Submitted:
12 March 2026
Posted:
13 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemical
2.2. Method
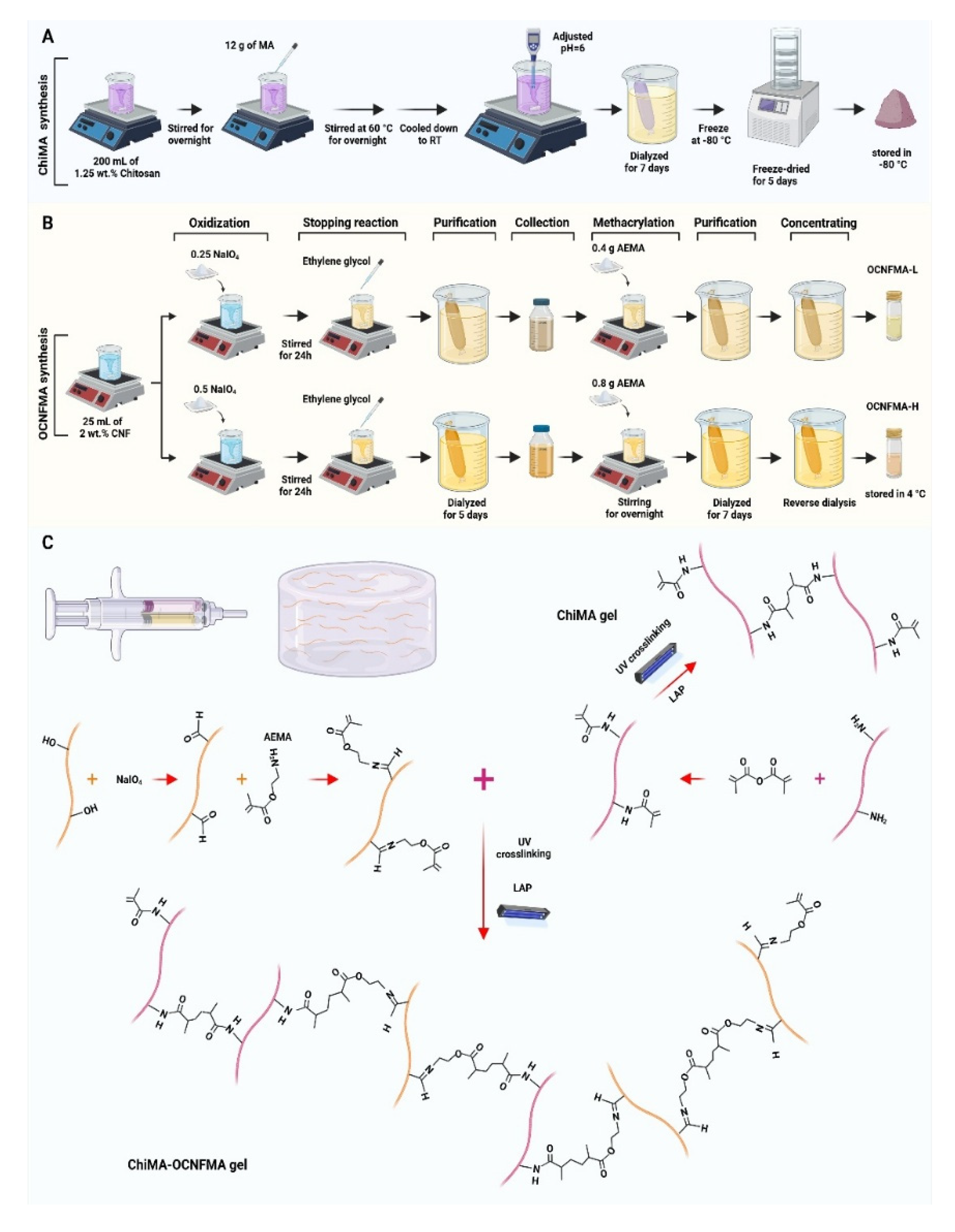
2.2.1. Cellulose Nanofiber (CNF) Functionalization
2.2.2. Chitosan Methacrylation
2.2.3. ChiMA-OCNFMA Composite Hydrogel Prepration
2.3. Characterization
2.3.1. Scanning Electron Microscopy (SEM)
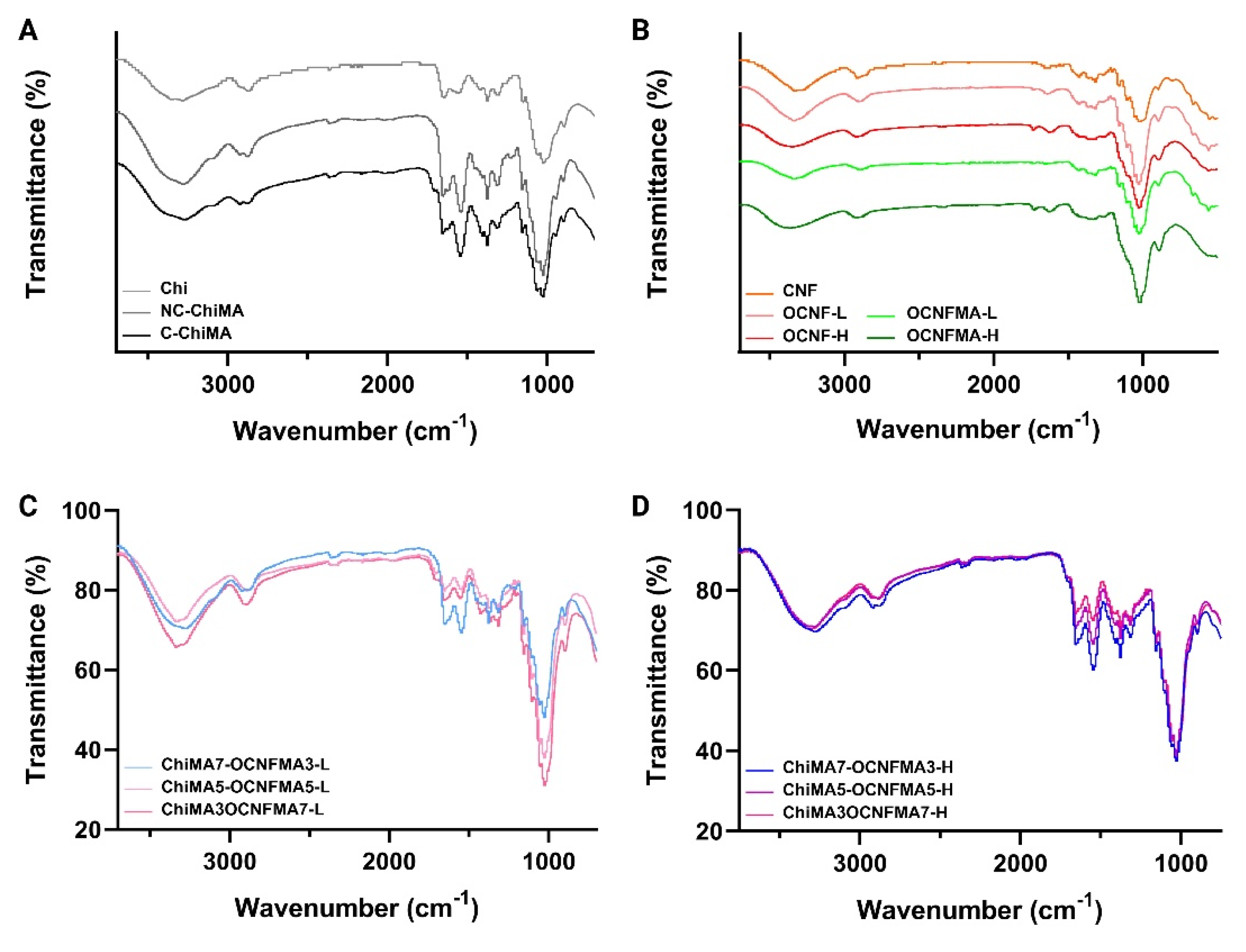
2.3.2. Fourier Transform Infrared (FTIR) Spectroscopy
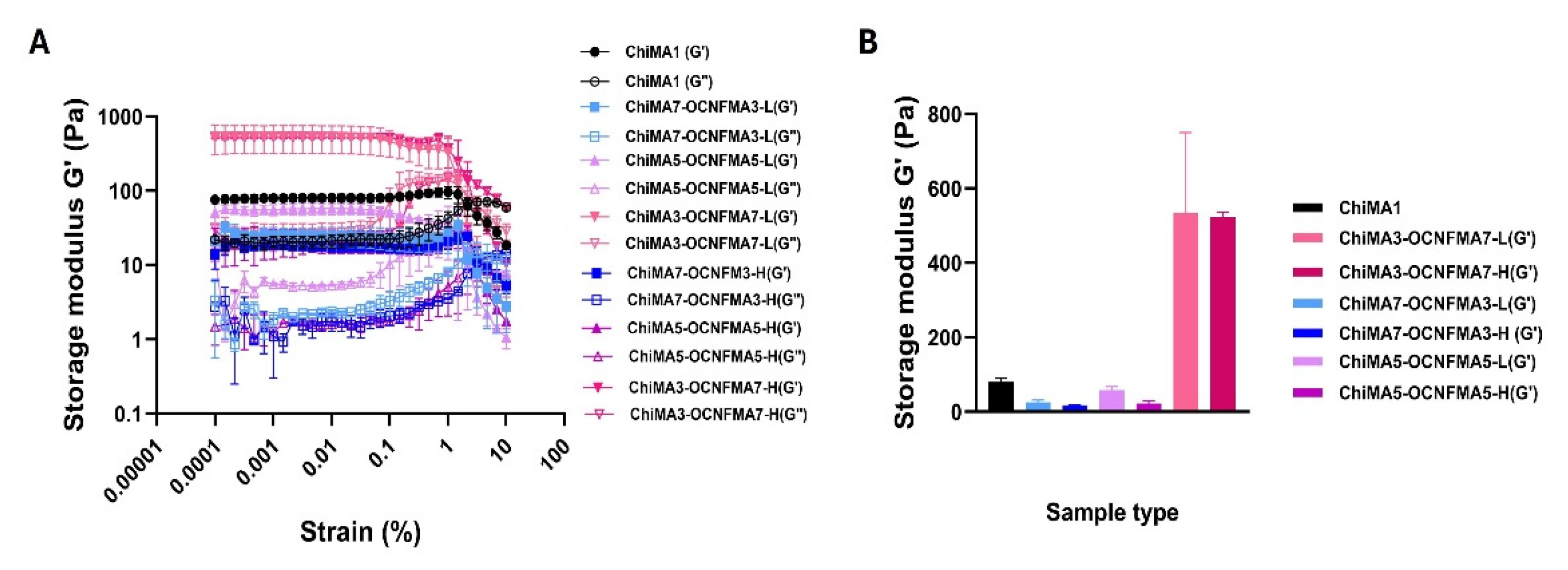
2.3.4. Rheology
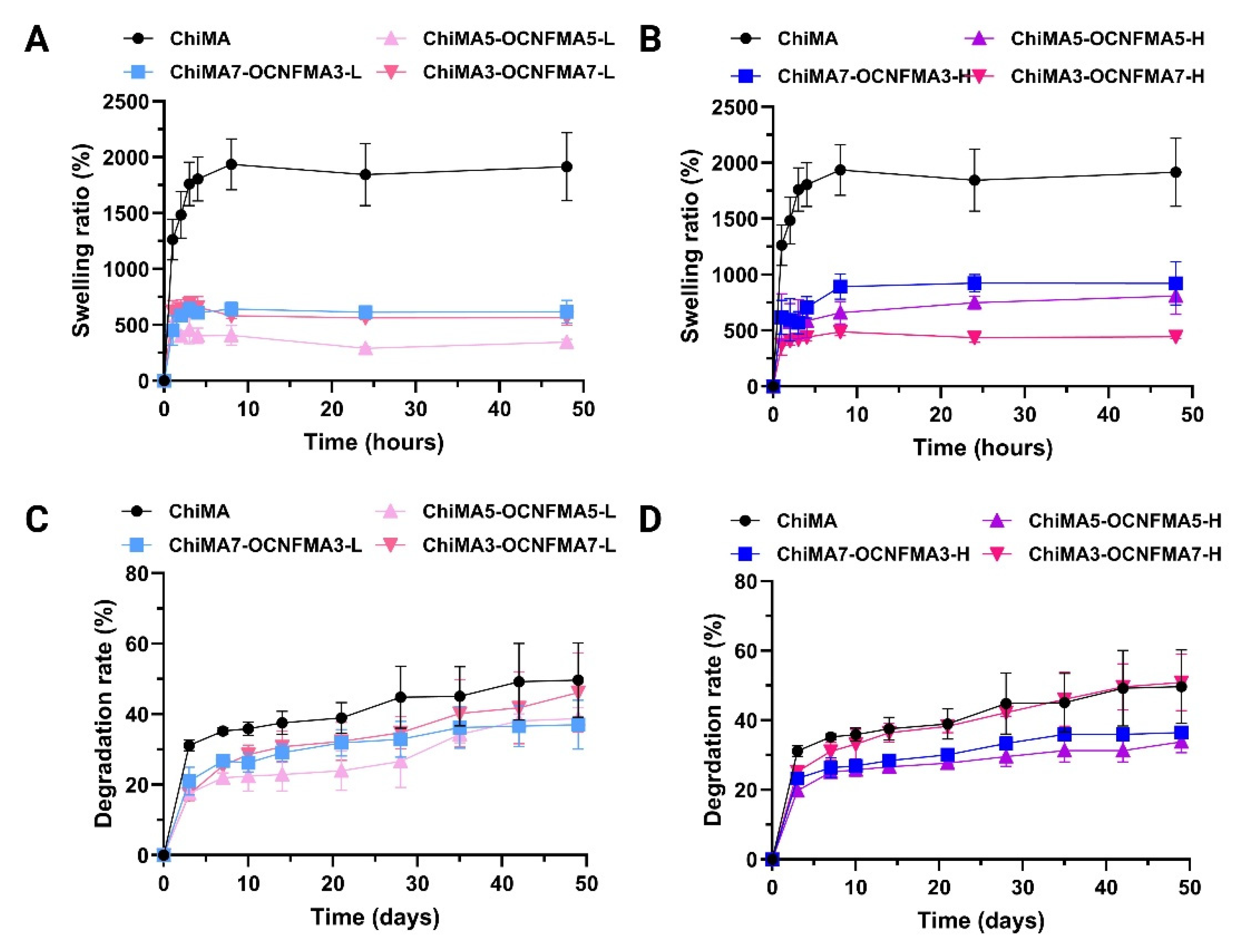
2.3.5. Swelling Behavior and Weight Loss
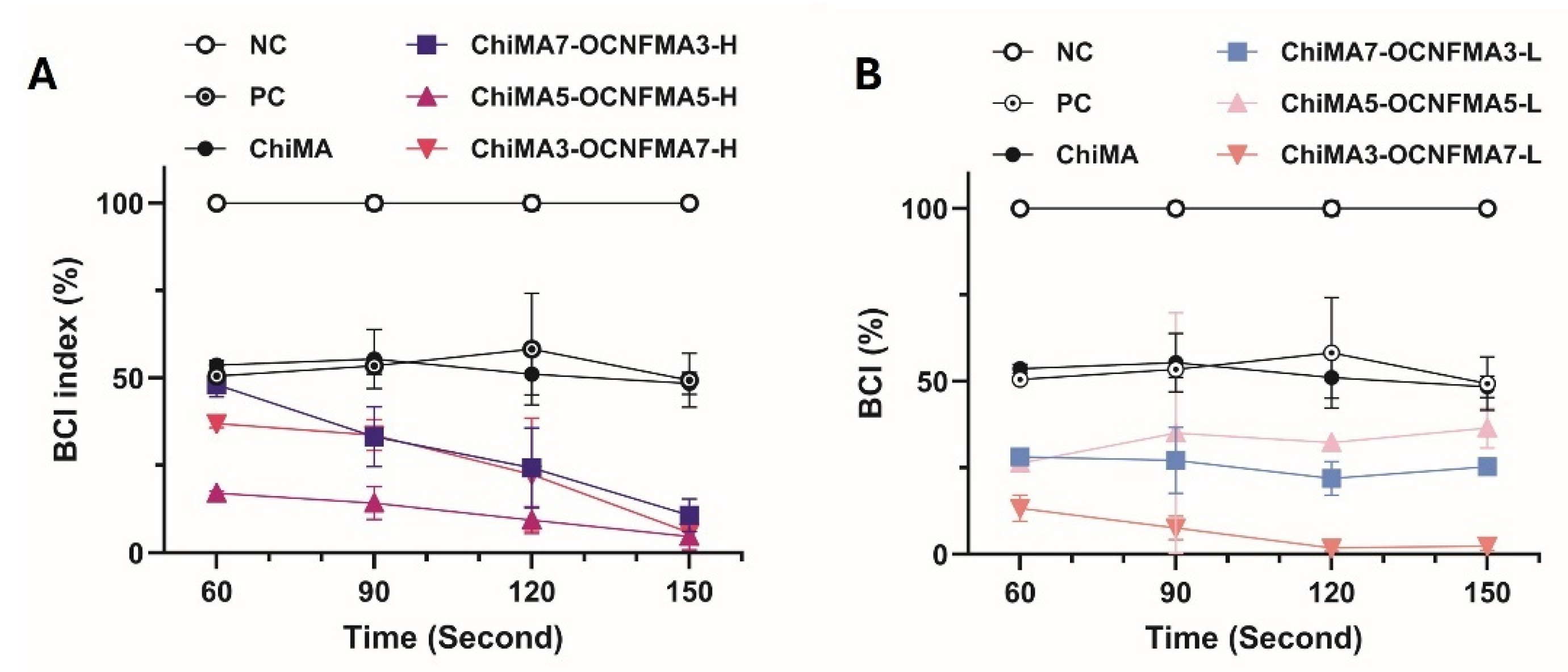
2.4. Blood Clotting Index (BCI) and Red Blood Cells (RBC) Attachment on Scaffolds
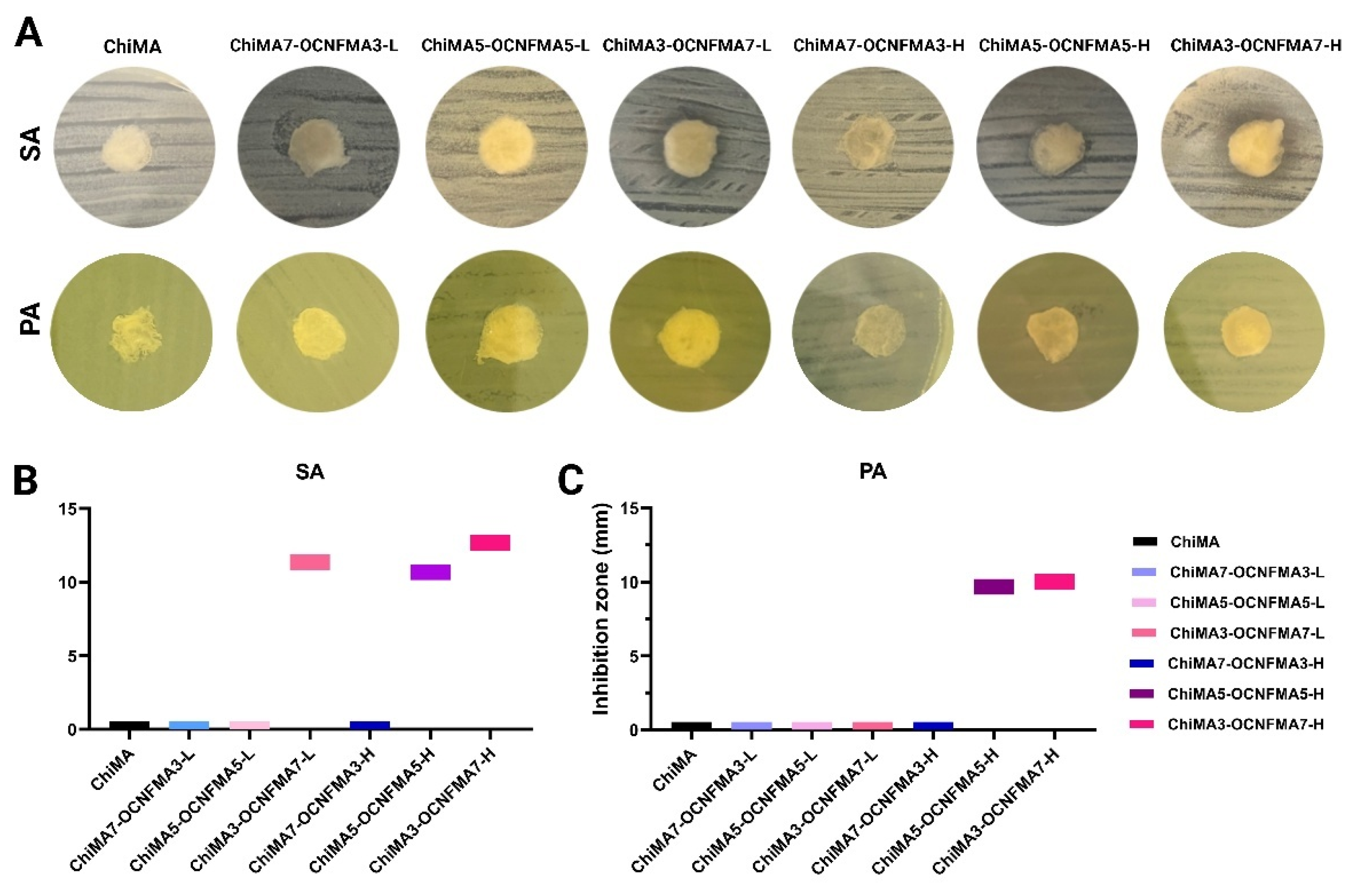
2.5. Antibacterial Properties
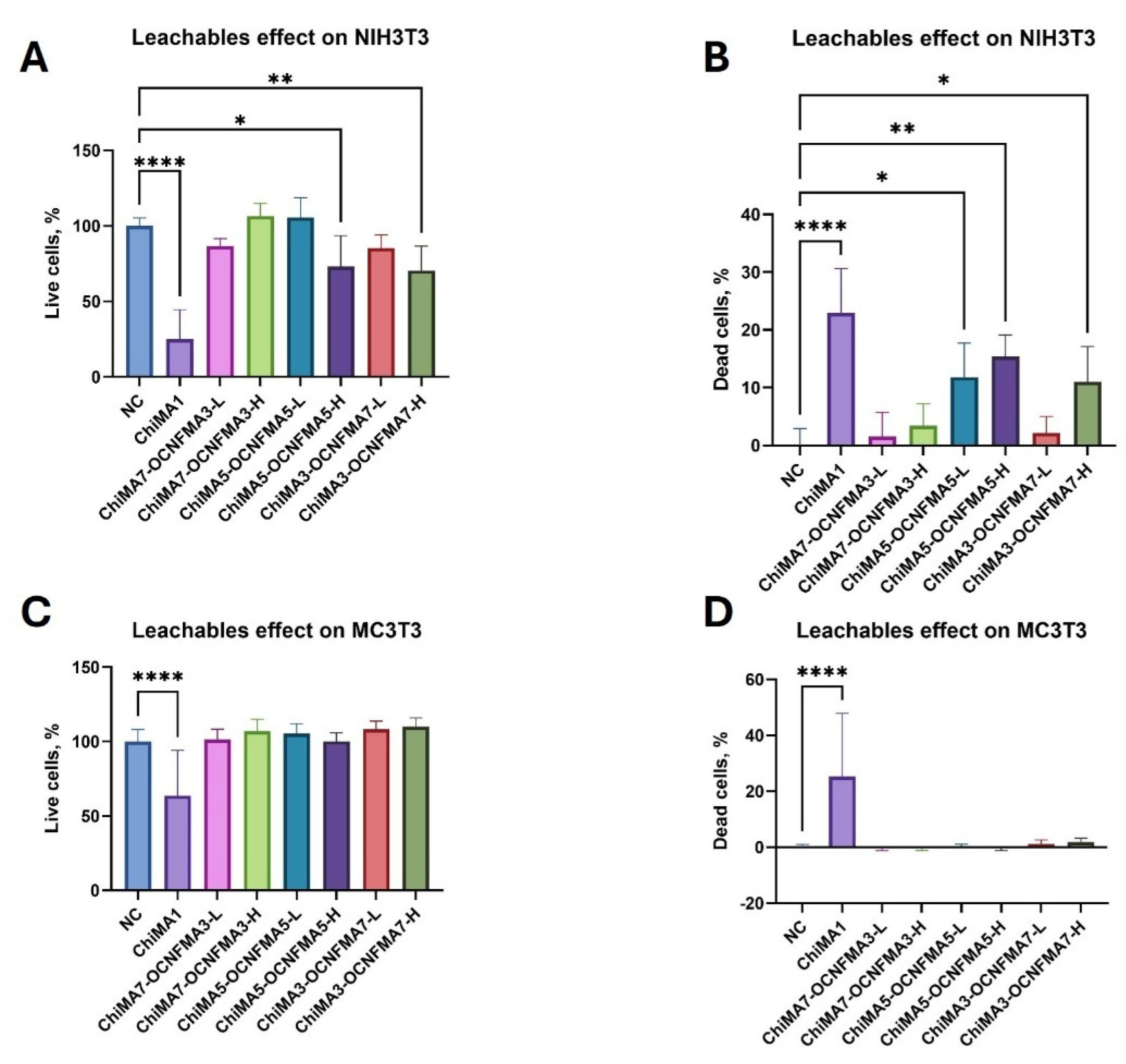
2.6. Cell Cultivation and Cytotoxicity Measurements by Lived Dead Assay for Leachable
Dual-Syringe Design and 3D Design
3. Results
4. Conclusions
Author Contributions
Funding
References
- Amirian, J.; Zeng, Y.; Shekh, M.I.; Sharma, G.; Stadler, F.J.; Song, J.; Du, B.; Zhu, Y. In-situ crosslinked hydrogel based on amidated pectin/oxidized chitosan as potential wound dressing for skin repairing. Carbohydrate Polymers 2021, 251, 117005. [Google Scholar] [CrossRef] [PubMed]
