Submitted:
13 March 2026
Posted:
16 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Expression and Purification of Recombinant Wild-Type Alpha-Synuclein and Its Mutant form A53T
2.2. Alpha-Synuclein Seeds Preparation
2.3. Alpha-Synuclein Fibrillation
2.4. Congo Red Fluorescence Spectroscopy
2.5. Dynamic Light Scattering
2.6. Transmission Electron Microscopy (TEM)
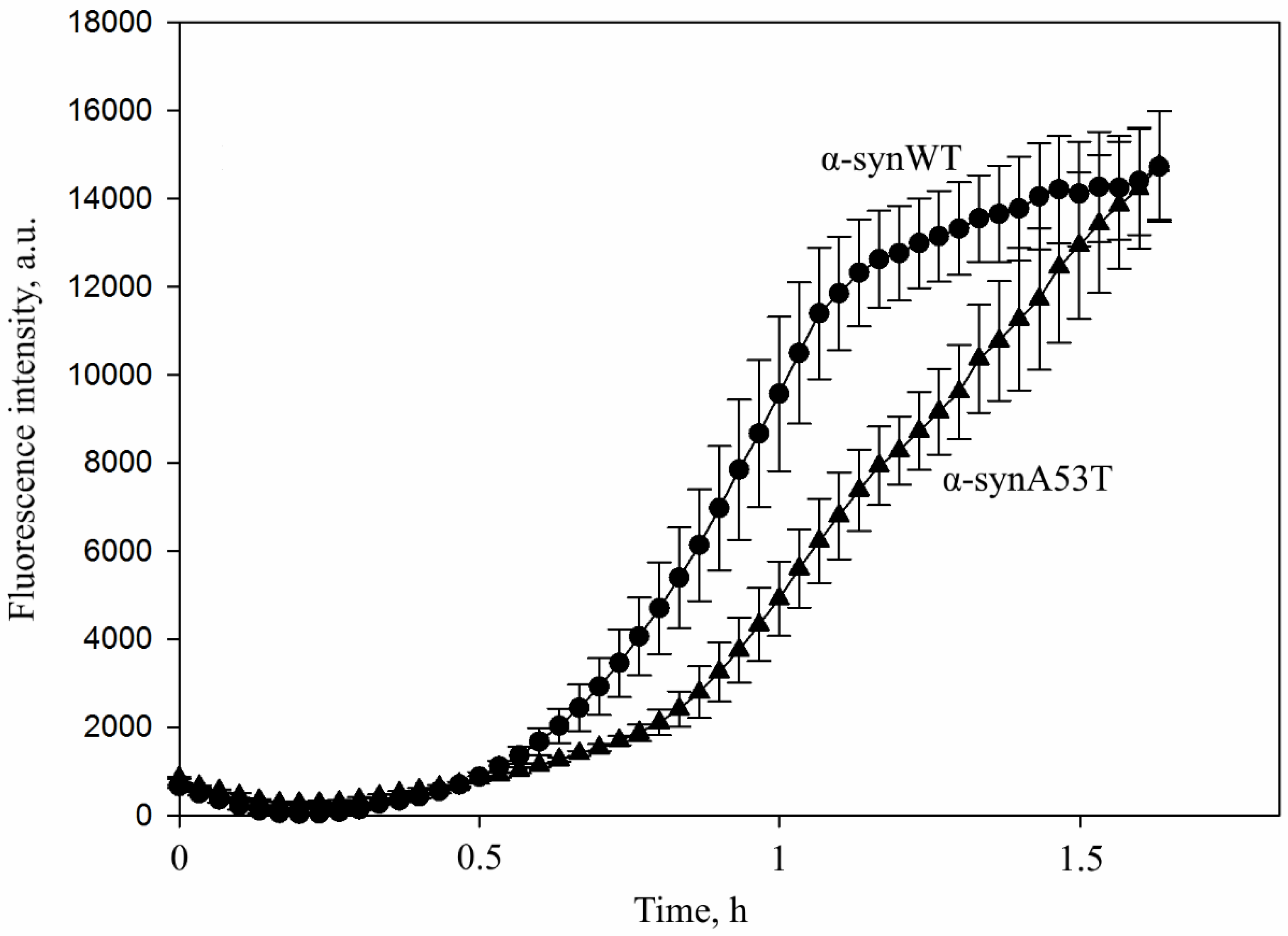
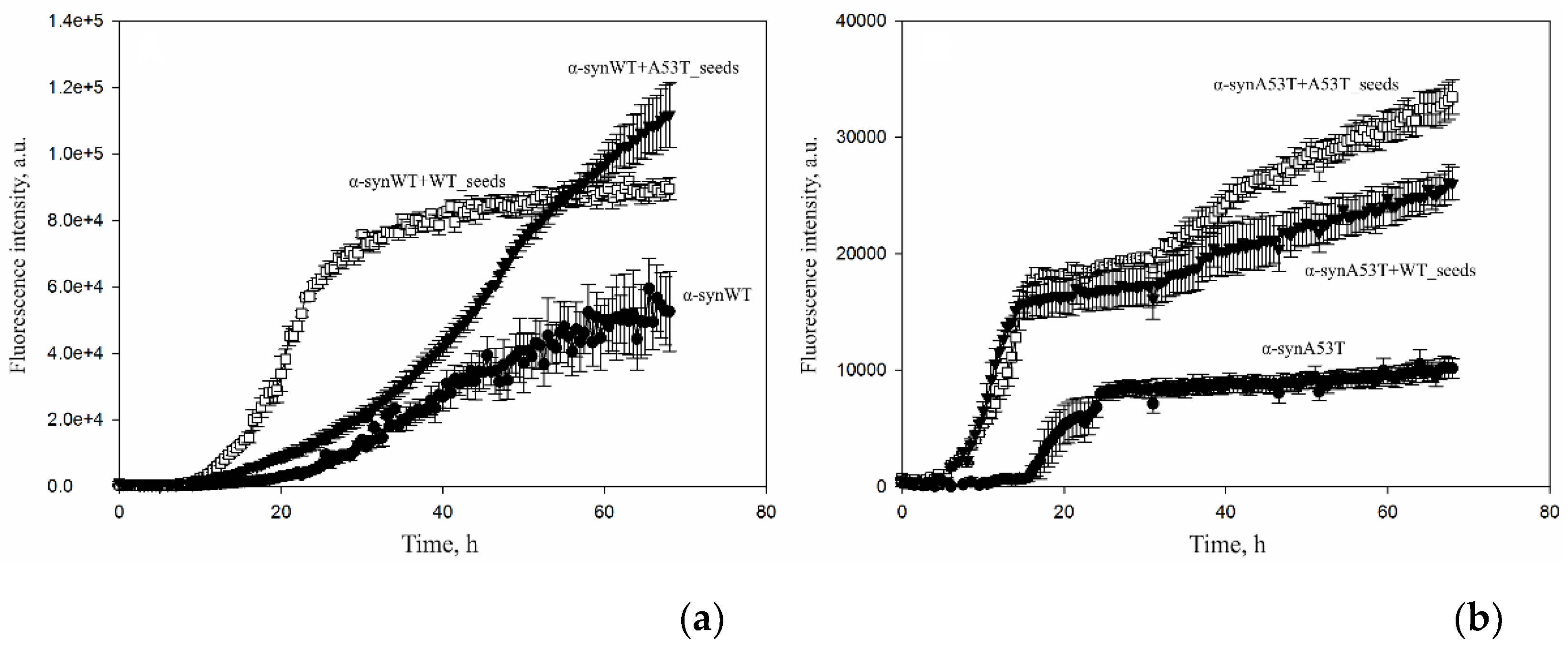
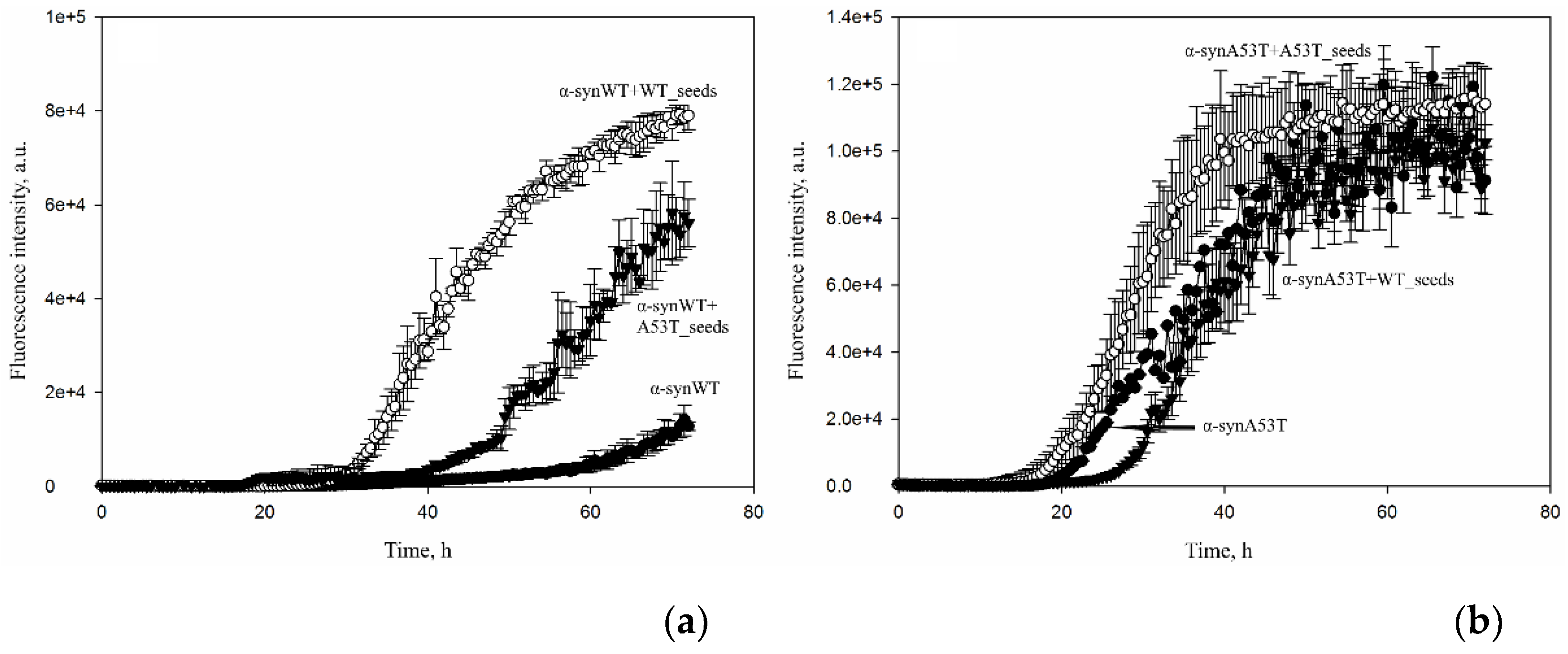
