Submitted:
12 March 2026
Posted:
16 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
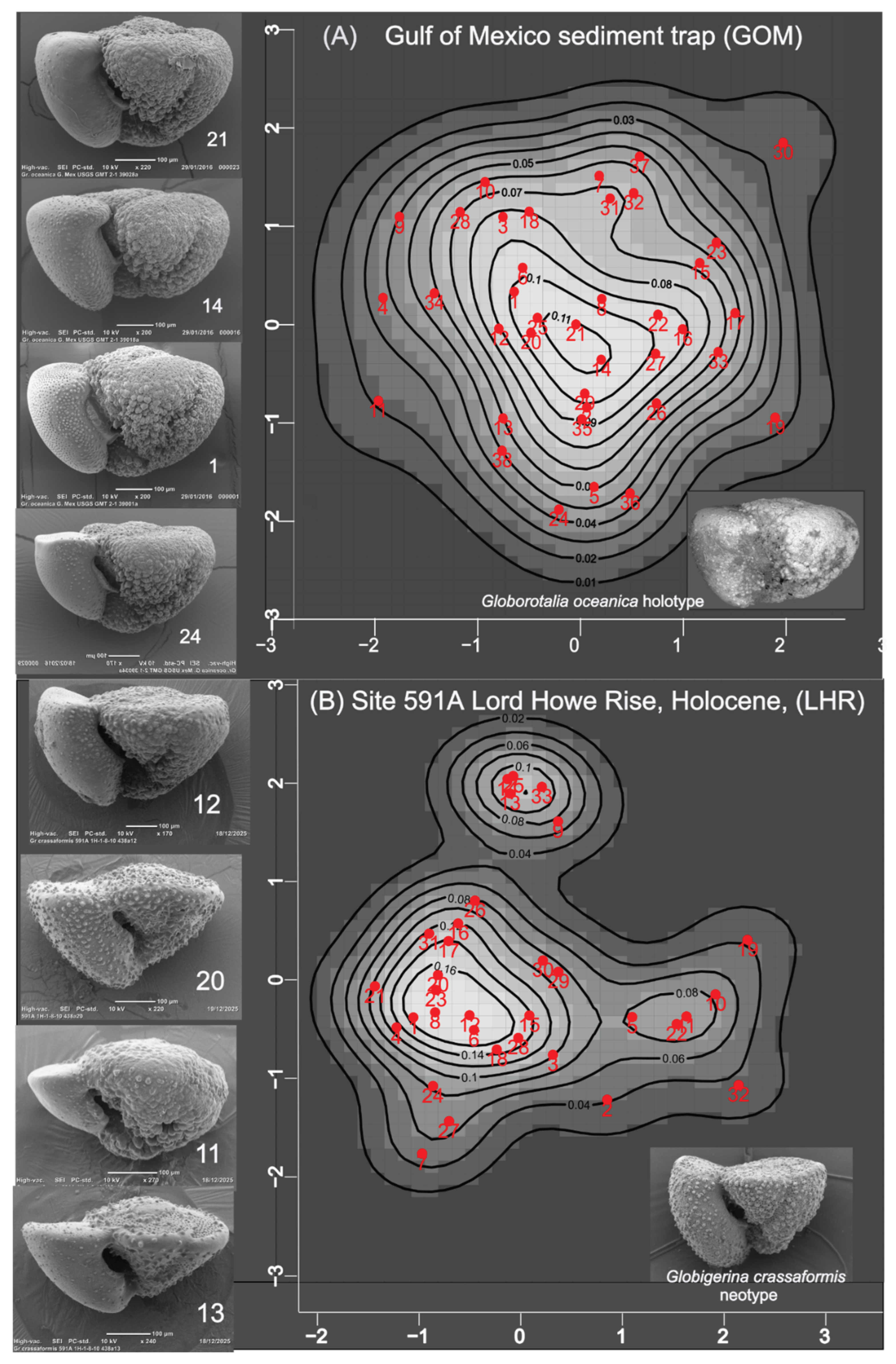
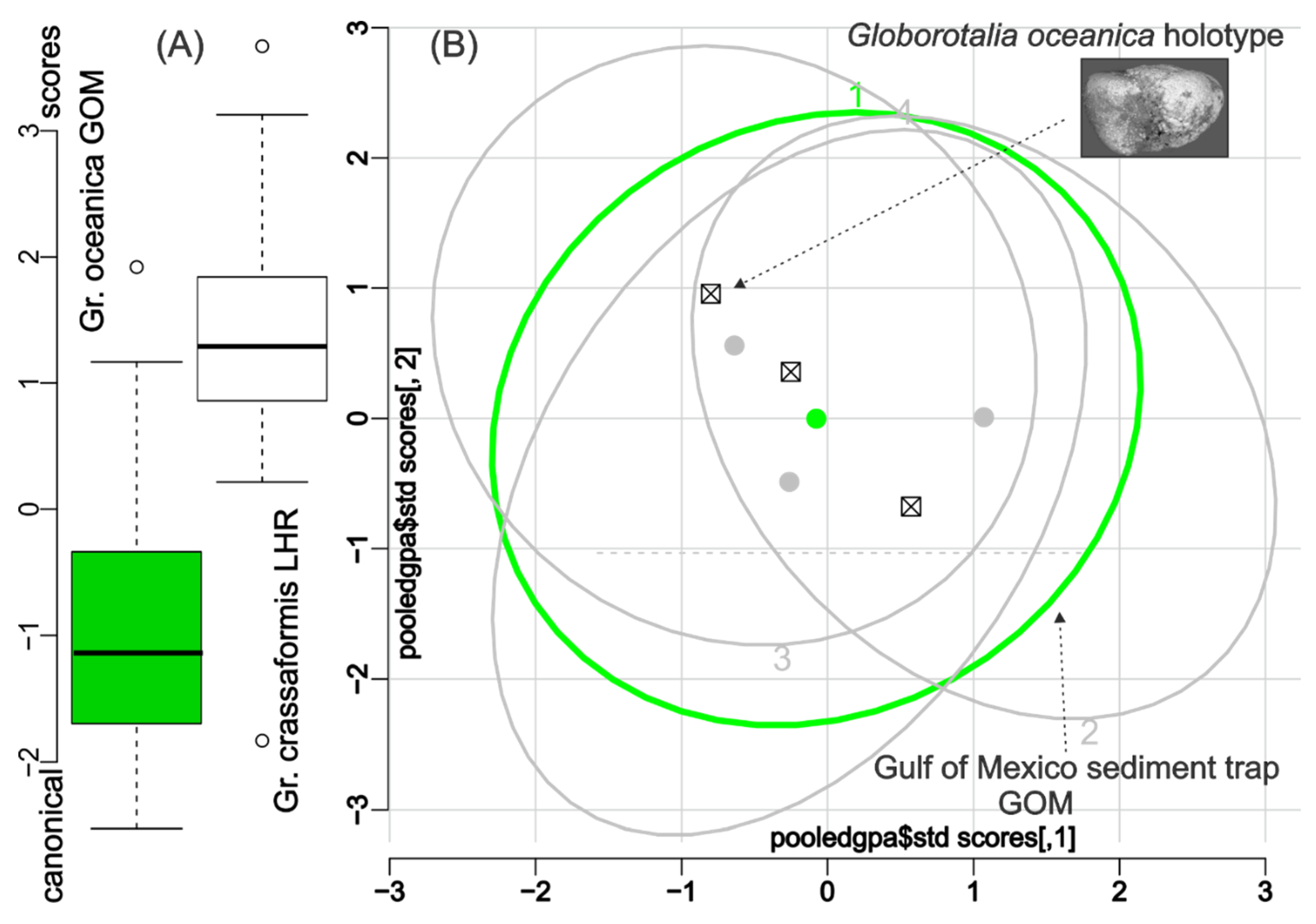
3. Results
4. Discussion
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Witteveen, J. Suppressing Synonymy with a Homonym: The Emergence of the Nomenclatural Type Concept in Nineteenth Century Natural History. J Hist Biol 2016, 49, 135–189. [Google Scholar] [CrossRef]
- Hooker, Joseph Dalton. Flora of New Zealand; Lovell Reeve: London, 1853. [Google Scholar]
- Strickland, Hugh Edward. On the Inexpediency of Altering Estabilished Terms in Natural History. Magazine of Natural History and Journal of Zoology, Botany, mineralogy, geology and meteorology 1837, n.s. 1, 127–131. [Google Scholar]
- International Commission on Zoological Nomenclature, Ride, W. D. L., International Trust for Zoological Nomenclature, Natural History Museum (London, England), International Union of Biological Sciences. International Code of Zoological Nomenclature: Code International de Nomenclature Zoologique, 4th ed.; International Trust for Zoological Nomenclature, Natural History Museum (London, England), International Union of Biological Sciences: London; International Trust for Zoological Nomenclature, c/o Natural History Museum, 1999. [Google Scholar]
- Burgin, C. J.; Zijlstra, J. S.; Becker, M. A.; Handika, H.; Alston, J. M.; Widness, J.; Liphardt, S.; Huckaby, D. G.; Upham, N. S. How Many Mammal Species Are There Now? Updates and Trends in Taxonomic, Nomenclatural, and Geographic Knowledge. Journal of Mammalogy 2025, 106, 1082–1117. [Google Scholar] [CrossRef]
- Alroy, J. How Many Named Species Are Valid? Proc. Natl. Acad. Sci. U.S.A. 2002, 99, 3706–3711. [Google Scholar] [CrossRef] [PubMed]
