Submitted:
13 March 2026
Posted:
16 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Insects
2.2. Insecticides and Dose Preparation
2.3. Leaf Disc Treatment
2.4. Experimental Design
2.5. Acute Toxicity Assay (0–24 h)
2.6. Residual Toxicity Assay (24–72 h and 72–96 h)
2.7. Data Processing and Statistical Analysis
3. Results
3.1. Acute Toxicity (0–24 h)
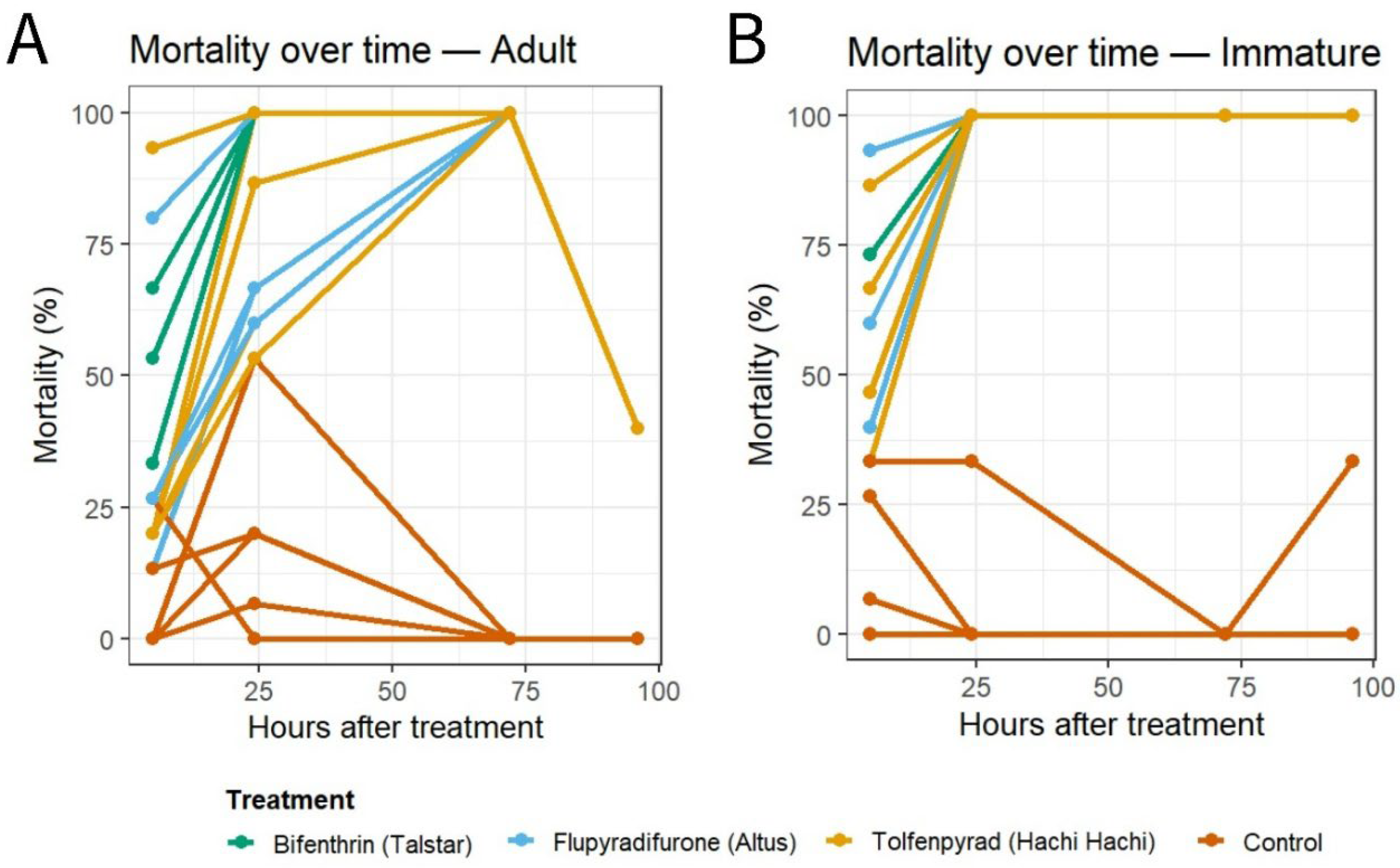
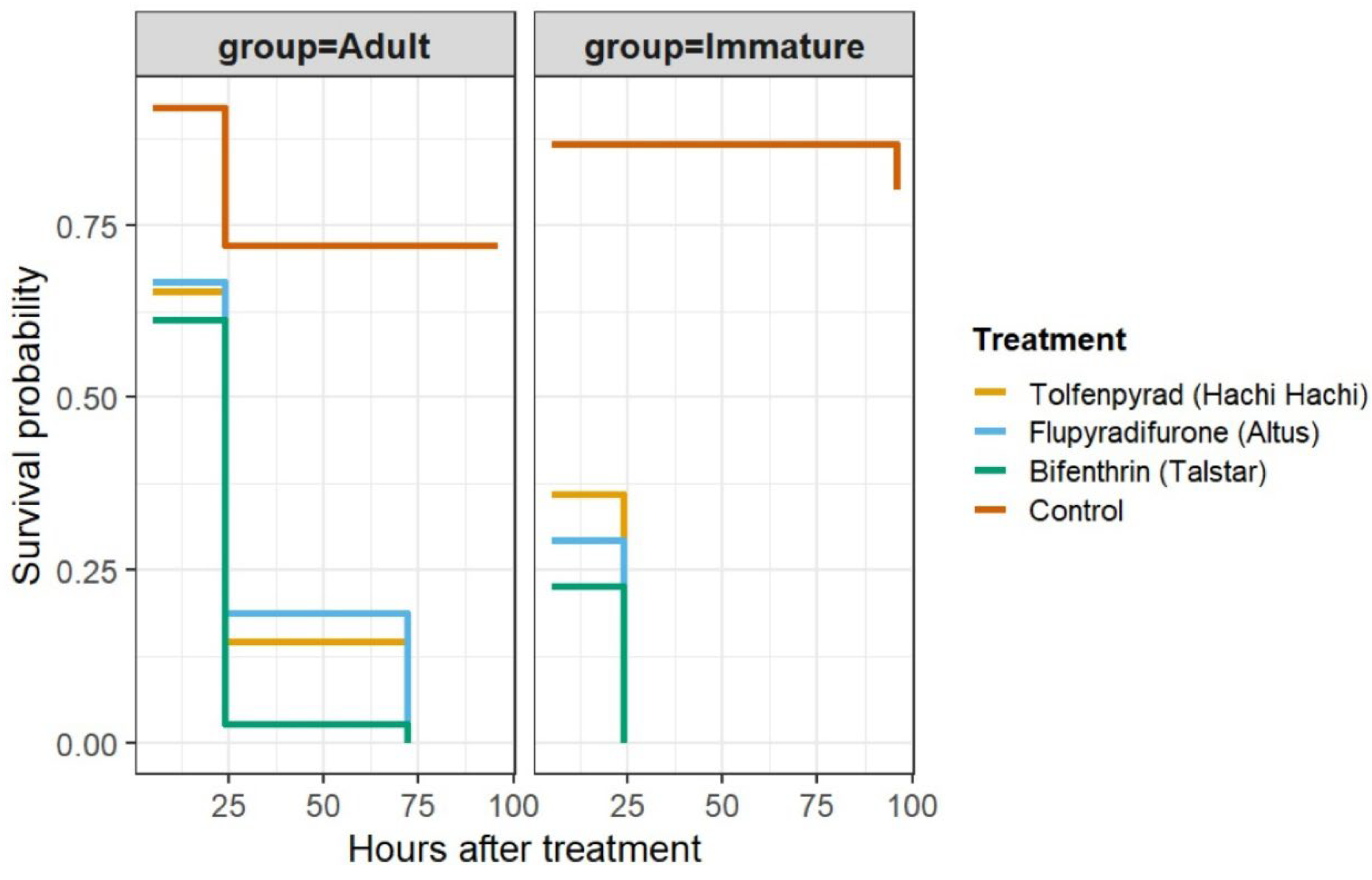
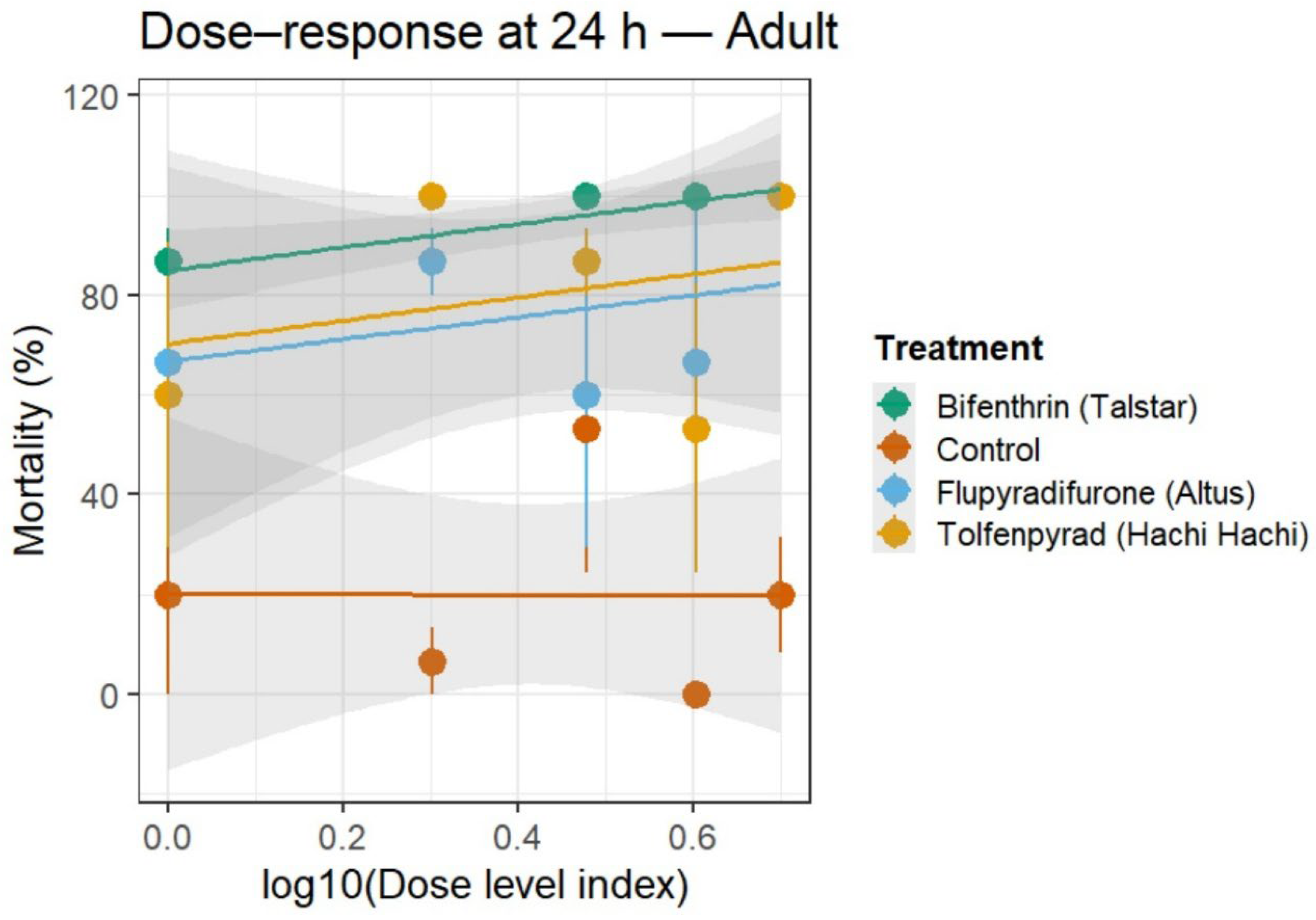
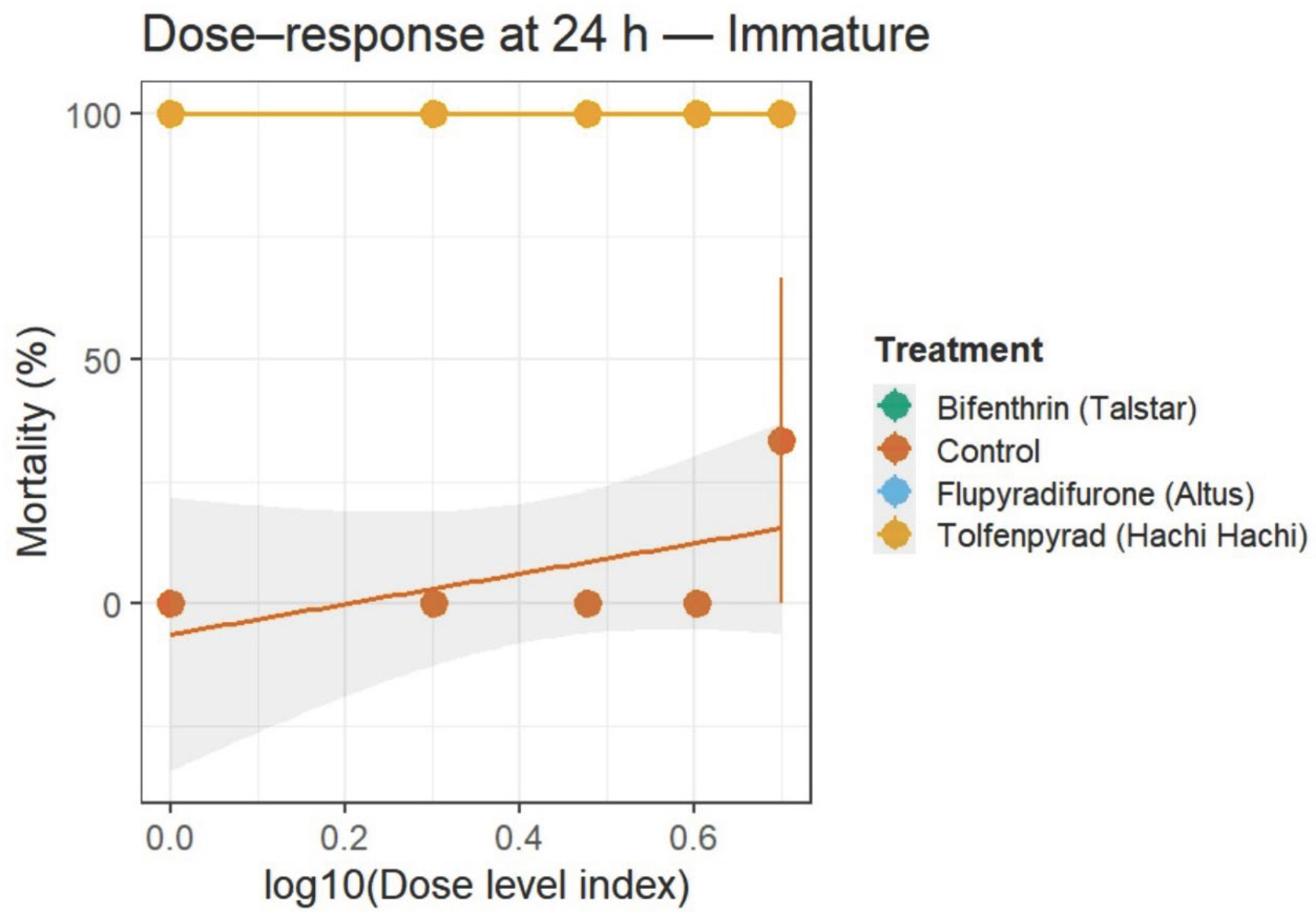
3.2. Residual Toxicity (24–72 h and 72–96 h)
3.3. Minimum Effective Dose (MED) and Time to 90% Mortality (T90)
3.4. Generalized Linear Model (GLM)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ahmed, Z.; Attique, M. R.; Rashid, A. An estimate