Submitted:
11 March 2026
Posted:
13 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
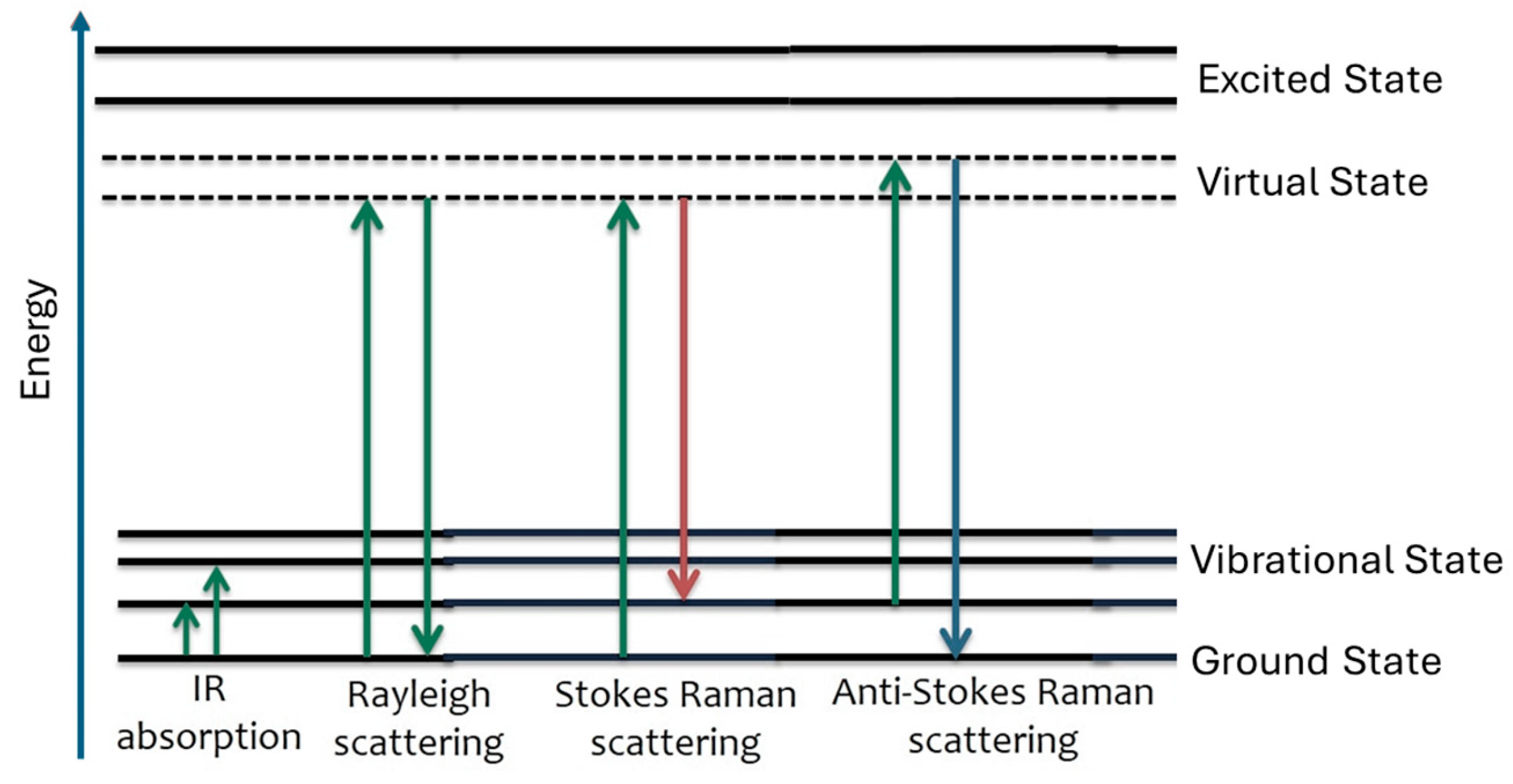
2. Raman Spectroscopy: Basic Principles
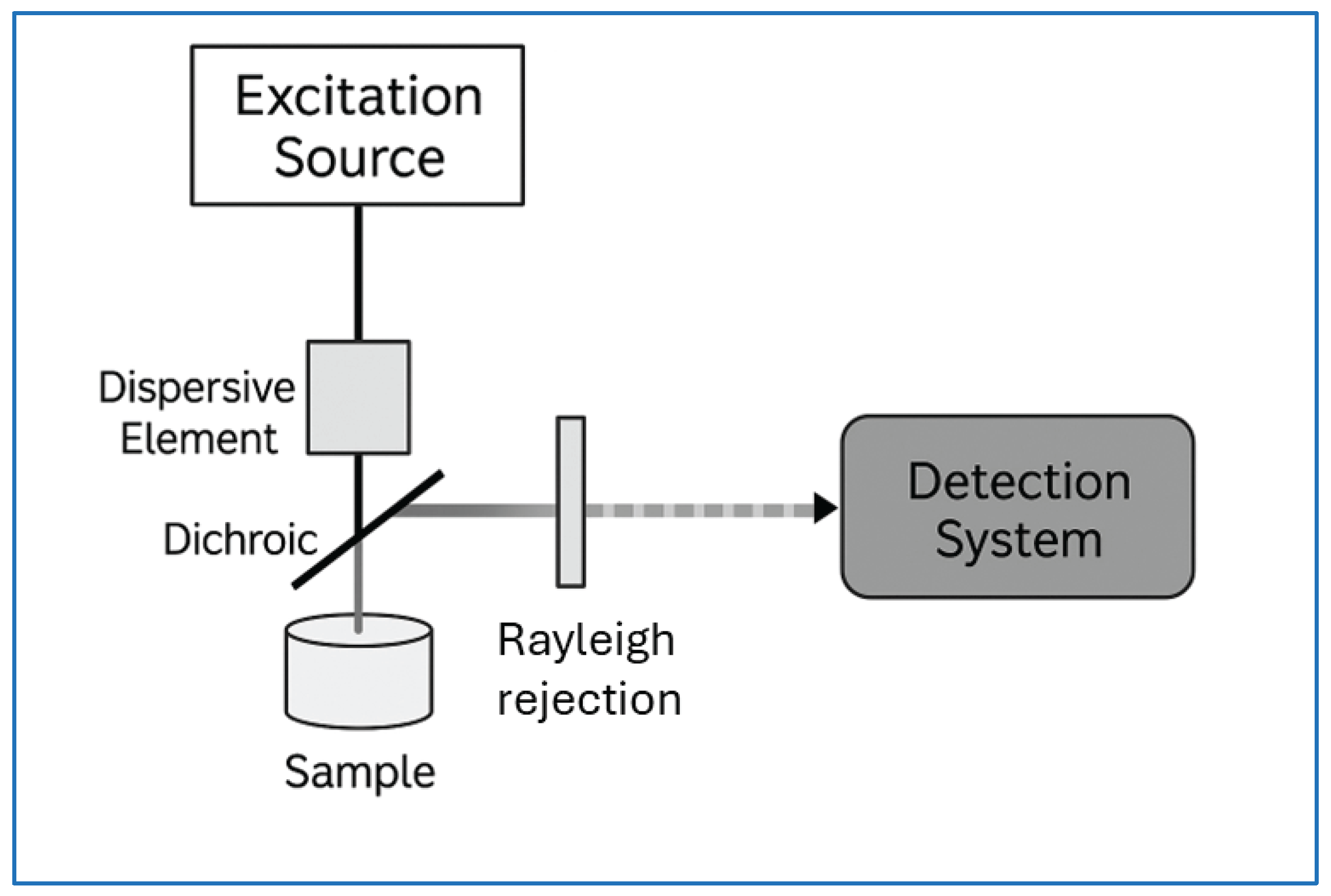
3. Methodology
3.1. Sample Selection
3.2. Spectral Acquisition Parameters
3.3. Spectra Preprocessing and Analyses
4. Applications of Raman Spectroscopy in Clinical Diagnosis
4.1. Oral Cancer
4.2. Breast and Cervical Cancers
4.3. Colorectal and Gastric Cancer
4.4. Brain Cancer
4.5. Other Cancers
4.6. Neurological/Neurodegenerative Disorders
4.7. Atherosclerosis
4.8. Infections and Pathogen Identification
5. Discussion
6. Conclusions
Author Contributions
Acknowledgments
References
- Rosenberg, A.; DeBusk, L.; Shah, M.; Burshtein, J.; Zakria, D.; Rigel, D. Updated Techniques for Melanoma Diagnosis. Dermatologic clinics 2025, 43, 433–442. [Google Scholar] [CrossRef] [PubMed]
- Hussain, S.; Mubeen, I.; Ullah, N.; Shah, S.S.U.D.; Khan, B.A.; Zahoor, M.; Ullah, R.; Khan, F.A.; Sultan, M.A. Modern diagnostic imaging technique applications and risk factors in the medical field: a review. BioMed research international 2022, 2022, 5164970. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Yao, Y.; Zhu, B.; Yu, Y.; Ren, R.; Hu, Y. The novel imaging methods in diagnosis and assessment of cerebrovascular diseases: an overview. Frontiers in medicine 2024, 11, 1269742–1269742. [Google Scholar] [CrossRef] [PubMed]
- Tseng, L.-J.; Matsuyama, A.; MacDonald-Dickinson, V. Histology: The gold standard for diagnosis? The Canadian veterinary journal = La revue veterinaire canadienne 2023, 64, 389–391. [Google Scholar] [PubMed]
- Coudray, N.; Ocampo, P.S.; Sakellaropoulos, T.; Narula, N.; Snuderl, M.; Fenyö, D.; Moreira, A.L.; Razavian, N.; Tsirigos, A. Classification and mutation prediction from non–small cell lung cancer histopathology images using deep learning. Nature Medicine 2018, 24, 1559–1567. [Google Scholar] [CrossRef]
- Haier, J.; Sleeman, J.; Schäfers, J. Editorial series: cancer care in low-and middle-income countries. Clinical & Experimental Metastasis 2019, 36, 477–480. [Google Scholar]
- Yadav, K.; Cree, I.; Field, A.; Vielh, P.; Mehrotra, R. Importance of cytopathologic diagnosis in early cancer diagnosis in resource-constrained countries. JCO Global Oncology 2022, 8, e2100337. [Google Scholar] [CrossRef]
- Kralova, K.; Vrtelka, O.; Fouskova, M.; Smirnova, T.A.; Michalkova, L.; Hribek, P.; Urbanek, P.; Kuckova, S.; Setnicka, V. Comprehensive spectroscopic, metabolomic, and proteomic liquid biopsy in the diagnostics of hepatocellular carcinoma. Talanta 2024, 270, 125527–125527. [Google Scholar] [CrossRef]
- Guo, M.; Hu, X.; Du, W. Near-Infrared-II Fluorescence Imaging of Tumors with Organic Small-Molecule Fluorophores. Sensors (Basel, Switzerland) 2025, 25. [Google Scholar] [CrossRef]
- Kumar, P.; Murali Krishna, C. Optical techniques: investigations in oral cancers. Oral cancer detection: novel strategies and clinical impact 2019, 167–187. [Google Scholar]
- Raman, C.V.; Krishnan, K.S. A new type of secondary radiation. Nature 1928, 121, 501–502. [Google Scholar] [CrossRef]
- Kouri, M.A.; Spyratou, E.; Karnachoriti, M.; Kalatzis, D.; Danias, N.; Arkadopoulos, N.; Seimenis, I.; Raptis, Y.S.; Kontos, A.G.; Efstathopoulos, E.P. Raman Spectroscopy: A Personalized Decision-Making Tool on Clinicians' Hands for In Situ Cancer Diagnosis and Surgery Guidance. Cancers 2022, 14. [Google Scholar] [CrossRef]
- Zhang, Y.; Yu, H.; Li, Y.; Xu, H.; Yang, L.; Shan, P.; Du, Y.; Yan, X.; Chen, X. Raman spectroscopy: A prospective intraoperative visualization technique for gliomas. Frontiers in oncology 2022, 12, 1086643–1086643. [Google Scholar] [CrossRef]
- Kumar, P.; Bhattacharjee, T.; Pandey, M.; Hole, A.; Ingle, A.; Murali Krishna, C. Raman spectroscopy in experimental oral carcinogenesis: investigation of abnormal changes in control tissues. J Raman Spectrosc 2016, 47, 1318–1326. [Google Scholar] [CrossRef]
- Kumar, P. Raman spectroscopy as a promising noninvasive tool in brain cancer detection. Journal of Innovative Optical Health Sciences 2017, 10, 1730012. [Google Scholar] [CrossRef]