- Garrity, G. M.; Lyons, C. Future-Proofing Biological Nomenclature. OMICS: A Journal of Integrative Biology 2003, 7, 31–33. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, J. Supporting Taxonomic Names in Cell and Molecular Biology Databases. OMICS: A Journal of Integrative Biology 2003, 7, 13–16. [Google Scholar] [CrossRef] [PubMed]
- Rao, M. K. V. The Importance of Botanical Nomenclature and Synonymy in Taxonomy and Biodiversity. Current Science 2004, 87, 602–606. [Google Scholar]
- Benton, M. J. How to Find a Dinosaur, and the Role of Synonymy in Biodiversity Studies. Paleobiology 2008, 34, 516–533. [Google Scholar] [CrossRef]
- Solow, A. R.; Benton, M. J. On the Flux Ratio Method and the Number of Valid Species Names. Paleobiology 2010, 36, 516–518. [Google Scholar] [CrossRef]
- Mora, C.; Tittensor, D. P.; Adl, S.; Simpson, A. G. B.; Worm, B. How Many Species Are There on Earth and in the Ocean? PLoS Biol 2011, 9, e1001127. [Google Scholar] [CrossRef]
- Garnett, S. T.; Christidis, L. Taxonomy Anarchy Hampers Conservation. Nature 2017, 546, 25–27. [Google Scholar] [CrossRef]
- Hobern, D.; Barik, S. K.; Christidis, L.; T.Garnett, S.; Kirk, P.; Orrell, T. M.; Pape, T.; Pyle, R. L.; Thiele, K. R.; Zachos, F. E.; Bánki, O. Towards a Global List of Accepted Species VI: The Catalogue of Life Checklist. Org Divers Evol 2021, 21, 677–690. [Google Scholar] [CrossRef]
- Garnett, S. T.; Bánki, O.; Barik, S. K.; Berryman, A. J.; Bouchard, P.; Buckeridge, J.; Christidis, L.; Cigliano, M. M.; Conix, S.; Crawford-Weaver, H.; Van Dijk, P. P.; Evenhuis, N. L.; Hilton-Taylor, C.; Hobern, D.; Johnston, C.; Klopper, R. R.; Kroh, A.; Le Roux, M.; Pape, T.; Pyle, R. L.; Raz, L.; Thomas, P.; Thomson, S.; Vandepitte, L.; Wambiji, N.; Zachos, F. E.; Lien, A. M. Measuring the Quality of Species List Governance. BioScience 2026, biaf071. [Google Scholar] [CrossRef]
