Submitted:
12 March 2026
Posted:
13 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Solid State Reaction
2.2. Coprecipitation Method
2.3. Hydrothermal/Solvothermal Method
2.4. Sol-Gel Route
2.5. Microwave-Assisted Method
3. Results
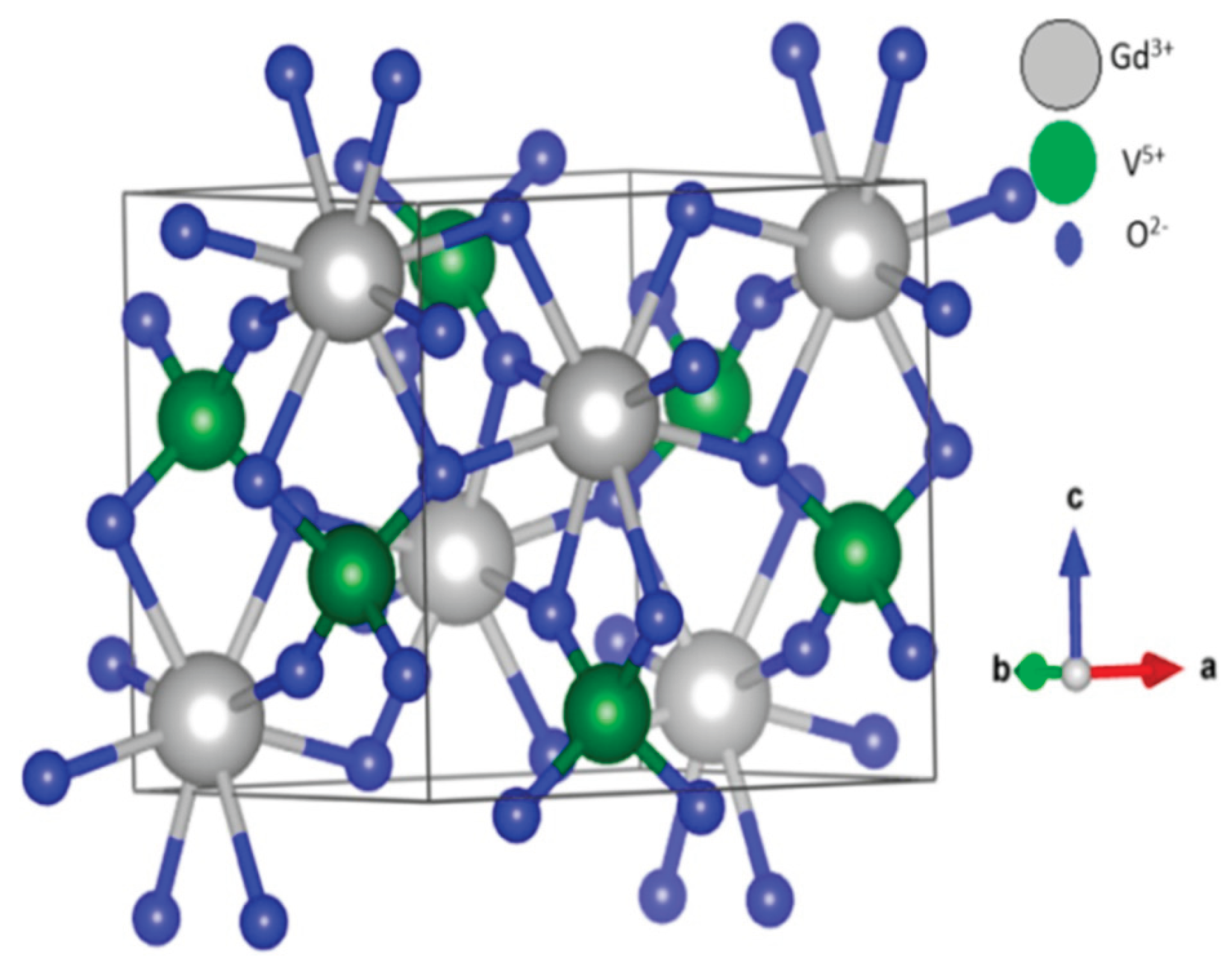
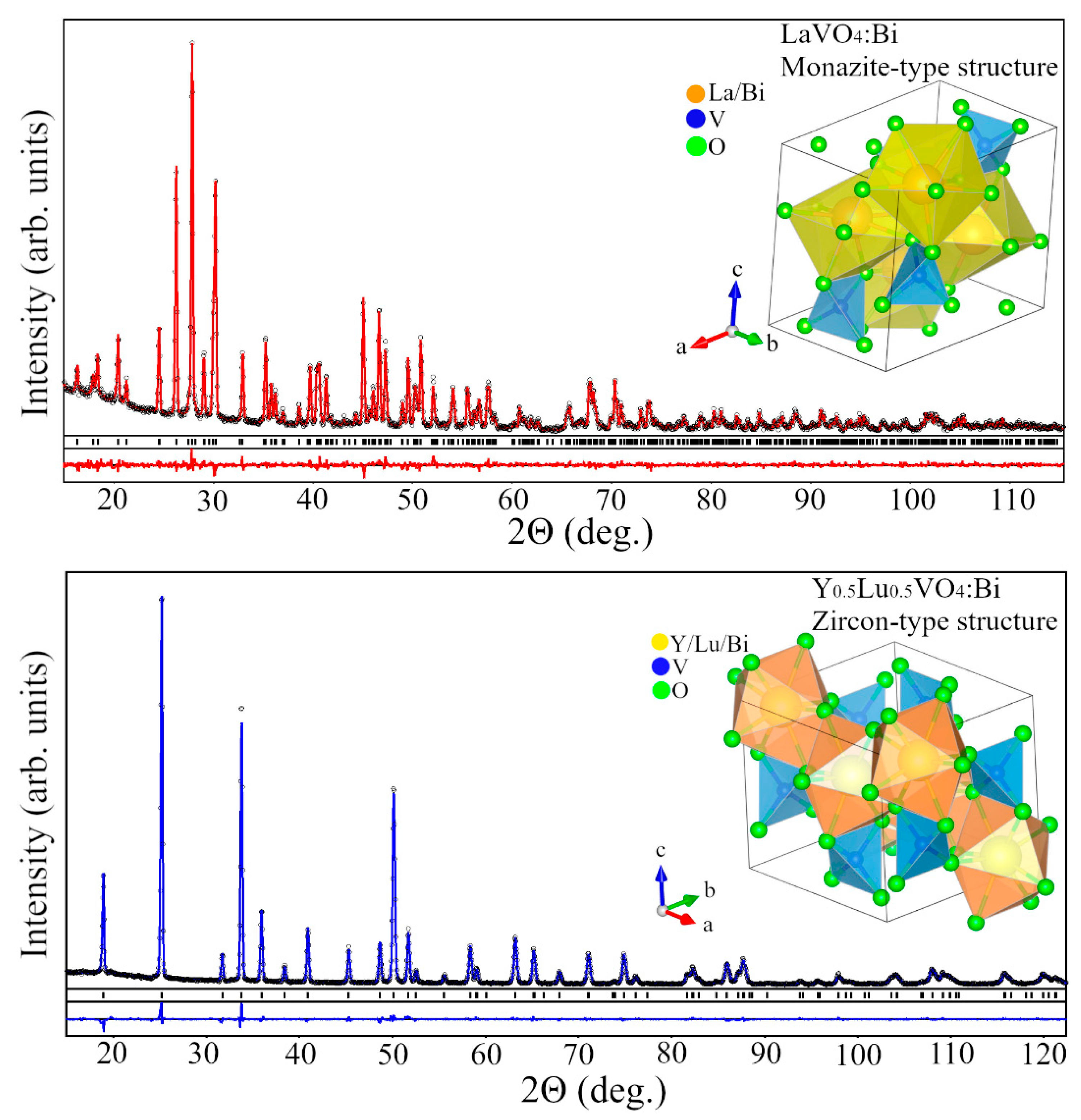
3.1. Crystal Structure of the AVO₄ Materials
3.2. Morphology of Luminescent REVO4 Materials
4. Discussion
4.1. Optical Properties of REVO4 Materials
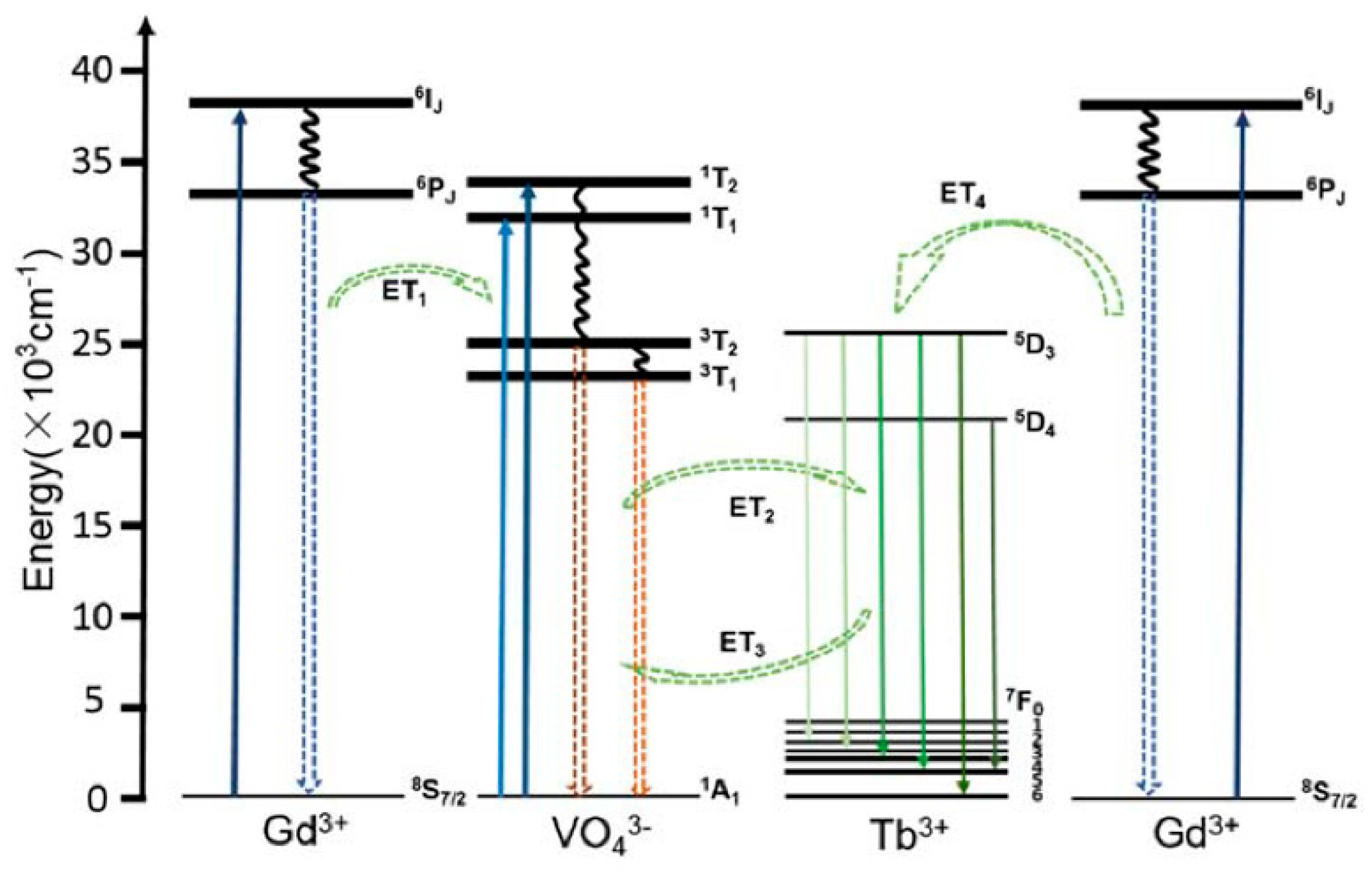
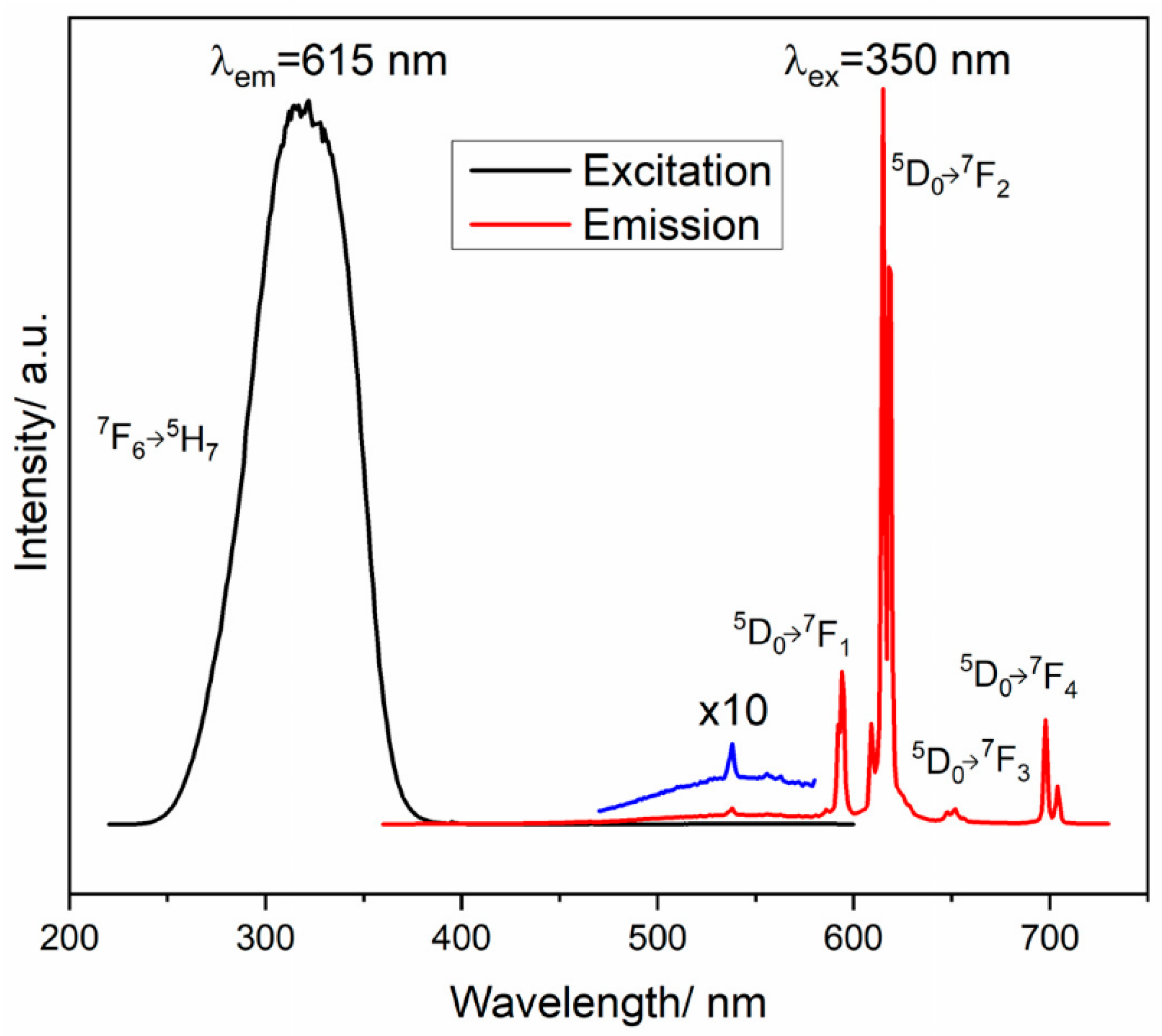
4.1.1. Downconversion REVO4-Based Luminescent Materials and Sensing Applications
4.1.2. Upconversion REVO4-Based Luminescent Materials and Sensing Applications
5. Sensing Applications: Recent Advances
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ma, X.-F.; Li, H.-Y.; Ren, W.; Gao, D.; Chen, F.; Diao, J.; Xie, B.; Huang, G.; Wang, J.; Pan, F. A critical review of vanadium-based electrode materials for rechargeable magnesium batteries. J. Mater. Sci. Tech. 2023, 153, 56–74. [Google Scholar] [CrossRef]
- Silva-Ramírez, G.A.; Garrido-Hernández, A.; Carrera-Jota, M.L.; García-Hernández, M.; Hernández-Fuentes, C.F.; Morales-Ramírez, A.J. Differences in structural and luminescent properties of GdVO4:Sm3+, Bi3+ powder and sol-gel derived films with varying Sm3+ contents and annealing temperatures. Ceram. Int. 2025, 51, 8913–8929. [Google Scholar] [CrossRef]
- Sisó-Moliné, R.; Cascales, C.; Méndez, M.; Fuentes-Rodriguez, L.; de Aquino, A.; Pujol, M.C.; Zaldo, C.; Carvajal, J.J. Preparation and characterization of GdVO4:Tb,Eu nanoparticles/carbon dots composites via hydrothermal deposition and physical mixing for luminescent nanothermometry. Adv. Mater. Interfaces 2025, 12, e00410. [Google Scholar] [CrossRef]
- Hrabovsky, J.; Kucera, M.; Palousova, L.; Zazvorka, J.; Kubat, J.; Bi, L.; Veis, M. Rapid and precise large area mapping of rare-earth doping homogeneity in luminescent materials. Commun. Mater. 2024, 5, 251. [Google Scholar] [CrossRef]
- Soharab, M.; Bhatt, R.; Benerji, N.S.; Bhaumik, I. Comparison of optical properties and self Q-switched lasing in Cr-Nd co-doped GdVO4 and YVO4 single crystals. Physica B: Condens. Matter 2025, 707, 417171. [Google Scholar] [CrossRef]
- Dahiya, B.; Nain, S. Structural and dielectric properties of rare earth-doped GdVO4/ZnO composites for opto-electronic applications. Ceram. Int. 2025, 51, 21448–21460. [Google Scholar] [CrossRef]
- Li, M.; Li, J.-G. Extensive tailoring of REPO4 and REVO4 crystallites via solution processing and luminescence. CrystEngComm 2022, 24, 4841–4852. [Google Scholar] [CrossRef]
- Yan, H.; Shao, E.; Qi, X.; Du, Y.; Wang, C. Low-temperature bonding of Nd: YVO4 crystals with superior interfaces for high-power solid-state lasers. Appl. Surf. Sci. 2025, 703, 163426. [Google Scholar] [CrossRef]
- Kruczek, M.; Talik, E.; Sakowska, H.; Ujma, Z.; Skrzypek, D. XPS investigations of YVO4:Tm,Yb single crystal. J. Crystal Growth 2005, 275, e1715–e1720. [Google Scholar] [CrossRef]
- Dai, W.; Liu, T.; Ding, Y.; Zhao, Y.; Zhang, Y. High-efficiency Nd:LuVO4 laser at 1343 nm recycling-pumped by a laser diode at 916 nm. Front. Phys. 2024, 12, 1388372. [Google Scholar] [CrossRef]
- Jovanović, D.J.; Chiappini, A.; Zur, L.; Gavrilović, T.V.; Tran, T.N.L.; Chiasera, A.; Lukowiak, A.; Smits, K.; Dramićanin, M.D.; Ferrari, M. Synthesis, structure and spectroscopic properties of luminescent GdVO4:Dy3+ and DyVO4 particles. Opt. Mater. 2018, 76, 308–316. [Google Scholar] [CrossRef]
- Antić, Ž.; Dramićanin, M.D.; Prashanthi, K.; Jovanović, D.; Kuzman, S.; Thundat, T. Pulsed laser deposited dysprosium-doped gadolinium–vanadate thin films for noncontact, self-referencing luminescence thermometry. Adv. Mater. 2016, 28, 7745–7752. [Google Scholar] [CrossRef] [PubMed]
