Submitted:
12 March 2026
Posted:
17 March 2026
You are already at the latest version
Abstract
Keywords:
Introduction
1. Part I: Local Sampling and Processing – Fast, Reliable Encoding of the Variable World
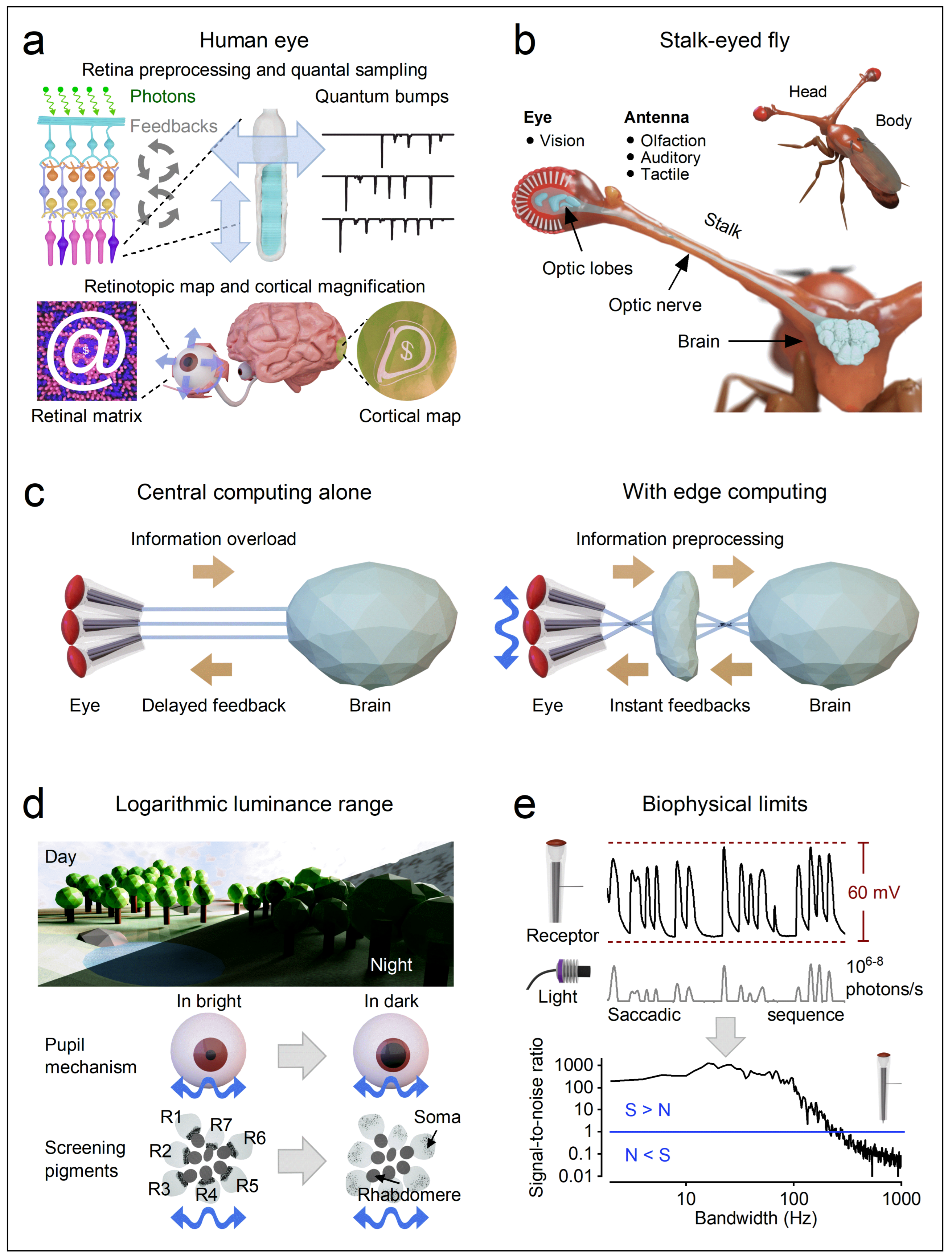
1.1. Historic Recap: Neural Models with Additive Noise and Stationarity Assumptions
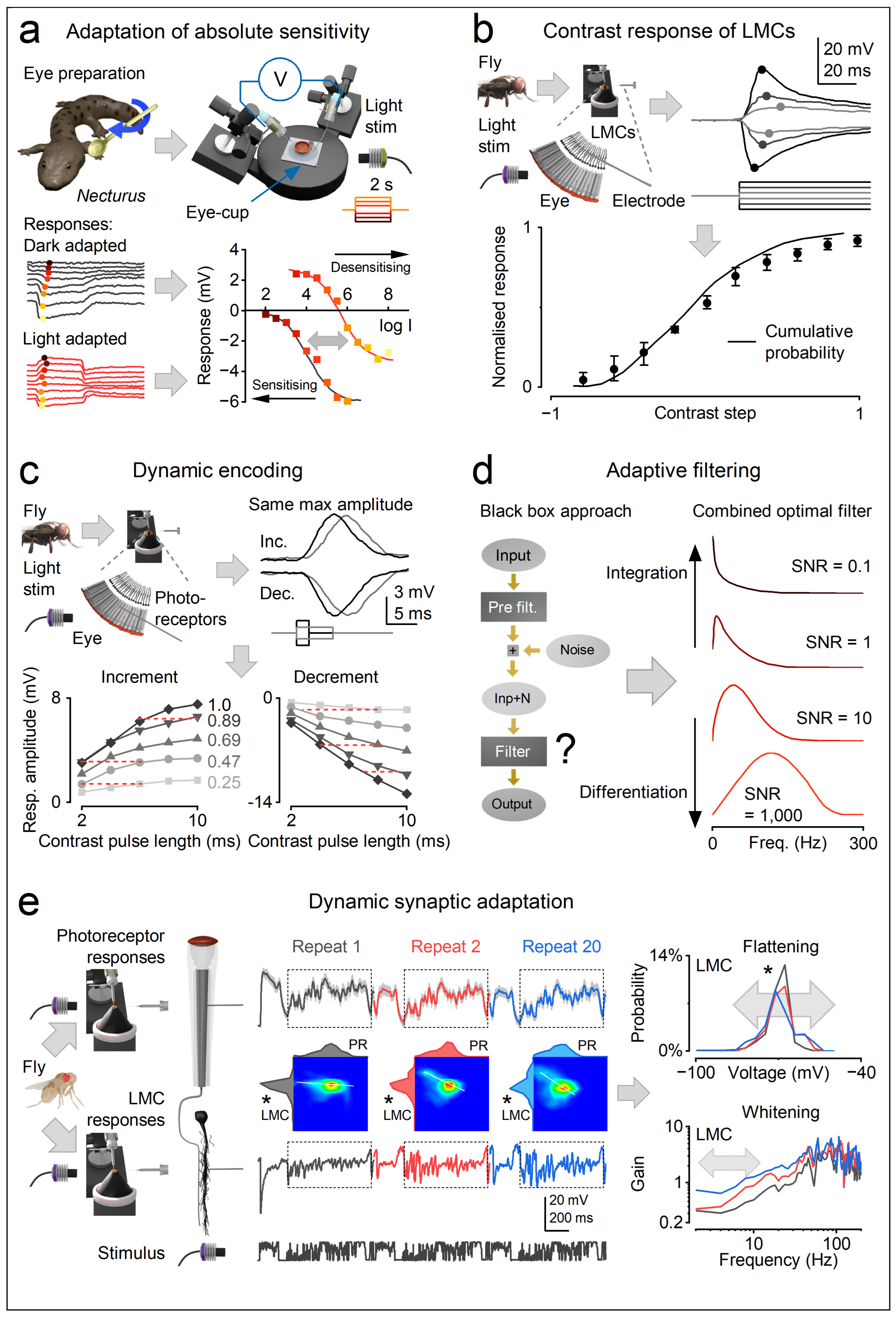
1.2. Information Capture Through Morphodynamic Parallel Sampling
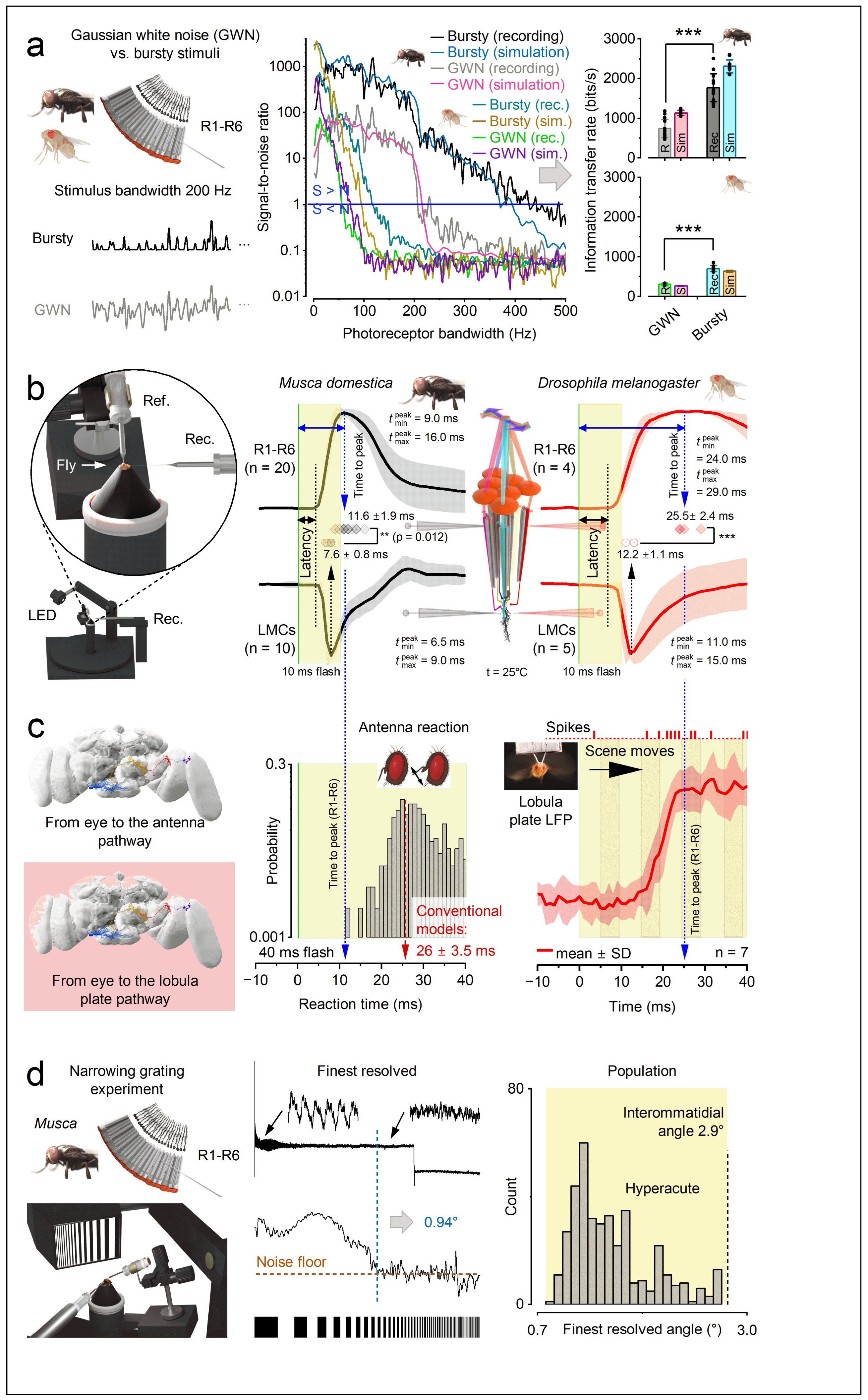
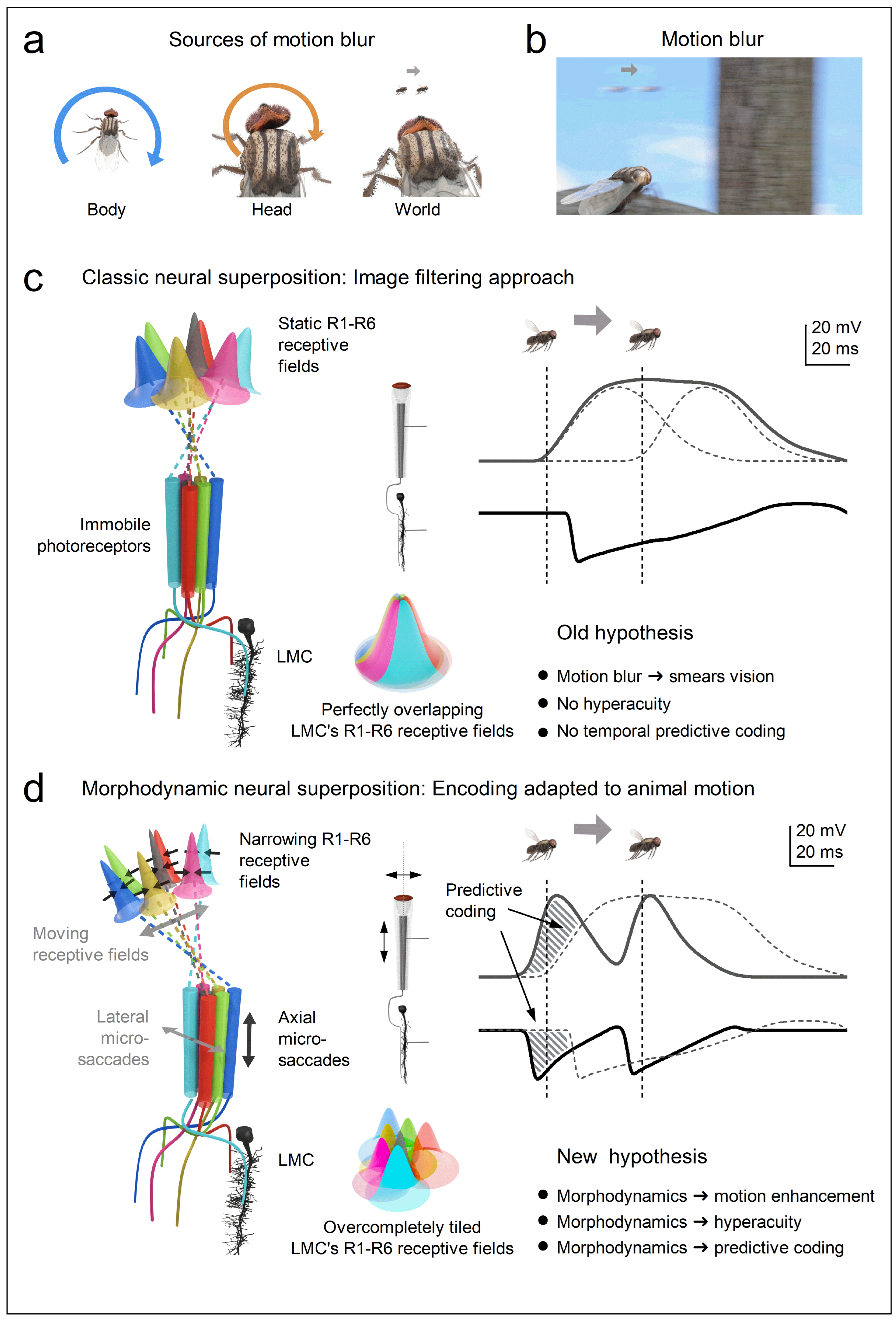
- Photomechanical transduction - Reactions inside microvilli induce photoreceptor microsaccades [23,24,26,129] that enhance visual acuity by encoding space in time through rapid, auto-regulated (adaptive) photomechanical motion [23,24,25,26,27,28,29,129]. The brighter the light change, the larger the movement [23,24,26,27]. These microsaccades shift and narrow the photoreceptor’s receptive field [24,26,27], thereby driving predictive coding (Figure 4c and Figure e), especially during saccadic behaviours [26,27].