- Hole, A.; Bhujbal, M.; Bendale, K.; Kumar, P.; Gera, P.; Krishna, C.M.; Chaudhari, P. Preliminary study of canine oral cancer by Raman spectroscopy. In Proceedings of the Optics and Biophotonics in Low-Resource Settings V, 2019; p. 1086917. [Google Scholar]
- Austin, C.; Kumar, P.; Carter, E.A.; Lee, J.; Smith, T.M.; Hinde, K.; Arora, M.; Lay, P.A. Stress exposure histories revealed by biochemical changes along accentuated lines in teeth. Chemosphere 2023, 329, 138673. [Google Scholar] [CrossRef]
- Kumar, P.; Austin, C. Signal collection through water-immersion objectives improves Raman spectral quality from dental tissues. In Proceedings of the Lasers in Dentistry XXVIII, 2022; p. 1194202. [Google Scholar]
- Kumar, P.; Sahu, A. Serum Raman spectroscopy-based biomedical diagnostics. In Spectroscopic Techniques for Cancer Diagnostics; IOP Publishing Bristol, UK, 2025; p. pp. 9–1–9–18. [Google Scholar]
- Kumar, P. Optoexposomics: Integrating biophotonics and exposomics towards precision health. In Proceedings of the Biomedical Vibrational Spectroscopy 2026: Advances in Research and Industry, San Francisco, 2026. [Google Scholar]
- Kumar, P.; Maroli, A.; Martinez, M.; Petrik, L.; Arora, M. Rapid screening of exposome in urban and semi-urban settings using silicone wrist bands. In Proceedings of the Optical Fibers and Sensors for Medical Diagnostics, Treatment, and Environmental Applications XXVI, San Francisco, 2026; p. 2026. [Google Scholar]
- Bhattacharjee, T.; Khan, A.; Kumar, P.; Ingle, A.; Maru, G.; Krishna, C.M. Raman spectroscopy of serum: A study on 'pre' and 'post' breast adenocarcinoma resection in rat models. J Biophotonics 2015, 8, 575–583. [Google Scholar] [CrossRef]
- Bhattacharjee, T.; Kumar, P.; Maru, G.; Ingle, A.; Krishna, C.M. Swiss bare mice: a suitable model for transcutaneous in vivo Raman spectroscopic studies of breast cancer. Lasers in medical science 2014, 29, 325–333. [Google Scholar] [CrossRef]
- Zhang, S.; Qi, Y.; Tan, S.P.H.; Bi, R.; Olivo, M. Molecular Fingerprint Detection Using Raman and Infrared Spectroscopy Technologies for Cancer Detection: A Progress Review. Biosensors (Basel) 2023, 13. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Liao, L.; Zhu, R.; Wang, Y.; Chen, K.; Hou, G.; Chen, S. Precise diagnosis of small cell and non-small cell lung cancer based on Raman spectroscopy. Photodiagnosis and photodynamic therapy 2025, 55, 104747–104747. [Google Scholar] [CrossRef]
- Kumamoto, Y.; Harada, Y.; Takamatsu, T.; Tanaka, H. Label-free Molecular Imaging and Analysis by Raman Spectroscopy. Acta histochemica et cytochemica 2018, 51, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Deshmukh, A.; Chaturvedi, P.; Murali Krishna, C. In vivo Raman spectroscopic identification of premalignant lesions in oral buccal mucosa. Journal of biomedical optics 2012, 17, 105002–105002. [Google Scholar] [CrossRef]
- Pilot, R.; Signorini, R.; Durante, C.; Orian, L.; Bhamidipati, M.; Fabris, L. A Review on Surface-Enhanced Raman Scattering. Biosensors 2019, 9. [Google Scholar] [CrossRef]
- Tahir, M.A.; Dina, N.E.; Cheng, H.; Valev, V.K.; Zhang, L. Surface-enhanced Raman spectroscopy for bioanalysis and diagnosis. Nanoscale 2021, 13, 11593–11634. [Google Scholar] [CrossRef] [PubMed]
- Djaker, N.; Lenne, P.-F.; Marguet, D.; Colonna, A.; Hadjur, C.; Rigneault, H. Coherent anti-Stokes Raman scattering microscopy (CARS): Instrumentation and applications. Nuclear Instruments and Methods in Physics Research Section A: Accelerators, Spectrometers, Detectors and Associated Equipment 2007, 571, 177–181. [Google Scholar] [CrossRef]
- Matousek, P. Spatially offset Raman spectroscopy for non-invasive analysis of turbid samples. TrAC Trends in Analytical Chemistry 2018, 103, 209–214. [Google Scholar] [CrossRef]
- Tipping, W.J.; Faulds, K.; Graham, D. Advances in Super-resolution Stimulated Raman Scattering Microscopy. Chemical & biomedical imaging 2024, 2, 733–743. [Google Scholar] [CrossRef]
- Itoh, T.; Procházka, M.; Dong, Z.-C.; Ji, W.; Yamamoto, Y.S.; Zhang, Y.; Ozaki, Y. Toward a new era of SERS and TERS at the nanometer scale: From fundamentals to innovative applications. Chemical reviews 2023, 123, 1552–1634. [Google Scholar] [CrossRef]
