Submitted:
11 March 2026
Posted:
11 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials and Animals
2.2. Methods
2.2.1. Synthesis of Camptothecin Nanowires
2.2.2. Preparation and Characterization of SA-SS-CPT Nanowires
2.2.3. Hemolysis Assay
2.2.4. Camptothecin Release
2.2.5. Pharmacokinetics Study
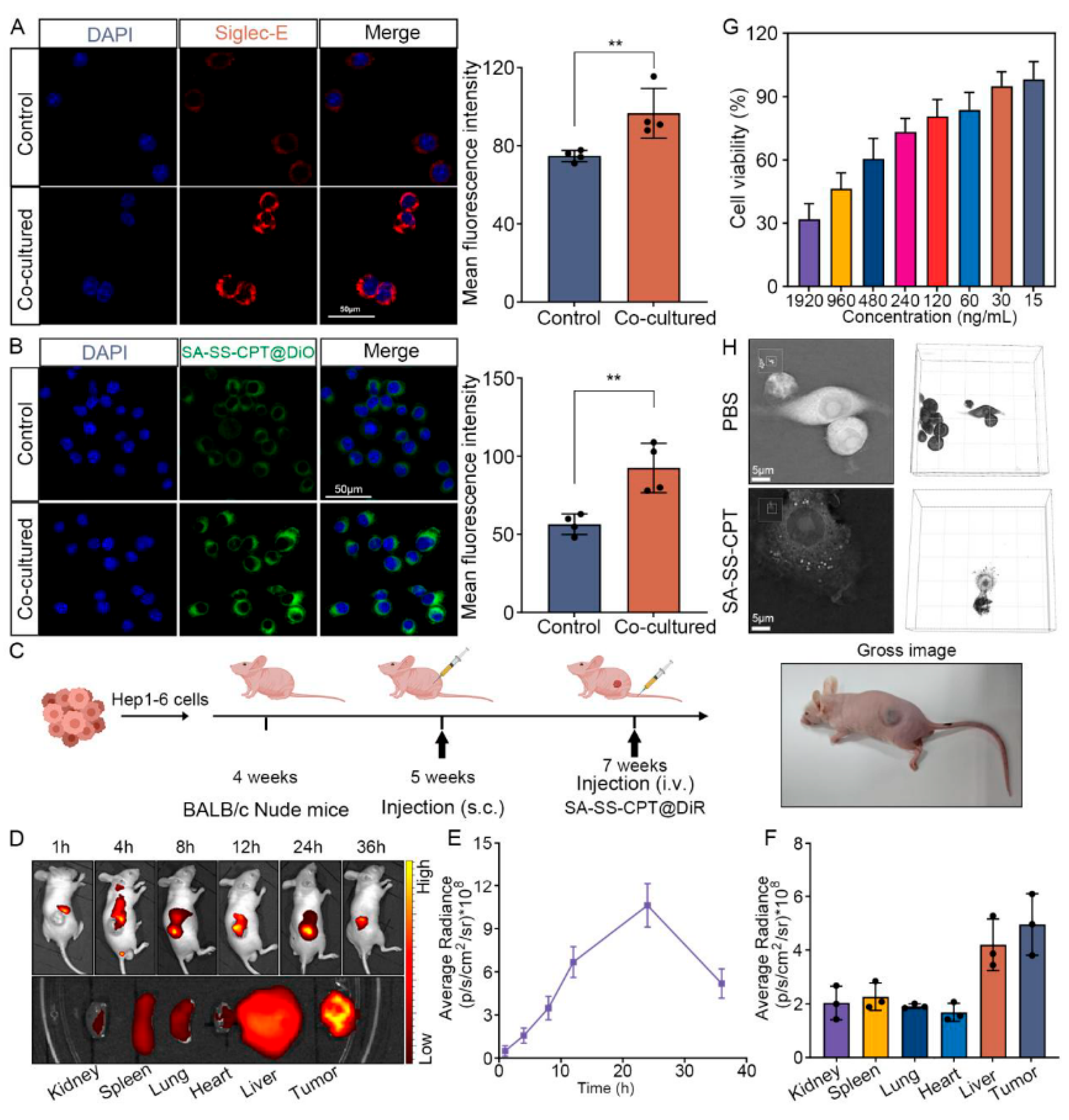
2.2.6. Immunofluorescence Analysis of Siglec-E Expression
2.2.7. Cellular Uptake of SA-SS-CPT
2.2.8. In Vivo Imaging
2.2.9. In Vitro Cytotoxicity of SA-SS-CPT
2.2.10. 3D Nanolive Imaging for Macrophage Polarization
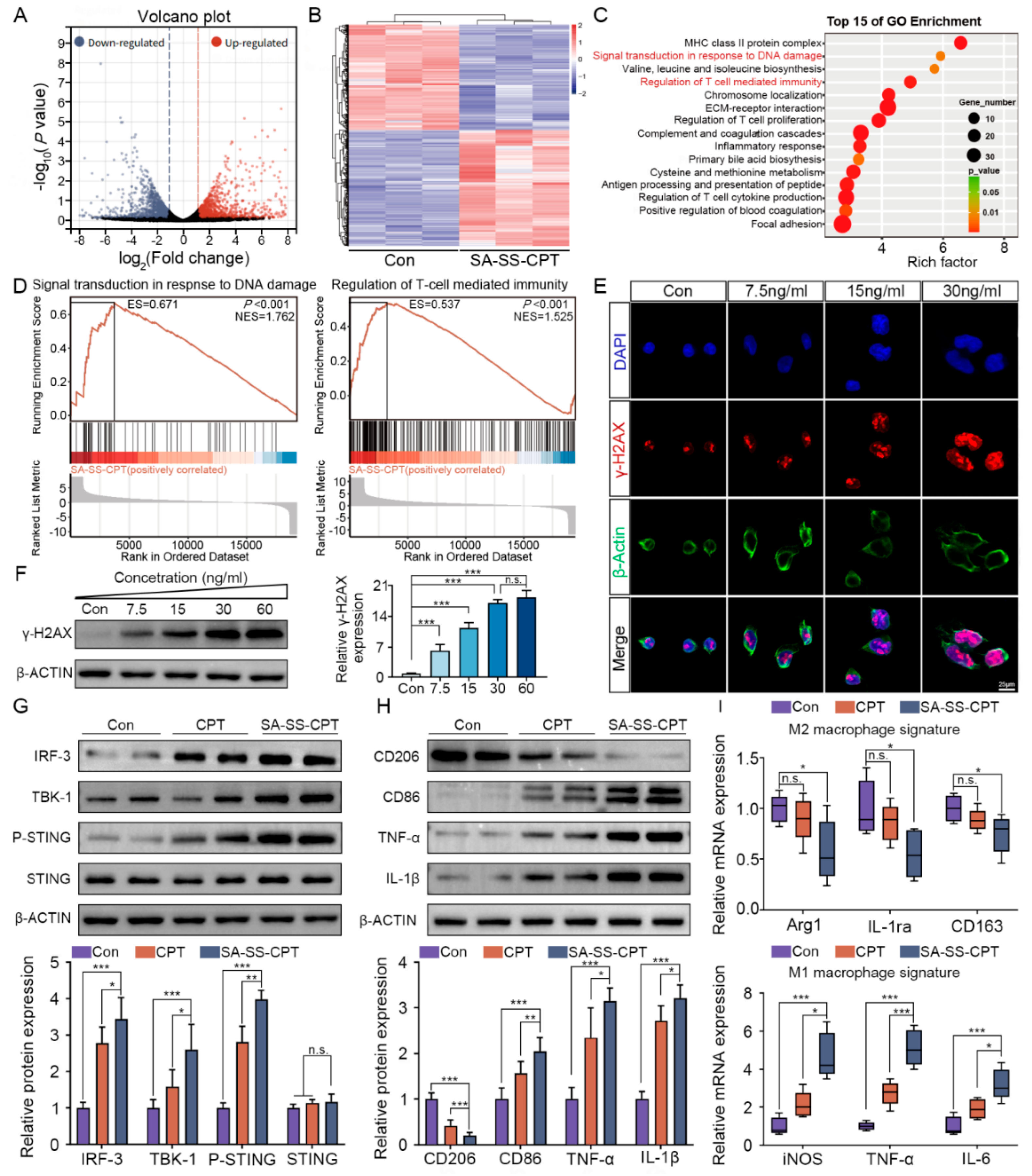
2.2.11. RNA-Seq Analysis
2.2.12. Immunofluorescence Analysis
2.2.13. Detection of DNA Damage and the cGAS-STING Pathway
2.2.14. RT-PCR Analysis of Inflammatory Factors
| Primers | Sequences (5’------3’) | |
| Arg-1 | Forward: | CAGCACTGAGGAAAGCTGGT |
| Reverse: | CAGACCGTGGGTTCTTCACA | |
| IL-1ra | Forward: | TAAGCAACCACCTTGAGCCT |
| Reverse: | CCAGCAGATTTCCATCCCGA | |
| CD163 | Forward: | TGCTGTCACTAACGCTCCTG |
| Reverse: | CATTGCATGCCAGGTCATCG | |
| iNOS | Forward: | TGTCGCAGCTCCCTATCTTG |
| Reverse: | CATTGGCCAGCTGCTTTTGC | |
| TNF-α | Forward: | TGTCTCAGCCTCTTCTCATTCC |
| Reverse: | TGAGGGTCTGGGCCATAGAAC | |
| IL-6 | Forward: | TAGTCCTTCCTACCCCAATTTCC |
| Reverse: | TTGGTCCTTAGCCACTCCTTC |
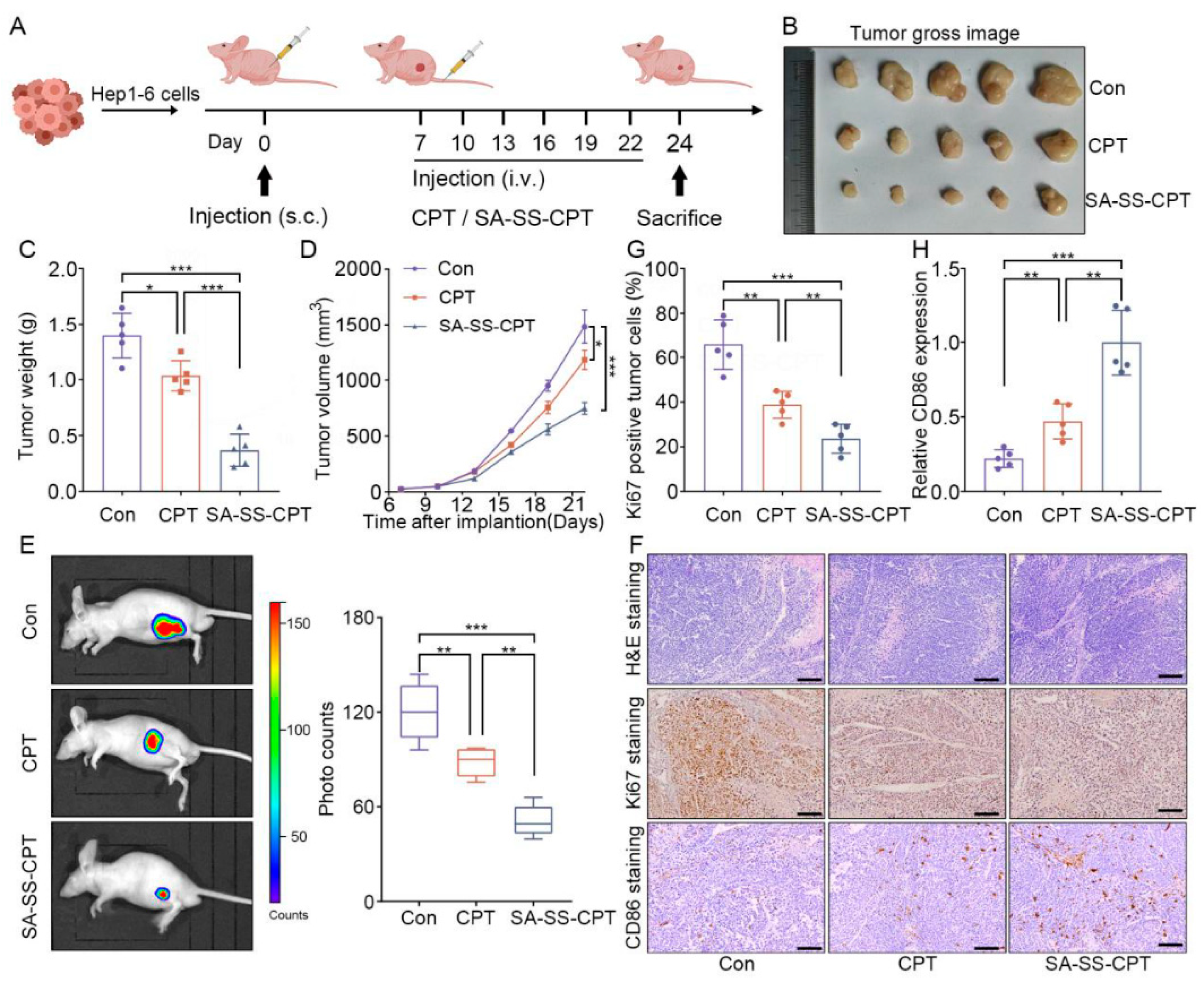
2.2.15. Establishment of Tumor-Bearing Mouse Model
2.2.16. Hematoxylin and Eosin (H&E) Staining
