Submitted:
09 March 2026
Posted:
11 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. On the Restartability of Hybrid Rocket Systems
1.2. Three-Dimensional Printing as an Enabling Technology for a Low-Power Hybrid Arc Ignition System
1.3. Using Nitrous Oxide as an Alternative Hybrid Rocket Oxidizer
1.3.1. Safety Concerns Associated with Using Pure Nitrous Oxide as a Hybird Rocket Oxidizer
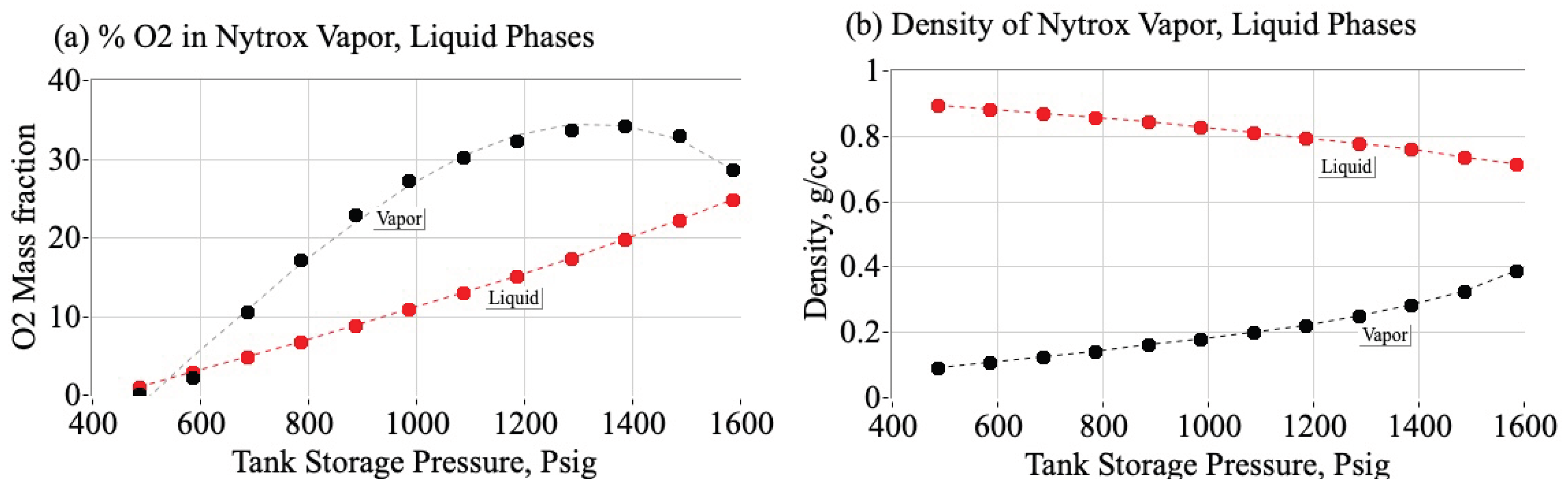
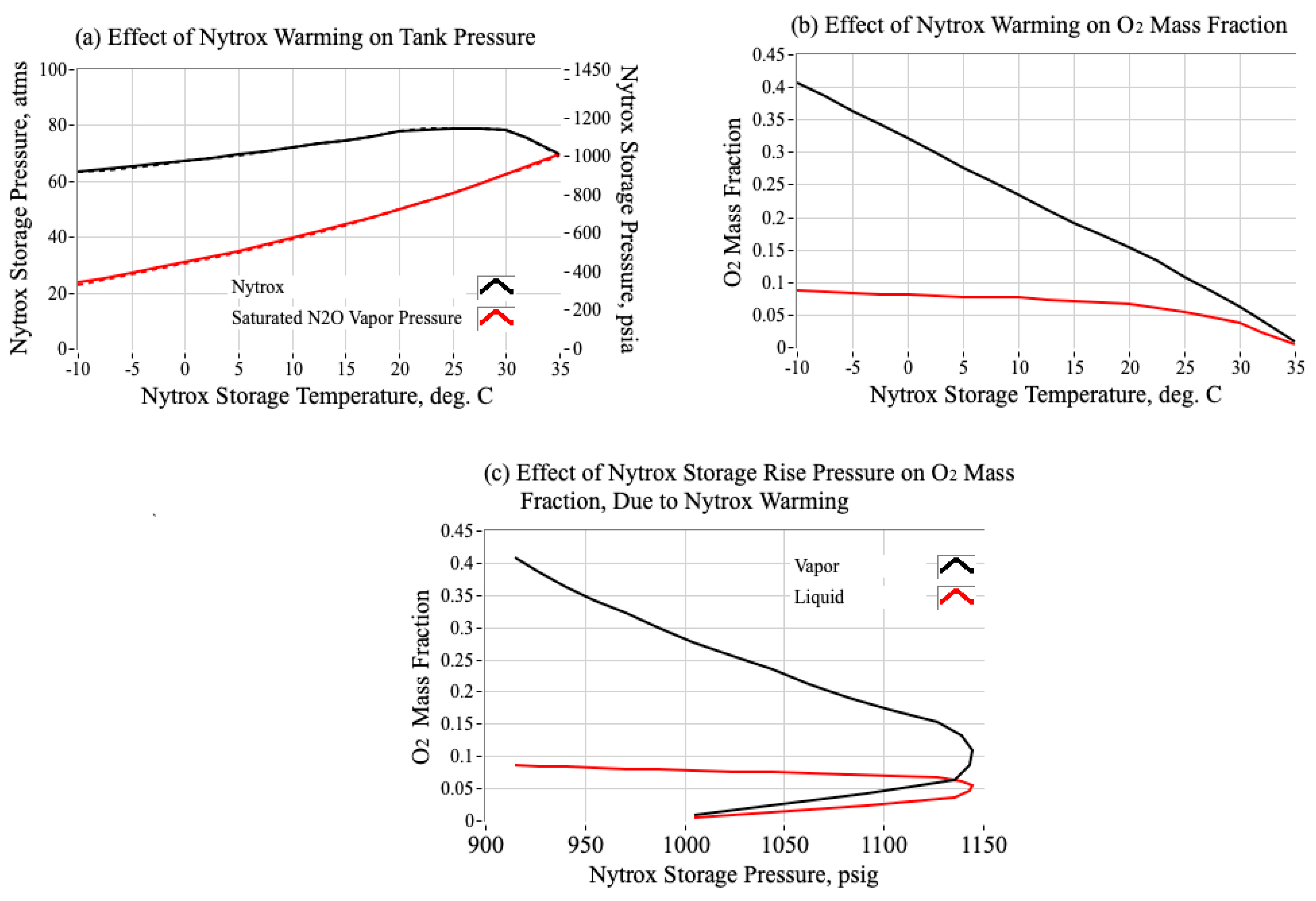
1.3.2. Using Nytrox as an Inherently Safe, Higher-Density, Alternative to Pure Nitrous Oxide
1.3.3. Arc-Ignition Reliability and Latency Issues Associated with Using Nytrox as a Hybrid Rocket Oxidizer
2. Materials and Methods
2.1. Test Systems Hardware
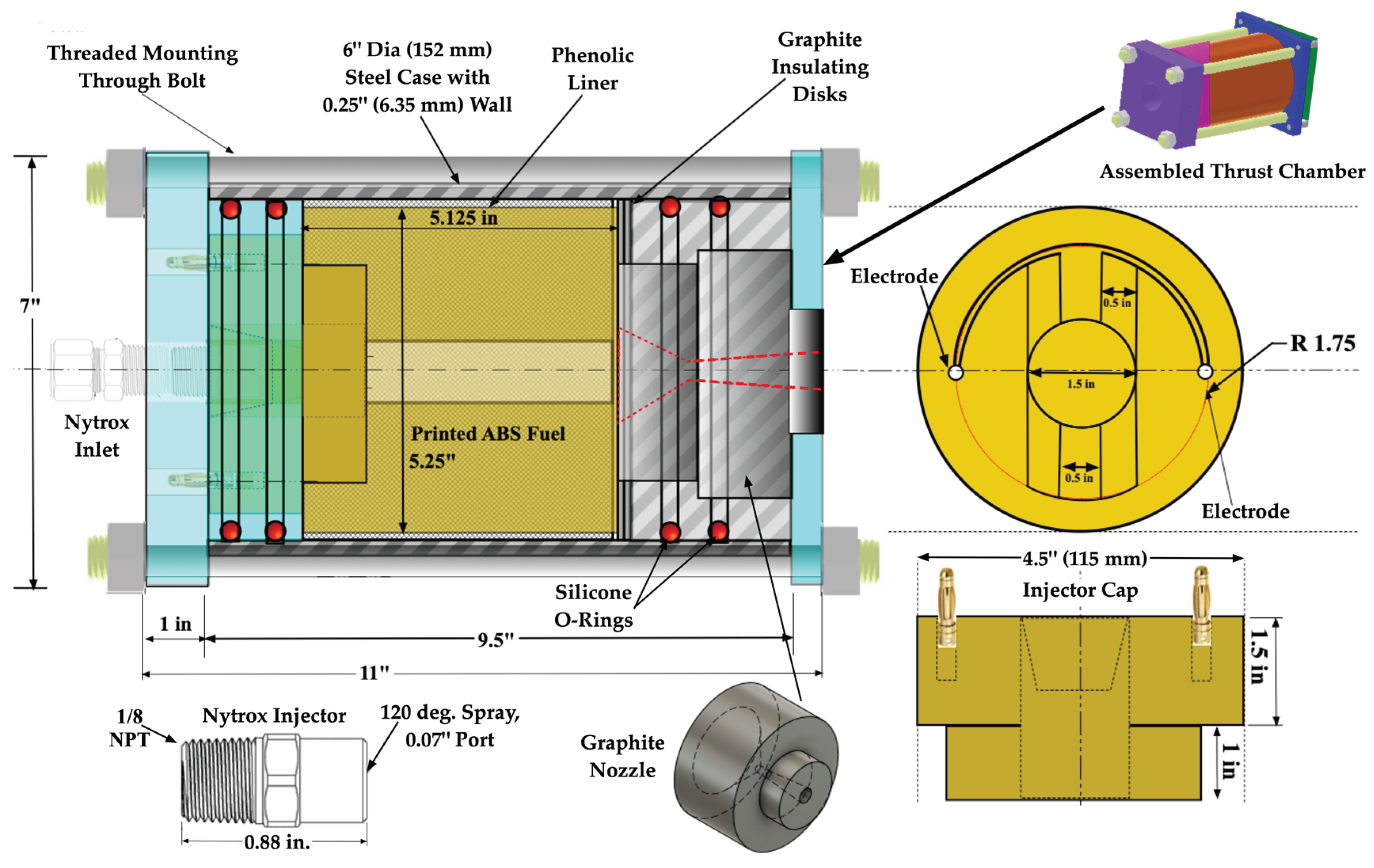
2.1.1. Thrust Chamber Design
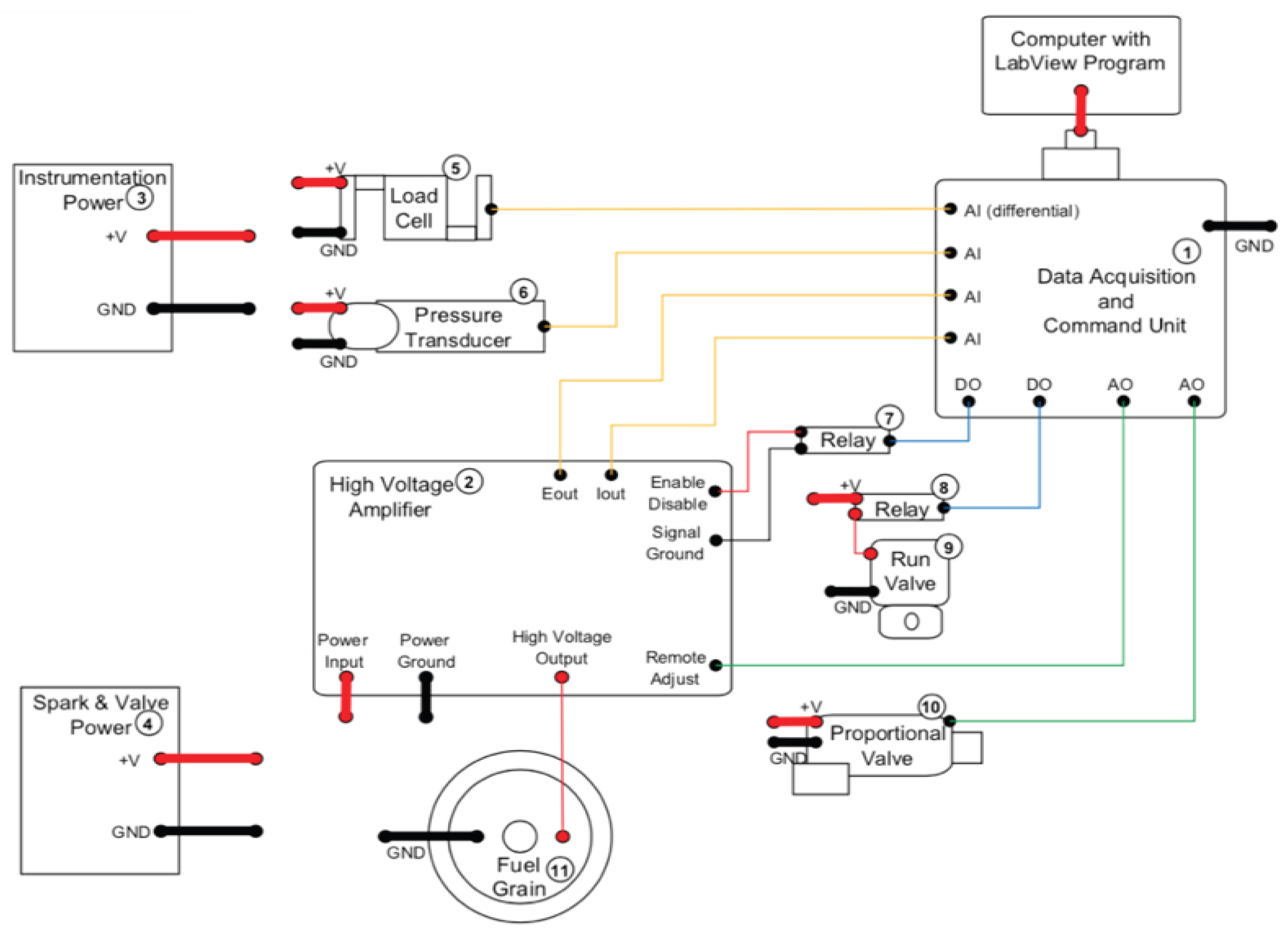
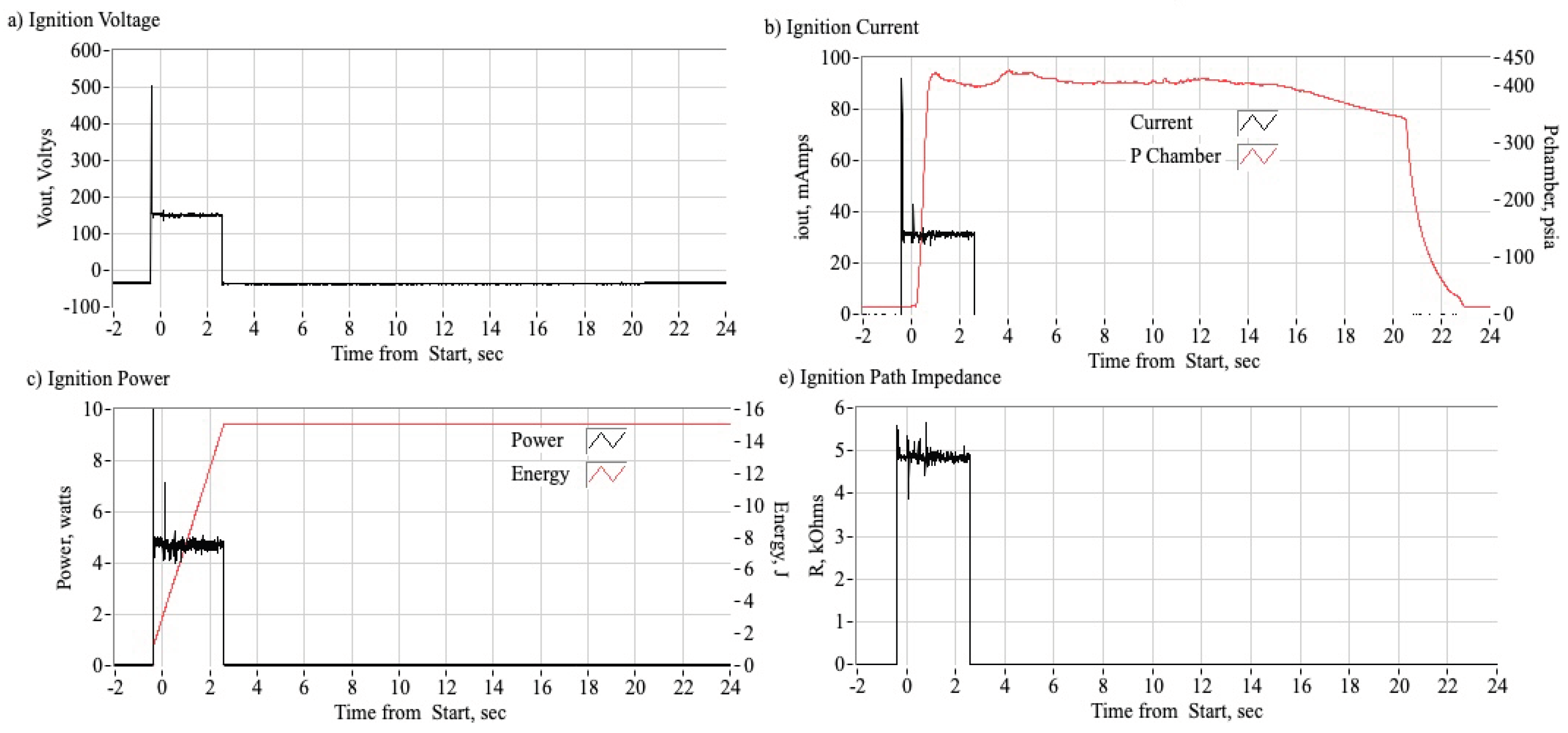