- Nishiyama, R.; Furuya, K.; Tamura, T.; Nakao, R.; Peterson, W.; Hiramatsu, K.; Ding, T.; Goda, K. Fourier Transform Coherent Anti-Stokes Raman Scattering Spectroscopy: A Comprehensive Review. Analytical Chemistry 2024, 96, 18322–18336. [Google Scholar] [CrossRef] [PubMed]
- Lux, A.; Conti, C.; Botteon, A.; Mosca, S.; Matousek, P. Theoretical and Practical Considerations of Spatially Offset Raman Spectroscopy (SORS) and Micro-SORS. Applied Spectroscopy 2025(79), 919–932. [CrossRef]
- Min, W.; Cheng, J.-X.; Ozeki, Y. Theory, innovations and applications of stimulated Raman scattering microscopy. Nature Photonics 2025, 19, 803–816. [Google Scholar] [CrossRef]
- Butler, H.J.; Ashton, L.; Bird, B.; Cinque, G.; Curtis, K.; Dorney, J.; Esmonde-White, K.; Fullwood, N.J.; Gardner, B.; Martin-Hirsch, P.L. Using Raman spectroscopy to characterize biological materials. Nature protocols 2016, 11, 664–687. [Google Scholar] [CrossRef]
- Traynor, D.; Behl, I.; O’dea, D.; Bonnier, F.; Nicholson, S.; O’connell, F.; Maguire, A.; Flint, S.; Galvin, S.; Healy, C.M. Raman spectral cytopathology for cancer diagnostic applications. Nature Protocols 2021, 16, 3716–3735. [Google Scholar] [CrossRef]
- Haugen, E.J.; Gautam, R.; Locke, A.K.; Mahadevan-Jansen, A. In vivo Raman spectroscopy for real-time biochemical assessment of tissue pathology and physiology. Nature Protocols 2026, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Fedorov Kukk, A.; Wu, D.; Gaffal, E.; Panzer, R.; Emmert, S.; Roth, B. Multimodal system for optical biopsy of melanoma with integrated ultrasound, optical coherence tomography and Raman spectroscopy. Journal of Biophotonics 2022, 15, e202200129. [Google Scholar] [CrossRef]
- Guo, S.; Popp, J.r.; Bocklitz, T. Key Steps in the Workflow to Analyze Raman Spectra. Spectroscopy 2023, 30–33. [Google Scholar] [CrossRef]
- Qi, Y.; Chen, E.X.; Hu, D.; Yang, Y.; Wu, Z.; Zheng, M.; Sadi, M.A.; Jiang, Y.; Zhang, K.; Chen, Z.; et al. Applications of Raman spectroscopy in clinical medicine. Food Frontiers 2024, 5, 392–419. [Google Scholar] [CrossRef]
- Wahl, J.; Klint, E.; Hallbeck, M.; Hillman, J.; Wårdell, K.; Ramser, K. Impact of preprocessing methods on the Raman spectra of brain tissue. Biomed. Opt. Express 2022, 13, 6763–6763. [Google Scholar] [CrossRef] [PubMed]
- Yan, C. A review on spectral data preprocessing techniques for machine learning and quantitative analysis. iScience 2025, 28, 112759–112759. [Google Scholar] [CrossRef]
- Boateng, D. Advances in deep learning-based applications for Raman spectroscopy analysis: A mini-review of the progress and challenges. Microchemical Journal 2025, 209, 112692–112692. [Google Scholar] [CrossRef]
- Guo, S.; Popp, J.; Bocklitz, T. Chemometric analysis in Raman spectroscopy from experimental design to machine learning–based modeling. Nature protocols 2021, 16, 5426–5459. [Google Scholar] [CrossRef]
- Luo, R.; Popp, J.; Bocklitz, T. Deep learning for Raman spectroscopy: A review. Analytica 2022, 3, 287–301. [Google Scholar] [CrossRef]
- Boateng, D. Advances in deep learning-based applications for Raman spectroscopy analysis: A mini-review of the progress and challenges. Microchemical Journal 2025, 112692. [CrossRef]
- Cui, S.; Zhang, S.; Yue, S. Raman Spectroscopy and Imaging for Cancer Diagnosis. Journal of Healthcare Engineering 2018, 2018, 1–11. [Google Scholar] [CrossRef]
- Blake, N.; Gaifulina, R.; Griffin, L.D.; Bell, I.M.; Thomas, G.M. Machine learning of Raman spectroscopy data for classifying cancers: a review of the recent literature. Diagnostics 2022, 12, 1491. [Google Scholar] [CrossRef] [PubMed]
- Auner, G.W.; Koya, S.K.; Huang, C.; Broadbent, B.; Trexler, M.; Auner, Z.; Elias, A.; Mehne, K.C.; Brusatori, M.A. Applications of Raman spectroscopy in cancer diagnosis. Cancer and Metastasis Reviews 2018, 37, 691–717. [Google Scholar] [CrossRef]
- Le Campion, A.C.O.V.; Ribeiro, C.M.B.; Luiz, R.R.; da Silva Júnior, F.F.; Barros, H.C.S.; Dos Santos, K.d.C.B.; Ferreira, S.J.; Gonçalves, L.S.; Ferreira, S.M.S. Low survival rates of oral and oropharyngeal squamous cell carcinoma. International journal of dentistry 2017, 2017, 5815493. [Google Scholar] [CrossRef] [PubMed]
- Zini, A.; Czerninski, R.; Sgan-Cohen, H.D. Oral cancer over four decades: epidemiology, trends, histology, and survival by anatomical sites. Journal of oral pathology & medicine 2010, 39, 299–305. [Google Scholar]
