Submitted:
11 March 2026
Posted:
12 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
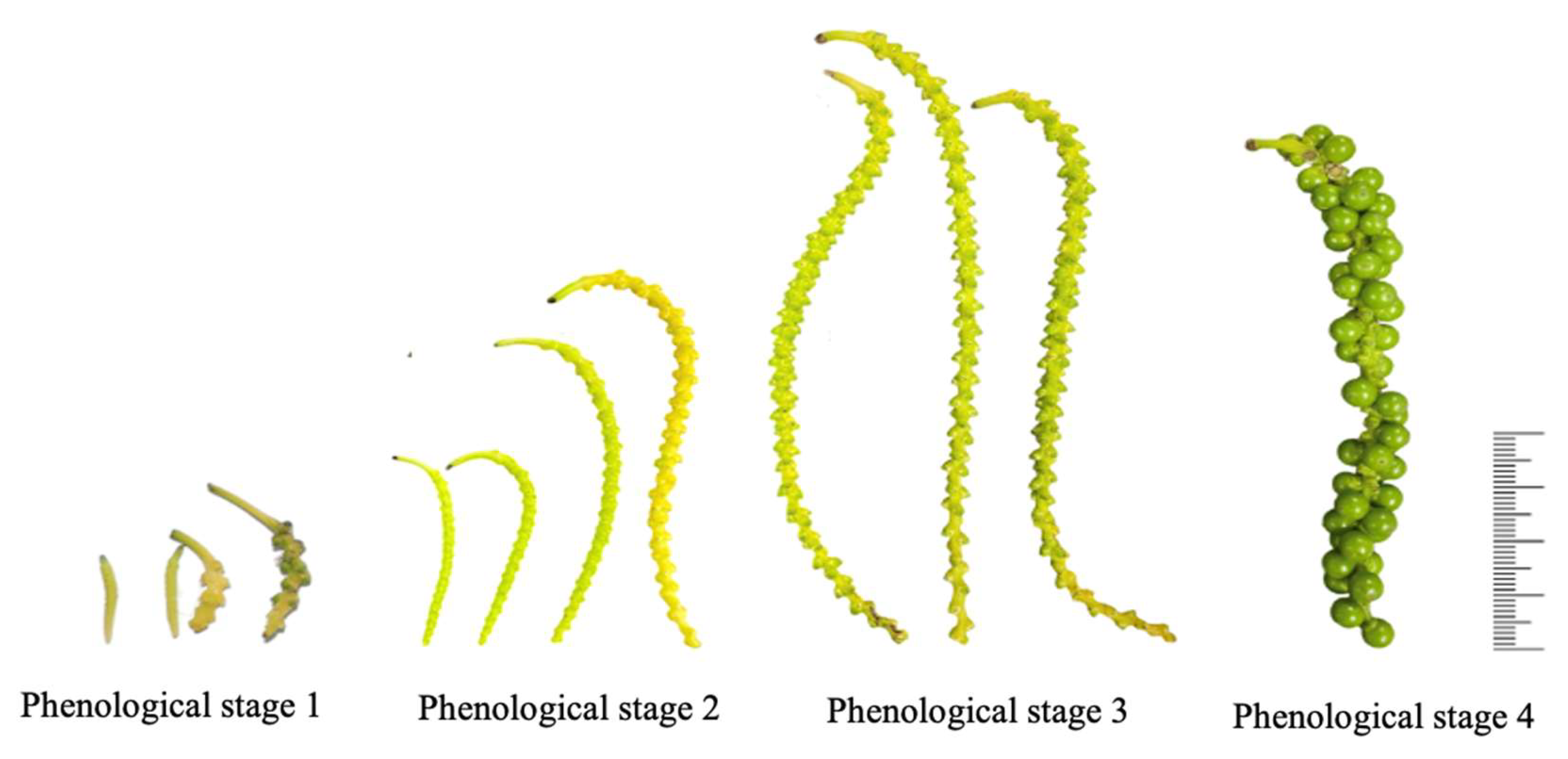
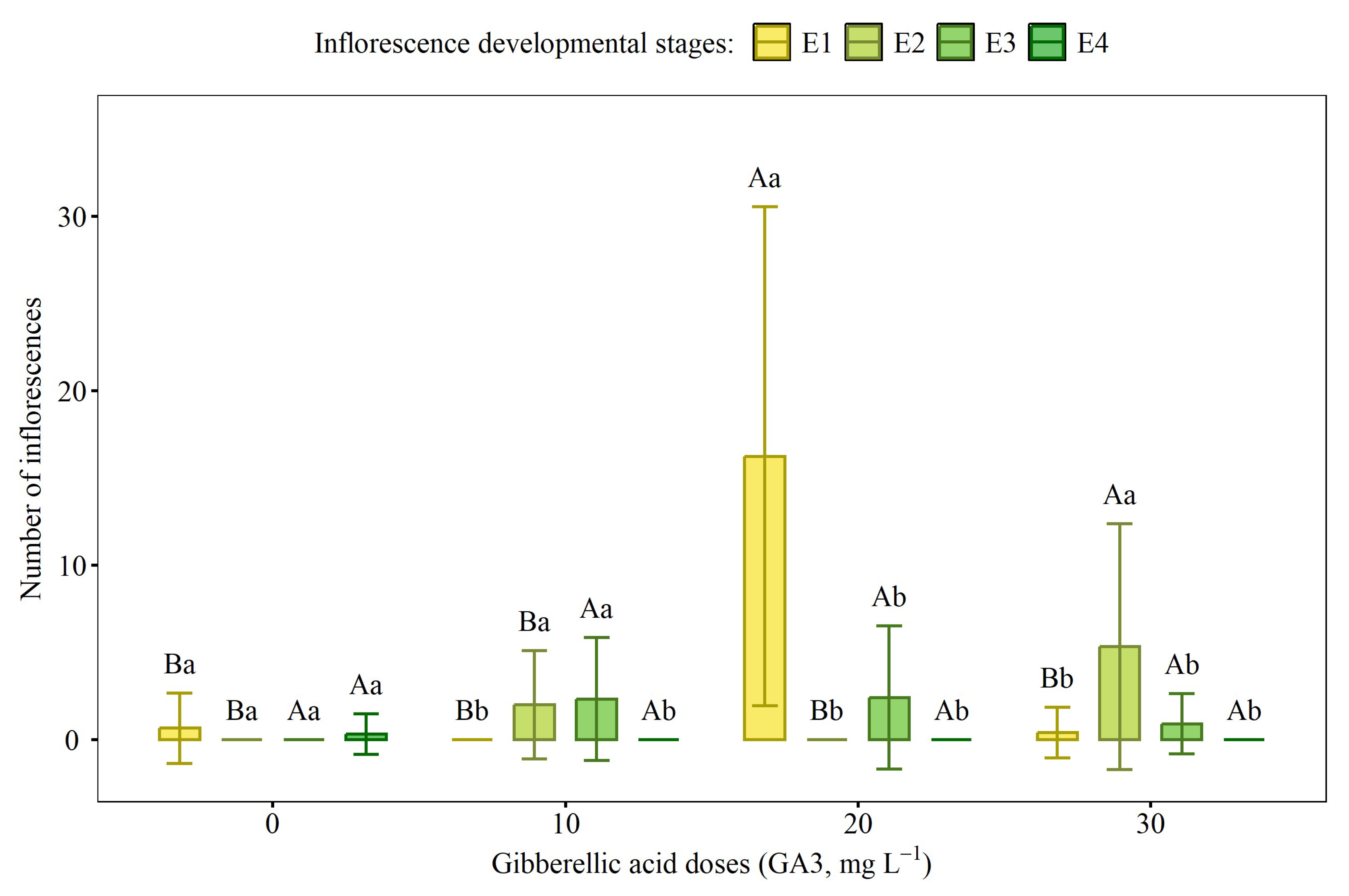
2.1. Flowering and Phenological Classification
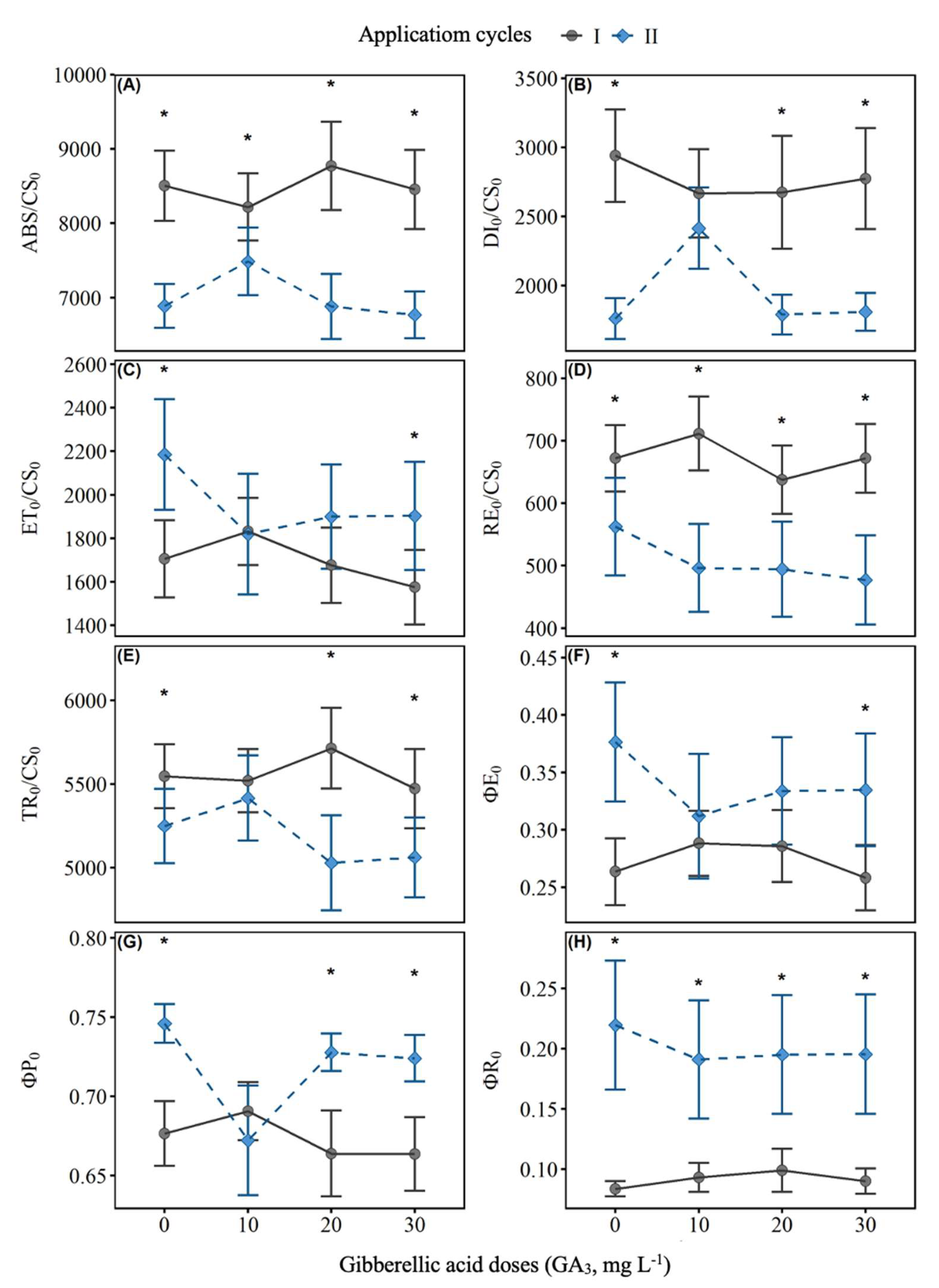
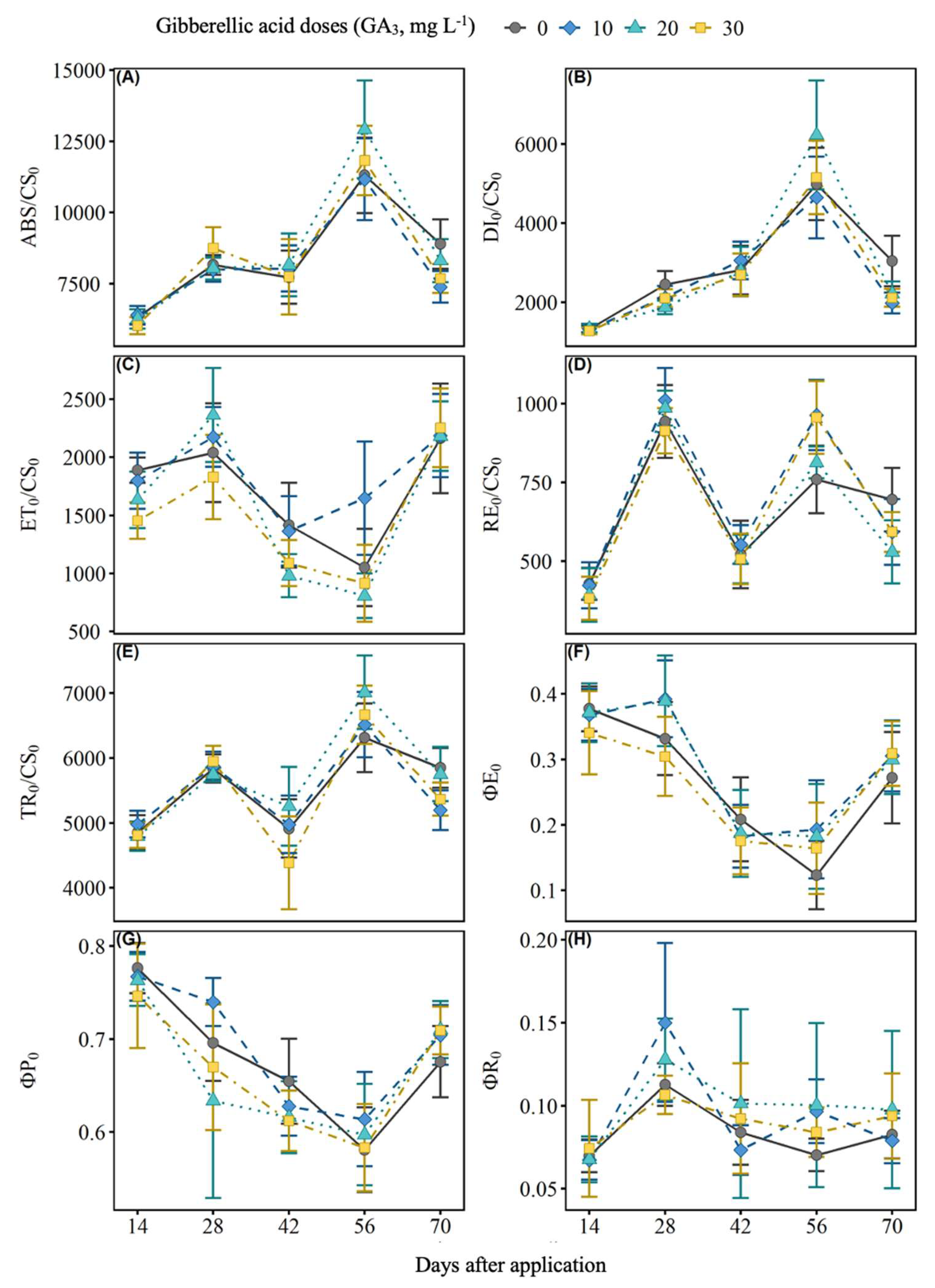
2.2. Chlorophyll a Fluorescence
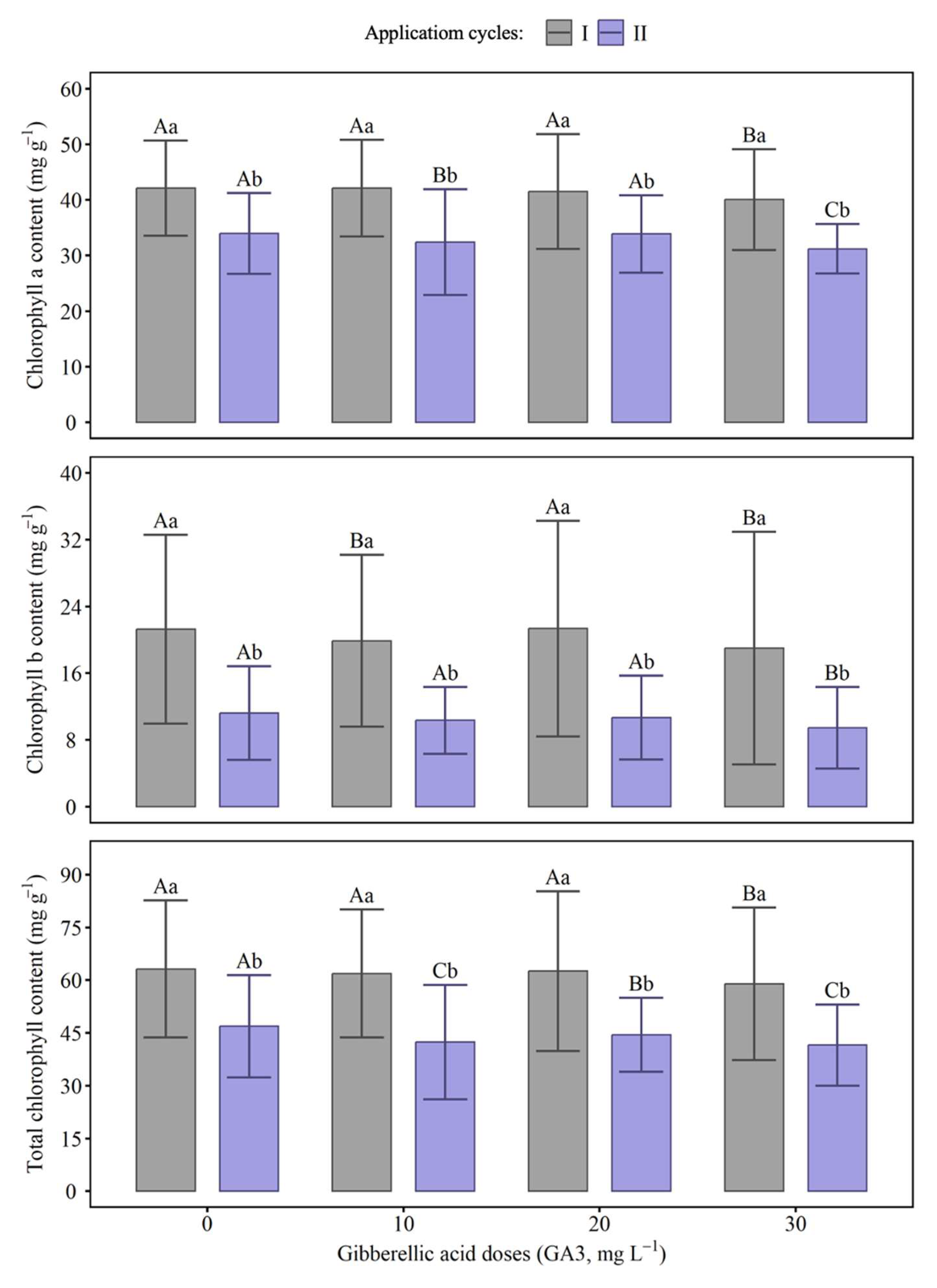
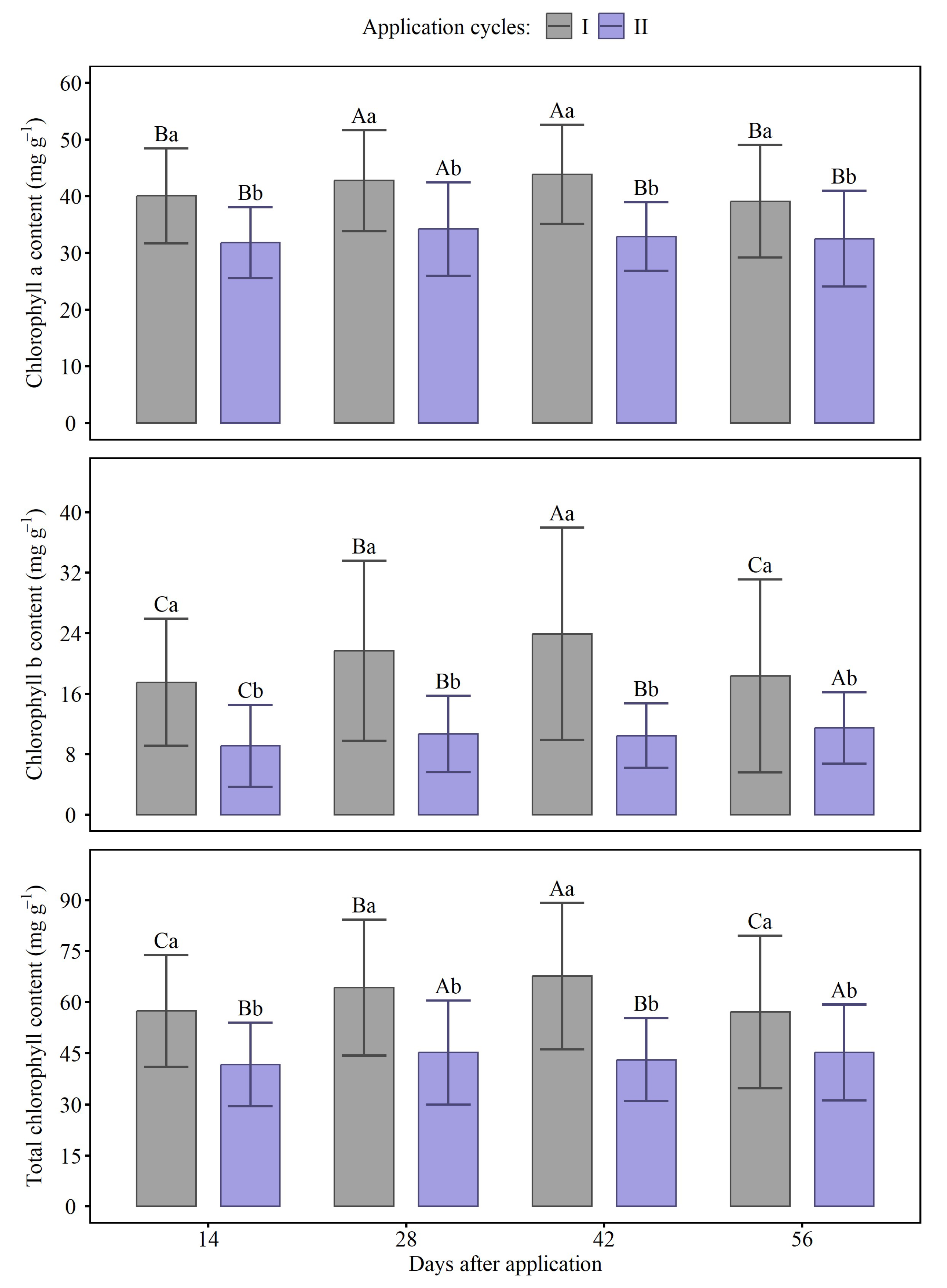
2.3. Chlorophyll Index
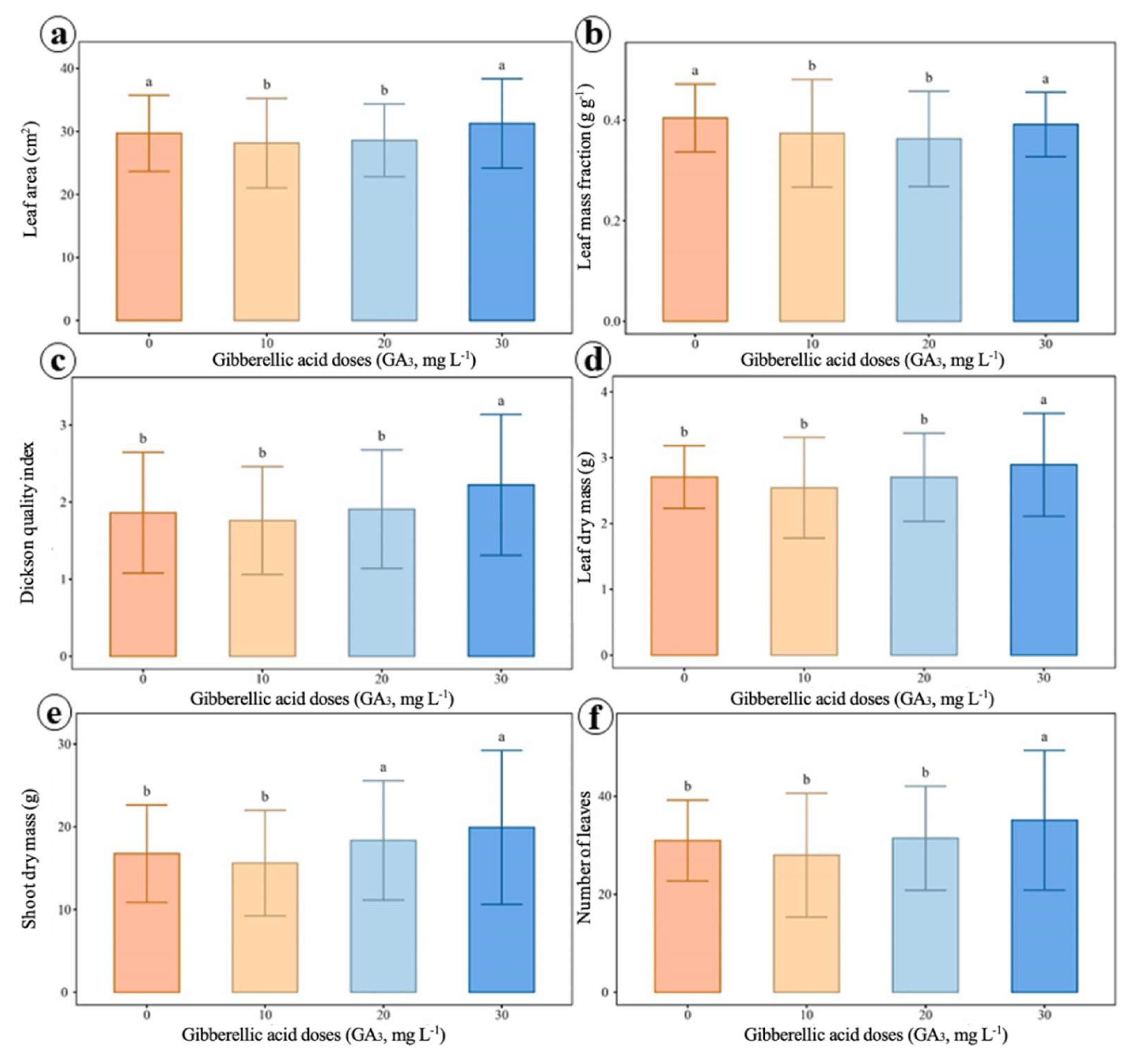
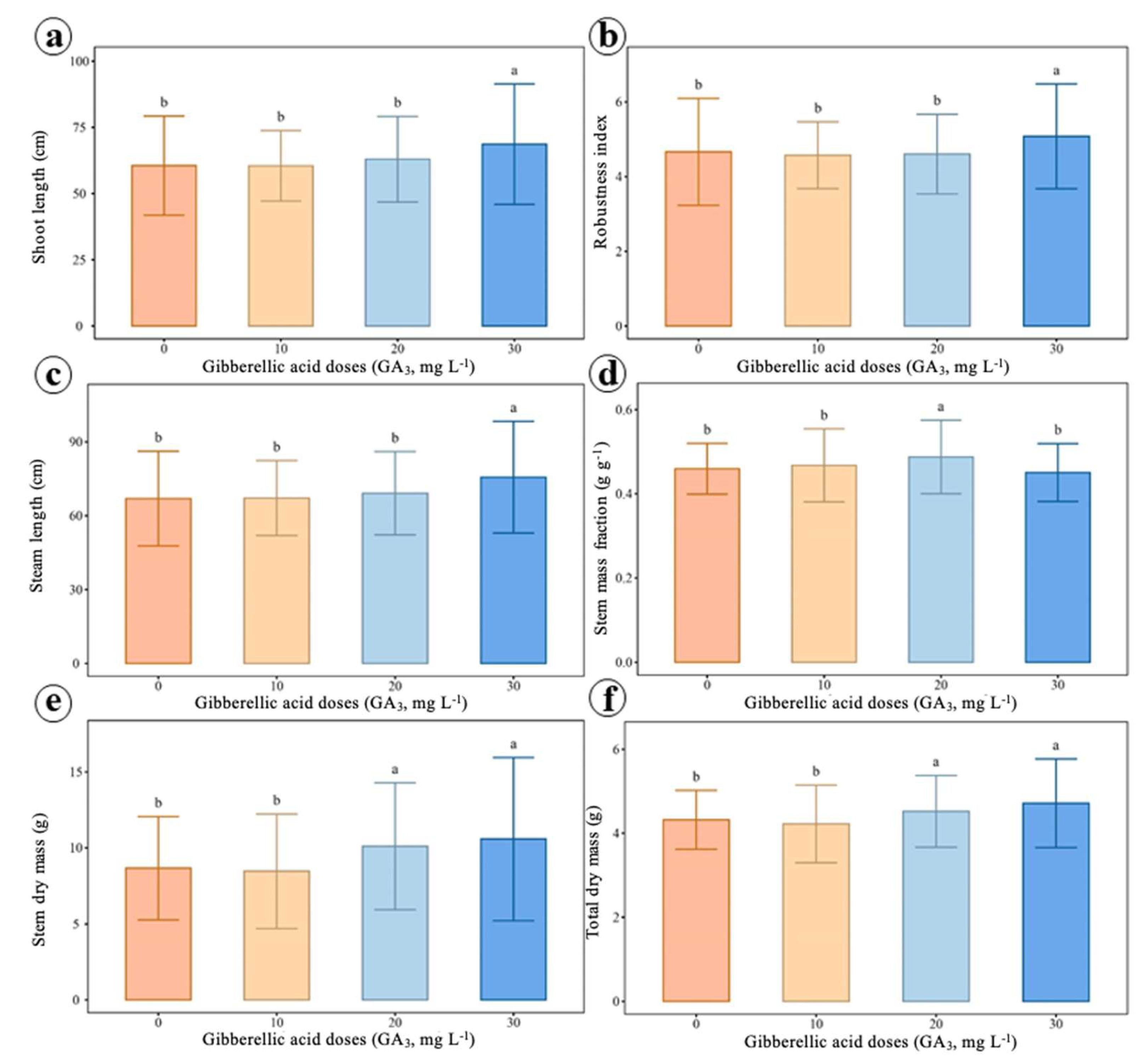
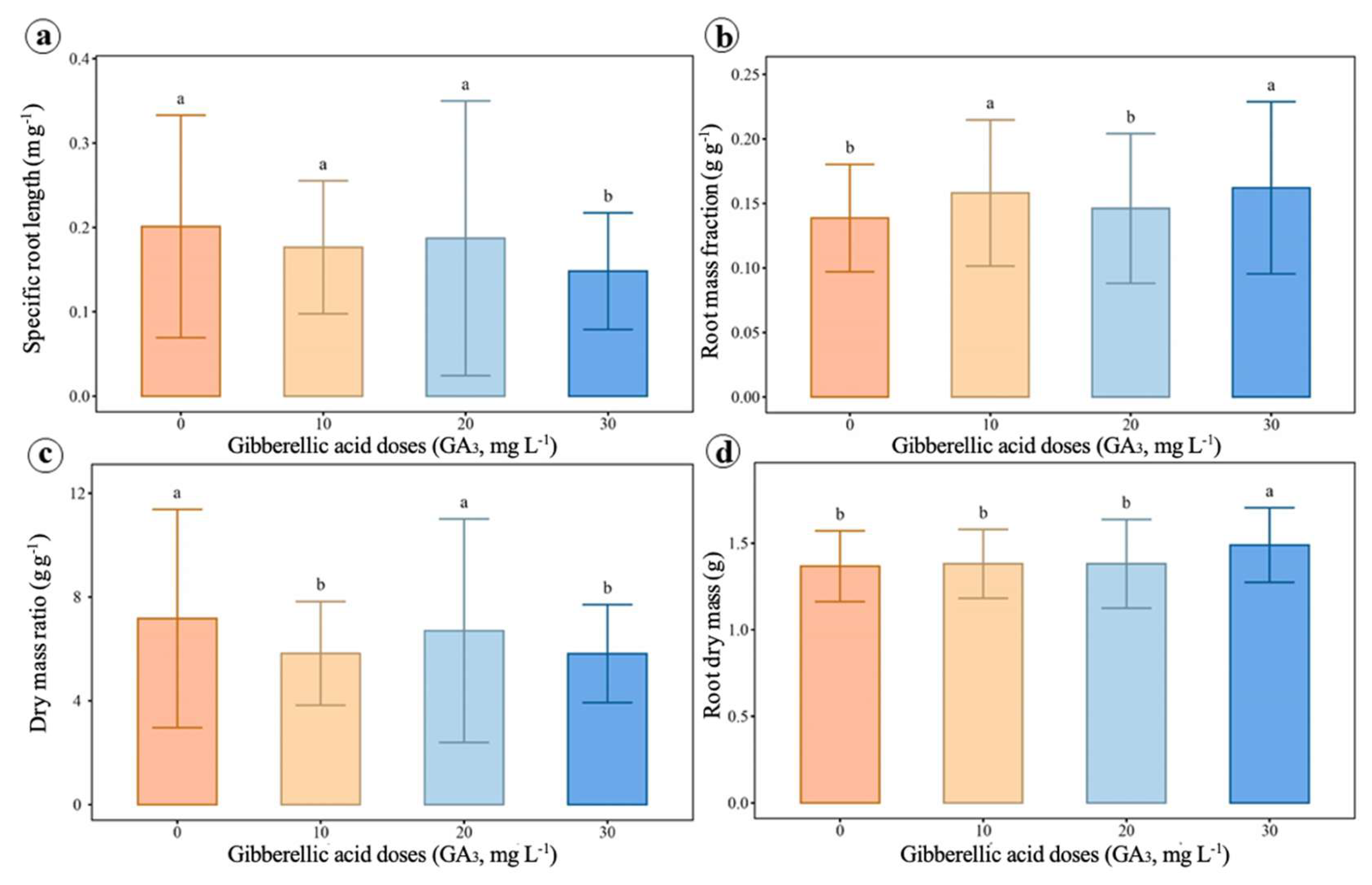
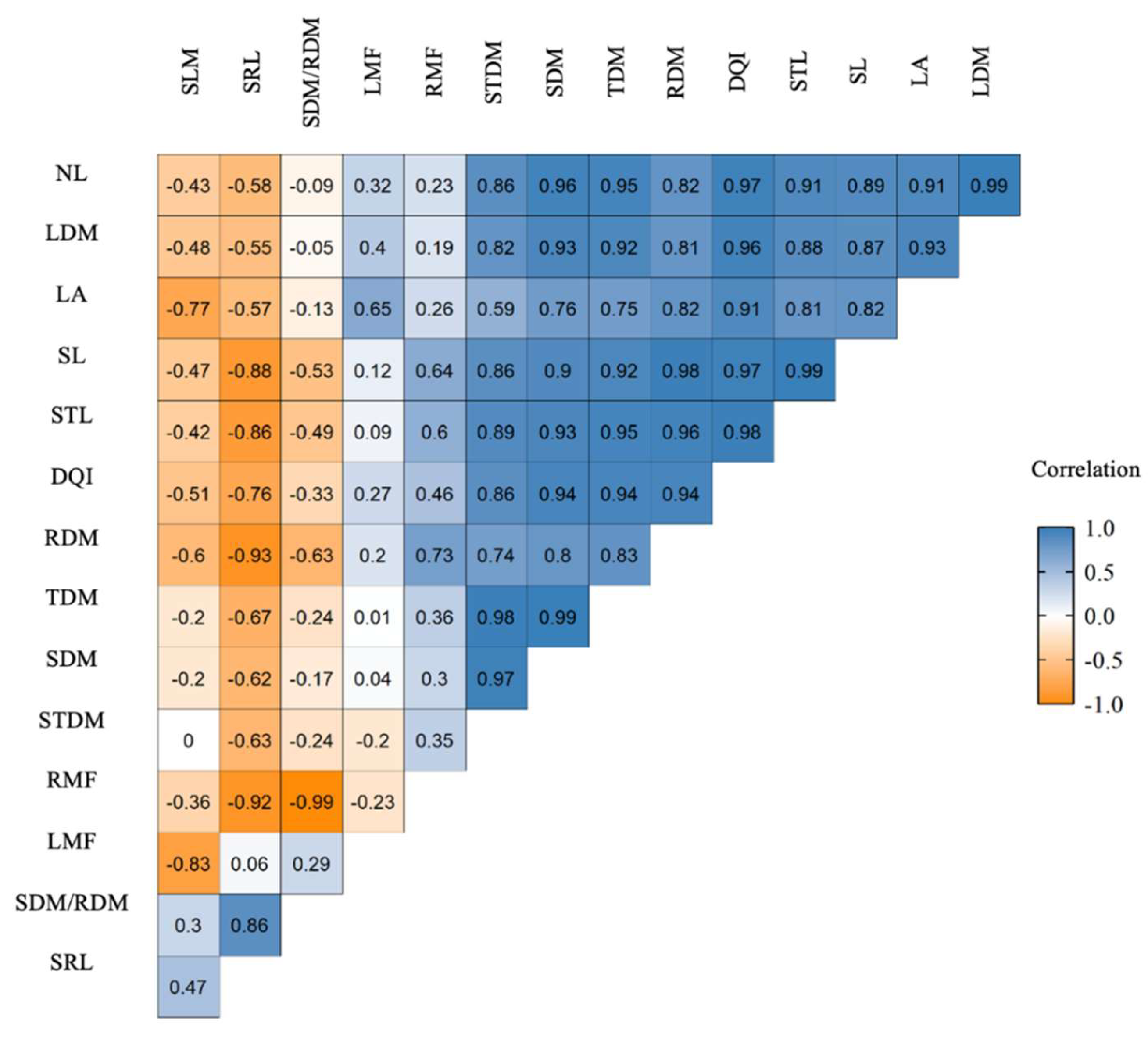
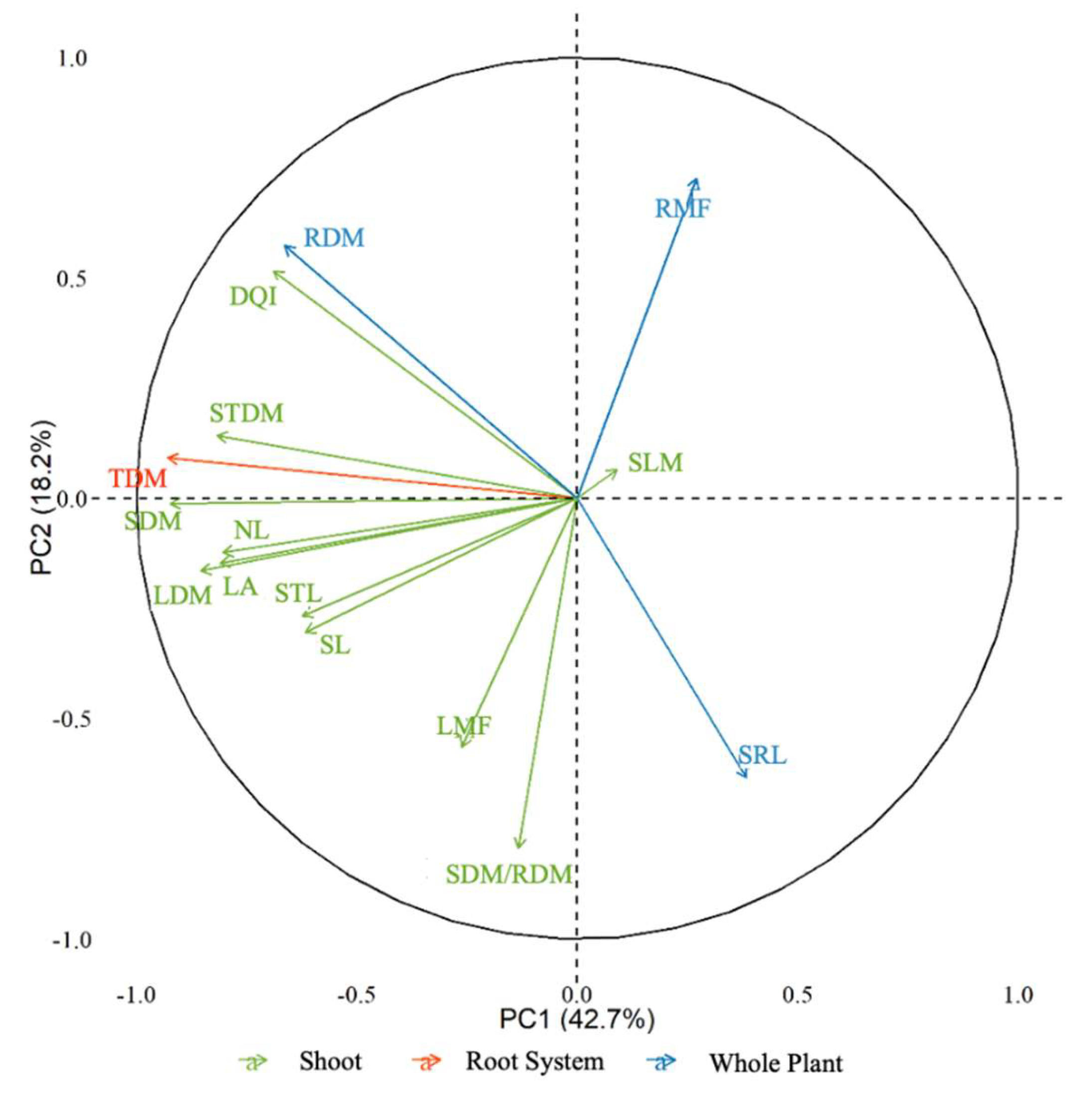
2.4. Growth and Biomass Allocation Analyses
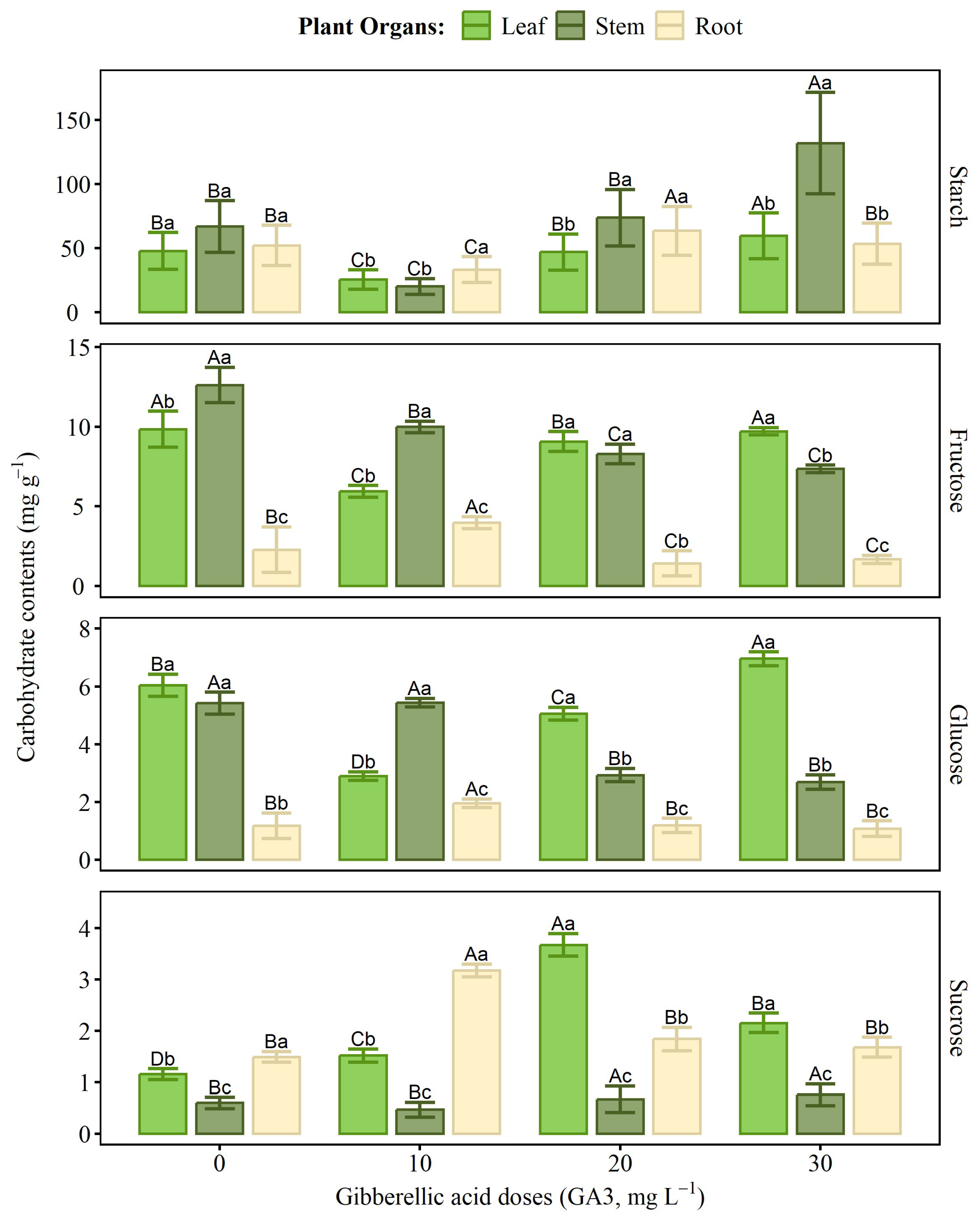