2.1.2. Arc-Ignition System
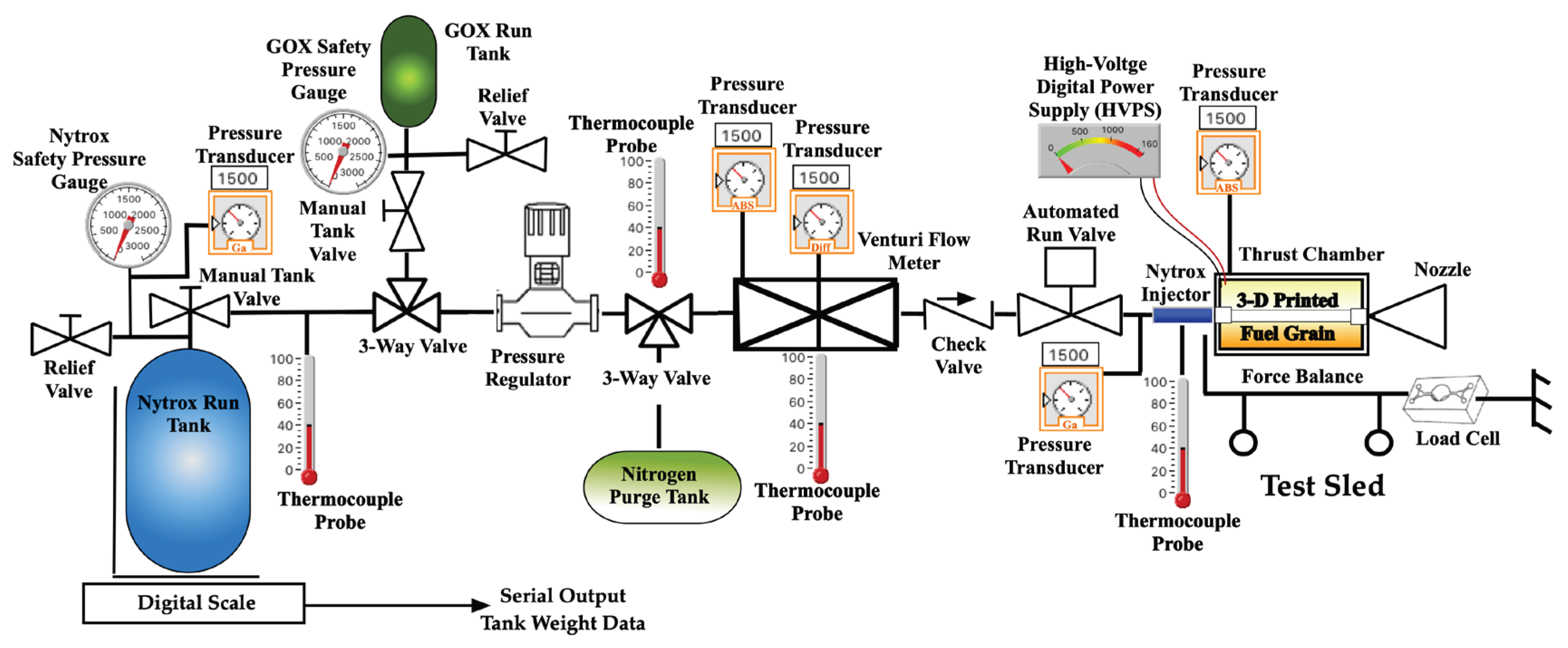
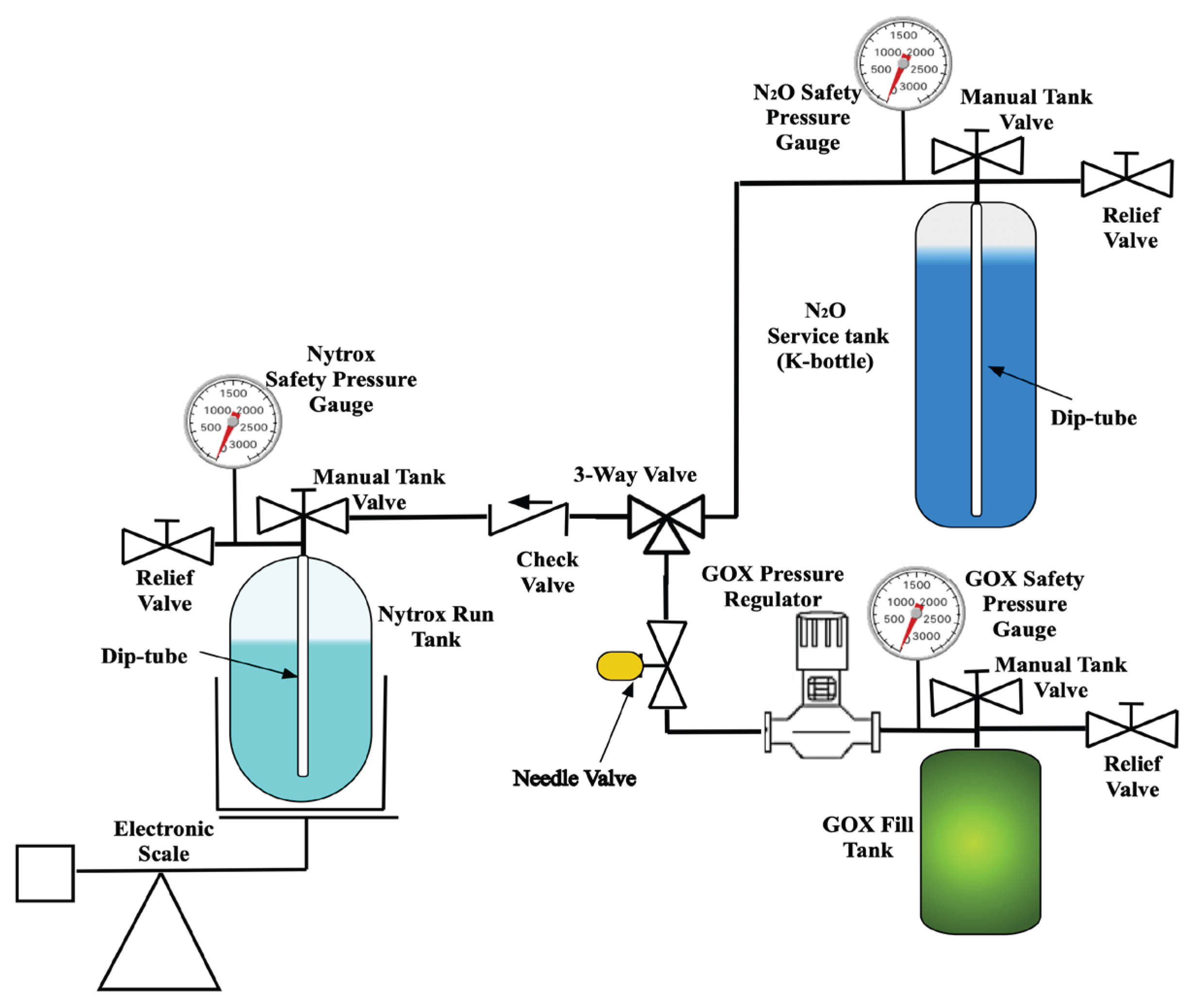
2.1.3. Motor Test Support Systems
2.2. Test Procedures
2.3. Making the Nytrox Solution
3. Results
3.1. Gox Prelead Time Tests
3.2. Nytrox-Only Ignition Tests
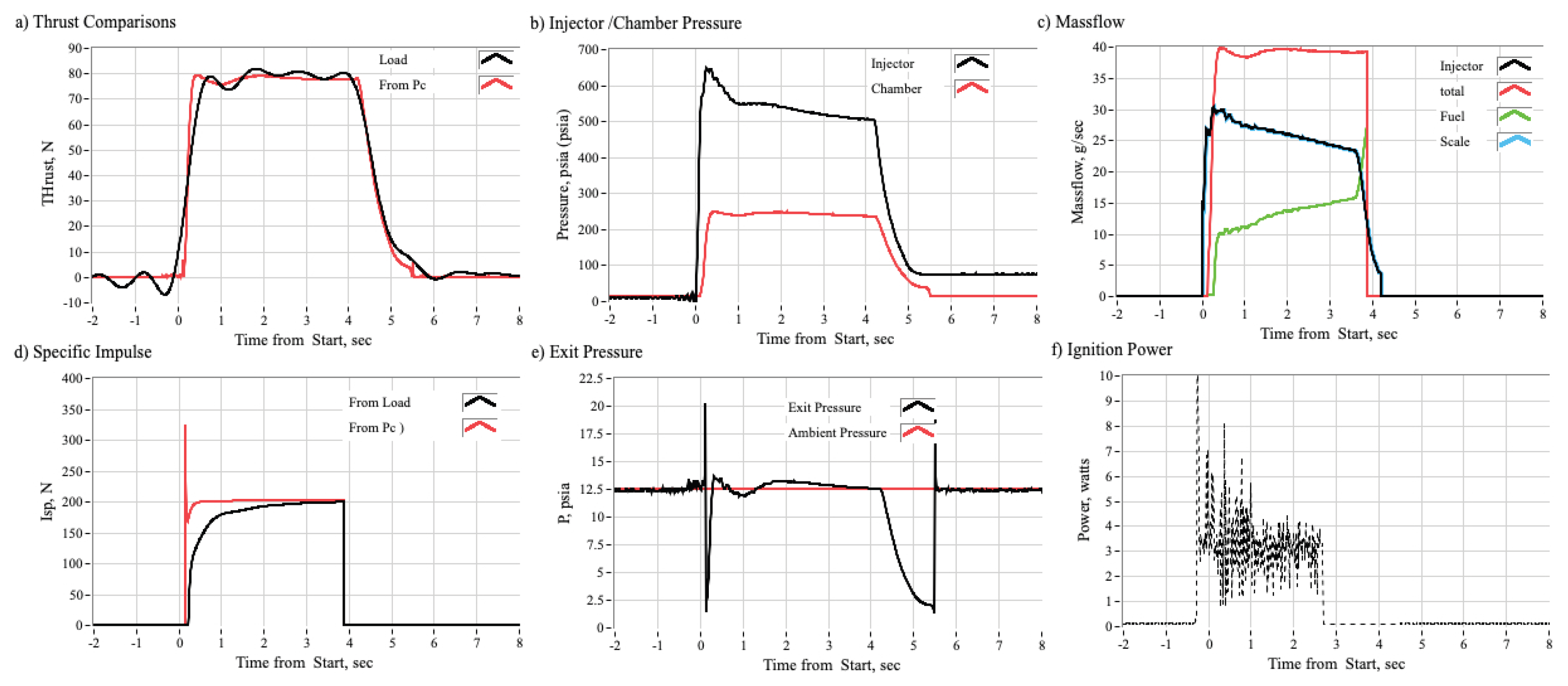
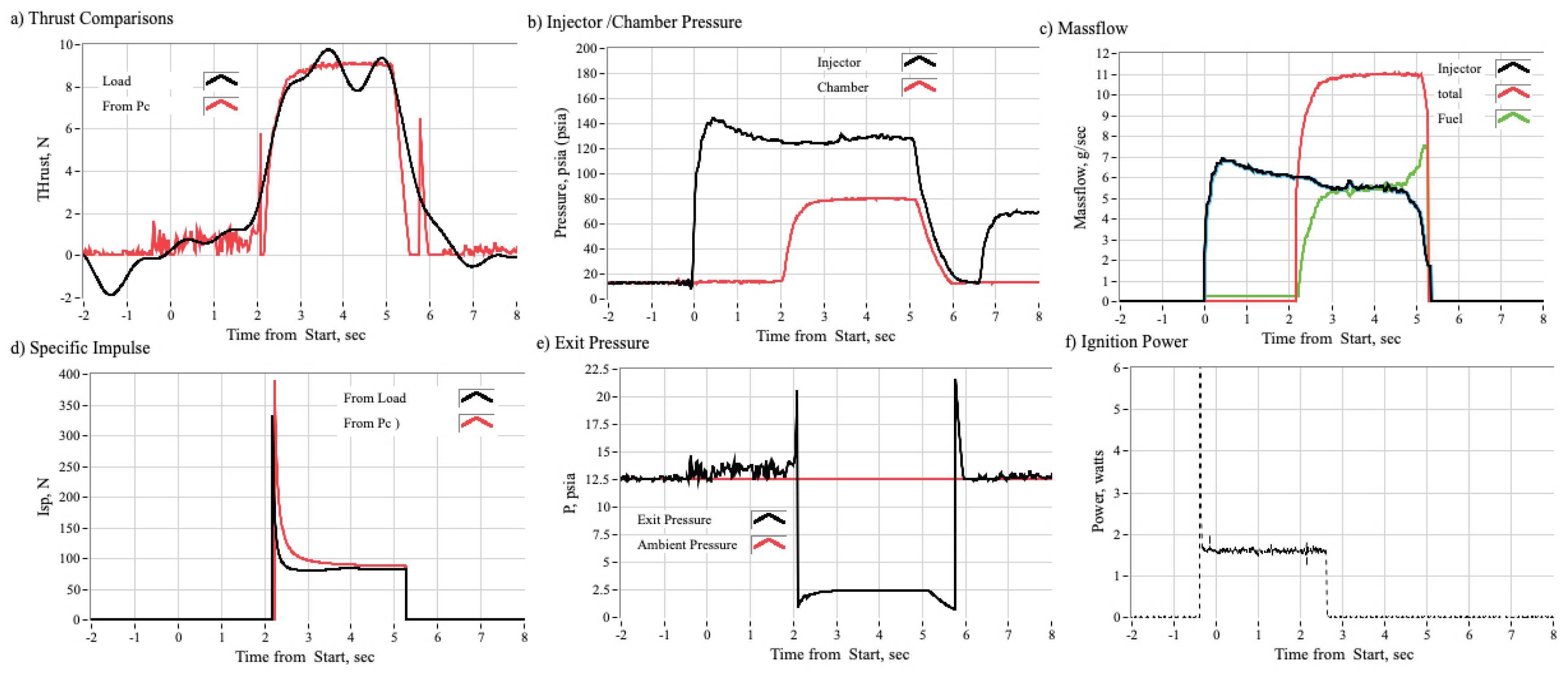
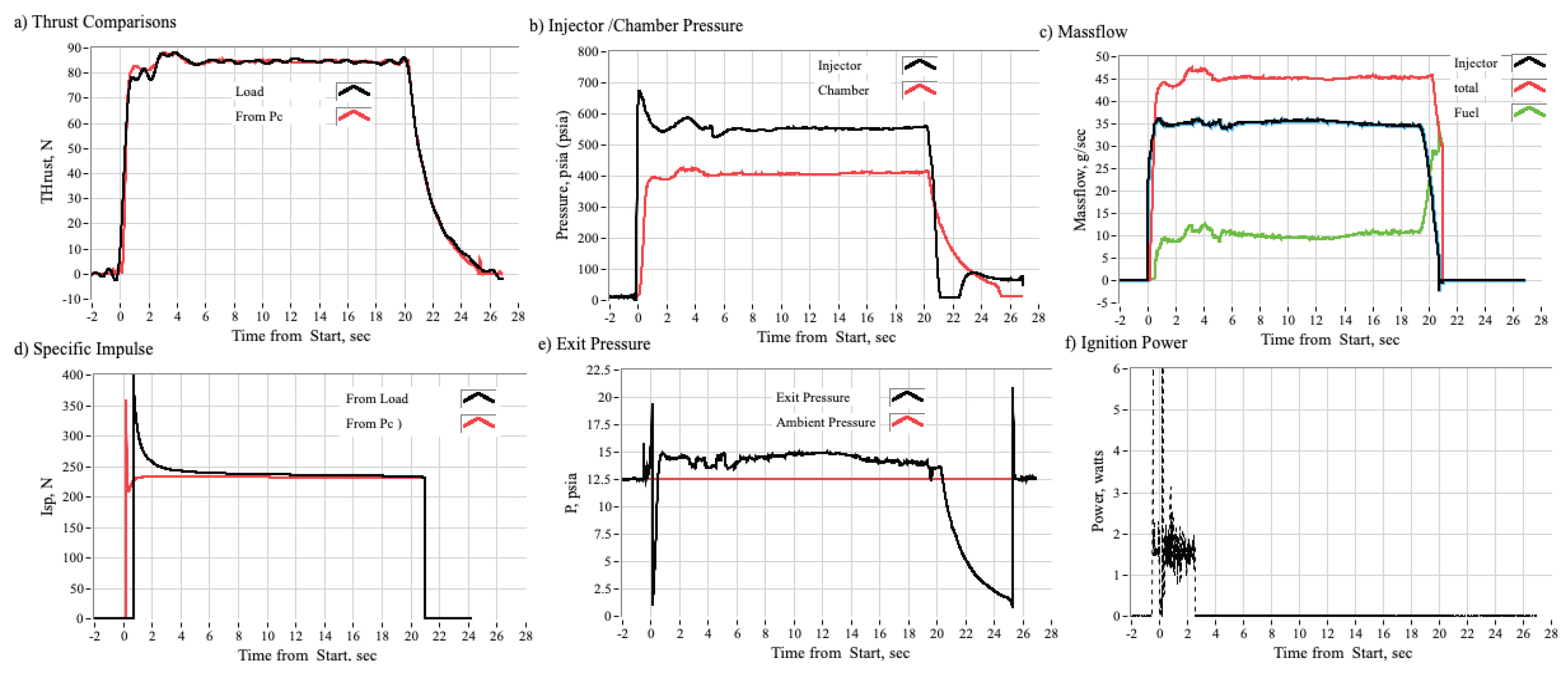
3.2.1. Time History Plots
3.2.2. Exhaust Plume Images
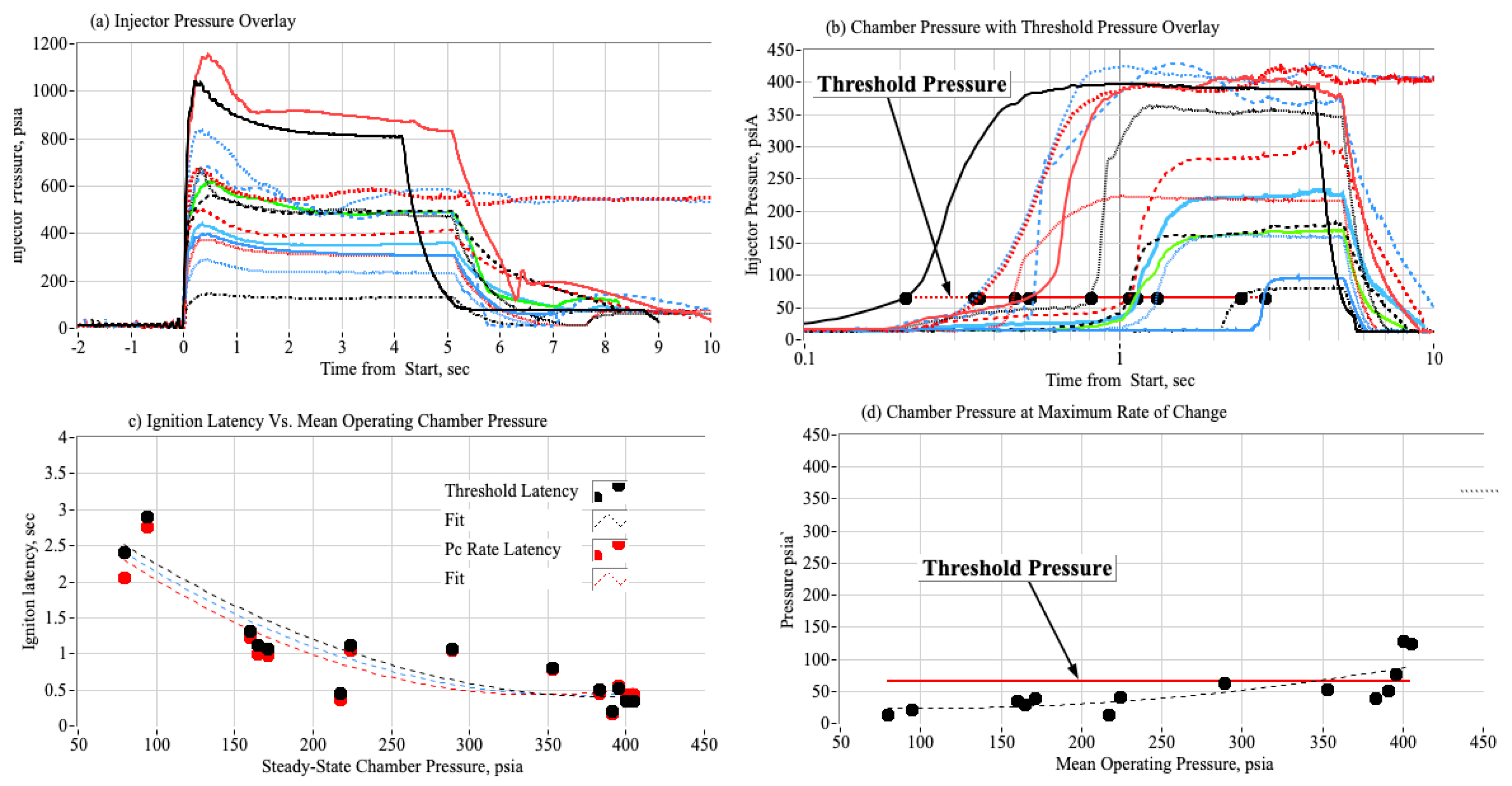
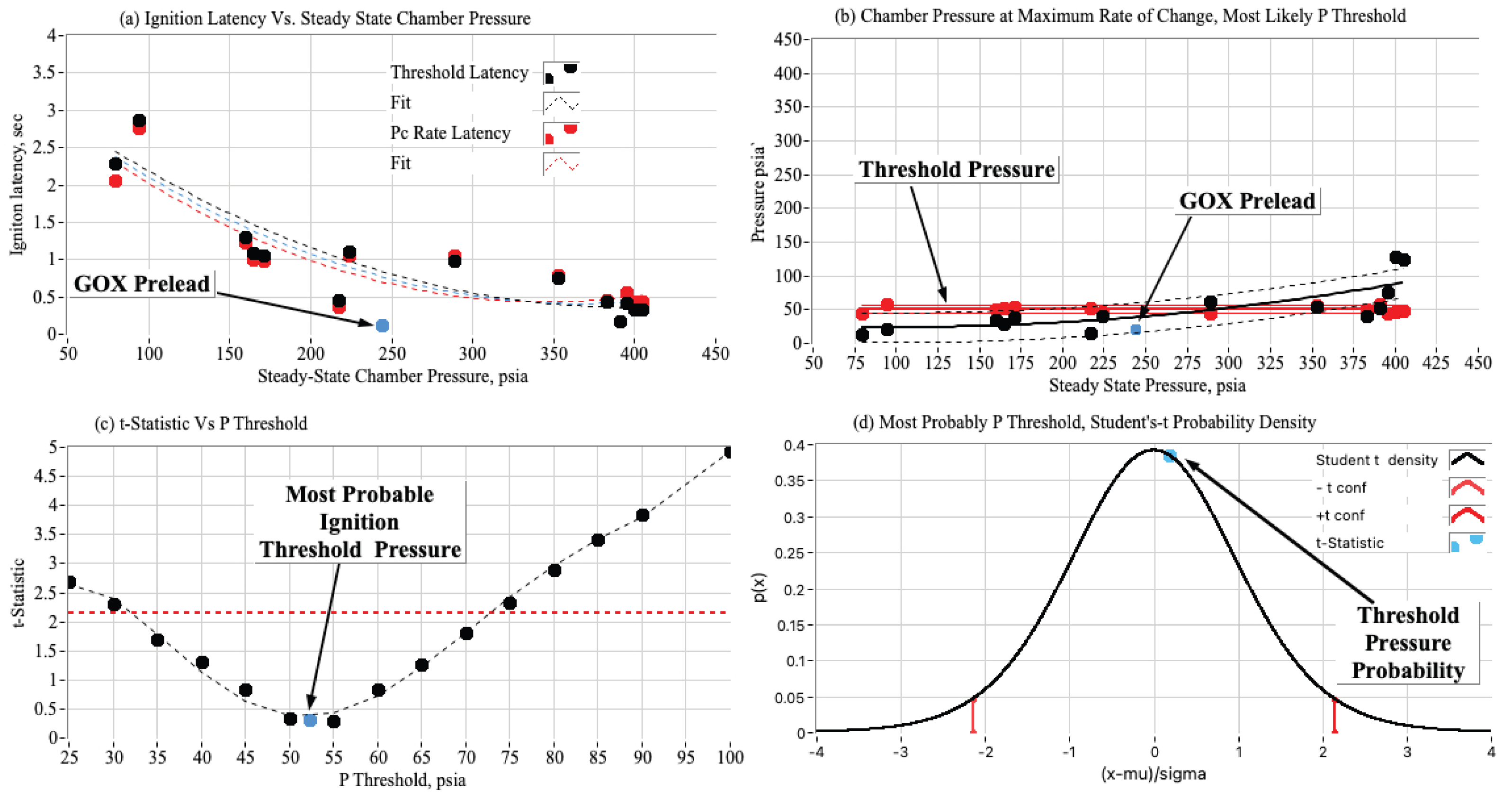
3.3. Ignition Test Summary Plots
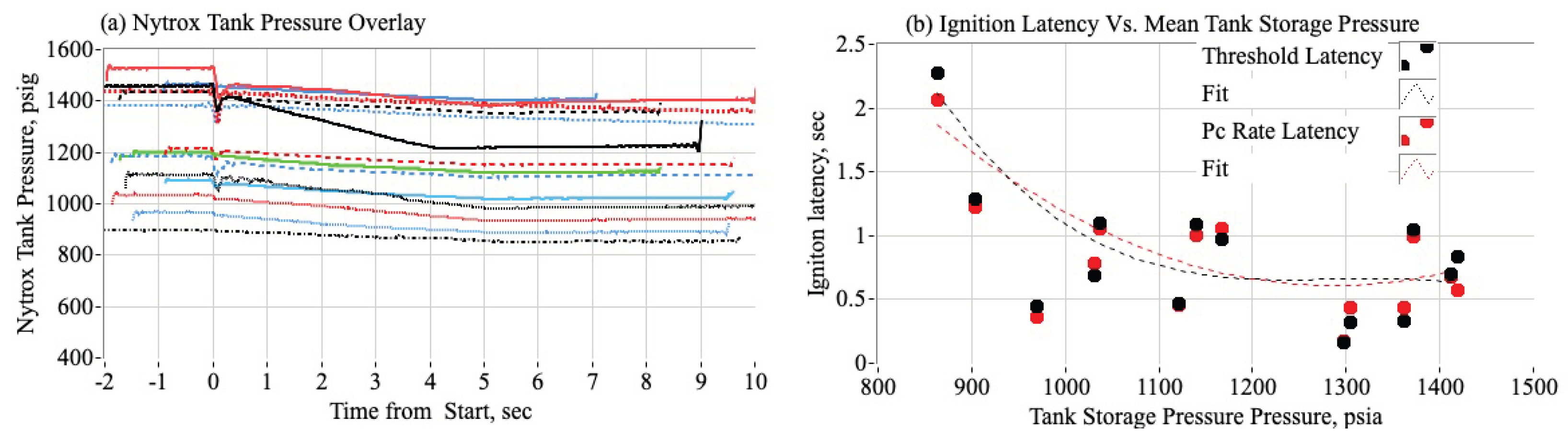
3.3.1. Effect of Chamber Pressure on Nytrox Ignition Latency
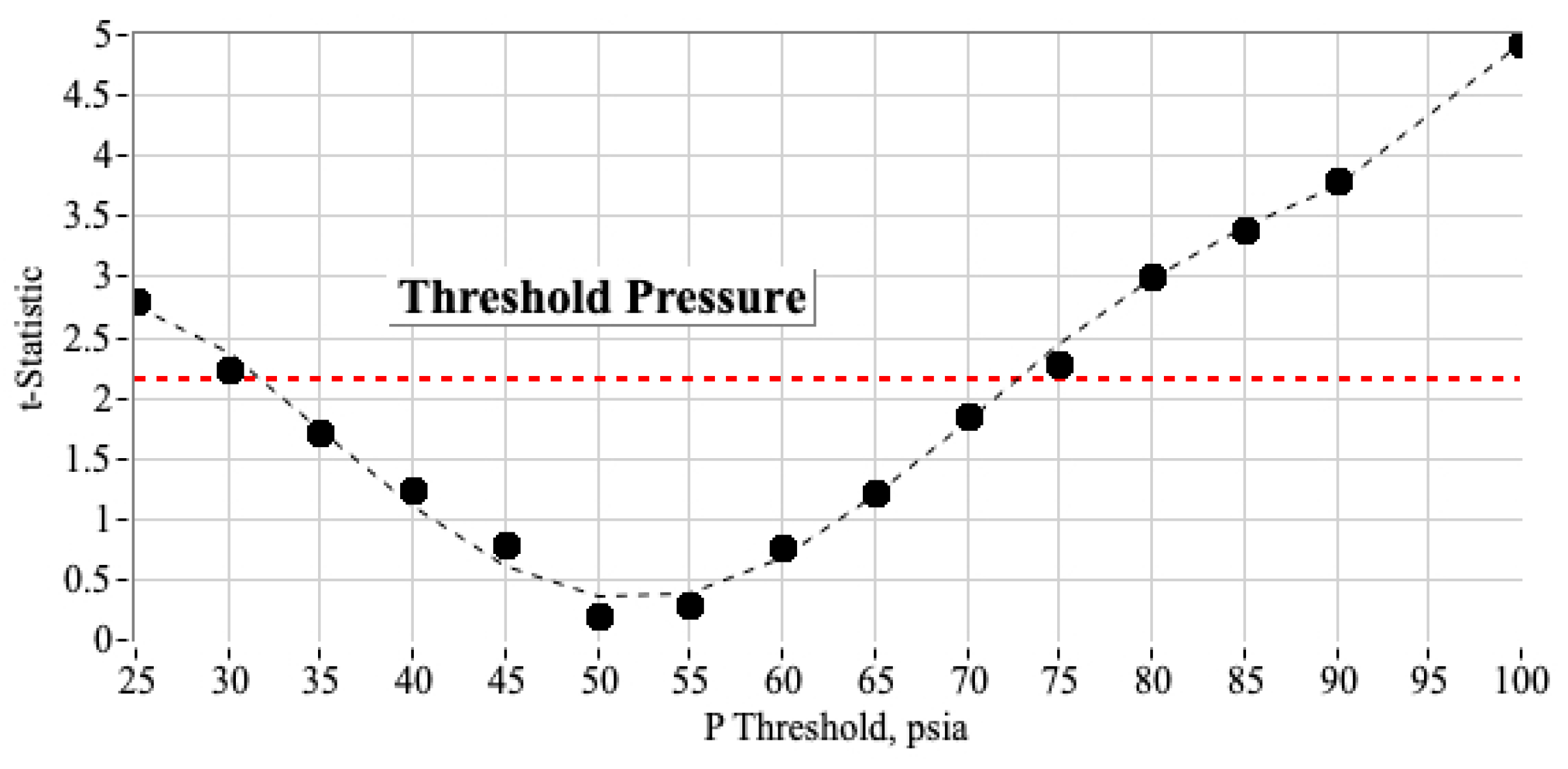
3.3.2. Estimating the Most Likely Ignition Threshold Pressure
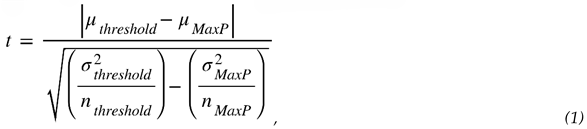
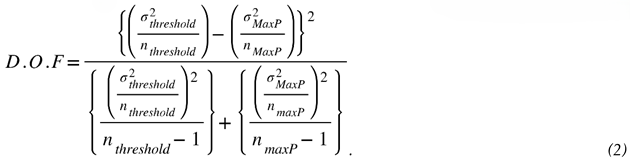
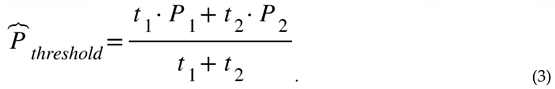
