Submitted:
09 March 2026
Posted:
11 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Thermo-Chemical Analysis of ABS Fuel Material
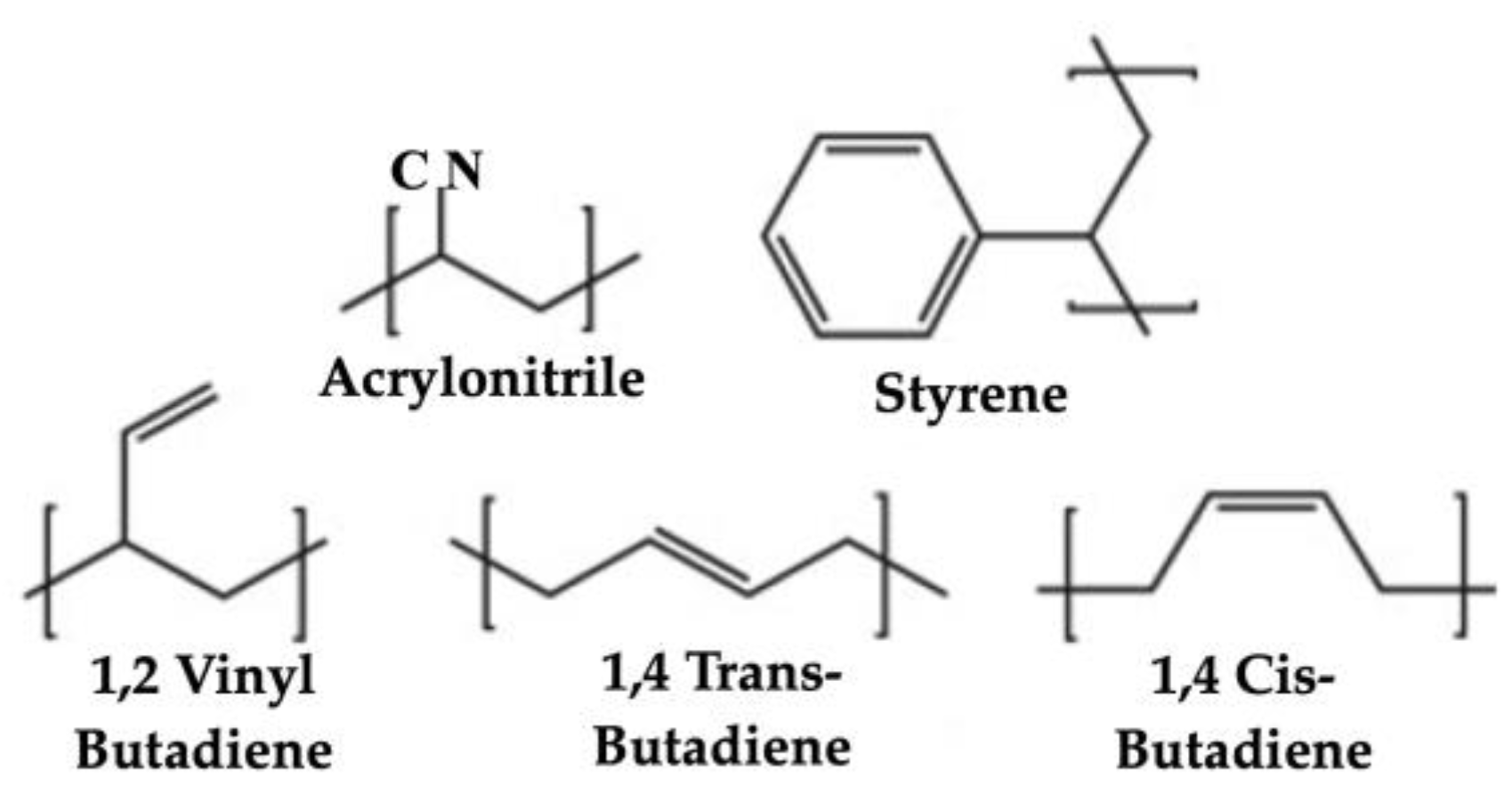
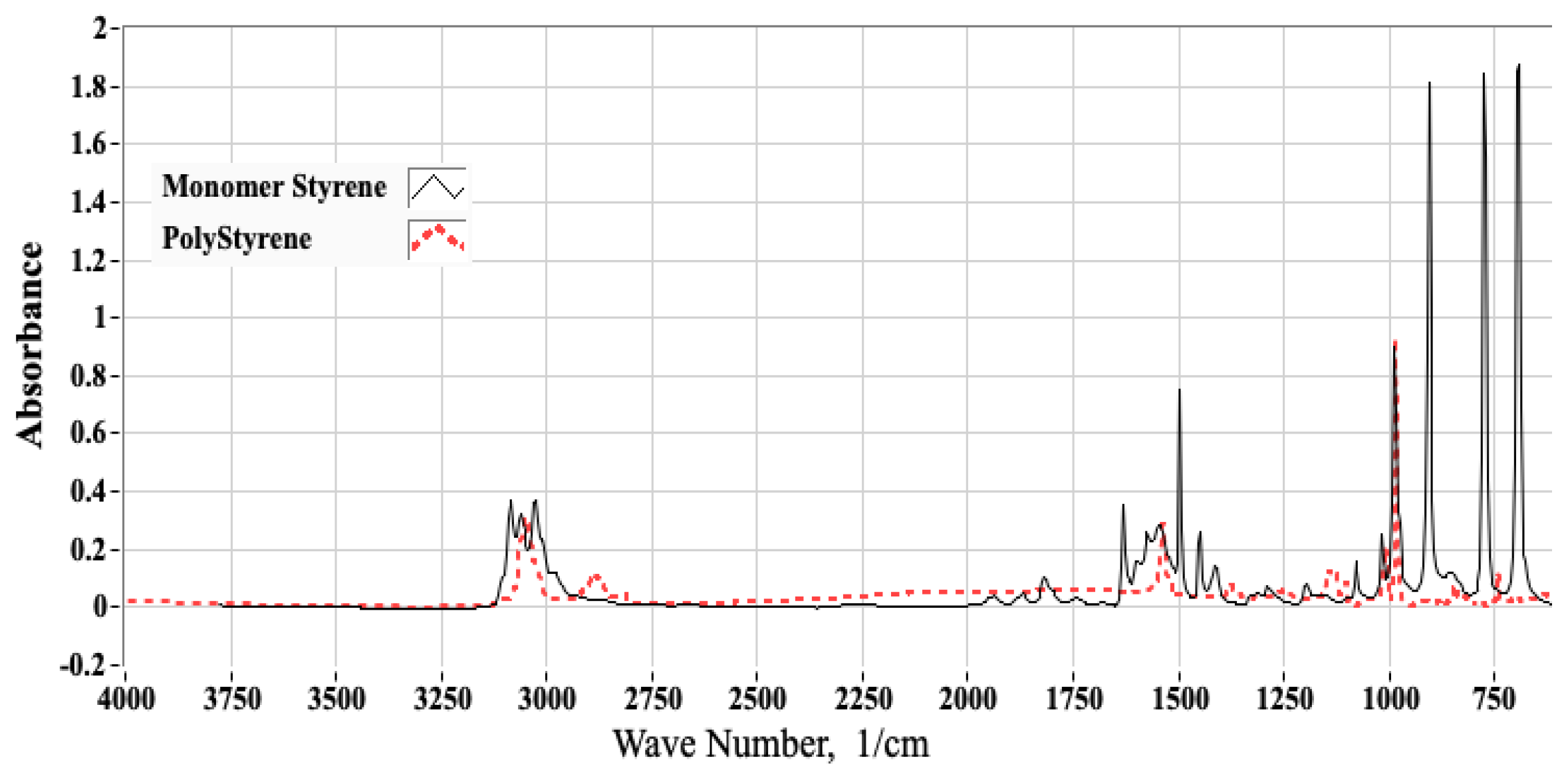
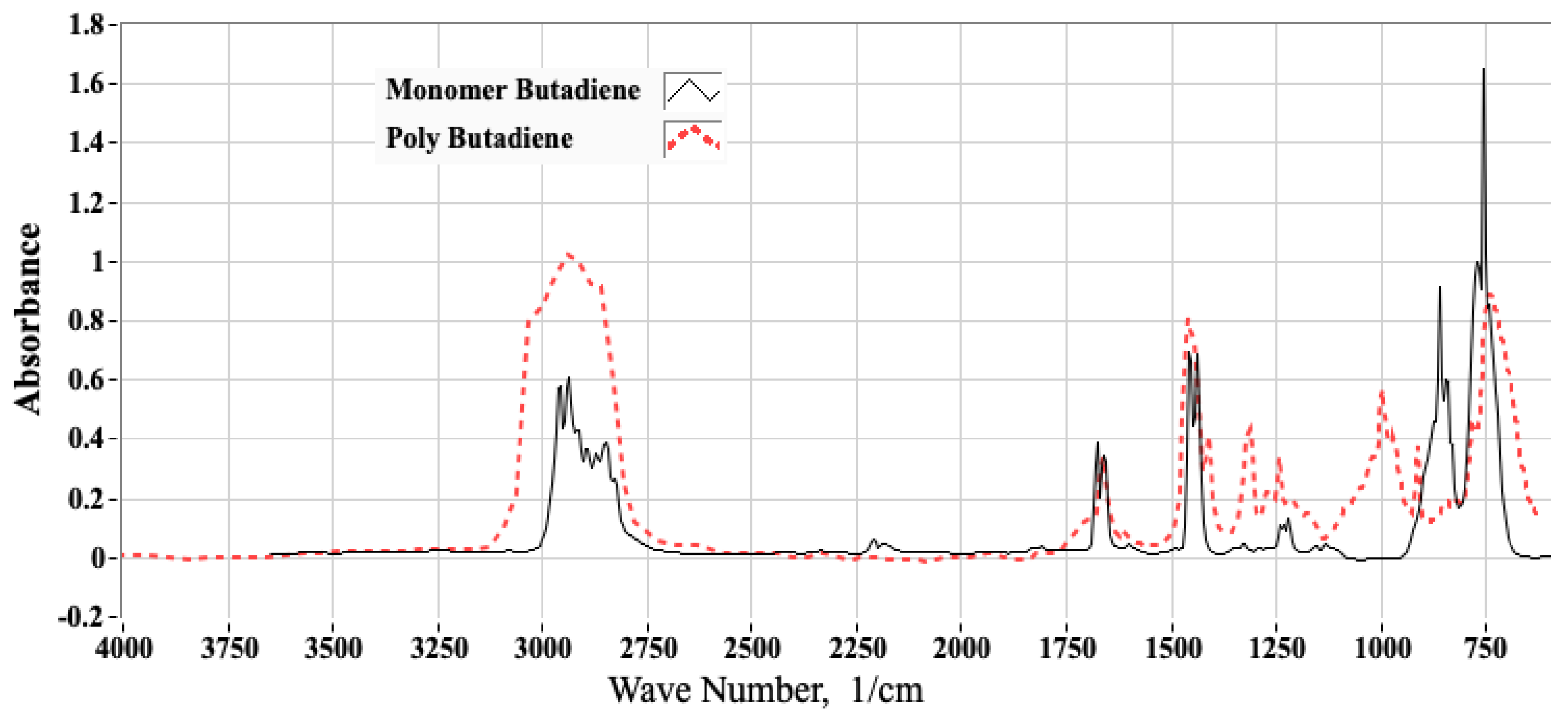
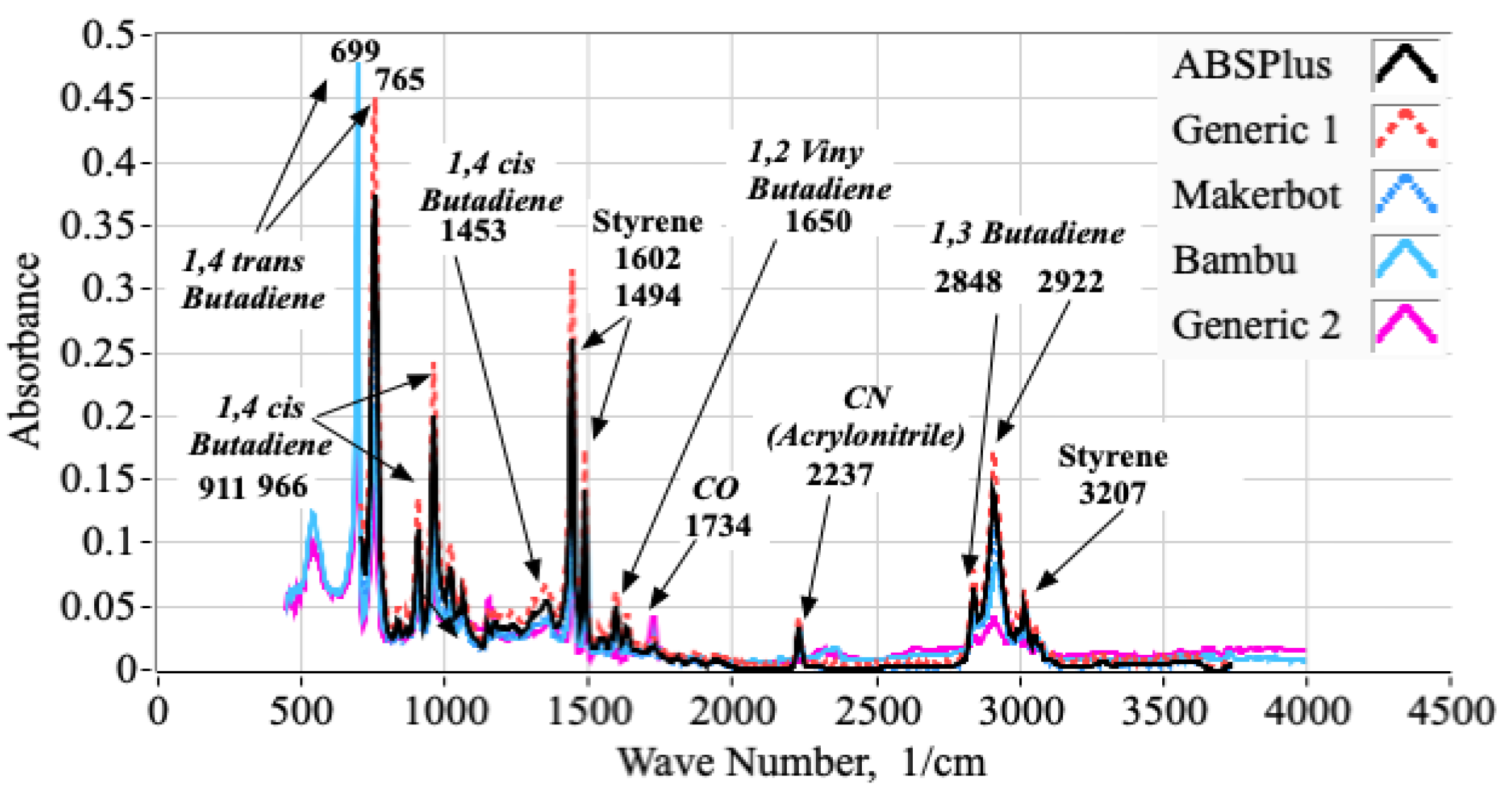
2.1.1. Analyses of ABS Samples Using Fourier Transform Infrared Spectroscopy (FTIR)
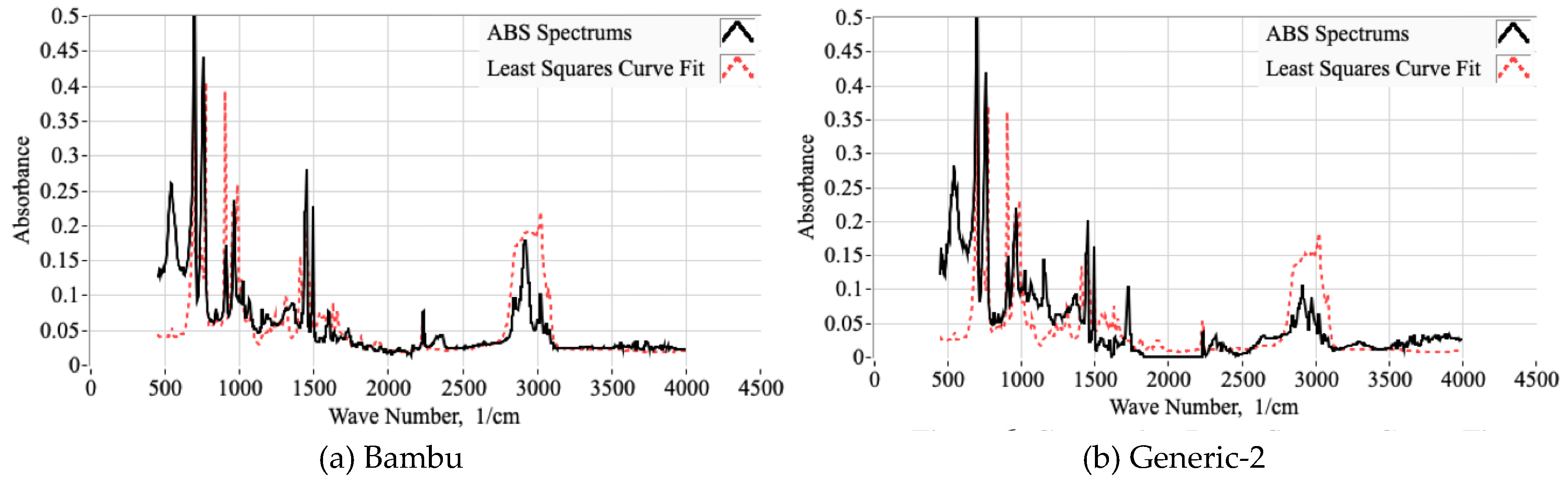
2.1.2. Using FTIR Spectrum to Estimate the Molar Proportions of Constituent Monomers
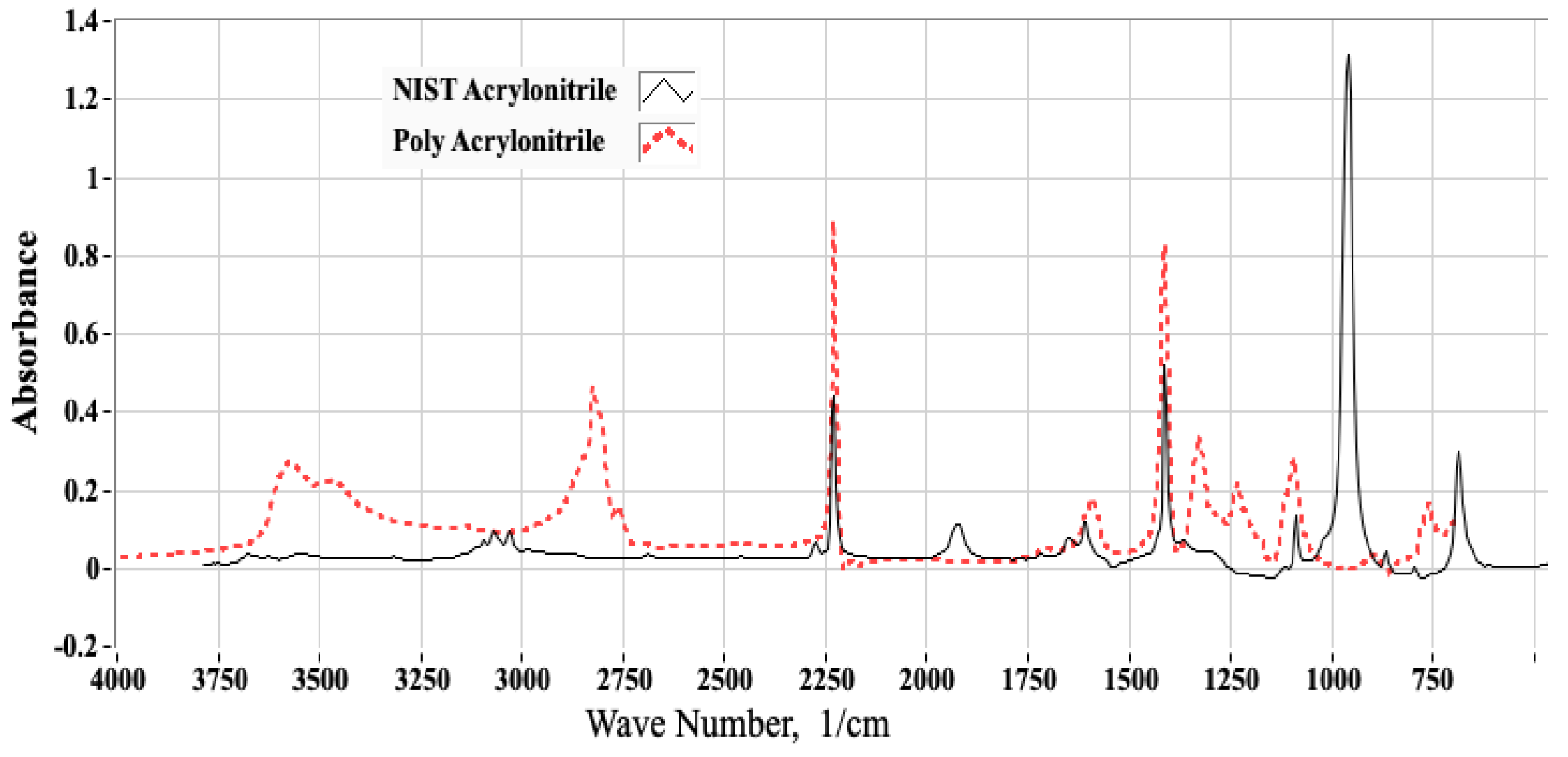
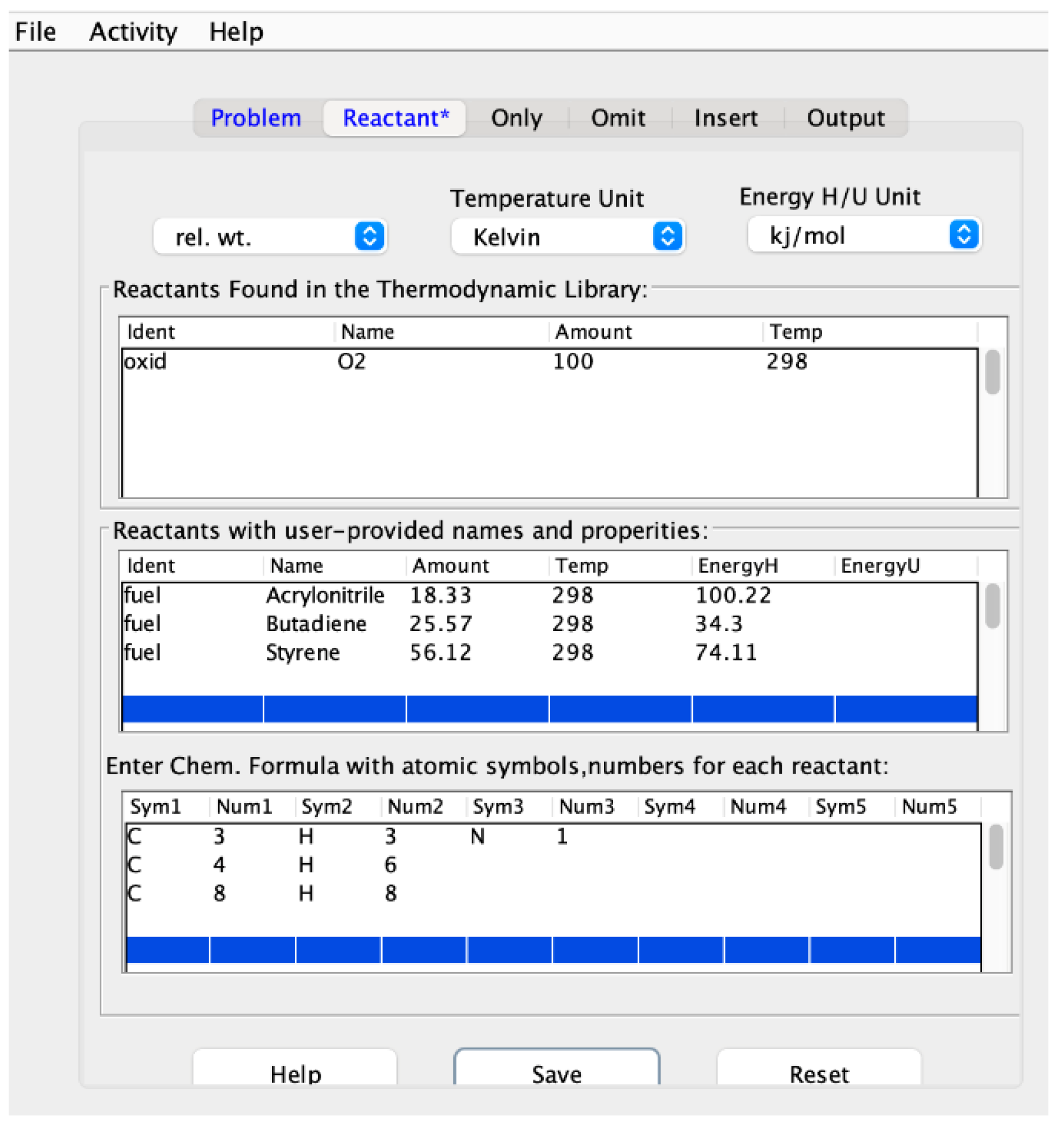
2.1.3. Selecting the Proper Reference Spectra
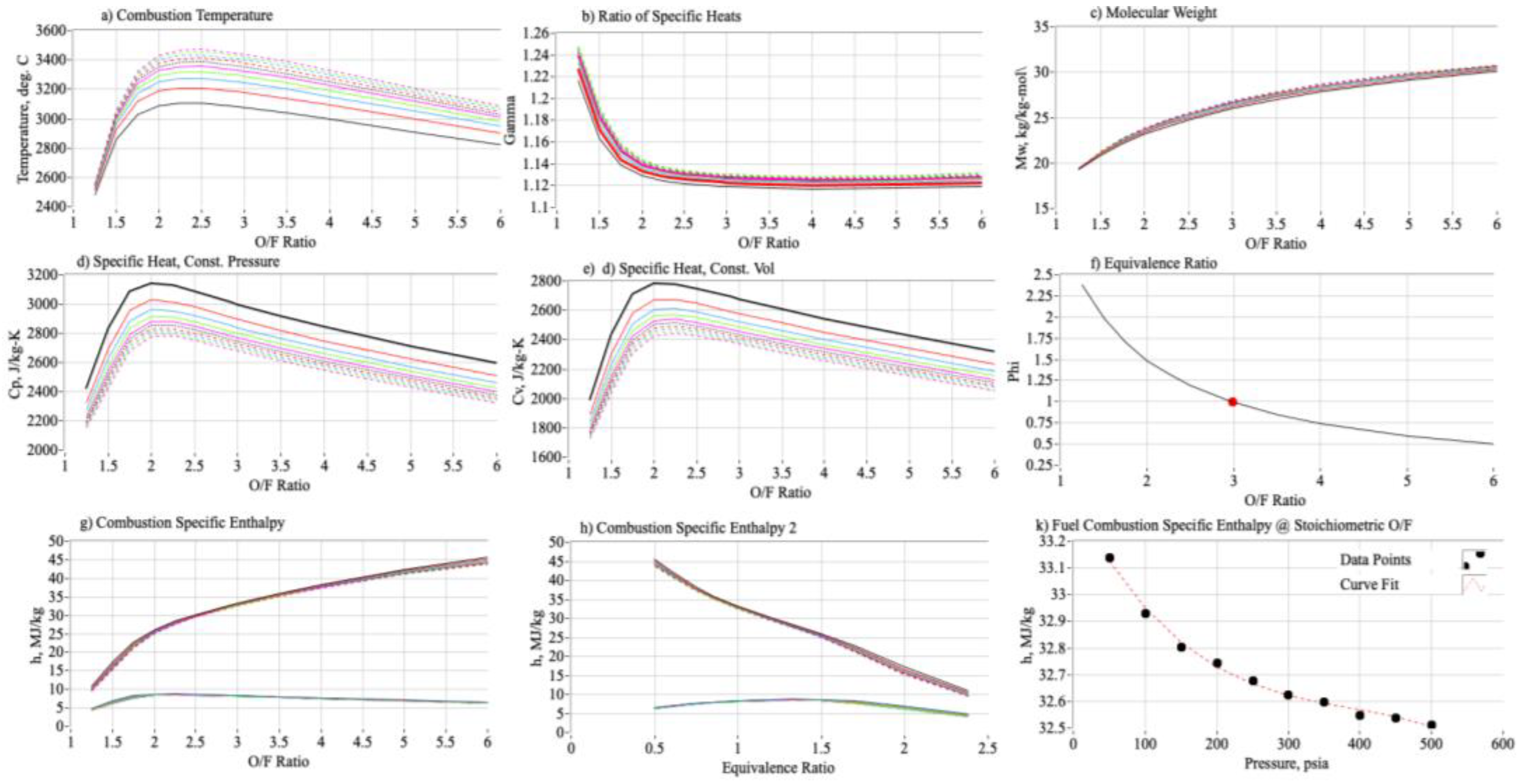
2.1.4. Using Mole-Fractions to Calculate ABS Properties of Combustion
2.2. Bomb Calorimeter Testing for ABS Enthalpies of Combustion
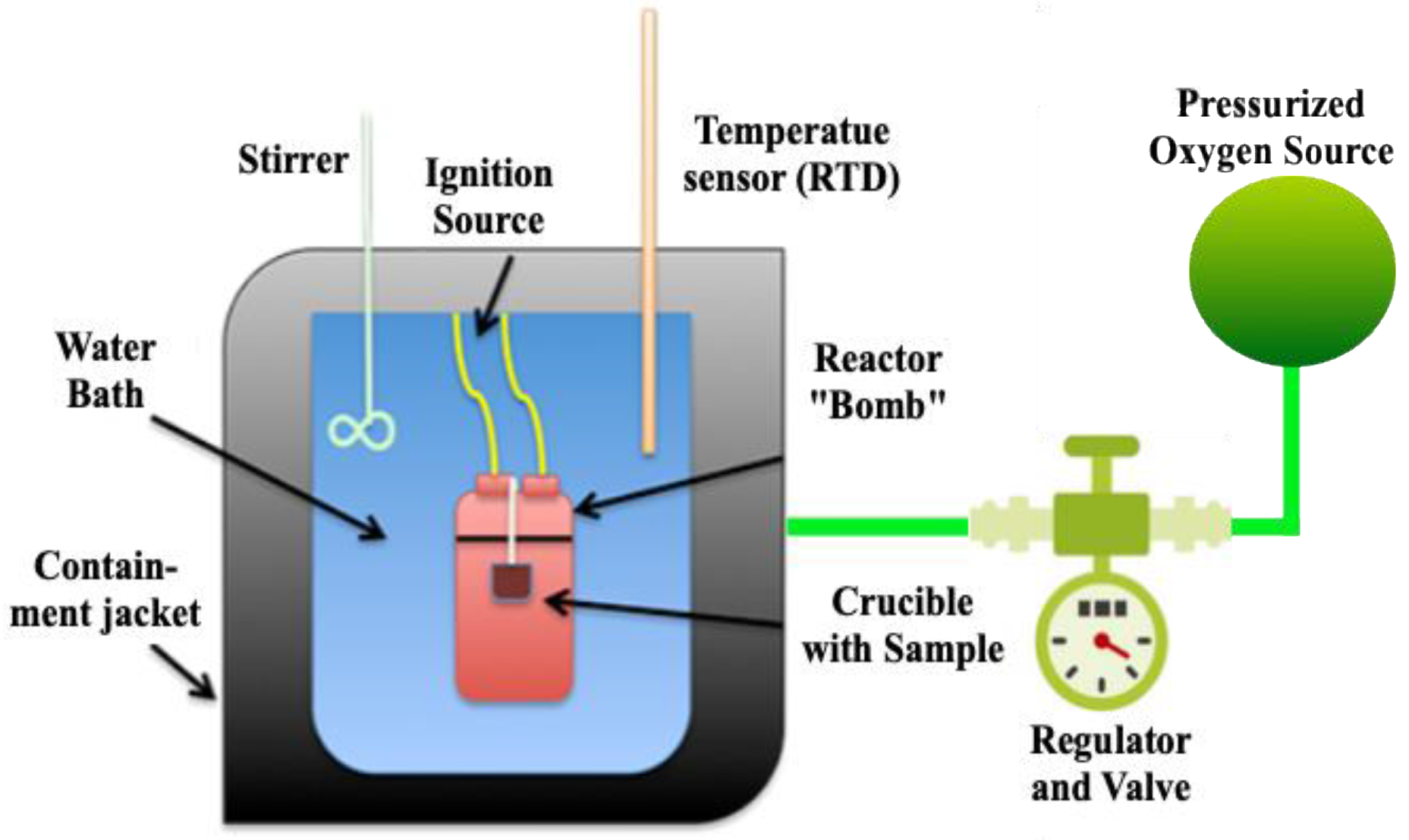
2.2.1. Bomb Calorimetry Test Apparatus
2.2.2. Bomb Calorimetry Test Procedures
2.2.3. Bomb Calorimeter Analysis Methods
3. Results
3.1. FTIR Testing Campaign and Associated Analyses
3.2. Calculating the Enthalpies of Formation Using FTIR Results
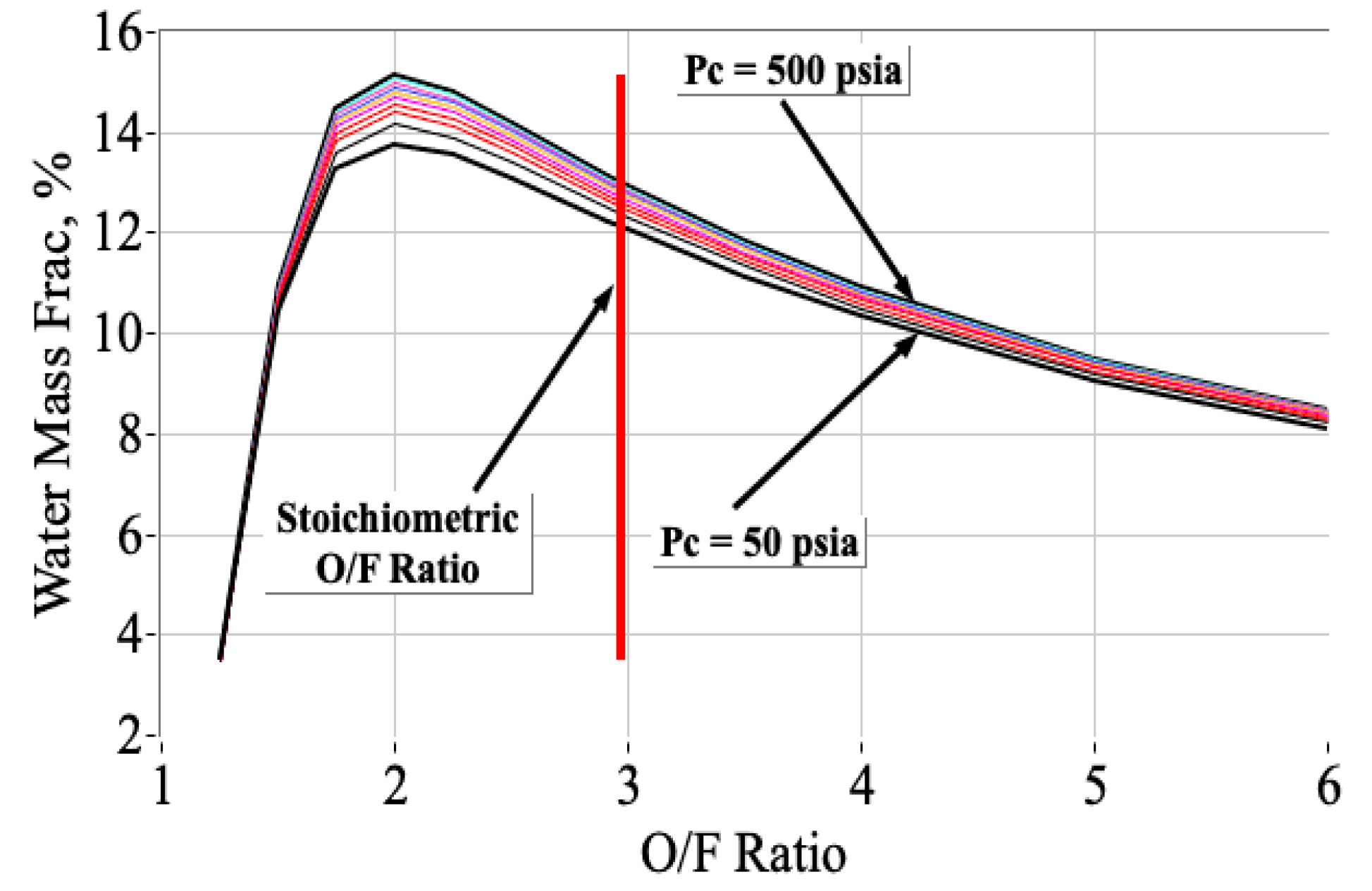
3.3. Calculating the Properties of Combustion
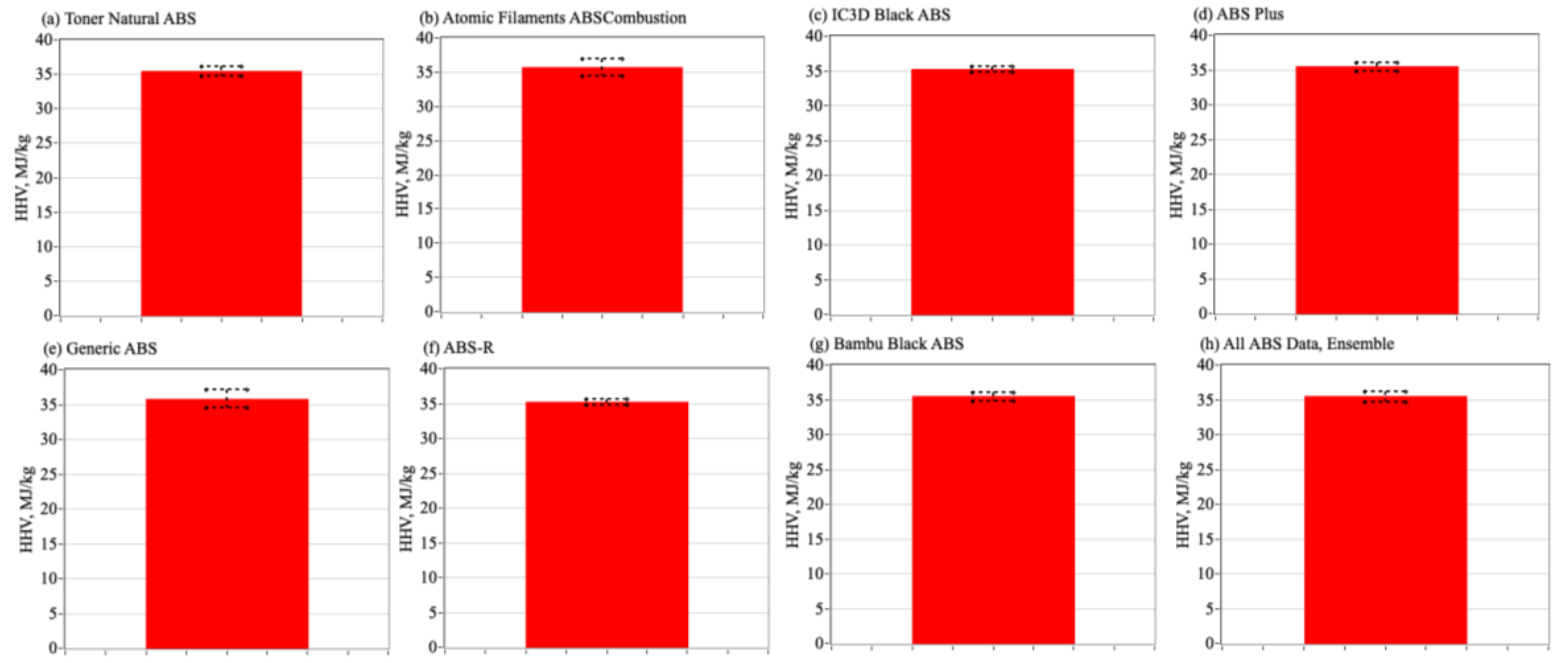
3.4. Bomb Calorimeter Testing Campaign and Associated Analyses
