Submitted:
12 March 2026
Posted:
12 March 2026
You are already at the latest version
Abstract
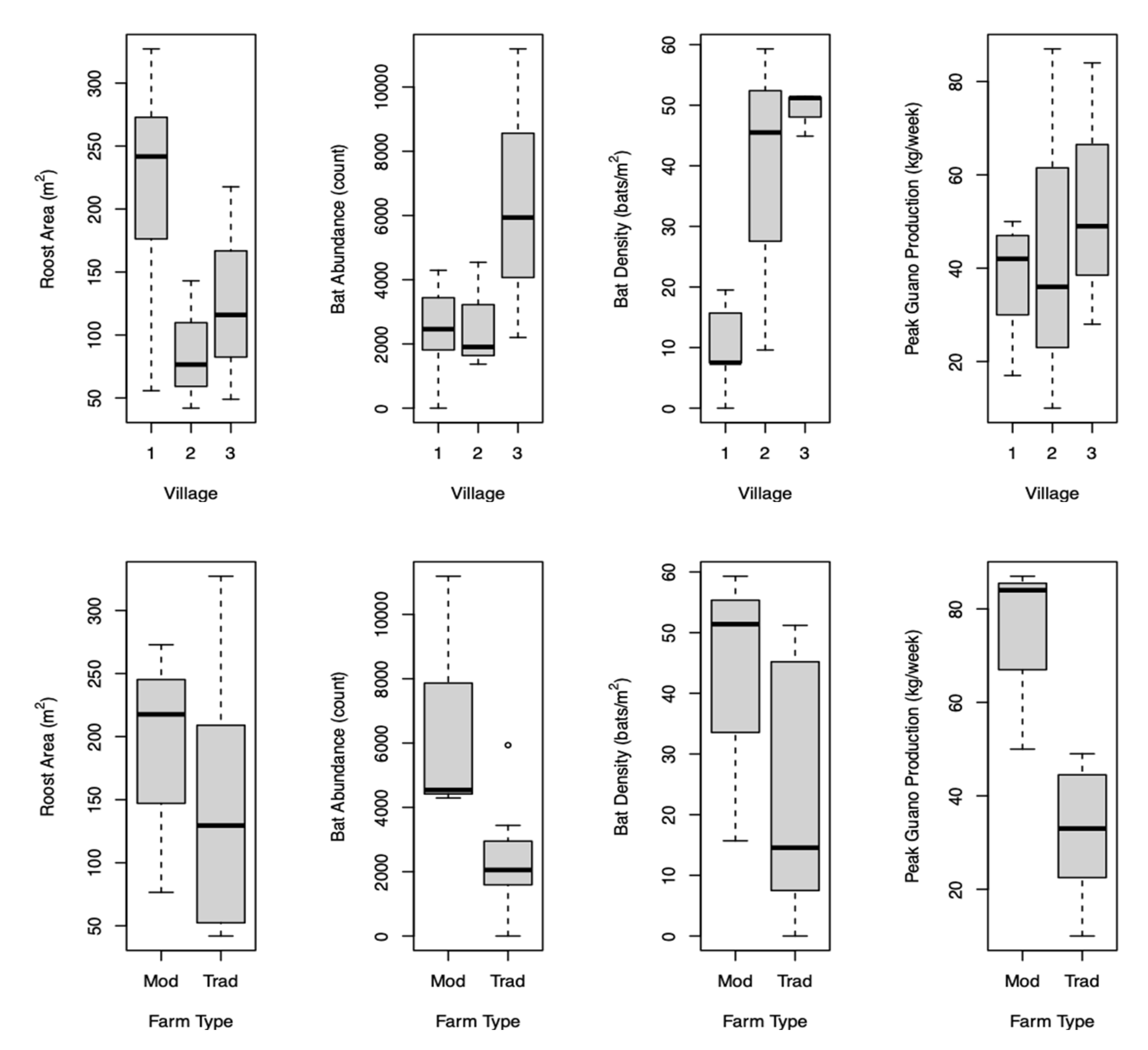
In Cambodia, farmers construct artificial household bat roosts to collect and sell guano as fertilizer. We investigated farming practices and attendant spillover risks using: 1) surveys on guano production; 2) estimating bat population size and species present using carcasses, visual identification, and audio recordings; 3) surveying guano-producing and neighbor households on water, sanitation, and hygiene practices; and 4) testing guano and household food, water and surfaces for coronaviruses by PCR. Bat roosts are constructed using dried palm leaves with coconut tree and/or steel/concrete supports. Roosting areas ranged from 42-327 m2, bat abundance varied from 0-11,187, guano production was 5-120 kg/week, guano yields were 0.15-0.4 kg/m2/week, and farmers earned ~100-200 USD/household/month. Higher guano production in peak (normally wet) season was associated with greater bat abundance (p=0.016). The lesser Asiatic yellow house bat (Scotophilus kuhlii) was the only bat species identified. Roosts were <20 m from guano-producing households. Neighbors and households’ hygiene risks included not having handwashing stations and not covering food in storage/while drying. Alphacoronaviruses or Infectious Bronchitis Virus were found in 14.6%, 17.3%, 2.9%, 1.4%, and 0.0% of guano, urine, surface, food, and water samples, respectively. While guano farming offers economic benefits, spillover risks exist. Safe guano collection and storage, handwashing, and food covering in guano-producing communities are necessary to mitigate spillover risks.