3.3.3. Effect of Nytrox Storage Pressure on Ignition Latency
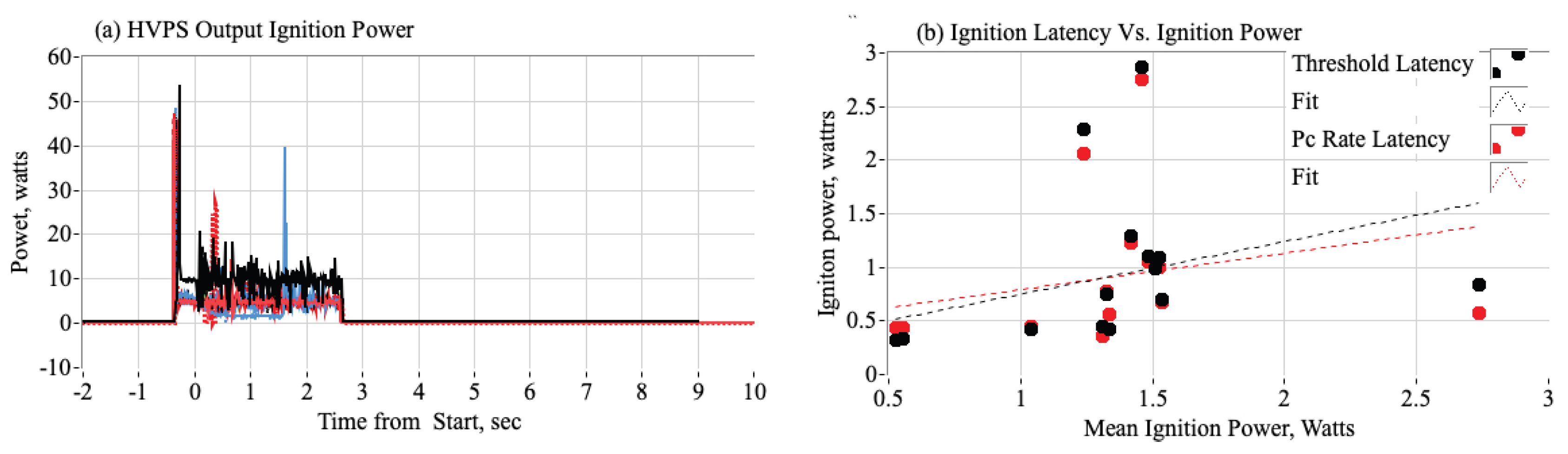
3.3.4. Effect of HVPS Ignition Power on Ignition Latency
4. Discussion
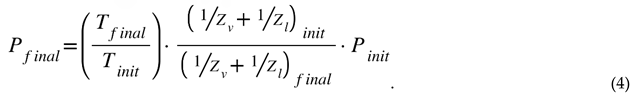
5. Conclusions
6. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Nomenclature
| Symbols |
| N2O = Chemical formula of nitrous oxide |
| n = Number of members in data set |
| nMaxP = Number of samples in max rate data set |
| nthreshold = Number of threshold pressure data samples |
| O2 = Chemical formula of molecular oxygen |
| P = Pressure, kPa |
| Pc = Combustion (chamber) pressure, kPa |
| Pmax = Most probably ignition threshold pressure, psia |
| Pthreshold = Threshold pressure for Nytrox Ignition, psia |
| t = Numerical statistic used for student’s-t hypothesis test |
| Z = Compressibility of a gas or fluid |
| μ = Sample mean |