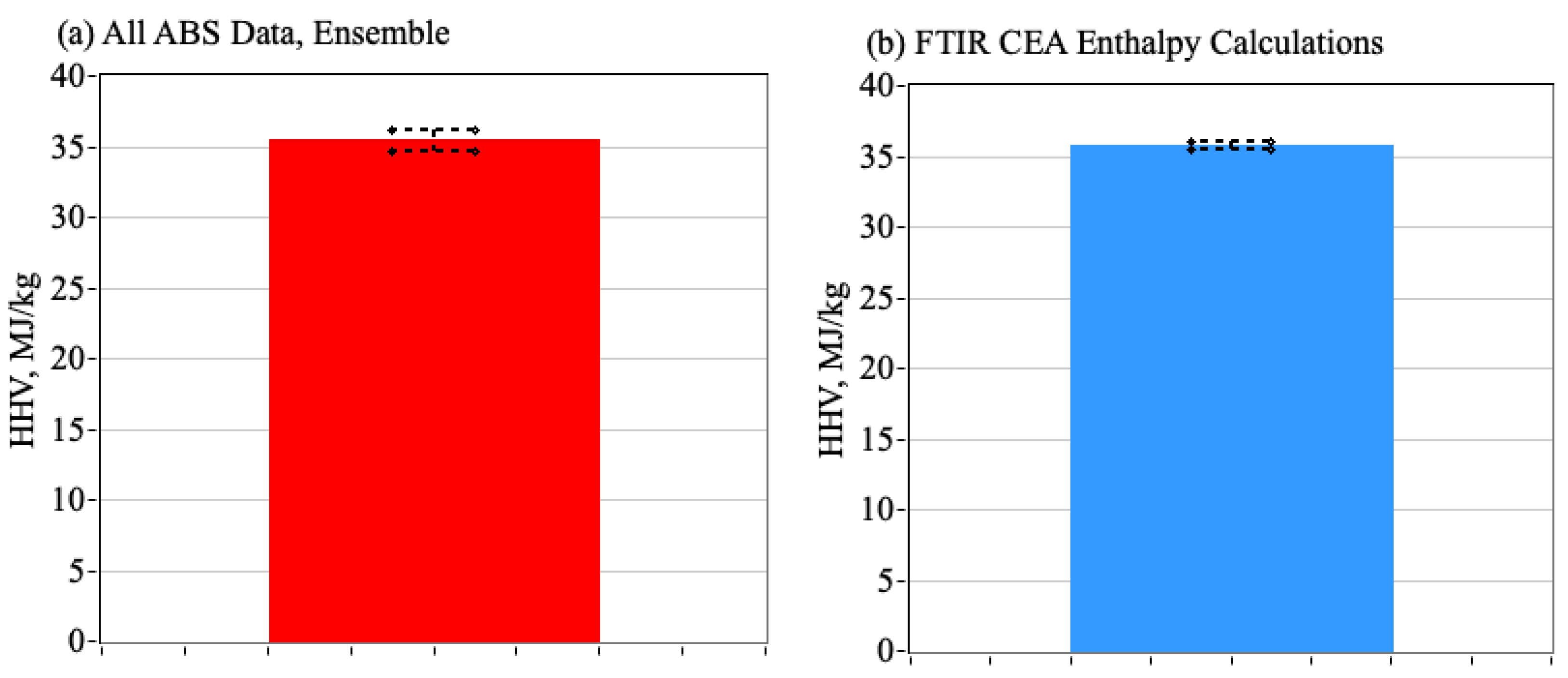
3.5. Comparing CEA Calculations to Bomb Calorimeter Test Results for ABS Samples
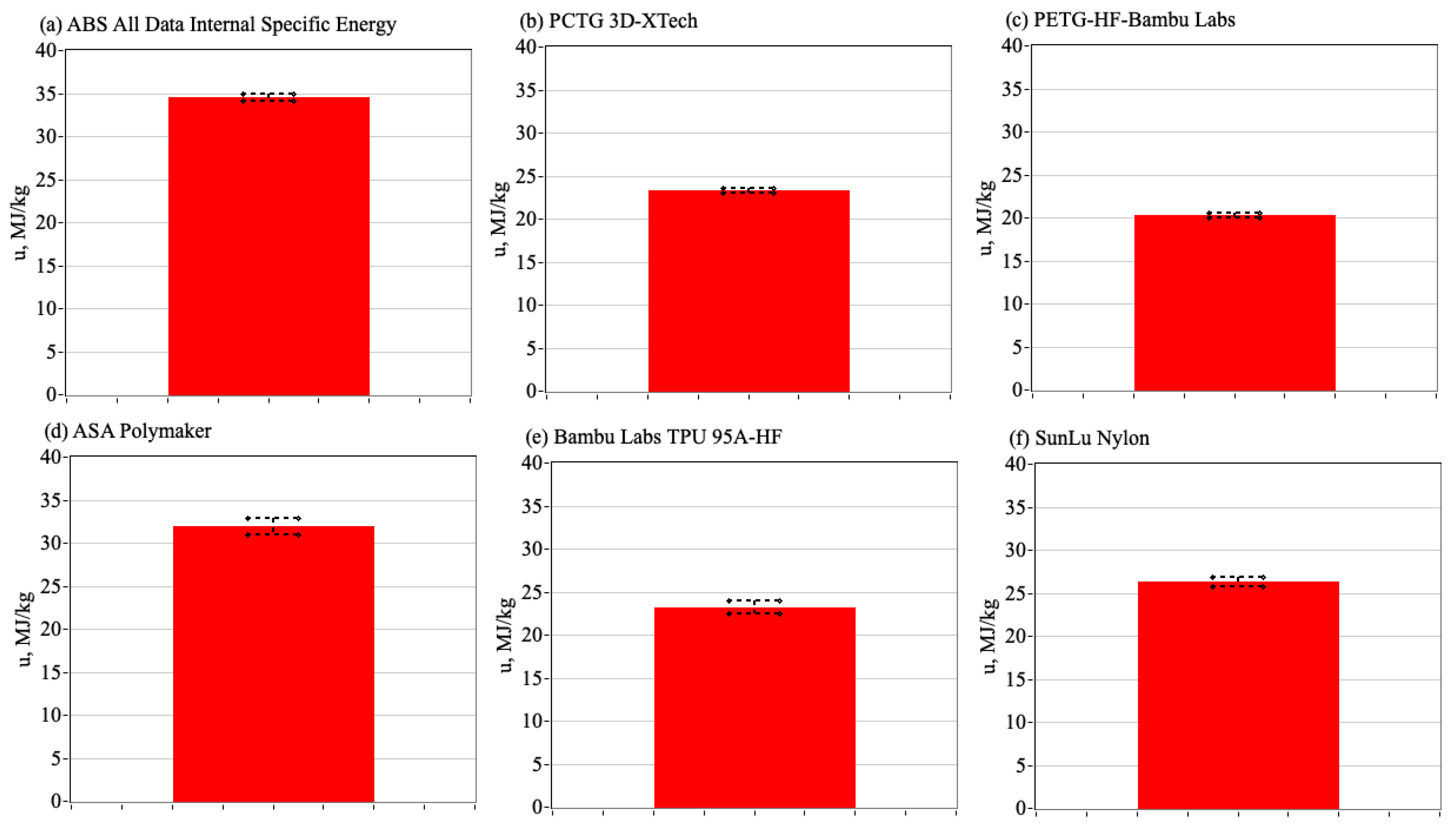
3.6. Comparing the Enthalpies of Combustion to ABS to Alternative Commercially Available 3-D Print Materials
4. Discussion
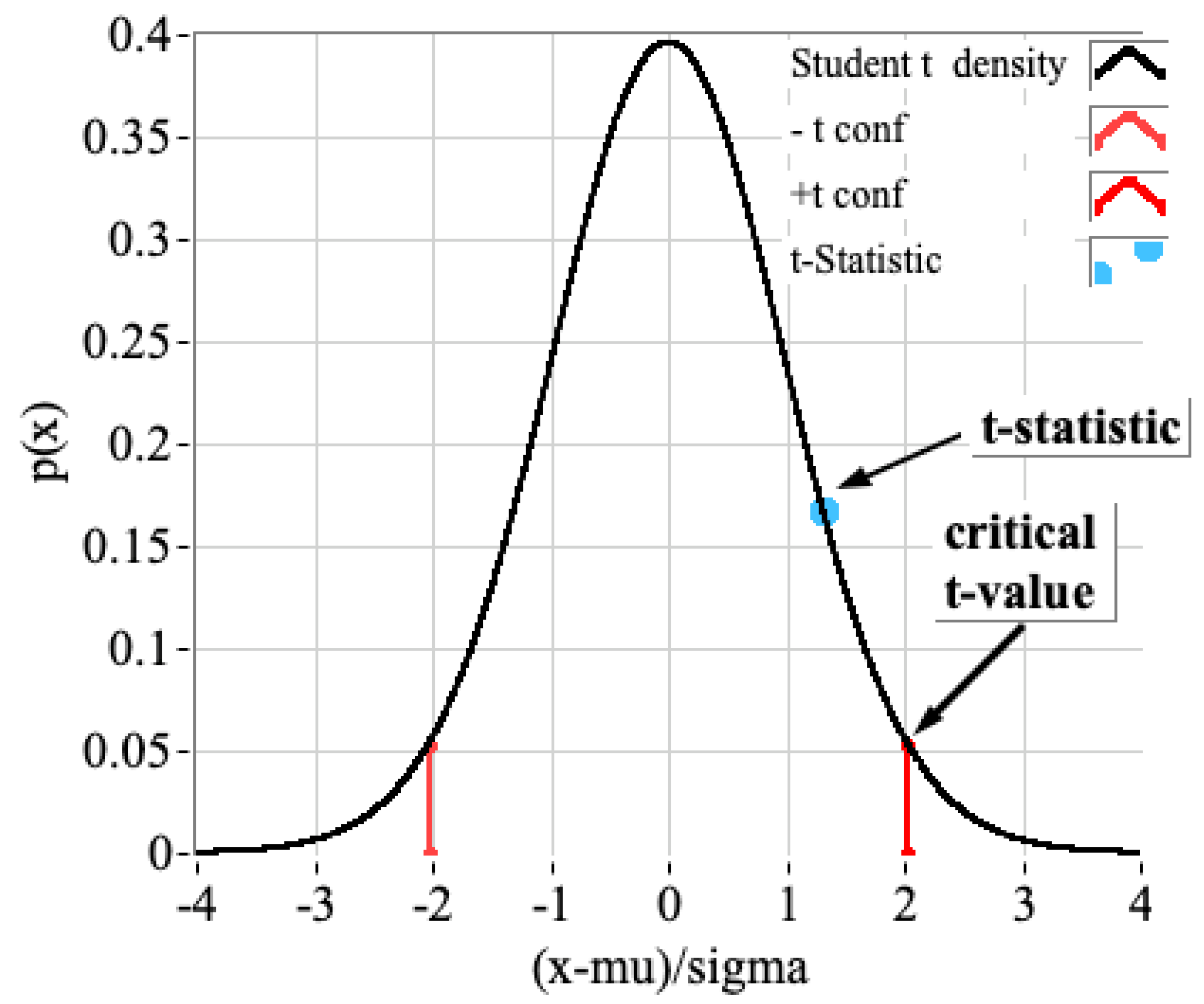
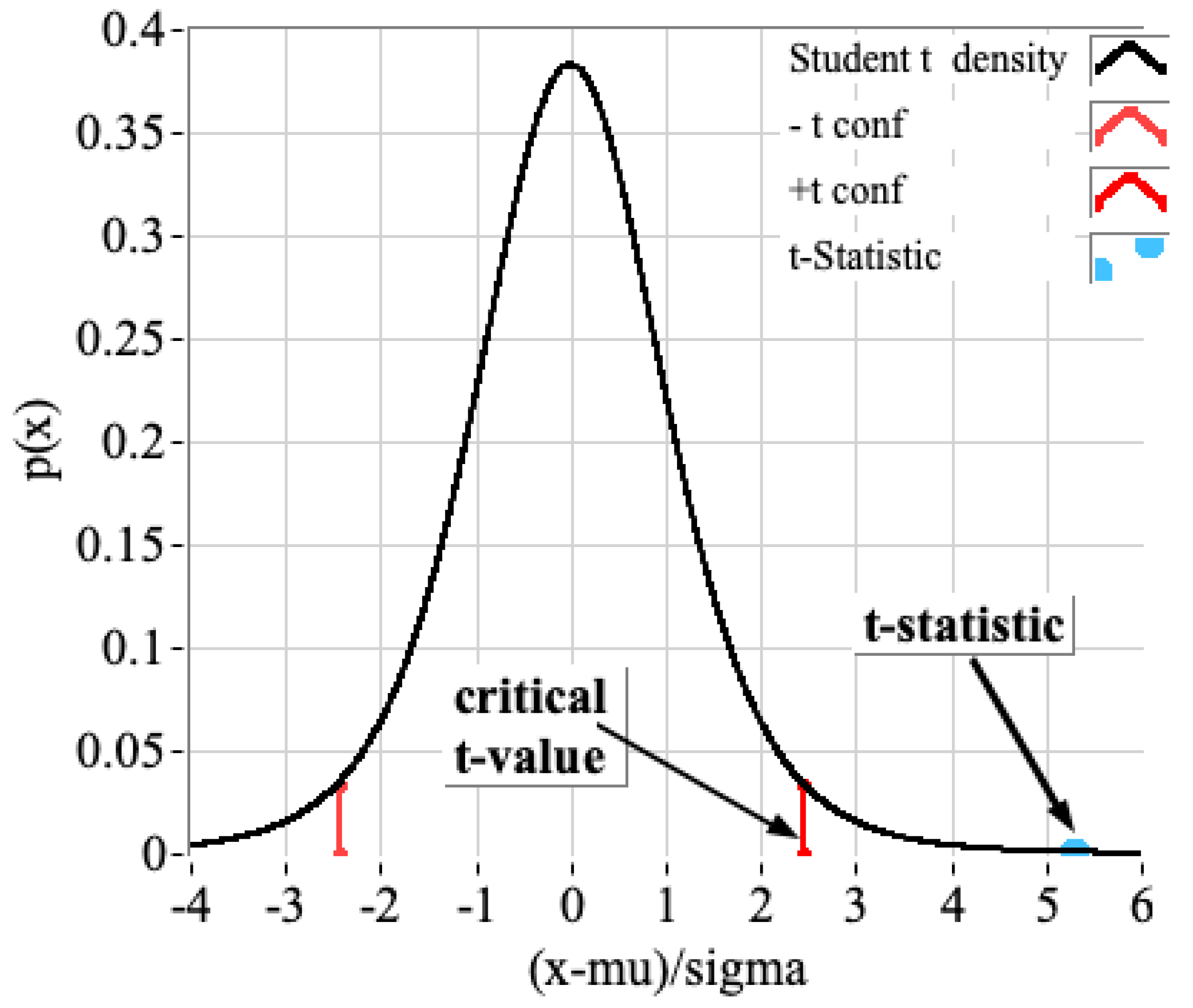
4.1. Statistical Assessment of FTIR/Bomb Calorimetry Results for ABS Samples
4.2. Statistical Assessment of Bomb Calorimetry Results, Comparing Mean ABS Result Against the Alternative Materials
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Nomenclature
| Symbols | ||
| = | Spectral absorbance of tested material | |
| {a,b,c} | = | FTIR least-squares curve fit coefficients |
| C | = | Symbol for atomic carbon |
| Cp | = | Specific heat at constant pressure, KJ/kg-K |
| Cv | = | Specific heat at constant volume, KJ/kg-K |
| Cbomb | = | Heat capacity of bomb calorimeter, KJ/K |
| ewire | = | energy content of bomb calorimeter fuse wire, J/cm |
| H | = | Symbol for atom hydrogen |
| DH | = | Total enthalpy change, MJ |
| DHc | = | Specific enthalpy of combustion, MJ |
| Hf | = | Molar enthalpy of formation, kJ/g-mol |
| DHLHV | = | Specific enthalpy changes due to condensed water vapor, MJ/kg |