- Shefa, A.A.; Amirian, J.; Kang, H.J.; Bae, S.H.; Jung, H.-I.; Choi, H.-j.; Lee, S.Y.; Lee, B.-T. In vitro and in vivo evaluation of effectiveness of a novel TEMPO-oxidized cellulose nanofiber-silk fibroin scaffold in wound healing. Carbohydrate Polymers 2017, 177, 284–296. [Google Scholar] [CrossRef] [PubMed]
- Shekh, M.I.; Amirian, J.; Stadler, F.J.; Du, B.; Zhu, Y. Oxidized chitosan modified electrospun scaffolds for controllable release of acyclovir. International Journal of Biological Macromolecules 2020, 151, 787–796. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, K.; Dezvare, Y.; Kamyab, S.; Amirian, J.; Brangule, A.; Bandere, D. Development of Composite Sponge Scaffolds Based on Carrageenan (CRG) and Cerium Oxide Nanoparticles (CeO2 NPs) for Hemostatic Applications. Biomimetics 2023. [Google Scholar] [CrossRef]
- Amirian, J.; Wychowaniec, J.K.; Amel Zendehdel, E.; Sharma, G.; Brangule, A.; Bandere, D. Versatile Potential of Photo-Cross-Linkable Silk Fibroin: Roadmap from Chemical Processing Toward Regenerative Medicine and Biofabrication Applications. Biomacromolecules 2023, 24, 2957–2981. [Google Scholar] [CrossRef]
