3.2. Batch Extraction Experiments
Figure 2 compares the Co(II) extraction yields using [Pn
3NC
2OC
2][Dca] with those obtained with [Pn
3NC
2OC
2][Br] under similar conditions.
Figure 3 illustrates the extraction yields of Co(II) and Ni(II) in [Pn
3NC
2OC
2][Dca] dissolved in MIBK, for varying NaCl concentrations.
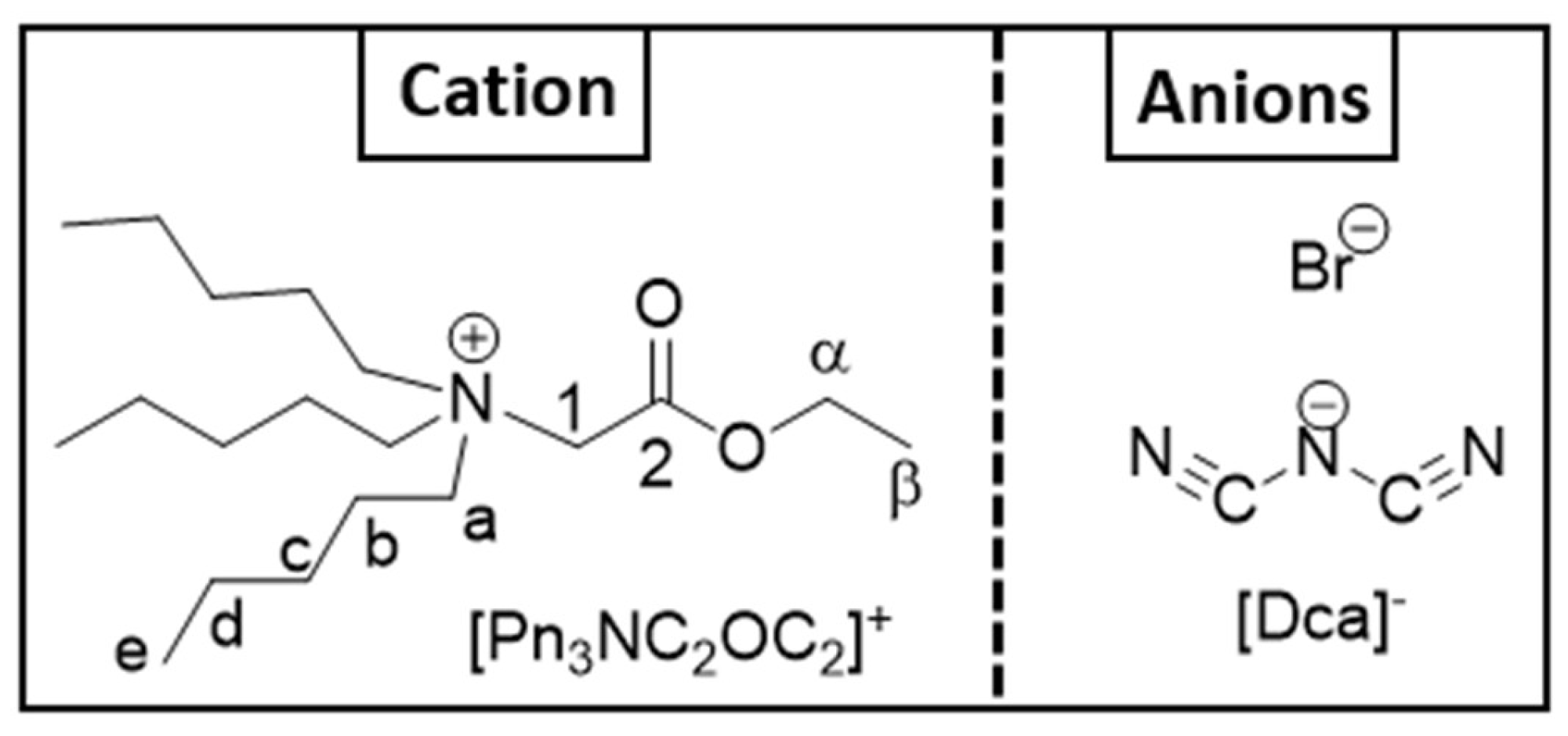
The ionic liquid [Pn3NC2OC2][Dca] was obtained from [Pn3NC2OC2][Br] through an anion-exchange reaction with NaDca. Consequently, [Pn3NC2OC2][Br] is considered the reference compound and serves as a control in this study for both batch and continuous processes.
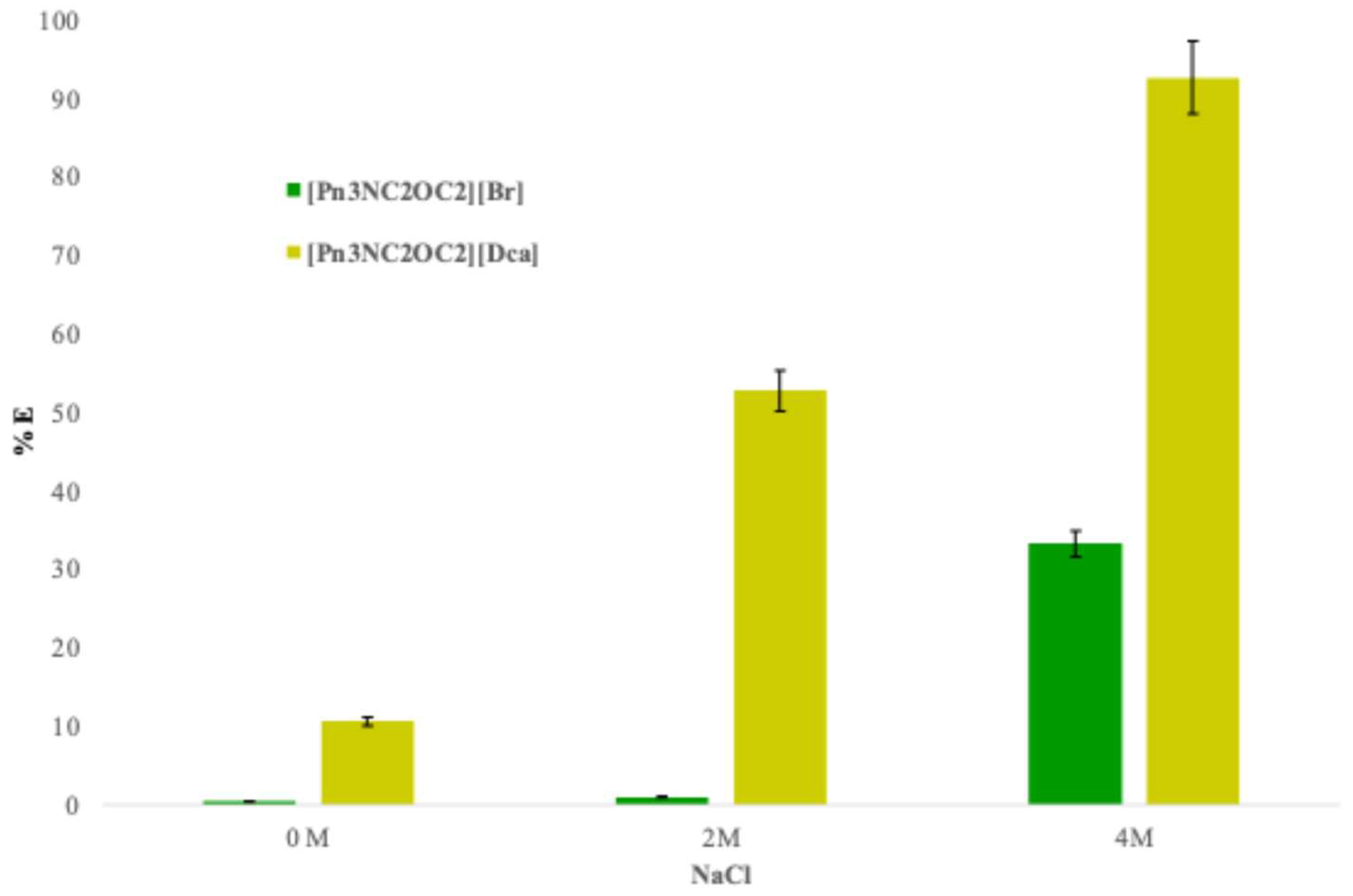
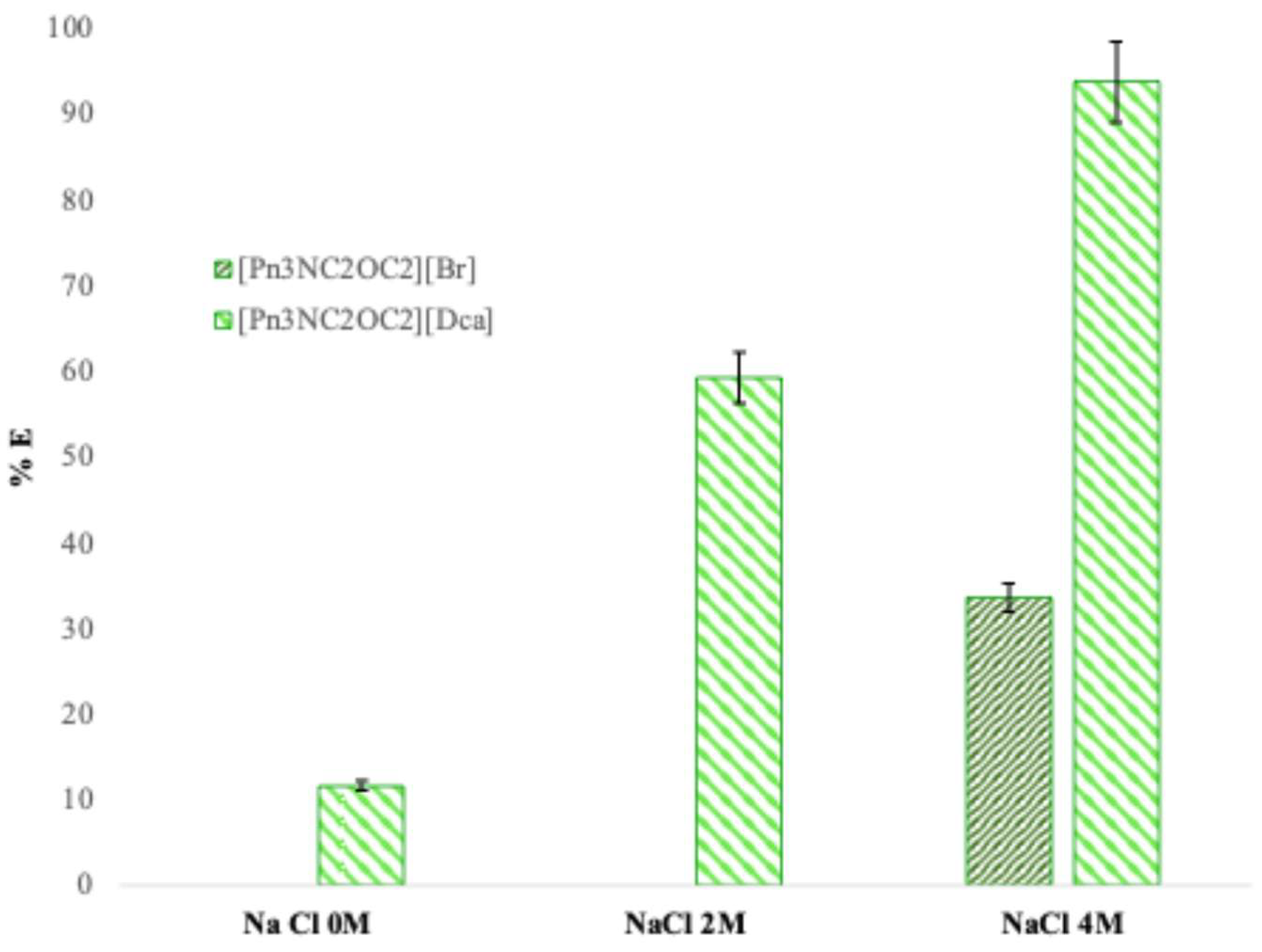
Figure 2 clearly highlights the benefit of performing an anionic metathesis on the brominated compound using a salt containing an anion with higher coordinating ability than bromide. When NaCl concentration increases from 0 to 4 M, the extraction yield rises from 0 to 33% with [Pn
3NC
2OC
2][Br], whereas it increases from 10% to 92% with [Pn
3NC
2OC
2][Dca]. This improvement is attributed to the strong coordinating power of the dicyanamide anion, which enhances the extraction efficiency of the ionic liquid. The dicyanamide anion integrates more readily into the coordination sphere of Co(II) compared to bromide.