| DHLHV | = | Specific enthalpy change used to calculate LHV, MJ/kg |
| L | = | Latent heat of vaporization, KJ/kg |
| DLfuse | = | Length of consumed fuel wire for bomb calorimetry tests, cm |
| Mf | = | Mass of fuel sample, g |
| MH2O | = | CEA plume of water mass fraction, kg/kg |
| Mox | = | Initial mass of oxygen in bomb calorimeter, g |
| Mw | = | Molecular weight, g/mol |
| Mwcomb | = | Molecular weight of combustion products, g/mol |
| Mwox | = | Molecular weight of O2, 31.9988 g/mol |
| = | Fuel mass flow rate, g/sec | |
| = | Oxygen mass flow rate, g/sec | |
| N | = | Symbol for atomic nitrogen |
| n | = | Number of samples in spectrum |
| n | = | Number of gaseous moles in bomb calorimeter, alternate definition |
| O | = | Symbol for atomic oxygen |
| O/F | = | Oxidizer to fuel ratio |
| O/Factual | = | Actual/achieved oxidizer to fuel ratio |
| O/Fstoich | = | Stoichiometric oxidizer to fuel ratio |
| P | = | Calorimeter bomb pressure, atms |
| Pc | = | Combustion (chamber) pressure, kPa |
| DQ | = | Total heat released during combustion, MJ |
| rc | = | Curve fit correlation index |
| T0 | = | Combustion flame temperature, K |
| DT | = | Temperature change during event |
| u | = | Specific internal energy of combustion, MJ/kg |