of the loss in cotton yield in Pakistan attributable to the jassid Amrasca devastans Dist. Crop Protection 1986, 5, 105–108. [Google Scholar] [CrossRef]
- Saeed, R.; Razaq, M.; Hardy, I. C. W. The importance of alternative host plants as reservoirs of the cotton leafhopper, Amrasca devastans, and its natural enemies. Journal of Pest Science 2015, 88, 517–531. [Google Scholar]
- Saeed, R.; Razaq, M.; Abbas, N.; Jan, M. T.; Naveed, M. Toxicity and resistance of the cotton leafhopper, Amrasca devastans (Distant), to neonicotinoid insecticides in Punjab, Pakistan. Crop Protection 2017, 93, 143–147. [Google Scholar]
- Abbas, N.; Ismail, M.; Ejaz, M.; Asghar, I.; Irum, A.; Shad, S. A.; Binyameen, M. Assessment of field-evolved resistance to some broad-spectrum insecticides in cotton jassid, Amrasca devastans (Distant). Phytoparasitica 2018, 46, 411–419. [Google Scholar]
- Hussain, S.; Freed, S.; Naeem, A.; Shah, M. S.; Nazar, M. Z.; Sumra, M. W. Monitoring and biochemical characterization of resistance against commonly used insecticides in cotton jassid, Amrasca biguttula (Ishida), from Punjab, Pakistan. International Journal of Tropical Insect Science 2025, 45, 361–373. [Google Scholar] [CrossRef]
- Cabrera Asencio, I.; Dietrich, C. H.; Zahniser, J. N. A new invasive pest in the Western Hemisphere: Amrasca biguttula (Hemiptera: Cicadellidae). Florida Entomologist 2023, 6 106, 263–266. [Google Scholar] [CrossRef]