- Contrast constancy (signal normalisation and response invariance) - Refractory quantal sampling implements divisive normalisation [31,32], maintaining consistent waveforms across wide (logarithmic) intensity ranges [49,80,115,130], so that photoreceptors evoke similarly shaped responses to contrast changes under different illumination conditions.
- Saliency enhancement - Because of refractory sampling units, novel or surprising inputs are naturally emphasised [3,7,31,32]. Events that cause the largest changes in microvillar activation (bump-rate increments or decrements), such as those triggered by saccadic eye movements, produce macroscopic voltage responses with the greatest amplitude and frequency utilisation, and thus the highest information content [3,7,31,115].
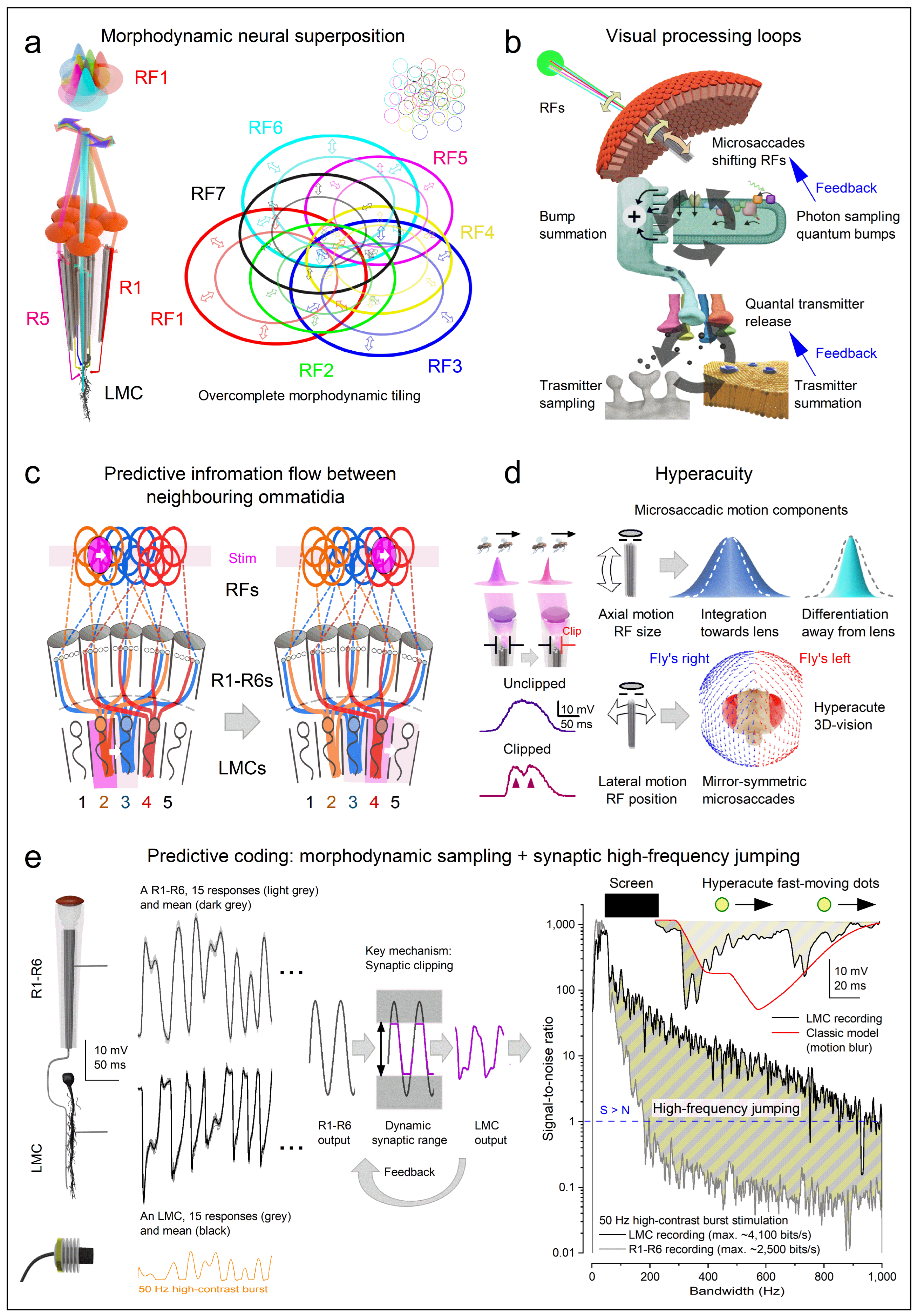
1.3. Structure and Function in Photoreceptors
1.3.1. Microvilli Are Photomechanical (Rapidly Moving) Sampling Units
1.3.2. Morphodynamic Rules for Photoreceptor Light Sampling
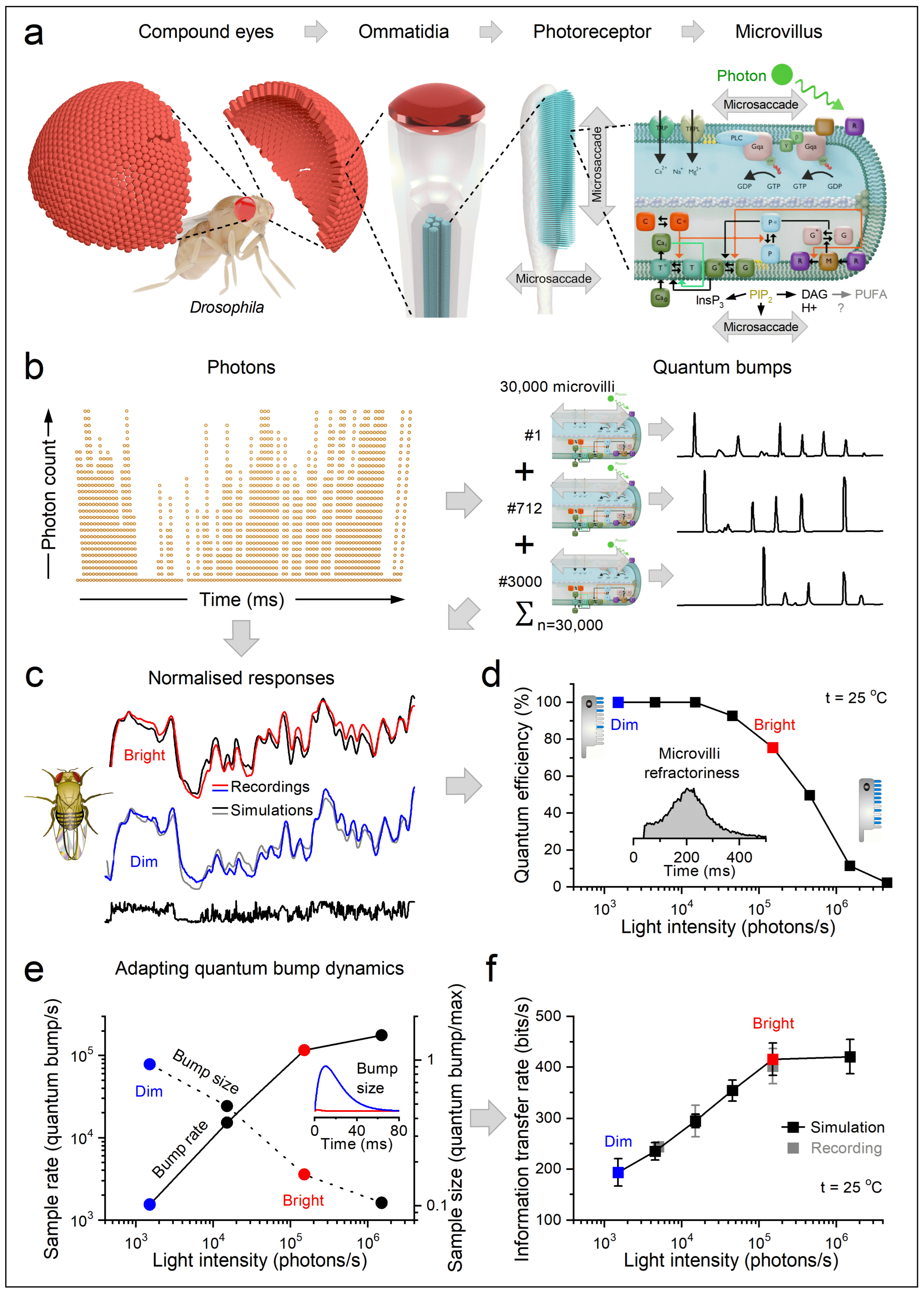
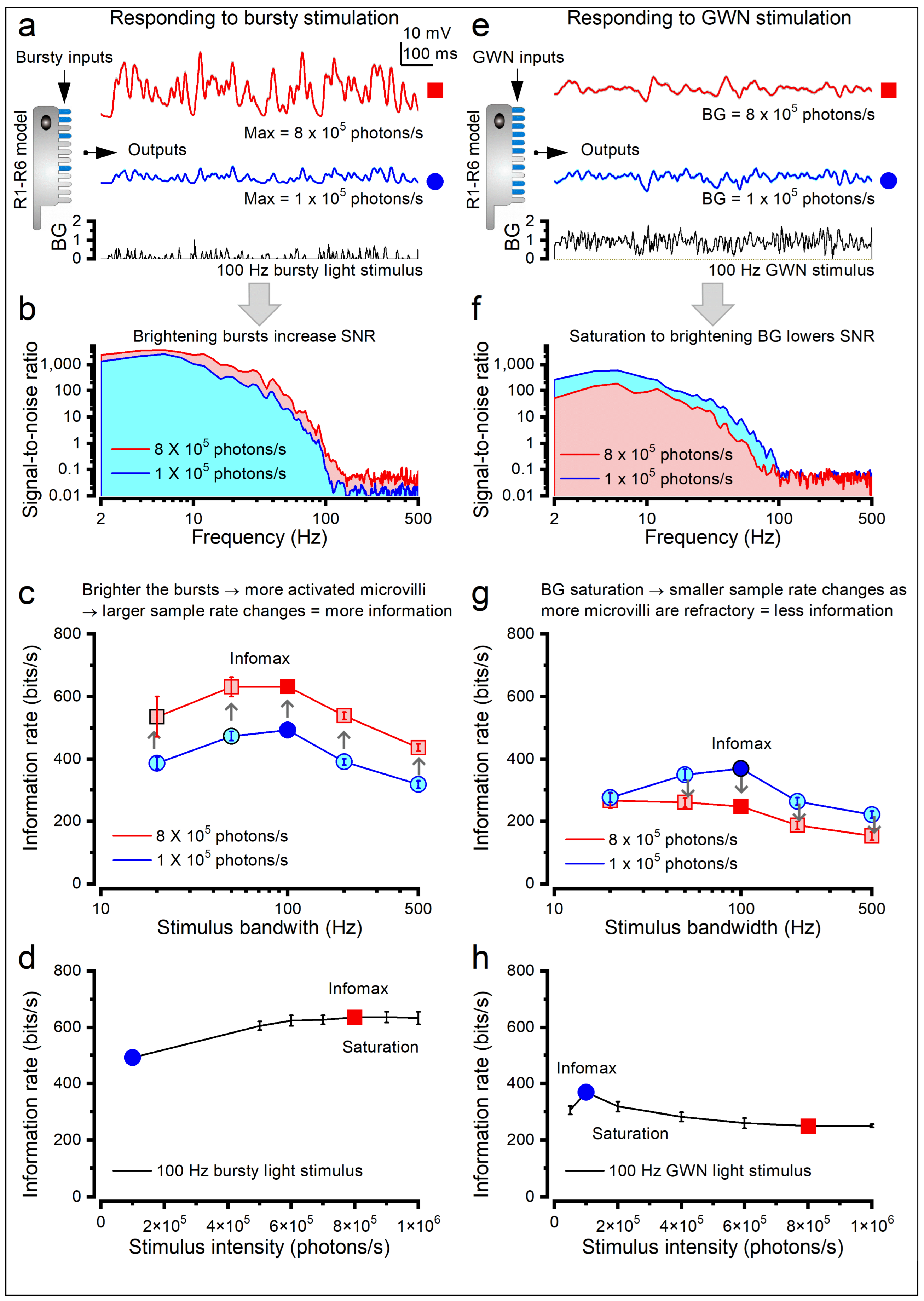
1.3.3. What Governs the Information Transfer Rate of Macroscopic Responses?
- Number of microvilli per rhabdomere (~30,000 in case of Drosophila R1-R6 photoreceptors)
- Quantum-bump waveform (average shape and duration at ambient intensity)
- Latency distribution (timing jitter before each bump)
- Refractoriness distribution (duration of microvillar unresponsiveness after a bump)
1.3.4. What Are the Benefits and Costs of Adaptive Stochastic Sampling?
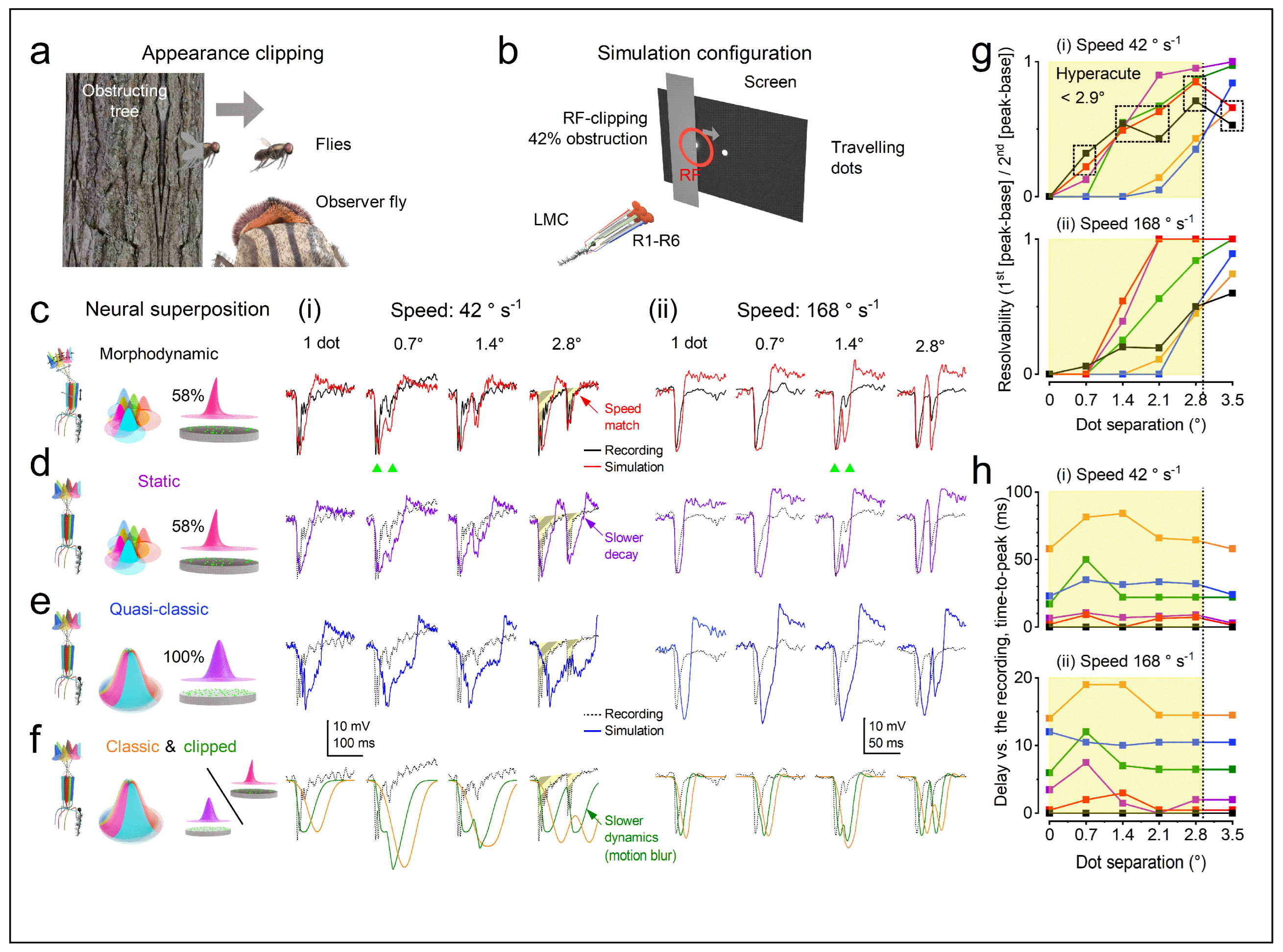
1.3.5. Photoreceptor Structure Defines Its Encoding Performance
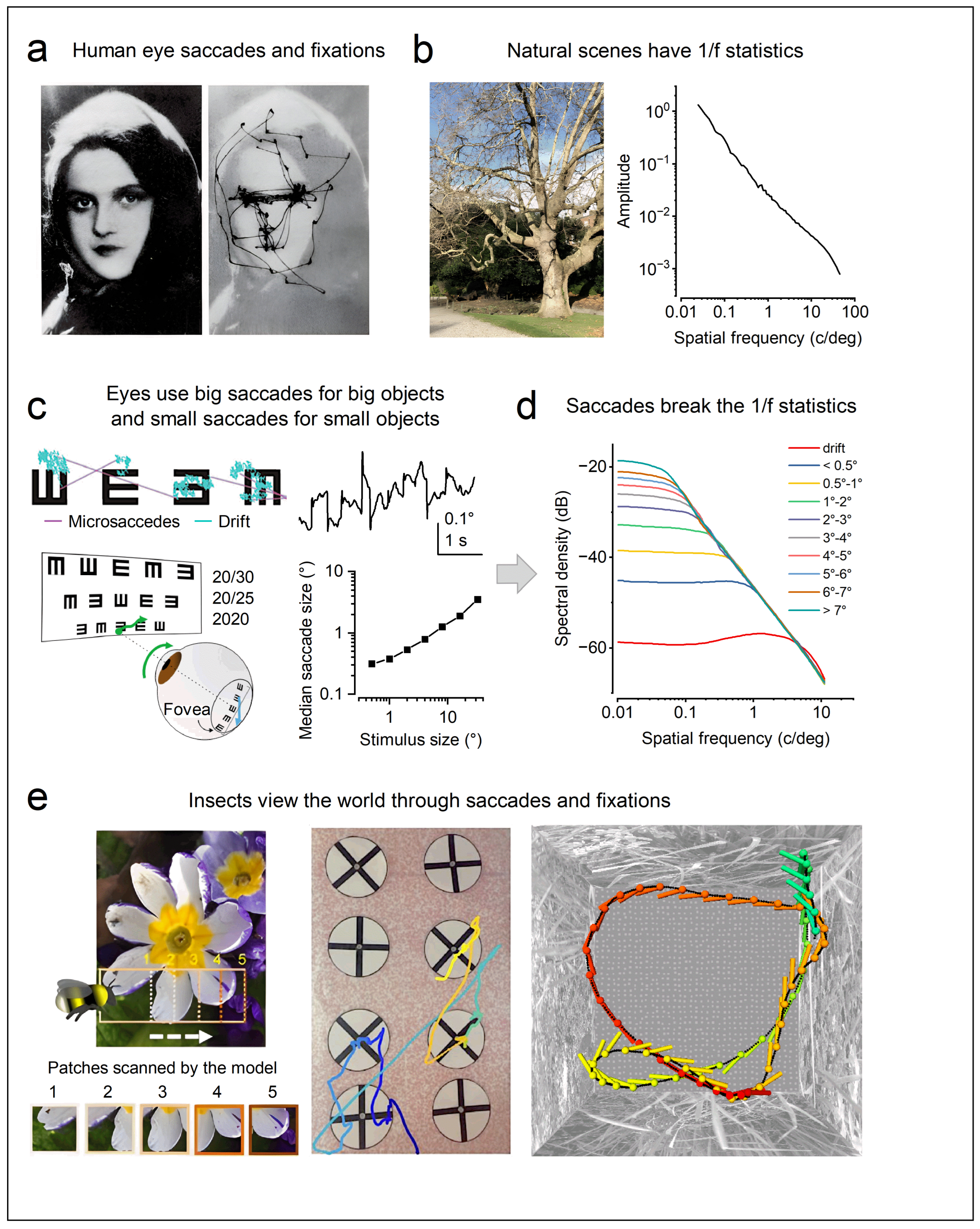
1.4. Active Sensing Drives Both Efficient Information Sampling and Predictive Coding
1.5. Active Sensing Adapts to the Natural Environment to Enhance Spatiotemporal Resolutions
2. Section II: Global Information Representation Ties Motion to Encoding and Perception
2.1. Genetic Scaffolding and Active Sensing Enable Semantic Encoding
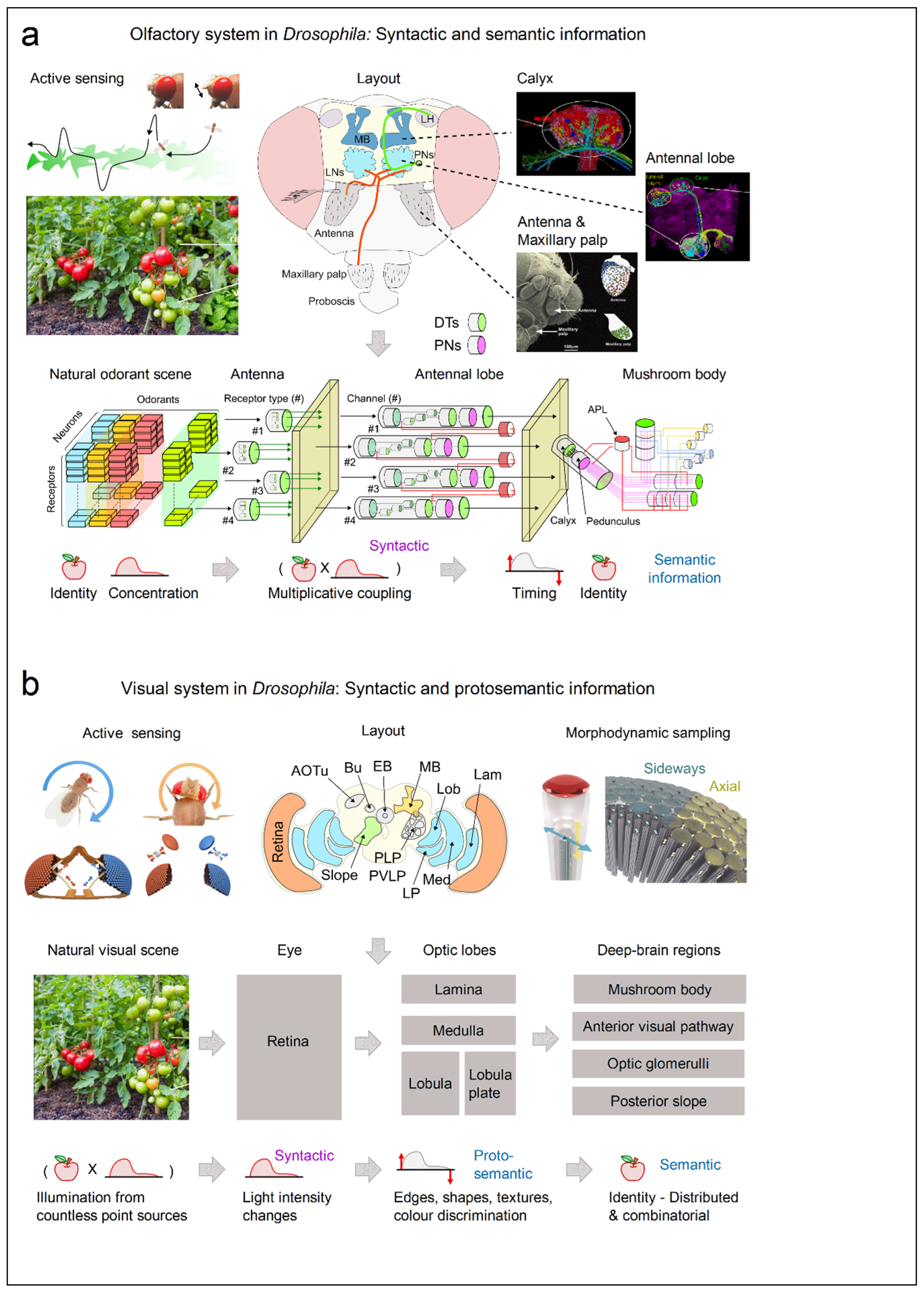
2.2. Shared Design Principles for Representing Semantic/Syntactic Sensory Information