| V | = | Calorimeter bomb volume, cm3 |
| g | = | Ratio of specific heats |
| m | = | Sample mean |
| | = | Equivalence ratio |
| s | = | Sample standard deviation |
| Acronyms and Abbreviations | ||
| ABS | = | Acrylonitrile butadiene styrene |
| ASA | = | Acrylonitrile Styrene Acrylate |
| ATR | = | Attenuated total reflection |
| CEA | = | Chemical equilibrium with applications (computer program) |
| D.O.F. | = | Degrees of freedom |
| FDM | = | Fused deposition modeling |
| FTIR | = | Fourier transform infrared spectroscopy |
| GOX | = | Gaseous oxygen |
| GUI | = | Graphical user interface |
| HPGHP | = | High-Performance Green Hybrid Propulsion |
| HHV | = | High heating value, MJ/kg |
| LHV | = | Low heating value, MJ/kg |
| PA6 | = | Polyamide-6 (Nylon-6) |
| PCTG | = | Polycyclohexylene Dimethylene Terephthalate Glycol |
| PETG | = | Polyethylene Terephthalate Glycol |
| PRL | = | Propulsion Research laboratory |
| Stoich | = | Stoichiometric O/F ratio |
| TPU | = | Thermoplastic Polyurethane |
References
- Bombelli, V. Economic Benefits for the Use of Non-toxic Monopropellants for Spacecraft Applications, AIAA-2003-4783. 39th AIAA/ASME/SAE/ASEE Joint Propulsion Conference and Exhibit, Huntsville, AL, July 2003. [Google Scholar] [CrossRef]