2.2.17. Ki67 Immunohistochemical Staining
2.2.18. Immunohistochemistry for CD86
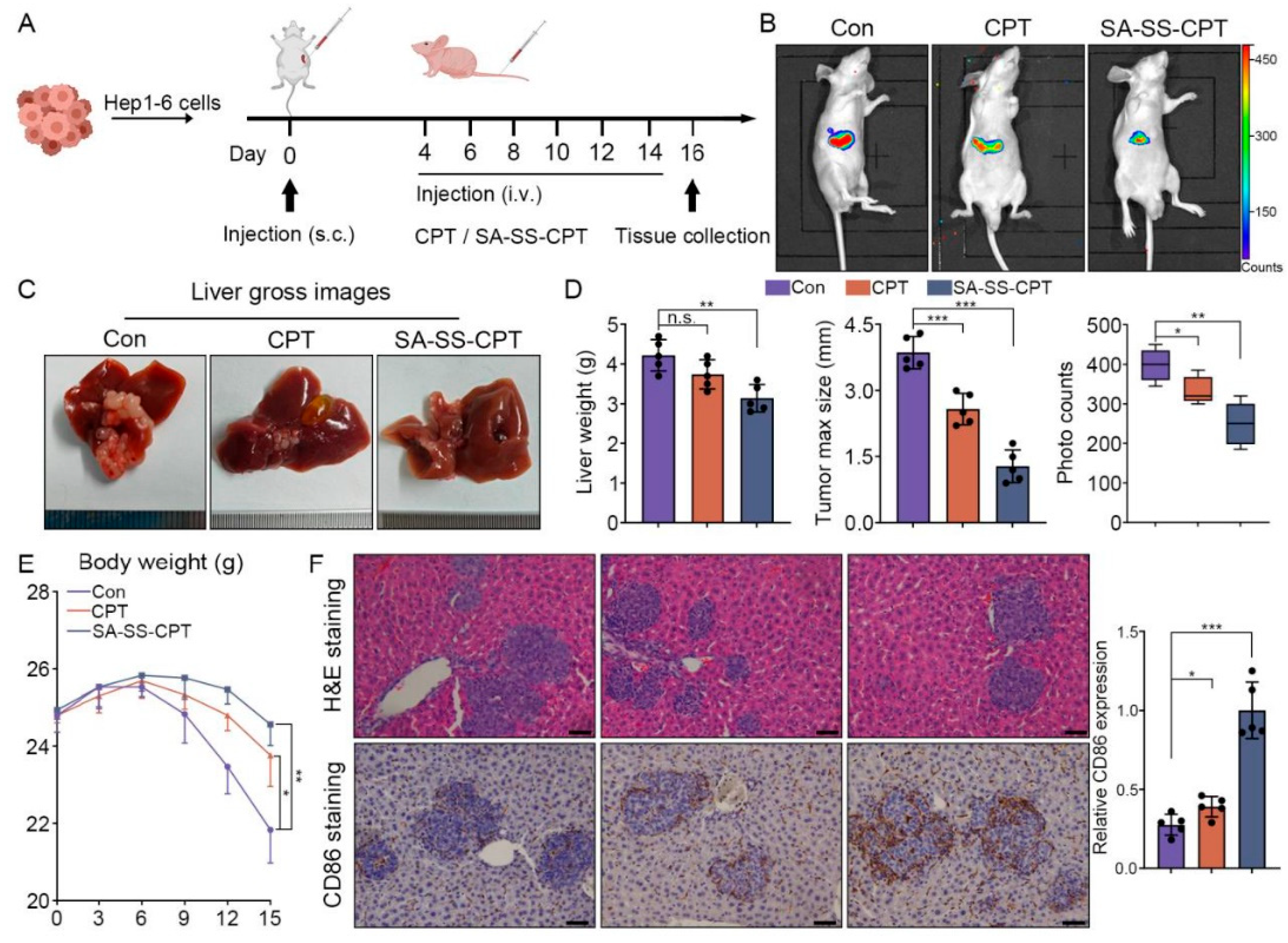
2.2.19. Analysis of Liver Metastasis
2.2.20. SA-SS-CPT Enhances the Immunotherapeutic Efficacy of PD-L1 in Mice
2.3. Statistical Analysis
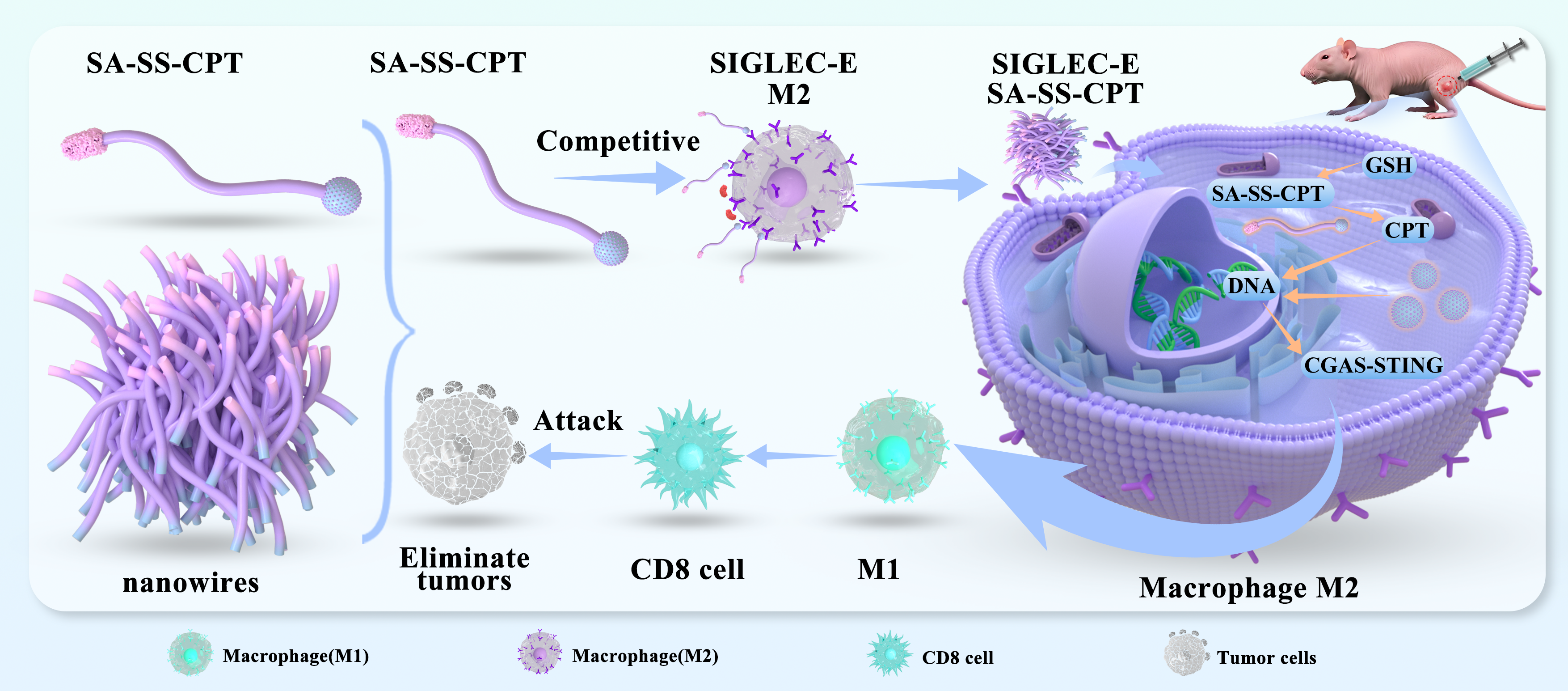
3. Results
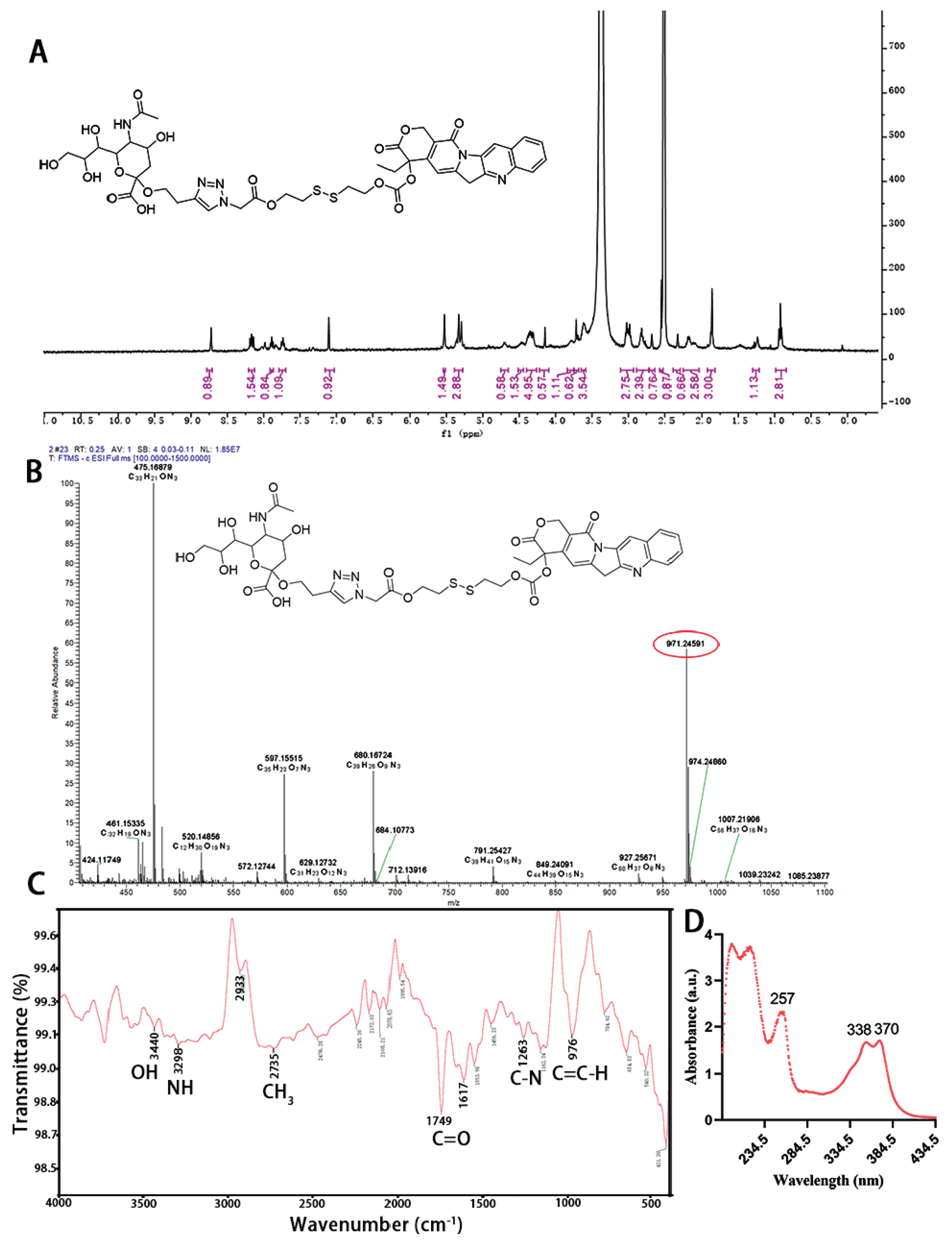
3.1. Synthesis of SA-SS-CPT
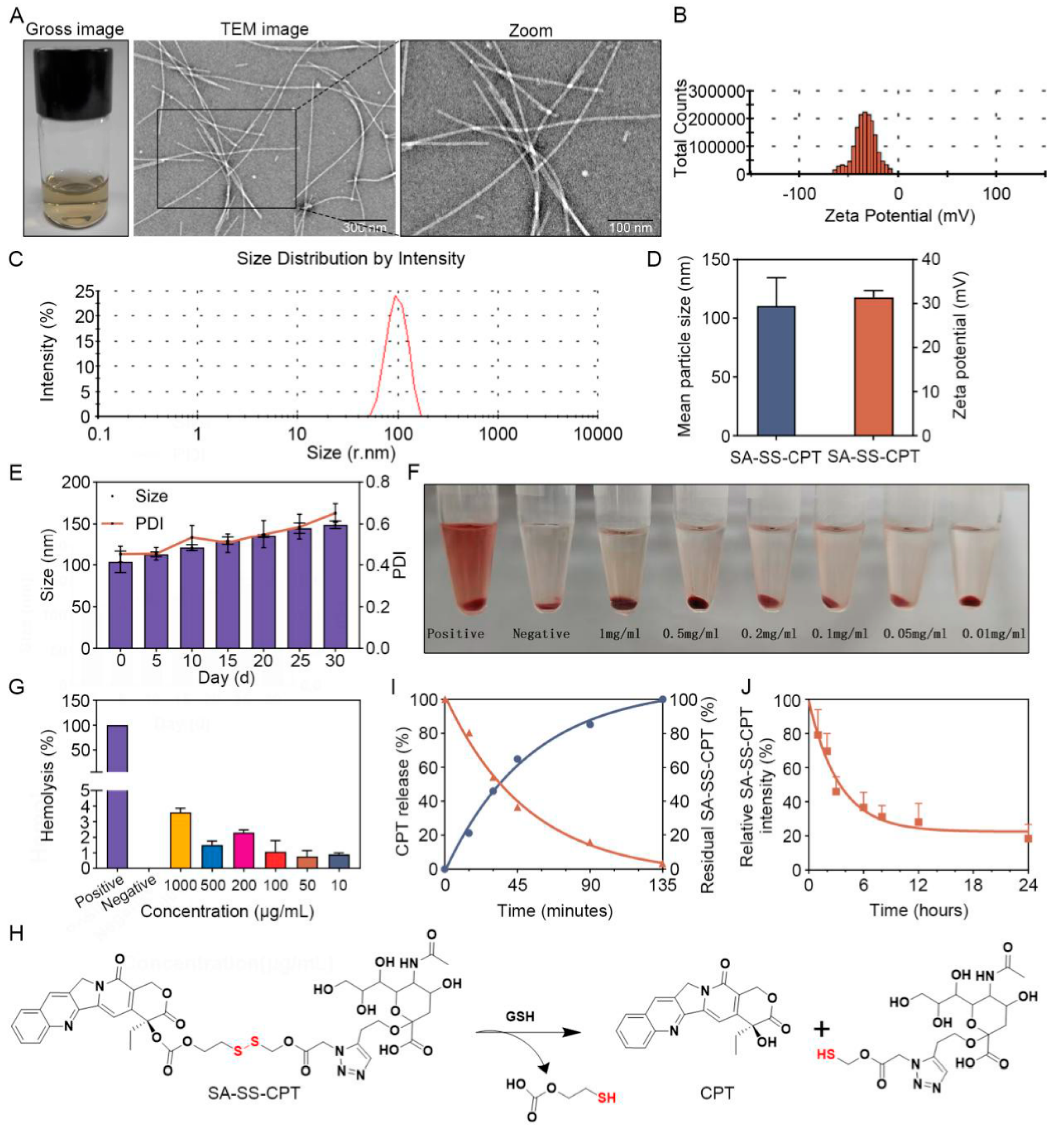
3.2. Characteristics of SA-SS-CPT Nanowires
3.3. Targeting Evaluation of SA-SS-CPT Nanowires
3.4. SA-SS-CTP Drived DNA Damage-Dependent M1 Macrophage Polarization
3.5. Tumor Proliferation Evaluation
3.6. Evaluation of Tumor Metastasis
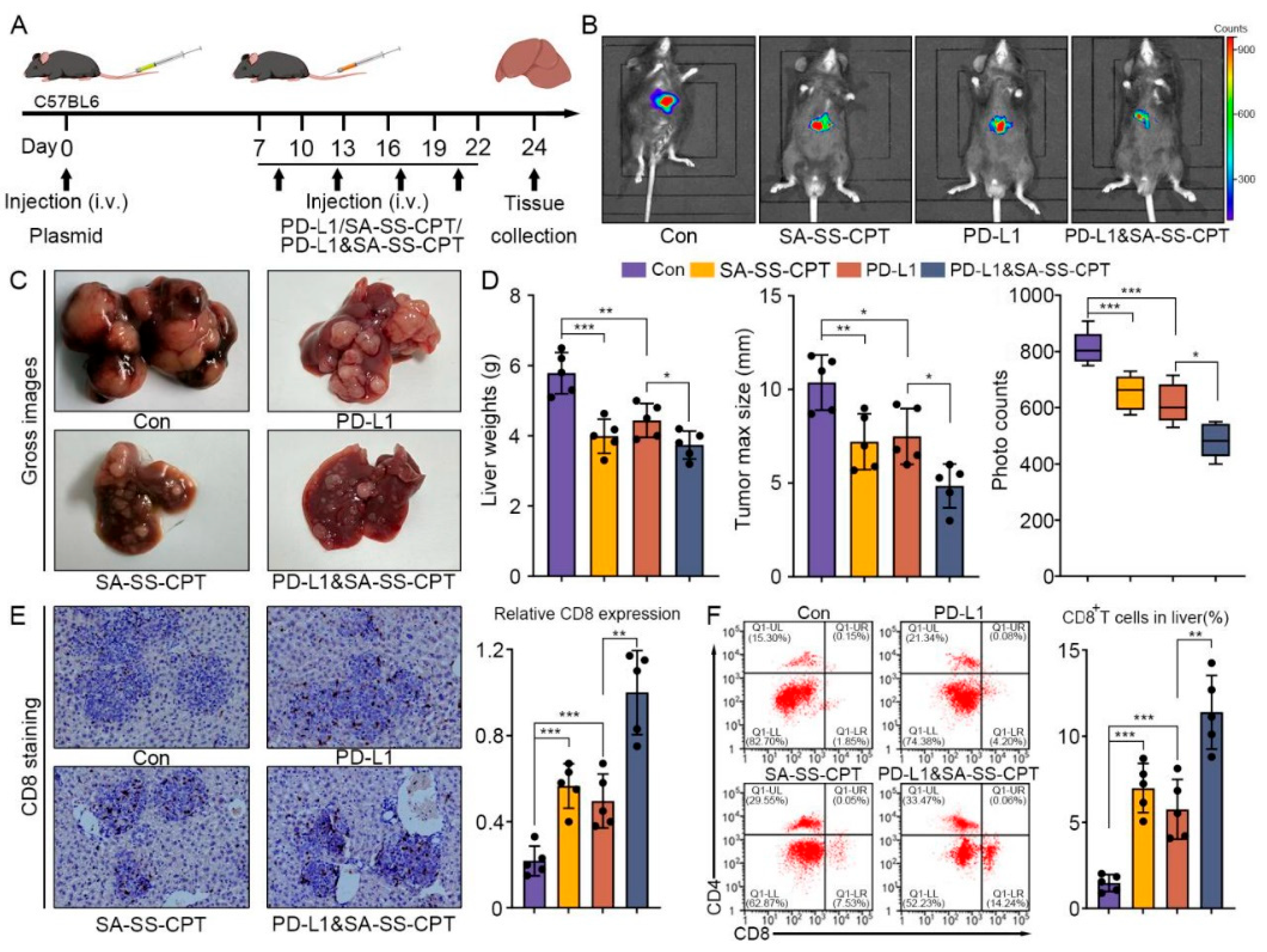
3.7. SA-SS-CPT Enhances the Immunotherapeutic Efficacy of PD-L1 in Mice
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, C.A.O.; Cheng, G.Y.; Zhang, S.; et al. Changing epidemiology of hepatocellular carcinoma in Asia. Liver Int. 2022, 42, 2029–2041. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Pinyol, R.; Yarchoan, M.; Singal, A.G.; Marron, T.U.; Schwartz, M.; Pikarsky, E.; Kudo, M.; Finn, R.S. Adjuvant and neoadjuvant immunotherapies in hepatocellular carcinoma. Nat Rev Clin Oncol. 2024, 21, 294–311. [Google Scholar] [CrossRef] [PubMed]
- Giulia, G.; Pierluigi, P.; Giuseppina, A.; et al. Modulating Tumor-Associated Macrophage Polarization by Synthetic and Natural PPARγ Ligands as a Potential Target in Breast Cancer. CELLS 2020, 5, 2. [Google Scholar]