- Sankaranarayanan, R.; Ramadas, K.; Thomas, G.; Muwonge, R.; Thara, S.; Mathew, B.; Rajan, B. Effect of screening on oral cancer mortality in Kerala, India: a cluster-randomised controlled trial. The Lancet 2005, 365, 1927–1933. [Google Scholar] [CrossRef]
- Venkatakrishna, K.; Kurien, J.; Pai, K.M.; Valiathan, M.; Kumar, N.N.; Murali Krishna, C.; Ullas, G.; Kartha, V. Optical pathology of oral tissue: a Raman spectroscopy diagnostic method. Current science 2001, 80, 665–669. [Google Scholar]
- Hanna, K.; Asiedu, A.-L.; Theurer, T.; Muirhead, D.; Speirs, V.; Oweis, Y.; Abu-Eid, R. Advances in Raman spectroscopy for characterising oral cancer and oral potentially malignant disorders. Expert reviews in molecular medicine 2024, 26, e25–e25. [Google Scholar] [CrossRef]
- Byrne, H.J.; Behl, I.; Calado, G.; Ibrahim, O.; Toner, M.; Galvin, S.; Healy, C.M.; Flint, S.; Lyng, F.M. Biomedical applications of vibrational spectroscopy: Oral cancer diagnostics. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy 2021, 252, 119470–119470. [Google Scholar] [CrossRef]
- Hanna, K.; Asiedu, A.-L.; Theurer, T.; Muirhead, D.; Speirs, V.; Oweis, Y.; Abu-Eid, R. Advances in Raman spectroscopy for characterising oral cancer and oral potentially malignant disorders. Expert Reviews in Molecular Medicine 2024, 26, e25. [Google Scholar] [CrossRef] [PubMed]
- Aaboubout, Y.; Soares, M.R.N.; Schut, T.C.B.; Barroso, E.M.; van der Wolf, M.; Sokolova, E.; Artyushenko, V.; Bocharnikov, A.; Usenov, I.; van Lanschot, C.G. Intraoperative assessment of resection margins by Raman spectroscopy to guide oral cancer surgery. Analyst 2023, 148, 4116–4126. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, L.; Sun, Q.; Chen, B.; Zhao, C.; Dong, Y.; Zhu, Z.; Zhao, R.; Ma, X.; Yu, M. Rapid multi-task diagnosis of oral cancer leveraging fiber-optic Raman spectroscopy and deep learning algorithms. Frontiers in Oncology 2023, 13, 1272305. [Google Scholar] [CrossRef] [PubMed]
- Behl, I.; Calado, G.; Vishwakarma, A.; Traynor, D.; Flint, S.; Galvin, S.; Healy, C.M.; Pimentel, M.L.; Malkin, A.; Byrne, H.J.; et al. Classification of cytological samples from oral potentially malignant lesions through Raman spectroscopy: A pilot study. Spectrochim Acta A Mol Biomol Spectrosc 2022, 266, 120437. [Google Scholar] [CrossRef]
- Behl, I.; Calado, G.; Vishwakarma, A.; Traynor, D.; Flint, S.; Galvin, S.; Healy, C.M.; Malkin, A.; Byrne, H.J.; Lyng, F.M. Identification of high-risk oral leukoplakia (OLK) using combined Raman spectroscopic analysis of brush biopsy and saliva samples: A proof of concept study. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy 2025(330), 125721. [CrossRef]
- Sahu, A.; Deshmukh, A.; Ghanate, A.; Singh, S.; Chaturvedi, P.; Krishna, C.M. Raman spectroscopy of oral buccal mucosa: a study on age-related physiological changes and tobacco-related pathological changes. Technology in cancer research & treatment 2012, 11, 529–541. [Google Scholar]
- Singh, S.; Deshmukh, A.; Chaturvedi, P.; Krishna, C.M. In vivo Raman spectroscopy for oral cancers diagnosis. In Proceedings of the SPIE BiOS, 2012; p. pp. 82190K–82190K–82196. [Google Scholar]
- Sahu, A.; Nandakumar, N.; Sawant, S.; Krishna, C.M. Recurrence prediction in oral cancers: a serum Raman spectroscopy study. Analyst 2015, 140, 2294–2301. [Google Scholar] [CrossRef]
- Sahu, A.; Sawant, S.; Talathi-Desai, S.; Murali Krishna, C. Raman spectroscopy of serum: a study on oral cancers. Biomedical Spectroscopy and Imaging 2015, 4, 171–187. [Google Scholar] [CrossRef]
- Kumar, P.; Bhattacharjee, T.; Ingle, A.; Maru, G.; Krishna, C.M. Raman Spectroscopy of Experimental Oral Carcinogenesis: Study on Sequential Cancer Progression in Hamster Buccal Pouch Model. Technol Cancer Res Treat 2016, 15, NP60–72. [Google Scholar] [CrossRef] [PubMed]
- Malik, A.; Sahu, A.; Singh, S.P.; Deshmukh, A.; Chaturvedi, P.; Nair, D.; Nair, S.; Murali Krishna, C. In vivo Raman spectroscopy-assisted early identification of potential second primary/recurrences in oral cancers: An exploratory study. Head Neck 2017, 39, 2216–2223. [Google Scholar] [CrossRef]