2.3. Active Sensing Enables Efficient Extraction of Phasic Information from Natural Images in Time
2.4. Multiscale Motion in Neural Code – A Foundation for Biological Language
2.5. Dimensionality Expansion in Brain Network Sampling Matrices and Resolution Limits
2.6. Single-Neuron Encoding Is Precise, Yet Every Neuron Responds Uniquely
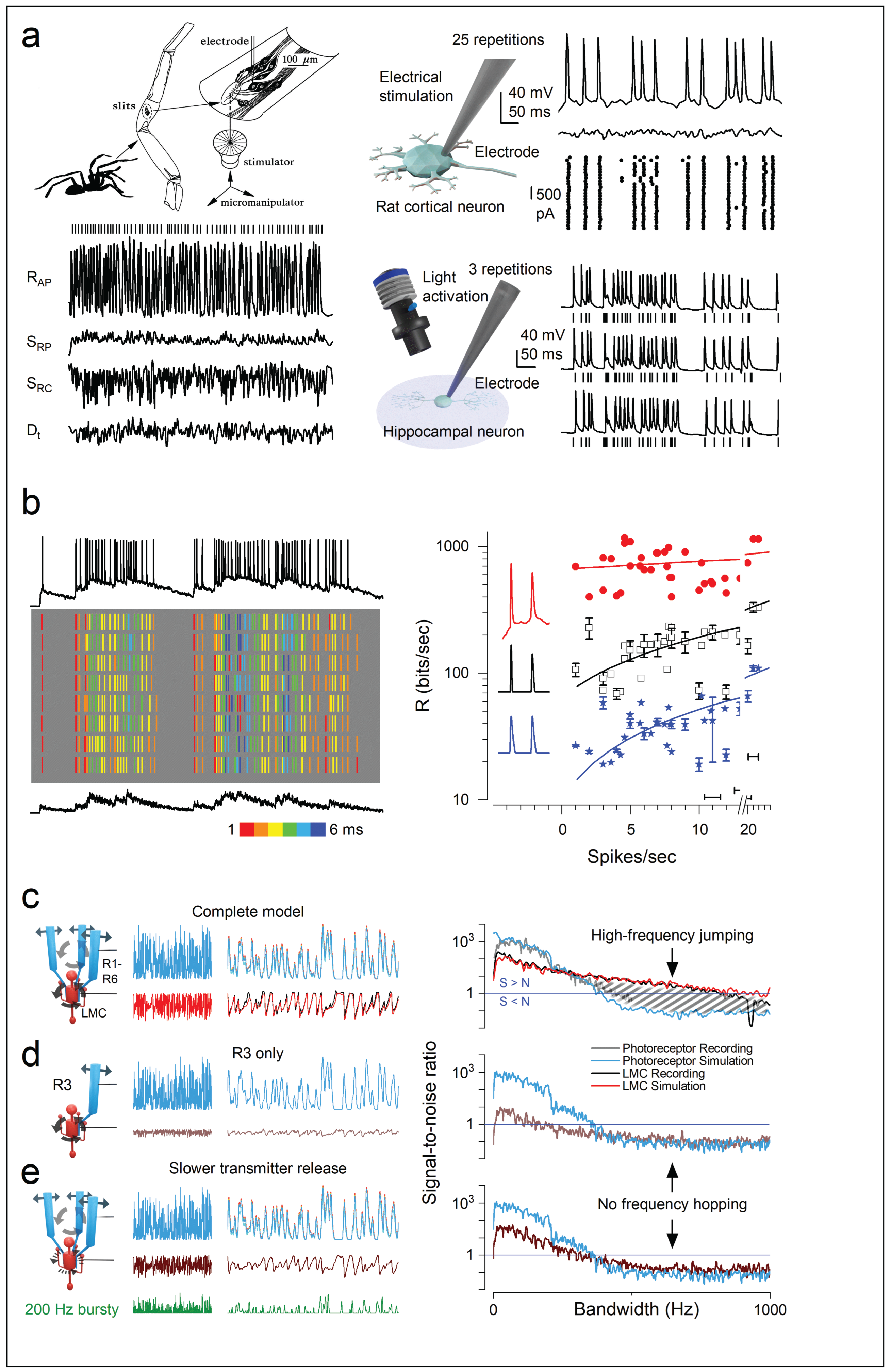
Conclusion
Funding
Acknowledgments
References
- Shannon CE. A mathematical theory of communication. Bell Syst Technic J. 1948;27:379–423, 623–56.
- Borst A, Theunissen FE. Information theory and neural coding. Nat Neurosci. 1999;2:947-57. [CrossRef]
- Juusola M, Takalo J, Kemppainen J, Razban Haghighi K, Scales B, McManus J, et al. Theory of morphodynamic information processing: linking sensing to behaviour. Vision Res. 2025;227.
- Perez-Escudero A, Rivera-Alba M, de Polavieja GG. Structure of deviations from optimality in biological systems. Proc Natl Acad Sci U S A. 2009;106:20544-9. [CrossRef]
- Rivera-Alba M, Vitaladevuni SN, Mischenko Y, Lu ZY, Takemura SY, Scheffer L, et al. Wiring economy and volume exclusion determine neuronal placement in the Drosophila brain. Curr Biol. 2011;21:2000-5. [CrossRef]
- de Polavieja GG. Errors drive the evolution of biological signalling to costly codes. J Theor Biol. 2002;214:657-64.
- Song Z, Juusola M. Refractory sampling links efficiency and costs of sensory encoding to stimulus statistics. J Neurosci. 2014;34:7216-37.
- Barlow HB. Possible principles underlying the transformations of sensory messages. In: Rosenblith W, editor. Sensory Communication: M.I.T. Press; 1961. p. 217-34.
- Darwin C. On the origin of species by means of natural selection, or the preservation of favoured races in the struggle for life. London: John Murray; 1859. [CrossRef]
- Juusola M, French AS, Uusitalo RO, Weckstrom M. Information processing by graded-potential transmission through tonically active synapses. Trends Neurosci. 1996;19:292-7. [CrossRef]
- Reshetniak S, Rizzoli SO. The vesicle cluster as a major organizer of synaptic composition in the short-term and long-term. Curr Opin Cell Biol. 2021;71:63-8.