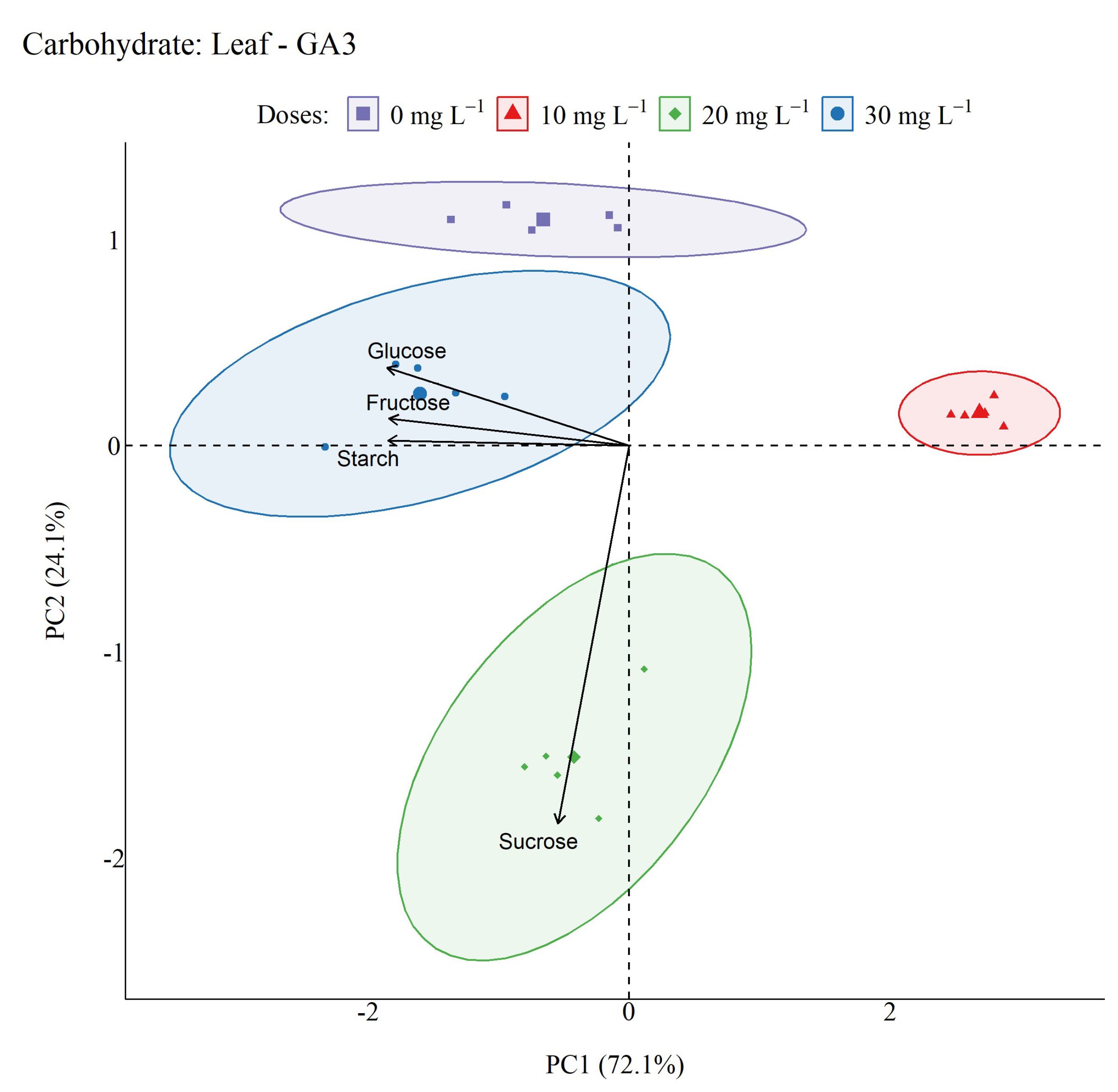
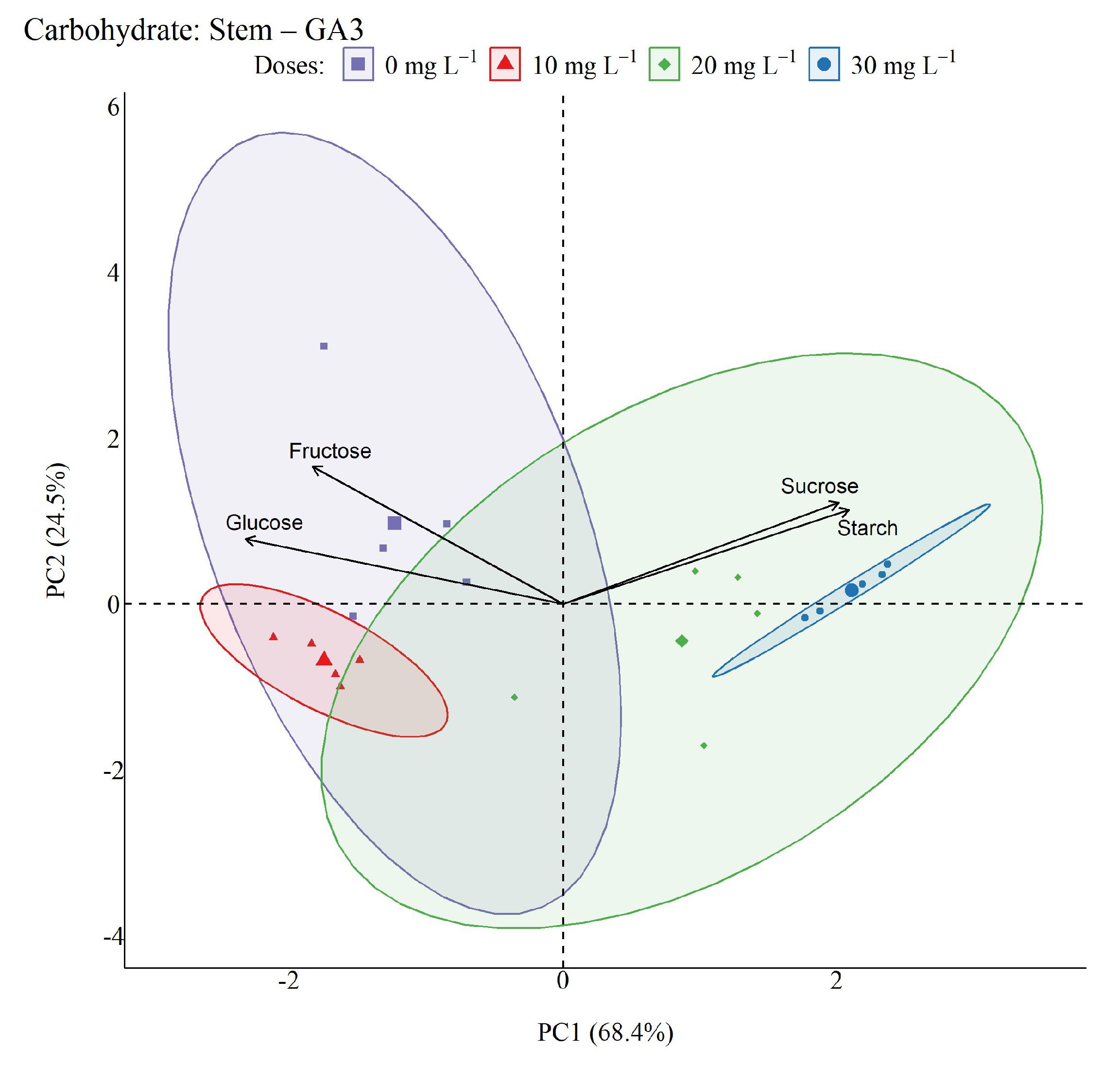
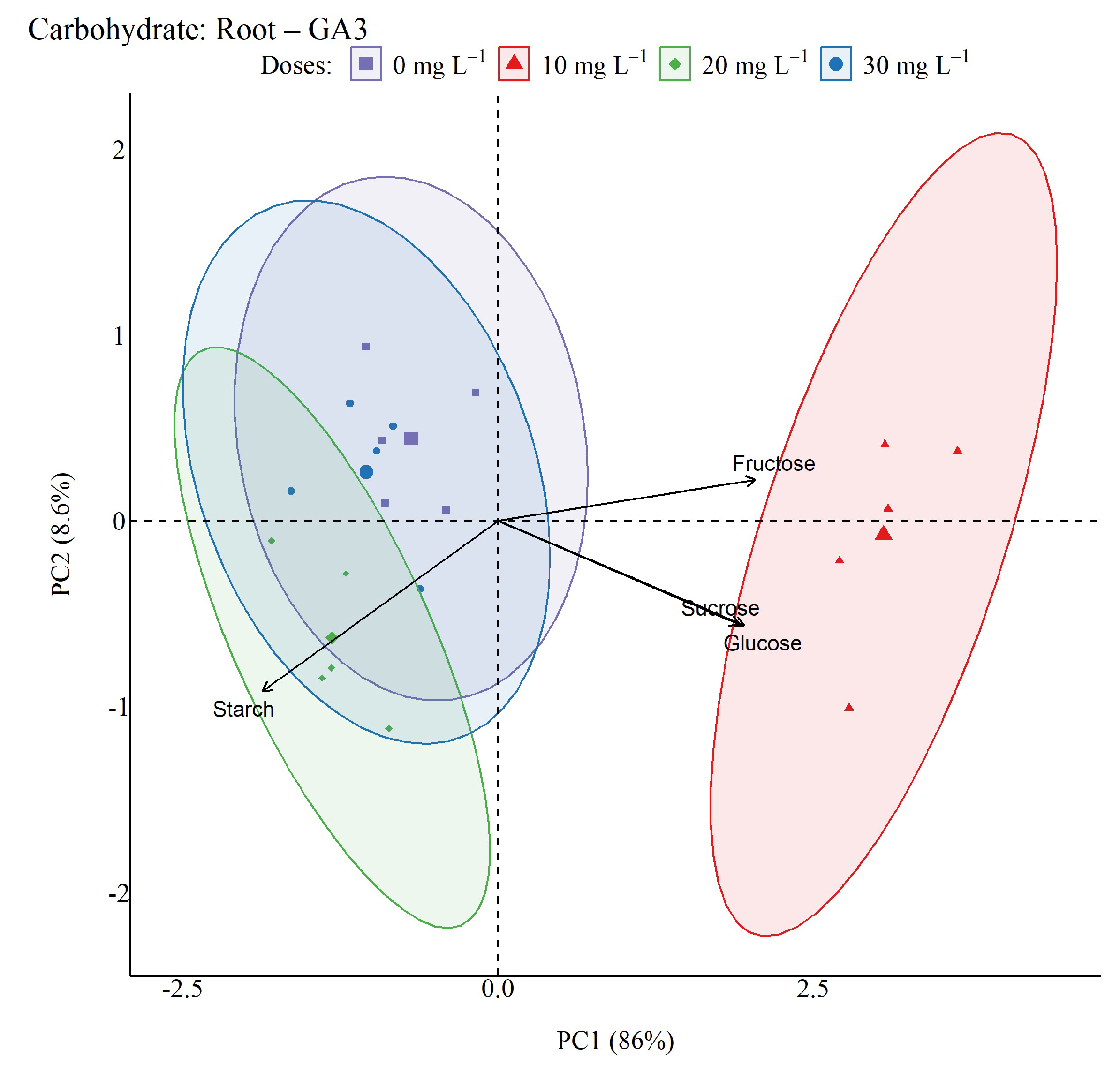
2.5. Non-Structural Carbohydrates
3. Discussion
4. Materials and Methods
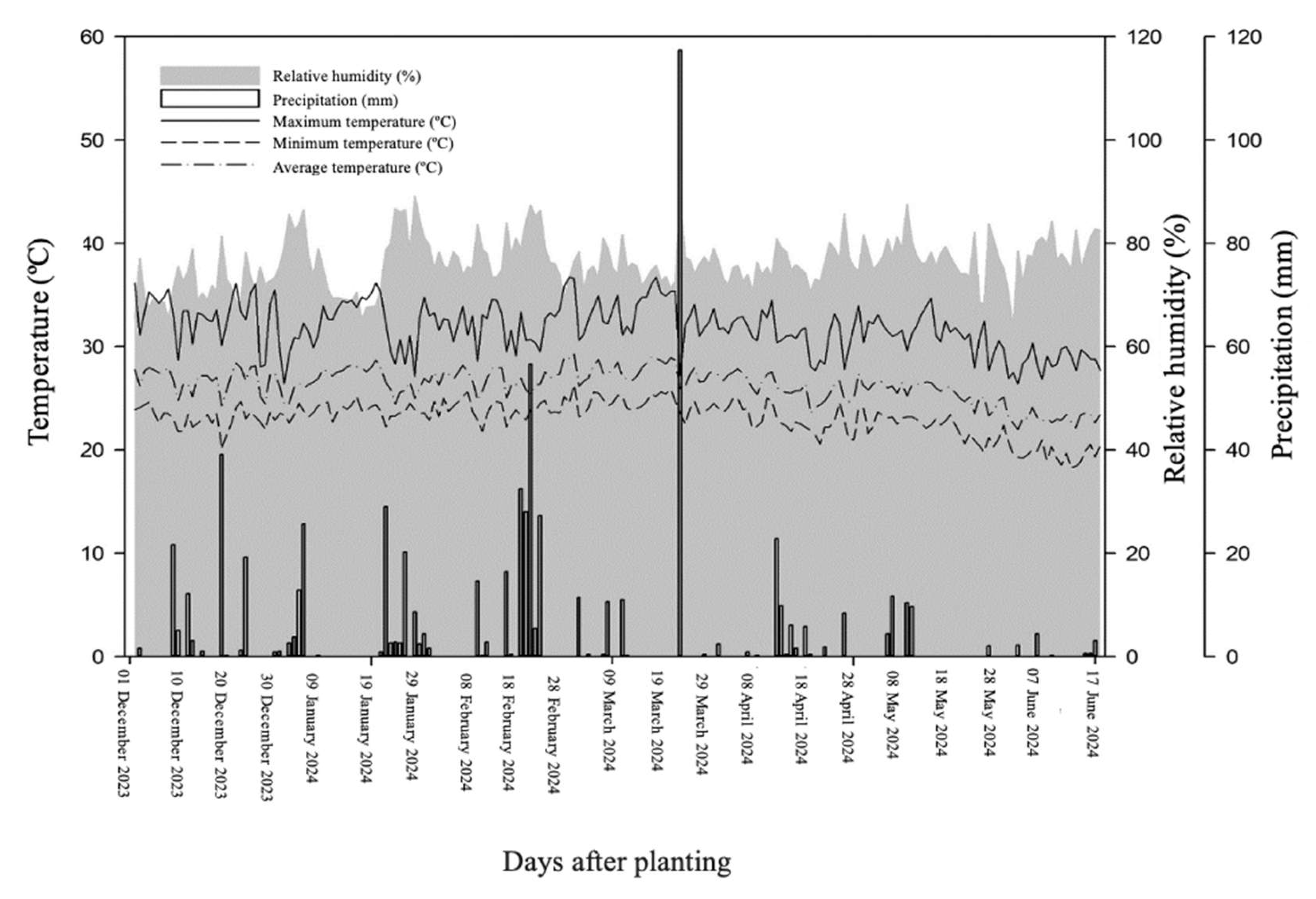
4.1. Experimental Area, Design, and Growing Conditions
4.2. Flowering and Phenological Classification
4.3. Chlorophyll a Fluorescence
4.4. Chlorophyll Index
4.5. Growth and Biomass Allocation Analysis
4.6. Non-structural Carbohydrates
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| ABS/CS0 | Energy absorption per reaction center |
| DI0/CS0 | Energy dissipation per reaction center |
| TR0/CS0 | Energy capture by the reaction center |
| ET0/CS0 | Electron transport rate beyond QA⁻ |