- Gavrilović, T.V.; Jovanović, D.J.; Smits, K.; Dramićanin, M.D. Multicolor upconversion luminescence of GdVO4:Ln3+/Yb3+ (Ln3+ = Ho3+, Er3+, Tm3+, Ho3+/Er3+/Tm3+) nanorods. Dyes Pigm 2016, 126, 1–7. [Google Scholar] [CrossRef]
- del Rosal, B.; Perez-Delgado, A.; Misiak, M.; Bednarkiewicz, A.; Vanetsev, A.S.; Orlovskii, Y.; Jovanović, D.J.; Dramićanin, M.D.; Rocha, U.; Kumar, K.U.; Jacinto, C.; Navarro, E.; Rodriguez, E.M.; Pedroni, M.; Speghini, A.; Hirata, G. A.; Martin, I.R.; Jaque, D. Neodymium-doped nanoparticles for infrared fluorescence bioimaging: The role of the host. J. Appl. Phys. 2015, 118, 143104. [Google Scholar] [CrossRef]
- Pan, X.; Hu, M.; Wu, L.; Wei, E.; Zhu, Q.; Lv, L.; Xv, X.; Dong, X.; Liu, H.; Liu, Y. Biomedical applications of gadolinium-containing biomaterials: Not only MRI contrast agent. Adv Sci. (Weinh) 2025, 12, 2501722. [Google Scholar] [CrossRef]
- Mahata, M.K.; Kumar, K.; Rai, V.K. Er3+–Yb3+ doped vanadate nanocrystals: A highly sensitive thermographic phosphor and its optical nanoheater behaviour. Sens. Actuators B Chem. 2015, 209, 775–780. [Google Scholar] [CrossRef]
- Kabbani, M.A.; Tiwary, C.S.; Autreto, P.A.S.; Brunetto, G.; Som, A.; Krishnadas, K.R.; Ozden, S.; Hackenberg, K.P.; Gong, Y.; Galvao, D.S.; Vajtai, R.; Kabbani, A.T.; Pradeep, T.; Ajayan, P.M. Ambient solid-state mechano-chemical reactions between functionalized carbon nanotubes. Nature Commun. 2015, 6, 7291. [Google Scholar] [CrossRef]
- Huang, Z.; Zhang, L.; Pan, W. Synthesis, lattice dynamics, and mechanical properties of a high-pressure scheelite phase of RVO4. Inorg. Chem. 2012, 51, 11235–11237. [Google Scholar] [CrossRef]
- Gavrilović, T.V.; Jovanović, D.J.; Trandafilović, L.V.; Dramićanin, M.D. Effects of Ho3+ and Yb3+ doping concentrations and Li+ co-doping on the luminescence of GdVO4 powders. Opt. Mater. 2015, 45, 76–81. [Google Scholar] [CrossRef]
- Huignard, A.; Gacoin, T.; Boilot, J.-P. Synthesis and luminescence properties of colloidal YVO4:Eu phosphors. Chem. Mater. 2000, 12, 1090–1094. [Google Scholar] [CrossRef]
- Tang, S.; Huang, M.; Wang, J.; Yu, F.; Shang, G.; Wu, J. Hydrothermal synthesis and luminescence properties of GdVO4:Ln3+ (Ln = Eu, Sm, Dy) phosphors. J. Alloys Compd. 2012, 513, 474–480. [Google Scholar] [CrossRef]
- Xu, Z.; Feng, B.; Gao, Y.; Zhao, Q.; Sun, D.; Gao, X.; Li, K.; Ding, F.; Sun, Y. Uniform and well-dispersed GdVO4 hierarchical architectures: hydrothermal synthesis, morphology evolution, and luminescence properties. CrystEngComm. 2012, 14, 145530–5538. [Google Scholar] [CrossRef]
- Li, X.-B.; Geng, Y.-C.; Wang, A.-Q.; Xi, B.-Y.; Zhang, J.-Q.; Dang, W.-Q.; Ling, W.-J.; Dong, Z. Preparation of zircon type GdVO and study of its gas sensitivity properties. Ceram. Int. 2025, 51, 59138–59148. [Google Scholar] [CrossRef]
- Silva-Ramírez, G.A.; Garrido-Hernández, A.; Carrera-Jota, M.L.; García-Hernández, M.; Hernández-Fuentes, C.F.; Morales-Ramírez, A.J. Differences in structural and luminescent properties of GdVO4:Sm3+, Bi3+ powder and sol-gel derived films with varying Sm3+ contents and annealing temperatures. Ceram. Int. 2025, 51, 8913–8929. [Google Scholar] [CrossRef]
- Roopana, S.M.; Chellapandia, Thangapandi; Shebeer, R.M.; Akhil, E.; Alappat, J.D.; Nair, N.R.; Madhusoodanan, M.; Chitra, D. Advances and prospects in the development of GdVO4-based photocatalysts for water pollutants removal activity: A review. Nano Mater. Sci. 2025, 7, 180–197. [Google Scholar] [CrossRef]
- Chumha, N.; Kittiwachana, S.; Thongtem, T.; Thongtem, S.; Kaowphon, S. Synthesis and characterization of GdVO4 nanostructures by a tartaric acid-assisted sol–gel method. Ceram. Int. 2014, 16337–16342. [Google Scholar] [CrossRef]
- Dhakal, D.R.; Kshetri, Y.K.; Chaudhary, B.; Choi, J.-h.; Murali, G.; Kim, T.H. Enhancement of upconversion luminescence in Yb3+/Er3+-doped BiVO4 hrough calcination. Mater. Today Commun. 2023, 37, 107258. [Google Scholar] [CrossRef]