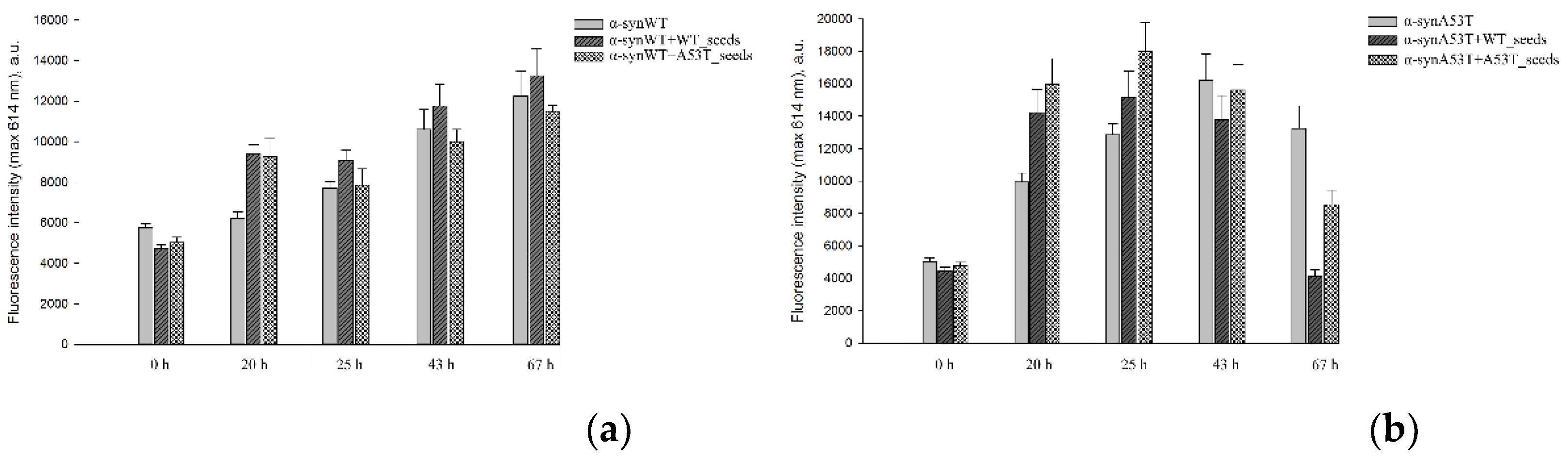
3. Results
4. Discussion
5. Conclusions
- The monomers of alpha-synuclein mutant form A53T are a promising candidate for use in test systems due to the significantly reduced lag period of fibrillation.