- Haeseler, D.; Bombelli, V.; Vuillermoz, P.; Lo, R.; Marée, T.; Caramelli, F. “Green Propellant Propulsion Concepts for Space Transportation and Technology Development Needs,” ESA SP-557. In Proceedings of the 2nd International Conference on Green Propellants for Space Propulsion, Cagliari, Sardinia, Italy, 7-8 June 2004. https://adsabs.harvard.edu/full/2004ESASP.557E...4H (accessed on 2 December 2025).
- Goldstein, E. The Greening of Satellite Propulsion. Aerospace America. 2012, pp. 26–28. https://uppsagd.files.wordpress.com/2012/05/aerospace-america (accessed on 2 December 2025).
- Hawkins, T. W.; Brand, A. J.; McKay, M.; Tinnirello, M. “Reduced Toxicity, High Performance Monopropellant at the U.S. Air Force Research Laboratory,” AFRL-RZ- ED-TP-2010-219. 4th International Association for the Advancement of Space Safety Conference, Huntsville, AL, May 2010; www.dtic.mil/dtic/tr/fulltext/u2/a522113.pdf (accessed on 2 December 2025).
- Dornheim, M.A. Reaching 100 km. Av. Week Space Technol. 2024, 2004, 45–46. https://www.researchgate.net/publication/292268464 (accessed on 29 Jan. 2026).
- Casalino, L.; Pastrone, D. Optimization of Hybrid Sounding Rockets for Hypersonic Testing. J. Propuls. Power 2012, 28, 405–411. [Google Scholar] [CrossRef]