For [Pn3NC2OC2][Br], the relatively low extraction in NaCl media below 4 M suggests that the process depends on the formation of the tetrahedral CoCl42- complex, which requires a high concentration of Cl− ions. In contrast, with [Pn3NC2OC2][Dca], the extraction mechanism involves the formation of more stable mixed Co/Dca/Cl complexes, explaining the significantly higher extraction yields.
Figure 3 illustrates that the addition of an electrolyte containing a chaotropic anion in the aqueous phase is a well-established strategy to enhance metal extraction yields [
32]. In a previous study [
32], we demonstrated that hydrophobic glycine-betaine-based Dca ionic liquids are highly effective for the selective separation of Co(II) and Ni(II) in NaCl media.
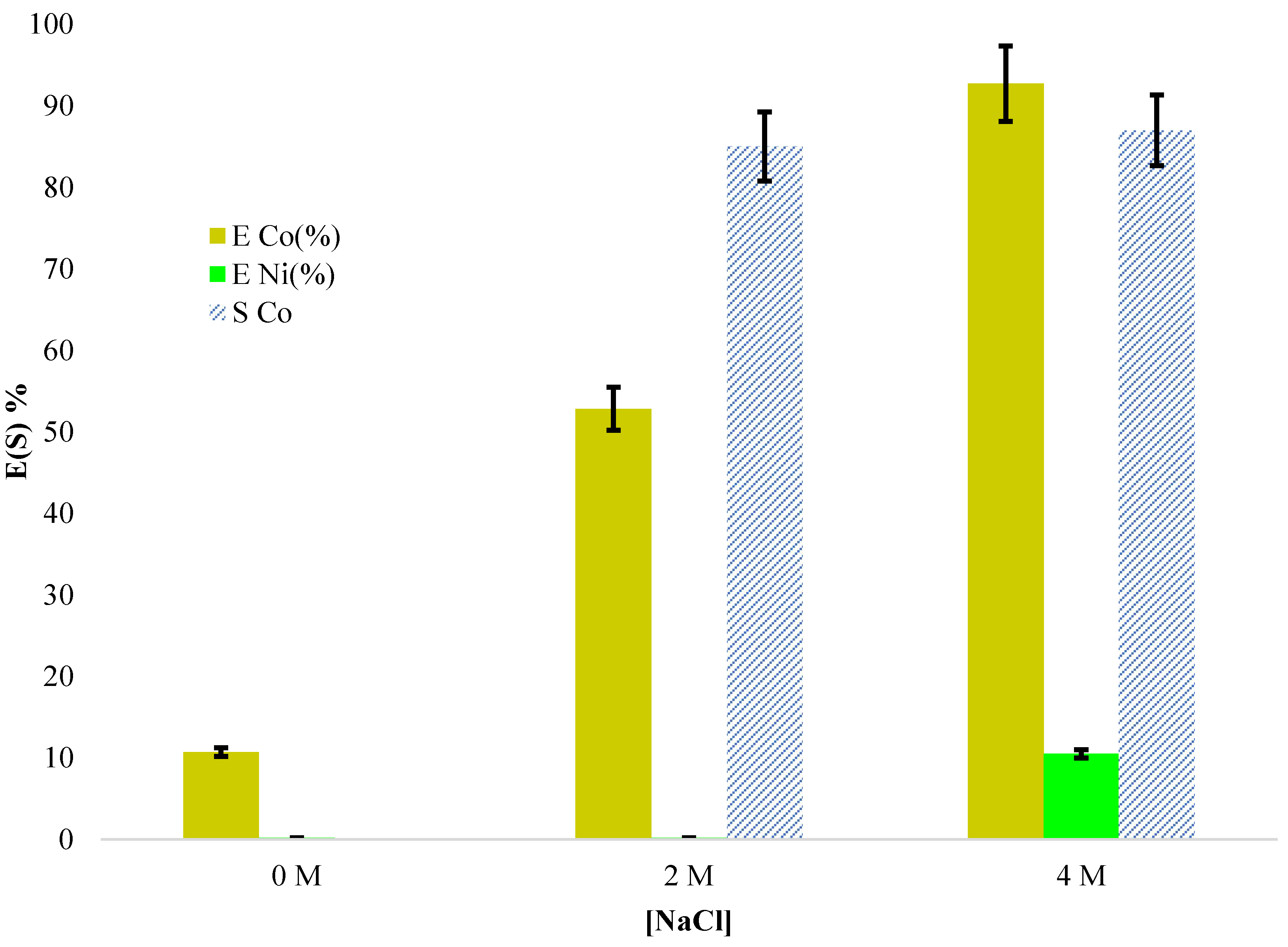
Under conditions similar to those used in dynamic experiments, a 4 M NaCl solution provides the optimal medium for Co/Ni separation, achieving extraction yields of 92% for Co(II) and 10.5% for Ni(II), resulting in a separation factor close to 100. At 2 M NaCl, the separation factor exceeds 100; however, the extraction yields decrease to 53% for Co(II) and only 0.2% for Ni(II). In the absence of NaCl, Ni(II) extraction is negligible, while Co(II) extraction remains limited to approximately 10%.