| σ = Sample standard deviation |
| Acronyms and Abbreviations |
| ABS = Acrylonitrile butadiene styrene |
| BLAST = Ballistics and Survivability Limits Testing (Laboratory) |
| CEA = Chemical Equilibrium with Applications, computer program. |
| D.O.F. = Degrees of freedom |
| FDM = Fused deposition modeling |
| GOX = Gaseous oxygen |
| HPGHP = High-Performance Green Hybrid Propulsion |
| NOS® = Nitrous Oxide Systems trade name |
| OSHA = Occupational Safety and Health Administration |
| RMS = Root mean squared statistic |
| TRL = Technology readiness level |
References
- Anon., “Hazard Analysis of Commercial Space Transportation; Vol. 1: Operations, Vol. 2: Hazards, Vol. 3: Risk Analysis,” U.S. Dept. of Transportation, PB93-199040, Accession No. 00620693, May 1988. h https://www.faa.gov/about/office_org/headquarters_offices/ast/licenses_permits/media/hazard.pdf , (accessed on 4 March 2026).
- Anon., “Department of Defense Interface Standard, Eletromagnetic Environmental Effects requirements for Systems, MIL-STD-464, March, 1997. http://www.tscm.com/MIL-STD-464.pdf, (accessed on 4 March 2026).
- Palermo, E. Fused Deposition Modeling: Most Common 3D Printing Method. LIVESCIENCE. 19 September 2013. Available online: https://www.livescience.com/39810-fused-deposition-modeling.html (accessed on 02 March 2026).
- Stephen A. Whitmore, Stephen L. Merkley, Louis Tonc, and Spencer D. Mathias, "Survey of Selected Additively Manufactured Propellants for Arc Ignition of Hybrid Rockets", J. Propulsion and Power, Vol. 32, No. 6, 2016, pp. 1494-1504. (accessed on 02 March 2026). [CrossRef]
- Whitmore, S. A., Mathias, S. D., and Harvey, R., “High Voltage Breakdown and Arc-Tracking Mechanism of Thermoplastics with Applications to Hybrid Rocket Arc- Ignition,” 