- Godinho, M.; Ribeiro, C.; Longo, E; Leite, E.R. Influence of microwave heating on the growth of gadolinium-doped cerium oxide nanorods. Cryst. Growth Des. 2008, 8, 384–386. [Google Scholar] [CrossRef]
- Ryu, Sung-Myung; Nam, Chunghee. Shape-controlled microwave hydrothermal synthesis of antiferromagnetic GdVO4 nanowires for magnetocaloric materials. Curr. Appl. Phys. 2020, 20, 715–719. [Google Scholar] [CrossRef]
- Thakur, H.; Singh, R.K.; Gathania, A.K. Synthesis and optical properties of GdVO4: Eu3+ phosphor. Mater. Res. Express 2021, 8, 026201. [Google Scholar] [CrossRef]
- Unoki, H. The secondary effect of the cooperative Jahn-Teller distortion in rare earth vanadate. J. Magn. Magn. Mater. 1983, 31-34, 1063–1064. [Google Scholar] [CrossRef]
- Lotfi, S.; Ouardi, M.E.; Ahsaine, H.A.; Madigou, V.; BaQais, A.; Assani, A.; Saadi, M.; Arab, M. Low-temperature synthesis, characterization and photocatalytic properties of lanthanum vanadate LaVO4. Heliyon 2023, 9, e17255. [Google Scholar] [CrossRef]
- Guo, H.; Lu, Q.; Xie, J.; Wu, D.; Chen, Z. Energy transfer mechanism and optical properties of Eu3+, Dy3+ co-doped LaVO4 phosphors. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2025, 339, 126292. [Google Scholar] [CrossRef]
- Vasylechko, L.; Tupys, A.; Hreb, V.; Tsiumra, V.; Lutsiuk, I.; Zhydachevskyy, Y. New mixed Y0.5R0.5VO4 and RVO4:Bi materials: Synthesis, crystal structure and some luminescence properties. Inorganics 2018, 6(3), 94. [Google Scholar] [CrossRef]
- Chen, B. Luminescent materials with advanced properties and applications. Inorganics 2024, 12(12), 306. [Google Scholar] [CrossRef]
- Li, J.; Tian, X.; Bao, W.; Zhao, J.; Jiang, Z. Recent advances in the development and applications of lanthanide-based luminescent materials: a comprehensive review. Discov Appl Sci. 2025, 7, 1310. [Google Scholar] [CrossRef]
- Eker, F.; Duman, H.; Akdaşçi, E.; Bolat, E.; Sarıtaş, S.; Karav, S.; Witkowska, A.M. A comprehensive review of nanoparticles: from classification to application and toxicity. Molecules 2024, 29(15), 3482. [Google Scholar] [CrossRef]
- Ragupathi, C.; Narayanan, S.; Tamizhdurai, P.; Sukantha, T.A.; Ramalingam, G.; Pachamuthu, M.P.; Mangesh, V.L.; Kumar, N.S.; Al-Fatesh, A.S.; Kasim, S.O. Correlation between the particle size, structural and photoluminescence spectra of nano NiCr2O4 and La doped NiCr2O4 materials. Heliyon 2023, 9(11), e21981. [Google Scholar] [CrossRef]
- Warutkar, G.; Ugemuge, N.; Ansari, E.; Pusdekar, A.; Dhale, S.; Tumram, P.; Patle, S.; Moharil, S.V. Synthesis and spectroscopic analysis in GdVO4:RE3+ (RE=Sm, Eu) by solid state metathesis for optical applications. J. Mol. Struct. 2025, 1332, 141591. [Google Scholar] [CrossRef]
- Kaowphong, S.; Chumha, N.; Nimmanpipug, P.; Kittiwachana, S. Nanosized GdVO4 powders synthesized by sol–gel method using different carboxylic acids. Rare Met. 2018, 37, 561–567. [Google Scholar] [CrossRef]
- Mazierski, P.; Sowik, J.; Miodyńska, M.; Trykowski, G.; Mikołajczyk, A.; Klimczuk, T.; Lisowski, W.; Nadolna, J.; Zaleska-Medynska, A. Shape-controllable synthesis of GdVO4 photocatalysts and their tunable properties in photocatalytic hydrogen generation. Dalton Trans. 2019, 48, 1662–1671. [Google Scholar] [CrossRef]
- Xu, H.Y.; Wang, H.; Meng, Y. Q.; Yan, H. Rapid synthesis of size-controllable YVO4 nanoparticles by microwave irradiation. Solid State Commun. 2004, 130, 465–468. [Google Scholar] [CrossRef]
- Tomina, E.V.; Sladkopevtsev, B.V.; Novikova, L.A.; Boykov, N.I.; Maltsev, S.A. Microwave and ultrasonic radiation-activated synthesis and luminescent properties of nanopowder YVO4:Bi3+,Eu3+. Russ Chem Bull 2023, 72, 1113–1121. [Google Scholar] [CrossRef]
- Zhu, Z.; Han, S.; Cao, Y.; Jiang, J. Synthesis of a novel photocatalyst MVO4/g-C3N4 (M = La, Gd) with better photocatalytic activity for tetracycline hydrochloride degradation under visible-light irradiation. Crystals 2021, 11(7), 756. [Google Scholar] [CrossRef]