- Liburd, O. E.; Revynthi, A.; Liu, Q.; Adkins, S. Detection of the two spot cotton leafhopper (Amrasca biguttula) in Florida and implications for specialty crop production. In UF/IFAS Extension Pest Alert, University of Florida; Gainesville, FL, 2024. [Google Scholar]
- Esquivel, I. L.; Bryant, T.; Malone, S.; Jacobson, A. L.; Graham, S. H.; Cremonez, P. S. Gimenez; Roberts, P.; Moreas, S. Paula; Reisig, D.; Huseth, A.; Greene, J.; Reay-Jones, F. P. F.; Taylor, S. First report of two spot cotton leafhopper (Amrasca biguttula) on commercial cotton in the southeastern United States. Insects 2025, 16, 966. [Google Scholar]
- Ahmed, M. Z.; Yadav, N.; Rustgi, S.; Saripalli, G.; Esquivel, I. L.; Bryant, T. B.; Graham, S.; Jacobson, A. L.; Gireesh, M.; Joseph, S. V.; Del Pozo-Valdivia, A.; Addesso, K. M.; Raszick, T. J.; Khan, R.; Rodrigues, J. C. V.; Bilbo, T. R.; Greene, J. K.; Reay-Jones, F. F. P. Global phylogeography of Amrasca biguttula (Hemiptera: Cicadellidae) across eight countries reveals a single-haplotype incursion into the United States beyond its putative native range. Journal of Applied Entomology 2024, 148, e70067. [Google Scholar] [CrossRef]
- USDA-APHIS. Integration of measures to manage Amrasca biguttula risk in nursery stock movement; U.S. Department of Agriculture, Animal and Plant Health Inspection Service, Plant Protection and Quarantine: Raleigh, NC, 2025; pp. 1–24 pp. [Google Scholar]
- Ahmed, M. Z. Chemical options for managing the invasive two-spot leafhopper on hibiscus. GrowerTalks. 2025. Available online: https://www.growertalks.com/Article/?articleid=27727 (accessed on 11 January 2026).