- Fibrillation of the alpha-synuclein A53T is accelerated by the addition of both wild-type and mutant alpha-synuclein seeds, which may facilitate the detection of various aberrant protein conformations in biological fluids.
- Low efficiency of wild-type alpha-synuclein fibrillation in the presence of alpha-synuclein A53T seeds may suggest that the features of cross-seeding cause a decrease in the effectiveness of current assays
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| α-synWT | recombinant human wild-type alpha-synuclein |
| α-synA53T | mutant form of recombinant human alpha-synuclein with replacement of the 53d alanine residue with a threonine one |
| CSF | cerebrospinal fluid |
| DLS | dynamic light scattering |
References
- Polymeropoulos, M. H.; Lavedan, C.; Leroy, E.; Ide, S. E.; Dehejia, A.; Dutra, A.; Pike, B.; Root, H.; Rubenstein, J.; Boyer, R.; Stenroos, E. S.; Chandrasekharappa, S.; Athanassiadou, A.; Papapetropoulos, T.; Johnson, W. G.; Lazzarini, A. M.; Duvoisin, R. C.; Di Iorio, G.; Golbe, L. I.; Nussbaum, R. L. Mutation in the Alpha-Synuclein Gene Identified in Families with Parkinson’s Disease. Science 1997, 276, 2045–2047. [Google Scholar] [CrossRef]
- Baba, M.; Nakajo, S.; Tu, P. H.; Tomita, T.; Nakaya, K.; Lee, V. M.; Trojanowski, J. Q.; Iwatsubo, T. Aggregation of Al-pha-Synuclein in Lewy Bodies of Sporadic Parkinson’s Disease and Dementia with Lewy Bodies. Am J Pathol 1998, 152, 879–884. [Google Scholar]
- Simon, D. K.; Tanner, C. M.; Brundin, P. Parkinson Disease Epidemiology, Pathology, Genetics, and Pathophysiology. Clin Geriatr Med 2020, 36, 1–12. [Google Scholar] [CrossRef]
- Koga, S.; Sekiya, H.; Kondru, N.; Ross, O. A.; Dickson, D. W. Neuropathology and molecular diagnosis of Synucleinopathies. Molecular neurodegeneration 2021, 16, 83. [Google Scholar] [CrossRef] [PubMed]