- Reshetniak S, Ussling JE, Perego E, Rammner B, Schikorski T, Fornasiero EF, et al. A comparative analysis of the mobility of 45 proteins in the synaptic bouton. Embo J. 2020;39.
- Rusakov DA, Savtchenko LP, Zheng KY, Henley JM. Shaping the synaptic signal: molecular mobility inside and outside the cleft. Trends Neurosci. 2011;34:359-69.
- Watanabe S, Rost BR, Camacho-Perez M, Davis MW, Sohl-Kielczynski B, Rosenmund C, et al. Ultrafast endocytosis at mouse hippocampal synapses. Nature. 2013;504:242-7.
- El Hady A, Machta BB. Mechanical surface waves accompany action potential propagation. Nat Commun. 2015;6. [CrossRef]
- Kasai H, Ucar H. Forceful synapses reveal mechanical interactions in the brain. Nature. 2021.
- Korkotian E, Segal M. Spike-associated fast contraction of dendritic spines in cultured hippocampal neurons. Neuron. 2001;30:751-8. [CrossRef]
- Majewska A, Sur M. Motility of dendritic spines in visual cortex in vivo: changes during the critical period and effects of visual deprivation. Proc Natl Acad Sci U S A. 2003;100:16024-9. [CrossRef]
- Ucar H, Watanabe S, Noguchi J, Morimoto Y, Iino Y, Yagishita S, et al. Mechanical actions of dendritic-spine enlargement on presynaptic exocytosis. Nature. 2021;600:686-9.
- Benoit CM, Ganea DA, Paricio-Montesinos R, Esser J, Thome C, Janssen JM, et al. Axon initial segment dynamics during associative fear learning. Nat Neurosci. 2025. [CrossRef]
- Yu WQ, Swanstrom R, Sigulinsky CL, Ahlquist RM, Knecht S, Jones BW, et al. Distinctive synaptic structural motifs link excitatory retinal interneurons to diverse postsynaptic partner types. Cell Rep. 2023;42:112006.
- Kennedy HJ, Crawford AC, Fettiplace R. Force generation by mammalian hair bundles supports a role in cochlear amplification. Nature. 2005;433:880-3.
- Hardie RC, Franze K. Photomechanical responses in Drosophila photoreceptors. Science. 2012;338:260-163. [CrossRef]
- Juusola M, Dau A, Song Z, Solanki N, Rien D, Jaciuch D, et al. Microsaccadic sampling of moving image information provides Drosophila hyperacute vision. Elife. 2017;6.