Keywords:
1. Introduction
2. Materials and Methods
3. Results
3.1. Farmer Surveys
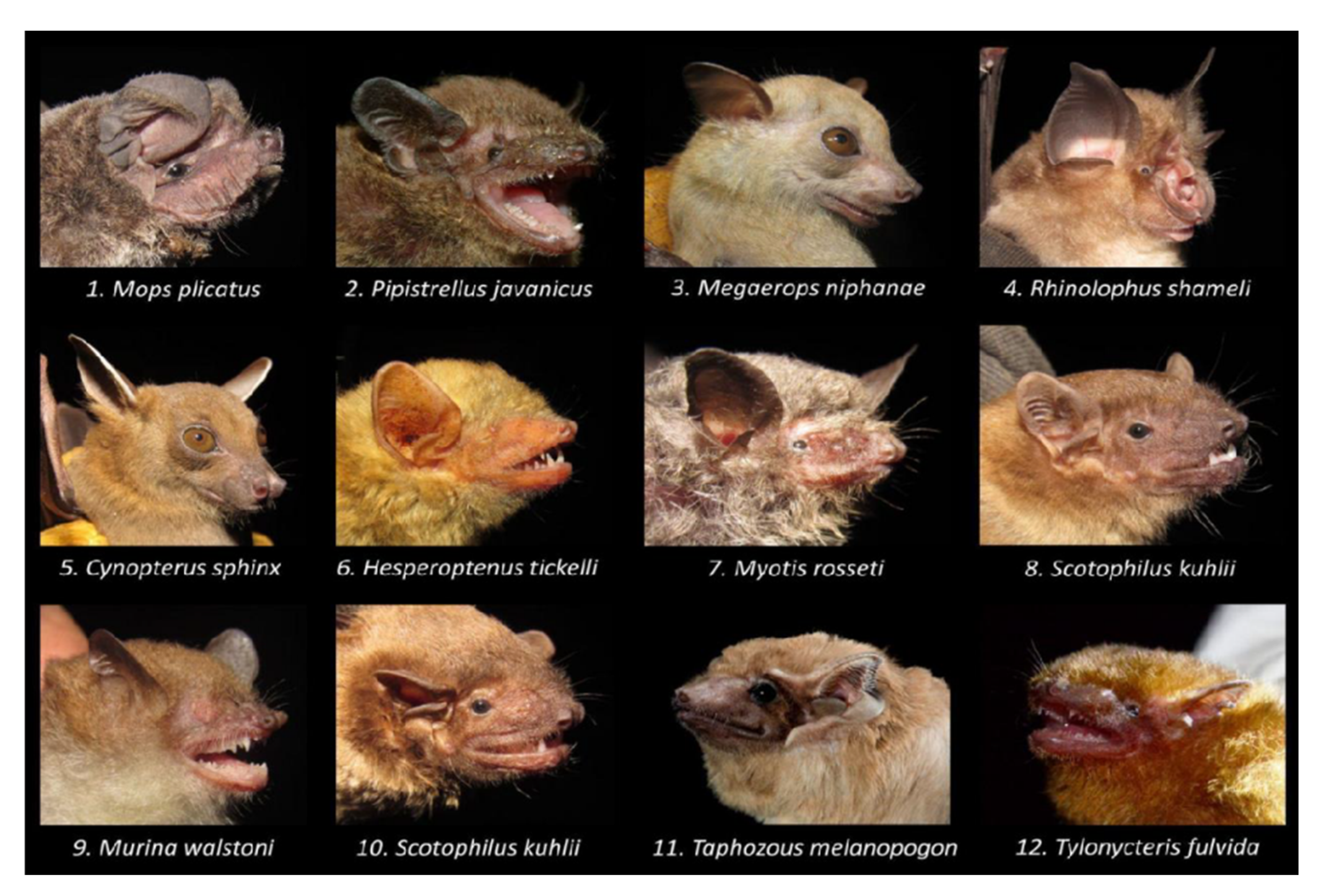
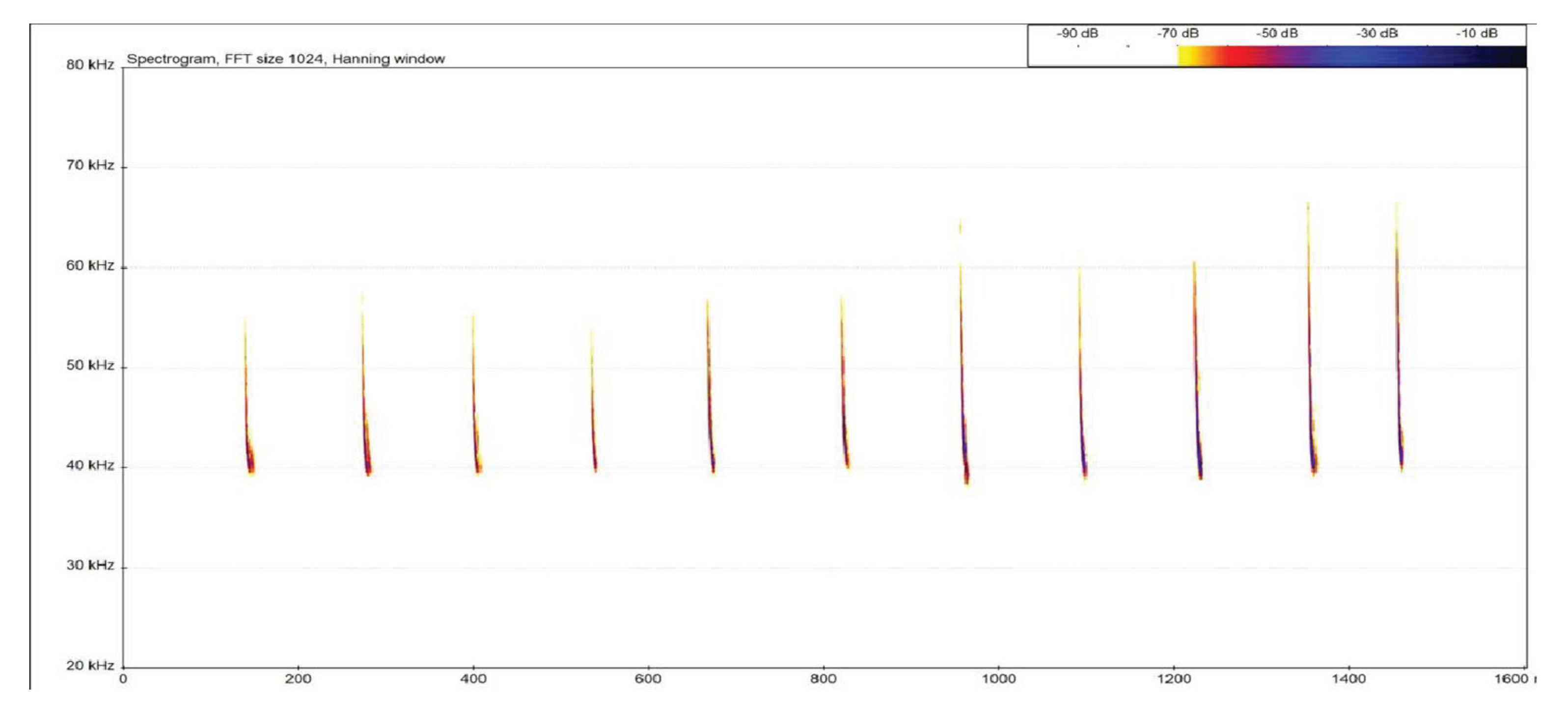
3.2. Bat Species and Behavior
3.3. Food, Water, and Surface Survey
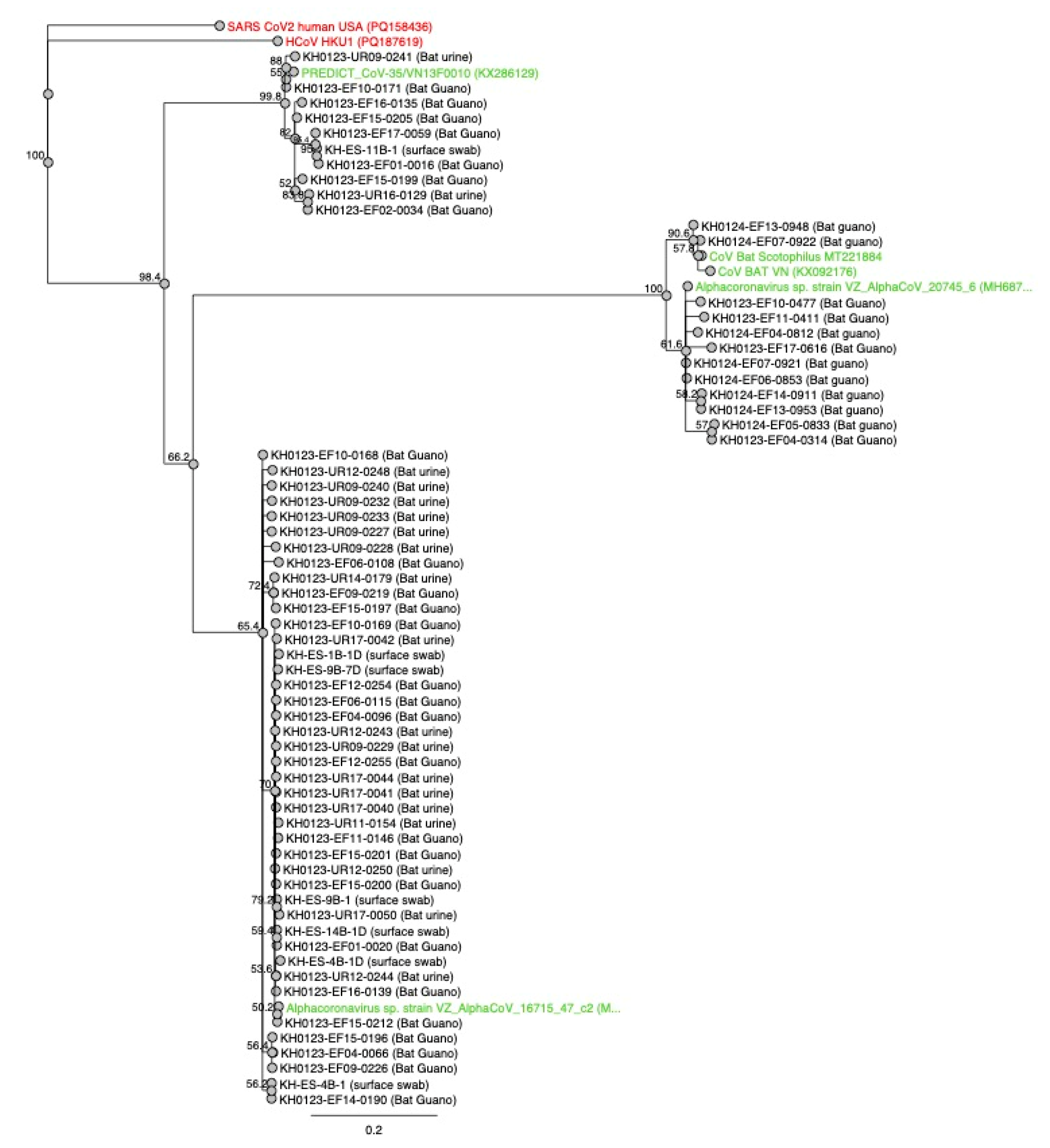
3.4. Guano, Urine, Food, Water, and Surface Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| IBV | Infectious Bronchitis Virus |
| NCBI | National Center for Biotechnology Information |
| PPE | Personal protective equipment |
| PCR | Polymerase chain reaction |
| WASH | Water, sanitation, and hygiene |
Appendix A
| ID Farm | Positive samples of feces and urine N (%) | ||
|---|---|---|---|
| Feces | Urine | Total | |
| 1 | 2 (0.13) | 0 (0.00) | 2 (0.07) |
| 2 | 1 (0.08) | 0 (0.00) | 1 (0.05) |
| 3 | 0 (0.00) | - | 0 (0.00) |
| 4 | 5 (0.14) | 0 (0.00) | 5 (0.10) |
| 5 | 1 (0.13) | - | 1 (0.13) |
| 6 | 3 (0.12) | 0 (0.00) | 3 (0.09) |
| 7 | 2 (0.33) | - | 2 (0.33) |
| 8 | 0 (0.00) | - | 0 (0.00) |
| 9 | 2 (0.12) | 7 (0.44) | 9 (0.27) |
| 10 | 3 (0.17) | 0 (0.00) | 3 (0.13) |
| 11 | 2 (0.09) | 1 (0.14) | 3 (0.10) |
| 12 | 2 (0.13) | 4 (0.40) | 6 (0.24) |
| 13 | 2 (0.50) | - | 2 (0.50) |
| 14 | 2 (0.25) | 1 (0.20) | 3 (0.23) |
| 15 | 7 (0.25) | 0 (0.00) | 7 (0.21) |
| 16 | 2 (0.13) | 1 (0.11) | 3 (0.13) |
| 17 | 2 (0.11) | 5 (0.45) | 7 (0.24) |
| Total | 38 (0.146) | 19 (0.173) | 57 (0.154) |
| No. | Virus Strain | Frequency |
|---|---|---|
| 1 | Alphacoronavirus sp. strain VZ_AlphaCoV_16715_47_c2, complete genome | 18 |
| 2 | Alphacoronavirus sp. strain VZ_AlphaCoV_16715_56, complete genome | 1 |
| 3 | Alphacoronavirus sp. strain VZ_AlphaCoV_16715_61, complete genome | 3 |
| 4 | Alphacoronavirus sp. strain VZ_AlphaCoV_16715_63, complete genome | 2 |
| 5 | Alphacoronavirus sp. strain VZ_AlphaCoV_16715_7, complete genome | 9 |
| 6 | Alphacoronavirus sp. strain VZ_AlphaCoV_16715_77, complete genome | 2 |