- Li, G.; Jiang, S.; Liu, A.; Ye, L.; Ke, J.; Liu, C.; Chen, L.; Liu, Y.; Hong, M. Proof of crystal-field-perturbation-enhanced luminescence of lanthanide-doped nanocrystals through interstitial H+ doping. Nat. Commun. 2023, 14, 5870. [Google Scholar] [CrossRef]
- Kimani, M.; Colin, M.; McMillen, D.; Kolis, J.W. Synthetic and spectroscopic studies of vanadate glaserites II: Photoluminescence studies of Ln:K3Y(VO4)2 (Ln=Eu, Er, Sm, Ho, or Tm). J. Solid State Chem. 2015, 226, 320–325. [Google Scholar] [CrossRef]
- Chen, Z.; Zhu, H.; Qian, J.; Li, Z.; Hu, X.; Guo, Y.; Fu, Y.; Zhu, H.; Nai, W.; Yang, Z.; Li, D.; Zhou, L. Rare earth ion doped luminescent materials: a review of up/downconversion luminescent mechanism, synthesis, and anti-counterfeiting application. Photonics 2023, 10, 1014. [Google Scholar] [CrossRef]
- Jovanović, D.J.; Gavrilović, T.V.; Dolić, S.D.; Marinović-Cincović, M.; Smits, K.; Dramićanin, M.D. Up-conversion luminescence of GdVO4:Nd3+/Er3+ and GdVO4:Nd3+/Ho3+ phosphors under 808 nm excitation. Opt. Mater. 2018, 82, 1–6. [Google Scholar] [CrossRef]
- Chen, Z.; Zhu, H.; Qian, J.; Li, Z.; Hu, X.; Guo, Y.; Fu, Y.; Zhu, H.; Nai, W.; Yang, Z.; Li, D.; Zhou, L. Rare earth ion doped luminescent materials: a review of up/downconversion luminescent mechanism, synthesis, and anti-counterfeiting application. Photonics 2023, 10, 1014. [Google Scholar] [CrossRef]
- Han, S.; Tao, Y.; Du, Y.; Yan, S.; Chen, Y.; Chen, D. Luminescence behavior of GdVO4:Tb nanocrystals in silica glass-ceramics. Crystals 2020, 10(5), 396. [Google Scholar] [CrossRef]
- Jovanović, D.J.; Antić, Ž.; Krsmanović, R.M.; Mitrić, M.; Ðorđević, V.; Bártová, B.; Dramićanin, M.D. Annealing effects on the microstructure and photoluminescence of Eu3+-doped GdVO4 powders. Opt. Mater. 2013, 35, 1797–1804. [Google Scholar] [CrossRef]
- González-Penguelly, B.; Pérez-Sánchez, G.G.; Medina-Velázquez, D.Y.; Martínez-Falcón, P.; Morales-Ramírez, A.J. Luminescence properties and energy transfer of Eu3+, Bi3+ co-doped LuVO4 films modified with Pluronic F-127 obtained by sol–gel. Materials 2023, 16(1), 146. [Google Scholar] [CrossRef] [PubMed]
- Chi, F.; Ji, Z.; Liu, Q.; Jiang, B.; Wang, B.; Cheng, J.; Li, B.; Liu, S.; Wei, X. Investigation of multicolor emitting Cs3GdGe3O9:Bi3+,Eu3+ phosphors via energy transfer for WLEDs. Dalton Trans. 2023, 52, 635–643. [Google Scholar] [CrossRef]
- Tu, D.; Zheng, W.; Huang, P.; Chen, X. Europium-activated luminescent nanoprobes: From fundamentals to bioapplications. Coord. Chem. Rev. 2019, 378, 104–120. [Google Scholar] [CrossRef]
- Tkachenko, A.; Yefimova, S.; Tryfonyuk, L.; Thomas, S. Nanostructured vanadates as Janus-faced redox-active nanomaterials: Biological effects and molecular mechanisms. In Biol. Trace Elem. Res.; In Press, 2026. [Google Scholar] [CrossRef]
- Kot, Y.; Klochkov, V.; Prokopiuk, V.; Sedyh, O.; Tryfonyuk, L.; Grygorova, G.; Karpenko, N.M.; Tomchuk, O.; Kot, K.; Onishchenko, A.; Yefimova, S.; Tkachenko, A. GdVO4:Eu3+ and LaVO4:Eu3+ nanoparticles exacerbate oxidative stress in L929 cells: potential implications for cancer therapy. Int. J. Mol. Sci. 2024, 25(21), 11687. [Google Scholar] [CrossRef]
- Hu, H.; Li, D.; Liu, S.; Wang, M.; Moats, R.; Conti, P.S.; Li, Z. Integrin α₂β₁ targeted GdVO4:Eu ultrathin nanosheet for multimodal PET/MR imaging. Biomaterials 2014, 35, 8649–8658. [Google Scholar] [CrossRef]
- Toro-González, M.; Dame, A.N.; Mirzadeh, S.; Rojas, J.V. Gadolinium vanadate nanocrystals as carriers of α-emitters (225Ac, 227Th) and contrast agents. J. Appl. Phys. 2019, 125, 214901. [Google Scholar] [CrossRef]
- Toro-González, M.; Copping, R.; Mirzadeh, S.; Rojas, J.V. Multifunctional GdVO4:Eu core–shell nanoparticles containing 225Ac for targeted alpha therapy and molecular imaging. J. Mater. Chem. B 2018, 6, 7985–7997. [Google Scholar] [CrossRef]