These results confirm the critical role of chloride concentration in promoting selective extraction, with higher ionic strength favoring the formation of mixed complexes that enhance cobalt recovery while maintaining nickel rejection.
In this study, we seek to investigate the impact of operational parameters such as aqueous and organic phase dilution on extraction yield, in order to better control the extraction process in dynamic mode and help define the optimal operating conditions for the transition from static to continuous mode. Our experiments will focus on the extraction of Co(II) in [Pn3NC2OC2][Dca] dissolved in MIBK.
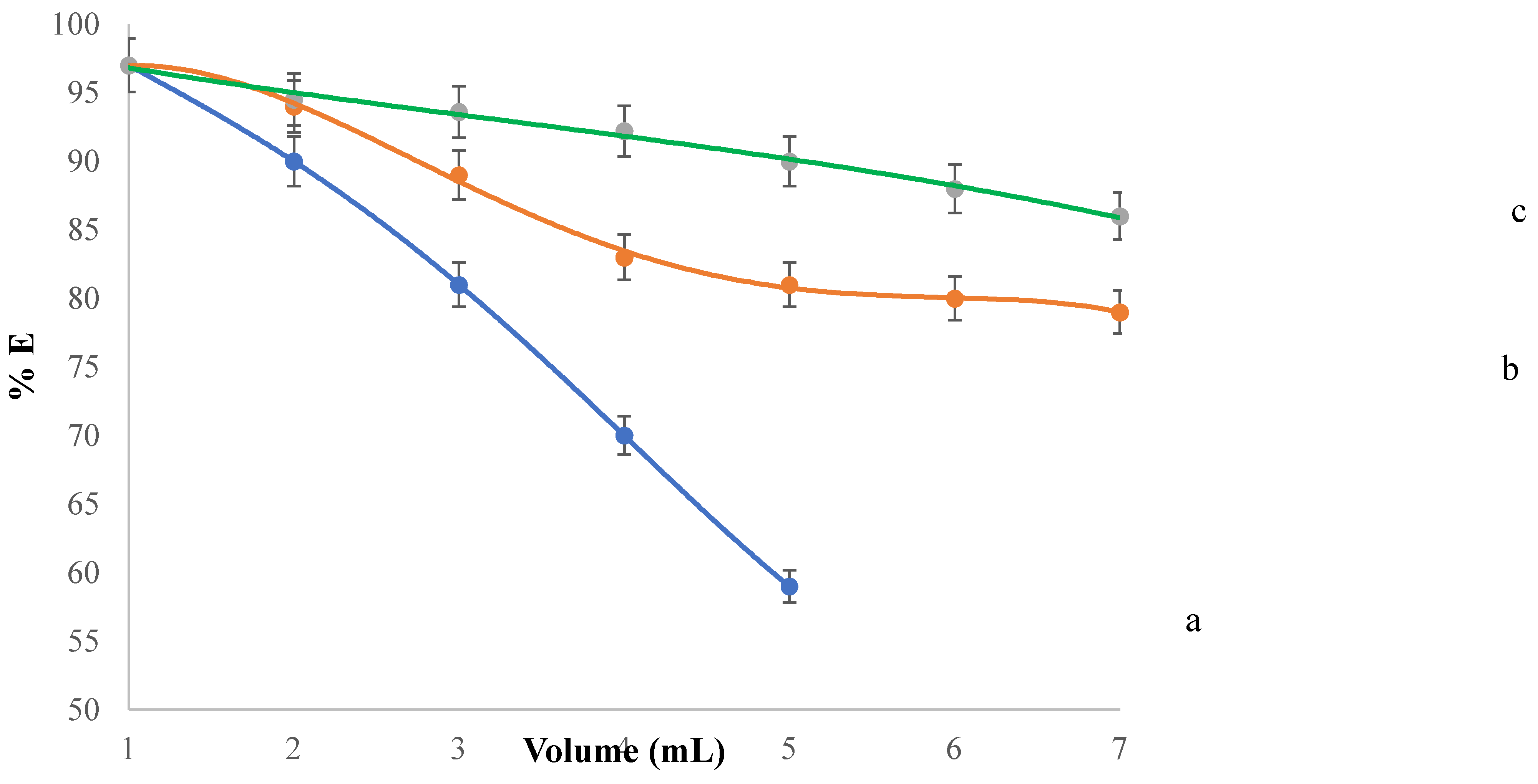
Figure 4 illustrates the variation in Co(II) ion extraction efficiency resulting from the dilution of (i) the aqueous phase, (ii) the organic phase, or (iii) both phases. Throughout the dilution process, the nIL/nCo ratio remains constant at 13.5. The graph shows that diluting either phase significantly decreases the extraction yield of Co(II) ions by [Pn
3NC
2OC
2]Dca]. The most pronounced effect occurs when the aqueous phase is diluted by a factor of 7, resulting in a 18% decrease in extraction yield (from 97% to 79%). In comparison, under the same dilution conditions, the organic phase shows a smaller decrease of 13% (from 97% to 86%). Simultaneous dilution of both phases leads to an overall reduction of more than 30% in extraction yield, up to a dilution factor of 5.
Several factors may account for this decline in extraction efficiency upon dilution: (i) increased exchange between dicyanamide anions and chloride ions, (ii) solvation of the ionic liquid anion in the organic phase due to IL dilution, and (iii) losses of IL caused by its dissolution in the aqueous phase. This raises an important question: how does the metal’s coordination sphere evolve under dilution, and do these behaviors follow the law of mass action? Our previous study [36] proposed the following equilibrium to describe Co(II) extraction in saline media:
This mechanism relies on: (i) the formation of anionic tetrahedral complexes [Co(Dca)
(2+(x+y)(Cl)
(2−(x+y)] in the organic phase, (ii) the ability of chloride ions to enter the Co(II) coordination sphere, and (iii) their capacity to form stable ion pairs with the cation in the organic phase, thereby shifting the equilibrium toward extraction. Here, x and y are complementary values whose sum equals two; a non-zero x implies y = 0 and vice versa, simplifying the expression of the mass action law. According to this hypothesis, a positive x leads to the formation of [Pn
3NC
2OC
2][Cl] species in the organic phase, whereas a non-zero y results in the release of free Dca
− ions into the aqueous phase. The formation of a tetrahedral complex, previously demonstrated extensively in our earlier work [
31,
32], produces an intense blue coloration of the organic phase, fully consistent with the measured spectral characteristics. Its formation is observed even at low Cl
− concentrations, which are incompatible with the formation of the tetrahedral CoCl
42− complex, meaning that dicyanamide ions are present in the coordination sphere of Co(II).
3.3. Determination of Extracted Species in the Organic Phase
Determining the nature of the species extracted in the organic phase is a delicate issue. To address this, the slope method was applied to study the influence of the concentrations of ionic liquid (IL), dicyanamide (Dca
-), and chloride ions, and to specify the stoichiometry of the Co–Dca complexes extracted in the organic phase [
26].
Our previous observations revealed no linear dependence between log D and log[Dca(Cl)], or log [IL], indicating the formation of a mixed tetrahedral complex Co[Dca]
(x)Cl
(4-x) with non-integer values of x. The Co(II) coordination sphere strongly depends on experimental conditions, and the Dca/Cl ratio, as these two ligands are easily interchangeable [
31].
However, as the experiments described in
Figure 4 were all carried out for an identical n
IL/n
Co ratio of 13.5, it may be interesting to see whether the trends observed can be predicted by a similar mass action law encompassing all the experimental data for the experiments depicted in
Figure 4.
