholar] [CrossRef] [PubMed]
- Neupane, K.; Manuel, A.; Woodside, M. Protein folding trajectories can be described quantitatively by one-dimensional diffusion over measured energy landscapes. Nature Phys 2016, 12, 700–703. [Google Scholar] [CrossRef]
- Pande, VS; Grosberg, AY; Tanaka, T; Rokhsar, DS. Pathways for protein folding: is a new view needed? Curr Opin Struct Biol 1998, 8(1), 68–79. [Google Scholar] [CrossRef]
- Plaxco, KW; Simons, KT; Ruczinski, I; Baker, D. Topology, stability, sequence, and length: defining the determinants of two-state protein folding kinetics. Biochemistry 2000, 37, 11177–11183. [Google Scholar] [CrossRef]
- Rooman, M; Dehouck, Y; Kwasigroch, JM; Biot, C; Gilis, D. What is paradoxical about Levinthal paradox? J Biomol Struct Dyn 2002, 20(3), 327–329. [Google Scholar] [CrossRef]
- Šali, A; Shakhnovich, E; Karplus, M. How does a protein fold? Nature 1994, 369(6477), 248–251. [Google Scholar] [CrossRef]
- Shakhnovich, E. Protein folding thermodynamics and dynamics: where physics, chemistry, and biology meet. Chem Rev 2006, 106, 1559–1588. [Google Scholar] [CrossRef]
- Simmons, W; Weiner, JL. The principle of stationary action in biophysics: stability in protein folding. J Biophys 2013, 2013, 697529. [Google Scholar] [CrossRef] [PubMed]
- Stiller, JB; Otten, R; Häussinger, D; Rieder, PS; Theobald, DL; Kern, D. Structure determination of high-energy states in a dynamic protein ensemble. Nature 2022, 603(7901), 528–535. [Google Scholar] [CrossRef]
- Thirumalai, D. From Minimal Models to Real Proteins: Time Scales for Protein Folding Kinetics. Journal de Physique I 1995, 5(11), 1457–1467. [Google Scholar] [CrossRef]
- Tsuboyama, K; Dauparas, J; Chen, J; Laine, E; Mohseni Behbahani, Y; Weinstein, JJ; Mangan, NM; Ovchinnikov, S; Rocklin, GJ. Mega-scale experimental analysis of protein folding stability in biology and design. Nature 2023, 620(7973), 434–444. [Google Scholar] [CrossRef] [PubMed]
- Vendruscolo, M; Paci, E; Dobson, CM; Karplus, M. Rare Fluctuations of Native Proteins Sampled by Equilibrium Hydrogen Exchange. J Am Chem Soc 2003, 125(51), 15686–15687. [Google Scholar] [CrossRef]
- Vila, JA; Ripoll, DR; Scheraga, HA. Atomically detailed folding simulation of the B domain of staphylococcal protein A from random structures. Proc Natl Acad Sci USA 2003, 100(25), 14812–14816. [Google Scholar] [CrossRef] [PubMed]
- Vila, JA. Analysis of proteins in the light of mutations. Eur Biophys J 2024b, 53(5-6), 255–265. [Google Scholar] [CrossRef]
- Vila, JA. Factors controlling protein evolvability—at the molecular scale. Eur Biophys J 2025, 55, 101–109. [Google Scholar] [CrossRef]
- Vila, JA. Forecasting the upper bound free energy difference between protein native-like structures. Physica A 2019, 533, 122053. [Google Scholar] [CrossRef]
- Vila, JA. Metamorphic Proteins in Light of Anfinsen's Dogma. J Phys Chem Lett 2020, 11(13), 4998–4999. [Google Scholar] [CrossRef]
- Vila, JA. Protein Evolution upon Point Mutations. ACS Omega 2022, 7, 14371–14376. [Google Scholar] [CrossRef]
- Vila, JA. Protein folding rate evolution upon mutations. Biophys Rev 2023a, 15, 661–669. [Google Scholar] [CrossRef]
- Vila, JA. Rethinking the protein folding problem from a new perspective. Eur Biophys J 2023b, 52(3), 189–193. [Google Scholar] [CrossRef]
- Vila, JA. The origin of mutational epistasis. Eur Biophys J 2024a, 53, 473–480. [Google Scholar] [CrossRef]
- Vila, JA. Thoughts on the Protein’s Native State. J Phys Chem Lett 2021, 12, 5963–5966. [Google Scholar] [CrossRef] [PubMed]
- Wolynes, PG; Onuchic, JN; Thirumalai, D. Navigating the folding routes. Science 1995, 267(5204), 1619–1620. [Google Scholar] [CrossRef] [PubMed]
- Wolynes, PG. Evolution, energy landscapes, and the paradoxes of protein folding. Biochimie 2015, 119, 218–230. [Google Scholar] [CrossRef] [PubMed]
- Zana, R. On the rate determining step for helix propagation in the helix-coil transition of polypeptides in solution. Biopolymers 1975, 14, 2425–2428. [Google Scholar] [CrossRef]
- Zhao, K; Xia, Y; Zhang, F; Zhou, X; Li, SZ; Zhang, G. Protein structure and folding pathway prediction based on remote homologs recognition using PAthreader. Commun Biol 2023, 6(1), 243. [Google Scholar] [CrossRef]
- Zhao, K; Zhao, P; Wang, S; Xia, Y; Zhang, G. FoldPAthreader: predicting protein folding pathway using a novel folding force field model derived from the known protein universe. Genome Biol 2024, 25(1), 152. [Google Scholar] [CrossRef]
- Zwanzig, R; Szabo, A; Bagchi, B. Levinthal’s paradox. Proc Natl Acad Sci USA 1992, 89, 20–22. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