- Kim, Hyunsub; Jeong, Heejin; Byeon, Song-Ho. Selective filter effect induced by Cu2+ adsorption on the fluorescence of a GdVO4:Eu nanoprobe. ACS Appl. Mater. Interfaces 2016, 8, 15497–15505. [Google Scholar] [CrossRef]
- Liang, Y.; Noh, H.M.; Park, S.H.; Choi, B.C.; Jeong, J.H. Colloidal GdVO4:Eu3+@SiO2 nanocrystals for highly selective and sensitive detection of Cu2+ ions. [CrossRef]
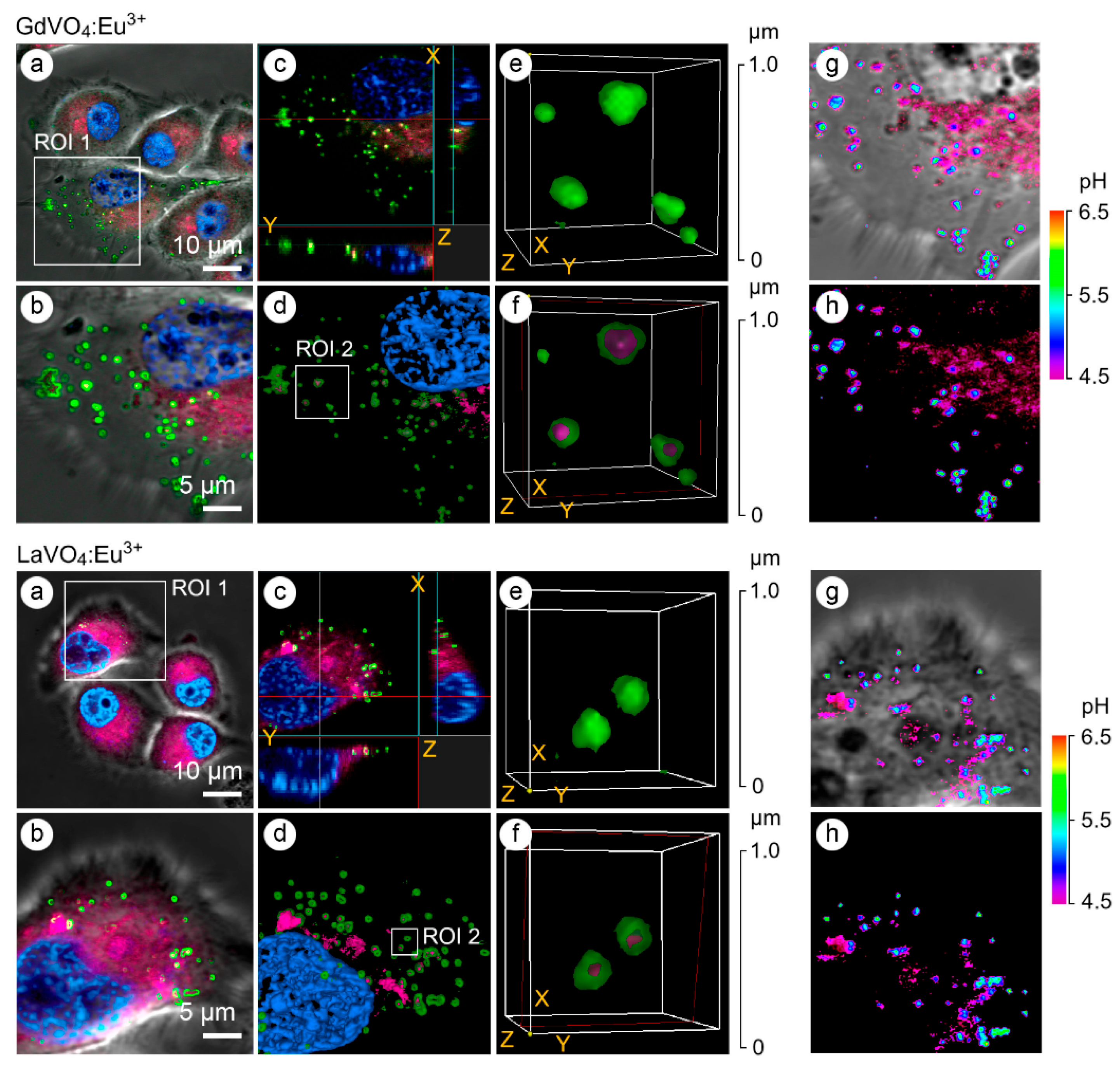
- Periša, J.; Antić, Ž.; Ma, C.-G.; Papan, J.; Jovanović, D.; Dramićanin, M.D. Pesticide-induced photoluminescence quenching of ultra-small Eu3+-activated phosphate and vanadate nanoparticles J. Mater. Sci. Technol. 2020, 38, 197–204. [Google Scholar] [CrossRef]
- Muhr, V.; Buchner, M.; Hirsch, T.; Jovanović, D.J.; Dolić, S.D.; Dramićanin, M.D.; Wolfbeis, O.S. Europium-doped GdVO4 nanocrystals as a luminescent probe for hydrogen peroxide and for enzymatic sensing of glucose. Sens. Actuator B Chem. 2017, 241, 349–356. [Google Scholar] [CrossRef]
- Maksimchuk, P.O.; Hubenko, K.O.; Knupfer, M.; Seminko, V.V.; Klochkov, V.K.; Sorokin, O.V.; Demchenko, L.D.; Yefimova, S.L. Microscopic mechanisms of luminescence quenching in Eu3+-doped GdVO4 nanoparticles under hydrogen peroxide decomposition. J. Mol. Liq. 2024, 400, 124510. [Google Scholar] [CrossRef]
- Proiou, E.; Pinakidou, F.; Paloura, E.C.; Pétri, N.; Gacoin, T.; Laplace-Builhé, C.; Schuck, G.; Alexandrou, A.; Katsikini, M. Spatial distribution and stability of Gd0.6Eu0.4VO4 nanoparticles injected in mouse ear pinnae. TrAC Trends Anal. Chem. 2025, 182, 118049. [Google Scholar] [CrossRef]
- Li, N.; Yang, B.; Guo, L.; Li, T. Na3Gd(VO4)2:Yb3+/(Er3+, Tm3+, Ho3+) and Na3Gd(VO4)2: Yb3+/Er3+/(La3+, Lu3+, Y3+): Upconversion luminescence and temperature sensitivity under single- and multi-wavelength excitation. Bull. Mater. Sci. 2023, 46, 222. [Google Scholar] [CrossRef]
- Min, B.H.; Jung, K.Y. Enhanced upconversion luminescence of GdVO4:Er3+/Yb3+ prepared by spray pyrolysis using organic additives. RSC Adv. 2019, 9, 20002–20008. [Google Scholar] [CrossRef]
- Prasad, M.; Rai, V.K. Thermally stable upconverting Na3Zr2(SiO4)2PO4:Er3+/Yb3+ phosphors for displays and optical thermometry. J. Alloys Compd. 2022, 911, 164968. [Google Scholar] [CrossRef]
- Gavrilović, T.V.; Jovanović, D.J.; Lojpur, V.; Dramićanin, M.D. Multifunctional Eu3+ and Er3+/Yb3+-doped GdVO4 nanoparticles synthesized by reverse micelle method. Sci. Rep. 2014, 4, 4209. [Google Scholar] [CrossRef]
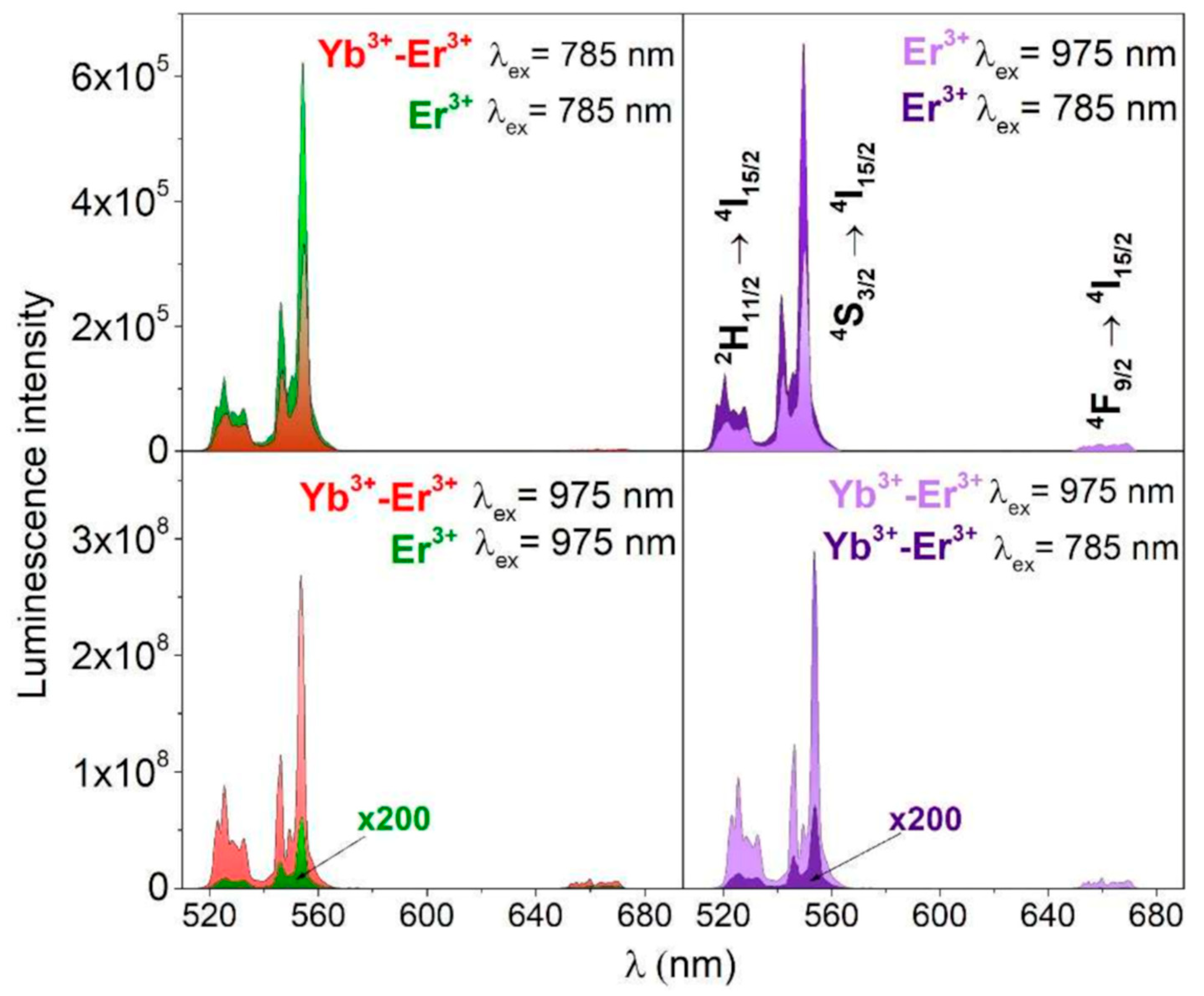
- Stopikowska, N.; Runowski, M.; Woźny, P.; Lis, S.; Du, P. Generation of pure green up-conversion luminescence in Er3+ doped and Yb3+-Er3+ co-doped YVO4 nanomaterials under 785 and 975 nm excitation. Nanomaterials 2022, 12(5), 799. [Google Scholar] [CrossRef]
- Zharkov, D.K.; Leontyev, A.V.; Shmelev, A.G.; Nurtdinova, L.A.; Chuklanov, A.P.; Nurgazizov, N.I.; Nikiforov, V. G. Upconversion luminescence response of a single YVO4:Yb, Er particle. Micromachines 2023, 14(5), 1075. [Google Scholar] [CrossRef]
- Maurya, A.; Yadav, R.S.; Yadav, R.V.; Rai, S.B.; Bahadur, A. Enhanced green upconversion photoluminescence from Ho3+/Yb3+ co-doped CaZrO3 phosphor via Mg2+ doping. RSC Adv. 2016, 6, 113469–113477. [Google Scholar] [CrossRef]
- Yadav, M.R.S.; Bahadur, A.; Rai, S.B. Near-infrared light excited highly pure green upconversion photoluminescence and intrinsic optical bistability sensing in a Ho3+/Yb3+ co-doped ZnGa2O4 phosphor through Li+ doping. J. Phys. Chem. C 2020, 124, 10117–10128. [Google Scholar] [CrossRef]
- Gavrilović, T.V.; Nikolić, M.G.; Jovanović, D.J.; Dramićanin, M.D. Up-conversion luminescence of Tm3+ sensitized by Yb3+ ions in GdVO4. Phys. Scr. T 2013, 157, 014055. [Google Scholar] [CrossRef]
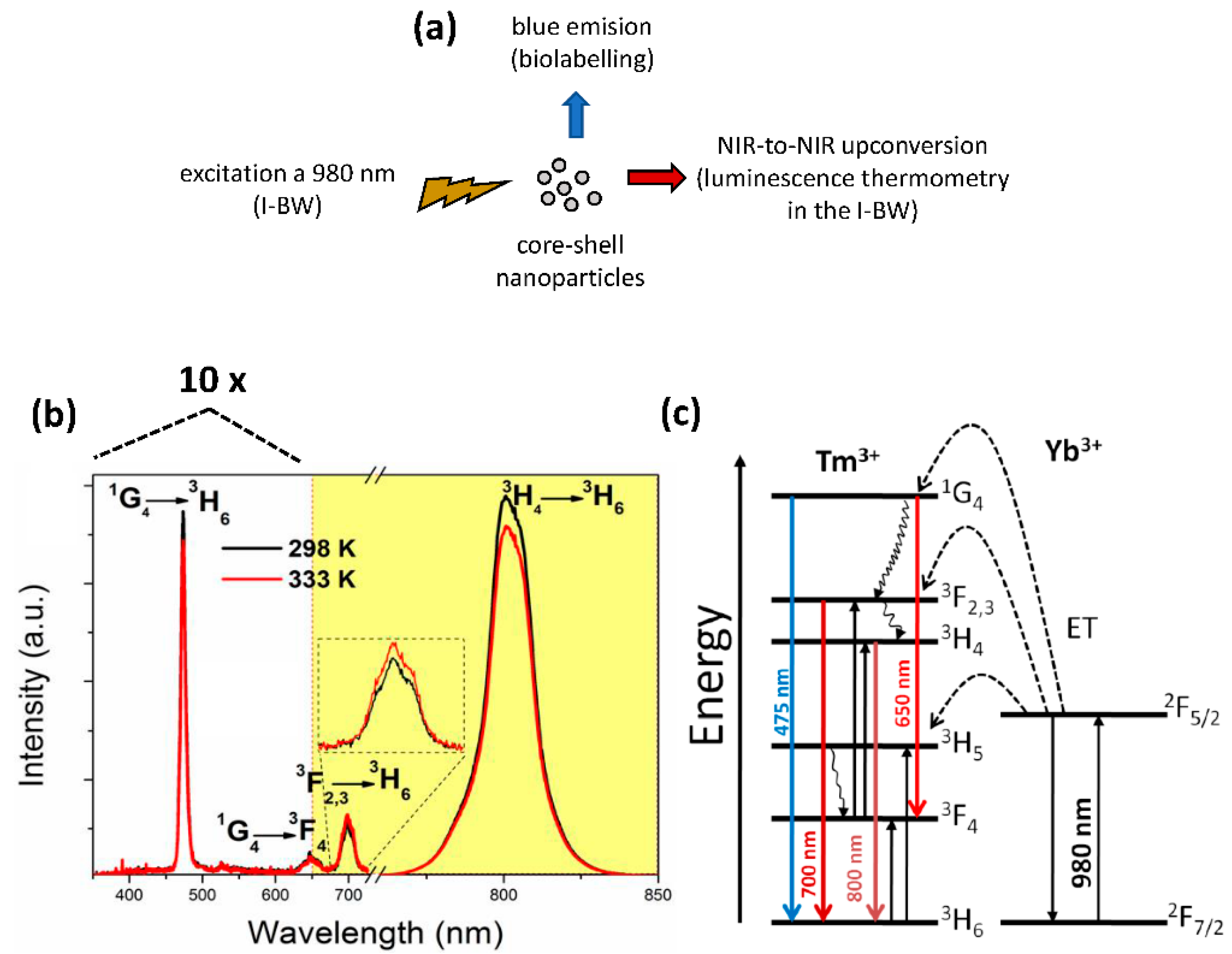