Since all experiments in
Figure 4 were conducted at an identical nIL/nCo ratio of 13.5, it is relevant to examine whether the observed trends can be predicted by a mass action law encompassing all experimental data. For each dataset:
Complex formation was considered for x values between 0 and 2 and y values between 0 and 2.
A mass balance was performed, and equilibrium constants were calculated relative to the proposed mass action laws ([Cl−] concentration was assumed constant at 4M NaCl and excluded from equilibrium constant calculations).
-
For each (x, y) pair:
- -
species concentrations at equilibrium were determined.
- -
The law of mass action was applied relative to equilibrium (1).
The average equilibrium constant for each hypothesis was used to recalculate extraction rates for each dataset.
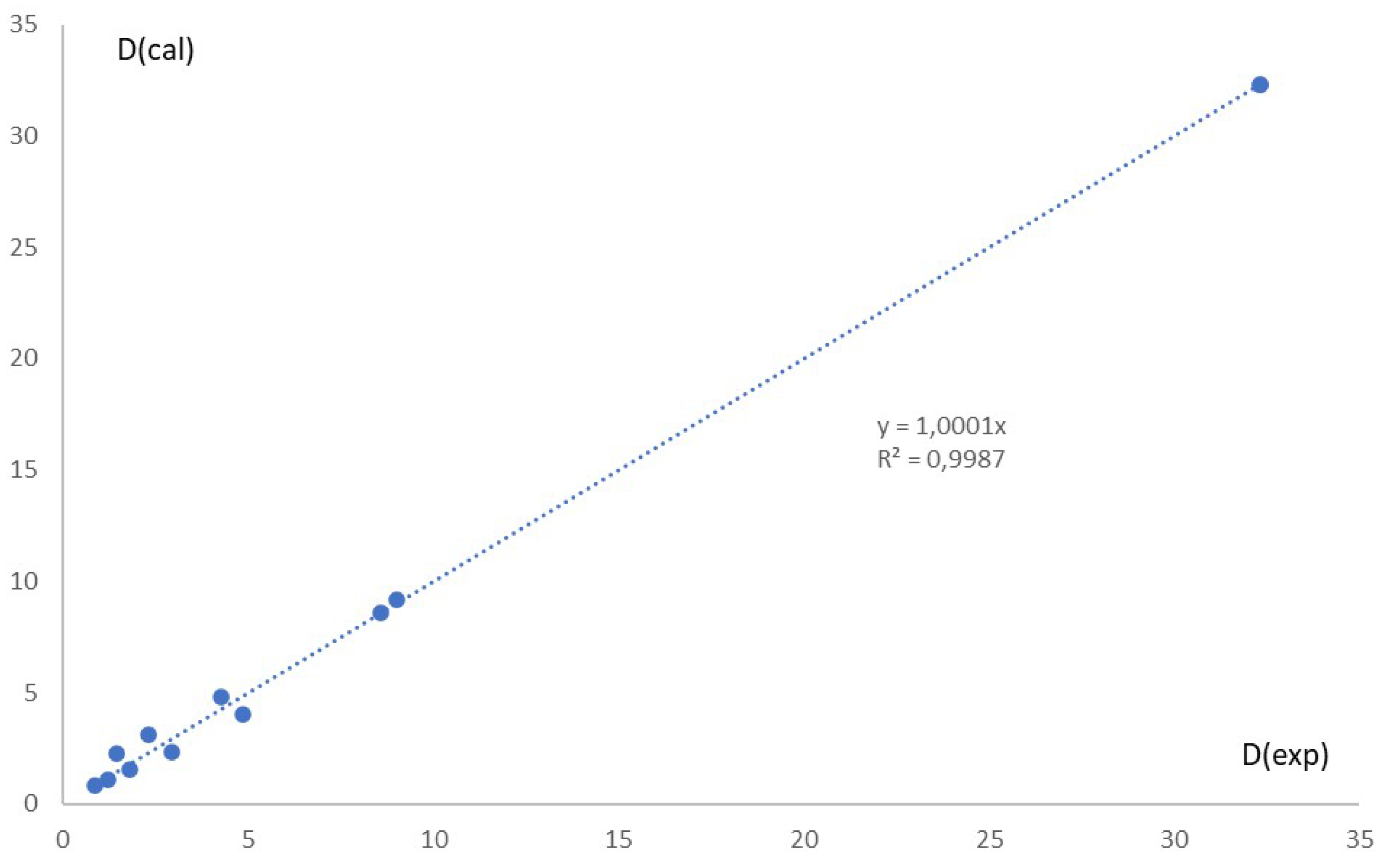
Calculated extraction rates were compared with experimental values using
Figure 5, which plots experimental vs. calculated extraction rates.
The relevance of each hypothesis was assessed using the Least squares method and based on the Slope (theoretical value = 1), and correlation coefficient values.
The Statistical analysis indicates that the best fit corresponds to: y = 0, and x = 1, suggesting a stoichiometry of Co/Dca/Cl ≈ 1/3/1, regardless of experimental conditions.
This is in agreement with the formation of [Pn
3NC
2OC
2]
2[Co(Dca)
3(Cl)], corresponding to the following equilibrium.
It should be noted that good correlations are nevertheless obtained for the value of x between 0.5 and 1.5 and that of y between 0.5 and 0 (slope values range between 1.02 and 1.03, and correlation coefficients are comprised between 0.98 and 0.99). These results corroborate the fact that the coordination sphere does not allow for the identification of a single, well-defined complex.
Instead, the stoichiometry of the complex in the organic phase can vary depending on experimental conditions, particularly the nIL/nCo ratio, which confirms our previous observations.
The equilibrium constant of equilibrium (3) could be determined and is equal to 11.53 ± 0.06. The extraction process can be reversed by changing NaCl with an electrolyte with a kosmotrope anion such as SO42-. Indeed, due to its high hydrophilicity, the sulfate anion is unable to form an ion-pair with the [Pn3NC2OC2]+ cation and allows a shift in equilibrium (1) to the left, leading to stripping of Co(II) in aqueous solution.
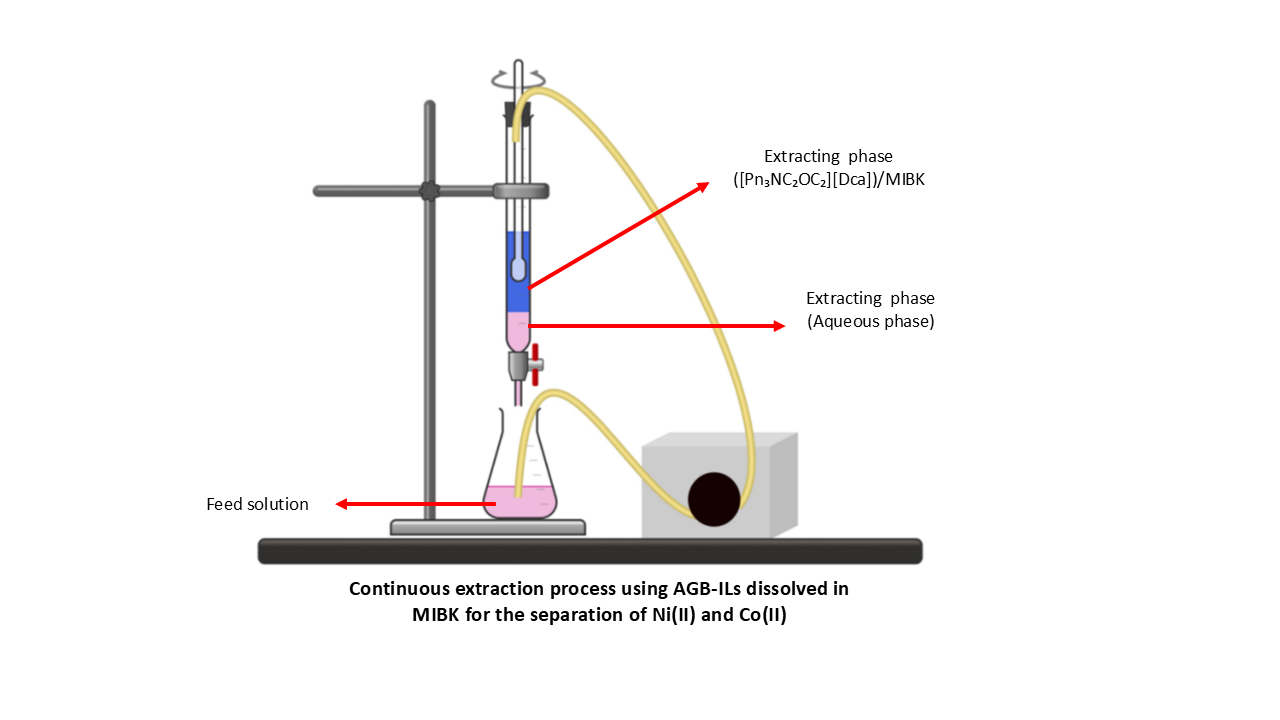
3.4. Development of a Continuous Extraction Protocol
Our objective is to develop a protocol using a single column and a peristaltic pump to continuously extract metals from synthetic effluents. To achieve this, we first evaluated the extractive performance of the organic phase for Co(II) extraction and stripping, and conducted experiments under closed- and open-circuit conditions:
- -
Open circuit: The effluent is collected at the bottom of the column and analyzed.