- Jens, E.; Karp, A.C.; Nakazono, B.; Eldred, D.B.; DeVost, M.E.; Vaughan, D. Design of a Hybrid CubeSat Orbit Insertion Motor. AIAA 2016-4961, 52nd AIAA/SAE/ASEE Joint Propulsion Conference, Salt Lake City, UT, USA, 25–27, July 2016. [Google Scholar] [CrossRef]
- Karp, A.C.; Nakazono, B.; Benito Manrique, J.; Shotwell, R.; Vaughan, D.; Story, G.T. A Hybrid Mars Ascent Vehicle Concept for Low Temperature Storage and Operation. AIAA 2016-496252nd AIAA/SAE/ASEE Joint Propulsion Conference, Salt Lake City, UT, USA, 25–27, July 2016. [Google Scholar] [CrossRef]
- Whitmore, S.A. “Three-Dimensional Printing of Green” Fuels for Low-Cost Small Spacecraft Propulsion Systems. J. Spacecraft Rockets 2018, 55. [Google Scholar] [CrossRef]
- Whitmore, S.A. Additive Manufacturing as an Enabling Technology for “Green” Hybrid Spacecraft Propulsion. In Proceedings of the RAST-1039, Conference on Recent Advances in Space Technology 2015, Istanbul Turkey, 2015; pp. 16–19. https://ieeexplore.ieee.org/document/7208305 (accessed on 5 December 2025).
- Whitmore, S.A. Additive Manufacturing as an Enabling Technology for “Green” Hybrid Spacecraft Propulsion. Istanbul Turkey, 16-19 June 2015. https://ieeexplore.ieee.org/document/7208305 (accessed on 5 December 2019).