- von Euler Chelpin, M.; Söderberg, L.; Fälting, J.; Möller, C.; Giorgetti, M.; Constantinescu, R.; Blennow, K.; Zetterberg, H.; Höglund, K. Alpha-Synuclein Protofibrils in Cerebrospinal Fluid: A Potential Biomarker for Parkinson’s Disease. J Parkinsons Dis 2020, 10, 1429–1442. [Google Scholar] [CrossRef]
- Outeiro, T. F. Alpha-Synuclein Antibody Characterization: Why Semantics Matters. Mol Neurobiol 2021, 58, 2202–2203. [Google Scholar] [CrossRef] [PubMed]
- Bengoa-Vergniory, N.; Roberts, R. F.; Wade-Martins, R.; Alegre-Abarrategui, J. Alpha-Synuclein Oligomers: A New Hope. Acta Neuropathol 2017, 134, 819–838. [Google Scholar] [CrossRef]
- Spillantini, M. G.; Schmidt, M. L.; Lee, V. M.; Trojanowski, J. Q.; Jakes, R.; Goedert, M. Alpha-Synuclein in Lewy Bodies. Nature 1997, 388, 839–840. [Google Scholar] [CrossRef]
- Burré, J.; Sharma, M.; Südhof, T. C. Cell Biology and Pathophysiology of α-Synuclein. Cold Spring Harb Perspect Med 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Moussaud, S.; Malany, S.; Mehta, A.; Vasile, S.; Smith, L. H.; McLean, P. J. Targeting α-Synuclein Oligomers by Pro-tein-Fragment Complementation for Drug Discovery in Synucleinopathies. Expert Opin Ther Targets 2015, 19, 589–603. [Google Scholar] [CrossRef]
- Srinivasan, E.; Chandrasekhar, G.; Chandrasekar, P.; Anbarasu, K.; Vickram, A. S.; Karunakaran, R.; Rajasekaran, R.; amp; Srikumar, P. S. Alpha-Synuclein Aggregation in Parkinson’s Disease. Frontiers in medicine 2021, 8, 736978. [Google Scholar] [CrossRef]
- Del Tredici, K.; Braak, H. Review: Sporadic Parkinson’s disease: development and distribution of α-synuclein pathology. Neuropathology and applied neurobiology 2016, 42, 33–50. [Google Scholar] [CrossRef]
- Morris, H. R.; Spillantini, M. G.; Sue, C. M.; Williams-Gray, C. H. The Pathogenesis of Parkinson’s Disease. Lancet 2024, 403, 293–304. [Google Scholar] [CrossRef]
- Chelban, V.; Vichayanrat, E.; Schottlaende, L.; Iodice, V.; Houlden, H. Autonomic Dysfunction in Genetic Forms of Synucle-inopathies. Mov Disord 2018, 33, 359–371. [Google Scholar] [CrossRef]