- Savchuk, O.; Marti, J.J.C.; Cascales, C.; Haro-Gonzalez, P.; Sanz-Rodríguez, F.; Aguilo, M.; Diaz, F. Bifunctional Tm3+,Yb3+:GdVO4@SiO2 core-shell nanoparticles in HeLa cells: Upconversion luminescence nanothermometry in the first biological window and biolabelling in the visible. Nanomaterials 2020, 10(5), 993. [Google Scholar] [CrossRef]
- Liu, B.; Li, C.; Yang, P.; Hou, Z.; Lin, J. 808-nm-light-excited lanthanide-doped nanoparticles: rational design, luminescence control and theranostic applications. Adv. Mater. 2017, 29, 1605434. [Google Scholar] [CrossRef]
- Zhai, T.; Zhang, Y.; Guan, D.; Yang, L.; Zhang, W.; Zhang, Y.; Liu, Q. Investigation of upconversion luminescence attenuation in aqueous solutions under 980 nm and 808 nm irradiation. J. Anal. Test. 2023, 7, 377–383. [Google Scholar] [CrossRef]
- Xu, C.; Wu, R.; Xiao, Z.; Xu, L. Thermal analysis of phonons assisted bright yellow upconversion emission and one photon near-infrared upconversion emission in Nd3+ doped GdVO4 single crystals. Ceram. Int. 2024, 50, 42619–42624. [Google Scholar] [CrossRef]
- Kolesnikov, I.E.; Kalinichev, A.A.; Kurochkin, M.A.; Golyeva, E.V.; Kolesnikov, E.Y.; Kurochkin, A.V.; Lähderanta, E.; Mikhailov, M.D. YVO4:Nd3+ nanophosphors as NIR-to-NIR thermal sensors in wide temperature range. Sci. Rep. 2017, 7, 18002. [Google Scholar] [CrossRef]
- Rosal, B. del.; Pérez-Delgado, A.; Carrasco, E.; Jovanović, D.J.; Dramićanin, M.D.; Dražić; de la Fuente, G.Á.J.; Sanz-Rodriguez, F.; Jaque, D. Neodymium-based stoichiometric ultrasmall nanoparticles for multifunctional deep-tissue photothermal therapy. Adv. Optical Mater. 2016, 4, 782–789. [Google Scholar] [CrossRef]
- Hong, G.; Antaris, A.; Dai, H. Near-infrared fluorophores for biomedical imaging. Nat. Biomed. Eng. 2017, 1, 0010. [Google Scholar] [CrossRef]
- Ansari, A.A.; Thakur, V.K.; Chen, G. Functionalized upconversion nanoparticles: New strategy towards FRET-based luminescence bio-sensing. Coord. Chem. Rev. 2021, 436, 213821. [Google Scholar] [CrossRef]
- Zajdel, K.; Janowska, J.; Frontczak-Baniewicz, M.; Sypecka, J.; Sikora, B. Upconverting nanoparticles as a new bio-imaging strategy—Investigating intracellular trafficking of endogenous processes in neural tissue. Int. J. Mol. Sci. 2023, 24(2), 1122. [Google Scholar] [CrossRef]
- Wang, W.; Shang, S.; Wang, Y.; Xu, B. Utilization of nanomaterials in MRI contrast agents and their role in therapy guided by imaging. Front. Bioeng. Biotechnol. 2024, 12, 1484577. [Google Scholar] [CrossRef]
- Zheng, W.T.; Chen, H.L.; Hou, X.T.; Abakirova, A.; Han, M.-Y.; Feng, Y.-Q.; Huang, W.-T.; Mo, X.-C.; Zhu, H.-C.; Huang, S.-K.; Lv, H.-D.; Yang, T.; Huang, S.; Shi, Z.-M. Research trends and hotspots of upconversion nanoparticles for drug delivery and biomedical targeting in photodynamic therapy. Discover. Nano 2025, 20, 235. [Google Scholar] [CrossRef]
- Thao, C.T.B.; Huy, B.T.; Sharipov, M.; Kim, J-I.; Dao, V-D.; Moon, J-Y.; Lee, Y-I. Yb3 +, Er3+, Eu3+-codoped YVO4 material for bioimaging with dual mode excitation. Mater. Sci. Engin. C 2017, 75, 990–997. [Google Scholar] [CrossRef]
- Lv, Z.; Jin, L.; Cao, Y.; Zhang, H.; Xue, D.; Yin, N.; Zhang, T.; Wang, Y.; Liu, J.; Liu, X.; Zhang, H. A nanotheranostic agent based on Nd3+-doped YVO4 with blood-brain-barrier permeability for NIR-II fluorescence imaging/magnetic resonance imaging and boosted sonodynamic therapy of orthotopic glioma. Light Sci. Appl. 2022, 29, 116. [Google Scholar] [CrossRef]
- Carbonati, T.; Ciontia, C.; Cosaert, E.; Nimmegeers, B.; Meronia, D.; Poelman, D. NIR emitting GdVO4:Nd nanoparticles for bioimaging: The role of the synthetic pathway. J. Alloys Compd. 2021, 862, 158413. [Google Scholar] [CrossRef]
- Yin, W.; Zhou, L.; Gu, Z.; Tian, G.; Jin, S.; Yan, L.; Liu, X.; Xing, G.; Ren, W.; Liu, F.; Pan, Z.; Zhao, Y. Lanthanide-doped GdVO4 upconversion nanophosphors with tunable emissions and their applications for biomedical imaging. J. Mater. Chem. 2012, 22, 6974–6981. [Google Scholar] [CrossRef]
