Submitted:
13 March 2026
Posted:
13 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Azziz, R.; Carmina, E.; Chen, Z.; Dunaif, A.; Laven, J.S.; Legro, R.S.; Lizneva, D.; Natterson-Horowtiz, B.; Teede, H.J.; Yildiz, B.O. Polycystic ovary syndrome. Nat Rev Dis Primers 2016, 2, 16057.
- Rotterdam, E.A.-S.P.c.w.g. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum Reprod 2004, 19, 41-47.
- Yildiz, B.O.; Bozdag, G.; Yapici, Z.; Esinler, I.; Yarali, H. Prevalence, phenotype and cardiometabolic risk of polycystic ovary syndrome under different diagnostic criteria. Hum Reprod 2012, 27, 3067-3073. [CrossRef]
- Balen, A.H.; Conway, G.S.; Kaltsas, G.; Techatrasak, K.; Manning, P.J.; West, C.; Jacobs, H.S. Polycystic ovary syndrome: the spectrum of the disorder in 1741 patients. Hum Reprod 1995, 10, 2107-2111. [CrossRef]
- Norman, R.J.; Masters, L.; Milner, C.R.; Wang, J.X.; Davies, M.J. Relative risk of conversion from normoglycaemia to impaired glucose tolerance or non-insulin dependent diabetes mellitus in polycystic ovarian syndrome. Hum Reprod 2001, 16, 1995-1998. [CrossRef]
- Legro, R.S.; Kunselman, A.R.; Dunaif, A. Prevalence and predictors of dyslipidemia in women with polycystic ovary syndrome. Am J Med 2001, 111, 607-613. [CrossRef]
- Guan, C.; Zahid, S.; Minhas, A.S.; Ouyang, P.; Vaught, A.; Baker, V.L.; Michos, E.D. Polycystic ovary syndrome: a "risk-enhancing" factor for cardiovascular disease. Fertil Steril 2022, 117, 924-935. [CrossRef]
- Gerstein, H.C.; Mann, J.F.; Yi, Q.; Zinman, B.; Dinneen, S.F.; Hoogwerf, B.; Halle, J.P.; Young, J.; Rashkow, A.; Joyce, C.; et al. Albuminuria and risk of cardiovascular events, death, and heart failure in diabetic and nondiabetic individuals. JAMA 2001, 286, 421-426. [CrossRef]
- Barzilay, J.I.; Farag, Y.M.; Durthaler, J. Albuminuria: an underappreciated risk factor for cardiovascular disease. Journal of the American Heart Association 2024, 13, e030131. [CrossRef]
- Boorsma, E.M.; Ter Maaten, J.M.; Damman, K.; Van Essen, B.J.; Zannad, F.; Van Veldhuisen, D.J.; Samani, N.J.; Dickstein, K.; Metra, M.; Filippatos, G. Albuminuria as a marker of systemic congestion in patients with heart failure. Eur Heart J 2023, 44, 368-380. [CrossRef]
- Koroshi, A. Microalbuminuria, is it so important? Hippokratia 2007, 11, 105-107.