- Fujita, K.; Homma, H.; Jin, M.; Yoshioka, Y.; Jin, X.; Saito, Y.; Tanaka, H.; Okazawa, H. Mutant α-Synuclein Propagates via the Lymphatic System of the Brain in the Monomeric State. Cell Rep 2023, 42, 112962. [Google Scholar] [CrossRef]
- De Luca, C. M. G.; Elia, A. E.; Portaleone, S. M.; Cazzaniga, F. A.; Rossi, M.; Bistaffa, E.; De Cecco, E.; Narkiewicz, J.; Salzano, G.; Carletta, O.; Romito, L.; Devigili, G.; Soliveri, P.; Tiraboschi, P.; Legname, G.; Tagliavini, F.; Eleopra, R.; Giaccone, G.; Moda, F. Efficient RT-QuIC Seeding Activity for α-Synuclein in Olfactory Mucosa Samples of Patients with Parkinson’s Disease and Multiple System Atrophy. Transl Neurodegener 2019, 8, 24. [Google Scholar] [CrossRef] [PubMed]
- Iranzo, A.; Mammana, A.; Muñoz-Lopetegi, A.; Dellavalle, S.; Mayà, G.; Rossi, M.; Serradell, M.; Baiardi, S.; Arqueros, A.; Quadalti, C.; Perissinotti, A.; Ruggeri, E.; Cano, JS.; Gaig, C.; Parchi, P. Misfolded α-Synuclein Assessment in the Skin and CSF by RT-QuIC in Isolated REM Sleep Behavior Disorder. Neurology 2023, 100, e1944–e1954. [Google Scholar] [CrossRef] [PubMed]
- https. [CrossRef]
- Fairfoul, G.; McGuire, L. I.; Pal, S.; Ironside, J. W.; Neumann, J.; Christie, S.; Joachim, C.; Esiri, M.; Evetts, S. G.; Rolinski, M.; Baig, F.; Ruffmann, C.; Wade-Martins, R.; Hu, M. T. M.; Parkkinen, L.; Green, A. J. E. Alpha-Synuclein RT-QuIC in the CSF of Patients with Alpha-Synucleinopathies. Ann Clin Transl Neurol 2016, 3, 812–818. [Google Scholar] [CrossRef]
- Orrú, CD.; Groveman, BR.; Hughson, AG.; Barrio, T.; Isiofia, K.; Race, B.; Ferreira, NC.; Gambetti, P.; Schneider, DA.; Masujin, K.; Miyazawa, K.; Ghetti, B.; Zanusso, G.; Caughey, B. Sensitive detection of pathological seeds of α-synuclein, tau and prion protein on solid surfaces. PLoS Pathog. 2024, 20, e1012175. [Google Scholar] [CrossRef]