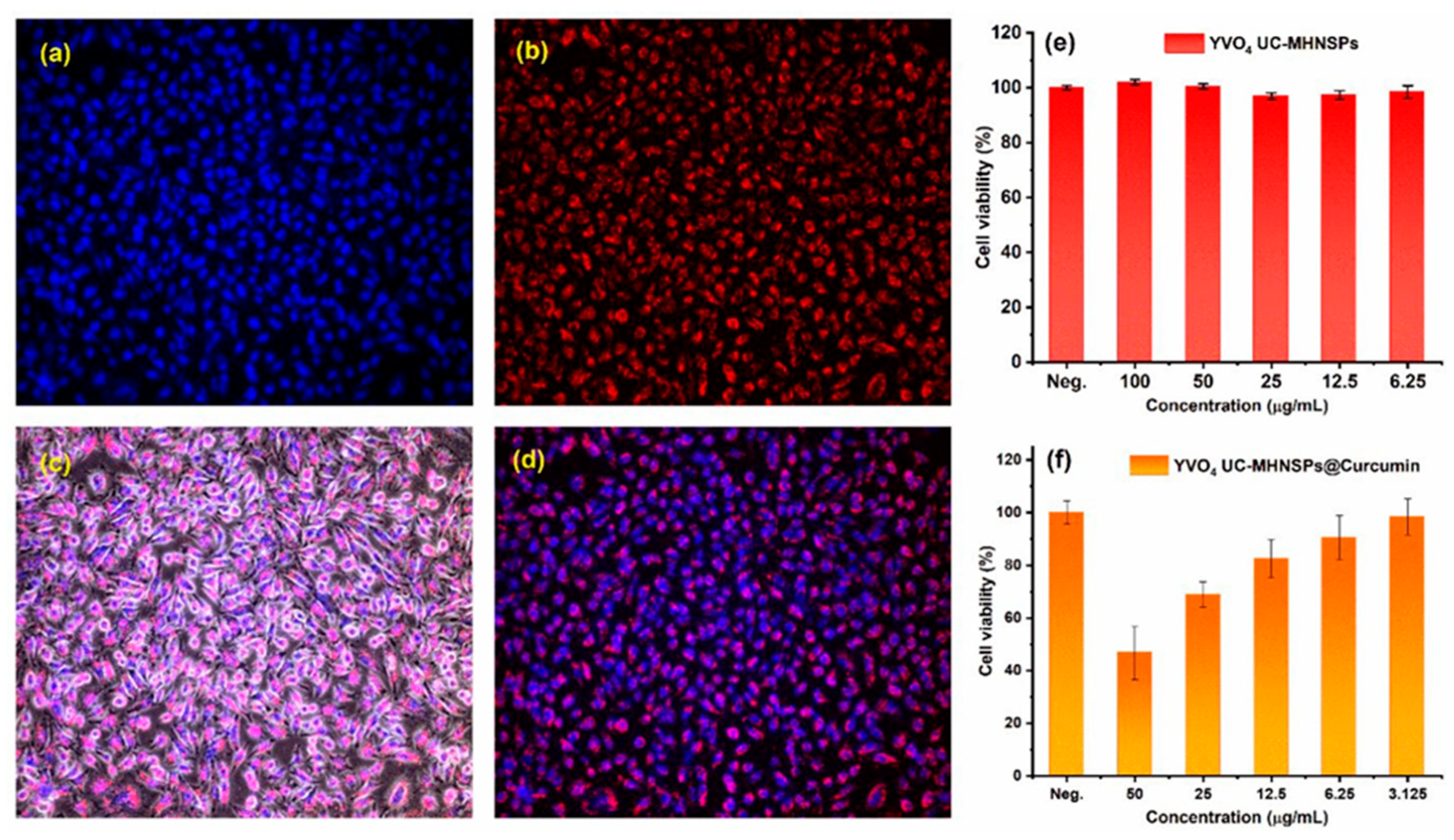
- Pavitra, E.; Lee, H.; Hwang, S.K.; Park, J.Y.; Han, Y-K.; Raju, G.S.R.; Huh, Y.S. Evolution of highly biocompatible and thermally stable YVO4:Er3+/Yb3+ upconversion mesoporous hollow nanospheriods as drug carriers for therapeutic applications. Nanomaterials 2022, 12, 2520. [Google Scholar] [CrossRef]
- Jaque, D.; Vetrone, F. Luminescence nanothermometry. Nanoscale 2012, 4, 4301–4326. [Google Scholar] [CrossRef]
- Mahata, M.K.; Koppe, T.; Kumar, K.; Hofsäss, H.; Vetter, U. Demonstration of temperature dependent energy migration in dual-mode YVO4:Ho3+/Yb3+ nanocrystals for low temperature thermometry. Sci Rep. 2016, 6, 36342. [Google Scholar] [CrossRef]
- Getz, M.N.; Nilsen, O.; Hansen, P.-A. Sensors for optical thermometry based on luminescence from layered YVO4:Ln3+ (Ln = Nd, Sm, Eu, Dy, Ho, Er, Tm, Yb) thin films made by atomic layer deposition. Sci Rep. 2019, 9, 10247. [Google Scholar] [CrossRef] [PubMed]
- Debasu, M.L.; Ananias, D.; Pastoriza-Santos, I.; Liz-Marzán, L.M.; Rocha, J.; Carlos, L.D. All-in-one optical heater-thermometer nanoplatform operative from 300 to 2000 K based on Er3+ emission and blackbody radiation. Adv. Mater. 2013, 25, 4868–4874. [Google Scholar] [CrossRef] [PubMed]
- Fischer, L.H.; Harms, G.S.; Wolfbeis, O.S. Upconverting nanoparticles for nanoscale thermometry. Angew. Chem. Int. Ed. Engl. 2011, 50, 4546–4551. [Google Scholar] [CrossRef]
- Alrammouz, R.; Lazerges, M.; Pironon, J.; Bin Taher, I.; Randi, A.; Halfaya, Y.; Gautier, S. V2O5 gas sensors: A review. Sens. Actuators A: Phys. 2021, 332, 113179. [Google Scholar] [CrossRef]
- Chackrabarti, S.; Zargar, R.A. Review: vanadium pentoxide thin films-advanced doping strategies and interface engineering for next-generation applications. J Mater Sci 2026, 61, 4894–4938. [Google Scholar] [CrossRef]
- Abdelrazek, MM; Abd-Elrazek, ME; El Refaay, DE; El-Desoky, M.M. Enhanced optical properties of V₂O₅ thin films through rare earth co-doping. Physica B 2025, 699, 416881. [Google Scholar] [CrossRef]
- Kishan Chand, T.R.; Kalpana, H.M.; Satish, T.N. Effect of thickness and annealing on electrical characteristics of cerium oxide-doped vanadium oxide (CeO2:V2O5) thin film for sensor application. J Mater Sci: Mater Electron 2024, 35, 616. [Google Scholar] [CrossRef]
- Marinković, D.; Righini, G.C.; Ferrari, M. Synthesis, optical, and photocatalytic properties of the BiVO4 semiconductor nanoparticles with tetragonal zircon-type structure. Photonics 2025, 12, 438. [Google Scholar] [CrossRef]
- Poovethamkandiyil, A.S.; Palliyalil, K.K.; Sujith, K.; Paravannoor, A.; Ganguly, P.; Vijayan, B. K. Emerging bismuth vanadate-based biosensor platforms for diagnostic, imaging, therapeutic and antibacterial applications. J. Mater. Chem. B 2025, 13, 13928–13953. [Google Scholar] [CrossRef] [PubMed]
- Sagadevan, S.; T. Soga, T. Morphology-dependent gas sensing properties of bismuth vanadate nano- materials: A promising approach for environmental monitoring. Inorg. Chem. Commun. 2024, 160, 111980. [Google Scholar] [CrossRef]
- Dwivedi, A.; Roy, A.; Rai, S. B. Photoluminescence behavior of rare earth doped self-activated phosphors (ie niobate and vanadate) and their applications. RSC Adv. 2023, 13, 16260–16271. [Google Scholar] [CrossRef]
- Kiran, R.; Kamath, N.; Sayyed, M. I.; Almuqrin, A. H.; Kamath, S. D. A review of recent developments in rare earth-doped nanophosphors for emerging technological applications. RSC Adv. 2025, 15, 20040–20060. [Google Scholar] [CrossRef]
- Ugemuge, N. S.; Bhagat, R. M.; Warutkar, G.; Dhale, S.; Pimpalkar, A.; Mistry, A.; Selokar, A.W.; Bishnoi, S.; Golhar, A.; Dhoble, S. J. Recent advances in vanadate phosphor materials: synthesis and optoelectronic applications: A comprehensive review. J. Electron. Mater. 2025, 54, 8185–8217. [Google Scholar] [CrossRef]