| 7 | Alphacoronavirus sp. strain VZ_AlphaCoV_16715_86, complete genome | 3 |
| 8 | Alphacoronavirus sp. strain VZ_AlphaCoV_16845_47, complete genome | 13 |
| 9 | Alphacoronavirus sp. strain VZ_AlphaCoV_17819_22, complete genome | 1 |
| 10 | Coronaviridae sp. isolate 75-55-L06-R2-CoV-BAT-VN RdRp gene, partial cds | 1 |
| 11 | Coronavirus PREDICT CoV-35 PREDICT_CoV-35/VN13F0010 F3R RNA-dependent RNA polymerase mRNA, partial cds | 2 |
| 12 | Scotophilus bat coronavirus 512 2005/PREDICT_VN13F0043 RNA-dependent RNA polymerase mRNA, partial cds | 1 |
| 13 | Scotophilus bat coronavirus 512 2005/PREDICT_VN13F0204 F1R RNA-dependent RNA polymerase mRNA, partial cds | 1 |
| Total | 57 | |
References
- Sakoui, S.; Derdak, R.; Addoum, B.; Serrano-Delgado, A.; Soukri, A.; El Khalfi, B. The Life Hidden Inside Caves: Ecological and Economic Importance of Bat Guano. International Journal of Ecology 2020. [Google Scholar] [CrossRef]
- Kunz, T.H.; de Torrez, E.B.; Bauer, D.; Lobova, T.; Fleming, T.H. Ecosystem services provided by bats. Ann N Y Acad Sci 2011, 1223, 1–38. [Google Scholar] [CrossRef] [PubMed]
- FAO. Investigating the Role of Bats in Emerging Zoonoses: Balancing ecology, conservation and public health interest, in Animal Production and Health Manual; Food and Agriculture Organization of the United Nations: Rome, Italy, 2011. [Google Scholar]
- Furey, N.M.; Racey, P.A. Conservation Ecology of Cave Bats, in Bats in the Anthropocene: Conservation of Bats in a Changing World; Voigt, C.C., Kingston, T., Eds.; Springer International Publishing: Cham, 2016; pp. 463–500. [Google Scholar]
- Kasso, M.; Balakrishnan, M. Ecological and Economic Importance of Bats (Order Chiroptera). International Scholarly Research Notices 2013. [Google Scholar] [CrossRef]
- Francis, C. A Guide to the Mammals of South-East Asia; Princeton University Press: Princeton, New Jersey, and Oxford, United Kingdom, 2008; Vol. 90. [Google Scholar]