- Okuzumi, A.; Hatano, T.; Fukuhara, T.; Ueno, S.; Nukina, N.; Imai, Y.; Hattori, N. α-Synuclein Seeding Assay Using RT-QuIC. Methods Mol Biol 2021, 2322, 3–16. [Google Scholar] [CrossRef]
- Bongianni, M.; Ladogana, A.; Capaldi, S.; Klotz, S.; Baiardi, S.; Cagnin, A.; Perra, D.; Fiorini, M.; Poleggi, A.; Legname, G.; Cattaruzza, T.; Janes, F.; Tabaton, M.; Ghetti, B.; Monaco, S.; Kovacs, G. G.; Parchi, P.; Pocchiari, M.; Zanusso, G. α-Synuclein RT-QuIC assay in cerebrospinal fluid of patients with dementia with Lewy bodies. Annals of clinical and translational neurology 2019, 6, 2120–2126. [Google Scholar] [CrossRef]
- Rossi, M.; Baiardi, S.; Teunissen, C. E.; Quadalti, C.; van de Beek, M.; Mammana, A.; Stanzani-Maserati, M.; Van der Flier, W. M.; Sambati, L.; Zenesini, C.; Caughey, B.; Capellari, S.; Lemstra, A. W.; Parchi, P. Diagnostic Value of the CSF α-Synuclein Re-al-Time Quaking-Induced Conversion Assay at the Prodromal MCI Stage of Dementia With Lewy Bodies. Neurology 2021, 97, e930–e940. [Google Scholar] [CrossRef] [PubMed]
- Garrido, A.; Fairfoul, G.; Tolosa, E. S.; Martí, M. J.; Green, A. Barcelona LRRK2 Study Group. α-Synuclein RT-QuIC in Cere-brospinal Fluid of LRRK2-Linked Parkinson’s Disease. Ann Clin Transl Neurol 2019, 6, 1024–1032. [Google Scholar] [CrossRef] [PubMed]
- Manne, S.; Kondru, N.; Hepker, M.; Jin, H.; Anantharam, V.; Lewis, M.; Huang, X.; Kanthasamy, A.; Kanthasamy, A. G. Ul-trasensitive Detection of Aggregated α-Synuclein in Glial Cells, Human Cerebrospinal Fluid, and Brain Tissue Using the RT-QuIC Assay: New High-Throughput Neuroimmune Biomarker Assay for Parkinsonian Disorders. J Neuroimmune Pharmacol 2019, 14, 423–435. [Google Scholar] [CrossRef]
- Barinova, K. V.; Kuravsky, M. L.; Arutyunyan, A. M.; Serebryakova, M. V.; Schmalhausen, E. V.; Muronetz, V. I. Dimerization of Tyr136Cys Alpha-Synuclein Prevents Amyloid Transformation of Wild Type Alpha-Synuclein. Int J Biol Macromol 2017, 96, 35–43. [Google Scholar] [CrossRef] [PubMed]