- Kemppainen J, Mansour N, Takalo J, Juusola M. High-speed imaging of light-induced photoreceptor microsaccades in compound eyes. Commun Biol. 2022;5:203.
- Kemppainen J, Scales B, Razban Haghighi K, Takalo J, Mansour N, McManus J, et al. Binocular mirror-symmetric microsaccadic sampling enables Drosophila hyperacute 3D vision. Proc Natl Acad Sci U S A. 2022;119:e2109717119. [CrossRef]
- Mansour N, Takalo J, Kemppainen J, Bridges AD, MaBouDi H, Bohra AA, et al. Synaptic high-frequency jumping synchronises vision to high-speed behaviour. BioRxiv. 2025.
- Bocchero U, Falleroni F, Mortal S, Li Y, Cojoc D, Lamb T, et al. Mechanosensitivity is an essential component of phototransduction in vertebrate rods. PLoS Biol. 2020;18:e3000750.
- Pandiyan VP, Maloney-Bertelli A, Kuchenbecker JA, Boyle KC, Ling T, Chen ZC, et al. The optoretinogram reveals the primary steps of phototransduction in the living human eye. Sci Adv. 2020;6.
- Joy MSH, Nall DL, Emon B, Lee KY, Barishman A, Ahmed M, et al. Synapses without tension fail to fire in an in vitro network of hippocampal neurons. Proc Natl Acad Sci U S A. 2023;120:e2311995120. [CrossRef]
- Juusola M, Song ZY. How a fly photoreceptor samples light information in time. J Physiol Lond. 2017;595:5427-37.
- Song Z, Postma M, Billings SA, Coca D, Hardie RC, Juusola M. Stochastic, adaptive sampling of information by microvilli in fly photoreceptors. Curr Biol. 2012;22:1371-80.
- Hubel DH, Wiesel TN. Visual area of the lateral suprasylvian gyrus (Clare-Bishop area) of the cat. J Physiol. 1969;202:251-60.
- Hubel DH, Wiesel TN. Stereoscopic vision in macaque monkey. Cells sensitive to binocular depth in area 18 of the macaque monkey cortex. Nature. 1970;225:41-2.
- Mountcastle VB. Modality and topographic properties of single neurons of cat’s somatic sensory cortex. J Neurophysiol. 1957;20:408-34. [CrossRef]
- Mountcastle VB. The columnar organization of the neocortex. Brain. 1997;120 ( Pt 4):701-22.
- Békésy Gv. Über die Schwingungen der Basilarmembran beim Hören. Physikalische Zeitschrift. 1928;29:793-810.
- Galambos R, Davis H. Electrophysiological evidence of tonotopic organization in the auditory cortex of the cat. Journal of Neurophysiology. 1948;11:431-9.
- Tunturi AR. Physiological determination of the tonotopic organization of the auditory cortex in the dog. American Journal of Physiology. 1950;162:562–8.
- Olson CR, Graybiel AM. Sensory maps in the claustrum of the cat. Nature. 1980;288:479-81.
- Lee KS, Loutit AJ, de Thomas Wagner D, Sanders M, Huber D. Emergence of a brainstem somatosensory tonotopic map for substrate vibration. Nat Neurosci. 2025;28:97-104.
- Li M, Liu F, Juusola M, Tang S. Perceptual color map in macaque visual area V4. J Neurosci. 2014;34:202-17.
- Merzenich MM, Knight PL, Roth GL. Cochleotopic organization of primary auditory cortex in the cat. Brain Res. 1973;63:343-6.
- Merzenich MM, Knight PL, Roth GL. Representation of cochlea within primary auditory cortex in the cat. J Neurophysiol. 1975;38:231-49.
- Kohonen T. Self-organizing neural projections. Neural Netw. 2006;19:723-33.
- Kohonen T, Hari R. Where the abstract feature maps of the brain might come from. Trends Neurosci. 1999;22:135-9. [CrossRef]
- Fernandes AR, Martins JP, Gomes ER, Mendes CS, Teodoro RO. Drosophila motor neuron boutons remodel through membrane blebbing coupled with muscle contraction. Nat Commun. 2023;14.
- Griswold JM, Bonilla-Quintana M, Pepper Rea. Membrane mechanics dictate axonal pearls-on-a-string morphology and function. Nat Neurosci. 2025;28:49-61. [CrossRef]
- Zheng L, de Polavieja GG, Wolfram V, Asyali MH, Hardie RC, Juusola M. Feedback network controls photoreceptor output at the layer of first visual synapses in Drosophila. J Gen Physiol. 2006;127:495-510.
- Wong F, Knight BW. Adapting-bump model for eccentric cells of Limulus. J Gen Physiol. 1980;76:539-57. [CrossRef]
- Wong F, Knight BW, Dodge FA. Dispersion of latencies in photoreceptors of Limulus and the adapting-bump model. J Gen Physiol. 1980;76:517-37.
- Baylor DA, Lamb TD, Yau KW. Responses of retinal rods to single photons. J Physiol. 1979;288:613-34.
- Faisal AA, Selen LPJ, Wolpert DM. Noise in the nervous system. Nat Rev Neurosci. 2008;9:292-303. [CrossRef]
- Laughlin SB. The role of sensory adaptation in the retina. J Exp Biol. 1989;146:39-62.
- Laughlin SB, Howard J, Blakeslee B. Synaptic limitations to contrast coding in the retina of the blowfly Calliphora. Proc R Soc Lond B Biol Sci. 1987;231:437-67.
- Laughlin SB, Lillywhite PG. Intrinsic noise in locust photoreceptors. J Physiol. 1982;332:25-45.
- Lillywhite PG, Laughlin SB. Transducer noise in a photoreceptor. Nature. 1979;277:569-72.
- Amari S, Takeuchi A. Mathematical theory on formation of category detecting nerve cells. Biol Cybern. 1978;29:127-36.
- Takeuchi A, Amari S. Formation of topographic maps and columnar microstructures in nerve fields. Biol Cybern. 1979;35:63-72.
- Sjoberg RL. Brain stimulation and elicited memories. Acta Neurochir (Wien). 2023;165:2737-45.
- Suthana N, Fried I. Percepts to recollections: insights from single neuron recordings in the human brain. Trends Cogn Sci. 2012;16:427-36.
- Panzeri S, Senatore R, Montemurro MA, Petersen RS. Correcting for the sampling bias problem in spike train information measures. Journal of Neurophysiology. 2007;98:1064-72.
- Zheng JY, Meister M. The unbearable slowness of being: Why do we live at 10 bits/s? Neuron. 2025;113:192-204.
- Rieke F, Bodnar DA, Bialek W. Naturalistic stimuli increase the rate and efficiency of information transmission by primary auditory afferents. Proc Biol Sci. 1995;262:259-65.
- Lazar AA, Liu TK, Yeh CH. The functional logic of odor information processing in the antennal lobe. Plos Comput Biol. 2023;19.
- Atick JJ, Redlich AN. Towards a theory of early visual processing. Neural Computation. 1990;2:308-20. [CrossRef]
- van Hateren JH. A theory of maximizing sensory information. Biol Cybern. 1992;68:23-9.
- Hubel DH, Wiesel TN. Uniformity of monkey striate cortex: a parallel relationship between field size, scatter, and magnification factor. J Comp Neurol. 1974;158:295-305.
- Hubel DH, Wiesel TN. Sequence regularity and geometry of orientation columns in the monkey striate cortex. J Comp Neurol. 1974;158:267-93.
- Ahissar E, Arieli A. Figuring space by time. Neuron. 2001;32:185-201.
- Casile A, Victor JD, Rucci M. Contrast sensitivity reveals an oculomotor strategy for temporally encoding space. Elife. 2019;8. [CrossRef]
- Intoy J, Li YH, Bowers NR, Victor JD, Poletti M, Rucci M. Consequences of eye movements for spatial selectivity. Current Biology. 2024;34. [CrossRef]
- Kuang X, Poletti M, Victor JD, Rucci M. Temporal encoding of spatial information during active visual fixation. Curr Biol. 2012;22:510-4.
- Rucci M, Ahissar E, Burr DC, Kagan I, Poletti M, Victor JD. The visual system does not operate like a camera. J Vis. 2025;25:2. [CrossRef]
- Rucci M, Victor JD. The unsteady eye: an information-processing stage, not a bug. Trends in Neurosciences. 2015;38:195-206.
- Rucci M, Iovin R, Poletti M, Santini F. Miniature eye movements enhance fine spatial detail. Nature. 2007;447:851-4. [CrossRef]
- Juusola M, Uusitalo RO, Weckstrom M. Transfer of graded potentials at the photoreceptor-interneuron synapse. J Gen Physiol. 1995;105:117-48. [CrossRef]
- Meinertzhagen IA, O’Neil SD. Synaptic organization of columnar elements in the lamina of the wild type in Drosophila melanogaster. J Comp Neurol. 1991;305:232-63.
- Nikolaev A, Zheng L, Wardill TJ, O’Kane CJ, de Polavieja GG, Juusola M. Network adaptation improves temporal representation of naturalistic stimuli in Drosophila eye: II mechanisms. PLoS One. 2009;4:e4306.
- Zheng L, Nikolaev A, Wardill TJ, O’Kane CJ, de Polavieja GG, Juusola M. Network adaptation improves temporal representation of naturalistic stimuli in Drosophila eye: I dynamics. PLoS One. 2009;4:e4307.
- Buschbeck EK, Hoy RR. Visual system of the stalk-eyed fly, Cyrtodiopsis quinqueguttata (Diopsidae, Diptera): an anatomical investigation of unusual eyes. J Neurobiol. 1998;37:449-68.
- Teich MC, Prucnal PR, Vannucci G, Breton ME, Mcgill WJ. Multiplication Noise in the Human Visual-System at Threshold .1. Quantum Fluctuations and Minimum Detectable Energy. J Opt Soc Am. 1982;72:419-31.
- Warrant EJ. The remarkable visual capacities of nocturnal insects: vision at the limits with small eyes and tiny brains. Philos T R Soc B. 2017;372.
- Tang S, Juusola M. Intrinsic activity in the fly brain gates visual information during behavioral choices. PLoS One. 2010;5:e14455. [CrossRef]
- Franceschini N. Combined optical neuroanatomical, electrophysiological and behavioural studies on signal processing in the fly compound eye. In: Taddei-Ferretti C, editor. Biocybernetics of Vision: Integrative Mechanisms and Cognitive Processes. Singapore: World Scientific; 1998. p. 341-61.
- Howard J, Blakeslee B, Laughlin SB. The intracellular pupil mechanism and photoreceptor signal: noise ratios in the fly Lucilia cuprina. Proc R Soc Lond B Biol Sci. 1987;231:415-35.
- Enroth-Cugell C, Robson JG. The contrast sensitivity of retinal ganglion cells of the cat. J Physiol. 1966;187:517-52. [CrossRef]
- Juusola M. Linear and nonlinear contrast coding in light-adapted blowfly photoreceptors. J Comp Physiol A. 1993;172:511-21.
- Hartline HK, Wagner HG, Ratliff F. Inhibition in the Eye of Limulus. Journal of General Physiology. 1956;39:651-73.
- Hartline HK, Graham CH. Nerve impulses from single receptors in the eye of limulus. P Soc Exp Biol Med. 1932;29:0613-5. [CrossRef]
- Livingstone MS, Hubel DH. Spatial relationship and extrafoveal vision. Nature. 1985;315:285. [CrossRef]
- Zhang SH, Zhao XN, Jiang DQ, Tang SM, Yu C. Ocular dominance-dependent binocular combination of monocular neuronal responses in macaque V1. Elife. 2024;13.
- Ju NS, Guan SC, Tao L, Tang SM, Yu C. Orientation Tuning and End-stopping in Macaque V1 Studied with Two-photon Calcium Imaging. Cereb Cortex. 2021;31:2085-97. [CrossRef]
- Gonzalez-Bellido PT, Wardill TJ, Juusola M. Compound eyes and retinal information processing in miniature dipteran species match their specific ecological demands. Proc Natl Acad Sci U S A. 2011;108:4224-9.
- van Hateren JH. Real and optimal neural images in early vision. Nature. 1992;360:68-70.
- Yau KW, Lamb TD, Baylor DA. Light-induced fluctuations in membrane current of single toad rod outer segments. Nature. 1977;269:78-80.
- Normann RA, Werblin FS. Control of retinal sensitivity. I. Light and dark adaptation of vertebrate rods and cones. J Gen Physiol. 1974;63:37-61.