- Huong, N.Q.; Nga, N.T.T.; Long, N.V.; Luu, B.D.; Latinne, A.; Pruvot, M.; Phuong, N.T.; Quang, L.T.V.; Hung, V.V.; Lan, N.T.; Hoa, N.T.; Minh, P.Q.; Diep, N.T.; Tung, N.; Ky, V.D.; Roberton, S.I.; Thuy, H.B.; Long, N.V.; Gilbert, M.; Wicker, L.; Mazet, J.A.K.; Johnson, C.K.; Goldstein, T.; Tremeau-Bravard, A.; Ontiveros, V.; Joly, D.O.; Walzer, C.; Fine, A.E.; Olson, S.H. Coronavirus testing indicates transmission risk increases along wildlife supply chains for human consumption in Viet Nam, 2013-2014. PLoS One 2020, 15, e0237129. [Google Scholar] [CrossRef] [PubMed]
- Dimkić, I.; Fira, D.; Janakiev, T.; Kabić, J.; Stupar, M.; Nenadić, M.; Unković, N.; Grbić, M.L. The microbiome of bat guano: for what is this knowledge important? Appl Microbiol Biotechnol 2021, 105, 1407–1419. [Google Scholar] [CrossRef]
- Ninsiima, L.R.; Nyakarahuka, L.; Kisaka, S.; Atuheire, C.G.K.; Mugisha, L.; Odoch, T.; Romano, J.S.; Klein, J.; Mor, S.M.; Kankya, C. Knowledge, perceptions, and exposure to bats in communities living around bat roosts in Bundibugyo district, Uganda: implications for viral haemorrhagic fever prevention and control. BMC Infectious Diseases 2024, 24, 311. [Google Scholar] [CrossRef]
- Robertson, K.; Lumlertdacha, B.; Franka, R.; Petersen, B.; Bhengsri, S.; Henchaichon, S.; Peruski, L.F.; Baggett, H.C.; Maloney, S.A.; Rupprecht, C.E. Rabies-related knowledge and practices among persons at risk of bat exposures in Thailand. PLoS Negl Trop Dis 2011, 5, e1054. [Google Scholar] [CrossRef]
- Shapiro, H.G.; Willcox, A.S.; Ader, D.R.; Willcox, E.V. Attitudes towards and Relationships with Cave-Roosting Bats in Northwest Cambodia. Journal of Ethnobiology 2021, 41, 87–104. [Google Scholar] [CrossRef]
- Furey, N.M.; Tu, V.T.; Hitch, A.T.; Pimsai, A.; Kimashalen, C.; Vuthy, B.; Raksmey, Y.; Sarak, C.; Borthwick, S.A.; Ch, L.; Sinly, S.; Csorba, G.; Saveng, I.; Smith, G.J.D.; Dany, C.; Mendenhall, I.H. First Records of Seemingly Rare Bats (Mammalia: Chiroptera) in Cambodia, with a Revised Checklist of Species for the Country. Acta Chiropterologica 2022, 23, 345–369, 25. [Google Scholar] [CrossRef]