- LAEMMLI, U. K. Cleavage of Structural Proteins during the Assembly of the Head of Bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Leisi, E. V.; Barinova, K. V.; Kudryavtseva, S. S.; Moiseenko, A. V.; Muronetz, V. I.; Kurochkina, L. P. Effect of Bacteriophage-Encoded Chaperonins on Amyloid Transformation of α-Synuclein. Biochem Biophys Res Commun 2022, 622, 136–142. [Google Scholar] [CrossRef]
- Sun, Y.; Hou, S.; Zhao, K.; Long, H.; Liu, Z.; Gao, J.; Zhang, Y.; Su, X.-D.; Li, D.; Liu, C. Cryo-EM Structure of Full-Length α-Synuclein Amyloid Fibril with Parkinson’s Disease Familial A53T Mutation. Cell Res 2020, 30, 360–362. [Google Scholar] [CrossRef]
- Sidhu, A.; Segers-Nolten, I.; Raussens, V.; Claessens, M. M. A. E.; Subramaniam, V. Distinct Mechanisms Determine α-Synuclein Fibril Morphology during Growth and Maturation. ACS Chem Neurosci 2017, 8, 538–547. [Google Scholar] [CrossRef]
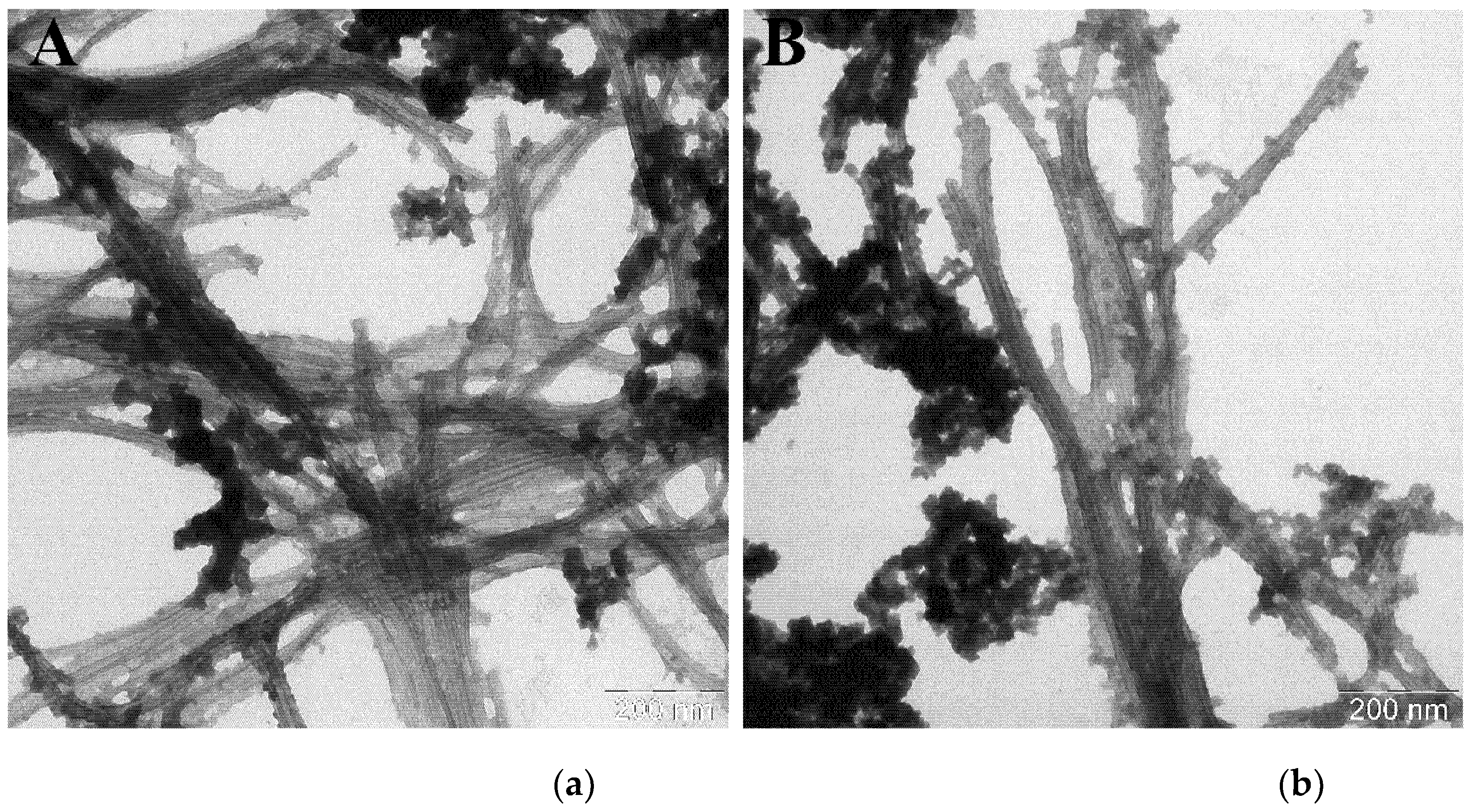
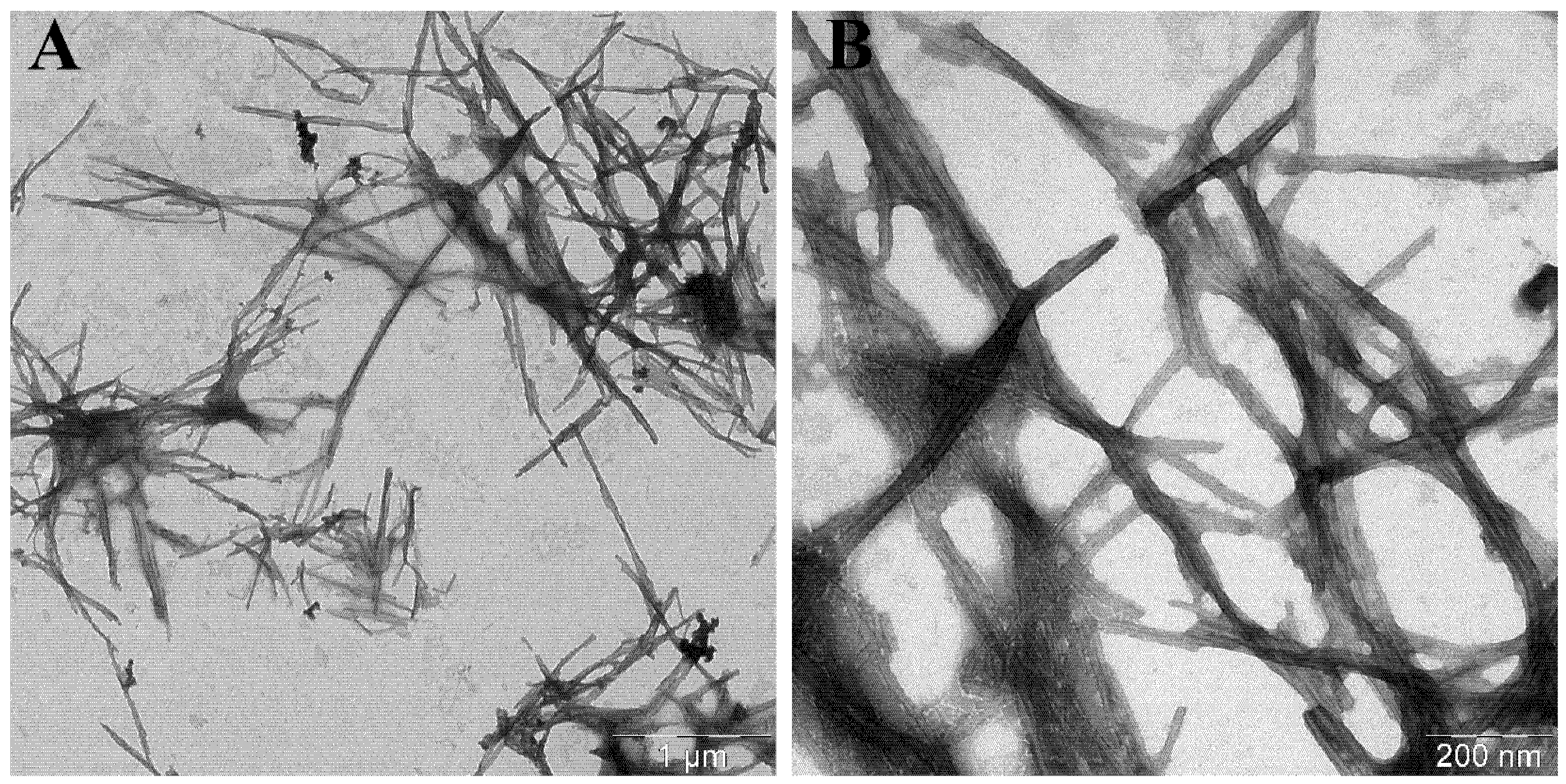



| 0 h | 20 h | 25 h | 43 h | 67 h | |
| α-synWT | ~2-7 | ~2-6; | ~1000-2500 | ~1000-2000 | ~3-16 |
| ~3000 | |||||
| α-synWT + WT_seeds | ~4-7 | ~1000-3000 | ~1-2; | ~1-2; | ~1-2; |
| ~300-800 | ~15-16; | ~4-8 | |||
| ~100-200; | |||||
| ~600-700 | |||||
| α-synWT + A53T_seeds | ~1-5 | ~1-5; | ~200-1300 | ~1-6; | ~1-2 |
| ~2000 | ~300 | ||||
| α-synA53T | ~1-2 | ~800-2500 | ~800-2500 | ~1500-4200 | ~600-1500 |
| α-synA53T + A53T_seeds | ~1-7 | ~1-7 | ~1-2; | ~700-3000 | ~10; |
| ~300-800 | ~800-2000 | ||||
| α-synA53T + WT_seeds | ~2; | ~3000-4000 | ~500-1800 | ~200-1300 | ~2000-4000 |
| ~2000-3000 |
| Average length, nm | Average width ± SD, nm | |
| α-synWT [27] | ~ 200 | 10.0 ± 1 |
| α-synWT + WT_seeds | 600 ± 50 | 12.9 ± 1.1 |
| α-synWT + А53Т_seeds | 770 ± 70 | 5.4 ± 1.0 |
| α-synA53T [28,29] | ~ 350 | 6.0 ± 0.6 |
| α-synA53T + А53Т_seeds | 550 ± 40 | 9.6 ± 1.1 |
| α-synA53T + WT_seeds | 1800 ± 140 | 11.7 ± 1.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