- Kumar, P.; Ingle, A.; Krishna, C.M. In vivo Raman spectroscopy: monitoring cancer progression post carcinogen withdrawal. In Proceedings of the Optical Imaging, Therapeutics, and Advanced Technology in Head and Neck Surgery and Otolaryngology, 2019, 2019; p. 108530K. [Google Scholar]
- Hole, A.; Tyagi, G.; Deshmukh, A.; Deshpande, R.; Gota, V.; Chaturvedi, P.; Krishna, C.M. Salivary Raman Spectroscopy: Standardization of Sampling Protocols and Stratification of Healthy and Oral Cancer Subjects. Appl Spectrosc 2021, 75, 581–588. [Google Scholar] [CrossRef]
- Saha, P.; Sawant, S.; Deshmukh, A.; Hole, A.; Murali Krishna, C. Serum Raman spectroscopy: Prognostic applications in oral cancers. Head & Neck 2023, 45, 1244–1254. [Google Scholar] [CrossRef] [PubMed]
- Hole, A.; Bendale, K.; Gera, P.; Chaudhari, P.; Krishna, C.M. Raman spectroscopy of canine cancer to differentiate oral malignancy. In Proceedings of the European Conference on Biomedical Optics, 2025; p. M4A. 4. [Google Scholar]
- Sahu, A.; Sawant, S.; Mamgain, H.; Krishna, C.M. Raman spectroscopy of serum: an exploratory study for detection of oral cancers. Analyst 2013, 138, 4161–4174. [Google Scholar] [CrossRef] [PubMed]
- Sahu, A.K.; Dhoot, S.; Singh, A.; Sawant, S.S.; Nandakumar, N.; Talathi-Desai, S.; Garud, M.; Pagare, S.; Srivastava, S.; Nair, S. Oral cancer screening: serum Raman spectroscopic approach. Journal of biomedical optics 2015, 20, 115006–115006. [Google Scholar] [CrossRef]
- Singh, S.; Sahu, A.; Deshmukh, A.; Chaturvedi, P.; Krishna, C.M. In vivo Raman spectroscopy of oral buccal mucosa: a study on malignancy associated changes (MAC)/cancer field effects (CFE). Analyst 2013, 138, 4175–4182. [Google Scholar] [CrossRef] [PubMed]
- Bray, F.; Laversanne, M.; Sung, H.; Ferlay, J.; Siegel, R.L.; Soerjomataram, I.; Jemal, A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: a cancer journal for clinicians 2024, 74, 229–263. [Google Scholar] [CrossRef]
- Hanna, K.; Krzoska, E.; Shaaban, A.M.; Muirhead, D.; Abu-Eid, R.; Speirs, V. Raman spectroscopy: current applications in breast cancer diagnosis, challenges and future prospects. British journal of cancer 2022, 126, 1125–1139. [Google Scholar] [CrossRef]
- Liu, C.; Alfano, R.; Sha, W.; Zhu, H.; Akins, D.; Cleary, J.; Prudente, R.; Cellmer, E. Human breast tissues studied by IR Fourier-transform Raman spectroscopy. In Proceedings of the Conference on lasers and electro-optics, 1991; p. CWF51. [Google Scholar]
- David, S.; Tran, T.; Dallaire, F.; Sheehy, G.; Azzi, F.; Trudel, D.; Tremblay, F.; Omeroglu, A.; Leblond, F.; Meterissian, S. In situ Raman spectroscopy and machine learning unveil biomolecular alterations in invasive breast cancer. Journal of Biomedical Optics 2023, 28, 036009–036009. [Google Scholar] [CrossRef]
- Li, J.; Wang, X.; Min, S.; Xia, J.; Li, J. Raman spectroscopy combined with convolutional neural network for the sub-types classification of breast cancer and critical feature visualization. Computer Methods and Programs in Biomedicine 2024, 255, 108361. [Google Scholar] [CrossRef] [PubMed]
- Mahadevan-Jansen, A.; Mitchell, M.F.; Ramanujamf, N.; Malpica, A.; Thomsen, S.; Utzinger, U.; Richards-Kortumt, R. Near-infrared Raman spectroscopy for in vitro detection of cervical precancers. Photochemistry and photobiology 1998, 68, 123–132. [Google Scholar]
- Kanter, E.M.; Vargis, E.; Majumder, S.; Keller, M.D.; Woeste, E.; Rao, G.G.; Mahadevan-Jansen, A. Application of Raman spectroscopy for cervical dysplasia diagnosis. Journal of biophotonics 2009, 2, 81–90. [Google Scholar] [CrossRef]
- Kanter, E.M.; Majumder, S.; Kanter, G.J.; Woeste, E.M.; Mahadevan-Jansen, A. Effect of hormonal variation on Raman spectra for cervical disease detection. Am J Obstet Gynecol 2009, 200, 512. e511–512. e515. [Google Scholar] [CrossRef] [PubMed]
- Mahadevan-Jansen, A.; Ramanujam, N.; Follen-Mitchell, M.; Malpica, A.; Thomsen, S.L.; Richards-Kortum, R.R. Optical techniques for diagnosis of cervical precancers: a comparison of Raman and fluorescence spectroscopies. In Proceedings of the Advances in fluorescence sensing technology II, 1995; pp. 110–120. [Google Scholar]