- -
Closed circuit: The effluent collected at the bottom is reinjected at the top of the column.
Operating under closed-circuit conditions simulates a batch process, allowing us to verify whether the information gathered in batch mode can be applied to a continuous process.
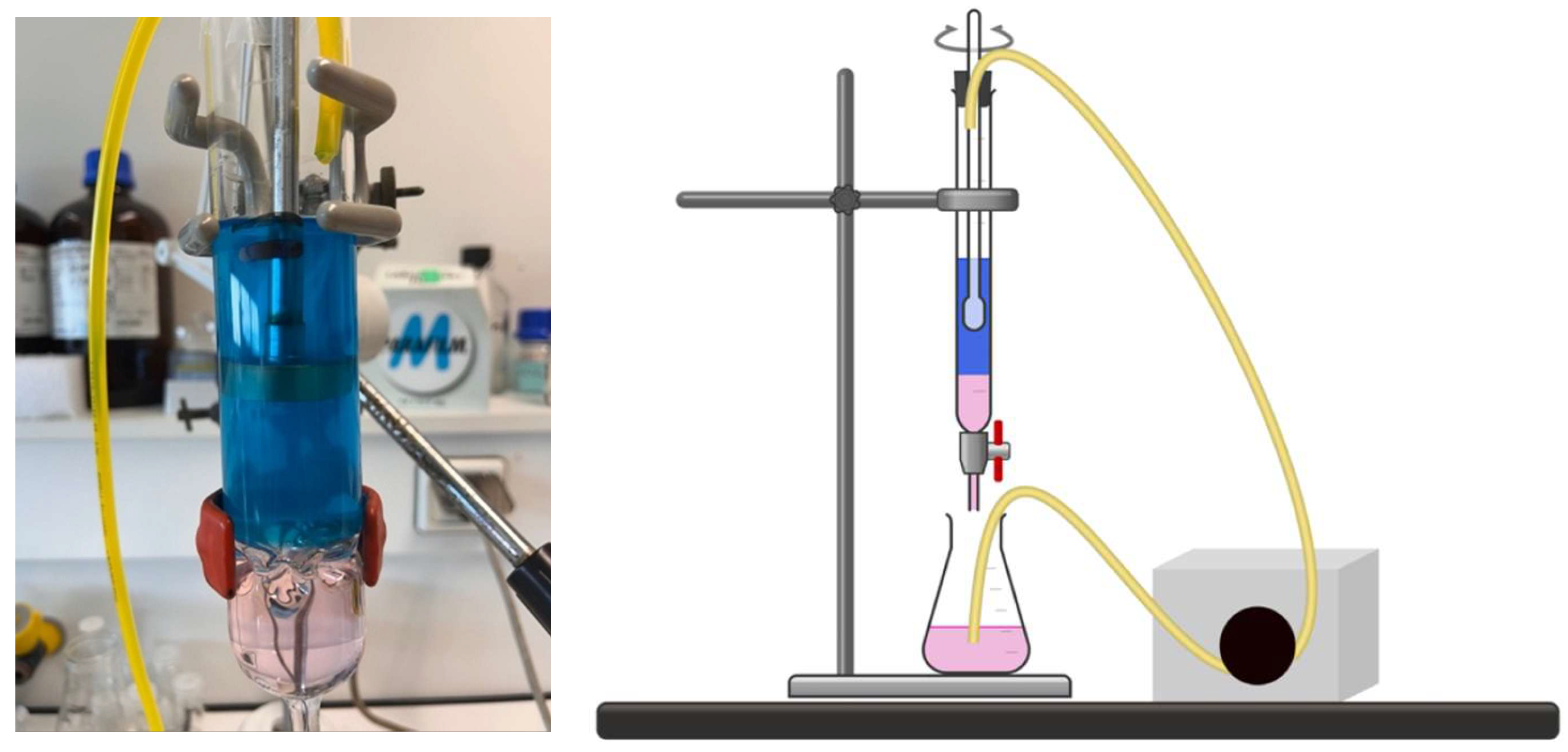
For continuous extraction experiments, we used a single type of column. This is a glass column with a height of 23 cm and a diameter of 4 cm, corresponding to a total volume capacity of 275 mL. The volume of the agitated phase is approximately 230 mL, while that of the non-agitated phase is approximately 50 mL. The agitated and non-agitated compartments are separated by horizontal permeable glass tubes that allow direct communication between them while preventing the agitation vortex from propagating. This setup enables an almost instantaneous transfer between the two compartments.
Experiments in open circuit were designed to assess the feasibility of separating Co(II) from Ni(II) using the proposed strategy.
We also investigated the reversibility and recyclability of the extraction process over four extraction/stripping cycles. To compensate for yield loss due to the larger organic phase volume, higher nIL/nCo ratios were used compared to batch conditions. The ratios tested were 13, 26, and 40, respectively
Since organic phase dilution negatively affects Co(II) extraction yield, the solvent volume was minimized to the smallest amount required to dissolve 15 g of ionic liquid (maximum quantity used), while maintaining acceptable viscosity. Thus, all experiments used 50 mL of MIBK. The standard experimental conditions to carry out extraction in continuous mode are as follows:
- -
Organic phase volume: 50 mL
- -
Ionic liquid mass (m IL): 5, 10, 15 g
- -
Effluent volume: 100 mL
- -
Stripping solution: H2O or aqueous Na2SO4 (1.5 M), V = 100 mL
- -
Stirring speed: 4 rpm·s-1
- -
Flow rate: 4 and 6 mL·min-1
These conditions were used to study the influence of ionic liquid concentration in the organic phase, the stirring speed, and the system performance over multiple extraction-stripping cycles.
After each experiment, organic phases were collected, combined, and evaporated. To recover the ionic liquid, an anion metathesis using aqueous NaDca was then performed under the same conditions as during synthesis to restore its effectiveness.
3.4.1. Influence on Stirring Speed
The influence of stirring speed on the extraction system’s performance was evaluated by determining the extraction yield of Co(II) through up to 5 elution cycles of 100 mL of the same aqueous Co(II) solution under stirring ranging from 0 to 10 rpm s-1. We show that without stirring, the extraction yields are less than 10% (not shown here), which means that the passage of the aqueous phase through the organic phase does not generate sufficient contact between the metal and the extractant to effectively transfer the metal from the aqueous phase to the organic phase. This indicates that in continuous mode, contact between the organic and aqueous phases should be enhanced on a very low timescale. The influence of stirring speed occurs between 2 and 4 rpm.s-1, the extraction speed significantly influences the extraction yield, which increases from 5 to 90%. For stirring speeds exceeding 4 rpm/s, the latter has no influence, with an extraction yield reaching its maximum value over 90%.