| RE0/CS0 | Reduction of final acceptors in the electron transport chain |
| φP0 | Quantum efficiency of primary photochemistry |
| φE0 | Quantum efficiency of electron transport |
| NL | Number of leaves |
| LDM | Leaf dry mass |
| LA | Leaf area |
| SL | Stem length |
| STL | Shoot length |
| IQD | Dickson quality index |
| RDM | Root dry mass |
| TDM | Total dry mass |
| SDM | Shoot dry mass |
| STDM | Stem dry mass |
| RMF | Root mass fraction |
| LMF | Leaf mass fraction |
| SRL | Specific root length |
| SD | Stem diameter |
| SLA | Specific leaf area |
| SLM | Stem mass fraction |
| RMF | Root mass fraction |
| SRL | Specific root length |
| RL | Root length |
| RTD | Root tissue density |
| FRV | Fresh root volume |
| RI | Robustness index |
| SSL | The specific shoot length |
Appendix A
Appendix A.1
| Months of evaluation | Cycle duration | Number of applications | ||
| Start | End | |||
| 1 | December to February | 01/12/2023 | 17/02/2024 | 1 |
| 2 | February to May | 18/02/2024 | a 18/05/2024 | 1 |
| Parameter | Formulas | Description |
| ABS/CS0 | Chl/CS | Energy absorption per reaction center |
| DI0/CS0 | ABS/RC – TR0/RC | Energy dissipation per reaction center |
| TR0/CS0 | ϕP0 (ABS/CS) | Energy capture by the reaction center |
| ET0/CS0 | ϕP0*Ψ0*(ABS/CS) | Electron transport rate beyond QA⁻ |
| RE0/CS0 | (RE0/ET0) – ET0/CS0) | Reduction of final acceptors in the electron transport chain |
| φP0 | TR0/ABS = [1 − (F0/Fm)] = FV/Fm | Quantum efficiency of primary photochemistry |