53rd AIAA/SAE/ASEE Joint Propulsion Conference, 10-12 July 2017, Atlanta, GA. AIAA 2017-4601. (accessed on 02 March 2026). [CrossRef]
- Hastings, J. K., Zuercher, J. C., Hetzmannseder, E., "Electrical Arcing and Material Ignition Levels," SAE 2004 World Congress & Exhibition, March 08, 004, ISSN: 0148-7191, e-ISSN: 2688-3627. (accessed on 02 March 2026). [CrossRef]
- Jiang, H. Q., Brazis, P. W., Navarro, N, "DC high-energy arcing ignition (HAI) resistance for polymeric materials: Part I: Consistency and repeatability of DC-HAI system," 2012 IEEE Symposium on Product Compliance Engineering Proceedings, Schematic Scholar, 2012. (accessed on 02 March 2026). [CrossRef]
- Whitmore, S. A., Bulcher, A. M., Lewis, Z., and Inkley, N., “Methods and Systems for Restartable Hybrid Rockets,” US Patent 10,774,789 B2, Sept. 15, 2020. https://patents.google.com/patent/US20150322892A1/en, (accessed on 02 March 2026).
- Whitmore, S. A., and Thibaudeau, R. J., “Development of a GOX/ABS Hybrid-Gas Generator System for the NASA Plume Surface Interaction (PSI) Experimental Campaign,” AIAA 2026-2871, AIAA SCITECH 2026 Forum, Orlando FL, Jan. 12-16, 2026. https://arc.aiaa.org/doi/abs/10.2514/6.2026-2871, (accessed on 25 Feb. 2026).
- Whitmore, S. A. Three-Dimensional Printing of “Green” Fuels for Low-Cost Small Spacecraft Propulsion Systems. J. Spacecraft and Rockets (accessed on 4 March 2026). 2017, Vol. 54(No. 6). [Google Scholar] [CrossRef]
- Whitmore, S. A., and Thibaudeau, R. J., “Development of a GOX/ABS Hybrid-Gas Generator System for the NASA Plume Surface Interaction (PSI) Experimental Campaign,” AIAA 2026-2871, AIAA SCITECH 2026 Forum, Orlando FL, Jan. 12-16, 2026. https://arc.aiaa.org/doi/abs/10.2514/6.2026-2871, (accessed on 25 Feb. 2026).