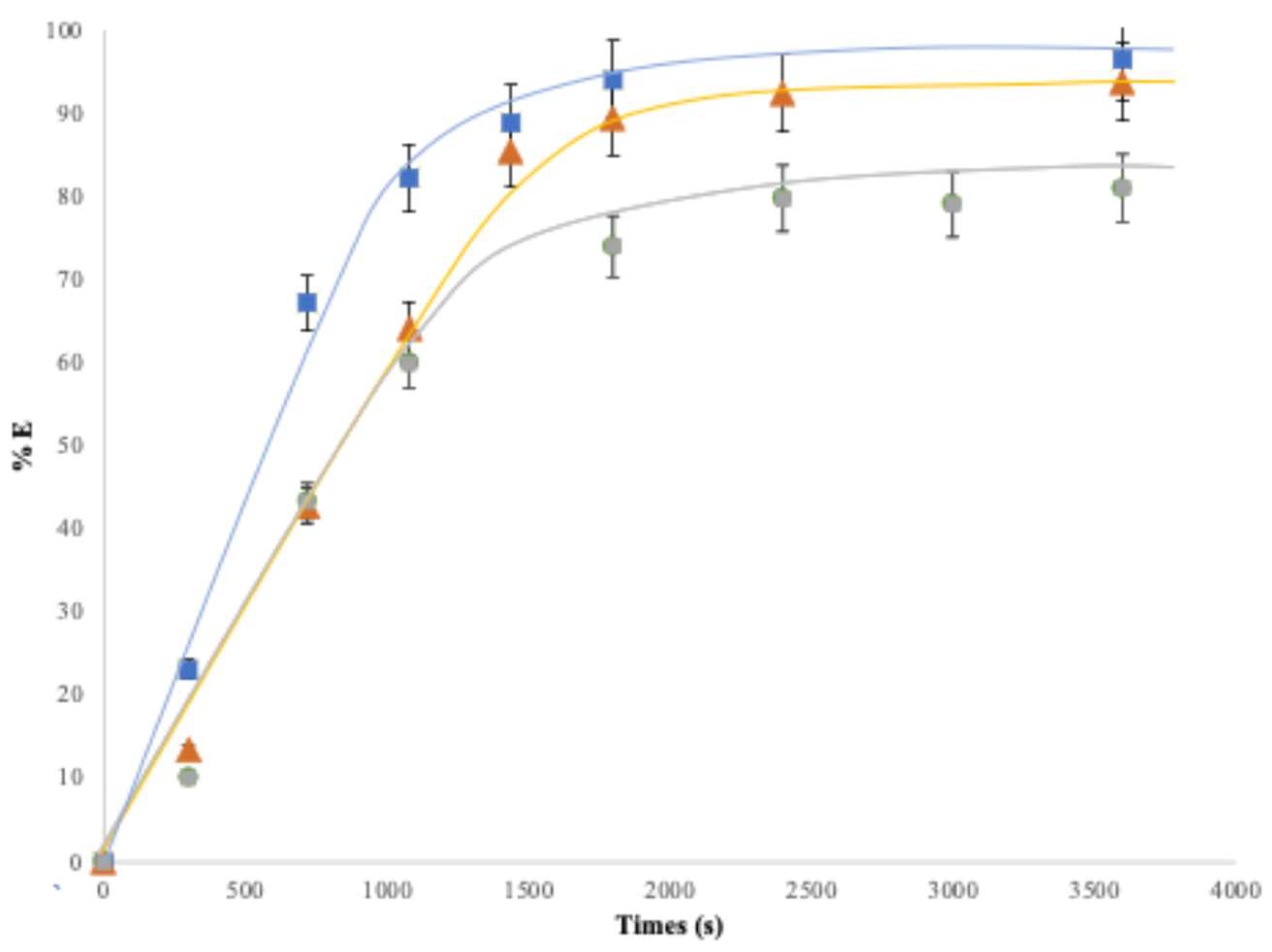
3.4.2. Influence on the Flow Rate
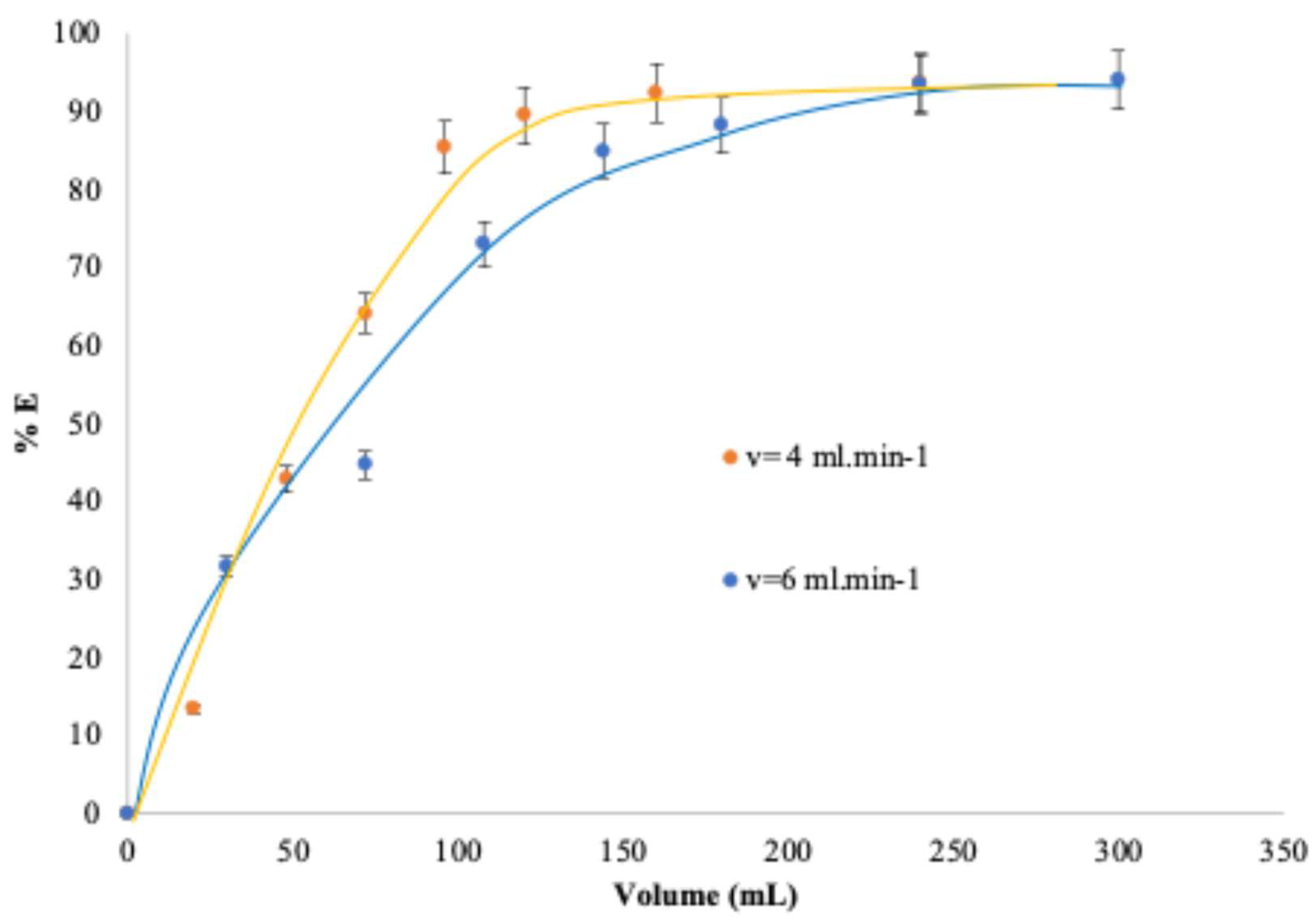
Figure 6 illustrates the effect of the feed solution flow rate on the extraction performance over time, for flow rates of 4 mL·min
-1 and 6 mL·min
-1, during periods shorter than 1500 s. The results show that a higher addition rate leads to a higher extraction yield.
The extraction yield reaches its maximum after approximately 1500 s, which corresponds to the passage of 100 mL and 150 mL of feed solution through the column at flow rates of 4 mL·min−1 and 6 mL·min−1, respectively. A flow rates of 4 mL.mn-1 allow a contact time between the feed solution and the extracting phase sufficient for the system to approach a near-equilibrium conditions with a single pass of the feed solution. This conclusion is supported by the fact that the extraction yields obtained in the column are comparable to those measured in batch mode, as will be discussed later in this article. However, at higher flow rates, the contact time becomes too short to reach equilibrium within a single pass, and achieving maximum yield would then require processing a volume equal to approximately 1.5 times the volume of the feed solution. A flow rate of 4 mL·min−1 (14.5 L·h−1) provides suitable reference conditions for semi-pilot-scale testing. At this stage of our investigation, further optimization is not required.