- Carmina, E.; Orio, F.; Palomba, S.; Longo, R.A.; Cascella, T.; Colao, A.; Lombardi, G.; Rini, G.B.; Lobo, R.A. Endothelial dysfunction in PCOS: role of obesity and adipose hormones. Am J Med 2006, 119, 356 e351-356. [CrossRef]
- Kelly, C.J.; Speirs, A.; Gould, G.W.; Petrie, J.R.; Lyall, H.; Connell, J.M. Altered vascular function in young women with polycystic ovary syndrome. J Clin Endocrinol Metab 2002, 87, 742-746. [CrossRef]
- Patel, A.A.; Bloomgarden, Z.T.; Futterweit, W. Premicroalbuminuria in women with polycystic ovary syndrome: a metabolic risk marker. Endocr Pract 2008, 14, 193-200. [CrossRef]
- Duleba, A.J.; Ahmed, I.M. Predictors of urinary albumin excretion in women with polycystic ovary syndrome. Fertil Steril 2010, 93, 2285-2290. [CrossRef]
- de Groot, P.C.; Dekkers, O.M.; Romijn, J.A.; Dieben, S.W.; Helmerhorst, F.M. PCOS, coronary heart disease, stroke and the influence of obesity: a systematic review and meta-analysis. Hum Reprod Update 2011, 17, 495-500. [CrossRef]
- Ziaee, A.; Oveisi, S.; Ghorbani, A.; Hashemipour, S.; Mirenayat, M. Association between metabolic syndrome and premicroalbuminuria among Iranian women with polycystic ovary syndrome: a case control study: met syn. and premicroalbuminuria in PCOS. Glob J Health Sci 2012, 5, 187. [CrossRef]
- Azziz, R. Epidemiology, phenotype, and genetics of the polycystic ovary syndrome in adults. Available online: https://www.uptodate.com/contents/epidemiology-phenotype-and-genetics-of-the-polycystic-ovary-syndrome-in-adults?search=polycystic%20ovarian%20syndrome%20subtype&source=search_result&selectedTitle=1%7E150&usage_type=default&display_rank=1 (accessed on November 21, 2023).
- Mitra, S.; Saharia, G.K.; Jena, S.K. Cardio-metabolic risk in Rotterdam clinical phenotypes of PCOS. In Proceedings of the Annales d'Endocrinologie, 2024; pp. 44-47. [CrossRef]
- Berbrier, D.E.; Leone, C.A.; Adler, T.E.; Bender, J.R.; Taylor, H.S.; Stachenfeld, N.S.; Usselman, C.W. Effects of androgen excess and body mass index on endothelial function in women with polycystic ovary syndrome. J Appl Physiol 2023, 134, 868-878. [CrossRef]
- Gungor, K.; Gungor, N.D.; Celik, O.; Ersahin, A.; Celik, N.; Yardim, M.; Yurci, A.; Kobaner, M.; Ilkov Maslarski, I. Polycystic ovary syndrome accompanied by hyperandrogenemia or metabolic syndrome triggers glomerular podocyte injury. Diagnostics 2024, 14, 2197. [CrossRef]
- Can, M.; Duran, C.; Guney, I.; Elmas, H.; Ayhan, M.; Erdem, S.S. The relationship between glomerular filtration rate, and metabolic and inflammatory parameters in obese and non-obese patients with polycystic ovary syndrome. Clin Investig Arterioscler (English Edition) 2020, 32, 256-262.
- Teede, H.J.; Tay, C.T.; Laven, J.J.; Dokras, A.; Moran, L.J.; Piltonen, T.T.; Costello, M.F.; Boivin, J.; Redman, L.M.; Boyle, J.A. Recommendations from the 2023 international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Eur J Endocrinol 2023, 189, G43-G64. [CrossRef]
- Kamrul-Hasan, A.B.; Aalpona, F.Z.; Chanda, P.K.; Ananya, K.F.; Kobra, T.; Miah, O.F.; Kazal, R.K.; Selim, S. Frequency and Correlates of Albuminuria in Adult Bangladeshi Women with Polycystic Ovary Syndrome. Mymensingh Med J 2020, 29, 234-240.