| φE0 | ET0/ABS = [1 – (F0/Fm)] ψ0 | Quantum efficiency of electron transport |
References
- Fao; Food and Agriculture Organization of the United Nations. FAOSTAT: Statistical Database. 2023. Available online: https://www.fao.org/faostat/en/ (accessed on 28 January 2025).
- Ibge. Produção Agrícola Municipal. Available online: https://sidra.ibge.gov.br/pesquisa/pam/tabelas (accessed on 04 February 2025).
- Khew, C.-Y.; Mori, I.C.; Matsuura, T.; Hirayama, T.; Harikrishna, J.A.; Lau, E-T.; Mercer, Z.J.A.; Hwang, S-S. Hormonal and transcriptional analyses of fruit development and ripening in different varieties of black pepper (Piper nigrum). J Plant Res. 2020, 133. [Google Scholar] [CrossRef] [PubMed]
- Teles, G.C.; Medici, L.O.; Valença, D.C.; Cruz, E.S.; Carvalho, D.F. Morphophysiological changes in black pepper under different water supplies. Acta Sci.-Agron. 2023, 45. [Google Scholar] [CrossRef]
- Wahyuni, S.; Susilawati, M.; Sirait, N.; Bermawie, N. Floral Morphological Variation in Black Pepper (Piper nigrum L.) Varieties and Hybrid Lines. Planta Tropika: Jurnal Agrosains. 2024, 12, 2. [Google Scholar] [CrossRef]
- Verma, V.M. Black Pepper: Health enefits, In Vitro Multiplication, and Commercial Cultivation; Springer: Cham, 2019. [Google Scholar] [CrossRef]
- Krishnamurthy, K.S.; Ankegowda, S.J.; Umadevi, P.; George, J.K. Black pepper and water stress; Springer: New Delhi, 2016; p. 2. [Google Scholar] [CrossRef]
- Zhang, J.; Cao, Y.; Tang, J.; He, X.; Li, M.; Li, C.; Ren, X.; Ding, Y. Physiology and Application of Gibberellins in Postharvest Horticultural Crops. Horticulturae 2023, 9, 6. [Google Scholar] [CrossRef]
- Shah, S.Z.H.; Kumar, R.; Bakshi, P.; Bhat, D.J.; Sinha, B.K.; Sharma, M.; Sharma, R. Influence of Gibberellic Acid on Fruit Crops: A Review. International Journal of Enviornment and Climate Change. 2023, 13, 8. [Google Scholar] [CrossRef]
- Singh, R.K.; Meena, M.L.; Verma, S.; Vilas, R.; Saurabh, V.; Mauriya, S.K.; Maurya, R.K.; Kumar, M. A review on performance of gibberellic acid on African marigold. Journal of Pharmacognosy and Phytochemistry. 2019, 8, 5. Available online: https://www.phytojournal.com/archives/2019.v8.i5.9521/a-review-on-performance-of-gibberellic-acid-on-african-marigold.