3.4.3. Effect of Ionic Liquid Dilution on Extraction Performance
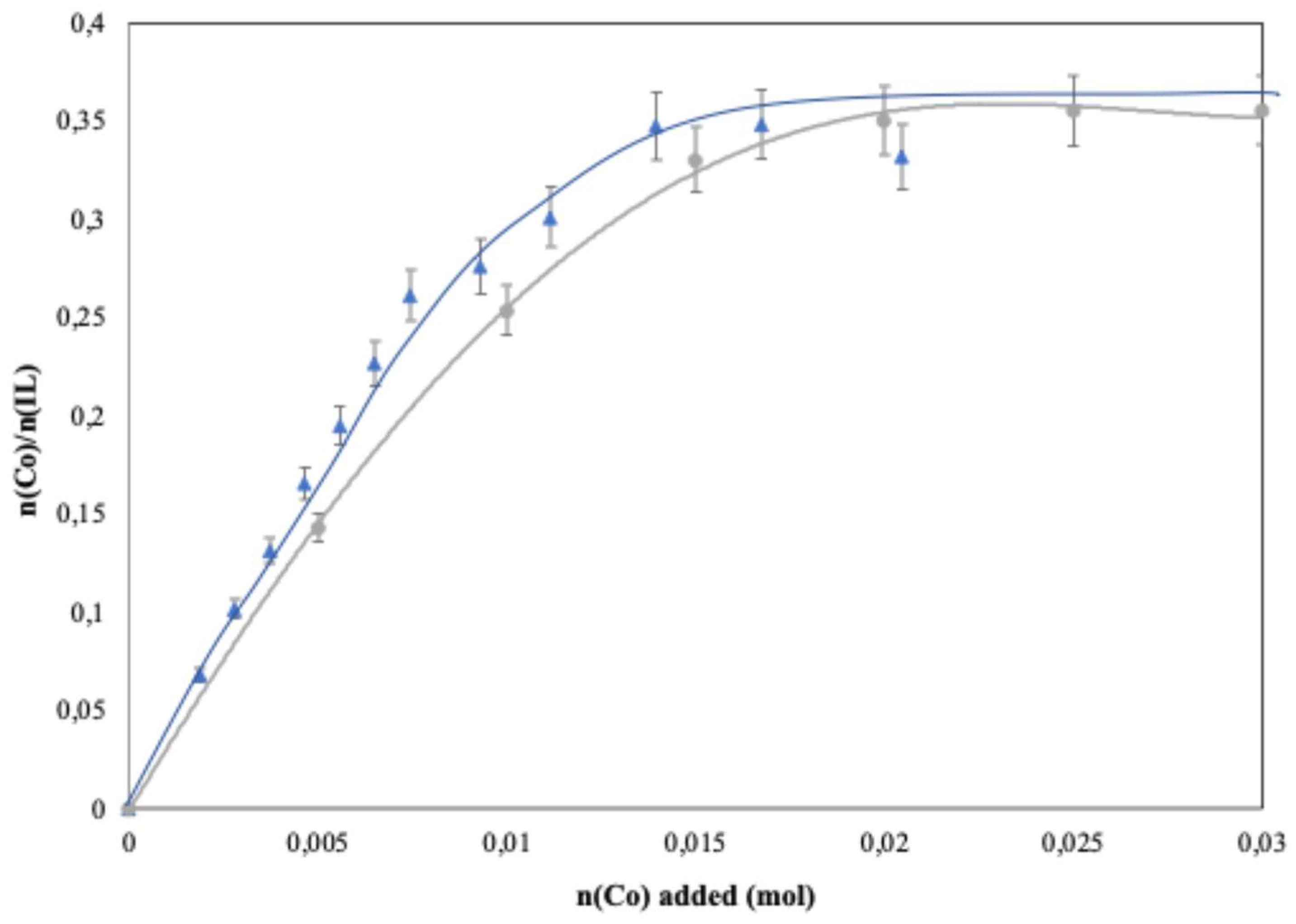
Dilution of the ionic liquid (IL) in the extracting phase affects its performance in terms of extraction yield. This suggests that maintaining system performance may require working with higher [IL]/[M(II)] ratios than those used in batch experiments. However, increasing the amount of ionic liquid raises questions about its impact on solution properties such as the viscosity of the extracting phase and on process recyclability.
Figure 7 presents the extraction yields obtained by passing 100 mL of a Co(II) solution (0.01 M) through 50 mL of organic phase containing 5, 10, or 15 g of [Pn
3NC
2OC
2][Dca] at a flow rate of 6 mL·min
−1. These masses correspond to nIL/nCo ratios of 13, 26, and 39.
Figure 7 shows that increasing the amount of ionic liquid dissolved in the organic phase significantly improves system efficiency, even under continuous extraction conditions. For example, increasing the IL content from 5 g to 15 g raises the extraction yield from 81% to 96.5%. To achieve quantitative extraction (yield > 90%), nIL/nCo ratios of at least 25 are required. This may not be a major drawback if process recyclability remains high.
The extraction yields obtained with a similar nIl/nCo ratio in batch experiments range between 76%, and 96% and are close to those obtained from continuous flow extraction experiments. This indicates that the contact time between the aqueous and organic phases in continuous mode is sufficient to approach equilibrium. The extraction rate confirms this after 1500 s, which corresponds to the time needed to elute the entire feed solution. At this point, the extraction rate reaches 91% of the equilibrium value.
3.4.4. Influence of the Saline Media in Continuous Mode
3.4.4.1. Comparison of Extraction Yields in Continuous and Batch Systems
Figure 8 presents the maximum Co(II) extraction yields obtained in continuous, closed-system experiments using [Pn
3NC
2OC
2][Dca] and [Pn
3NC
2OC
2][Br] as extractant phases. To enable comparison with batch experiments, the operating conditions were kept similar: an nIL/nCo ratio of 26 and a volume ratio of 2 between the aqueous and organic phases.
The results obtained in continuous mode confirm the trend observed in batch mode: extraction yield increases with salt concentration, and [Pn3NC2OC2][Dca] exhibits higher extraction efficiency than [[Pn3NC2OC2][Br]. The most notable observation is the strong similarity between the extraction results in continuous and batch experiments. Under comparable conditions, the yields differ by no more than 5%, suggesting rapid, almost instantaneous, transfer kinetics during continuous operation.
Under the experimental conditions used, the system reaches equilibrium within 30 to 40 minutes, corresponding to approximately two complete passes of the solution.
3.4.5. Recyclability of the IL
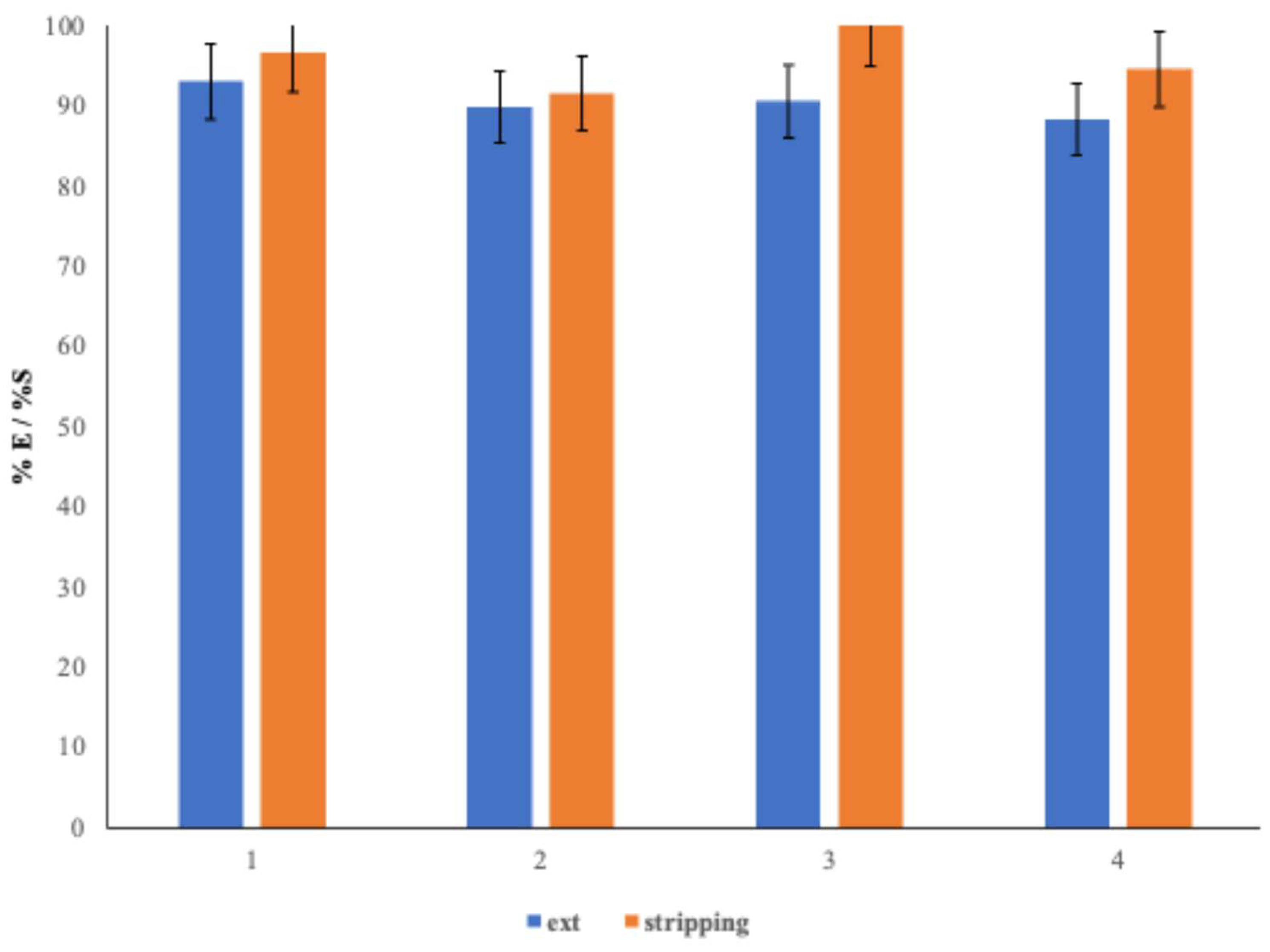
The recyclability of the ionic liquid (IL) in continuous mode was evaluated over four extraction–stripping cycles using 100 mL of a 0.01 mol·L−1 Co(II) solution. At the end of each cycle, the aqueous phase was analyzed to determine the concentration of the organic cation and estimate the cumulative losses of the IL.