- Nguyen, H.T.T.; Lindahl, J.F.; Bett, B.; Nguyen-Viet, H.; Lâm, S.; Nguyen-Tien, T.; Unger, F.; Dang-Xuan, S.; Bui, T.X.; Le, H.T.; Lundkvist, Å.; Ling, J.; Lee, H.S. Understanding zoonotic pathogens and risk factors from wildlife in Southeast Asia: a systematic literature review. Veterinary Quarterly 2025, 45, 1–17. [Google Scholar] [CrossRef]
- Cappelle, J.; Hoem, T.; Hul, V.; Furey, N.; Nguon, K.; Prigent, S.; Dupon, L.; Ken, S.; Neung, C.; Hok, V. Nipah virus circulation at human–bat interfaces, Cambodia. Bulletin of the World Health Organization 2020, 98, 539. [Google Scholar] [CrossRef]
- Cappelle, J.; Furey, N.; Hoem, T.; Ou, T.P.; Lim, T.; Hul, V.; Heng, O.; Chevalier, V.; Dussart, P.; Duong, V. Longitudinal monitoring in Cambodia suggests higher circulation of alpha and betacoronaviruses in juvenile and immature bats of three species. Scientific Reports 2021, 11, 24145. [Google Scholar] [CrossRef]
- Drexler, J.F.; Corman, V.M.; Wegner, T.; Tateno, A.F.; Zerbinati, R.M.; Gloza-Rausch, F.; Seebens, A.; Müller, M.; Drosten, C. Amplification of Emerging Viruses in a Bat Colony. Emerging Infectious Disease journal 2011, 17, 449. [Google Scholar] [CrossRef]
- Delaune, D.; Hul, V.; Karlsson, E.A.; Hassanin, A.; Ou, T.P.; Baidaliuk, A.; Gámbaro, F.; Prot, M.; Tu, V.T.; Chea, S.; Keatts, L.; Mazet, J.; Johnson, C.K.; Buchy, P.; Dussart, P.; Goldstein, T.; Simon-Lorière, E.; Duong, V. A novel SARS-CoV-2 related coronavirus in bats from Cambodia. Nat Commun 2021, 12, 6563. [Google Scholar] [CrossRef]
- Sánchez, C.A.; Li, H.; Phelps, K.L.; Zambrana-Torrelio, C.; Wang, L.-F.; Zhou, P.; Shi, Z.-L.; Olival, K.J.; Daszak, P. A strategy to assess spillover risk of bat SARS-related coronaviruses in Southeast Asia. Nature Communications 2022, 13, 4380. [Google Scholar] [CrossRef]