- Tay, C.T.; Mousa, A.; Vyas, A.; Pattuwage, L.; Tehrani, F.R.; Teede, H. 2023 International Evidence-Based Polycystic Ovary Syndrome Guideline Update: insights from a systematic review and meta-analysis on elevated clinical cardiovascular disease in polycystic ovary syndrome. J Am Heart Assoc 2024, 13, e033572. [CrossRef]
- Moriconi, D.; Nannipieri, M.; Jadoon, M.; Solini, A.; Bruno, R.M. Albumin-To-Creatinine Ratio Underestimates True 24-Hour Albuminuria in Obesity: Clinical Relevance for Vascular Risk Stratification. Diabetes Metab Res Rev 2025, 41, e70064. [CrossRef]
- Moriconi, D.; Nannipieri, M.; Jadoon, M.; Solini, A.; Bruno, R. Albumin-To-Creatinine Ratio Underestimates True 24-Hour Albuminuria in Obesity: Clinical Relevance for Vascular Risk Stratification. Diabetes Metab Res Rev 2025, 41, e70064. [CrossRef]
- Borzan, V.; Lerchbaum, E.; Missbrenner, C.; Heijboer, A.C.; Goschnik, M.; Trummer, C.; Theiler-Schwetz, V.; Haudum, C.; Gumpold, R.; Schweighofer, N.; et al. Risk of Insulin Resistance and Metabolic Syndrome in Women with Hyperandrogenemia: A Comparison between PCOS Phenotypes and Beyond. J Clin Med 2021, 10. [CrossRef]
- Marchesan, L.B.; Ramos, R.B.; Spritzer, P.M. Metabolic Features of Women With Polycystic Ovary Syndrome in Latin America: A Systematic Review. Front Endocrinol (Lausanne) 2021, 12, 759835. [CrossRef]
- Walford, H.; Tyler, B.; Abbara, A.; Clarke, S.; Talaulikar, V.; Wattar, B.A. Biomarkers to inform the management of polycystic ovary syndrome: A review of systematic reviews. Clin Endocrinol 2024, 101, 535-548. [CrossRef]