- Haley, N.; Revynthi, A. M.; Abu, H.; Blare, T. Profitability and cost estimates for producing tropical hibiscus in South Florida: FE1158. EDIS 2024. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2024. [Google Scholar]
- Abbott, W. S. A method of computing the effectiveness of an insecticide. Journal of Economic Entomology 1925, 14 18, 265–267. [Google Scholar] [CrossRef]
| Treatment | Dose | 5 | 24 | 72 | 96 |
| Bifenthrin (Talstar) | D1 | 60.0±20.0 a | 100.0±0.0 a | 100.0±0.0 a | 100.0±0.0 a |
| D2 | 73.3±11.5 a | 100.0±0.0 a | 100.0±0.0 a | 100.0±0.0 a | |
| D3 | 73.3±23.1 a | 100.0±0.0 a | 100.0±0.0 a | 100.0±0.0 a | |
| D4 | 93.3±11.5 a | 100.0±0.0 a | 100.0±0.0 a | 100.0±0.0 a | |
| D5 | 86.7±23.1 a | 100.0±0.0 a | 100.0±0.0 a | 100.0±0.0 a | |
| Control | D1 | 0.0±0.0 b | 0.0±0.0 b | 0.0±0.0 b | 0.0±0.0 b |
| D2 | 6.7±11.5 b | 0.0±0.0 b | 0.0±0.0 b | 33.3±57.7 b | |
| D3 | 26.7±46.2 b | 0.0±0.0 b | 0.0±0.0 b | 0.0±0.0 b | |
| D4 | 0.0±0.0 b | 0.0±0.0 b | 0.0±0.0 b | 0.0±0.0 b | |
| D5 | 33.3±57.7 b | 33.3±57.7 b | 0.0±0.0 b | 0.0±0.0 b | |
| Flupyradifurone (Altus) | D1 | 60.0±34.6 a | 100.0±0.0 a | 100.0±0.0 a | 100.0±0.0 a |
| D2 | 40.0±34.6 a | 100.0±0.0 a | 100.0±0.0 a | 100.0±0.0 a | |
| D3 | 66.7±30.6 a | 100.0±0.0 a | 100.0±0.0 a | 100.0±0.0 a | |
| D4 | 93.3±11.5 a | 100.0±0.0 a | 100.0±0.0 a | 100.0±0.0 a | |
| D5 | 93.3±11.5 a | 100.0±0.0 a | 100.0±0.0 a | 100.0±0.0 a | |
| Tolfenpyrad (Hachi Hachi) | D1 | 33.3±23.1 a | 100.0±0.0 a | 100.0±0.0 a | 100.0±0.0 a |
| D2 | 46.7±30.6 a | 100.0±0.0 a | 100.0±0.0 a | 100.0±0.0 a | |
| D3 | 86.7±11.5 a | 100.0±0.0 a | 100.0±0.0 a | 100.0±0.0 a | |
| D4 | 66.7±23.1 a | 100.0±0.0 a | 100.0±0.0 a | 100.0±0.0 a | |
| D5 | 86.7±11.5 a | 100.0±0.0 a | 100.0±0.0 a | 100.0±0.0 a | |
| Statistical analysis (Treatment effect) |
F = 19.35, P < 0.0001 |
F = 196.00, P < 0.0001 |
F = 2.66 × 10³¹, P < 0.0001 |
F = 196.00, P < 0.0001 |
| Treatment | Dose | 5 | 24 | 72 | 96 |