- Laughlin SB, Hardie RC. Common Strategies for Light Adaptation in the Peripheral Visual Systems of Fly and Dragonfly. J Comp Physiol. 1978;128:319-40.
- Baylor DA, Hodgkin AL, Lamb TD. Reconstruction of the electrical responses of turtle cones to flashes and steps of light. J Physiol. 1974;242:759-91. [CrossRef]
- Baylor DA, Hodgkin AL, Lamb TD. The electrical response of turtle cones to flashes and steps of light. J Physiol. 1974;242:685-727.
- Juusola M, Hardie RC. Light adaptation in Drosophila photoreceptors: II. Rising temperature increases the bandwidth of reliable signaling J Gen Physiol. 2001;117:27–42.
- Saviane C, Silver RA. Fast vesicle reloading and a large pool sustain high bandwidth transmission at a central synapse. Nature. 2006;439:983-7.
- Laughlin SB. A simple coding procedure enhances a neuron’s information capacity. Zeitschrift für Naturforschung C. 1981;36:910-2.
- van Hateren JH. Processing of natural time series of intensities by the visual system of the blowfly. Vision Res. 1997;37:3407-16.
- van Hateren JH. Theoretical predictions of spatiotemporal receptive fields of fly LMCs, and experimental validation. J Comp Physiol A. 1992;171:157-70.
- Brenner N, Bialek W, de Ruyter van Steveninck R. Adaptive rescaling maximizes information transmission. Neuron. 2000;26:695-702. [CrossRef]
- Attneave F. Some informational aspects of visual perception. Psychol Rev. 1954;61:183-93.
- Juusola M, Weckstrom M, Uusitalo RO, Korenberg MJ, French AS. Nonlinear models of the first synapse in the light-adapted fly retina. J Neurophysiol. 1995;74:2538-47.
- Juusola M, Kouvalainen E, Jarvilehto M, Weckstrom M. Contrast gain, signal-to-noise ratio, and linearity in light-adapted blowfly photoreceptors. J Gen Physiol. 1994;104:593-621.
- van Hateren JH, Schilstra C. Blowfly flight and optic flow II. Head movements during flight. J Exp Biol. 1999;202:1491-500.
- de Ruyter van Steveninck RR, Laughlin SB. The rate of information transfer at graded-potential synapses. Nature. 1996:642-5. [CrossRef]
- Juusola M, Hardie RC. Light Adaptation in Drosophila Photoreceptors: I. Response Dynamics and Signaling Efficiency at 25 °C. J Gen Physiol. 2001;117:3-25.
- van Hateren JH, Snippe HP. Information theoretical evaluation of parametric models of gain control in blowfly photoreceptor cells. Vision Res. 2001;41:1851-65.
- van Hateren JH, Snippe HP. Phototransduction in primate cones and blowfly photoreceptors: different mechanisms, different algorithms, similar response. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2006;192:187-97. [CrossRef]
- Juusola M, de Polavieja GG. The rate of information transfer of naturalistic stimulation by graded potentials. J Gen Physiol. 2003;122:191-206.
- Blaj G, van Hateren JH. Saccadic head and thorax movements in freely walking blowflies. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2004;190:861-8.
- Lindemann JP, Kern R, Michaelis C, Meyer P, van Hateren JH, Egelhaaf M. FliMax, a novel stimulus device for panoramic and highspeed presentation of behaviourally generated optic flow. Vision Res. 2003;43:779-91.
- Schilstra C, Van Hateren JH. Blowfly flight and optic flow I. Thorax kinematics and flight dynamics. J Exp Biol. 1999;202:1481-90.
- van Hateren JH, van der Schaaf A. Independent component filters of natural images compared with simple cells in primary visual cortex. Proc Roy Soc Lond B. 1998;265:359-66.
- Atick JJ. Could information theory provide an ecological theory of sensory processing? Network. 2011;22:4-44. [CrossRef]
- Dan Y, Atick JJ, Reid RC. Efficient coding of natural scenes in the lateral geniculate nucleus: experimental test of a computational theory. J Neurosci. 1996;16:3351-62.
- van Hateren JH, Ruttiger L, Sun H, Lee BB. Processing of natural temporal stimuli by macaque retinal ganglion cells. J Neurosci. 2002;22:9945-60.
- Schilstra C, van Hateren JH. Using miniature sensor coils for simultaneous measurement of orientation and position of small, fast-moving animals. J Neurosci Methods. 1998;83:125-31.
- Fenk LM, Avritzer SC, Weisman JL, Nair A, Randt LD, Mohren TL, et al. Muscles that move the retina augment compound eye vision in Drosophila. Nature. 2022;612:116-22.
- Franceschini N, Chagneux R, Kirschfeld K. Gaze control in flies by co-ordinated action of eye muscles. In: Elsner N, Menzel R, editors. Gottingen Neurobiology Report1995. p. 401.
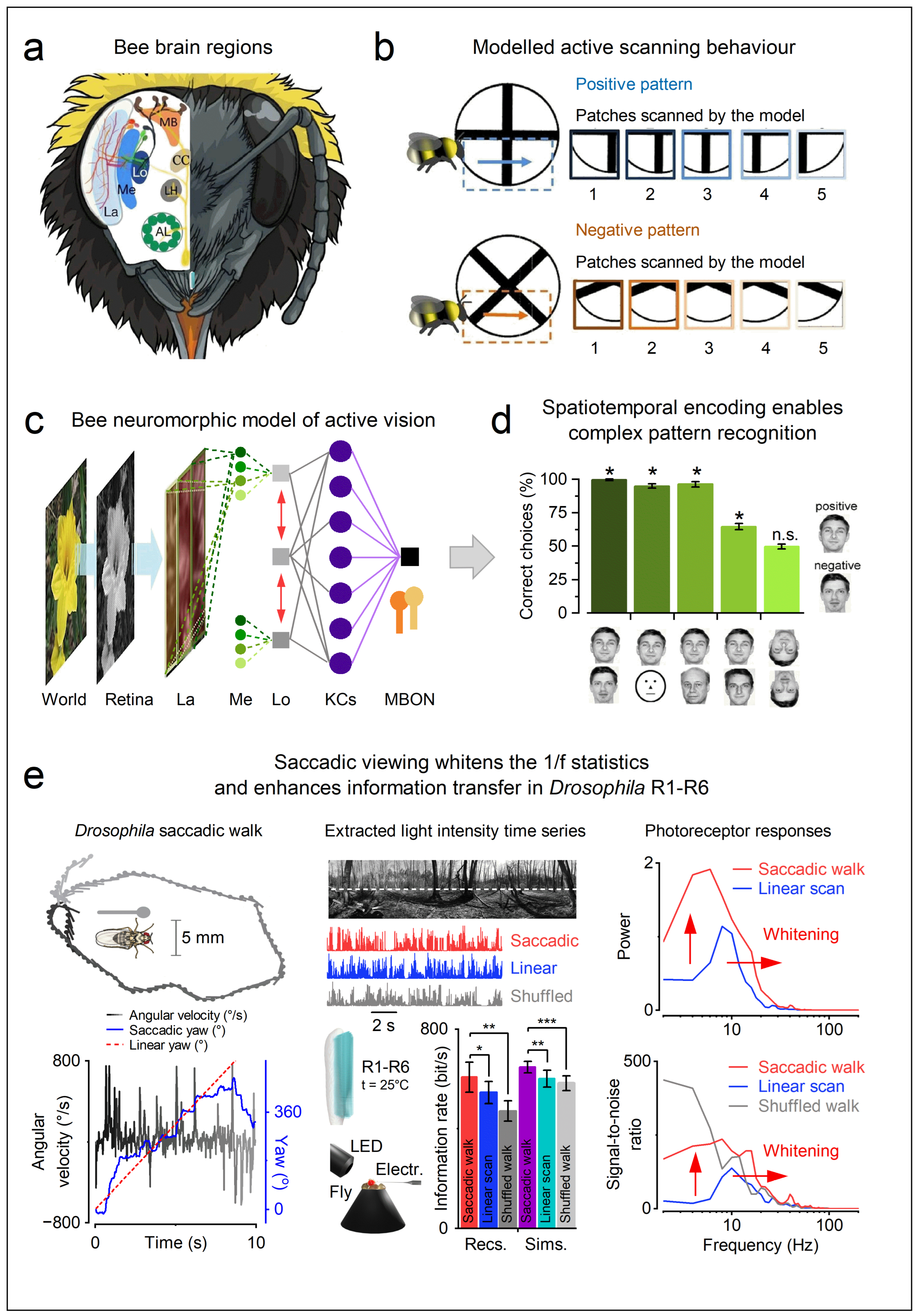
- MaBouDi H, Roper M, Guiraud M-G, Chittka L, Marshall JA. A neuromorphic model of active vision shows how spatiotemporal encoding in lobula neurons can aid pattern recognition in bees. eLife. 2023;14. [CrossRef]
- Juusola M, Song Z, Hardie RC. Phototransduction Biophysics. In: Jaeger D, Jung R, editors. Encyclopedia of Computational Neuroscience. New York, NY: Springer; 2022. p. 2758-76.
- Song Z, Zhou Y, Feng J, Juusola M. Multiscale ‘whole-cell’ models to study neural information processing - New insights from fly photoreceptor studies. J Neurosci Methods. 2021;357:109156.
- Hardie RC, Juusola M. Phototransduction in Drosophila. Curr Opin Neurobiol. 2015;34:37-45.
- Faivre O, Juusola M. Visual coding in locust photoreceptors. Plos One. 2008;3.
- Bastos AM, Usrey WM, Adams RA, Mangun GR, Fries P, Friston KJ. Canonical Microcircuits for Predictive Coding. Neuron. 2012;76:695-711. [CrossRef]
- Rao RPN, Ballard DH. Predictive coding in the visual cortex: a functional interpretation of some extra-classical receptive-field effects. Nat Neurosci. 1999;2:79-87.
- Friston KJ. A theory of cortical responses. Philos T R Soc B. 2005;360:815-36. [CrossRef]
- Sommer MA, Wurtz RH. Brain circuits for the internal monitoring of movements. Annual Review of Neuroscience. 2008;31:317-38.
- Sperry RW. Neural Basis of the Spontaneous Optokinetic Response Produced by Visual Inversion. J Comp Physiol Psych. 1950;43:482-9.
- von Holst E, Mittelstaedt H. Das Reafferenzprinzip - (Wechselwirkungen Zwischen Zentralnervensystem Und Peripherie). Naturwissenschaften. 1950;37:464-76.
- Skingsley DR, Laughlin SB, Hardie RC. Properties of Histamine-Activated Chloride Channels in the Large Monopolar Cells of the Dipteran Compound Eye - a Comparative-Study. J Comp Physiol A. 1995;176:611-23. [CrossRef]
- Hardie RC. A Histamine-Activated Chloride Channel Involved in Neurotransmission at a Photoreceptor Synapse. Nature. 1989;339:704-6.
- Hardie RC. Effects of Antagonists on Putative Histamine-Receptors in the 1st Visual Neuropil of the Housefly (Musca-Domestica). J Exp Biol. 1988;138:221-41.
- Nassel DR, Holmqvist MH, Hardie RC, Hakanson R, Sundler F. Histamine-Like Immunoreactivity in Photoreceptors of the Compound Eyes and Ocelli of the Flies Calliphora-Erythrocephala and Musca-Domestica. Cell Tissue Res. 1988;253:639-46.
- Hardie RC. Is Histamine a Neurotransmitter in Insect Photoreceptors. J Comp Physiol A. 1987;161:201-13.
- Dau A, Friederich U, Dongre S, Li X, Bollepalli MK, Hardie RC, et al. Evidence for Dynamic Network Regulation of Drosophila Photoreceptor Function from Mutants Lacking the Neurotransmitter Histamine. Front Neural Circuits. 2016;10:19.
- Yau KW, Hardie RC. Phototransduction motifs and variations. Cell. 2009;139:246-64.
- Fain GL, Hardie R, Laughlin SB. Phototransduction and the evolution of photoreceptors. Curr Biol. 2010;20:R114-24. [CrossRef]
- Wardill TJ, List O, Li X, Dongre S, McCulloch M, Ting CY, et al. Multiple spectral inputs improve motion discrimination in the Drosophila visual system. Science. 2012;336:925-31.