- Whitmore, S. A., and Bulcher, A. M., "A Green Hybrid Thruster Using Moderately Enriched Compressed Air as the Oxidizer", AIAA 2018-4841, 2018 Joint Propulsion Conference, AIAA Propulsion and Energy Forum. (accessed on 02 March 2026). [CrossRef]
- Anon, Nitrous Oxide, NIST Chemistry WebBook, SRD 69, National Institute of Standards and Technology, U.S. Dept. of Commerce. Available online: https://webbook.nist.gov/cgi/cbook.cgi?ID=10024-97-2 (accessed on 25 Feb. 2025).
- Large-Scale Hybrid Motor Testing. In Fundamentals of Hybrid Rocket Combustion and Propulsion; Story, G., Arves, J., Kuo, K. K., Chiaverini, M. J., Eds.; American Institute of Aeronautics and Astronautics: Reston, VA, USA, 2007; pp. 513–552. Available online: https://arc.aiaa.org/doi/10.2514/4.866876 (accessed on 4 March 2026)ISBN (print): 978-1-56347-703-4, eISBN: 978-1-60086-687-6.
- Anon, “Occupational Safety and Health Guideline for Nitrous Oxide,” US Department of Labor, Occupational Health and Safety Administration. 7 March 1996. Available online: https://www.osha.gov/laws-regs/regulations/standardnumber/1910/1910.105 (accessed on 4 March 2026).
- Rhodes, G., W., “Investigation of Decomposition Characteristics of Gaseous and Liquid Nitrous Oxide,” Air Force Weapons Laboratory, Report AD-784 602, Kirtland AFB, New Mexico, July 1974. https://www.freelists.org/archives/arocket/01-2014/pdfEE82jaPU9W.pdf, (accessed on 4 March 2026).
- Bracken, A.B.; Broughton, G.B.; Hill, W. Equilibria for Mixtures of Oxygen and Nitrous Oxide and Carbon Dioxide and Their Relevance to the Storage of N2O/O2 Cylinders for Use in Analgesia. J. Phys. D, Applied Physics (accessed on 4 March 2026). 1970, Vol. 3, 1747–1758. [Google Scholar] [CrossRef]
- Karabeyoglu, M., A., Nitrous Oxide and Oxygen Mixtures (Nytrox) as Oxidizers for Rocket Propulsion Applications. J. Prop. Power (accessed on 4 March 2026). 2014, Vol. 30(No. 3), 696–706. [CrossRef]
- Whitmore, S. A. Nytrox as “Drop-in” Replacement for Gaseous Oxygen in SmallSat Hybrid Propulsion Systems. Aerospace (accessed on 4 March 2026). 2020, Vol. 7(No. 43), 1–38. [Google Scholar] [CrossRef]
- Whitmore, S. A.; Stoddard, R.L. N2O/O2 blends safe and volumetrically efficient oxidizers for small spacecraft hybrid propulsion. Aeronaut. Aerosp. Open Access J. 2019, Vol.2(No. 3). Available online: https://medcraveonline.com/AAOAJ/AAOAJ-03-00097.pdf. (accessed on 4 March 2026). [CrossRef]
- Stoddard, R. L., "Experimental Investigation of N2O/O2 Mixtures as Volumetrically Efficient Oxidizers for Small Spacecraft Hybrid Propulsion Systems," Master Deg. Thesis, Utah State University Digital Commons, December 2019. https://digitalcommons.usu.edu/. , (accessed on 4 March 2026).
- Whitmore, S. A., and Frischkorn, C. I., “Analyzing and Reducing Ignition Latency of a Nytrox/ABS Hybrid Propulsion System,” AIAA 2020-3754, AIAA Propulsion and Energy 2020 Forum Virtual Conference, August 24-28, 2020. https://arc.aiaa.org/doi/abs/10.2514/6.2020-3754, (accessed on 4 March 2026).
- Anon, Full-Cone Spray Nozzle Brass, 1/8 NPT Male, 0.5 gal/min @At 20 PSI, 120 Degree Angle,“ McMaster-Carr. Available online: https://www.mcmaster.com/32885K131/ (accessed on 4 March 2026).
- Whitmore, S. A., and Anthony M. Bulcher, A. M., "Vacuum Test of a Novel Green-Propellant Thruster for Small Spacecraft", AIAA 2017-5044. 53rd AIAA/SAE/ASEE Joint Propulsion Conference, AIAA Propulsion and Energy Forum, 2017. (accessed on 4 March 2026). [CrossRef]
- Anon, ULTRAVOLT C Series High Voltage CAP-Charging Supplies. Advanced Energy, Inc. Available online: https://www.advancedenergy.com/globalassets/resources-root/data-sheets/ultravolt-c-series-data-sheet.pdf (accessed on 4 March 2026).
- Whitmore, S. A., Babb, R. S., Gardner, T. J., Lloyd, K. P, and Stephens, J. C., “Pyrolytic Graphite and Boron Nitride as Low-Erosion Nozzle Materials for Long-Duration Hybrid Rocket Testing, AIAA 2020-3740, AIAA Propulsion and Energy 2020 Forum, August 2020. https://arc.aiaa.org/doi/abs/10.2514/6.2020-3740, (accessed on 4 March 2026).
- Holley, Nitrous Oxide Systems. Available online: https://www.holley.com/brands/nos/ (accessed on 4 March 2026).
- Anderson, J. D., Modern Compressible Flow, 3rd Edition, New York: The McGraw Hill Companies, Inc., 2003, Chapter 4, pp. 127-187. ISBN-13: 978-0072424430. https://libcat.lib.usu.edu/search/i0070016542.
- Gordon, S., and McBride, B. J., “Computer Program for Calculation of Complex Chemical Equilibrium Compositions and Applications, I. Analysis” National Aeronautics and Space Administration, Cleveland, Ohio, Tech. Rep. NASA RP-1311, 1994. https://ntrs.nasa.gov/api/citations/19950013764/downloads/19950013764.pdf, (accessed on 23 Jan. 2026).
- Gordon, S., and McBride, B. J., “Computer Program for Calculation of Complex Chemical Equilibrium Compositions and Applications, II. User’s Manual and Program Description” National Aeronautics and Space Administration, Cleveland, Ohio, Tech. Rep. NASA RP-1311-2, 1994. https://ntrs.nasa.gov/api/citations/19960044559/downloads/19960044559.pdf, (accessed on 23 Jan. 2026).
- Anon. ,“18.6: Factors Affecting Reaction Rate,“ LibreTexts TM, Chemistry. https://chem.libretexts.org/Bookshelves/Introductory_Chemistry/Introductory_Chemistry_(CK-12)/18%3A_Kinetics/18.06%3A_Factors_Affecting_Reaction_Rate, (Accesses 5 March 2025.).
- Otnes, R. K.; Enochson, L. Applied Time Series Analysis; (accessed on 5 March 2026); Wiley: New York, 1978; pp. 219–260. [Google Scholar] [CrossRef]
- Beckwith, T. G., Marangoni, R. D., and Lienhard V, J. H., Mechanical Measurements, 6th Ed., Prentice Hall, 2006, pp.43- 73. ISBN-13: 978-0201847659, ISBN-10: 0201847655. https://archive.org/details/mechanicalmeasur0000beck_q2k3, (accessed 5 March 2026).
- Peng, D.-Y.; Robinson, D. B. A New Two Constant Equation of State. Industrial and Engineering Chemistry Fundamentals (accessed on 23 Jan. 2026).. 1976, Vol. 15(No. 1), 59–64. [Google Scholar] [CrossRef]
- Zudkevitch, D.; Joffe, J. Correlation and Prediction of Vapor- Liquid Equilibria with the Redlich-Kwong Equation of State. AIChE Journal (accessed on 23 Jan. 2026). 1970, Vol. 16(No. 1), 112–119. [Google Scholar] [CrossRef]
- Anon., NATIONAL POLICY ON THE TRANSFER OF SCIENTIFIC, TECHNICAL AND ENGINEERING INFORMATION, National Security Decision Directives (NSDDs), 1985. https://irp.fas.org/offdocs/nsdd/nsdd- 189.htm. (Accessed 7 Feb. 2026).



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