- Whitmore, Stephen A. Additively Manufactured Acrylonitrile-Butadiene-Styrene–Nitrous-Oxide Hybrid Rocket Motor with Electrostatic Igniter. J. Prop. Power 2015, 31, 1217–1220. [Google Scholar] [CrossRef]
- Whitmore, S.A.; Inkley, N.R.; Merkley, D. P.; Judson, M.I. Development of a Power-Efficient, Restart Capable Arc Ignitor for Hybrid Rockets. J. Prop. Power 2015, 31, 1739–1749. [Google Scholar] [CrossRef]
- Wright, P. K. 21st Century Manufacturing; Prentice–Hall: Upper Saddle, River, NJ, 2001; pp. 7–67. ISBN -10 0130956015, ISBN-13 978-0130956019. [Google Scholar]
- Fuller, J. K.; Ehrlich, D. A.; Lu, P. C.; Jansen, R. P.; Hoffman, J. D. Advantages of Rapid Prototyping for Hybrid Rocket Motor Fuel Grain Fabrication. AIAA 2011-5909, 47th AIAA/ASME/SAE/ASEE Joint Propulsion Conference & Exhibit, San Diego, CA, 2011. [Google Scholar] [CrossRef]
- Whitmore, S. A.; Walker, S. D.; Merkley, D. P.; Sobbi, M. High Regression Rate Hybrid Rocket Fuel Grains with Helical Port Structures. J. Prop. Power 2015, 31, 1727–1738. [Google Scholar] [CrossRef]
- Whitmore, S. A.; Peterson, Z.; Eilers, S. D. “Analytical and Experimental Comparisons of HTPB and ABS as Hybrid Rocket Fuels,” AIAA 2011-5909. 47th AIAA/ASME/SAE/ASEE Joint Propulsion Conference & Exhibit, July 2011; https://arc.aiaa.org/doi/abs/10.2514/6.2011-5909.
- Whitmore, S. A.; Peterson, Z.; Eilers, S. D. “Comparing of Hydroxyl Terminated Poly Butadiene and Acrylonitrile Butadiene as Hybrid Rocket Fuels,”. J. Prop. Power 2012, 29, 582–592. [Google Scholar] [CrossRef]
- Cha, J. Co-authors, Engineering Plastics Handbook, Chap. 6, "Acrylonitrile-Butadiene-Styrene (ABS) Resin"; Margolis, J. M., Ed.; McGraw-Hill, 2006; pp. 101–130. ISBN 0071457674, ISBN-13: 9780071457675. [Google Scholar]
- Anon. National Institute for Standards in Technology (NIST), Standard Reference Database Number 69. http://webbook.nist.gov/chemistry (accessed on 25 June 2025).
- Othmer, K. Butadiene. In Encyclopedia of Chemical Technology; John Wiley & Sons, Inc.: New York, 2006; (accessed on 25 June 2025). [Google Scholar] [CrossRef]
- Anon. The Basics of Infrared Spectrophotometry. 10 October 2019. https://conductscience.com/the-basics-of-infrared-spectrophotometry/ (accessed on 10 January 2026).
- Bradley, M. FTIR Sample Techniques: Attenuated Total Reflection (ATR)," ThermoFisher Scientific Technical Note. https://www.thermofisher.com/us/en/home/industrial/spectroscopy (accessed on 1 June 2025).
- Whitmore, Stephen A. Plume Contamination Measurements of an Additively Printed, Green-Propellant Hybrid Thruster. J. of Prop. and Power 2022, 38, 671–685. [Google Scholar] [CrossRef]
- Ball, D. W. Beer’s Law, Field Guide to Spectroscopy. International Society for Optics and Photonics, SPIE. 2006, p. 66. https://spie.org/publications (accessed on 30 January 2026).
- Anon. Polyacrylonitrile, Millipore Sigma, 2026, Merck KGaA, Darmstadt, Germany and/or its affiliates. https://www.sigmaaldrich.com/US/en/product/aldrich/181315 (accessed on 25 February 2026).
- Anon. 2-Propenenitrile, NIST Chemistry WebBook, SRD 69, National Institute of Standards and Technology, U.S. Dept. of Commerce. https://webbook.nist.gov/cgi/cbook.cgi?ID=C107131&Mask=80#IR-Spec (accessed on 25 February 2026).
- Anon. Polyacrylonitrile, (25014-41-9) IR1, Chemical Book. https://www.chemicalbook.com/SpectrumEN_25014-41-9_IR1.htm (accessed on 25 February 2026).
- Anon, Styrene. NIST Chemistry WebBook, SRD 69, National Institute of Standards and Technology, U.S. Dept. of Commerce. https://webbook.nist.gov/cgi/cbook.cgi?ID=C100425&Type=IR-SPEC&Index=2#IR-SPEC (accessed on 25 February 2026).
- Anon. Polystyrene, (9003-56-6) IR1, Chemical Book. https://www.chemicalbook.com/SpectrumEN_9003-53-6_IR1.htm (accessed on 25 February 2026).
- Anon, 1-3 Butadiene, NIST Chemistry WebBook, SRD 69, National Institute of Standards and Technology, U.S. Dept. of Commerce. https://webbook.nist.gov/cgi/cbook.cgi?ID=C106990&Type=IR-SPEC&Index=1 (accessed on 25 February 2026).
- Anon. Cis-Polybutadiene NIST Chemistry WebBook, SRD 69, National Institute of Standards and Technology, U.S. Dept. of Commerce. https://webbook.nist.gov/cgi/cbook.cgi?ID=B6002924&Mask=80 (accessed on 25 February 2026).
- Gordon, S.; McBride, B. J. “Computer Program for Calculation of Complex Chemical Equilibrium Compositions and Applications, I. Analysis” National Aeronautics and Space Administration, Cleveland, Ohio, Tech. Rep. NASA RP-1311, 1994. https://ntrs.nasa.gov/api/citations/19950013764/downloads/19950013764.pdf (accessed on 23 January 2026).
- Gordon, S.; McBride, B. J. “Computer Program for Calculation of Complex Chemical Equilibrium Compositions and Applications, II. User’s Manual and Program Description” National Aeronautics and Space Administration, Cleveland, Ohio, Tech. Rep. NASA RP-1311-2, 1994. https://ntrs.nasa.gov/api/citations/19960044559/downloads/19960044559.pdf (accessed on 23 January 2026).
- Leader, M. K.; Lavelle, T. M.; Wang, X.J.; Dickens, K. W.; McTague, M. CEA2022: A Modernization of NASA Glenn’s Software CEA (Chemical Equilibrium with Applications), Thermal and Fluids Analysis Workshop (TFAWS), 2024. https://ntrs.nasa.gov/api/citations/20240009728/downloads/TFAWS_2024_CEA.pdf (accessed on 23 January 2026).
- McBride, B. J.; Zehe, M. J.; Sanford Gordon, S. NASA Glenn Coefficients for Calculating Thermodynamic Properties of Individual Species,“ NASA/TP—2002-211556, 2002. https://ntrs.nasa.gov/api/citations/20020085330/downloads/20020085330.pdf (accessed on 23 January 2026).
- Svehla, R. A. Transport Coefficients for the NASA Lewis Chemical Equilibrium Program. https://ntrs.nasa.gov/api/citations/19950021761/downloads/19950021761.pdf (accessed on 23 January 2026).
- Anon. Model 6200 Calorimeter. Preiser Scientific. https://preiser-my.sharepoint.com (accessed on 2 February 2026).
- Anon. “Introduction to Bomb Calorimetry,” 483M. Parr Instrument Co. https://www.scimed.co.uk/wp-content/uploads/2024/02/Introduction-to-bomb-calorimetry.pdf (accessed on 2 February 2026).
- ASTM International. Standard Test Method for Heat of Combustion of Liquid Hydrocarbon Fuels by Bomb Calorimeter (ASTM D240-19); ASTM International, 2025. https://store.astm.org/d0240-19.html (accessed on 2 February 2026).
- Whitmore, S. A.; Brewer, D. L. Plume Contamination Measurements of an Additively-Printed GOX/ABS Hybrid Thruster. AIAA 2017- 4982, 53rd AIAA/SAE/ASEE Joint Propulsion Conference, Atlanta GA, USA, 10-12 July 2017. [Google Scholar] [CrossRef]
- Whitmore, S. A. Plume Contamination Measurements of an Additively Printed, Green-Propellant Hybrid Thruster. J. of Prop. Power 2022, Vol. 38(No. 4). https://arc.aiaa.org/doi/abs/10.2514/1.B38612 (accessed on 2 March 2026). [CrossRef]
- Hu, K-H.; Kao, C-S.; Duh, Y-S. Studies on the Runaway Reaction of ABS Polymerization Process. J. Haz. Materials 2008, 159, 1–186. http://lib3.dss.go.th/fulltext/e_content/0304-3894/2008v.159n.1.pdf. [CrossRef] [PubMed]
- Anon. “ABS poly-acrylonitrile-co-butadiene-co-styrene,” Specification Sheet. http://www.polympart.com (accessed on 30 January 2026).
- Anon. “Molar Heat of Polymerization of ABS,” AI Search. https://www.google.com/search?q=molar+heat+of+polymerization+of+ABS (accessed on 30 January 2026).
- Baxendale, J. H.; Madaras, G. W. Kinetics and Heats of Copolymerization of Acrylonitrile and Methyl Methacrylate, J. Polymer Science 1956, 19, 171–179. [Google Scholar] [CrossRef]
- Van Krevelen, D. W.; Jijenhuis, K. Properties of Polymers: Their Correlation with Chemical Structure; Their Numerical Estimation and Prediction from Additive Group Contributions, Chapt. 20, 4th ed.; Elsevier Science Ltd., 2009; https://www.amazon.com/Properties-Polymers-Correlation-Estimation-Contributions/dp/0080548199.