- Chen, G.; Fu, Y.; Guo, E.S.; et al. BRD4 Inhibition by AZD5153 Promotes Antitumor Immunity via Depolarizing M2 Macrophages. FRONTIERS IN IMMUNOLOGY 2020, 5, 9. [Google Scholar]
- Enyong, D.; Leng, H.; Jiao, L.; et al. Autophagy-dependent ferroptosis drives tumor-associated macrophage polarization via release and uptake of oncogenic KRAS protein. AUTOPHAGY 2020, 14, 3. [Google Scholar]
- Qian, C.; Lu, J.; Man, L.S.; et al. RIPK3 Orchestrates Fatty Acid Metabolism in Tumor-Associated Macrophages and Hepatocarcinogenesis. CANCER IMMUNOLOGY RESEARCH 2020, 8, 2. [Google Scholar]
- Zhang, J.; Zhou, X.; Hao, H. Macrophage phenotype-switching in cancer. European Journal of Pharmacology 2022, 931, 175229. [Google Scholar] [CrossRef]
- Visser, E.A.; Moons, S.J.; Timmermans, S.; et al. Sialic acid O-acetylation: From biosynthesis to roles in health and disease. J Biol Chem. 2021, 297, 100906. [Google Scholar] [CrossRef]
- Zhang, X.; Dou, P.; Akhtar, M.L.; et al. NEU4 inhibits motility of HCC cells by cleaving sialic acids on CD44. Oncogene 2021, 40, 5427–5440. [Google Scholar] [CrossRef]
- Tang, X.; Sui, D.; Liu, M.; et al. Targeted delivery of zoledronic acid through the sialic acid-Siglec axis for killing and reversal of M2 phenotypic tumor-associated macrophages-A promising cancer immunotherapy. International Journal of Pharmaceutics 2020, 590, 119929. [Google Scholar] [CrossRef]