- Poulet JF, Hedwig B. New insights into corollary discharges mediated by identified neural pathways. Trends Neurosci. 2007;30:14-21.
- Hochstrate P, Hamdorf K. Microvillar components of light adaptation in blowflies. J Gen Physiol. 1990;95:891-910. [CrossRef]
- Pumir A, Graves J, Ranganathan R, Shraiman BI. Systems analysis of the single photon response in invertebrate photoreceptors. Proc Natl Acad Sci U S A. 2008;105:10354-9.
- Liu CH, Satoh AK, Postma M, Huang J, Ready DF, Hardie RC. Ca2+-dependent metarhodopsin inactivation mediated by calmodulin and NINAC myosin III. Neuron. 2008;59:778-89.
- Mishra P, Socolich M, Wall MA, Graves J, Wang Z, Ranganathan R. Dynamic scaffolding in a G protein-coupled signaling system. Cell. 2007;131:80-92.
- Scott K, Sun Y, Beckingham K, Zuker CS. Calmodulin regulation of Drosophila light-activated channels and receptor function mediates termination of the light response in vivo. Cell. 1997;91:375-83.
- Yarbus AL. Eye movements and vision. New York: Plenum; 1967.
- Buracas GT, Albright TD. Gauging sensory representations in the brain. Trends in Neurosciences. 1999;22:303-9. [CrossRef]
- Farroni T, Csibra G, Simion G, Johnson MH. Eye contact detection in humans from birth. P Natl Acad Sci USA. 2002;99:9602-5.
- Bushnell IWR. Mother’s face recognition in newborn infants: Learning and memory. Infant Child Dev. 2001;10:67-74.
- Lewkowicz DJ, Hansen-Tift AM. Infants deploy selective attention to the mouth of a talking face when learning speech. P Natl Acad Sci USA. 2012;109:1431-6.
- Cover TM, Thomas JA. Elements of Information Theory. 2nd ed. Hoboken: John Wiley & Sons; 2005.
- Li X, Abou Tayoun A, Song Z, Dau A, Rien D, Jaciuch D, et al. Ca2+-activated K+ channels reduce network excitability, improving adaptability and energetics for transmitting and perceiving sensory information. J Neurosci. 2019;39:7132-54.
- Pick B. Specific Misalignments of Rhabdomere Visual Axes in Neural Superposition Eye of Dipteran Flies. Biol Cybern. 1977;26:215-24.
- Dippé MAZ, Wold EH. Antialiasing through stochastic sampling. ACM SIGGRAPH Computer Graphics. 1985;19:69-78.
- Yellott JI. Spectral-analysis of spatial sampling by photoreceptors - topological disorder prevents aliasing. Vis Res. 1982;22:1205-10. [CrossRef]
- Yellott JI. Spectral consequences of photoreceptor sampling in the rhesus retina. Science. 1983;221:382-5.
- Galton F. Vox populi. Nature 1907:450-1.
- Laughlin SB, van Steveninck RRD, Anderson JC. The metabolic cost of neural information. Nat Neurosci. 1998;1:36-41.
- Land MF. Visual acuity in insects. Ann Rev Entomol. 1997;42:147-77.
- MaBouDi H, Richter J, Guiraud MG, Roper M, Marshall JAR, Chittka L. Active vision of bees in a simple pattern discrimination task. Elife. 2025;14.
- MaBouDi H, Roper M, Guiraud M, Marshall JA, Chittka L. Automated video tracking and flight analysis show how bumblebees solve a pattern discrimination task using active vision. bioRxiv. 2021.
- Maboudi H, Roper M, Guiraud MG, Juusola M, Chittka L, Marshall JAR, et al. A neuromorphic model of active vision shows how spatiotemporal encoding in lobula neurons can aid pattern recognition in bees. Elife. 2025;14. [CrossRef]
- Hardie RC. Functional organization of the fly retina. Progress in Sensory Physiology: Springer; 1985. p. 1-79.
- Srinivasan MV, Bernard GD. Effect of Motion on Visual-Acuity of Compound Eye - Theoretical-Analysis. Vision Res. 1975;15:515-25.
- Juusola M, French AS. Visual acuity for moving objects in first- and second-order neurons of the fly compound eye. Journal of Neurophysiology. 1997;77:1487-95.
- Riggs LA, Ratliff F. The effects of counteracting the normal movements of the eye. J Opt Soc Am. 1952;42:872-3.
- Riggs LA, Ratliff F, Cornsweet JC, Cornsweet TN. The Disappearance of Steadily Fixated Visual Test Objects. J Opt Soc Am. 1953;43:495-501.
- Martinez-Conde S, Macknik SL, Hubel DH. The role of fixational eye movements in visual perception. Nat Rev Neurosci. 2004;5:229-40. [CrossRef]
- Kahneman D. Thinking, fast and slow: Farrar, Straus and Giroux.; 2011.
- Simoncelli EP, Olshausen BA. Natural image statistics and neural representation. Annu Rev Neurosci. 2001;24:1193-216.
- Hubel DH, Wiesel TN. The period of susceptibility to the physiological effects of unilateral eye closure in kittens. J Physiol. 1970;206:419-36. [CrossRef]
- Wechsler SP, Bhandawat V. Behavioral algorithms and neural mechanisms underlying odor-modulated locomotion in insects. J Exp Biol. 2023;226.
- Budick SA, Dickinson MH. Free-flight responses of Drosophila melanogaster to attractive odors. J Exp Biol. 2006;209:3001-17.
- Stupski SD, van Breugel F. Wind gates olfaction-driven search states in free flight. Current Biology. 2024;34.
- Lazar AA, Liu T, Yeh CH, Zhou Y. Modeling and characterization of pure and odorant mixture processing in the Drosophila mushroom body calyx. Front Physiol. 2024;15:1410946.
- Lazar AA, Yeh CH. A molecular odorant transduction model and the complexity of spatio-temporal encoding in the antenna. Plos Comput Biol. 2020;16.
- Behnia R, Desplan C. Visual circuits in flies: beginning to see the whole picture. Curr Opin Neurobiol. 2015;34:125-32. [CrossRef]
- Matsliah A, Yu SC, Kruk K, Bland D, Burke AT, Gager J, et al. Neuronal parts list and wiring diagram for a visual system. Nature. 2024;634:166-80.
- Wachowiak M. All in a sniff: olfaction as a model for active sensing. Neuron. 2011;71:962-73.
- Kepecs A, Uchida N, Mainen ZF. The sniff as a unit of olfactory processing. Chem Senses. 2006;31:167-79.
- Harvey J, Rinberg D. Olfaction: Source separation in a single sniff. Curr Biol. 2021;31:R1051-R3. [CrossRef]
- Karadas M, Gill JV, Ceballo S, Shoham S, Rinberg D. Rapid temporal processing in the olfactory bulb underlies concentration invariant odor identification and signal decorrelation. Res Sq. 2025.
- Shusterman R, Smear MC, Koulakov AA, Rinberg D. Precise olfactory responses tile the sniff cycle. Nat Neurosci. 2011;14:1039–44.
- Smear M, Shusterman R, O’Connor R, Bozza T, Rinberg D. Perception of sniff phase in mouse olfaction. Nature. 2011;479:397–400.
- Brainard DH, Wandell BA. Asymmetric Color Matching - How Color Appearance Depends on the Illuminant. Journal of the Optical Society of America a-Optics Image Science and Vision. 1992;9:1433-48.
- Rushton WAH. Pigments and Signals in Color-Vision. J Physiol-London. 1972;220:P1-&.
- Bandyopadhyay P, Sachse S. Mixing things up! - how odor blends are processed in Drosophila. Curr Opin Insect Sci. 2023;59:101099.
- Blazing RM, Franks KM. Odor coding in piriform cortex: mechanistic insights into distributed coding. Curr Opin Neurobiol. 2020;64:96-102.
- Endo K, Kazama H. Central organization of a high-dimensional odor space. Curr Opin Neurobiol. 2022;73:102528. [CrossRef]
- Intoy J, Rucci M. Finely tuned eye movements enhance visual acuity. Nat Commun. 2020;11. [CrossRef]
- Rucci M, Ahissar E, Burr D. Temporal Coding of Visual Space. Trends Cogn Sci. 2018;22:883-95. [CrossRef]
- Pisokas I, Heinze S, Webb B. The head direction circuit of two insect species. Elife. 2020;9.
- Seelig JD, Jayaraman V. Feature detection and orientation tuning in the Drosophila central complex. Nature. 2013;503:262-6.
- Cope AJ, Sabo C, Vasilaki E, Barron AB, Marshall JAR. A computational model of the integration of landmarks and motion in the insect central complex. Plos One. 2017;12.
- Dan C, Hulse BK, Kappagantula R, Jayaraman V, Hermundstad AM. A neural circuit architecture for rapid learning in goal-directed navigation. Neuron. 2024;112:2581-99 e23.
- Garner D, Kind E, Lai JYH, Nern A, Zhao A, Houghton L, et al. Connectomic reconstruction predicts visual features used for navigation. Nature. 2024;634:181-90. [CrossRef]
- Goulard R, Buehlmann C, Niven JE, Graham P, Webb B. A unified mechanism for innate and learned visual landmark guidance in the insect central complex. Plos Comput Biol. 2021;17:e1009383.
- Grabowska MJ, Jeans R, Steeves J, van Swinderen B. Oscillations in the central brain of Drosophila are phase locked to attended visual features. Proc Natl Acad Sci U S A. 2020;117:29925-36.
- Green J, Adachi A, Shah KK, Hirokawa JD, Magani PS, Maimon G. A neural circuit architecture for angular integration in Drosophila. Nature. 2017;546:101-6. [CrossRef]
- Heinze S. Mapping the fly’s ‘brain in the brain’. Elife. 2021;10.
- Heinze S, Homberg U. Linking the input to the output: new sets of neurons complement the polarization vision network in the locust central complex. J Neurosci. 2009;29:4911-21.
- Hulse BK, Haberkern H, Franconville R, Turner-Evans D, Takemura SY, Wolff T, et al. A connectome of the Drosophila central complex reveals network motifs suitable for flexible navigation and context-dependent action selection. Elife. 2021;10.
- Li F, Lindsey J, Marin EC, Otto N, Dreher M, Dempsey G, et al. The connectome of the adult mushroom body provides insights into function. Elife. 2020;9.
- Pfeiffer K, Homberg U. Organization and functional roles of the central complex in the insect brain. Annu Rev Entomol. 2014;59:165-U787.
- Eichler K, Li F, Litwin-Kumar A, Park Y, Andrade I, Chneider-Mizell CMS, et al. The complete connectome of a learning and memory centre in an insect brain. Nature. 2017;548:175-82.
- MaBouDi H, Marshall JAR, Barron AB. Honeybees solve a multi-comparison ranking task by probability matching. P Roy Soc B-Biol Sci. 2020;287.
- Nityananda V, Chittka L. Modality-specific attention in foraging bumblebees. Roy Soc Open Sci. 2015;2.
- Sorribes A, Armendariz BG, Lopez-Pigozzi D, Murga C, de Polavieja GG. The origin of behavioral bursts in decision-making circuitry. Plos Comput Biol. 2011;7:e1002075.
- Takemura SY, Aso Y, Hige T, Wong A, Lu Z, Xu CS, et al. A connectome of a learning and memory center in the adult Drosophila brain. Elife. 2017;6.
- Mostofi N, Zhao Z, Intoy J, Boi M, Victor JD, Rucci M. Spatiotemporal Content of Saccade Transients. Curr Biol. 2020;30:3999-4008 e2. [CrossRef]
- Barlow HB. Eye movements during fixation. J Physiol. 1952;116:290-306. [CrossRef]
- Ahissar E, Arieli A. Seeing via Miniature Eye Movements: A Dynamic Hypothesis for Vision. Front Comput Neurosci. 2012;6:89. [CrossRef]
- Aytekin M, Victor JD, Rucci M. The visual input to the retina during natural head-free fixation. J Neurosci. 2014;34:12701-15.
- Rucci M, Poletti M. Control and Functions of Fixational Eye Movements. Annual Review of Vision Science, Vol 1. 2015;1:499-518. [CrossRef]
- Zhao Z, Ahissar E, Victor JD, Rucci M. Inferring visual space from ultra-fine extra-retinal knowledge of gaze position. Nat Commun. 2023;14:269.