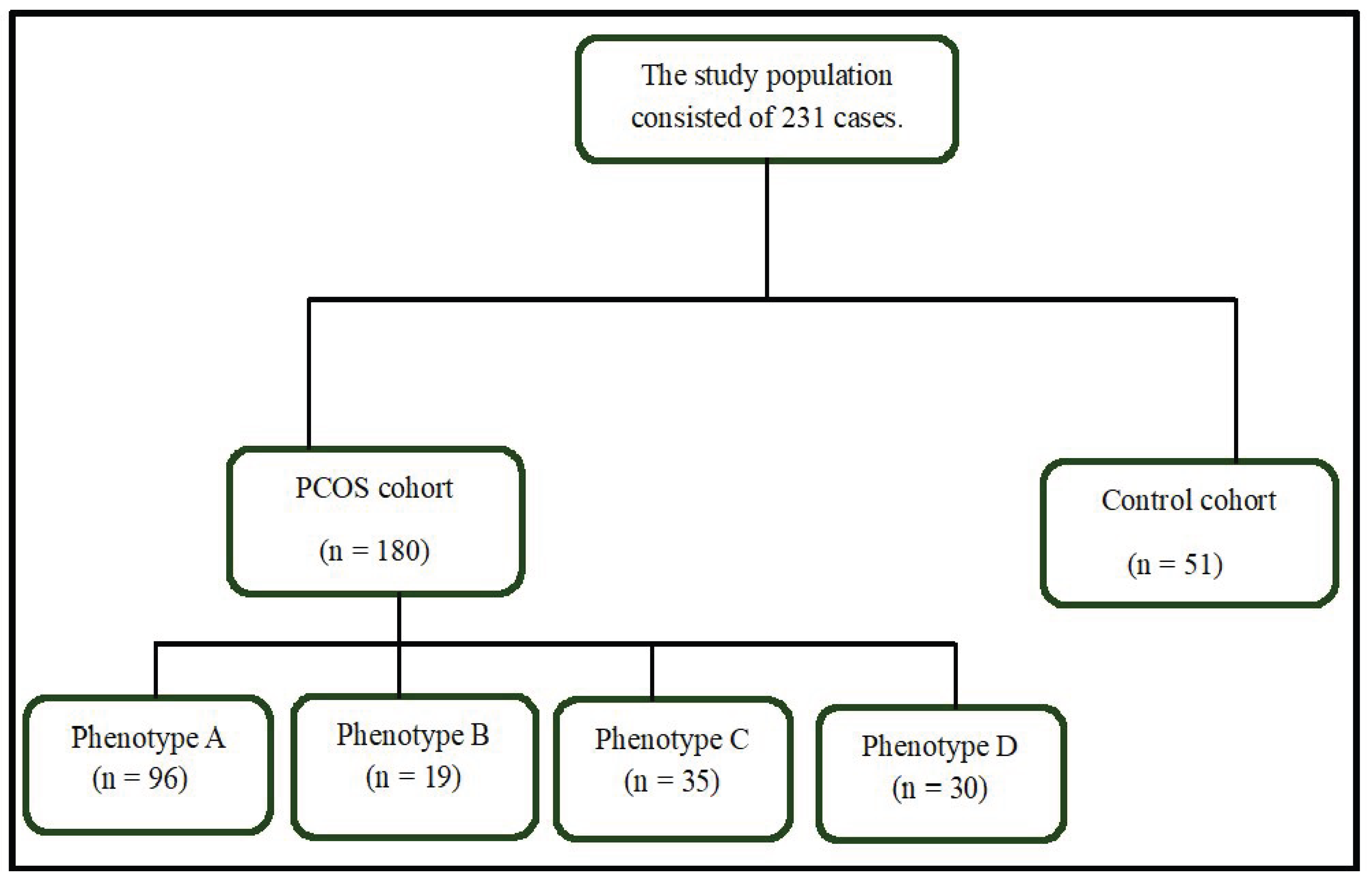
| Control Group (n = 51) |
PCOS Subtype 1 (n = 96) | PCOS Subtype 2 (n = 19) | PCOS Subtype 3 (n = 35) | PCOS Subtype 4 (n = 30) | PCOS (n = 180) |
p1 | p2 | p3 | p4 | p5 | p6 | p7 | p8 | p9 | p10 | p11 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Age (year) | 23 (22-33) | 25 (18-35) | 22 (18-34) | 25 (18-35) | 24 (20-35) | 24 (18-35) | 0.191 | 0.191 | 0.950 | 0.408 | 0.103 | 0.353 | 0.954 | 0.322 | 0.099 | 0.500 | 0.457 |
| BMI (kg/m²) | 21.48 (17.78-27.34) | 27.19 (17.15-49.77) | 24.83 (15.78-39.84) | 22.14 (15.59-34.08) | 24.88 (17.19-50.22) | 25,49 (15,59-50,22) | < 0.001 | 0.028 | 0.146 | 0.006 | 0.111 | < 0.001 | 0.031 | 0.174 | 0.766 | 0.184 | < 0.001 |
| WC (cm) | 75 (59-100) | 90 (61-129) | 82 (60-120) | 77 (58-105) | 78 (60-116) | 84 (58-129) | < 0.001 | 0.041 | 0.237 | 0.160 | 0.065 | < 0.001 | 0.005 | 0.269 | 0.689 | 0.562 | < 0.001 |
| SBP (mmHg) | 110 (90-130) | 110 (70-150) | 110 (102-160) | 110 (94-140) | 110 (90-140) | 110 (70-160) | 0.076 | 0.152 | 0.774 | 0.728 | 0.783 | 0.199 | 0.065 | 0.185 | 0.043 | 0.544 | 0.194 |
| DBP (mmHg) | 70 (50-90) | 70 (50-93) | 70 (60-90) | 70 (50-90) | 70 (60-90) | 70 (50-93) | 0.048 | 0.227 | 0.799 | 0.580 | 0.846 | 0.040 | 0.035 | 0.146 | 0.125 | 0.721 | 0.239 |
| Control Group (n = 51) |
PCOS Subtype 1 (n = 96) | PCOS Subtype 2 (n = 19) | PCOS Subtype 3 (n = 35) | PCOS Subtype 4 (n = 30) | PCOS (n = 180) |