- Häuselmann, I.; Borsig, L. Altered tumor-cell glycosylation promotes metastasis. Front Oncol. 2014, 4, 28. [Google Scholar] [CrossRef]
- Läubli, H.; Kawanishi, K.; George Vazhappilly, C.; et al. Tools to study and target the Siglec-sialic acid axis in cancer. FEBS J. 2021, 288, 6206–6225. [Google Scholar] [CrossRef]
- Xiao, N.; Zhu, X.; Li, K.; et al. Blocking siglec-10(hi) tumor-associated macrophages improves anti-tumor immunity and enhances immunotherapy for hepatocellular carcinoma. Exp Hematol Oncol. 2021, 10, 36. [Google Scholar] [CrossRef]
- Dusoswa, S.A.; Horrevorts, S.K.; Ambrosini, M.; et al. Glycan modification of glioblastoma-derived extracellular vesicles enhances receptor-mediated targeting of dendritic cells. J Extracell Vesicles 2019, 8, 1648995. [Google Scholar] [CrossRef] [PubMed]
- Adams, O.J.; Stanczak, M.A.; Von Gunten, S.; et al. Targeting sialic acid-Siglec interactions to reverse immune suppression in cancer. Glycobiology 2018, 28, 640–647. [Google Scholar] [CrossRef] [PubMed]
- Boudjedir, A.; Kraim, K.; Saihi, Y.; et al. A computational molecular docking study of camptothecin similars as inhibitors for topoisomerase 1. Structural Chemistry 2021, 32, 689–97. [Google Scholar] [CrossRef]
- Tomicic, M.T.; Kaina, B. Topoisomerase degradation, DSB repair, p53 and IAPs in cancer cell resistance to camptothecin-like topoisomerase I inhibitors. Biochim Biophys Acta 2013, 1835, 11–27. [Google Scholar] [CrossRef]