- Bachhar, A.; Dalapati, P.; Bose, A.; Barman, S.; Roy, D.; Mandal, S. Intelligent Self-Healing Hydrogels: AI-Empowered Design, Biomedical Innovation, and Future Horizons. Journal of Drug Delivery Science and Technology 2026, 108215. [Google Scholar] [CrossRef]
- Qi, Y.; Wang, F.; Liu, J.; Wang, C.; Liu, Y. Enzyme-mediated hydrogelation for biomedical applications: A review. International Journal of Biological Macromolecules 2025, 311, 143379. [Google Scholar] [CrossRef]
- Elham, B.; Hosseini, M.; Mohajer, M.; Hassanzadeh, S.; Saghati, S.; Hilborn, J.; Khanmohammadi, M. Enzymatic Crosslinked Hydrogels for Biomedical Application. Polymer Science, Series A 2021, 63, S1–S22. [Google Scholar] [CrossRef]
- Amirian, J.; Wychowaniec, J.K.; D′Este, M.; Vernengo, A.J.; Metlova, A.; Sizovs, A.; Brangule, A.; Bandere, D. Preparation and Characterization of Photo-Cross-Linkable Methacrylated Silk Fibroin and Methacrylated Hyaluronic Acid Composite Hydrogels. Biomacromolecules 2024, 25, 7078–7097. [Google Scholar] [CrossRef]