- Kern R, van Hateren JH, Michaelis C, Lindemann JP, Egelhaaf M. Function of a fly motion-sensitive neuron matches eye movements during free flight. PLoS Biol. 2005;3:e171.
- Field DJ. Relations between the statistics of natural images and the response properties of cortical cells. J Opt Soc Am A. 1987;4:2379-94.
- Martinez-Conde S, Otero-Millan J, Macknik SL. The impact of microsaccades on vision: towards a unified theory of saccadic function. Nat Rev Neurosci. 2013;14:83-96.
- Noton D, Stark L. Scanpaths in Saccadic Eye Movements While Viewing and Recognizing Patterns. Vision Res. 1971;11:929-&. [CrossRef]
- Noton D, Stark L. Scanpaths in Eye Movements during Pattern Perception. Science. 1971;171:308-&. [CrossRef]
- Najemnik J, Geisler WS. Optimal eye movement strategies in visual search. Nature. 2005;434:387-91.
- Rothkopf CA, Ballard DH, Hayhoe MM. Task and context determine where you look. J Vision. 2007;7.
- Guiraud M, Roper M, Chittka L. High-Speed Videography Reveals How Honeybees Can Turn a Spatial Concept Learning Task Into a Simple Discrimination Task by Stereotyped Flight Movements and Sequential Inspection of Pattern Elements. Front Psychol. 2018;9.
- Land MF, Collett TS. Chasing Behavior of Houseflies (Fannia-Canicularis) - Description and Analysis. J Comp Physiol. 1974;89:331-57.
- Boeddeker N, Kern R, Egelhaaf M. Chasing a dummy target: smooth pursuit and velocity control in male blowflies. P Roy Soc B-Biol Sci. 2003;270:393-9.
- Atick JJ, Redlich AN. What Does the Retina Know About Natural Scenes. Neural Computation. 1992;4:196-210.
- Olshausen BA. Learning sparse, overcomplete representations of time-varying natural images. Ieee Image Proc. 2003:41-4.
- MaBouDi H, Marshall JAR, Dearden N, Barron AB. How honey bees make fast and accurate decisions. BioRxiv. 2023.
- Gold JI, Shadlen MN. The neural basis of decision making. Annual Review of Neuroscience. 2007;30:535-74. [CrossRef]
- Heitz RP, Schall JD. Neural Mechanisms of Speed-Accuracy Tradeoff. Neuron. 2012;76:616-28.
- Hanks TD, Kiani R, Shadlen MN. A neural mechanism of speed-accuracy tradeoff in macaque area LIP. Elife. 2014;3.
- Bogacz R. Optimal decision-making theories: linking neurobiology with behaviour. Trends in Cognitive Sciences. 2007;11:118-25.
- Chittka L, Skorupski P, Raine NE. Speed-accuracy tradeoffs in animal decision making. Trends Ecol Evol. 2009;24:400-7.
- Marshall JAR, Dornhaus A, Franks NR, Kovacs T. Noise, cost and speed-accuracy trade-offs: decision-making in a decentralized system. J R Soc Interface. 2006;3:243-54.
- Ruderman DL, Cronin TW, Chiao CC. Statistics of cone responses to natural images: implications for visual coding. Journal of the Optical Society of America a-Optics Image Science and Vision. 1998;15:2036-45.
- Geurten BRH, Jahde P, Corthals K, Gopfert MC. Saccadic body turns in walking Drosophila. Front Behav Neurosci. 2014;8. [CrossRef]
- von Frisch K. The dance language and orientation of bees. Cambridge: Harvard University Press; 1967.
- Seeley TD. The Wisdom of the Hive: The Social Physiology of Honey Bee Colonies. Cambridge: Harvard University Press; 1995.
- Hadjitofi A, Webb B. Dynamic antennal positioning allows honeybee followers to decode the dance. Curr Biol. 2024;34:1772-9 e4. [CrossRef]
- Pavlou HJ, Goodwin SF. Courtship behavior in Drosophila melanogaster: towards a ‘courtship connectome’. Curr Opin Neurobiol. 2013;23:76-83.
- Coen P, Murthy M. Singing on the fly: sensorimotor integration and acoustic communication in Drosophila. Curr Opin Neurobiol. 2016;38:38-45.
- Levy WB, Baxter RA. Energy efficient neural codes. Neural Computation. 1996;8:531-43.
- Attwell D, Laughlin SB. An energy budget for signaling in the grey matter of the brain. J Cerebr Blood F Met. 2001;21:1133-45. [CrossRef]
- Ma WJ, Beck JM, Latham PE, Pouget A. Bayesian inference with probabilistic population codes. Nat Neurosci. 2006;9:1432-8.
- Zhang WH, Wu S, Josic K, Doiron B. Sampling-based Bayesian inference in recurrent circuits of stochastic spiking neurons. Nat Commun. 2023;14:7074.
- Knill DC, Pouget A. The Bayesian brain: the role of uncertainty in neural coding and computation. Trends Neurosci. 2004;27:712-9. [CrossRef]
- Schultz W, Dayan P, Montague PR. A neural substrate of prediction and reward. Science. 1997;275:1593-9.
- Lanore F, Cayco-Gajic NA, Gurnani H, Coyle D, Silver RA. Cerebellar granule cell axons support high-dimensional representations. Nat Neurosci. 2021;24:1142-50.
- Silver RA, Lubke J, Sakmann B, Feldmeyer D. High-probability uniquantal transmission at excitatory synapses in barrel cortex. Science. 2003;302:1981-4.
- Alle H, Geiger JR. Combined analog and action potential coding in hippocampal mossy fibers. Science. 2006;311:1290-3. [CrossRef]
- de Polavieja GG, Harsch A, Kleppe I, Robinson HP, Juusola M. Stimulus history reliably shapes action potential waveforms of cortical neurons. J Neurosci. 2005;25:5657-65.
- Juusola M, Robinson HP, de Polavieja GG. Coding with spike shapes and graded potentials in cortical networks. Bioessays. 2007;29:178-87.
- Shu Y, Hasenstaub A, Duque A, Yu Y, McCormick DA. Modulation of intracortical synaptic potentials by presynaptic somatic membrane potential. Nature. 2006;441:761-5.
- de Kock CPJ, Feldmeyer D. Shared and divergent principles of synaptic transmission between cortical excitatory neurons in rodent and human brain. Front Synaptic Neuro. 2023;15.
- Eyal G, Verhoog MB, Testa-Silva G, Deitcher Y, Benavides-Piccione R, DeFelipe J, et al. Human Cortical Pyramidal Neurons: From Spines to Spikes via Models. Front Cell Neurosci. 2018;12. [CrossRef]
- Leleo EG, Segev I. Burst control: Synaptic conditions for burst generation in cortical layer 5 pyramidal neurons. Plos Comput Biol. 2021;17.
- Laughlin SB, Sejnowski TJ. Communication in neuronal networks. Science. 2003;301:1870-4.
- Pillow JW, Shlens J, Paninski L, Sher A, Litke AM, Chichilnisky EJ, et al. Spatio-temporal correlations and visual signalling in a complete neuronal population. Nature. 2008;454:995-9. [CrossRef]
- Juusola M, French AS. The efficiency of sensory information coding by mechanoreceptor neurons. Neuron. 1997;18:959-68.
- Mainen ZF, Sejnowski TJ. Reliability of Spike Timing in Neocortical Neurons. Science. 1995;268:1503-6. [CrossRef]
- Boyden ES, Zhang F, Bamberg E, Nagel G, Deisseroth K. Millisecond-timescale, genetically targeted optical control of neural activity. Nat Neurosci. 2005;8:1263-8.
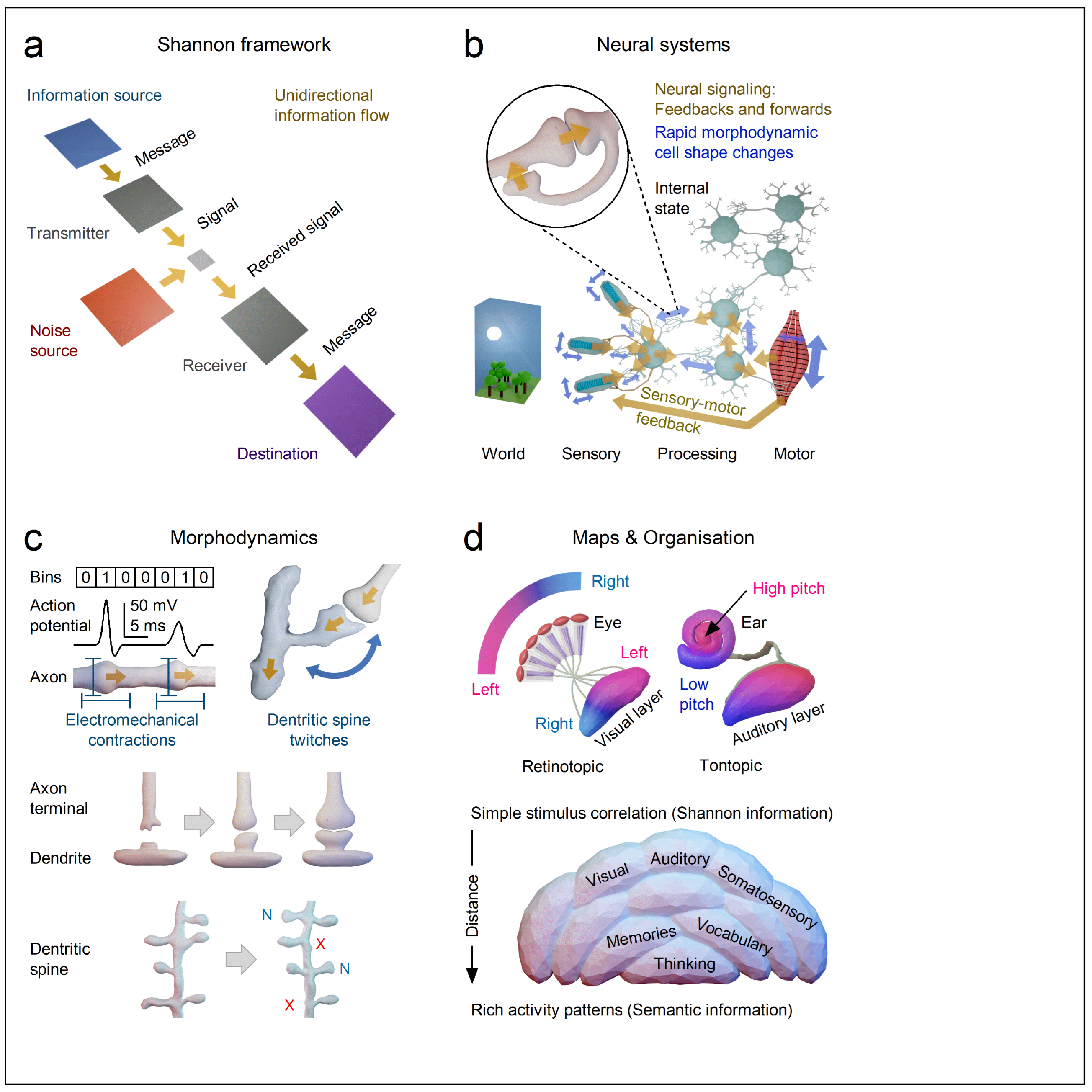
| Syntactic Information | |
| Definition: | Concerns the form or structure of signals, independent of what they mean. |
| In Neuroscience: | Corresponds to Shannon information - bits of data that quantify uncertainty or variability in signals. |
| Key Question: | How much information is transmitted? |
| Example: | Measuring how unpredictable a neuron’s spike train is, or how many distinct voltage patterns it can encode, regardless of what those spikes represent. |
| Analogy: | The grammar and word count of a sentence without understanding its meaning - e.g., “The cat sat on the mat” and “The mat sat on the cat” have the same syntactic elements (words, structure) but very different semantics. |
| Semantic Information | |
| Definition: | Concerns the meaning or interpretation of signals within a given context or system of understanding. |
| In Neuroscience | Refers to what the signals stand for - their relevance to perception, expectation, or action. |
| Key question: | What does the information mean to the organism? |
| Example: | A spike in a neuron participating in the representation of a visual object - say, a basketball - at a precise phase of a saccade could indicate that the ball is moving along a specific trajectory toward the basket, prompting a blocking response. |
| Analogy: | Understanding the meaning of the sentence - not just its syntax - and using it to act appropriately. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