- Chiou, W.F.; Chou, C.J.; Chen, C.F. Camptothecin suppresses nitric oxide biosynthesis in RAW 264.7 macrophages. Life Sciences 2001, 69, 625–635. [Google Scholar] [CrossRef]
- Mollica, A.; Stefanucci, A.; Fau-Feliciani, F.; Cacciatore, I.; et al. Delivery methods of camptothecin and its hydrosoluble analogue irinotecan for treatment of colorectal cancer. Curr Drug Deliv. 2012, 9, 122–31. [Google Scholar] [CrossRef]
- Bansal, A.A.-O.; Simon, M.A.-O.X. Glutathione metabolism in cancer progression and treatment resistance. J Cell Biol. 2018, 217, 2291–2298. [Google Scholar] [CrossRef]
- MA, Z.; GAO, X.; RAZA, F.A.-O.; et al. Design of GSH-Responsive Curcumin Nanomicelles for Oesophageal Cancer Therapy. Pharmaceutics 2022, 14, 1802. [Google Scholar] [CrossRef]
- Hiroshi, H.; Toru, S.; Shinji, I.; et al. Constitutively expressed Siglec-E inhibits LPS-induced CCR7, but enhances IL-4-induced CD200R expression in human macrophages. BIOSCIENCE BIOTECHNOLOGY AND BIOCHEMISTRY 2016, 1, 3. [Google Scholar]
- Rifeng, C.; Di, Y.; Linxia, S. Overexpression of CD86 enhances the ability of THP-1 macrophages to defend against Talaromyces marneffei. IMMUNITY INFLAMMATION AND DISEASE 2022, 2, 7. [Google Scholar]
- Schmidt Keith, T.; Peer Cody, J.; Huitema Alwin, D.R.; et al. Measurement of NLG207 (formerly CRLX101) nanoparticle-bound and released camptothecin in human plasma. JOURNAL OF PHARMACEUTICAL AND BIOMEDICAL ANALYSIS 2020, 3, 1. [Google Scholar] [CrossRef] [PubMed]