- Kim, H.; Jeong, J.H.; Fendereski, M.; Lee, H.-S.; Kang, D.Y.; Hur, S.S.; Amirian, J.; Kim, Y.; Pham, N.T.; Suh, N.; Hwang, N.S.; Ryu, S.; Yoon, J.K.; Hwang, Y. Heparin-Mimicking Polymer-Based In Vitro Platform Recapitulates In Vivo Muscle Atrophy Phenotypes. International Journal of Molecular Sciences 2021. [Google Scholar] [CrossRef]
- Sarker, A.; Amirian, J.; Min, Y.K.; Lee, B.T. HAp granules encapsulated oxidized alginate–gelatin–biphasic calcium phosphate hydrogel for bone regeneration. International Journal of Biological Macromolecules 2015, 81, 898–911. [Google Scholar] [CrossRef] [PubMed]
- Skadiņš; Labsvārds, K.D.; Grava, A.; Amirian, J.; Tomsone, L.E.; Ruško, J.; Viksna, A.; Bandere, D.; Brangule, A. Antimicrobial and Antibiofilm Properties of Latvian Honey against Causative Agents of Wound Infections. Antibiotics 2023. [Google Scholar] [CrossRef] [PubMed]
- Bashabsheh, R.H.F.; Al-Fawares, O.; Natsheh, I.; Bdeir, R.; Al-Khreshieh, R.O.; Bashabsheh, H.H.F. Staphylococcus aureus epidemiology, pathophysiology, clinical manifestations and application of nano-therapeutics as a promising approach to combat methicillin resistant Staphylococcus aureus. Pathog Glob Health 2024, 118, 209–231. [Google Scholar] [CrossRef] [PubMed]