- Prosen, E.J.; Rossini, F.D. Heats of Formation and Combustion of 1,3-Butadiene and Styrene. J. Research, NIST (formerly NBS) 1945, 34, 59–63. https://nvlpubs.nist.gov/nistpubs/jres/34/jresv34n1p59_A1b.pdf. [CrossRef]
- Anon. NATIONAL POLICY ON THE TRANSFER OF SCIENTIFIC, TECHNICAL AND ENGINEERING INFORMATION, National Security Decision Directives (NSDDs), 1985. https://irp.fas.org/offdocs/nsdd/nsdd-189.htm (accessed on 7 November 2024).
| Material | ABS-Plus | Generic-1 | ABS-R | Bambu | Generic-2 |
|---|---|---|---|---|---|
| ABS-Plus | 1.000 | 0.931 | 0.951 | 0.905 | 0.901 |
| Generic-1 | 0.931 | 1.000 | 0.950 | 0.925 | 0.905 |
| ABS-R | 0.951 | 0.950 | 1.000 | 0.911 | 0.866 |
| Bambu | 0.905 | 0.925 | 0.911 | 1.000 | 0.964 |
| Generic-2 | 0.901 | 0.905 | 0.905 | 0.964 | 1.000 |
| Material | Acrylonitrile | Butadiene | Styrene | Curve Fit rc | |||
|---|---|---|---|---|---|---|---|
| Mole Frac. % | Mass Frac. % | Mole Frac. % | Mass Frac. % | Mole Frac. % | Mass Frac. % | ||
| ABS-Plus | 23.77 | 16.90 | 34.60 | 25.06 | 41.63 | 58.06 | 0.885 |
| Generic-1 | 24.32 | 16.93 | 30.95 | 22.00 | 44.73 | 61.11 | 0.826 |
| ABS-R | 28.39 | 21.81 | 41.09 | 32.18 | 30.52 | 46.02 | 0.871 |
| Bambu | 24.01 | 17.18 | 35.40 | 25.82 | 40.59 | 57.00 | 0.865 |
| Generic-2 | 26.53 | 18.82 | 31.50 | 22.77 | 41.97 | 58.42 | 0.815 |
| Mean, m | 25.40 | 18.33 | 34.71 | 25.57 | 39.89 | 56.12 | 0.869 |
| Std. Dev. s | 2.00 | 2.10 | 4.05 | 4.02 | 5.46 | 5.85 | 0.015 |
| Monomer | Chemical Formula | Mw g/mol |
Hf monomer kJ/g-mol |
Qp polymer kJ/g-mol |
Net Hf kJ/g-mol | Mole Fraction | Mass Fraction | Net Enthalpy Contribution kJ/g-mol |
|
|---|---|---|---|---|---|---|---|---|---|
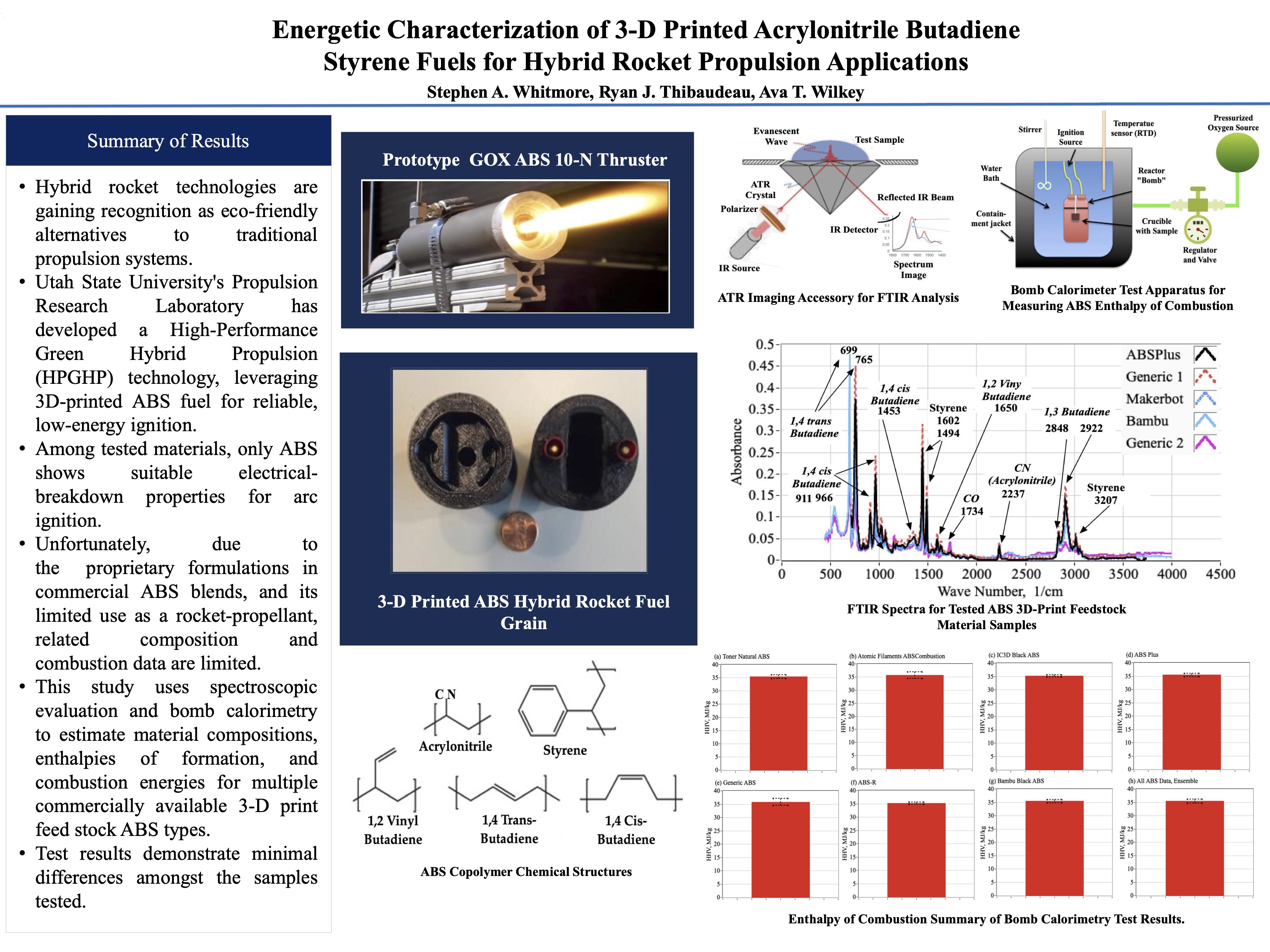
| Acrylo-nitrile | C3H3N | 53.06 | 172.62 [46] | 72.4 | 100.22 | 0.2540 | 0.1826 | 25.456 | |
| Butadiene | C4H6 | 54.09 | 104.10 [47] | 69.8 | 34.30 | 0.3471 | 0.2544 | 11.906 | |
| Styrene | C8H8 | 104.15 | 146.91 [48] | 72.8 | 74.11 | 0.3989 | 0.5630 | 29.563 | |
| ABS Total | C5.3416H6.0358 N0.254 | 74.164 | 1.000 | 1.000 | 66.924 | ||||
| Net Enthalpy Contribution kJ/g | 0.9068 | ||||||||
| Material | Hf(Molar) kJ/g-mol | Hf(Mass) kJ/g | Mw g/mol | Monomer Mass Fractions (%) | Chemical Formula | ||
| C3H3N | C4H6 | C8H8 | |||||
| ABS-Plus | 66.542 | 0.8910 | 74.687 | 16.90 | 25.06 | 58.06 | C5.428H6.120N0.238 |
| Generic-1 | 68.139 | 0.8938 | 76.233 | 16.93 | 22.00 | 61.11 | C5.546H6.165N0.243 |
| ABS-R | 65.165 | 0.9434 | 69.078 | 21.81 | 32.18 | 46.02 | C4.937H5.759N0.284 |
| Bambu | 66.286 | 0.8937 | 74.164 | 17.18 | 25.82 | 57.00 | C5.384H6.092N0.240 |
| Generic-2 | 67.296 | 0.9148 | 73.5671 | 18.82 | 22.77 | 58.42 | C5.315H6.002N0.262 |
| Mean, m | 66.924 | 0.9068 | 73.799 | 18.26 | 25.44 | 56.30 | C5.3416H6.0358N0.254 |
| Std. Dev. s | 1.155 | 0.0223 | 2.686 | 2.10 | 4.02 | 5.85 | - |
|
95% Student-t Conf. Interval |
1.384 | 0.0277 | 3.333 | 2.61 | 4.99 | 7.25 | |
| Parameter | Sample Mass , g | DT Bomb, oC | P final, atms | DQ, kJ | u, MJ/kg | HHV, MJ/kg |
|---|---|---|---|---|---|---|
| Test 1 | 0.970 | 3.70 | 26.86 | 33.011 | 34.032 | 34.992 |
| Test 2 | 0.960 | 3.80 | 26.84 | 33.892 | 35.304 | 36.273 |
| Test 3 | 0.970 | 3.80 | 26.87 | 33.894 | 34.942 | 35.902 |
| Test 4 | 0.970 | 3.70 | 26.86 | 33.002 | 34.022 | 34.982 |
| Test 5 | 0.970 | 3.70 | 26.86 | 33.016 | 34.997 | 34.997 |
| m | 0.968 | 3.74 | 26.86 | 33.363 | 34.468 | 35.429 |
| s | 0.0045 | 0.055 | 0.010 | 0.4838 | 0.612 | 0.615 |
| 95% Conf. Intvl. | 0.0047 | 0.057 | 0.011 | 0.5073 | 0.642 | 0.645 |
| Test Sample | Degrees of Freedom |
DQ, kJ Mean (m) |
DQ, kJ Std. Dev. (s) |
u, MJ/kg Mean |
u, MJ/kg Std. Dev. |
HHV, MJ/kg Mean |
HHV, MJ/kg Std. Dev. |
|---|---|---|---|---|---|---|---|
| Toner Plastics | 4 | 33.36 | 0.484 | 34.47 | 0.612 | 35.43 | 0.615 |
| Atomic Filaments | 4 | 34.25 | 1.011 | 34.75 | 1.218 | 35.69 | 1.224 |
| IC3D Black | 4 | 33.00 | 0.012 | 34.24 | 0.324 | 35.20 | 0.333 |
| ABSPlus | 4 | 33.34 | 0.032 | 34.32 | 0.341 | 35.29 | 0.349 |
| Generic Natural | 4 | 34.25 | 0.450 | 34.40 | 0.515 | 35.36 | 0.517 |
| ABS-R | 4 | 33.03 | 0.045 | 34.32 | 0.261 | 35.28 | 0.267 |
| Bambu Labs Black | 4 | 33.39 | 0.462 | 34.51 | 0.580 | 35.48 | 0.583 |
| m | 34 | 33.53 | 34.496 | 35.424 | |||
| s | 0.665 | 1.519 | 1.475 | ||||
| 95% Conf. Intvl. | 0.337 | 0.61 | 0.253 | ||||
| Material | Hf (Mass), kJ/g | MH2O, % Concentration |
Hc (Mass), kJ/g LHV |
Hc (Mass), kJ/g HHV |
|---|---|---|---|---|
| ABS-Plus | 0.891 | 24.26 | 32.54 | 35.81 |
| Generic-1 | 0.894 | 23.99 | 32.43 | 35.70 |
| ABS-R | 0.944 | 24.29 | 32.47 | 35.87 |
| Bambu | 0.894 | 24.73 | 32.55 | 35.88 |
| Generic-2 | 0.915 | 24.00 | 32.33 | 35.61 |
| Mean, m | 0.907 | 24.25 | 32.46 | 35.77 |
| Std. Dev. s | 0.022 | 0.300 | 0.221 | 0.232 |
|
95% Student-t conf. Interval |
0.028 | 0.373 | 0.150 | 0.144 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