- Nasim, A.; Mehriar, A.; Faramarz, M. Molecular Insight into the Interaction between Camptothecin and Acyclic Cucurbit[4]urils as Efficient Nanocontainers in Comparison with Cucurbit[7]uril: Molecular Docking and Molecular Dynamics Simulation. JOURNAL OF CHEMICAL INFORMATION AND MODELING 2020, 5, 3. [Google Scholar]
- Diana, C.; Laura, F.; Nuno, R.F. Cyclodextrin-based delivery systems for in vivo-tested anticancer therapies. DRUG DELIVERY AND TRANSLATIONAL RESEARCH 2020, 5, 5. [Google Scholar]
- Jie, C.; Xiaoheng, F.; Shangcong, H. Novel polymeric micelles as enzyme-sensitive nuclear-targeted dual-functional drug delivery vehicles for enhanced 9-nitro-20(S)-camptothecin delivery and antitumor efficacy. NANOSCALE 2020, 5, 1. [Google Scholar]
- Brian, K.W.; Prud'homme, R.K. Encapsulation and Controlled Release of a Camptothecin Prodrug from Nanocarriers and Microgels: Tuning Release Rate with Nanocarrier Excipient Composition. MOLECULAR PHARMACEUTICS 2021, 4, 5. [Google Scholar]
- Bing, X.; Mengmeng, Y.; Fei, Z.; et al. Prostate-Specific Membrane Antigen and Esterase Dual Responsive Camptothecin-Oligopeptide Self-Assembled Nanoparticles for Efficient Anticancer Drug Delivery. INTERNATIONAL JOURNAL OF NANOMEDICINE 2021, 6, 5. [Google Scholar]
- Avelino, C.; Pablo, B.; Eva, R.-B. Silica-Based Stimuli-Responsive Systems for Antitumor Drug Delivery and Controlled Release. PHARMACEUTICS 2022, 5, 5. [Google Scholar]