- Proietti, A.; De Angelis, E.; Buccini, L.; Leopizzi, M.; Pernazza, A.; Mura, F.; Accorinti, A.; Sbardella, G.; La Penna, G.; Maria Arseni, R. A pilot multimodal study of cervical cancer: Raman spectroscopy as a molecular fingerprint tool. PloS one 2026, 21, e0327286. [Google Scholar] [CrossRef]
- Fousková, M.; Vališ, J.; Synytsya, A.; Habartová, L.; Petrtýl, J.; Petruželka, L.; Setnička, V. In vivo Raman spectroscopy in the diagnostics of colon cancer. The Analyst 2023, 148, 2518–2526. [Google Scholar] [CrossRef]
- Liu, K.; Zhao, Q.; Li, B.; Zhao, X. Raman Spectroscopy: A Novel Technology for Gastric Cancer Diagnosis. Frontiers in bioengineering and biotechnology 2022, 10, 856591–856591. [Google Scholar] [CrossRef]
- Li, X.; Yang, T.; Yu, T.; Li, S. Discrimination of serum Raman spectroscopy between normal and colorectal cancer. In Proceedings of the European Conference on Biomedical Optics, 2011; p. 808727. [Google Scholar]
- Ito, H.; Uragami, N.; Miyazaki, T.; Yang, W.; Issha, K.; Matsuo, K.; Kimura, S.; Arai, Y.; Tokunaga, H.; Okada, S.; et al. Highly accurate colorectal cancer prediction model based on Raman spectroscopy using patient serum. World J Gastrointest Oncol 2020, 12, 1311–1324. [Google Scholar] [CrossRef] [PubMed]
- Bahreini, M.; Hosseinzadegan, A.; Rashidi, A.; Miri, S.R.; Mirzaei, H.R.; Hajian, P. A Raman-based serum constituents’ analysis for gastric cancer diagnosis: In vitro study. Talanta 2019, 204, 826–832. [Google Scholar] [CrossRef]
- Jermyn, M.; Mok, K.; Mercier, J.; Desroches, J.; Pichette, J.; Saint-Arnaud, K.; Bernstein, L.; Guiot, M.-C.; Petrecca, K.; Leblond, F. Intraoperative brain cancer detection with Raman spectroscopy in humans. Science translational medicine 2015, 7, 274ra219–274ra219. [Google Scholar] [CrossRef]
- Lemoine, É.; Dallaire, F.; Yadav, R.; Agarwal, R.; Kadoury, S.; Trudel, D.; Guiot, M.-C.; Petrecca, K.; Leblond, F. Feature engineering applied to intraoperative in vivo Raman spectroscopy sheds light on molecular processes in brain cancer: a retrospective study of 65 patients. Analyst 2019, 144, 6517–6532. [Google Scholar] [CrossRef]
- Leblond, F.; Dallaire, F.; Ember, K.; Le Moël, A.; Blanquez-Yeste, V.; Tavera, H.; Sheehy, G.; Tran, T.; Guiot, M.-C.; Weil, A.G. Quantitative assessment of the generalizability of a brain tumor Raman spectroscopy machine learning model to various tumor types including astrocytoma and oligodendroglioma. Journal of Biomedical Optics 2025, 30, 010501–010501. [Google Scholar] [CrossRef]
- Daoust, F.; Dallaire, F.; Tavera, H.; Ember, K.; Guiot, M.-C.; Petrecca, K.; Leblond, F. Preliminary study demonstrating cancer cells detection at the margins of whole glioblastoma specimens with Raman spectroscopy imaging. Scientific Reports 2025, 15, 6453. [Google Scholar] [CrossRef]
- Dulude, J.-P.; Le Moël, A.; Dallaire, F.; Doyon, J.; Urmey, K.; Marple, E.; Leblanc, G.; Basile, G.; Mottard, S.; Isler, M. Intraoperative use of high-speed Raman spectroscopy during soft tissue sarcoma resection. Scientific Reports 2025, 15, 8789. [Google Scholar] [CrossRef]
- Blanquez-Yeste, V.; Janelle, F.; Tran, T.; Ember, K.; Sheehy, G.; Dallaire, F.; Marple, E.; Urmey, K.; Labidi, M.; Leblond, F. Development and preclinical evaluation of an endonasal Raman spectroscopy probe for transsphenoidal pituitary adenoma surgery. Journal of Biomedical Optics 2025, 30, 035004–035004. [Google Scholar] [CrossRef] [PubMed]
- Kaur, E.; Sahu, A.; Hole, A.R.; Rajendra, J.; Chaubal, R.; Gardi, N.; Dutt, A.; Moiyadi, A.; Krishna, C.M.; Dutt, S. Unique spectral markers discern recurrent Glioblastoma cells from heterogeneous parent population. Scientific reports 2016, 6, 26538–26538. [Google Scholar] [CrossRef]
- Mehta, K.; Atak, A.; Sahu, A.; Srivastava, S.; M.K., C. An early investigative serum Raman spectroscopy study of meningioma. Analyst 2018, 143, 1916–1923. [Google Scholar] [CrossRef]
- Lui, H.; Zhao, J.; McLean, D.; Zeng, H. Real-time Raman spectroscopy for in vivo skin cancer diagnosis. Cancer research 2012, 72, 2491–2500. [Google Scholar] [CrossRef] [PubMed]