- Tan, C.S.; Noni, V.; Anwarali-Khan, F.A.; Denel, A.; Sathyiya-Seelan, J.S. The discovery of novel coronaviruses in bat guano, Sarawak, Malaysian Borneo. International Journal of Infectious Diseases 2022, 116, S102–S103. [Google Scholar] [CrossRef]
- NIS, General population census of the kingdom of Cambodia 2019; Ministry of Planning, National Institute of Statistics.
- Jiao, X.; Smith-Hall, C.; Theilade, I. Rural household incomes and land grabbing in Cambodia. Land Use Policy 2015, 48, 317–328. [Google Scholar] [CrossRef]
- Rydell, J.; Entwistle, A.; Racey, P.A. Timing of Foraging Flights of Three Species of Bats in Relation to Insect Activity and Predation Risk. Oikos 1996, 76, 243–252. [Google Scholar] [CrossRef]
- Rohr, J.R.; Barrett, C.B.; Civitello, D.J.; Craft, M.E.; Delius, B.; DeLeo, G.A.; Hudson, P.J.; Jouanard, N.; Nguyen, K.H.; Ostfeld, R.S.; Remais, J.V.; Riveau, G.; Sokolow, S.H.; Tilman, D. Emerging human infectious diseases and the links to global food production. Nat Sustain 2019, 2, 445–456. [Google Scholar] [CrossRef] [PubMed]
- Francis, C. Field Guide to the Mammals of South-east Asia (2nd Edition); Bloomsbury Wildlife, 2019. [Google Scholar]
- Quan, P.-L.; Firth, C.; Street, C.; Jose, A.H.; Petrosov, A.; Tashmukhamedova, A.; Stephen, K.H.; Egholm, M.; Modupe, O.V.O.; Niezgoda, M.; Albert, B.O.; Briese, T.; Charles, E.R.; Lipkin, W.I. Identification of a Severe Acute Respiratory Syndrome Coronavirus-Like Virus in a Leaf-Nosed Bat in Nigeria. mBio 2010, 1. [Google Scholar] [CrossRef]
- Rocha, R.; Fernández-Llamazares, Á.; López-Baucells, A.; Andriamitandrina, S.F.M.; Andriatafika, Z.E.; Temba, E.M.; Torrent, L.; Burgas, D.; Cabeza, M. Human-Bat Interactions in Rural Southwestern Madagascar through a Biocultural Lens. Journal of Ethnobiology 2021, 41, 53–69. [Google Scholar] [CrossRef]