- Xiaoqing, Z.; Xiaomei, R.; Jiayin, T.; et al. Hyaluronic acid reduction-sensitive polymeric micelles achieving co-delivery of tumor-targeting paclitaxel/apatinib effectively reverse cancer multidrug resistance. In DRUG DELIVERY; 2020. [Google Scholar]
- Preeti, K.; Milan, P.; Himanshu, B.; et al. Chlorin e6 Conjugated Methoxy-Poly(Ethylene Glycol)-Poly(D,L-Lactide) Glutathione Sensitive Micelles for Photodynamic Therapy. PHARMACEUTICAL RESEARCH 2020, 4, 3. [Google Scholar]
- Roshni, I.; Tam, N.; Dona, P.; et al. Glutathione-responsive biodegradable polyurethane nanoparticles for lung cancer treatment. JOURNAL OF CONTROLLED RELEASE 2020, 11, 5. [Google Scholar]
- Ying, P.; Youqing, S.; Jianbin, T.; et al. Glutathione-Specific and Intracellularly Labile Polymeric Nanocarrier for Efficient and Safe Cancer Gene Delivery. ACS APPLIED MATERIALS & INTERFACES 2020, 8, 2. [Google Scholar]
- Hyukjin, C.; Julie M, L.; Henghong, L.; et al. Wip1 directly dephosphorylates gamma-H2AX and attenuates the DNA damage response. CANCER RESEARCH 2010, 16, 6. [Google Scholar]
- Tuo, L.; Zhijian, J.C. The cGAS-cGAMP-STING pathway connects DNA damage to inflammation, senescence, and cancer. JOURNAL OF EXPERIMENTAL MEDICINE 2018, 10, 6. [Google Scholar]
- Rong, S.; Disheng, L.; Xiaoning, W.; et al. DNA Damage and Activation of cGAS/STING Pathway Induce Tumor Microenvironment Remodeling. FRONTIERS IN CELL AND DEVELOPMENTAL BIOLOGY 2022, 4, 3. [Google Scholar]
