Submitted:
13 March 2026
Posted:
17 March 2026
You are already at the latest version
Abstract
Keywords:
Biosynthetic and Phylogenetic Evidence of Mutual Pigment Exclusion
Questionable Claims of the Presence of Anthocyanins in Well-Known Betalain-Pigmented Plants
- (1)
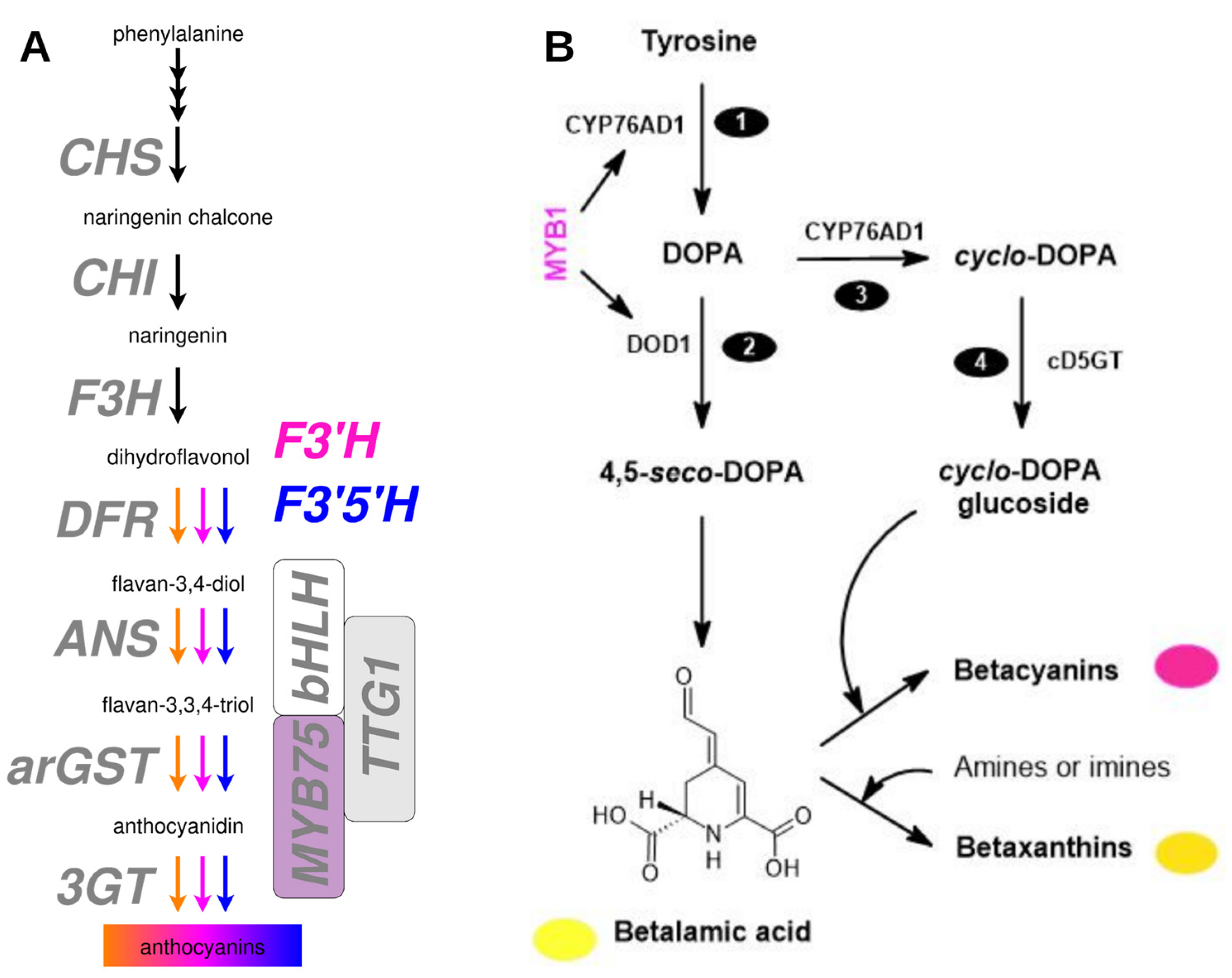
- A recurring issue in all the studies listed in Table S1 is the use of inappropriate analytical approaches or the lack of thorough methods. Spectrophotometric quantification without further confirmation using HPLC-MS/MS following the guidelines laid down by Metabolomics Standard Initiative (Sumner et al., 2007) is a frequent issue (Table S1). Authentication of the pigment identity is required, because anthocyanins and betalains absorb light around 530 ± 10 nm and both pigments are hydrophilic and show color at pH 5-6 or pH ﹤4 (Stintzing and Carle, 2004). A simple approach to differentiate the two pigments is based on the knowledge that anthocyanin extracts turn blue in alkaline pH, whereas betalain extracts are yellow/brown at the same pH. This can be easily carried out by adding a small amount of NaOH to the extract. Further, when the pH is made acidic, anthocyanin extracts regain the original extract color instantaneously, whereas betalain extracts do not do so immediately. Given that betalains would be an adequate explanation for the coloration of the plant species listed in Table S1, claims regarding the presence of anthocyanins in these betalain-pigmented taxa remain unsubstantiated and are inconsistent with the compelling evidence in the scientific literature.