p1 | p2 | p3 | p4 | p5 | p6 | p7 | p8 | p9 | p10 | p11 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Glucose (mg/dL) | 87 (73-98) | 90 ((56-142) | 88 (75-109) | 88 (60-103) | 88 (78-103) | 89 (56-142) | 0.54 | 0.204 | 0.588 | 0.327 | 0.976 | 0.280 | 0.521 | 0.462 | 0.622 | 0.732 | 0.082 |
| Insulin (μU/mL) | 7.32 (2.80-22.00) | 13.70 (3.60-100.50) | 13.60 (4.40-35.70) | 10.10 (2.50-37.80) | 9.60 (2.00-30.50) | 12.59 (2.00-100.50) | < 0.001 | < 0.001 | 0.010 | 0.024 | 0.810 | 0.009 | 0.006 | 0.094 | 0.071 | 0.797 | < 0.001 |
| HOMA-IR | 1.63 (0.58-4.35) | 2.96 (0.77-25.57 | 3.06 (0.92-7.31) | 2.30 (0.55-7.54) | 2.13 (0.38-6.55) | 2.75 (0.38-25.57) | < 0.001 | < 0.001 | 0.120 | 0.024 | 0.775 | 0.008 | 0.008 | 0.094 | 0.071 | 0.813 | < 0.001 |
| PC (mg/dl) | 0.65 (0.60-0.90) | 0.66 (0.50-1.00) | 0.66 (0.60-0.80) | 0.66 (0.50-0.80) | 0.64 (0.50-0.90) | 0.66 (0.50-1.00) | 0.190 | 0.439 | 0.529 | 0.499 | 0.859 | 0.073 | 0.105 | 0.264 | 0.366 | 0.787 | 0.569 |
| CKD-EPI | 122.31 (90.12-129.38) | 121.79 (78.24-137.38) | 119.58 (96.19-132.14) | 123.17 (105.39-137.38) | 123.42 (82.84-136.42) | 122.10 (78.20-137.40) | 0.034 | 0.551 | 0.788 | 0.914 | 0.904 | 0.030 | 0.180 | 0.379 | 0.704 | 0.906 | 0.213 |
| UC (mg/dL) | 103.87 (6.50-381.10) | 126.31 (15.30-419.30) | 120.30 (14.00-330.70) | 96.42 (8.10-246.40) | 137.82 (6.70-395.00) | 122.62 (6.70-419.30) | 0.221 | 0.797 | 0.676 | 0.207 | 0.588 | 0.104 | 0.735 | 0.520 | 0.552 | 0.117 | 0.340 |