- Can, L.; Weiwei, L.; Fang, W.; et al. DNA damage-triggered activation of cGAS-STING pathway induces apoptosis in human keratinocyte HaCaT cells. MOLECULAR IMMUNOLOGY 2021, 3. [Google Scholar]
- Boyi, Y.; Xuanyi, L.; Xianglong, F.; et al. Gadolinium Oxide Nanoparticles Reinforce the Fractionated Radiotherapy-Induced Immune Response in Tri-Negative Breast Cancer via cGAS-STING Pathway. INTERNATIONAL JOURNAL OF NANOMEDICINE 2023, 6, 5. [Google Scholar]
- Ming, T.; Shasha, Y.; Hongliang, Z.; et al. The P286R mutation of DNA polymerase ε activates cancer-cell-intrinsic immunity and suppresses endometrial tumorigenesis via the cGAS-STING pathway. CELL DEATH & DISEASE 2024, 9, 6. [Google Scholar]
- Ganesh, R.; Khatri, L.; Martina, O.; et al. Anti-PD-1 Induces M1 Polarization in the Glioma Microenvironment and Exerts Therapeutic Efficacy in the Absence of CD8 Cytotoxic T Cells. CLINICAL CANCER RESEARCH 2020, 10, 2. [Google Scholar]
- Lin, X.; Jiang, L.; JunYan, L.; et al. Methionine restriction promotes the polarization of macrophages towards M1 and the immunotherapy effect of PD-L1/PD-1 blockades by inhibiting the secretion of MIF by gastric carcinoma cells. TRANSLATIONAL ONCOLOGY 2024, 4, 1. [Google Scholar]
- Lili, H.; Jingbo, Y.; Jinjin, Z.; et al. PD-L1 in melanoma and extracellular vesicles promotes local and regional immune suppression through M2-like macrophage polarization. AMERICAN JOURNAL OF PATHOLOGY 2024, 3, 6. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