- Schiebel, R. Planktic Foraminiferal Sedimentation and the Marine Calcite Budget. Global Biogeochemical Cycles 2002, 16. [Google Scholar] [CrossRef]
- Schiebel, R.; Hemleben, C. Planktic Foraminifers in the Modern Ocean; Springer: Berlin, 2017. [Google Scholar]
- Brummer, G-A.; Kučera, M. Taxonomic Review of Living Planktonic Foraminifera. Journal of Micropalaeontology 2022, 41, 29–74. [Google Scholar] [CrossRef]
- Cushman, J.A.; Bermudez, P.J. Some Cuban Species of Globorotalia. Contributions from the Cushman Laboratory for Foraminiferal Research 1949, 25, 26–45. [Google Scholar]
- Galloway, J.J.; Wissler, S.G. Pleistocene Foraminifera from the Lomita Quarry, Palos Verde Hills, California. Journal of Paleontology 1927, 1, 35–87. [Google Scholar]
- Richey, J.; Reynolds, C.; Thunell, R. Weekly Resolution Particulate Flux from a Sediment Trap in the Northern Gulf of Mexico, 2008-2012. USGS Open-File Report 2014, 2014–1035, 9. [Google Scholar]
- Nelson, C.S.; Hendy, C.H.; Cuthbertson, A.M. Compendium of Stable Oxygen and Carbon Isotope Data for the Late Quaternary Interval of Deep-Sea Cores from the New Zealand Sector of the Tasman Sea and Southwest Pacific Ocean; Occasional Report; University of Waikato, Department of Earth Sciences: Hamilton, New Zealand, 1993. [Google Scholar]
- Scott, G. H. Representing Zooplankters: An Example from the Foraminifera. Geosciences 2024, 14, 169. [Google Scholar] [CrossRef]
- Beckner, M. The Biological Way of Thought; Columbia University Press, 1959. [Google Scholar] [CrossRef]
- Boyd, R. Homeostasis, Species, and Higher Taxa. In Species; Wilson, R. A., Ed.; The MIT Press, 1999; pp. pp 141–186. [Google Scholar] [CrossRef]