| Bifenthrin (Talstar) | D1 | 20.0±20.0 a | 86.7±11.5 a | 100.0±0.0 a | 40.0±0.0 a |
| D2 | 20.0±20.0 a | 86.7±11.5 a | 100.0±0.0 a | 40.0±0.0 a | |
| D3 | 33.3±11.5 a | 100.0±0.0 a | 100.0±0.0 a | 40.0±0.0 a | |
| D4 | 53.3±30.6 a | 100.0±0.0 a | 100.0±0.0 a | 40.0±0.0 a | |
| D5 | 66.7±41.6 a | 100.0±0.0 a | 100.0±0.0 a | 40.0±0.0 a | |
| Control | D1 | 0.0±0.0 b | 20.0±34.6 b | 0.0±0.0 b | 0.0±0.0 b |
| D2 | 0.0±0.0 b | 6.7±11.5 b | 0.0±0.0 b | 0.0±0.0 b | |
| D3 | 0.0±0.0 b | 53.3±50.3 b | 0.0±0.0 b | 0.0±0.0 b | |
| D4 | 26.7±46.2 b | 0.0±0.0 b | 0.0±0.0 b | 0.0±0.0 b | |
| D5 | 13.3±23.1 b | 20.0±20.0 b | 0.0±0.0 b | 0.0±0.0 b | |
| Flupyradifurone (Altus) | D1 | 13.3±11.5 a | 66.7±23.1 a | 100.0±0.0 a | 40.0±0.0 a |
| D2 | 20.0±20.0 a | 86.7±11.5 a | 100.0±0.0 a | 40.0±0.0 a | |
| D3 | 26.7±30.6 a | 60.0±52.9 a | 100.0±0.0 a | 40.0±0.0 a | |
| D4 | 26.7±23.1 a | 66.7±57.7 a | 100.0±0.0 a | 40.0±0.0 a | |
| D5 | 80.0±34.6 a | 100.0±0.0 a | 100.0±0.0 a | 40.0±0.0 a | |
| Tolfenpyrad (Hachi Hachi) | D1 | 20.0±34.6 a | 60.0±52.9 a | 100.0±0.0 a | 40.0±0.0 a |
| D2 | 20.0±20.0 a | 100.0±0.0 a | 100.0±0.0 a | 40.0±0.0 a | |
| D3 | 20.0±20.0 a | 86.7±11.5 a | 100.0±0.0 a | 40.0±0.0 a | |
| D4 | 20.0±34.6 a | 53.3±50.3 a | 100.0±0.0 a | 40.0±0.0 a | |
| D5 | 93.3±11.5 a | 100.0±0.0 a | 100.0±0.0 a | 40.0±0.0 a | |
| Statistical analysis (Treatment effect) |
F = 4.57, P = 0.0076 |
F = 19.18, P < 0.0001 |
F = 2.66 × 10³¹, P < 0.0001 |
F = 1.63 × 10³⁰, P < 0.0001 |
| Life Stage | Treatment | MED | T90 |
| Adult | Bifenthrin (Talstar) | — | 24 |
| Adult | Control | — | — |
| Adult | Flupyradifurone (Altus) | — | 72 |
| Adult | Tolfenpyrad (Hachi Hachi) | — | 72 |
| Immature | Bifenthrin (Talstar) | D1 | 24 |
| Immature | Control | — | — |
| Immature | Flupyradifurone (Altus) | D1 | 24 |
| Immature | Tolfenpyrad (Hachi Hachi) | D3 | 24 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