The stripping step was carried out by passing 100 mL of either pure water or an aqueous Na2SO4 solution (1.5 mol·L−1) through the organic phase under the same conditions as those used for Co(II) extraction.
Figure 10 presents the evolution of extraction and stripping yields across the cycles, along with the concentrations of dissolved organic cation.
The system maintained high performance after four back-extraction cycles, with extraction yields remaining above 88%. The ionic liquid demonstrated good recyclability, with stripping yields exceeding 90% even after five cycles under similar conditions. In contrast, when water was used instead of Na2SO4 solution, the stripping yield did not exceed 80%.
This difference is directly related to the lower IL loss observed with each cycle, as shown by the concentration of organic cation solubilized after each stripping step. The release of organic cations into the aqueous phase increases with each cycle due to the Dca−/Cl− exchange occurring during the extraction step. This substitution renders the IL more hydrophilic, increasing its solubility in water—[Pn3NC2OC2][Cl] being more water-soluble than [Pn3NC2OC2][Dca].
After five cycles, the cumulative IL loss was 21% and only 3% when stripping was performed with water and Na2SO4 solution, respectively.
The chemical characterization (NMR spectroscopies and elemental analysis) of the IL recovered after each extraction and stripping cycle shows that the IL has not undergone any alteration. In addition, the extraction performance obtained for each of the five cycles confirms that the IL can be successfully recovered and reused without losses on the systems extraction performance
3.4.6. Co(II)/Ni(II) Separation in Saline Media
The efficiency of [Pn3NC2OC2][Dca], diluted in MIBK, was evaluated for the separation of cobalt and nickel in an equimolar aqueous Co(II)/Ni(II) solution, as well as in a 4 M NaCl aqueous medium. The total metal concentration was maintained at 0.02 mol·L−1. Experimental conditions were consistent with those previously used for metal extraction from monometallic solutions.
Under these conditions, the extraction yields for Co(II) and Ni(II) were 93.1% and 1.79%, respectively, resulting in a separation factor exceeding 500. These yields are comparable to those obtained from monometallic solutions, with an even higher separation factor observed in the bimetallic system. This outcome highlights the effectiveness of the column-based continuous extraction system for selectively separating Co(II) from Ni(II).
These findings provide a strong proof of concept for the use of saline media in continuous extraction processes aimed at the strategic separation of 3d transition metals. Future work will focus on optimizing the operational parameters of the process, with particular attention to enhancing extraction efficiency, improving Co/Ni selectivity, and increasing the recyclability of the system.
Potential avenues for improvement include the use of extractants with greater hydrophobicity than [Pn3NC2OC2][Dca], which could reduce ionic liquid losses and enhance process sustainability. Additionally, optimizing the ionic liquid/solvent ratio and selecting more environmentally friendly solvent systems will be explored to develop an extraction phase with tailored properties. Adjustments to the column design, such as reducing its diameter while maintaining a constant volume, will also be investigated to increase the contact time between the aqueous and organic phases, thereby improving overall process performance.
3.4.7. Comparison with Other Existing Processes
The extraction and separation of cobalt(II) and nickel(II) using ionic liquids primarily rely on two distinct strategies, each offering specific advantages depending on the operating conditions:
a) Cation-Exchange
This approach employs ionic liquids functionalized with complexing groups, such as organophosphorus acids (e.g., Cyanex 272). These extractants operate via a cation-exchange mechanism involving proton displacement. Here, pH control is critical, as it directly influences the relative affinity of Co(II) and Ni(II) for the extractant, thereby governing their separation. While ionic liquids based on carboxylate anions (e.g., [P8888][Oleate]) can effectively bind both metals, their selectivity remains limited, making selective separation challenging [
27].
- a)
Anion-exchange
This method leverages the formation of anionic metal complexes, such as CoCl42−, which interact with the ionic liquid’s anion (typically Cl−). Common extractants include Aliquat 336, Cyphos IL 101, [P8888][Cl], and [P44414][Cl]. The formation of CoCl42− requires highly chlorinated environments (e.g., HCl > 6 M). Under these conditions, Ni(II) does not form stable chlorocomplexes, enabling efficient Co/Ni separation.Performance and limitations of existing systems
Most reported systems have been optimized at the laboratory scale, achieving extraction yields of 90% to 100% in batch mode [
30]. However, continuous processes remain rare, complicating direct performance comparisons. Notable exceptions include:
- a reactor developed by Wellens et al. [
33] using a stationary [P66614][Cl] phase, which achieved separation factors exceeding 52,000 in 4 M NaCl solutions with metal concentrations of 1–2 mmol·L
−1.
- a multi-stage mixer–settler process based on Cyanex 272, enabling Ni/Co separation at moderate pH (5–6), though requiring large volumes of organic phase and precise pH control [
16,
34].
- a supported membrane extraction system proposed by Zante et al. [
28]using [P66614][Cl], which facilitates Ni(II)/Co(II) separation, although Co(II) extraction does not exceed 75% at 9–10 mol·L
−1 HCl.
Our approach may constitute an innovative alternative, since it relies on the formation of mixed anionic complexes Co(Dca)4₋ₓClₓ2− using based Dca ionic liquids. This strategy offers several advantages:
- an effective Co/Ni separation at Cl− concentrations above 2 M,
- an independence from pH, unlike commercial ionic liquids (e.g., Aliquat 336 or Cyphos IL 101), which require highly acidic conditions (HCl > 6 M). Batch experiments confirm that our method remains efficient even in moderately acidic media (pH > 2). Since Dca− exhibits negligible acid–base behavior, strict pH control is unnecessary.
Our process achieves results comparable to those reported in the literature in a single-step extraction while using moderate NaCl concentrations, making it suitable for scale-up. While our study focuses on demonstrating proof of concept, the process still offers significant potential for optimization.
3.4.8. Economic Advantages and Outlook
Although the high cost of ionic liquids often limits their industrial deployment, our ionic liquids can be synthesized in only 2–3 steps with high yields. This enables straightforward production of up to 100 g at the laboratory scale, at costs roughly an order of magnitude lower than those of commercially available ionic liquids. Such cost efficiency strongly supports the economic viability of future scale-up.
Based on reagent costs, the estimated synthesis cost for 100 g of [Pn3NC2OC2][Dca] is approximately € 50. In contrast, 100 g of Cyphos IL 101 and [P66614][Cl] cost approximately € 240 and € 347, respectively (Sigma-Aldrich pricing). When molecular weight is taken into account, the economic advantage becomes even more pronounced—exceeding a factor of 8 relative to Cyphos IL 101 and a factor of 17 relative to [P66614][Cl].