- Shapiro, H.G.; Willcox, A.S.; Tate, M.; Willcox, E.V. Can farmers and bats co-exist? Farmer attitudes, knowledge, and experiences with bats in Belize. Human-Wildlife Interactions 2020, 14, 5–15. [Google Scholar]
- Jenkins, M.W.; Anand, A.R.; Revell, G.; Sobsey, M.D. Opportunities to improve domestic hygiene practices through new enabling products: a study of handwashing practices and equipment in rural Cambodia. International Health 2013, 5, 295–301. [Google Scholar] [CrossRef]
- Osbjer, K.; Boqvist, S.; Sokerya, S.; Kannarath, C.; San, S.; Davun, H.; Magnusson, U. Household practices related to disease transmission between animals and humans in rural Cambodia. BMC Public Health 2015, 15, 476. [Google Scholar] [CrossRef]
- Olival, K.J.; Hosseini, P.R.; Zambrana-Torrelio, C.; Ross, N.; Bogich, T.L.; Daszak, P. Host and viral traits predict zoonotic spillover from mammals. Nature 2017, 546, 646–650. [Google Scholar] [CrossRef]
- Hu, B.; Ge, X.; Wang, L.F.; Shi, Z. Bat origin of human coronaviruses. Virol J 2015, 12, 221. [Google Scholar] [CrossRef]
- Wong, A.C.P.; Li, X.; Lau, S.K.P.; Wo, P.C.Y. Global Epidemiology of Bat Coronaviruses. Viruses 2019, 11(2). [Google Scholar] [CrossRef]
- Li, W.; Shi, Z.; Yu, M.; Ren, W.; Smith, C.; Epstein, J.H.; Wang, H.; Crameri, G.; Hu, Z.; Zhang, H.; Zhang, J.; McEachern, J.; Field, H.; Daszak, P.; Eaton, B.T.; Zhang, S.; Wang, L.F. Bats are natural reservoirs of SARS-like coronaviruses. Science 2005, 310, 676–9. [Google Scholar] [CrossRef] [PubMed]
- Valitutto, M.T.; Aung, O.; Tun, K.Y.N.; Vodzak, M.E.; Zimmerman, D.; Yu, J.H.; Win, Y.T.; Maw, M.T.; Thein, W.Z.; Win, H.H.; Dhanota, J.; Ontiveros, V.; Smith, B.; Tremeau-Brevard, A.; Goldstein, T.; Johnson, C.K.; Murray, S.; Mazet, J. Detection of novel coronaviruses in bats in Myanmar. PLoS One 2020, 15, e0230802. [Google Scholar] [CrossRef]