- Simpson, G. G. Principles of Animal Taxonomy; Columbia University Press, 1961. [Google Scholar] [CrossRef]
- Karlin, S. R. A. Fisher and Evolutionary Theory. Statist. Sci. 1992, 7. [Google Scholar] [CrossRef]
- Mayr, E. Principles of Systematic Zoology; McGraw-Hill: New York, 1969. [Google Scholar]
- Aze, T.; Ezard, T. H. G.; Purvis, A.; Coxall, H. K.; Stewart, D. R. M.; Wade, B. S.; Pearson, P. N. A Phylogeny of Cenozoic Macroperforate Planktonic Foraminifera from Fossil Data. Biol Rev Camb Philos Soc 2011, 86, 900–927. [Google Scholar] [CrossRef] [PubMed]
- Walton, D. Argumentation Theory: A Very Short Introduction. In Argumentation in Artificial Intelligence; Simari, G., Rahwan, I., Eds.; Springer US: Boston, MA, 2009; pp. pp 1–22. [Google Scholar] [CrossRef]
- Walton, D. Examination Dialogue: An Argumentation Framework for Critically Questioning an Expert Opinion. Journal of Pragmatics 2006, 38, 745–777. [Google Scholar] [CrossRef]
- Caley, M. J.; O’Leary, R. A.; Fisher, R.; Low-Choy, S.; Johnson, S.; Mengersen, K. What Is an Expert? A Systems Perspective on Expertise. Ecology and Evolution 2014, 4, 231–242. [Google Scholar] [CrossRef]
- Vandepitte, L.; Dekeyzer, S.; Decock, W.; Delgat, L.; Boydens, B.; Tyberghein, L.; Vanhoorne, B.; Hernandez, F.; Mees, J.; Horton, T.; Kroh, A.; Boyko, C. B.; Ahyong, S. T. The World Register of Marine Species (WoRMS) through the Looking Glass: Insights from the Data Management Team in Light of the Crystal Anniversary of WoRMS. Hydrobiologia 2025, 852, 1–22. [Google Scholar] [CrossRef]
- Huber, R.; Klump, J. Charting Taxonomic Knowledge through Ontologies and Ranking Algorithms. Computers & Geosciences 2009, 35, 862–868. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