- (2)
- Support for the presence of anthocyanins in betalain-pigmented lineages is often drawn from controversial or retracted publications. For example, reports claiming the presence of anthocyanins in betalain-pigmented pitayas have been criticized(Pucker et al., 2021; Khan, 2022; Pucker and Brockington, 2022) and are retracted or in the retraction process, but are still frequently cited.
- (3)
- Mis-interpretation of transcriptomic data. For example, a truncated transcript of an anthocyanidin synthase is highly abundant in the betalain-pigmented Mirabilis jalapa even though it does not result in a functional enzyme (Polturak et al., 2018). Some genes of the anthocyanin biosynthesis are still present and even expressed in betalain-pigmented lineages, but with at least one gene missing the anthocyanin biosynthesis is blocked (Pucker et al., 2024). The detection of transcripts of genes from the wider flavonoid biosynthesis is often mistaken for evidence of an active anthocyanin biosynthesis, because the activity of these genes in other pathways is ignored.
- (4)
- The questionable studies were published between 2008 and 2026 in journals operating under standard peer-review procedures. While these outlets are established, they are not generally regarded as leading or highly selective journals. The acceptance of claims that contradict the well-documented mutual exclusivity of anthocyanins and betalains raises concerns regarding the consistency and subject-specific rigor of the review process. In particular, it suggests that the evaluation may not have consistently involved reviewers with specialized expertise.
Concluding Remarks
References
- Grünig, N., Horz, J. M., and Pucker, B. (2025). Diversity and ecological functions of anthocyanins. BMC Plant Biol. 26:146.
- Hatlestad, G. J., Akhavan, N. A., Sunnadeniya, R. M., Elam, L., Cargile, S., Hembd, A., Gonzalez, A., McGrath, J. M., and Lloyd, A. M. (2015). The beet Y locus encodes an anthocyanin MYB-like protein that activates the betalain red pigment pathway. Nat. Genet. 47:92–96.
- Khan, M. I. (2022). The reported colour formation mechanism in pitaya fruit through co-accumulation of anthocyanins and betalains is inconsistent and fails to establish the co-accumulation. BMC Genomics 23:740.
- Khan, M. I., and Giridhar, P. (2015). Plant betalains: Chemistry and biochemistry. Phytochemistry 117:267–295.
- Lopez-Nieves, S., Yang, Y., Timoneda, A., Wang, M., Feng, T., Smith, S. A., Brockington, S. F., and Maeda, H. A. (2018). Relaxation of tyrosine pathway regulation underlies the evolution of betalain pigmentation in Caryophyllales. New Phytol. 217:896–908.
- Polturak, G., Heinig, U., Grossman, N., Battat, M., Leshkowitz, D., Malitsky, S., Rogachev, I., and Aharoni, A. (2018). Transcriptome and Metabolic Profiling Provides Insights into Betalain Biosynthesis and Evolution in Mirabilis jalapa. Mol. Plant 11:189–204.
- Pucker, B., and Brockington, S. F. (2022). The evidence for anthocyanins in the betalain-pigmented genus Hylocereus is weak. BMC Genomics 23:739.
- Pucker, B., Singh, H. B., Kumari, M., Khan, M. I., and Brockington, S. F. (2021). The report of anthocyanins in the betalain-pigmented genus Hylocereus is not well evidenced and is not a strong basis to refute the mutual exclusion paradigm. BMC Plant Biol. 21:297.
- Pucker, B., Walker-Hale, N., Dzurlic, J., Yim, W. C., Cushman, J. C., Crum, A., Yang, Y., and Brockington, S. F. (2024). Multiple mechanisms explain loss of anthocyanins from betalain-pigmented Caryophyllales, including repeated wholesale loss of a key anthocyanidin synthesis enzyme. New Phytol. 241:471–489.
- Sheehan, H., Feng, T., Walker-Hale, N., Lopez-Nieves, S., Pucker, B., Guo, R., Yim, W. C., Badgami, R., Timoneda, A., Zhao, L., et al. (2020). Evolution of l-DOPA 4,5-dioxygenase activity allows for recurrent specialisation to betalain pigmentation in Caryophyllales. New Phytol. 227:914–929.
- Stintzing, F. C., and Carle, R. (2004). Functional properties of anthocyanins and betalains in plants, food, and in human nutrition. Trends Food Sci. Technol. 15:19–38.
- Sumner, L. W., Amberg, A., Barrett, D., Beale, M. H., Beger, R., Daykin, C. A., Fan, T. W.-M., Fiehn, O., Goodacre, R., Griffin, J. L., et al. (2007). Proposed minimum reporting standards for chemical analysis Chemical Analysis Working Group (CAWG) Metabolomics Standards Initiative (MSI). Metabolomics Off. J. Metabolomic Soc. 3:211–221.
- Timoneda, A., Feng, T., Sheehan, H., Walker-Hale, N., Pucker, B., Lopez-Nieves, S., Guo, R., and Brockington, S. (2019). The evolution of betalain biosynthesis in Caryophyllales. New Phytol. 224:71–85.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