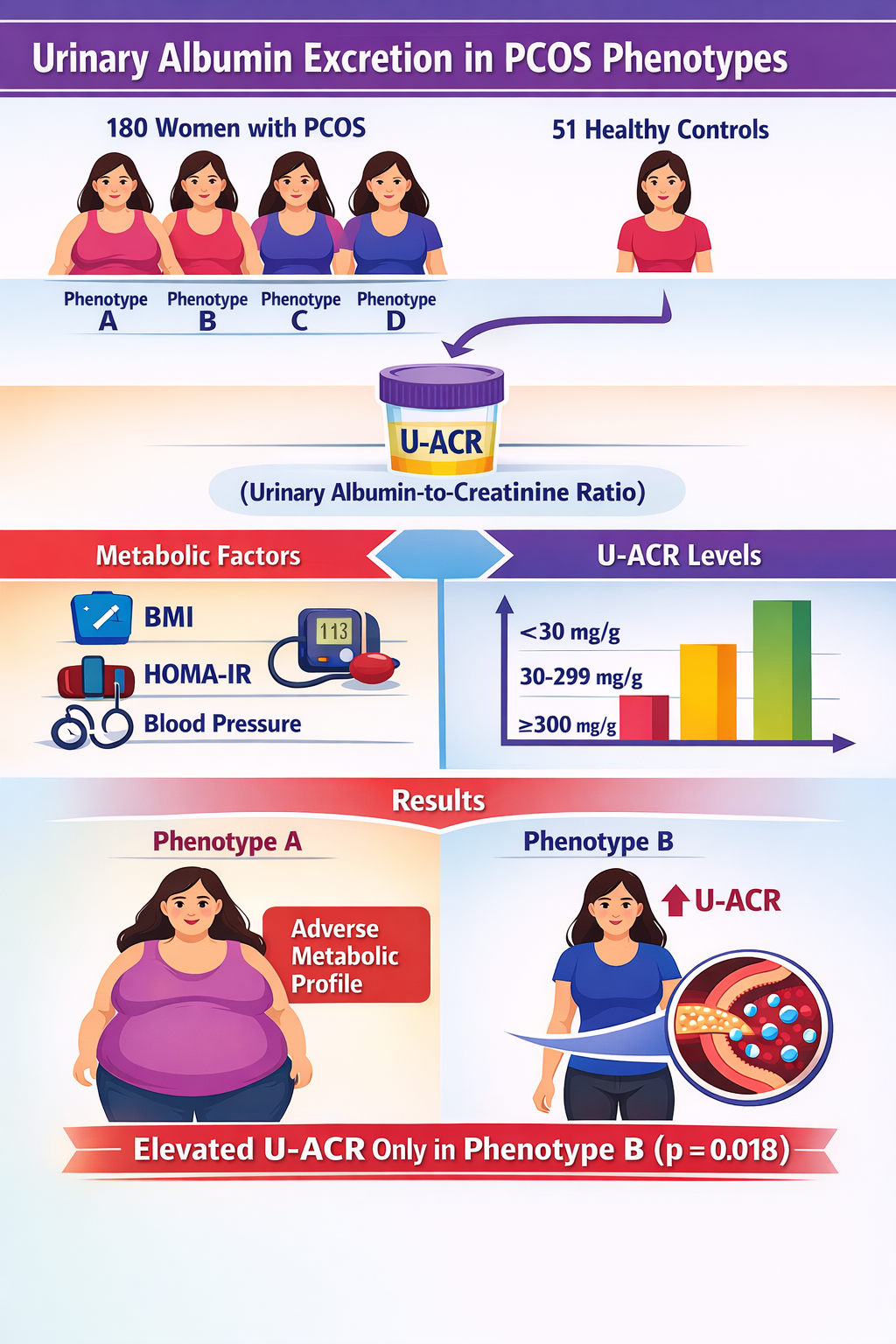
| UA mg/L | 15 (5-468) | 22 (5-616) | 45 (5-433) | 12 (5-253) | 15 (4-159) | 21 (4-616) | 0.014 | 0.007 | 0.965 | 0.691 | 0.352 | 0.022 | 0.111 | 0.008 | 0.047 | 0.663 | 0.045 |
| U-ACR mg/g creatinin | 12.38 (5.25-426.62) | 22.81 (3.97-334.43) | 38.95 (5.49-164.72) | 17.78 (5.13-172.63) | 13.62 (2.52-152.99) | 19.66 (2.52-426.62) | 0.143 | 0.018 | 0.513 | 0.876 | 0.096 | 0.526 | 0.135 | 0.056 | 0.016 | 0.445 | 0.156 |
| U-ACR category | The Control Group (n = 51) |
Phenotype A (n = 96) |
Phenotype B (n = 19) |
Phenotype C (n = 35) |
Phenotype D (n = 30) |
Total PCOS (n = 180) |
p1 | p2 |
|---|---|---|---|---|---|---|---|---|
| < 29.9 mg/g creatinine | 37 (72.5) | 59 (61.5) | 7 (36.8) | 22 (62.9) | 22 (73.3) | 110 (61.1) | p = 0.105 | p = 0.84 |
| 30-299 mg/g creatinine | 12 (23.5) | 35 (36.5) | 12 (63.2) | 13 (37.1) | 8 (26.7) | 68 (37.8) | ||
| > 300 mg/g creatinine | 2 (3.9) | 2 (2.1) | 0 (0) | 0 (0) | 0 (0) | 2 (1.1) |
| Age (year) | BMI (kg/m2) | HOMA-IR | U-ACR (mg/g creatinine) | |
|---|---|---|---|---|
| Age (year) | r = 1 | r = 0.235 p < 0.001 |
r = 0.011 p = 0.872 |
r = -0.100 p = 0.128 |
| BMI (kg/m2) | r = 0.235 p < 0.001 |
r = 1 | r = 0.537 p < 0.001 |
r = -0.022 p = 0.738 |
| HOMA-IR | r = 0.011 p = 0.872 |
r = 0.537 p < 0.001 |
r = 1 | r = -0.007 p = 0.920 |
| U-ACR (mg/g creatinine) | r = -0.100 p = 0.128 |
r = -0.022 p = 0.738 |
r = -0.007 p = 0.920 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