- Qin, S.; Xiao, W.; Zhou, C.; Pu, Q.; Deng, X.; Lan, L.; Liang, H.; Song, X.; Wu, M. Pseudomonas aeruginosa: Pathogenesis, virulence factors, antibiotic resistance, interaction with host, technology advances and emerging therapeutics. Signal Transduct Target Ther 2022, 7, 199. [Google Scholar] [CrossRef]
- Edo, G.I.; Ndudi, W.; Ali, A.B.M.; Yousif, E.; Zainulabdeen, K.; Akpoghelie, P.O.; Isoje, E.F.; Igbuku, U.A.; Opiti, R.A.; Athan Essaghah, A.E.; Ahmed, D.S.; Umar, H.; Alamiery, A.A. Chitosan: An overview of its properties, solubility, functional technologies, food and health applications. Carbohydrate Research 2025, 550, 109409. [Google Scholar] [CrossRef]
- Zeng; Zeng, Z.; Cheng, Z.; Wang, Y.; Wang, X.; Wang, B.; Gao, W. Cellulose nanofibrils manufactured by various methods with application as paper strength additives. Scientific Reports 2021, 11, 11918. [Google Scholar] [CrossRef]
- Antony Jose, S.; Cowan, N.; Davidson, M.; Godina, G.; Smith, I.; Xin, J.; Menezes, P.L. A Comprehensive Review on Cellulose Nanofibers, Nanomaterials, and Composites: Manufacturing, Properties, and Applications. Nanomaterials 2025, 356. [Google Scholar] [CrossRef]
- Lu, Z.; Zhang, H.; Toivakka, M.; Xu, C. Current progress in functionalization of cellulose nanofibers (CNFs) for active food packaging. International Journal of Biological Macromolecules 2024, 267, 131490. [Google Scholar] [CrossRef]