- Li, W.-F.; Zhou, Q.; Ma, Z.; Zuo, C.-W.; Chu, M.-Y.; Mao, J.; Chen, B. Regulatory mechanism of GA3 application on grape (Vitis vinifera L.) berry size. Plant Physiology and Biochemistry. 2024, 210, 108543. [Google Scholar] [CrossRef]
- Sharma, A.; Dilta, B.; Sharma, C.L.; Kumar, M.; Sharma, S. Effect of Foliar Application of Gibberellic Acid on Plant Growth, Flowering and Yield Attributes in Pansy (Viola × wittrockiana Gams.). International Journal of Plant and Soil Science. 2023, 35, 17. [Google Scholar] [CrossRef]
- Prodhan, Md.M.H.; Sarker, U.; Hoque, Md. A.; Biswas, Md. S.; Ercisli, S.; Assouguem, A.; Ullah, R.; Almutairi, M.; Hamad Mohamed, H.R.; Najda, A. Foliar Application of GA3 Stimulates Seed Production in Cauliflower. Agronomy. 2022, 12, 6. [Google Scholar] [CrossRef]
- Haifaa, M.D.I.; Moses, C.K. Effects of Foliar and Soil Application of Gibberellic Acid (GA3) at Different Growth Stages on Agronomic Traits and Yield of Rice (Oryza sativa L.). Journal of Agricultural Science. 2022, 14, 6. [Google Scholar] [CrossRef]
- Murcia, G.; Pontin, M.; Pontin, M.; Reinoso, H.; Baraldi, R.; Bertazza, G.; Gomez-Talquenca, S.; Bottini, R.; Piccoli, P. ABA and GA3 increase carbon allocation in different organs of grapevine plants by inducing accumulation of non-structural carbohydrates in leaves, enhancement of phloem area and expression of sugar transporters. Physiologia Plantarum. 2016, 156, 3. [Google Scholar] [CrossRef]
- Hui, J.-A.; Zhou, Y.-J. Effect of GA₃ spraying on the physiological and biochemical indexes of Aglaonema under drought treatment. HortScience. 2024. [Google Scholar] [CrossRef]
- Kahraman, N.D.; Okumus, O. Role of Gibberellic Acid (GA3) in Improving Salt Stress Tolerance of Wheat (Triticum aestivum). Erciyes Tarım ve Hayvan Bilimleri Dergisi. 2024. [Google Scholar] [CrossRef]
- Luo, J.; Wang, X.; Pang, W.; Jiang, J. GA₃-induced SlXTH19 expression enhances cell wall remodeling and plant height in tomatoes. Plants. 2024, 13, 24. [Google Scholar] [CrossRef]
- Pawar, S.S.; Gawankar, M.S.; Gajbhiye, R.; Dhopavkar, R.V.; Dalvi, N.V. Effect of GA₃ and nutrient sprays on growth of kokum (Garcinia indica Choisy) seedlings. Asian Research Journal of Agriculture. 2024, 17, 4. [Google Scholar] [CrossRef]
- Sari, Ö. Determination of the effect of GA₃ applications on plant development, nutrient content change and analysis of root architectural features using ML artificial neural network modelling in Tulipa saxatilis. Folia Horticulturae. 2024. [Google Scholar] [CrossRef]
- Pradeepkumar, C.M.; Chandrashekar, S.Y.; Kavana, G.B.; Supriya, B.V. A review on role and use of gibberellic acid (GA3) in flower production. International Journal of Chemical Studies. 2020. [Google Scholar] [CrossRef]
- Qu, Y.; Chen, X.-L.; Mao, X.; Huang, P.-Z.; Fu, X. Transcriptome Analysis Reveals the Role of GA3 in Regulating the Asynchronism of Floral Bud Differentiation and Development in Heterodichogamous Cyclocarya paliurus (Batal.) Iljinskaja. International Journal of Molecular Sciences. 2022, 23, 12. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Zhao, Y.; Cai, N.; Guan, J.; Liu, Z.; Liu, Z.; Feng, H.; Zhang, Y. Characterization and Transcriptome Analysis Reveal Exogenous GA3 Inhibited Rosette Branching via Altering Auxin Approach in Flowering Chinese Cabbage. Agronomy. 2024, 14, 4. [Google Scholar] [CrossRef]
- Lamb, R.S.; Hill, T.A.; Tan, Q.K.G.; Irish, V.F. Regulation of APETALA3 floral homeotic gene expression by meristem identity genes. Development. 2002, 129, 9. [Google Scholar] [CrossRef]
- Peña, L.; Martín-Trillo, M.; Juárez, J.; Pina, J. A.; Navarro, L.; Martínez-Zapater, J.M. Constitutive expression of Arabidopsis LEAFY or APETALA1 genes in citrus reduces their generation time. Nature biotechnology 2001, 19, 3. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.J.; Li, L.; Wang, S.; Yu, N.; Shan, H.-S.; Shi, Z.; Li, F.H.; Zhong, X.M. Effect of gibberellic acid on photosynthesis and oxidative stress response in maize under weak light conditions. Frontiers in Plant Science 2023, 14. [Google Scholar] [CrossRef] [PubMed]
- Bassi, R.; Dall’osto, L. Dissipation of light energy absorbed in excess: the molecular mechanisms. Annual review of plant biology 2021, 72, 1. [Google Scholar] [CrossRef]
- Yang, G.; Tatara, G. A quantum picture of light-suppressed photosynthetic charge transfer. Chem. Phys. Lett. 2024. [Google Scholar] [CrossRef]
- Qiu, L.; Rongfa, C.; Hanmin, L.; Yegeng, F.; Huang, X.; Junxian, L.; Faqian, X.; Huiwen, Z.; Chongkun, G.; Jianming, W.; Li, Y.-R. Effects of Exogenous GA3 and DPC Treatments on Levels of Endogenous Hormone and Expression of Key Gibberellin Biosynthesis Pathway Genes During Stem Elongation in Sugarcane. Sugar Tech. 2019, 21, 6. [Google Scholar] [CrossRef]
- Müller, M.; Munné-Bosch, S. Hormonal impact on photosynthesis and photoprotection in plants. Plant Physiology 2021, 185, 4. [Google Scholar] [CrossRef]
- Monge, E.; Aguirre, R.; Blanco, A. Application of paclobutrazol and GA3 to adult peach trees: Effects on nutritional status and photosynthetic pigments. Journal of Plant Growth Regulation 1994, 13, 1. [Google Scholar] [CrossRef]
- Cruciol, G.C.D.; Koyanagui, M.T.; Batista, T.B.; Da Silva Binotti, F.F.; Costa, M.L.N. Aplicação de ácido giberélico e paclobutrazol na cultura da soja. Revista de Agricultura Neotropical 2014, 1, 2. [Google Scholar] [CrossRef]
- Boyers, R.E.; Costa, G.; Vizzotto, E.G. Desbaste de flores e frutos de Peach e outros Prunus. J Amer Soci Hort Sci. 2003, 28. [Google Scholar]
- Taiz, L.; Zeiger, E.; Moller, I.M.; Murphy, A. Fisiologia e Desenvolvimento Vegetal, 6 ed.; ArtMed: Porto Alegre, 2017. [Google Scholar]
- Ghorbani, J. M.; Hoseinifard, M.; Allahdadi, I.; Soltani, E. Hormonal priming with BAP and GA₃ induces improving yield and quality of saffron flower through promotion of carbohydrate accumulation in corm. Journal of Plant Growth Regulation 2021, 41. [Google Scholar] [CrossRef]
- Moreno, D.; Berli, F.J.; Piccoli, P.; Bottini, R. Gibberellins and Abscisic Acid Promote Carbon Allocation in Roots and Berries of Grapevines. Journal of Plant Growth Regulation 2011, 30, 2. [Google Scholar] [CrossRef]
- Chandra, A.; Roopendra, K.; Verma, I. Transcriptome analysis of the effect of GA₃ in sugarcane culm. 3 Biotech. 2019, 9, 10. [Google Scholar] [CrossRef]
- Koch, F.; Aisenberg, G.R.; Monteiro, M.A.; Pedó, T.; Zimmer, P.D.; Villela, F.A.; Aumonde, T.Z. Growth of wheat plants submitted to the application of the growth regulator trinexapac-ethyl and vigor of the produced seeds. Agrociencia Uruguay 2017, 21, 1. [Google Scholar] [CrossRef]
- Alvares, C.A.; Stape, J.L.; Sentelhas, P.C.; Gonçalves, J.L.M.; Sparovek, G. Köppen’s climate classification map for Brazil. Meteorologische Zeitschrift 2014, 22, 6. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, W.R.; Cerri Neto, B.; Crasque, J.; Ferreira, T.R.; Souza, T.C.; Falqueto, A.R.; Arantes, L.O.; Dousseau-Arantes, S. Black pepper grafting in Piper wild species. Bragantia 2023, 82, e20230105. [Google Scholar] [CrossRef]
- Alexandre, R.S.; Giles, J.A.D.; Siqueira, A.L.; Rodrigues, J.P.; Araujo, C.P.; Mello, T.; Rosa, T.L.M.; Lopes, J.C. Production of clonal seedlings of black pepper cv. Bragantina under doses of controlled-release fertilizer. Comunicata Scientiae 2022, 13, e3639. [Google Scholar] [CrossRef]
- Silva, T.R.G.; Costa, M.L.A.; Farias, L.R.A.; Santos, M.A.; Rocha, J.J.L.; Silva, J.V. Abiotic factors in plant growth and flowering. Pesquisa, sociedade e desenvolvimento 2021, 10, 4. [Google Scholar] [CrossRef]
- Lekha, S. Molecular analysis of floral meristem identity genes in black pepper (Piper nigrum L.). Tese (Doutorado em Biotecnologia Vegetal) – Department of Plant Biotechnology, College of Agriculture: Vellayani, 2014. Available online: http://14.139.185.57:8080/jspui/bitstream/123456789/1681/1/173351.pdf.
- Poorter, H.; Niklas, K.J.; Reich, P.B.; Oleksyn, J.; Poot, P.; Mommer, L. Biomass allocation to leaves, stems and roots: meta-analyses of interspecific variation and environmental control. New Phytologist 2011, 193, 1. [Google Scholar] [CrossRef]
- Kramer--Walter, K. R.; Bellingham, J.; Millar, T.R.; Smissen, R.D.; Richardson, S.J.; Laughlin, D.C. Root traits are multidimensional: specific root length is independent of root tissue density and plant economy spectrum. Ecology 2016, 104, 5. [Google Scholar] [CrossRef]
- Popp, M.; Lied, W.; Meyer, A. J.; Richter, A.; Schiller, P.; Schwitte, H. Sample pres ervation fordetermination of organic compounds: microwave versusfreeze-drying. J. Exp. Bot. 1996, 47, 303. [Google Scholar] [CrossRef]
- Box, G.E.P.; Hunter, J.S.; Hunter, W.G. Statistics for experimenters: design, innovation, and discovery; John Wiley & Sons, 2005. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