- Fedorov Kukk, A.; Wu, D.; Panzer, R.; Emmert, S.; Roth, B. Four-modal device comprising optical coherence tomography, photoacoustic tomography, ultrasound, and Raman spectroscopy developed for in vivo skin lesion assessment. Biomed. Opt. Express 2025, 16, 1792–1806. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Sun, H.; Sun, L.; Shi, K.; Chen, Y.; Ren, X.; Ge, Y.; Jiang, D.; Liu, X.; Knoll, W.; et al. Rapid, label-free histopathological diagnosis of liver cancer based on Raman spectroscopy and deep learning. Nature communications 2023, 14, 48–48. [Google Scholar] [CrossRef] [PubMed]
- Paraskevaidi, M.; Morais, C.L.M.; Halliwell, D.E.; Mann, D.M.A.; Allsop, D.; Martin-Hirsch, P.L.; Martin, F.L. Raman Spectroscopy to Diagnose Alzheimer's Disease and Dementia with Lewy Bodies in Blood. ACS chemical neuroscience 2018, 9, 2786–2794. [Google Scholar] [CrossRef] [PubMed]
- Stevens, A.R.; Stickland, C.A.; Harris, G.; Ahmed, Z.; Goldberg Oppenheimer, P.; Belli, A.; Davies, D.J. Raman Spectroscopy as a Neuromonitoring Tool in Traumatic Brain Injury: A Systematic Review and Clinical Perspectives. Cells 2022, 11. [Google Scholar] [CrossRef]
- Alix, J.J.P.; Verber, N.S.; Schooling, C.N.; Kadirkamanathan, V.; Turner, M.R.; Malaspina, A.; Day, J.C.C.; Shaw, P.J. Label-free fibre optic Raman spectroscopy with bounded simplex-structured matrix factorization for the serial study of serum in amyotrophic lateral sclerosis. Analyst 2022, 147, 5113–5120. [Google Scholar] [CrossRef]
- Dogan, G.Y.; Halimoglu, G.; Kaplanoglu, D.; Aksoy, U.M.; Kandeger, A.; Yavuz, E.; Kartal, S.; Fausto, R.; Ildiz, G.O. Raman Spectra of Blood Serum as Holistic Biomarker for Differential Auxiliary Diagnoses of Attention Deficit and Hyperactivity Disorder (ADHD) in Adults. Spectroscopy Journal 2024, 2, 53–67. [Google Scholar] [CrossRef]
- Matthäus, C.; Dochow, S.; Bergner, G.; Lattermann, A.; Romeike, B.F.M.; Marple, E.T.; Krafft, C.; Dietzek, B.; Brehm, B.R.; Popp, J. In vivo characterization of atherosclerotic plaque depositions by Raman-probe spectroscopy and in vitro coherent anti-stokes Raman scattering microscopic imaging on a rabbit model. Analytical chemistry 2012, 84, 7845–7851. [Google Scholar] [CrossRef] [PubMed]
- van de Poll, S.W.E.; Kastelijn, K.; Bakker Schut, T.C.; Strijder, C.; Pasterkamp, G.; Puppels, G.J.; van der Laarse, A. On-line detection of cholesterol and calcification by catheter based Raman spectroscopy in human atherosclerotic plaque ex vivo. Heart (British Cardiac Society) 2003, 89, 1078–1082. [Google Scholar] [CrossRef]
- Kęsik, J.J.; Paja, W.; Terlecki, P.; Iłżecki, M.; Klebowski, B.; Depciuch, J. Raman spectroscopy combined with machine learning and chemometrics analyses as a tool for identification atherosclerotic carotid stenosis from serum. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy 2025, 326, 125198. [Google Scholar] [CrossRef] [PubMed]
- Ho, C.-S.; Jean, N.; Hogan, C.A.; Blackmon, L.; Jeffrey, S.S.; Holodniy, M.; Banaei, N.; Saleh, A.A.E.; Ermon, S.; Dionne, J. Rapid identification of pathogenic bacteria using Raman spectroscopy and deep learning. Nature communications 2019, 10, 4927–4927. [Google Scholar] [CrossRef]
- Salbreiter, M.; Frempong, S.B.; Even, S.; Wagenhaus, A.; Girnus, S.; Rösch, P.; Popp, J. Lighting the Path: Raman Spectroscopy’s Journey Through the Microbial Maze. Molecules 2024, 29, 5956. [Google Scholar] [CrossRef] [PubMed]
- Stöckel, S.; Kirchhoff, J.; Neugebauer, U.; Rösch, P.; Popp, J. The application of Raman spectroscopy for the detection and identification of microorganisms. J Raman Spectrosc 2016, 47, 89–109. [Google Scholar] [CrossRef]
- Paz-Bailey, G.; Adams, L.E.; Deen, J.; Anderson, K.B.; Katzelnick, L.C. Dengue. The Lancet 2024, 403, 667–682. [Google Scholar] [CrossRef]
- Jacob, S.S.; Lukose, J.; Bankapur, A.; Mithun, N.; Vani Lakshmi, R.; Acharya, M.; Rao, P.; Kamath, A.; Baby, P.M.; Rao, R.K.; et al. Micro-Raman spectroscopy study of optically trapped erythrocytes in malaria, dengue and leptospirosis infections. Frontiers in medicine 2022, 9, 858776–858776. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.K.; Rajora, N.; Kumar, S.; Sahu, A.; Kochar, S.K.; Krishna, C.M.; Srivastava, S. Rapid discrimination of malaria-and dengue-infected patients sera using raman spectroscopy. Analytical chemistry 2019, 91, 7054–7062. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Ullah, R.; Saleem, M.; Bilal, M.; Rashid, R.; Khan, I.; Mahmood, A.; Nawaz, M. Raman spectroscopic analysis of dengue virus infection in human blood sera. Optik 2016, 127, 2086–2088. [Google Scholar] [CrossRef]