- Anthony, S.J.; Johnson, C.K.; Greig, D.J.; Kramer, S.; Che, X.; Wells, H.; Hicks, A.L.; Joly, D.O.; Wolfe, N.D.; Daszak, P.; Karesh, W.; Lipkin, W.I.; Morse, S.S.; Consortium, P.; Mazet, J.A.K.; Goldstein, T. Global patterns in coronavirus diversity. Virus Evolution 2017, 3(1). [Google Scholar] [CrossRef]
- Niu, Y.; McKee, C.D. Bat Viral Shedding: A Review of Seasonal Patterns and Risk Factors. Vector Borne Zoonotic Dis 2025, 25, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Wacharapluesadee, S.; Duengkae, P.; Chaiyes, A.; Kaewpom, T.; Rodpan, A.; Yingsakmongkon, S.; Petcharat, S.; Phengsakul, P.; Maneeorn, P.; Hemachudha, T. Longitudinal study of age-specific pattern of coronavirus infection in Lyle’s flying fox (Pteropus lylei) in Thailand. Virology Journal 2018, 15, 38. [Google Scholar] [CrossRef] [PubMed]
- Joffrin, L.; Hoarau, A.O.G.; Lagadec, E.; Torrontegi, O.; Köster, M.; Le Minter, G.; Dietrich, M.; Mavingui, P.; Lebarbenchon, C. Seasonality of coronavirus shedding in tropical bats. R Soc Open Sci 2022, 9, 211600. [Google Scholar] [CrossRef]
- Escudero-Pérez, B.; Lalande, A.; Mathieu, C.; Lawrence, P. Host–Pathogen Interactions Influencing Zoonotic Spillover Potential and Transmission in Humans. Viruses 2023, 15, 599. [Google Scholar] [CrossRef] [PubMed]

| Sample Type | Quantity | Collection Date |
|---|---|---|
| Farmer survey | 17 farms | April-May 2023 |
| Household and neighbor WASH survey | 20 farms | April 2023 |
| Bat abundance | 11 farms | April-May 2023 August 2023 December 2023 March 2024 |
| Bat species identification | 7 carcasses (6 farms) | April-May 2023 |
| Bat feces samples | 261 samples (17 farms) | April-May 2023 August 2023 December 2023 March 2024 |
| Bat urine samples | 219 samples (17 farms) | April-May 2023 August 2023 December 2023 March 2024 |
| Household surface samples | 346 samples (10 farms) | April 2023 |
| Food samples | 70 samples (10 farms) | April 2023 |
| Water samples | 75 samples (10 farms) | April 2023 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