- Liu, J.; Huang, Z.; Zhang, Z.; Zhu, D.; Yue, C.; Li, K. Near-ultraviolet activated dual-functional Na3Sc2(VO4)3 phosphors with Eu3+/Sm3+ dopant systems for multimodal anti-counterfeiting and ratiometric optical thermal sensing. J. Alloys Compd. 2025, 1031, 181107. [Google Scholar] [CrossRef]
- Ye, Y.; Liang, J.; Zhang, W.; Xie, W.; Feng, Z.; Hu, Z.; Xiong, G.; Liu, Z. Synthesis and luminescence properties of Bi3+ and Re3+ (Re= Eu, Dy, Tm) co-doped GdVO4 phosphors for latent fingerprint detection. J. Phys.: Conf. Ser. 2024, 012002. [Google Scholar] [CrossRef]
- Devi, P.; Dalal, H.; Sehrawat, N.; Solanki, D.; Malik, R. K. Phase identification and photophysical characteristics of vanadate-based nanophosphors for lighting and latent fingerprinting applications. Inorg. Chem. Commun. 2024, 168, 112885. [Google Scholar] [CrossRef]
- Sehrawat, N.; Devi, P.; Solanki, D.; Kumari, N.; Punia, R.; Malik, R. K.; Garg, S. Crystal structure and photophysical analysis of red emitting Eu3+ doped vanadate based nanomaterials for innovative optoelectronic devices and latent fingerprinting applications. Inorg. Chem. Commun. 2026, 186, 116318. [Google Scholar] [CrossRef]
- Xie, X.; Ge, W.; Tian, Y.; Zhang, Q.; Yang, M.; Wu, C.; He, P.; Yin, H. La3Sc2Ga3O12:Cr3+, Nd3+ near-infrared phosphor for nondestructive detection and luminescence thermometry. Ceram. Int. 2024, 50(22), 46098–46106. [Google Scholar] [CrossRef]
- Mahata, M. K.; Koppe, T.; Kumar, K.; Hofsäss, H.; Rai, V. K.; Schneider, J. Demonstration of temperature dependent energy migration in dual-mode YVO₄:Ho³⁺/Yb³⁺ nanocrystals for low temperature thermometry. Sci. Rep. 2016, 6, 36342. [Google Scholar] [CrossRef]
- Paz-Buclatin, F.; Rivera-Lopez, F.; Gonzalez, O.; Martin, I.R.; Martin, L.L.; Jovanovic, D.J. GdVO4:Er3+/Yb3+ nanocrystalline powder as fluorescence temperature sensor. Application to monitor the temperature of an electrical component. Sens. Actuators A: Phys. 2019, 299, 111628. [Google Scholar] [CrossRef]
- Perrella, R. V.; de Sousa Filho, P. C. High-sensitivity dual UV/NIR-excited luminescence thermometry by rare earth vanadate nanoparticles . Dalton Trans. 2020, 49, 911–921. [Google Scholar] [CrossRef]
- Ma, Y.; Zhou, X.; Wu, J.; Dong, Z.; Cui, L.; Wang, Y.; Meijerink, A. Luminescence thermometry via multiparameter sensing in YV1–xPxO4:Eu3+, Er3+. J. Am. Chem. Soc. 2025, 147, 12925–12936. [Google Scholar] [CrossRef]
- Vieira Perrella, R.; Derroso, G.; de Sousa Filho, P.C. Improper background treatment underestimates thermometric performance of rare earth vanadate and phosphovanadate nanocrystals. ACS Omega 2024, 9, 34974–34980. [Google Scholar] [CrossRef] [PubMed]
- Tripathy, A.; Kumari, K.H.; Pati, S.; Veldurthi, N.K. Visible-light driven multifunctional Ca2V2O7 photocatalyst for sustainable hydrogen generation and degradation of organic pollutants. Photochem Photobiol Sci 2026. [Google Scholar] [CrossRef] [PubMed]
- Sriram, B.; Baby, J. N.; Hsu, Y.-F.; Wang, S.-F.; George, M. Scheelite-type rare earth vanadates TVO₄ (T = Ho, Y, Dy) electrocatalysts: Investigation and comparison of T site variations towards bifunctional electrochemical sensing application. Chem. Engin. J. 2023, 451, 138694. [Google Scholar] [CrossRef]
- Bharathi, P.; Wang, S.F. Rare earth orthovanadate (REM-VO4; REM = Pr, Gd, and Sm)-based sensors for selective and simultaneous detection of furazolidone and metronidazole. Nanoscale 2025, 17, 5907–5924. [Google Scholar] [CrossRef]
- Bhagat, N.; Niharika; Devi, S.; Singhaal, R.; Sen, C.; Sheik, H.N. Hydrothermal synthesis and dual functional performance of NaDy(MoO₄)₂:Tb3⁺, NaDy(WO₄)₂:Tb3⁺, and Na₃Dy(VO₄)₂:Tb3⁺ nanomaterials for nitrofurantoin sensing and photocatalytic degradation. J. Mater. Sci: Mater. Electron. 2025, 36, 1736. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