- Khan, S.; Ullah, R.; Khan, A.; Wahab, N.; Bilal, M.; Ahmed, M. Analysis of dengue infection based on Raman spectroscopy and support vector machine (SVM). Biomed. Opt. Express 2016, 7, 2249–2256. [Google Scholar] [CrossRef]
- Khan, S.; Ullah, R.; Khan, A.; Sohail, A.; Wahab, N.; Bilal, M.; Ahmed, M. Random Forest-Based Evaluation of Raman Spectroscopy for Dengue Fever Analysis. Applied Spectroscopy 2017, 71, 2111–2117. [Google Scholar] [CrossRef]
- Yin, G.; Li, L.; Lu, S.; Yin, Y.; Su, Y.; Zeng, Y.; Luo, M.; Ma, M.; Zhou, H.; Orlandini, L. An efficient primary screening of COVID-19 by serum Raman spectroscopy. J Raman Spectrosc 2021, 52, 949–958. [Google Scholar] [CrossRef] [PubMed]
- Goulart, A.C.C.; Silveira, L.; Carvalho, H.C.; Dorta, C.B.; Pacheco, M.T.T.; Zângaro, R.A. Diagnosing COVID-19 in human serum using Raman spectroscopy. Lasers in Medical Science 2022, 37, 2217–2226. [Google Scholar] [CrossRef]
- Guleken, Z.; Tuyji Tok, Y.; Jakubczyk, P.; Paja, W.; Pancerz, K.; Shpotyuk, Y.; Cebulski, J.; Depciuch, J. Development of novel spectroscopic and machine learning methods for the measurement of periodic changes in COVID-19 antibody level. Measurement 2022, 196, 111258. [Google Scholar] [CrossRef]
- Singh, R. C. V. Raman and the Discovery of the Raman Effect. Physics in Perspective (PIP) 2002, 4, 399–420. [Google Scholar] [CrossRef]
- Wang, Y.; Fang, L.; Wang, Y.; Xiong, Z. Current Trends of Raman Spectroscopy in Clinic Settings: Opportunities and Challenges. Advanced science (Weinheim, Baden-Wurttemberg, Germany) 2024, 11, e2300668–e2300668. [Google Scholar] [CrossRef] [PubMed]
| Condition | Title | Author | Year | Sample / Modality | Raman Technique | Main Findings |
|---|---|---|---|---|---|---|
| Cervical cancer | Near-infrared Raman spectroscopy for in vitro detection of cervical precancers | Mahadevan-Jansen A | 1998 | Ex vivo cervical tissue | Near-infrared Raman | Differentiated normal vs precancerous lesions using biochemical fingerprints |
| Skin cancer | Melanoma diagnosis by Raman spectroscopy and neural networks : Structure Alterations in Proteins and Lipids in Intact Cancer Tissue | Gniadecka M | 2004 | Excised skin lesions | Near-infrared Fourier transform Raman spectra with neural network | Distinguished melanoma melanoma could be differentiated from pigmented nevi, basal cell carcinoma, seborrheic keratoses, and normal skin |
| Colorectal cancer | Classification of colonic tissues using near-infrared Raman spectroscopy and support vector machines | Widjaja E | 2008 | Ex vivo colon tissue | Near-infrared Raman | Support vector machines classification of normal vs cancerous colon tissue |
| Gastric cancer | Diagnostic potential of near-infrared Raman spectroscopy in the stomach: differentiating dysplasia from normal tissue | Teh SK | 2008 | Biopsy tissue | Near-infrared Raman | Differentiated dysplasia from normal gastric mucosa |
| Pancreatic cancer | Evaluation of pancreatic cancer with Raman spectroscopy in a mouse model | Pandya AK | 2008 | Animal pancreatic tissue | Spontaneous Raman | Identified chemical changes in normal and malignant tissue. |
| Brain tumors | Intraoperative brain cancer detection with Raman spectroscopy | Jermyn M | 2015 | In vivo during neurosurgery | Hand-held fiber optic probe Raman | Real-time discrimination of tumor vs normal brain |
| Traumatic brain injury – development | Development of Raman probe device toward neuromonitoring of Traumatic Brain Injury | Mowbray M | 2021 | Animal brain | Intracranial Raman probe | Detected biochemical alterations after Traumatic Brain Injury |
| Atherosclerosis | Determination of human coronary artery composition by Raman spectroscopy | Brennan JF | 1997 | Human artery tissue | Near-infrared Raman | Identified cholesterol, lipids and calcification in plaques |
| Liver fibrosis / cancer | Evaluation of liver fibrosis using Raman spectroscopy and infrared thermography: A pilot study | Ramírez-Elías MG | 2017 | Rat liver tissue | Raman with PCA- Linear Discriminant Analysis (LDA) | Classified normal vs fibrotic liver |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