- Fernández-Santos; Valls, C.; Cusola, O.; Roncero, M.B. Periodate oxidation of nanofibrillated cellulose films for active packaging applications. International Journal of Biological Macromolecules 2024, 267, 131553. [Google Scholar] [CrossRef]
- Lamm, M.E.; Li, K.; Ker, D.; Zhao, X.; Hinton, H.E.; Copenhaver, K.; Tekinalp, H.; Ozcan, S. Exploiting chitosan to improve the interface of nanocellulose reinforced polymer composites. Cellulose 2022, 29, 3859–3870. [Google Scholar] [CrossRef]
- Bucciarelli, A.; Selicato, N.; Coricciati, C.; Rainer, A.; Capodilupo, A.L.; Gigli, G.; Moroni, L.; Polini, A.; Gervaso, F. Modelling methacrylated chitosan hydrogel properties through an experimental design approach: From composition to material properties. Journal of Materials Chemistry B 2024, 12, 10221–10240. [Google Scholar] [CrossRef]
- Kolawole, O.M.; Lau, W.M.; Khutoryanskiy, V.V. Methacrylated chitosan as a polymer with enhanced mucoadhesive properties for transmucosal drug delivery. International Journal of Pharmaceutics 2018, 550, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Sayyar, S.; Gambhir, S.; Chung, J.; Officer, D.L.; Wallace, G.G. 3D printable conducting hydrogels containing chemically converted graphene. Nanoscale 2017, 9, 2038–2050. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Guo, B.; Wu, H.; Liang, Y.; Ma, P.X. Injectable antibacterial conductive nanocomposite cryogels with rapid shape recovery for noncompressible hemorrhage and wound healing. Nature Communications 2018, 9, 2784. [Google Scholar] [CrossRef] [PubMed]
- Visan, A.I.; Popescu-Pelin, G.; Socol, G. Degradation Behavior of Polymers Used as Coating Materials for Drug Delivery-A Basic Review. Polymers (Basel) 2021, 13. [Google Scholar] [CrossRef]
- Catoira, M.C.; Fusaro, L.; Di Francesco, D.; Ramella, M.; Boccafoschi, F. Overview of natural hydrogels for regenerative medicine applications. Journal of Materials Science: Materials in Medicine 2019, 30, 115. [Google Scholar] [CrossRef]
- Wojciechowski; Gutarowicz, M.; Mierzejewska, J.; Parzuchowski, P. Antimicrobial films of poly(2-aminoethyl methacrylate) and its copolymers doped with TiO2 and CaCO3. Colloids and Surfaces B: Biointerfaces 2020, 185, 110605. [Google Scholar] [CrossRef]
- Punia, A.; Debata, P.R.; Banerjee, P.; Yang, N.-L. Structure–property relationships of antibacterial amphiphilic polymers derived from 2-aminoethyl acrylate. RSC Advances 2015, 5, 95300–95306. [Google Scholar] [CrossRef]
- Lyu, X.; Cui, F.; Zhou, H.; Cao, B.; Zhang, X.; Cai, M.; Yang, S.; Sun, B.; Li, G. 3D co-culture of macrophages and fibroblasts in a sessile drop array for unveiling the role of macrophages in skin wound-healing. Biosensors and Bioelectronics 2023, 225, 115111. [Google Scholar] [CrossRef]
- Amirian, J.; Linh, N.T.B.; Min, Y.K.; Lee, B.-T. The effect of BMP-2 and VEGF loading of gelatin-pectin-BCP scaffolds to enhance osteoblast proliferation. Journal of Applied Polymer Science 2015, 132. [Google Scholar] [CrossRef]


| Sample name | ChiMA (wt.%) | OCNFMA (wt.%) | Title 3 |
|---|---|---|---|
| ChiMA | 100 | 0 | ----- |
| ChiMA7–OCNFMA3–L | 70 | 30 | Low methacrylation (L) |
| ChiMA5–OCNFMA5–L | 50 | 50 | Low methacrylation (L) |
| ChiMA3–OCNFMA7–L | 30 | 70 | Low methacrylation (L) |
| ChiMA7–OCNFMA3–H | 70 | 30 | High methacrylation (H) |
| ChiMA5–OCNFMA5–H | 50 | 50 | High methacrylation (H) |
| ChiMA3–OCNFMA7–H | 30 | 70 | High methacrylation (H) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




